Abstract
Treatment with an anti-inflammatory Salmonella vaccine expressing enterotoxigenic E. coli colonization factor antigen 1 (CFA/I) proved effective in stimulating protective, potent CD25+ CD4+ T (Treg) cells in susceptible mice challenged with experimental autoimmune encephalomyelitis (EAE). Since the Salmonella vector was considerably less protective, we questioned whether altering the fimbrial subunit expression to resemble conventional Salmonella expression may impact Treg cell potency. The Salmonella-CFA/I vaccine was modified to limit the fimbrial subunit expression to the intracellular compartment (Salmonella-CFA/IIC). SJL mice were challenged with proteolipid protein (PLP)139–151 to induce EAE and orally treated with one of three Salmonella vaccines six days post-challenge. Treatment with Salmonella-CFA/IIC greatly reduced clinical disease, similar to Salmonella-CFA/I, by subduing IL-17 and IL-21; however, mechanisms of protection differed, as evident by increased IL-13 and IFN-γ, but diminished TGF-β production by Treg cells from Salmonella-CFA/IIC-treated mice. Adoptive transfer of Treg cells from both CFA/I-expressing constructs was equivalent in protecting against EAE, showing minimal disease. While not as potent in its protection, CD25−CD4+ T cells from Salmonella-CFA/IIC showed minimal Th2 cells, but this vaccine did prime for Th2 cells subsequent EAE challenge. In vivo IL-13, but not IFN-γ neutralization, compromised protection conferred by adoptive transfer with Salmonella-CFA/IIC-induced Treg cells. Thus, the Salmonella-CFA/IIC vaccine elicits Treg cells with attributes from both the Salmonella vector and Salmonella-CFA/I vaccines. Importantly, these Treg cells can be induced to high potency by simply vaccinating against irrelevant Ags, offering a novel approach to treat autoimmune diseases independently of the auto-Ag.
Keywords: vaccine, mucosa, tolerance, bacterial, autoimmunity, IL-13
Introduction
Immunity to Salmonella is typically Th1 cell-dependent (1, 2). Consequently, Th1 cell-dominating responses are also elicited when using conventional Salmonella vaccine vectors, which result in elevated IFN-γ-regulated IgG2a Ab production, as well as suboptimal mucosal IgA Abs toward both Salmonella and passenger Ags (1–5). Previous works performed in our laboratory demonstrated that when mice are orally immunized with a Salmonella vaccine vector expressing the enterotoxigenic E. coli fimbriae, colonization factor Ag I (CFA/I)4, on its cell surface, elevated serum IgG1 and mucosal IgA Ab titers are stimulated (6). This Ab production is supported by a biphasic Th cell response in which Th2 cells are rapidly induced preceding the development of the Th1 cells (6). In addition to stimulating a Th2 cell bias, the Salmonella-CFA/I vaccine failed to elicit proinflammatory cytokines, TNF-α, IL-1α, IL-1β, and IL-6, following infection of macrophages, unlike the isogenic Salmonella vector strain H647, which readily produced these cytokines (7).
Experimental autoimmune encephalomyelitis (EAE) is an animal model of multiple sclerosis (MS) that shares many features with this human neurodegenerative disease (8). EAE is a Th1/Th17 cell-mediated autoimmune disease directed against protein components of CNS myelin, resulting in neurological damage and paralysis. In EAE and MS, encephalitogenic CD4+ T cells react against CNS auto-antigens and secrete proinflammatory cytokines such as IFN-γ, TNF-α, IL-2, and IL-17 (9). These proinflammatory components induce the activation of microglia and macrophages, the infiltration of inflammatory cells into the CNS, and the eventual axonal damage and demyelination (10–12). In addition to IL-23, IL-21 might play a role in the amplification of Th17 effector responses (13–15). A recent report showed that myeloid dendritic cells (DCs) react to proteolipid protein (PLP), leading to Th17, and not Th1 cell differentiation, thus, increasing the expression of TGF-β and IL-6 and IL-23 (16). Recent data have shown that the combination of IL-6 with TGF-β drives the development of pathogenic Th17 cells (12, 17).
Recently, we described that the anti-inflammatory properties of the Salmonella-CFA/I vaccine confer prophylactic (18) and therapeutic (19) protection against EAE in SJL mice. While not nearly as effective, the isogenic Salmonella vector was able to modestly reduce the EAE clinical scores. Infection with the attenuated Salmonella-CFA/I vaccine elicited FoxP3+ CD25+CD4+ regulatory T (Treg) cells, producing protective levels of TGF-β (19). Although the Salmonella vector was also able to induce Treg cells, the co-stimulation by immune deviation obtained with the Salmonella-CFA/I was not evident, and consequently, protection against EAE was substantially reduced. Neutralization of Treg cell function in mice immunized with Salmonella-CFA/I provoked a more severe EAE, and adoptive transfer of Salmonella-CFA/I-induced Treg cells, but not naïve Treg cells, protected against EAE. Adoptive transfer of Treg cells from immunized mice with Salmonella vector protected against EAE, but to a lesser degree. We concluded that the immunization with attenuated strains of Salmonella expressing irrelevant, non-self antigens induced and expanded Treg cells that were protective against EAE (19).
In this current work, we queried whether differences in the observed Treg cell potency were attributed to the stimulation of Th2 cells, resulting in immune deviation, since these latter T cells were not elicited by the Salmonella vector. A Salmonella vaccine limiting the CFA/I subunit to its expression in the periplasmic/intracellular compartment (Salmonella-CFA/IIC) was developed to determine if the CFA/I fimbrial subunit could then stimulate Th1, rather than Th2 cells, and whether this shift in CD4+ T cell response would impact the development of potent Treg cells to treat EAE. In other words, would the Salmonella-CFA/IIC strain remain anti-inflammatory or would it resemble the proinflammatory, isogenic Salmonella vector? It was found that treatment with Salmonella-CFA/IIC greatly reduced the EAE clinical scores as effectively as Salmonella-CFA/I and better than the Salmonella vector-treated mice. Both CFA/I vaccines reduced the IFN-γ produced by CD25−CD4+ T cells, but, interestingly, the Salmonella- CFA/IIC stimulated the generation of IL-13- and IFN-γ-producing Treg cells that appeared to protect against EAE. In vivo blockade of IL-13 reversed protection conferred by Salmonella-CFA/IIC induced-Treg cells, whereas the in vivo blockade of IFN-γ did not. As evident from this study, oral immunization with Salmonella vaccines enhances multiple subsets of Treg cells with different regulatory profiles for which their induction is dependent upon the presence and location of heterologous Ags.
Materials and Methods
Mice
Female six wk old SJL mice were obtained from Frederick Cancer Research Facility, National Cancer Institute (Frederick, MD), and The Jackson Laboratories (Bar Harbor, ME). All mice were maintained at Montana State University Animal Resources Center under pathogen-free conditions in individual ventilated cages under HEPA-filtered barrier conditions and were fed sterile food and water ad libitum. The mice were free of bacterial and viral pathogens, as determined by Ab screening and histopathological analysis of major organs and tissues. All animal care and procedures were in accordance with institutional policies for animal health and well-being.
Salmonella-CFA/IIC (AP331) construction
The pMA20-asd+ recombinant plasmid was constructed by deleting the 2.0-kb Sac I DNA fragment from the CFA/I operon (cfaABCE) from pJGX15C-asd+ (20). The deleted fragment encodes for an outer membrane usher protein (CfaC), which mediates the ordered assembly of the filamentous heteropolymer and a minor component protein (CfaE) present as a single tip subunit (21). The remaining cfaAB genes are involved in the expression of the major structural subunit (CfaB) and the chaperone (CfaA) promoting subunit folding and transport in periplasmic and intracellular compartments. The ligated plasmid construct was electroporated into E. coli H681 Δasd strain. Clones containing the selected plasmid construct were detected and screened by colony immunoblotting, in vivo antigen/Ab slide agglutination, and plasmid miniprep-restriction map protocols. The purified plasmids obtained were then electroporated into Salmonella enterica serovar Typhimurium H683 ΔaroA Δasd strain to obtain the desired balanced lethal Salmonella-CFA/IIC (AP331) construct. Recombinant strains and control strain H647 were cultured by using both Luria-Bertani (LB) and nutrient agar or broth media without antibiotics or diaminopimelic acid, as previously described (20). Neither H681 nor H683 grows on these media unless the asd+ allele is supplied in trans.
Detection of CFA/I fimbrial subunit in Salmonella strains by Western blot
The absence or presence of CFA/I fimbrial subunit in S. Typhimurium H647, AP331, and H696 was analyzed by SDS-PAGE and Western blot. SDS-PAGE gel (15% [wt/vol] polyacrylamide) was transferred to a 0.2-mm-pore-size nitrocellulose membrane (Bio-Rad Laboratories, Hercules, CA). The membrane was incubated in rabbit anti-CFA/I antiserum (produced in-house; 1:2,000 diluted in PBS-Tween 20) overnight at 4 ºC. After four washes in PBS-Tween 20, a second incubation of 90 min with HRP conjugated rat anti-rabbit IgG (Southern Biotechnology Associates, Inc., Birmingham, AL) diluted (1:1,000) in PBS-Tween 20 was performed. Detection of CFA/I fimbrial subunit, after four new washes in PBS-Tween 20, was achieved upon development with the substrate 4-chloro-1-naphthol chromogen and H2O2 (Sigma-Aldrich Chemical Co., St. Louis, MO). The apparent molecular masses were determined by comparing their electrophoretic mobility with that of the following molecular mass markers (Amersham Pharmacia Biotech, Freiburg, Germany): myosin [220 kDa]; phosphorylase b [97 kDa]; bovine serum albumin [66 kDa]; ovalbumin [45 kDa]; carbonic anhydrase [30 kDa]; trypsin inhibitor [20.1 kDa]; lysozyme [14.3 kDa].
AP331 subcellular fraction and isolation studies were performed to quantify the level of CfaB expression. AP331 was cultured in LB medium, as described above, and bacteria were harvested by centrifugation. Subcellular fractionation was performed according to manufacturer’s directions using the PeriPreps Periplasting Kit (Epicentre, Madison, WI), as previously described (22, 23). The different subcellular components were subjected to 15% SDS-PAGE and subsequently transferred to a nitrocellulose membrane for Western blot analysis. A rabbit anti-CFA/I serum was used as primary Ab, and bound Ab was detected using a HRP-conjugated goat anti-rabbit-IgG Ab (Southern Biotechnology Associates, Inc.) and substrate, as described above.
PLP139–151 challenge and oral Salmonella vaccination
The encephalitogenic PLP peptide (PLP139–151; HSLGKWLGHPDKF) was synthesized by Global Peptide Services, LLC, (Ft. Collins, CO), and HPLC-purified to > 90%. For each experiment, female SJL mice (5/group) were challenged s.c. with 200 μg PLP139–151 in 200 μl of complete Freund’s adjuvant (18). On days 0 and 2 post-challenge, mice received i.p. 200 ng of Bordetella pertussis toxin (PT; List Biological Laboratories, Campbell, CA). Six days after PLP139–151 challenge, mice were given a single oral dose of 5 × 109 colony-forming units (CFU) of the Salmonella-CFA/I vaccine (6,19; ΔaroA Typhimurium-CFA/I vector vaccine, strain H696, expressing functional CFA/I fimbriae from E. coli), Salmonella-CFA/IIC (ΔaroA Typhimurium-CFA/I vector vaccine, strain AP331, expressing CFA/I in the periplasmic/intracellular compartment), or its isogenic control strain H647 (Salmonella empty vector) (6, 19). Fimbrial expression was maintained by a plasmid bearing a functional asd gene to complement the lethal chromosomal Δasd mutation in the parent Salmonella strain. Control groups were treated with PBS. Mice were monitored and scored daily for disease progression (18): 0, normal; 1, a limp tail; 2, hind limb weakness; 3, hind limb paresis; 4, quadriplegia; 5, death.
Mice were also evaluated for extent of colonization by AP331 and H696 vaccine strains. Mice (5/group) were orally dosed with 5 × 109 CFU of AP331 or H696, and spleens and Peyer’s patches were harvested 1 wk later. Tissues were weighed and homogenized in sterile water for CFU enumeration on MacConkey’s agar (Difco, Detroit, MI) and incubated overnight at 37°C (24).
Ab ELISA
CFA/I-specific endpoint titers from dilution of immune sera or fecal extracts were measured by an ELISA, as previously described, using purified fimbrial Ag (6) as a coating Ag. Specific reactivity to CFA/I fimbriae was determined using HRP conjugates of goat anti-mouse IgG-, IgG1-, IgG2a-, IgG2b- and IgA-specific Abs (1 μg/ml; Southern Biotechnology Associates), and ABTS (Moss Inc., Pasadena, CA) enzyme substrate was used to develop the reaction. The absorbance was measured at 415 nm on a Bio-Tek Instruments ELx808 microtiter plate reader (Winooski, VT). Endpoint titers were expressed as the reciprocal dilution of the last sample dilution, giving an absorbance of 0.1 OD units above OD415 of negative controls after 1 h incubation.
Histological evaluation of spinal cords
For histological evaluation of tissue pathology, spinal cords were removed 14 days after challenge and fixed with neutral buffered formalin (VWR International, West Chester, PA), embedded into paraffin, and sectioned at 5 μm. Transverse sections of spinal cords were stained with H&E for pathological changes and inflammatory cell infiltration. Adjacent sections were stained with luxol fast blue (LFB) and examined for loss of myelin. Pathological manifestations were scored separately for cell infiltrates and demyelination. Each H&E section was scored from 0 to 4: 0, normal; 1, cell infiltrate into the meninges; 2, one to four small focal perivascular infiltrates; 3, five or more small focal perivascular infiltrates and/or one or more large infiltrates invading the parenchyma; 4, extensive cell infiltrates involving 20% or more of the white matter (18,19). In each LFB stained section, myelin was also scored from 0 to 4: 0, normal; 1, one small focal area of demyelination; 2, two or three small focal areas of demyelination; 3, one to two large areas of demyelination; 4, extensive demyelination involving 20% or more of white matter (18, 19).
