Abstract
Human papilloma virus (HPV) infection of the uterine cervix is linked to the pathogenesis of cervical cancer. Preclinical in vitro and in vivo studies using HPV-containing human cervical carcinoma cell lines have shown that the mammalian target of rapamycin (mTOR) inhibitor, rapamycin, and epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor, erlotinib, can induce growth delay of xenografts. Activation of Akt and mTOR are also observed in cervical squamous cell carcinoma and, the expression of phosphorylated mTOR was reported to serve as a marker to predict response to chemotherapy and survival of cervical cancer patients. Therefore, we investigated: a) the expression level of EGFR in cervical squamous cell carcinoma (SCC) and high-grade squamous intraepithelial lesions (HSIL) versus non-neoplastic cervical squamous epithelium; b) the state of activation of the mTOR pathway in these same tissues; and c) any impact of these signal transduction molecules on cell cycle. Formalin-fixed paraffin-embedded tissue microarray blocks containing 20 samples each of normal cervix, HSIL and invasive SCC, derived from a total of 60 cases of cervical biopsies and cervical conizations were examined. Immunohistochemistry was utilized to detect the following antigens: EGFR; mTOR pathway markers, phosphorylated (p)-mTOR (Ser2448) and p-p70S6K (Thr389); and cell cycle associated proteins, Ki-67 and S phase kinase-associated protein (Skp)2. Protein compartmentalization and expression were quantified in regard to proportion (0-100%) and intensity (0-3+). Mitotic index (MI) was also assessed. An expression index (EI) for pmTOR, p-p70S6K and EGFR, respectively was calculated by taking the product of intensity score and proportion of positively staining cells. We found that plasmalemmal EGFR expression was limited to the basal/parabasal cells (2-3+, EI = 67) in normal cervical epithelium (NL), but was diffusely positive in all HSIL (EI = 237) and SCC (EI 226). The pattern of cytoplasmic p-mTOR and nuclear p-p70S6K expression was similar to that of EGFR; all showed a significantly increased EI in HSIL/SCC versus NL (p<0.02). Nuclear translocation of p-mTOR was observed in all SCC lesions (EI = 202) and was significantly increased versus both HSIL (EI = 89) and NL (EI = 54) with p<0.015 and p<0.0001, respectively. Concomitant increases in MI and proportion of nuclear Ki-67 and Skp2 expression were noted in HSIL and SCC. In conclusion, morphoproteomic analysis reveals constitutive activation and overexpression of the mTOR pathway in HSIL and SCC as evidenced by: increased nuclear translocation of pmTOR and p-p70S6K, phosphorylated at putative sites of activation, Ser2448 and Thr389, respectively; correlative overexpression of the upstream signal transducer, EGFR, and increases in cell cycle correlates, Skp2 and mitotic indices. These results suggest that the mTOR pathway plays a key role in cervical carcinogenesis and targeted therapies may be developed for SCC as well as its precursor lesion, HSIL.
Keywords: mTOR pathway, cervical squamous carcinoma, high grade SIL, morphoproteomics
Introduction
Squamous cell carcinoma (SCC) of the uterine cervix is the most common malignant tumor of the female genital tract worldwide [1]. It is well known that the human papilloma virus (HPV), especially the high risk variants HPV 16 and 18, is the single most important etiologic agent in the pathogenesis of cervical carcinoma [1]. HPV-encoded oncoproteins, E5, E6 and E7 all possess immortalizing activity via their respective direct or indirect interactions with epidermal growth factor receptor (EGFR), p53 and retinoblastoma (Rb) proteins. Past studies have shown that the expression of E7 is directly linked to the growth and survival of the HPV-associated cancer cells [2–5]. Furthermore, studies based on cell culture have also shown that the cellular translation machinery, namely the downstream effector of the mammalian target of rapamycin (mTOR) pathway, eukaryotic initiation factor (eIF) 4E via mTOR’s interaction with translation inhibitor 4E-BP1, plays a significant role in maintaining a high level of E7 protein expression [6]. Additionally, E5 is shown to activate EGFR and delay its degradation to promote cellular proliferation [7].
The serine/threonine kinase mTOR pathway is an appealing therapeutic target because it has an identified inhibitor, rapamycin, an analogue of which (temsirolimus) has been utilized in phase II and III clinical trials for metastatic renal cell carcinoma [8, 9]. The mTOR pathway has been shown in preclinical studies to play a role in the carcinogenesis of breast, ovary and prostate as well as gastrointestinal malignancies. Studies have also demonstrated that the phosphorylative activation of p70S6K via the mTOR pathway resulted in increased G1 cell cycle progression, cell survival, and tumor cell proliferation [10–12]. A study from Japan showed that Akt and mTOR are activated in advanced stage of cervical squamous cell carcinoma and the activation was observed to serve as a marker to predict response to chemotherapy and survival of cervical cancer patients [13]. The receptor tyrosine kinase (RTK), epidermal growth factor receptor (EGFR) is involved in the regulation of cell proliferation, survival, mobility, and differentiation [14,15]. Its expression in a majority of cervical SCCs and encouraging results from preclinical trials in various tumors of the lung, pancreas, central nervous system and head and neck has made it an attractive target for targeted therapy using EGFR inhibitors, such as erlotinib, which also show synergistic response with the mTOR pathway inhibitors, rapamycin and temsirolimus [14–24].
The goal of this study is to characterize the expression levels of EGFR, the activation of the mTOR pathway and the proliferative cell cycle markers in the same tissues of cervical SCC and high-grade squamous intraepithelial lesions (HSIL) versus non-neoplastic cervical squamous epithelium.
Materials and Methods
Study Group
The pathology database at Lyndon B. Johnson Hospital was retrospectively reviewed and a total of 60 cases of normal cervix, HSIL or cervical dysplasia and invasive cervical SCC with adequate tissue were selected with IRB approval. All identifiers were removed to protect patient’s confidentiality. Three tissue microarray blocks containing 20 tissue samples each of normal cervix, HSIL and invasive cervical SCC were created by manually re-embedding archived paraffin-embedded tissues.
Immunohistochemical Staining
Tissue microarray sections, 4 μm thick, were deparaffinized in xylene and rehydrated in a graded series of ethanols. Heat-induced epitope retrieval was performed. Phospho-specific probes include antibodies directed against mTOR and p70S6K, phosphorylated at putative sites of activation, serine (Ser) 2448 and threonine (Thr) 389, respectively (Cell Signaling Technology, Inc., Beverly, MA, U.S.A.). The tissue was then treated with 3% H2O2 and rinsed with Tween-20 (TBST) buffer. A few drops of diluted normal blocking serum were placed on the tissue and incubated at room temperature. The serum was then blotted off and the slides were incubated with primary antibody overnight at 4°C. The rest of the staining procedure took place on a DAKO Autostainer programmed to treat each slide with diluted biotinylated secondary antibody for 30 minutes. The slides were then rinsed and incubated with DAB (3, 3’-diaminobenzidine chromogen solution, EnVision+ System Kit, DAKO) for 10 minutes. The slides were rinsed again and counterstained with Gill II hematoxylin, treated with xylene and cover slipped. The remaining immunohistochemical staining for EGFR (monoclonal, 1:50, 30 minute incubation at room temperature, 28-8005, Zymed, San Francisco, CA, U.S.A.), Ki-67 (1:500, 30 minute incubation at