Cytokine ELISA
Spleens, mesenteric lymph nodes (MLNs), and head and neck LNs (HNLNs) were aseptically removed 14 days after challenge from PBS-, Salmonella vector-, Salmonella-CFA/IIC-, and Salmonella-CFA/I-treated groups of mice. Lymphocytes were prepared, as previously described (19), and resuspended in complete medium (CM): RPMI 1640 medium supplemented with 1 mM sodium pyruvate, 1 mM nonessential amino acids, penicillin/streptomycin (10 U/ml), and 10% fetal bovine serum (Atlanta Biologicals, Lawrenceville, GA). Lymphocytes were cultured in 24-well tissue plates at 5 × 106 cells/ml in CM alone or in the presence of OVA (Sigma-Aldrich; 10 μg/ml), purified CFA/I fimbriae (10 μg/ml), or PLP139–151 peptide (30 μg/ml) in a total volume of 1 ml for 60 h at 37º C. The supernatants were collected by centrifugation and stored at −80º C. Capture ELISA was employed to quantify, on duplicate sets of samples, the levels of IFN-γ, IL-4, IL-10, IL-13, IL-17, and TGF-β produced by lymphocytes, as previously described (19). For detection of IL-21 and IL-22, microtiter wells were coated with 2 μg/ml of purified goat anti-mouse IL-21 Ab, or goat anti-mouse IL-22 Ab, respectively, (R&D Systems, Minneapolis, MN). After blocking with PBS + 1% BSA for 2 h at 37ºC, washed wells were incubated with cell culture supernatants at 4º C for 24 h. After washing, 0.5 μg/ml biotinylated rat anti-mouse IL-21 mAb or biotinylated goat anti-mouse IL-22 Ab (R&D Systems) was added, respectively, for 90 min at 37º C. Following washing, 1:500 HRP-goat anti-biotin Ab (Vector Laboratories, Inc., Burlingame, CA) was added for 1 h at room temperature. After washing, ABTS peroxidase substrate (Moss, Inc.) was added to develop the reaction.
FACS analysis
Lymphocytes from the HNLNs, MLNs, and spleens were isolated 14 days after challenge, and single cell preparations were prepared, as described above (19). To obtain lymphocytes from spinal cords, mice were perfused through the left ventricle with 20 ml of cold PBS, and spinal cords were removed by flushing the vertebral canal with media and prepared, as previously described (19).
Cells were stained for FACS analysis using conventional methods. To distinguish among neutrophils, monocytes/macrophages, and lymphocytes, staining for CD45 and MHC class II was done, as previously described (19). Leukocyte gates were set within the forward and side scatter profiles to exclude resting microglia cells from the spinal cord preparations. To detect neutrophils, cells were stained with SK208 mAb (18,19), followed by FITC-donkey anti-rat anti-IgG (Jackson ImmunoResearch Laboratories, West Grove, PA) and fluorochrome-conjugated anti-CD11b (BD Pharmingen); for macrophages, fluorochrome-conjugated mAbs (BD Pharmingen) were used: CD45 (clone 30-F11), I-As (clone 10–3.6), CD11b, and PE-F4/80 mAb (Serotec, Oxford, UK). T cell subsets were analyzed using fluorochrome-conjugated mAbs (BD Pharmingen) for CD4, CD25, TCRβ, CD8, GITR, CCR6, CTLA-4, and RANKL, and biotinylated TGF-β (R&D Systems). Intracellular staining for FoxP3 was done using fluorochrome labeled-anti-Foxp3 mAb (clone FJK-16s; eBioscience, San Diego, CA), FITC or PE-anti-IFN-γ Ab, (BD Pharmingen), and biotinylated anti-IL-13 Ab (R&D Systems). Bound fluorescence was analyzed with a FACS Canto (BD Biosciences, Mountain View, CA).
Adoptive transfer studies
Fourteen days after oral immunization with H647, AP331, or H696, total CD4+ T cells from spleens, HNLNs, and MLNs were obtained (negative CD4+ T cell isolation kit, Dynal Biotech ASA, Oslo, Norway). CD25+CD4+ and CD25−CD4+ T cells were isolated to > 93% and 99%, respectively, by cell-sorting (FACSVantage with Turbo-Sort, BD Biosciences) of stained T cells. To test Treg cell efficacy, 6 × 105 CD25−CD4+ T cells or CD25+CD4+ T cells were i.v. injected into naïve recipients. One day after the adoptive transfer of T cell subsets, mice were challenged with PLP139–151.
In vivo blockade of IFN-γ and IL-13
To block IFN-γ, mice were given i.p. 0.5 mg of anti-IFN-γ mAb purified from hybridoma cell line (clone R4 6A2; ATCC, Manassas, VA) on day -1 (before), and +1 and +5 (after) EAE challenge with PLP139–151. One day before the challenge, AP331- and H647- induced CD25+ CD4+ Treg cells were adoptively transferred into mice. Control groups received 0.5 mg of purified rat IgG Ab (AbD Serotec, Oxford, UK). A separate control group was immunized with PBS one day prior to EAE challenge. All mice were monitored daily for development of EAE.
To block IL-13, mice were given i.p. 0.5 ml of serum from rabbits immunized with rat IL-13 (25) on day -1 before challenge, and 0.25 ml of serum on days +1 and +5 after EAE challenge with PLP139–151. The same adoptive transfer protocols were used in this study, and control mice were given equivalent volumes of normal rabbit serum (NRS). All mice were monitored daily for development of EAE.
In vitro T cell assays
To assess cytokine production by Treg and effector T cells, CD25+CD4+ T cells and CD25−CD4+ T cells (2 × 105) were stimulated in vitro with anti-CD3 mAb-coated wells (10 μg/ml; BD Pharmingen), plus the soluble anti-CD28 mAb (5.0 μg/ml; BD Pharmingen) or with 30 μg/ml PLP139–151 for 5 days in CM (final volume of 300 μl in 48-wells plate). Capture ELISA was employed to quantify triplicate sets of samples to measure cytokines.
Statistical analysis
The ANOVA followed by posthoc Tukey test was applied to show differences in clinical scores in treated vs. PBS mice and in the Treg cell kinetic experiments. The student t test was used to evaluate the differences between variations in cytokine level production, and P-values < 0.05 are indicated.
RESULTS
Oral immunization with Salmonella-CFA/IIC protects against EAE
Previously, it was shown that oral treatment with a single dose of a S. Typhimurium, expressing the CFA/I fimbriae from enterotoxigenic E. coli (Salmonella-CFA/I, strain H696), was able to reduce inflammatory cell infiltration, demyelination, and EAE clinical scores in SJL mice (19). This particular vaccine was exquisite in stimulating elevated levels of Treg cells, in part, by immune deviation obtained with CD25− Th2-type cells. While Treg cells were also induced upon vaccination with the Salmonella empty vector, the co-stimulation of the CD25− Th2-type cells was not induced, resulting in much less protection (19). Thus, the empty vector was unable to elicit the production of anti-inflammatory cytokines, although it elicited the expansion of Treg cells, but this was insufficient for protection against EAE when compared to Salmonella-CFA/I. Given these findings, we queried that if the CFA/I fimbrial subunit was not expressed on the Salmonella’s cell surface and made to resemble the Salmonella empty vector would the protective efficacy be compromised since this construct would be predicted to lack the co-induced anti-inflammatory response by CD25−CD4+ T cells.
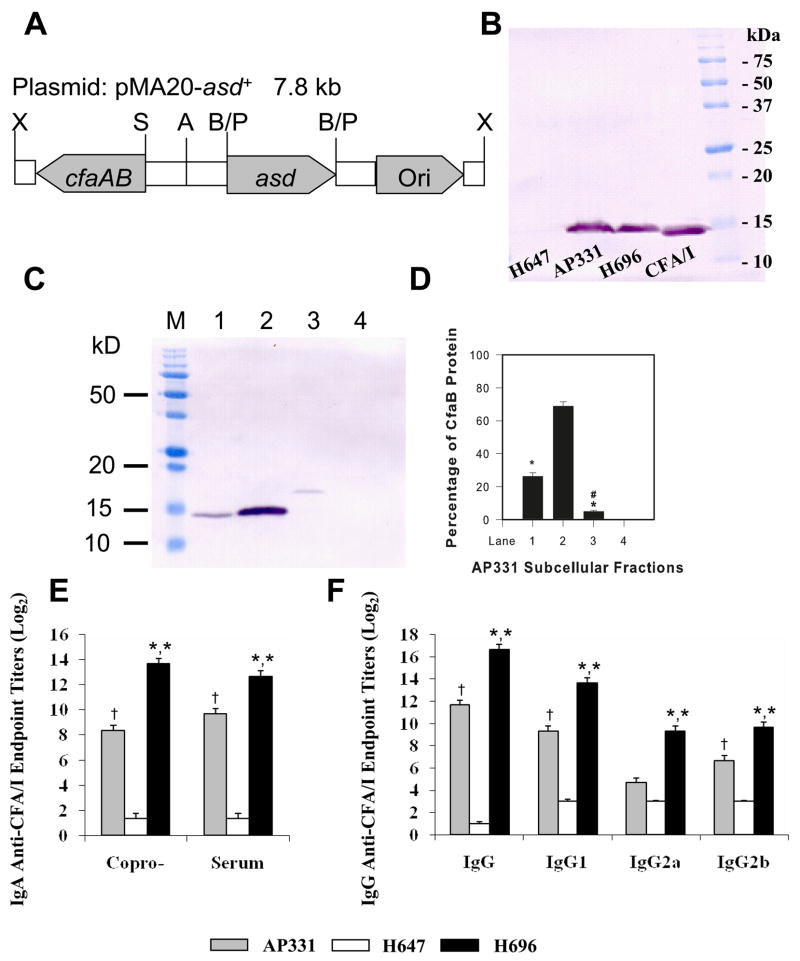
To address this question, a S. Typhimurium strain that expresses the CFA/I fimbrial subunit in its periplasmic/intracellular compartment (Salmonella-CFA/IIC, strain AP331) was constructed (Fig. 1A) by deleting the presumed usher protein and minor subunit (21). To demonstrate expression of CFA/I fimbrial subunit by AP331, a Western blot was performed using a rabbit polyclonal Ab against CFA/I fimbriae (Fig. 1B) and compared to expression by Salmonella-CFA/I (H696). Similar amounts of CFA/I fimbrial subunit were observed in whole cell extracts from AP331 and H696 when compared to a purified sample of CFA/I fimbriae, and no expression of the fimbriae was observed by the Salmonella empty vector (H647). The CfaB subunit fractionated mostly with the periplasm and some with the spheroplast with negligible levels associated with the outer membrane and cell wall (Fig. 1C, D). Oral vaccination of SJL mice with AP331 elicited serum and mucosal Ab titers against CFA/I fimbriae, as demonstrated by ELISA 14 days after vaccination, but was not as great as with Salmonella-CFA/I (P < 0.001; Fig. 1E and F). Differences in these Ab titers did not appear to be attributed to differences in colonization by AP331- or H696-dosed mice. Splenic and Peyer’s patch weights, one week after infection, were identical, as was splenic colonization; however, AP331-infected mice showed ~50% more reduction (P < 0.001) in colonization of the Peyer’s patches than did the H696-infected mice.
FIGURE 1.
Expression of CFA/I fimbrial subunit by Salmonella vaccines are equivalent, but render different Ab titers. (A) The cfaABCE operon for the expression of the CFA/I fimbriae was modified by deleting the cfaCE genes but leaving the fimbrial subunit (CfaB) and its chaperone (CfaA) intact. Restriction sites include: X: Xho I; S: Sac I; A: Aat II; B: Bgl II; and P: Pvu I. (B) Equivalent whole cell extracts from Salmonella-CFA/I (H696) and Salmonella-CFA/IIC (AP331), when subjected to SDS-PAGE and Western blot analysis, showed similar amounts of CFA/I fimbrial subunit being produced. Purified CFA/I fimbriae were used as a positive migration control, and whole cell extracts from Salmonella empty vector (H647) did not react against the polyclonal rabbit anti-CFA/I fimbriae Abs. (C, D) Subcellular fractionation of S. Typhimurium strain AP331. AP331 cells were fractionated and enriched for Lane 1: spheroplasts; Lane 2: periplasmic membranes; Lane 3: outer membranes and cell wall; and Lane 4: cell culture supernatant and (D) quantified by densiometric scanning. Lane M is the prestained protein M.W. standards. Presented are the results from one of the three experiments. The majority of the CfaB subunit was present in the periplasm (Lane 2), some in the spheroplasts (Lane 1), and a minimal amount associated with the outer membrane (Lane 3). None was secreted (lane 4). *P < 0.001, represents statistical differences between the periplasmic fraction versus each fraction; #P < 0.001, represents the statistical difference between the spheroplasm fraction versus the outer membrane and cell wall fraction. (E, F) A single oral immunization with Salmonella-CFA/I or Salmonella-CFA/IIC vaccine increased (E) mucosal and serum IgA and (F) IgG responses to CFA/I fimbriae, but not the Salmonella empty vector (H647). The AP331 strain stimulated less mucosal and serum Abs than the H696 strain. Analysis of IgG subclasses showed significant increases in IgG1, IgG2a, and IgG2b (P < 0.001) in H696-vaccinated mice when compared to AP331-vaccinated mice. Depicted are the means ± SEM from three separate experiments for a total of 15 mice/group. *, P < 0.001 for H696- vs. AP331- and H696- vs. H647-vaccinated groups. †, P < 0.001 for AP331- vs. H647-vaccinated groups.
To test whether the therapeutic potential of CFA/I fimbriae was lost by altered expression in Salmonella, SJL mice were s.c. challenged with PLP139–151 per standard procedures, and 6 days after challenge, a single oral dose of H647, AP331, H696, or PBS was administered. PBS-dosed mice showed the expected disease course with a mean onset of disease of 9.2 ± 0.4 (Table I) with a maximum clinical score of 5. Disease peaked 16 days after challenge (Fig. 2). Mice orally treated with the Salmonella-CFA/I (H696) showed a significant delay in the onset of disease (10.9 ± 0.4; P < 0.001) and a significant reduction in the cumulative score (13.2 versus 54.2 by PBS-treated group; P < 0.001). When mice were treated with Salmonella-CFA/IIC, onset of disease was also delayed significantly (11.2 ± 1.3; P < 0.001), and the cumulative scores were reduced significantly (11.6; P < 0.001) when compared to the PBS-treated group. Maximum scores of 2 were observed after vaccination with H696 and AP331. Treatment with the Salmonella empty vector (H647) also caused a significant delay on the onset of disease (10.8 ± 0.4; P < 0.001) and a reduction of the cumulative (25.0; P < 0.001) when compared to the PBS-treated group. However, the maximum clinical score was the same as that obtained with the PBS group, and the reduction of the clinical scores was significantly lower than that observed after treatment with AP331 (25.0 vs. 11.6; P < 0.001) or H696 (25.0 vs. 13.2; P < 0.001). These results confirmed the previously published data about the Salmonella empty vector and the Salmonella-CFA/I strain (18, 19) and demonstrated that the altered presence of CFA/I in Salmonella did not reduce its ability to protect against EAE in susceptible SJL mice.
Table I.
Therapeutic treatment with Salmonella vaccines after PLP139–151 challenge protects SJL/J mice from EAEa
| Treatmentb | EAE/Totalc | Onsetd | Max. Scoree | CSf | Inflammationg | Demyelinationh |
|---|---|---|---|---|---|---|
| PBS | 15/15 | 9.2 ± 0.4 | 5 | 54.0 ± 2.8 | 2.0 ± 0.9 | 2.9 ± 0.31 |
| AP331 | 15/15 | 11.2 ± 1.3 | 2 | 11.6 ± 0.4*,* | 0.8 ± 0.3*,* | 1.1 ± 0.2*,* |
| H647 | 15/15 | 10.8 ± 0.4 | 5 | 25.0 ± 1.5* | 2.1 ± 0.1 | 2.4 ± 0.1 |
| H696 | 15/15 | 10.9 ± 0.4 | 2 | 13.2± 0.4 *,* | 0.7 ± 0.3*,* | 1.0 ± 0.1*,* |
SJL/J mice were challenged s.c. with 200 μg PLP139–151 in complete Freund’s adjuvant plus 200 ng PT i.p. on days 0 and 2.
Mice were immunized 6 days post-challenge with PBS or 5 × 109 CFU of S. enterica Typhimurium H647 (vector), H696 (CFA/I fimbriae),
or AP331 (CFA/IIC fimbriae).
Number of mice with EAE/total in group.
Mean day ± SD of clinical disease onset.
Maximum (Max.) daily clinical score.
Cumulative scores (CS) were calculated as the sum ± SEM of all scores from disease onset to day 26 post-challenge, divided by the number of mice in each group. * P < 0.001 for PBS vs. treated groups; H647- vs. H696-treated groups; and H647- vs. AP331-treated groups.
Mean score ± SEM of inflammation: the infiltration of nucleated cells into spinal cords was scored from 0 to 4 in each mouse separately, and the mean score ± SEM was calculated. *, P < 0.001 for PBS vs. AP331- or H696-treated groups; H647- vs. H696-treated groups; and H647- vs. AP331-treated groups.
Mean score ± SEM of demyelination: of spinal cords was scored from 0 to 4 in each mouse separately, and the mean score ± SEM was calculated. *, P < 0.001 for PBS vs. H696- or AP331-treated groups; H647- vs. H696-treated groups; and H647- vs. AP331-treated groups.
% CD4+ TCRβ+ T cells from the total cells in spinal cords, analyzed by FACS (Fig. 1C).*, P < 0.001 for PBS vs. treated groups; H647- vs. H696-treated groups; and H647- vs. AP331-treated groups.
FIGURE 2.
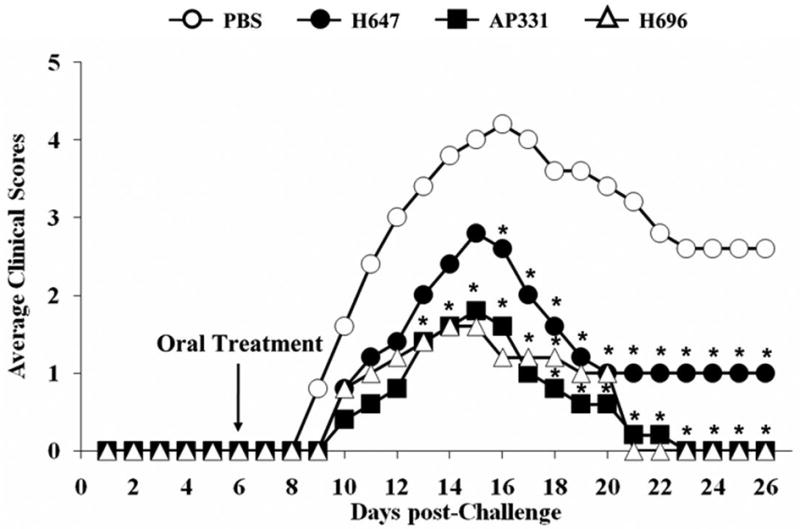
Oral Salmonella-CFA/I (H696) and Salmonella-CFA/IIC (AP331) treatments reduced EAE clinical scores in PLP139–151-challenged SJL mice. Mice therapeutically treated 6 days after EAE challenge with H696 or AP331 vaccines showed a significant reduction in their clinical scores with all mice recovering from EAE. Salmonella vector (strain H647)-treated mice showed some recovery, but presented greater clinical scores when compared to PBS-treated mice. Depicted are the combined results from three separate experiments for a total of 15 mice/group: *, P < 0.001 for PBS- vs. H647-, H696-, or AP331-treated mice.
FACS analysis of inflammatory cells obtained from spinal cords from treated mice was performed 6, 10, and 14 days after challenge with PLP139–151. H696- and AP331-treated mice at 10 and 14 days after challenge (4 and 8 days after treatment respectively) failed to show inflammatory cell infiltration (Table II). PBS- and Salmonella vector-treated mice showed marked infiltration by neutrophils (CD11b+ SK208+), CD11b+ F4/80+ macrophages, and T cells (TCRβ+ CD4+ and TCRβ+ CD4−). Although the H647 showed a reduced clinical score, it failed to prevent inflammatory cell infiltration by day 14 after challenge.
Table II.
Treatment with Salmonella vaccines reduces inflammatory cell infiltrationa into the spinal cords
| MHC class II+ CD45 high infiltrated cells
| ||||||||
|---|---|---|---|---|---|---|---|---|
| % Infiltration | % CD4+TCR-β+ | % CD11b+ SK208+ | % CD11b+ F4/80+ | |||||
| Day 6 | 0.95 ± 0.2 | 0.37 ± 0.04 | 0.49 ± 0.1 | 0.01 ± 0.01 | ||||
| Day 10 | Day 14 | Day 10 | Day 14 | Day 10 | Day 14 | Day 10 | Day 14 | |
| PBS | 3.53 ± 0.08 | 3.9 ± 0.12 | 2.19 ± 0.8 | 1.78 ± 0.05 | 1.87 ± 0.3 | 1.95 ± 0.2 | 1.43 ± 0.1 | 0.96 ± 0.6 |
| AP331 | 0.2 ± 0.1*,* | 0.4 ± 0.05*,* | 0.16 ± 0.4 | 0.3 ± 0.02* | 0.1 ± 0.2*,* | 0.9 ± 0.2*,* | 0.0 ± 0.0*,* | 0.0 ± 0.1* |
| H647 | 2.73 ± 0.06 | 1.3 ± 0.2 | 0.16 ± 0.4 | 1.4 ± 0.06 | 0.6 ± 0.01 | 1.85 ± 0.1 | 0.27 ± 0.5 | 0.15 ± 0.1 |
| H696 | 0.4 ± 0.1*,* | 0.37 ± 0.1*,* | 0.17 ± 0.04 | 0.2 ± 0.06* | 0.0 ± 0.2*,* | 0.7 ± 0.1*,* | 0.1 ± 0.02*,* | 0.0 ± 0*,* |
Results are shown in percentage of MHC class II+ CD45high cells from the total cells in spinal cords analyzed by FACS (Fig. 4).
P < 0.001 for PBS vs. AP331- or H696-treated groups; H647- vs. AP331-treated groups; and H647- vs. H696-treated groups.
Salmonella-CFA/IIC reduces inflammatory PLP139–151-specific CNS damage without eliciting Th2-type cytokines
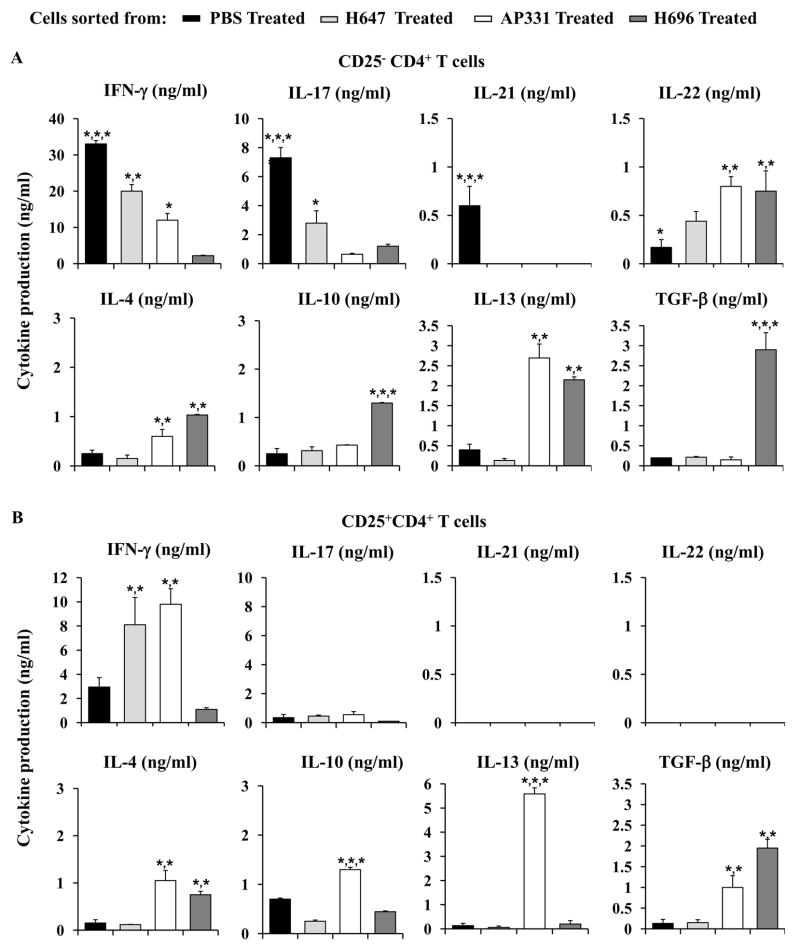
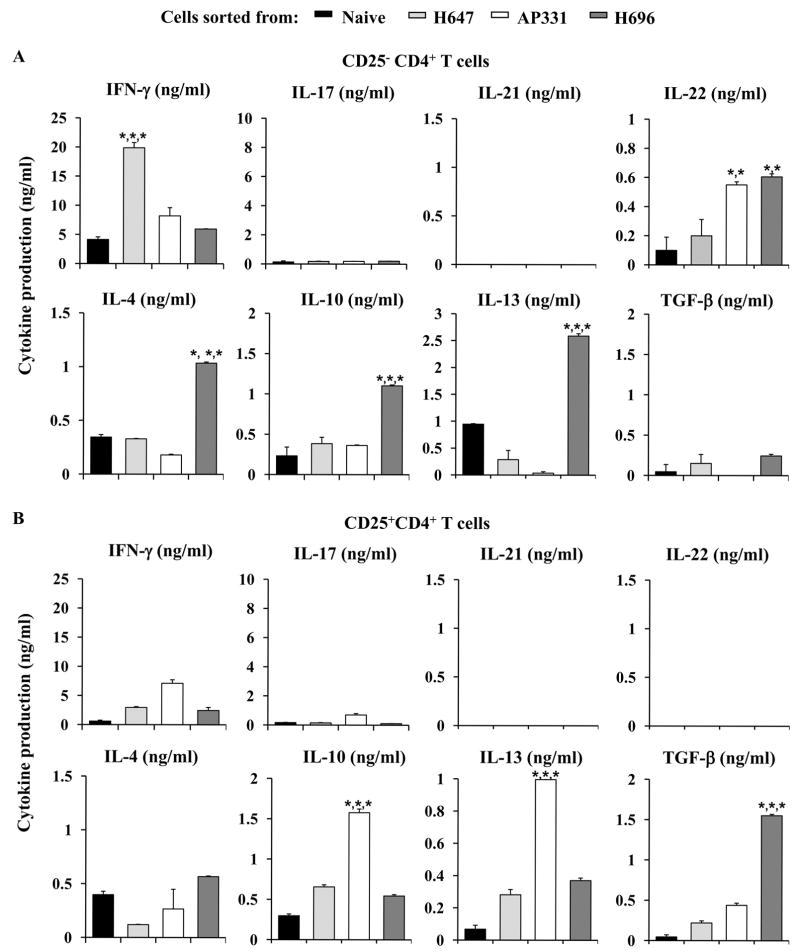
Cytokine production was analyzed for sorted CD25−CD4+ T cells from PBS-, H647-, AP331-, and H696-treated mice, 14 days post-EAE challenge. Cells were sorted (purity of isolated cells, determined by FACS analysis: > 95%) and in vitro co-stimulated with anti-CD3 and anti-CD28 mAbs (Fig. 3). CD25−CD4+ T cells from PBS-treated mice produced significantly elevated levels of IFN-γ, IL-17, and IL-21 (P < 0.001) than CD25−CD4+ T cells obtained from H647-, AP331-, or H696-treated mice (Fig. 3A). IFN-γ and IL-17 production by CD25−CD4+ T cells isolated from H647-treated mice was also significantly enhanced (P < 0.001) when compared to AP331- and H696-treated mice. In contrast, CD25−CD4+ T cells from H696-treated mice produced significantly greater levels (P < 0.001) of anti-inflammatory cytokines, IL-4, IL-10 and TGF-β, than those obtained from PBS-, H647-, or AP331-treated mice and IL-13 and IL-22 when compared to PBS- and H647-treated mice (P < 0.001). CD25−CD4+ T cells from AP331-treated mice showed significantly higher productions (P < 0.001) of IL-4, IL-13, and IL-22 than those obtained from PBS- and H647-treated mice
FIGURE 3.
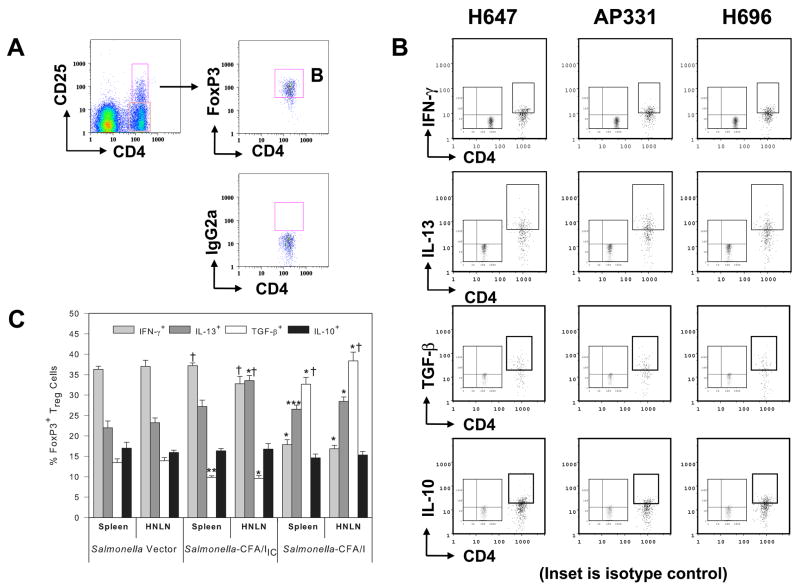
Salmonella-CFA/IIC vaccine therapy elicits IL-13- and IFN-γ-producing Treg and CD25−CD4+ T cells in contrast to Salmonella-CFA/I therapy that stimulates TGF-β-producing Treg and CD25− Th2 cells subsequent to EAE challenge. Cell-sorted (A) CD25−CD4+ T cells and (B) Treg cells from EAE-challenged SJL mice treated with H647, AP331, H696, or PBS were purified on day 14 post-challenge with PLP139–151 and co-stimulated in vitro with anti-CD3 and anti-CD28 mAbs. Cytokine production was measured from supernatants after 72 h of culture. (A) CD25−CD4+ T cells from diseased (PBS-treated) mice produced elevated levels of IFN-γ, IL-17, and IL-21 with more depressed IL-4, IL-10, IL-13, TGF-β, and IL-22 than any of the Salmonella-treated groups. IFN-γ and IL-17 production by H647-treated mice was also significantly enhanced when compared to AP331- and H696-treated mice, and AP331 CD25−CD4+ T cells produced more IFN-γ than H696. CD25−CD4+ T cells from H696-and AP331-treated mice produced more IL-4, IL-13, and IL-22 than PBS- or H647-treated mice; H696 also produced more IL-10 and TGF-β than any of the treatment groups. (B) CD25+CD4+ T cells sorted from H647- and AP331-treated mice produced elevated levels of IFN-γ when compared to cells sorted from PBS-treated or H696-treated mice (P < 0.001). Treg cells from PBS- and H647-treated groups produced minimal levels of the anti-inflammatory cytokines, IL-4, IL-10, IL-13, and TGF-β. Treg cells sorted from any of the treatment groups produced no to minimal amounts of IL-17, IL-21, and IL-22. While both produced equivalent IL-4, AP331-induced Treg cells showed strikingly enhanced IL-13 while H696-induced Treg cells produced elevated levels of TGF-β. Depicted are the combined results from three separate experiments for a total of 15 mice/group ± SEM. *, P < 0.001 represents differences in cytokine production between PBS-, H647-, AP331-, and H696-treated groups.
Treg cells were also sorted and evaluated for their production of cytokines (purity of > 93%; Fig. 3B). Treg cells sorted from H647-treated mice showed only an increased production of IFN-γ when compared to those sorted from PBS-treated mice (P < 0.001). Treg cells sorted from H696-treated mice produced minimal to no levels of IFN-γ and IL-17, but enhanced levels of IL-4 when compared to those obtained from PBS- and H647-treated mice (P < 0.001) and significantly greater levels of TGF-β than cells sorted from PBS-, H647-, and AP331-treated mice (P < 0.001). CD25+CD4+ T cells sorted from AP331-treated mice produced significantly enhanced levels of IFN-γ when compared to PBS- and H696-derived Treg cells (P < 0.001); enhanced IL-4 and TGF-β when compared to PBS- and H647-derived Treg cells (P < 0.001); and enhanced IL-10 and IL-13 when compared with all treatment groups (P < 0.001). IL-17 production was not different in any of the groups. None of the Treg cells produced any IL-21 or IL-22.
Ag-specific cytokine evaluations were also performed on the purified Treg and CD25− CD4+ T cells. As with the anti-CD3 plus anti-CD28 stimulation (Fig. 3), AP331 vaccine-induced Treg cells produced elevated IL-13 and IFN-γ with lesser amounts of TGF-β, but similar levels of IL-4 and IL-10 following in vitro restimulation with PLP139–151 (Fig. 4B). For the CD25− CD4+ T cells, similar trends obtained with PLP139–151 restimulation as with the anti-CD3 plus anti-CD28 stimulations observed in Fig. 3A were evident (Fig. 4A). Peptide restimulation notably induced increased IL-13 with similar IL-4, IL-10, and TGFβ and depressed IFN-γ and IL-17 production (Fig. 4A). IL-21 was only produced by CD25− CD4+ T cells from PBS-dosed and challenged mice.
FIGURE 4.
Salmonella-CFA/IIC vaccine elicits IL-13- and IFN-γ-producing Treg cells, as evidenced by PLP139–151 restimulation. Sorted (A) Treg cells and (B) CD25−CD4+ T cells from SJL mice that were orally dosed with H647, AP331, H696, or PBS on day -14, EAE-challenged on day 0, and were purified 14 days after EAE challenge and co-stimulated in vitro with 30 μg PLP139–151 peptide/ml of cultured cells. Cytokine production was measured from supernatants after 5 days of culture. (A) CD25−CD4+ T cells from diseased (PBS-treated) mice produced elevated levels of IFN-γ, IL-17, and IL-21 with minimal IL-4, IL-10, IL-13, and TGF-β than any of the Salmonella-infected groups. IFN-γ and IL-17 production by H647-dosed mice was also significantly enhanced when compared to AP331- and H696-dosed mice, and more IFN-γ was produced by AP331- than H696-dosed mice. CD25−CD4+ T cells from H696-dosed mice produced more IL-4 and IL-10 than PBS- or H647-dosed mice, while CD25−CD4+ T cells from AP331-dosed mice produced strikingly more IL-13 than any of the groups; H696-dosed mice produced more TGF-β than any of the treatment groups. (B) CD25+CD4+ T cells sorted from H647- and AP331-dosed mice produced elevated levels of IFN-γ when compared to cells sorted from PBS- or H696-dosed mice (P < 0.001). Treg cells from PBS- and H647-dosed groups produced minimal levels of the anti-inflammatory cytokines, IL-4, IL-10, IL-13, and TGF-β. Treg cells sorted from any of the treatment groups produced minimal amounts of IL-17 and IL-21. Both produced equivalent IL-4 and IL-10 and AP331-induced Treg cells showed strikingly enhanced IL-13 while H696-induced Treg cells produced elevated levels of TGF-β. Cytokine production after stimulation with PLP139–151 peptide is depicted as the means of 10 mice/group ± SEM corrected for cytokine production by unstimulated cells. *, P ≤ 0.001; **, P ≤ 0.05, represents differences in cytokine production between PBS-, H647-, AP331-, vs. H696-treated groups.
Oral vaccination with Salmonella-CFA/IIC induces IL-13- and IFN-γ-producing FoxP3+ Treg cells
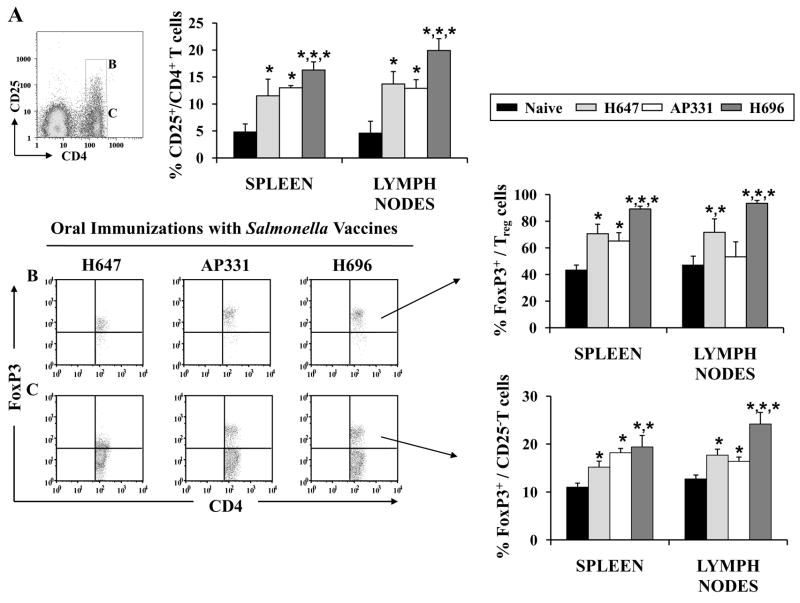
To ascertain whether the altered expression of CFA/I fimbrial subunit in Salmonella would modify the induction of Treg cells, spleens, MLNs, and HNLNs were harvested from SJL mice 14 days post-vaccination with H647, AP331, and H696, as well as from naïve SJL mice. The presence of Treg cells was analyzed by FACS (Fig 5A). A single oral dose with the Salmonella vaccines induced significant increases in the percentages of CD25+CD4+ T cells in spleens and LNs when compared to percentages obtained from naïve SJL mice (Fig. 5A and Tables III and IV; P < 0.001). Increased FoxP3+ CD25+ T cells were obtained in mice vaccinated with H696 when compared to the same cells induced in spleens and LNs of H647- and AP331-vaccinated mice (Fig. 5B and Tables III and IV; P < 0.001). In fact, the percentages of induced FoxP3+ Treg cells by Salmonella-CFA/IIC more resembled the Salmonella vector than Salmonella-CFA/I.
FIGURE 5.
Oral vaccination with Salmonella vaccines stimulates the induction of FoxP3+ Treg cells in mice. (A) Spleens and lymph nodes were harvested from SJL mice 14 days after immunization with Salmonella vaccines, and CD25+CD4+ T cells were analyzed by flow cytometry. Each of the Salmonella vaccines stimulated enhanced expression of Treg cells when compared to naïve mice, albeit, vaccination with H696 induced the greatest increases in the CD25+CD4+ T cells, as well as (B) the percentages of FoxP3+ CD25+CD4+ T cells. (C) The percentages of FoxP3+ CD25−CD4+ T cells were also enhanced after oral vaccination with each of the Salmonella strains when compared to naïve mice. Depicted are the combined results from two separate experiments for a total of 12 samples/group ± SEM *, P < 0.001 for naïve vs. treated groups, and differences between treated groups.
Table III.
Characterization of splenic CD4+ T cells from naïve, H647-, AP331-, and H696- immunized SJL mice
| T cells | H647 | AP331 | H696 | Naïve |
|---|---|---|---|---|
| CD25+CD4+ | 11.5 ± 3.1 | 13.0 ± 0.4 * | 16.3 ± 1.5 *,*,* | 4.8 ± 5.5 |
| FoxP3+ | 70.6 ± 7.2 *,* | 65.2 ± 6.2 * | 89.2 ± 7.4 *,*,* | 43.3 ± 3.8 |
| TGF-β+ | 15.9 ± 5.0 *,* | 10.7 ± 0.5 | 22.2 ± 5.1 *,*,* | 3.5 ± 1.5 |
| GITR+ | 38.1 ± 4.2 | 36.5 ± 2.6 | 35.8 ± 7.4 | 33.3 ± 2.1 |
| CCR6+ | 42.1 ± 5.5 *,*,* | 31.2 ± 3.4 *,* | 26.4 ± 4. | 25.2 ± 0.2 |
| CTLA-4+ | 31.3 ± 3.8 | 32.4 ± 8.6 | 61.5 ± 7.5 *,*,* | 34.3 ± 5.2 |
| RANKL+ | 3.8 ± 0.02 | 3.3 ± 2.5 | 3.8 ± 4.2 | 3.0 ± 5.5 |
|
| ||||
| CD25−CD4+ | ||||
| FoxP3+ | 15.2 ± 2.4 * | 18.2 ± 2.3 *,* | 19.4 ± 0.6 *,*,* | 11.0 ± 2.1 |
| TGF-β+ | 1.5 ± 0.0 | 1.5 ± 0.2 | 1.9 ± 0.1 | 1.2 ± 0.2 |
| GITR+ | 11.2 ± 0.7 | 10.6 ± 0.6 | 10.9 ± 0.4 | 9.9 ± 0.1 |
| CCR6+ | 12.5 ± 0.4 | 10.9 ± 0.5 | 12.6 ± 0.3 | 9.6 ± 0.2 |
| CTLA-4+ | 4.4 ± 0.1 | 5.3 ± 0.2 | 13.3 ± 1.6 *,*,* | 3.3 ± 0.3 |
| RANKL+ | 0.9 ± 0.1 | 0.4 ± 0.3 | 0.9 ± 0.2 | 0.3 ± 0.5 |
,P < 0.001 for naïve vs. treated groups; H696- vs. AP331-treated groups; and H647- vs. AP331-treated groups.
Table IV.
Characterization of LN CD4+ T cells from naïve, H647-, AP331-, and H696- immunized SJL mice
| T cells | H647 | AP331 | H696 | Naïve |
|---|---|---|---|---|
| CD25+CD4+ | 13.7 ± 2.3 * | 12.9 ± 1.6 * | 19.9 ± 2.2 *,*,* | 4.6 ± 2.2 |
| FoxP3+ | 71.7 ± 10.0 | 53.3 ± 11.3 | 93.5 ± 7.1 *,*,* | 47.1 ± 6.7 |
| TGF-β+ | 21.7 ± 3.0 *,* | 14.0 ± 7.1 * | 32.0 ± 4.3 *,* | 9.8 ± 2.6 |
| GITR+ | 33.1 ± 7.6 *,* | 32.0 ± 6.0 *,* | 25.0 ± 1.3 * | 21.2 ± 3.0 |
| CCR6+ | 19.0 ± 1.5 *,* | 16.0 ± 2.1 * | 15.5 ± 1.7 * | 12.3 ± 2.1 |
| CTLA-4+ | 24.6 ± 6.5 * | 19.1 ± 4.5 | 51.9 ± 7.6 *,*,* | 24.8 ± 3.1 |
| RANKL+ | 12.2 ± 2.0 | 10.9 ± 3.6 | 20.2 ± 1.6 *,*,* | 6.5 ± 5.5 |
|
| ||||
| CD25−CD4+ | ||||
| FoxP3+ | 17.7 ± 2.3 * | 16.4 ± 2.7 * | 24.2 ± 1.2 *,*,* | 12.7 ± 1.5 |
| TGF-β+ | 2.1 ± 0.4 | 1.65 ± 0.6 | 3.16 ± 0.8 | 1.1 ± 0.1 |
| GITR+ | 6.8 ± 0.5 | 6.8 ± 0.2 | 8.22 ± 1.05 | 5.5 ± 0.9 |
| CCR6+ | 9.73 ± 0.5 | 10.9 ± 0.7 | 10.4 ± 0.8 | 8.8 ± 0.5 |
| CTLA-4+ | 2.3 ± 0.6 | 5.3 ± 1.1 | 5.2 ± 1.2 | 2.5 ± 0.7 |
| RANKL+ | 1.7 ± 1.1 | 2.5 ± 1.2 | 15.3 ± 5.2 *,*,* | 0.5 ± 1.5 |
,P < 0.001 for naïve vs. treated groups; H696- vs. AP331-treated groups; and H647- vs. AP331-treated groups.
Since AP331 and H696 vaccines were equivalent in their protective efficacy for EAE, subsequent phenotypic analyses were performed to determine their differences and similarities among the Treg cell subsets. One aspect that defined Treg cells from AP331-dosed mice was the preponderance of IL-13+ Treg cells when compared to Salmonella vector- or Salmonella-CFA/I-dosed mice (Fig. 6C). Similar to H647-derived Treg cells, AP331-immunized mice showed enhanced IFN-γ+ Treg cells (Fig. 6C), but the percentages of TGF-β+ Treg cells were significantly higher in spleens and LNs of mice immunized with H647 and H696 when compared to AP331 (Fig. 6C and Tables III and IV; P < 0.001).
FIGURE 6.
Increased expression of IL-13+ and IFN-γ+ FoxP3+ CD25+ CD4+ T cells subsequent oral vaccination with Salmonella-CFA/IIC (AP331). Spleens and HNLNs from SJL mice immunized with Salmonella vector (H647), Salmonella-CFA/I (H696), and AP331 strains were harvested 14 days post-immunization, and (A) FoxP3+ CD25+CD4+ T cells were analyzed by FACS (B, C) to determine their cytokine profiles by intracellular staining for IFN-γ, IL-10, IL-13, or cell-surface TGF-β expression. Splenic and HNLN FoxP3+ Treg cells obtained from AP331-dosed mice shared features of both H647- and H696-immunized mice. Resembling H647-dosed mice, an elevated percentage of IFN-γ+ Treg cells and a reduced level of TGF-β+ Treg cells were obtained; resembling H696-dosed mice, an elevated percentage of IL-13+ Treg cells was observed. The percent IL-10+ Treg cells were the same for each immunized group and tissue evaluated. Insets in B represent the isotype control for intracellular cytokine staining. Depicted are the results from 6 – 9 mice/group: *, P < 0.001, **, P = 0.006, ***, P = 0.040, represent differences in cytokine-positive FoxP3+ Treg cells from H647-vaccinated mice versus AP331- or H696-vaccinated mice. †, P < 0.001, represents differences in cytokine-positive FoxP3+ Treg cells from AP331-vaccinated mice versus H696-vaccinated mice.
Other potential surface phenotypic markers for Treg cells were evaluated, including CTLA-4, GITR, and CCR6. Immunization of mice with H696 induced a significant enhancement (P < 0.001) of CTLA-4 expression by Treg cells with 61.5% in the spleen (Table III) and 51.9% in LNs (Table IV). Only half as many Treg cells expressed CTLA-4 in naïve, H647-, or AP331-dosed mice. Interestingly, splenic and LN H647-Treg cells showed significantly higher levels of CCR6 than naïve, AP331, and H696 Treg cells (P < 0.001; Tables III and IV). Expression of CCR6 by splenic AP331 Treg cells was also slightly greater than H696 or naïve Treg cells (P < 0.001; Table III). Expression of GITR seemed to correlate less among the evaluated groups, although H647-Treg cells also showed the highest GITR levels.
Similar evaluations were performed with CD25−CD4+ T cells. Clearly, a subset of these cells from mice immunized with H696 showed enhanced expression of FoxP3 when compared to naïve-, H647-, and AP331-CD25−CD4+ T cells in spleens and LNs (P < 0.001; Fig. 5C, Table III and IV, respectively). Interestingly, RANKL was found to be associated mostly with LN Treg cells and CD25−CD4+ T cells and not with splenic CD4+ T cells (Tables III and IV). Past studies have shown these RANKL+ T cells to be mostly associated with pathogenic CD4+ T cells (26, 27).
Adoptive transfer of CFA/I fimbriae-induced Treg cells protects against EAE
Previous studies have shown that Treg cells obtained from EAE-induced mice were able to protect when adoptively transferred into susceptible naïve mice and when subsequently challenged with myelin antigens to induce EAE (28–30). In contrast to innate Treg cells, it was previously shown that Salmonella-CFA/I-induced Treg cells were able to potently protect against EAE (19). To understand differences in the mechanism of protection conferred by Salmonella-CFA/IIC by Salmonella-CFA/I immunization, cell-sorting experiments were performed 14 days post-immunization with H647, AP331, or H696 Salmonella strains to obtain Treg cell and CD25−CD4+ T cell subsets to evaluate their cytokine profile after in vitro stimulation with anti-CD3 and anti-CD28 mAbs. Treg cells sorted from Salmonella-CFA/IIC-immunized mice produced significantly (P < 0.001) greater levels of IFN-γ when compared to H647- or H696-induced or naïve Treg cells (Fig. 7B). AP331 induced-Treg cells also produced significantly greater IL-10 and IL-13 when compared to naïve, H647-, and H696-induced-Treg cells (P < 0.001). In contrast, H696 induced-Treg cells produced significantly more TGF-β and IL-4, which is consistent with that previously reported (19). Minimal to no IL-17, IL-21, or IL-22 was observed with any of the vaccinated or naïve groups.
FIGURE 7.
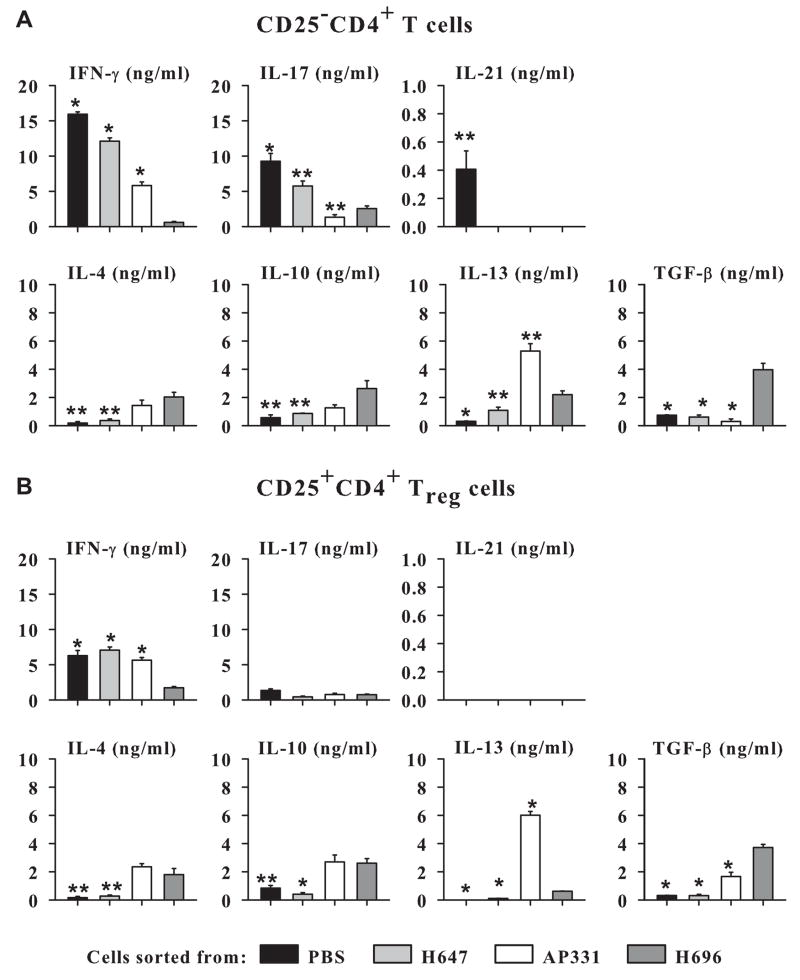
Oral immunization of SJL mice with Salmonella-CFA/IIC induces Treg cells to produce elevated levels of IFN-γ and IL-13, but does not induce immune deviation. (A) CD25−CD4+ T cells and (B) CD25+CD4+ Treg cells from SJL mice vaccinated with H647, AP331, or H696, and from naïve mice were co-stimulated in vitro with anti-CD3 and anti-CD28 mAbs, and cytokine production was measured from culture supernatants after 72 h. (A) CD25−CD4+ T cells from AP331- vaccinated mice failed to show any significant difference in cytokine production from naïve mice. H647-CD25−CD4+ T cells showed enhanced IFN-γ, and H696-CD25−CD4+ T cells showed elevated production of IL-4, IL-10, and IL-13. (B) Treg cells elicited by AP331 vaccination produced enhanced levels of IFN-γ, IL-10, and IL-13 when compared to Treg cells H647- or H696- vaccinated or naïve mice. In contrast, H696 induced-Treg cells produced elevated levels of IL-4 and TGF-β. Depicted are the combined results from two separate experiments for a total of 12 samples/group ± SEM. *, P < 0.001 represents differences in cytokine production between Salmonella-vaccinated groups and naïve mice.
Cytokine profiles for CD25−CD4+ T cells were also performed. Strikingly elevated IFN-γ was produced by H647-CD25−CD4+ T cells with the production of IL-4, IL-10, IL-13, and IL-22 not being different from naïve mice (Fig. 7A). In contrast, H696 immunization of SJL mice produced a significant Th2-type response evident by elevated IL-4 and IL-13 with a modest IL-10 production (Fig. 7A). The CD25−CD4+ T cells obtained from the AP331-dosed mice displayed aspects of both H647- or H696-vaccinated mice. Similar to H647-induced responses, minimal to no Th2-type cytokines were produced, and similar to H696-induced responses, diminished IFN-γ and modest increases in IL-22 were obtained with the AP331-immunized mice (Fig. 7A). Thus, unlike H696, the Salmonella-CFA/IIC lacks the ability to stimulate immune deviation by CD25−CD4+ T cells producing Th2-type cytokines, although these vaccinated mice clearly are able to produce regulatory cytokines by their Treg cell subset.
In order to determine the potency of Treg cells induced by Salmonella-CFA/IIC when compared to Salmonella-CFA/I, adoptive transfer experiments were performed. SJL mice were orally immunized with a single dose of H647, AP331, or H696, and 14 days after immunization, CD25− CD4+ T cells and Treg cells were sorted from spleens and HNLNs (purity of isolated cells was determined by FACS analysis: > 90% for CD25+CD4+ Treg cells and 99% for CD25− CD4+ T cells; Fig. 8A). 6 × 105 Treg or CD25−CD4+ T cells were i.v. injected into naïve recipient mice, and one day later, mice were challenged with PLP135–151. Adoptive transfer of mice with CD25− CD4+ T cells from H647-dosed mice failed to confer protection against EAE challenge, and no significant differences were observed when compared to the PBS-treated control mice that developed the expected EAE disease (Fig. 8B). Recipient mice given CD25− CD4+ T cells from AP331 donor mice showed a significant delay in the EAE clinical onset when compared to H696-induced CD25−CD4+ T cells (P < 0.001); however, 15 days post-challenge, no significant differences were observed between both groups. Thus, recipient mice given CD25−CD4+ T cells obtained from mice dosed with either CFA/I vaccines were equally protected. In a similar fashion, donor Treg cells obtained from H696- or AP331-mice and adoptively transferred into challenged recipient mice were able to confer nearly complete protection (Fig. 8B), albeit, via different mechanisms. In both recipient groups, a significant reduction in the EAE clinical scores was observed in challenged mice when compared to the challenged PBS-treated mice. No statistical differences were observed in the minimal to no clinical scores of mice treated with AP331- and H696-Treg cells.
FIGURE 8.
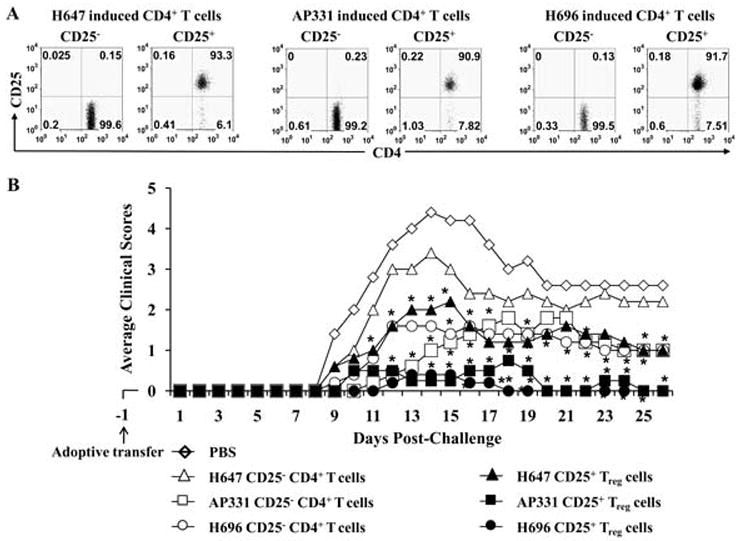
Adoptive transfer of Treg cells and, to a lesser extent, CD25−CD4+ T cells from mice orally immunized with Salmonella-CFA/IIC (AP331) conferred protection against EAE. (A) CD25−CD4+ T cells and CD25+CD4+ T cells were sorted from spleens and HNLN of SJL mice 14 days post-immunization with AP331, Salmonella-CFA/I (H696), or Salmonella vector (H647). Purity of isolated cells was determined by FACS analysis (> 90% for Treg cells and 99% for CD25−CD4+ T cells). (B) 6 × 105 Treg or CD25−CD4+ T cells from each Salmonella vaccination group were i.v. injected into naïve recipient mice, and one day later, mice were challenged with PLP135–151. Adoptive transfer of Treg cells from AP331- and H696-dosed mice conferred nearly complete protection against EAE when compared to the PBS-treated mice, while H647-Treg cells were only partially protective. Adoptive transfer of CD25−CD4+ T cells from AP331- and H696-dosed mice were partially protective, although the AP331-induced CD25−CD4+ T cells showed a significant delay in the EAE clinical outcome when compared to H696-induced CD25−CD4+ T cells (P < 0.001), but by 15 days post-challenge, no significant differences were observed between these groups. H647-induced CD25−CD4+ T cells did not confer any protection against EAE. Depicted results are from a total of 5 mice/group. *, P < 0.001, for adoptively transferred CD25+ CD4+ T cells vs. PBS-dosed mice; †, P < 0.001 for adoptively transferred CD25−CD4+ T cells vs. PBS-dosed mice.
Salmonella-CFA/IIC-induced Treg cells protect via IL-13
Evaluation of cytokines produced by Treg cells subsequent to oral immunization with Salmonella-CFA/IIC suggested that IL-13 and IFN-γ (Figs. 6 and 7) may be important for the observed protective responses obtained in the adoptive transfer studies (Fig. 8). In order to determine the relative importance of IL-13 and IFN-γ in the protection conferred by AP331-induced Treg cells, in vivo cytokine neutralization studies were performed at the time of adoptively transferred Treg cells to assess their impact upon protection against EAE (Figs. 9 and 10). As before, 6 × 105 Treg cells sorted from PBS-, H647-, and AP331-immunized SJL mice were adoptively transferred (i.v.) into naïve SJL recipient mice, and one day after, EAE was induced. For IL-13 neutralization, 0.5 ml of rabbit anti-rat IL-13 serum (25) was administered one day before EAE induction, and two additional doses of 0.25 ml were each given on days 1 and 5 post-EAE challenge (Fig. 9). In vivo neutralization of IL-13 resulted in the loss of protection conferred by adoptively transferred Treg cells obtained from AP331. Treatment with anti-IL-13 or NRS upon EAE challenged mice given Treg cells obtained from H647-immunized mice had no significant effect upon the disease course. Likewise, treatment of PBS-group with anti-IL-13 or NRS did not impact EAE clinical scores. These results demonstrate that IL-13 is essential for protection conferred by Treg cells induced upon immunization with the AP331 vaccine.
FIGURE 9.
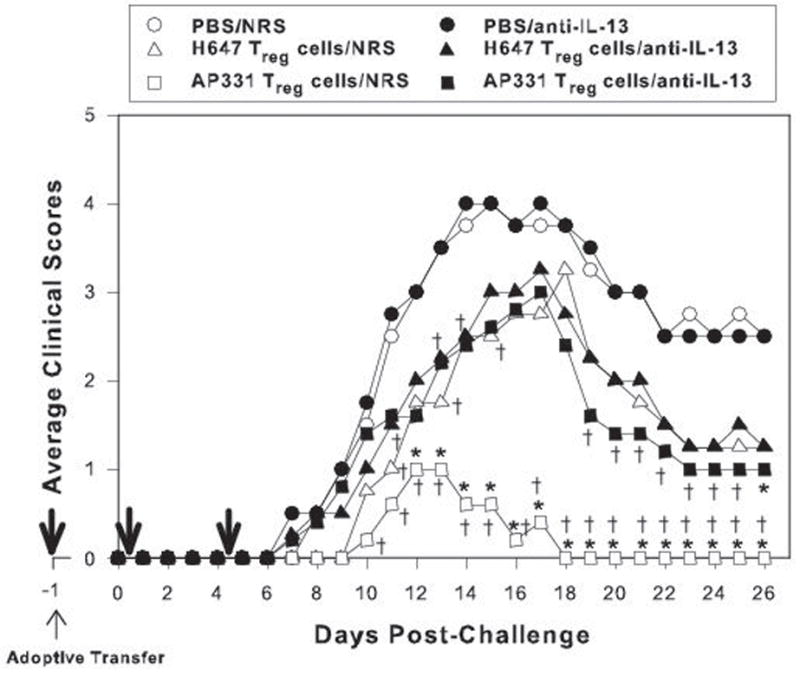
IL-13 produced from mice adoptively transferred with Treg cells from Salmonella-CFA/IIC (AP331)-immunized mice protects against EAE. Cell-sorted 6 × 105 Treg cells from Salmonella vector (H647)- and AP331-immunized SJL mice were adoptively transferred (i.v.) into naïve SJL recipient mice, and one day later, EAE was induced with PLP139–151. To neutralize IL-13, rabbit antiserum (0.5 ml) was given one day before EAE induction, and two additional doses (0.25 ml each) were given on days 1 and 5 post-EAE challenge (thick arrows). Equivalent volumes of normal rabbit serum (NRS) were given to control groups. In vivo neutralization of IL-13 of recipient mice given AP331-induced Treg cells reversed the protection conferred by these T cells, and their clinical scores resembled mice adoptively transferred with H647-induced Treg cells. Anti-IL-13 treatment had no impact upon EAE in PBS-treated or mice adoptively transferred with Treg cells from H647-immunized mice. NRS treatments had no negative impact upon the various treated groups. Depicted results are from a total of 5 mice/group. *, P < 0.001, for anti-IL-13 vs. NRS treatment with the adoptively transferred Treg cell groups; †, P < 0.001 for PBS vs. anti-IL-13 and NRS treatment with the adoptively transferred Treg cell groups.
FIGURE 10.
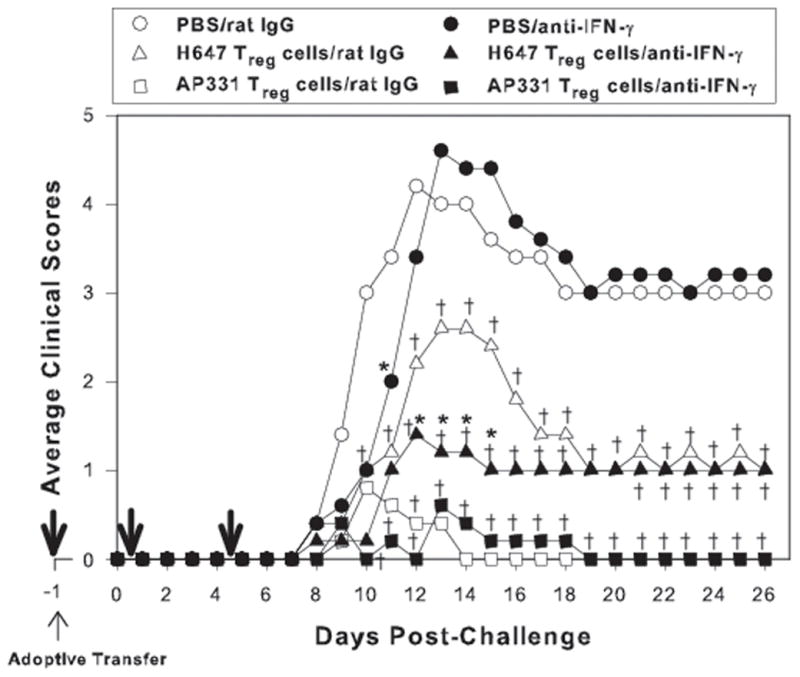
In vivo neutralization of IFN-γ fails to augment clinical disease in mice adoptively transferred with CD25+CD4+ T (Treg) cells from Salmonella-CFA/IIC (AP331)-vaccinated mice. Cell-sorted 6 × 105 Treg cells obtained from Salmonella vector (H647)- or AP331-vaccinated SJL mice were adoptively transferred (i.v.) into naïve SJL recipient mice, and one day later, EAE was induced with PLP139–151. To neutralize IFN-γ, 0.5 mg of anti-mouse IFN-γ mAb was given one day before EAE induction, and two additional doses of 0.25 mg were given on days 1 and 5 post-EAE challenge (thick arrows). Equivalent amounts of normal rat IgG Ab were given to control groups. In vivo` neutralization of IFN-γ of recipient mice given AP331-induced Treg cells only delayed EAE onset and did not cause a loss in the ability to confer protection when compared similarly treated mice given rat IgG, suggesting that IFN-γ is not required for protection by the AP331 vaccine. In contrast, in vivo neutralization of IFN-γ neutralization of recipient mice given H647-induced Treg cells significantly reduced their clinical scores when compared to similarly treated mice given rat IgG, suggesting that IFN-γ contributes to EAE when using the Salmonella vector vaccine. In vivo IFN-γ neutralization showed a slight delay in PBS-treated mice, but similar EAE clinical outcomes when compared to rat IgG-treated mice were observed. Depicted results are from a total of 5 mice/group. *, P < 0.001, for anti-IFN-γ treatment vs. IgG treatment within the same adoptively transferred Treg cell groups; †, P < 0.001 for PBS vs. anti-IFN-γ- or rat IgG-treated Treg cell groups.
To assess the role of IFN-γ, in vivo neutralization was performed by the administration of anti-IFN-γ mAb on days −1, +1, and +5 post-EAE induction with PLP139–151 (Fig. 10). In vivo blockade of IFN-γ in mice treated with PBS did not provoke significant differences in the EAE clinical outcome when compared to rat IgG-treated mice. The neutralization of IFN-γ significantly reduced the clinical scores of H647-derived Treg cells when compared to the similar groups treated with rat IgG Ab. No difference in protection was obtained using the AP331-derived Treg cells, further suggesting that the Treg cells producing IL-13 are mediating protection. Thus, these studies show that in vivo reduction of IFN-γ has minimal impact upon protection conferred by AP331-induced CD25+ CD4+ T cells, but enhances the protection by H647-induced Treg cells.
Discussion
In EAE, encephalitogenic T cells secrete inflammatory cytokines, inducing the activation of macrophages and microglial cells and the infiltration of inflammatory cells from peripheral lymphoid tissues (10, 11). Recent studies have reported the potential for developing therapeutic Treg cells independently of auto-Ag for treating EAE (18, 19) by down-regulating inflammatory mediators and inhibiting autoimmune aggressive reactions. Alternatively, significant efforts have focused to treat autoreactive T cells by vaccinating with altered peptide ligands (30, 31), isolating Treg cells from diseased animals (29), or stimulating Treg cells in vitro (30). Since it is often unclear to which myelin epitope(s) MS patients may be reacting, a strategy not relying upon known epitopes may be advantageous. Along these lines, we demonstrated that by using a live attenuated Salmonella Typhimurium strain expressing on its cells’ surface, the enterotoxigenic E. coli CFA/I fimbriae was able to protect SJL mice against EAE by preventing encephalitogenic T cells from entering the CNS (18). Likewise, oral treatment with the vaccine ceased subsequent demyelination by the stimulation of FoxP3+ CD25+CD4+ T cells (19).
In an attempt to disrupt the therapeutic potential of Salmonella-CFA/I vaccine, this current work queried whether altering the expression of the CFA/I fimbrial subunit within Salmonella’s periplasmic/cytoplasmic compartments would dampen protective immunity to EAE. Instead, these studies revealed the stimulation of different Treg cell subsets became evident by differences in cytokine profiles and cell surface expression of various regulatory-associated molecules. Nonetheless, the protective efficacy against EAE was conserved, but via different mechanisms. One advantage of this new construct, Salmonella-CFA/IIC (strain AP331), is that the host response against the fimbrial subunit was dampened (lessened Ab titers), but still retained protective qualities. It is not as evident that the presence of a CD25− Th2 cell subset was responsible for immune deviation as was found with the intact Salmonella-CFA/I vaccine. In this latter construct, there is a dominance of IL-4-, IL-10-, IL-13-, and TGF-β-producing Th2 cells by CD25−CD4+ T cells, and while not as protective as their induced Treg cells, these Th2 cells clearly contribute to protection against EAE (this study and in 19). However, the AP331-induced CD25−CD4+ T cells do seem to prime for immune deviation, as evident by the 73-fold increased production of IL-13 following challenge. Likewise, immunization with Salmonella-CFA/IIC enhanced the production of IL-13 by Treg cells by 5.6-fold subsequent EAE challenge. Despite these differences in capacity to produce IL-13, it was quite evident that nearly all of the protective activity is with the AP331-induced Treg cells since adoptive transfer of these Treg cells, not their corresponding CD25−CD4+ T cells, was capable of imparting nearly full protection against EAE. Given this finding, in vivo neutralization of IL-13 upon adoptive transfer of AP331-induced Treg cells reversed its ability to protect showing clinical EAE, and the manifestation of EAE in these mice was similar to that induced in mice adoptively transferred with Salmonella vector-induced Treg cells. Although a portion of the Salmonella-CFA/IIC-induced Treg cells produced IFN-γ, in vivo neutralization of IFN-γ upon adoptive transfer of AP331-induced Treg cells had no effect upon the development of EAE. Thus, the protection conferred by Salmonella-CFA/IIC is mostly served by IL-13-producing Treg cells.
To aid in discerning how the two CFA/I-expressing Salmonella vaccines differed, studies were performed to assess potential phenotypic differences between the induced Treg cells. The Salmonella-CFA/I (strain H696) vaccine clearly elicited >90% of the CD25+CD4+ T cells being FoxP3+, whereas CD25+CD4+ T cells from Salmonella-CFA/IIC varied from 40 – 70%; yet both vaccines induced similar splenic levels of FoxP3+ CD25−CD4+ T cells. CTLA-4 has also been shown to be expressed by Treg cells (32–34). In some cases, activated Treg cells express higher levels of CTLA-4, down-regulating T cell proliferative responses induced by dendritic cells by modifying their B7 co-stimulatory pathways (35). Our results showed that CTLA-4 was more prevalent on Salmonella-CFA/I-induced Treg cells than in Treg cells from naïve mice or those induced by the Salmonella empty vector or Salmonella-CFA/IIC. Other regulatory-associated molecules were also evaluated, including GITR (29, 36) and CCR6 (37, 38). GITR expression was only modestly enhanced on LN Treg cells from Salmonella vector- or by AP331-immunized mice; however, CCR6 expression was elevated for splenic Treg cells and again from H647- and AP331-immunized mice. RANKL expression was examined as a possible means to help identify pathogenic CD4+ T cells, as others have shown for periodontal disease (26) and diabetes (27). Instead, higher percentages of LN RANKL+ Treg cells were found with those from H696-vaccinated mice, as well as with LN CD25−CD4+ T cells. Thus, while Treg cells from Salmonella-CFA/I-vaccinated mice showed distinctive expression of FoxP3 and CTLA-4, a distinctive phenotype for the Treg cells from Salmonella-CFA/IIC was not as evident.
The relevance of IL-13 upon protection against EAE was quite evident from its in vivo neutralization, showing loss of protection by the Salmonella-CFA/IIC-induced Treg cells. Clearly, the restimulation assays of CD4+ T cell subsets, as well as FACS analysis of FoxP3+ Treg cells, showed dominance of IL-13. Only a few studies have evaluated the relevance of IL-13 to EAE. Using recombinant TCRs with covalently bound encephalitogenic peptide, it was shown to be partially protective against EAE, which was, in part, attributed to enhanced IL-13 production, as well as other anti-inflammatory cytokines (39). The observed induction of IL-13 may be IL-25-dependent since a recent study has shown that IL-4 and IL-13 are depressed in IL-25-deficient mice challenged with EAE (40). Moreover, they showed that the protection conferred by IL-25 was lost in IL-13-deficient mice (40). These collective findings, along with our observations, showed that IL-13 represents an alternative therapeutic for treating autoimmune diseases. Of interest, IL-22 was induced upon oral vaccination with either CFA/I fimbrial subunit-expressing Salmonella vaccines, less in naïve mice or mice orally vaccinated with the Salmonella empty vector, and none was detected after EAE challenge. This association with an anti-inflammatory response has been previously reported, showing that IL-22-deficient mice are susceptible to ConA-induced hepatocyte inflammation (41). While in our study, only minimal IL-22 levels were detected. IL-22 has been recently shown to be produced during myelin oligodendrocyte glycoprotein-induced EAE, but development of EAE in IL-22-deficient mice was not affected (42); thus, its presence may have minimal impact upon EAE.
Another feature of the Salmonella-CFA/IIC vaccine was its ability to stimulate IFN-γ+ Treg cells, and thus, we questioned the significance of this finding. In vivo neutralization of IFN-γ did not affect the potency of the adoptively transferred Treg cells from the Salmonella-CFA/IIC- vaccinated mice. The potency of these IFN-γ+ Treg cells to treat EAE might not have been affected because of the presence of other Treg cells producing IL-10 and IL-13, and these Treg cells may limit the action of the IFN-γ+ Treg cells. Such observations contrasted to what was obtained when similar treatment paradigm was applied to mice adoptively transferred with Treg cells from Salmonella vector-immunized mice. Instead of being minimally therapeutic, protection was now conferred by Salmonella vector-induced Treg cells, as noted by the reduced clinical scores subsequent PLP139–151 challenge. Thus, the in vivo neutralization of IFN-γ significantly augmented the protection by the Salmonella vector-induced Treg cells. These variable results obtained after IFN-γ blockade might be related to our experimental approach, since in vivo neutralization of IFN-γ not only might neutralize potential production of IFN-γ by Treg cells or effect their activation or potency, but also reduce the biological activities of the cytokine during the entire course of the disease. The role of IFN-γ in the development or even in the resolution of EAE has remained in a quagmire until the recent discovery of IL-17, subsequently identified as the primary inflammatory mediator responsible for the development of EAE (43–45). Neutralization of IFN-γ via mAbs (46, 47), IFN-γ-deficient mice (48, 49), or IFN-γ receptor-deficient mice (50) has resulted in enhanced EAE, suggesting that IFN-γ is important for protection by possibly dampening IL-17 generation, as recently suggested (11, 51, 52). In contrast, delayed neutralization of IFN-γ after and not upon initial EAE challenge with anti-IFN-γ mAbs protected EAE-susceptible animals (53, 54). Examination of what may be occurring upon IFN-γ neutralization during adoptive transfer with AP331-induced Treg cells revealed a notable absence of an effect, suggesting that the FoxP3+ IFN-γ+ Treg cells are not pathogenic and that the produced IFN-γ has a minimal impact in the presence of the co-produced regulatory cytokines. In contrast, since the Salmonella vector produces minimal amounts of regulatory cytokines, its production of IFN-γ may be more pathogenic in this vaccination scheme, and upon neutralization of IFN-γ during adoptive transfer with H647-induced Treg cells, enhanced protection is obtained.
It was originally hypothesized that altering the expression of the CfaB fimbrial subunit could impact the types of Th cells induced, as previously observed in altering FanC expression for K99 fimbriae by Salmonella (23). While the induced Th cells were altered, as described here with the AP331 strain, protection against EAE was still maintained. Both AP331 and H696 strains produced similar levels of CfaB subunit despite differences in location of expression, e.g., AP331 that localized CfaB to cytoplasm/periplasm as determined in this study and H696 localized CfaB to cell surface as determined by electron microscopy (6). Thus, the differences in Th cell responses must be attributed to differences in the mode of Ag recognition and how APCs are recognizing/displaying CfaB subunit. Future studies will address this observation in greater detail since these studies could reveal potential mechanisms of action by simply altering the compartment by which passenger Ags are expressed within Salmonella vaccine vectors.
The described work suggests that regulatory T cells, in particular, CD25+CD4+ T cells, have varied patterns of cytokine production displaying the conventional pattern of regulatory cytokines, as well as displaying IFN-γ. Not surprisingly, this latter finding may be expected when adapting live attenuated Salmonella vaccines. Again, in the absence of auto-Ag, the varied Salmonella vaccines are protective against EAE, and the mechanism of protection is also varied. The data show that mode of CFA/I fimbrial subunit expression impacts the type of Treg cells induced. When the fimbrial subunit is on the cell surface, >90% of CD25+CD4+ T cells are FoxP3+ and produce a number of regulatory cytokines, including IL-4, IL-10, IL-13, and TGF-β. In addition, an impact via immune deviation is also induced by the stimulation of CD25− Th2 cells. Upon internalization of the fimbrial subunit, there is a reduction in the percentage of FoxP3+ Treg cells, as well as a notable bias for IL-13. The stimulation of CD25− Th2 cells is less noticeable, but the data suggest there is a priming effect for the generation of IL-13 that is greatly augmented during EAE challenge. Despite these differences in mechanisms, the Salmonella-CFA/IIC vaccine is equally protective against EAE, as is the Salmonella-CFA/I vaccine. Added to this mix, the Salmonella vector is also protective against EAE when IFN-γ is neutralized. Thus, these findings further expand the potential repertoire of Treg cell subtypes that can be induced by Salmonella vaccines and may be beneficial for a variety of other autoimmune diseases. Moreover, these studies suggest vaccines can be designed to elicit the desired Treg cell subtype.
Acknowledgments
We thank Dr. Mark. A. Jutila, VMB, Montana State University, Bozeman, MT, for kindly providing the SK208 mAb, Ms. Larrisa Jackiw for the cell-sorting experiments, and Ms. Nancy Kommers for her assistance in preparing this manuscript.
Footnotes
This work was supported by Public Health Service grant AI-41123 and, in part, by Montana Agricultural Station and U.S. Department of Agriculture Formula Funds. The VMB flow cytometry facility was, in part, supported by NIH/National Center for Research Resources, Centers of Biomedical Excellence P20 RR-020185, and an equipment grant from the M.J. Murdock Charitable Trust.
Abbreviations used in this paper: CFA/I, colonization factor antigen I; CFA/IIC, CFA/I expressed in the periplasmic/intracellular compartment; CLNs, cervical lymph nodes; CM, complete medium; DC, dendritic cell; EAE, experimental autoimmune encephalomyelitis; GITR, glucocorticoid-induced TNF receptor; HNLNs, head and neck lymph nodes; LFB, luxol fast blue; MLNs, mesenteric lymph nodes; MS, multiple sclerosis; NRS, normal rabbit serum; PLP, protein proteolipid; RANKL, receptor activator of nuclear factor kappaB ligand; Treg, regulatory T cells.
References
- 1.Sztein MB. Cell-mediated immunity and antibody responses elicited by attenuated Salmonella enterica Serovar Typhi strains used as live oral vaccines in humans. Clin Infect Dis. 2007;45:S15–S19. doi: 10.1086/518140. [DOI] [PubMed] [Google Scholar]
- 2.Pascual DW, Ochoa-Repáraz J, Rynda A, Yang X. Tolerance in the absence of auto-antigen. Endocr Metab Immune Disord Drug Targets. 2007;7:203–210. doi: 10.2174/187153007781662549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mastroeni P, Villarreal-Ramos B, Hormaeche CE. Role of T cells, TNF-alpha and IFN-gamma in recall of immunity to oral challenge with virulent salmonellae in mice vaccinated with live attenuated aro- Salmonella vaccines. Microb Pathog. 1992;13:477–491. doi: 10.1016/0882-4010(92)90014-f. [DOI] [PubMed] [Google Scholar]
- 4.Okahashi N, Yamamoto M, Vancott JL, Chatfield SN, Roberts M, Bluethmann H, Hiroi T, Kiyono H, McGhee JR. Oral immunization of interleukin-4 (IL-4) knockout mice with a recombinant Salmonella strain or cholera toxin reveals that CD4+ Th2 cells producing IL-6 and IL-10 are associated with mucosal immunoglobulin A responses. Infect Immun. 1996;64:1516–1525. doi: 10.1128/iai.64.5.1516-1525.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.VanCott JL, Staats HF, Pascual DW, Roberts M, Chatfield SN, Yamamoto M, Coste M, Carter PB, Kiyono H, McGhee JR. Regulation of mucosal and systemic antibody responses by T helper cell subsets, macrophages, and derived cytokines following oral immunization with live recombinant Salmonella. J Immunol. 1996;156:1504–1514. [PubMed] [Google Scholar]
- 6.Pascual DW, Hone DM, Hall S, van Ginkel FW, Yamamoto M, Walters N, Fujihashi K, Powell RJ, Wu S, Vancott JL, Kiyono H, McGhee JR. Expression of recombinant enterotoxigenic Escherichia coli colonization factor antigen I by Salmonella typhimurium elicits a biphasic T helper cell response. Infect Immun. 1999;67:6249–6256. doi: 10.1128/iai.67.12.6249-6256.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pascual DW, Trunkle T, Sura J. Fimbriated Salmonella enterica serovar Typhimurium abates initial inflammatory responses by macrophages. Infect Immun. 2002;70:4273–4281. doi: 10.1128/IAI.70.8.4273-4281.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol. 2005;23:683–747. doi: 10.1146/annurev.immunol.23.021704.115707. [DOI] [PubMed] [Google Scholar]
- 9.McFarland HF, Martin R. Multiple sclerosis: a complicated picture of autoimmunity. Nat Immunol. 2007;8:913–919. doi: 10.1038/ni1507. [DOI] [PubMed] [Google Scholar]
- 10.Juedes AE, Hjelmstrom P, Bergman CM, Neild AL, Ruddle NH. Kinetics and cellular origin of cytokines in the central nervous system: insight into mechanisms of myelin oligodendrocyte glycoprotein-induced experimental autoimmune encephalomyelitis. J Immunol. 2000;164:419–426. doi: 10.4049/jimmunol.164.1.419. [DOI] [PubMed] [Google Scholar]
- 11.Karpus WJ, Ransohoff RM. Chemokine regulation of experimental autoimmune encephalomyelitis: temporal and spatial expression patterns govern disease pathogenesis. J Immunol. 1998;161:2667–2671. [PubMed] [Google Scholar]
- 12.Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, Weiner HL, Kuchroo VK. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 13.Nurieva R, Yang XO, Martinez G, Zhang Y, Panopoulos AD, Ma L, Schluns K, Tian Q, Watowich SS, Jetten AM, Dong C. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature. 2007;448:480–483. doi: 10.1038/nature05969. [DOI] [PubMed] [Google Scholar]
- 14.Korn T, Bettelli E, Gao W, Awasthi A, Jäger S, Strom TB, Oukka M, Kuchroo VK. IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature. 2007;448:484–487. doi: 10.1038/nature05970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bettelli E, Korn T, Kuchroo VK. Th17: the third member of the effector T cell trilogy. Curr Opin Immunol. 2007;19:652–657. doi: 10.1016/j.coi.2007.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bailey SL, Schreiner B, McMahon EJ, Miller SD. CNS myeloid DCs presenting endogenous myelin peptides 'preferentially' polarize CD4+ T(H)-17 cells in relapsing EAE. Nature Immunol. 2007;8:172–180. doi: 10.1038/ni1430. [DOI] [PubMed] [Google Scholar]
- 17.Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity. 2006;24:179–189. doi: 10.1016/j.immuni.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 18.Jun S, Gilmore W, Callis G, Rynda A, Haddad A, Pascual DW. A live diarrheal vaccine imprints a Th2 cell bias and acts as an anti-inflammatory vaccine. J Immunol. 2005;175:6733–6740. doi: 10.4049/jimmunol.175.10.6733. [DOI] [PubMed] [Google Scholar]
- 19.Ochoa-Repáraz J, Riccardi C, Rynda A, Jun S, Callis G, Pascual DW. Regulatory T cell vaccination without autoantigen protects against experimental autoimmune encephalomyelitis. J Immunol. 2007;178:1791–1799. doi: 10.4049/jimmunol.178.3.1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu S, Pascual DW, VanCott JL, McGhee JR, Maneval DR, Jr, Levine MM, Hone DM. Immune responses to novel Escherichia coli and Salmonella typhimurium vectors that express colonization factor antigen I (CFA/I) of enterotoxigenic E. coli in the absence of the CFA/I positive regulator cfaR. Infect Immun. 1995;63:4933–4938. doi: 10.1128/iai.63.12.4933-4938.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sakellaris H, Munson GP, Scott JR. A conserved residue in the tip proteins of CS1 and CFA/I pili of enterotoxigenic Escherichia coli that is essential for adherence. Proc Natl Acad Sci U S A. 1999;96:12828–12832. doi: 10.1073/pnas.96.22.12828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang X, Hinnebusch BJ, Trunkle T, Bosio CM, Suo Z, Tighe M, Harmsen A, Becker T, Crist K, Walters N, Avci R, Pascual DW. Oral vaccination with Salmonella simultaneously expressing Yersinia pestis F1 and V antigens protects against bubonic and pneumonic plague. J Immunol. 2007;178:1059–1067. doi: 10.4049/jimmunol.178.2.1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ascón MA, Ochoa-Repáraz J, Walters N, Pascual DW. Partially assembled K99 fimbriae required for protection. Infect Immun. 2005;73:7274–7280. doi: 10.1128/IAI.73.11.7274-7280.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pascual DW, White M, Larson T, Walters N. Impaired mucosal immunity in L-selectin-deficient mice orally immunized with a Salmonella vaccine vector. J Immunol. 2001;167:407–415. doi: 10.4049/jimmunol.167.1.407. [DOI] [PubMed] [Google Scholar]
- 25.Lakkis FG, Cruet EN, Nassar GM, Badr KF, Pascual DW. Expression of recombinant rat interleukin-13 and generation of a neutralizing rat IL-13 antiserum. Biochem Biophys Res Commun. 1997;235:529–532. doi: 10.1006/bbrc.1997.6832. [DOI] [PubMed] [Google Scholar]
- 26.Teng YT, Mahamed D, Singh B. Gamma interferon positively modulates Actinobacillus actinomycetemcomitans-specific RANKL+ CD4+ Th-cell-mediated alveolar bone destruction in vivo. Infect Immun. 2005;73:3453–3461. doi: 10.1128/IAI.73.6.3453-3461.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mahamed DA, Marleau A, Alnaeeli M, Singh B, Zhang X, Penninger JM, Teng YT. G(−) anaerobes-reactive CD4+ T-cells trigger RANKL-mediated enhanced alveolar bone loss in diabetic NOD mice. Diabetes. 2005;54:1477–1486. doi: 10.2337/diabetes.54.5.1477. [DOI] [PubMed] [Google Scholar]
- 28.Kohm AP, Carpentier PA, Anger HA, Miller SD. Cutting edge: CD4+CD25+ regulatory T cells suppress antigen-specific autoreactive immune responses and central nervous system inflammation during active experimental autoimmune encephalomyelitis. J Immunol. 2002;169:4712–4716. doi: 10.4049/jimmunol.169.9.4712. [DOI] [PubMed] [Google Scholar]
- 29.McGeachy MJ, Stephens LA, Anderton SM. Natural recovery and protection from autoimmune encephalomyelitis: contribution of CD4+CD25+ regulatory cells within the central nervous system. J Immunol. 2005;175:3025–3032. doi: 10.4049/jimmunol.175.5.3025. [DOI] [PubMed] [Google Scholar]
- 30.Yu P, Gregg RK, Bell JJ, Ellis JS, Divekar R, Lee HH, Jain R, Waldner H, Hardaway JC, Collins M, Kuchroo VK, Zaghouani H. Specific T regulatory cells display broad suppressive functions against experimental allergic encephalomyelitis upon activation with cognate antigen. J Immunol. 2005;174:6772–6780. doi: 10.4049/jimmunol.174.11.6772. [DOI] [PubMed] [Google Scholar]
- 31.Young DA, Lowe LD, Booth SS, Whitters MJ, Nicholson L, Kuchroo VK, Collins M. IL-4, IL-10, IL-13, and TGF-β from an altered peptide ligand-specific Th2 cell clone down-regulate adoptive transfer of experimental autoimmune encephalomyelitis. J Immunol. 2000;164:3563–3572. doi: 10.4049/jimmunol.164.7.3563. [DOI] [PubMed] [Google Scholar]
- 32.Salomon B, Lenschow DJ, Rhee L, Ashourian N, Singh B, Sharpe A, Bluestone JA. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T cells that control autoimmune diabetes. Immunity. 2000;12:431–440. doi: 10.1016/s1074-7613(00)80195-8. [DOI] [PubMed] [Google Scholar]
- 33.Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J Exp Med. 2000;192:295–302. doi: 10.1084/jem.192.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Takahashi T, Kuniyasu Y, Toda M, Sakaguchi N, Itoh M, Iwata M, Shimizu J, Sakaguchi S. Immunologic self-tolerance maintained by CD25+CD4+ naturally anergic and suppressive T cells: induction of autoimmune disease by breaking their anergic/suppressive state. Int Immunol. 1998;10:1969–1980. doi: 10.1093/intimm/10.12.1969. [DOI] [PubMed] [Google Scholar]
- 35.Oderup C, Cederbom L, Makowska A, Cilio CM, Ivars F. Cytotoxic T lymphocyte antigen-4-dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory T-cell-mediated suppression. Immunol. 2006;118:240–249. doi: 10.1111/j.1365-2567.2006.02362.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ono M, Shimizu J, Miyachi Y, Sakaguchi S. Control of autoimmune myocarditis and multiorgan inflammation by glucocorticoid-induced TNF receptor family-related proteinhigh, Foxp3-expressing CD25+ and CD25− regulatory T cells. J Immunol. 2006;176:4748–4756. doi: 10.4049/jimmunol.176.8.4748. [DOI] [PubMed] [Google Scholar]
- 37.Lügering A, Floer M, Maaser C, Spahn TW, Schmidt MA, Domschke W, Williams IR, Kucharzik T. Absence of CCR6 inhibits CD4+ regulatory T-cell development and M-cell formation inside Peyer's patches. Am J Pathol. 2005;166:1647–1654. doi: 10.1016/S0002-9440(10)62475-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hirahara KL, Liu Clark RA, Yamanaka K, Fuhlbrigge RC, Kupper TS. The majority of human peripheral blood CD4+CD25high Foxp3+ regulatory T cells bear functional skin-homing receptors. J Immunol. 2006;177:4488–4494. doi: 10.4049/jimmunol.177.7.4488. [DOI] [PubMed] [Google Scholar]
- 39.Offner H, Subramanian S, Wang C, Afentoulis M, Vandenbark AA, Huan J, Burrows GG. Treatment of passive experimental autoimmune encephalomyelitis in SJL mice with a recombinant TCR ligand induces IL-13 and prevents axonal injury. J Immunol. 2005;175:4103–4111. doi: 10.4049/jimmunol.175.6.4103. [DOI] [PubMed] [Google Scholar]
- 40.Kleinschek MA, Owyang AM, Joyce-Shaikh B, Langrish CL, Chen Y, Gorman DM, Blumenschein WM, McClanahan T, Brombacher F, Hurst SD, Kastelein RA, Cua DJ. IL-25 regulates Th17 function in autoimmune inflammation. J Exp Med. 2007;204:161–170. doi: 10.1084/jem.20061738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zenewicz LA, Yancopoulos GD, Valenzuela DM, Murphy AJ, Karow M, Flavell RA. Interleukin-22 but not interleukin-17 provides protection to hepatocytes during acute liver inflammation. Immunity. 2007;27:647–659. doi: 10.1016/j.immuni.2007.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kreymborg K, Etzensperger R, Dumoutier L, Haak S, Rebollo A, Buch T, Heppner FL, Renauld JC, Becher B. IL-22 is expressed by Th17 cells in an IL-23-dependent fashion, but not required for the development of autoimmune encephalomyelitis. J Immunol. 2007;179:8098–8104. doi: 10.4049/jimmunol.179.12.8098. [DOI] [PubMed] [Google Scholar]
- 43.Hofstetter HH, Ibrahim SM, Koczan D, Kruse N, Weishaupt A, Toyka KV, Gold R. Therapeutic efficacy of IL-17 neutralization in murine experimental autoimmune encephalomyelitis. Cell Immunol. 2005;237:123–130. doi: 10.1016/j.cellimm.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 44.Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol. 2006;177:566–573. doi: 10.4049/jimmunol.177.1.566. [DOI] [PubMed] [Google Scholar]
- 45.Sutton C, Brereton C, Keogh B, Mills KH, Lavelle EC. A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med. 2006;203:1685–1691. doi: 10.1084/jem.20060285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Billiau AH, Heremans H, Vandekerckhove F, Kijkmans R, Sobis I, Meulepas E, Carton H. Enhancement of experimental allergic encephalomyelitis in mice by antibodies against IFN-γ. J Immunol. 1988;140:1506–1510. [PubMed] [Google Scholar]
- 47.Duong TT, Finkelman FD, Singh B, Strejan GH. Effect of anti-interferon-gamma monoclonal antibody treatment on the development of experimental allergic encephalomyelitis in resistant mouse strains. J Neuroimmunol. 1994;53:101–107. doi: 10.1016/0165-5728(94)90069-8. [DOI] [PubMed] [Google Scholar]
- 48.Ferber IA, Brocke S, Taylor-Edwards C, Ridgway W, Dinisco C, Steinman L, Dalton D, Fathman CG. Cutting Edge: Mice with a disrupted IFN-γ gene are susceptible to the induction of experimental autoimmune encephalomyelitis (EAE) J Immunol. 1996;156:5–7. [PubMed] [Google Scholar]
- 49.Chu CQ, Wittmer S, Dalton DK. Failure to suppress the expansion of the activated CD4 T cell population in interferon γ-deficient mice leads to exacerbation of experimental autoimmune encephalomyelitis. J Exp Med. 2000;192:123–128. doi: 10.1084/jem.192.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Willenborg DO, Fordham S, Bernard CC, Cowden WB, Ramshaw IA. Cutting Edge: IFN-γ plays a critical down-regulatory role in the induction and effector phase of myelin oligodendrocyte glycoprotein-induced autoimmune encephalomyelitis. J Immunol. 1996;157:3223–3227. [PubMed] [Google Scholar]
- 51.Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol. 2005;6:1123–1132. doi: 10.1038/ni1254. [DOI] [PubMed] [Google Scholar]
- 52.Park H, Li Z, Yang XO, Chang SH, Nurieva R, Wang YH, Wang Y, Hood L, Zhu Z, Tian Q, Dong C. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol. 2005;6:1133–1141. doi: 10.1038/ni1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Voorthuis JA, Uitdehaagh BM, DeGroot CJ, Goede PH, Vander Meide PH, Dijkstra CD. Suppression of experimental allergic encephalomyelitis by intraventricular administration of interferon-gamma in Lewis rats. Clin Exp Immunol. 1990;81:183–188. doi: 10.1111/j.1365-2249.1990.tb03315.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Espejo C, Penkowa M, Sáez-Torres I, Xaus J, Celada A, X Montalban, Martinez-Cáceres EM. Treatment with anti-interferon-γ monoclonal antibodies modifies experimental autoimmune encephalomyelitis in interferon-γ receptor knockout mice. Exp Neurol. 2001;172:460–468. doi: 10.1006/exnr.2001.7815. [DOI] [PubMed] [Google Scholar]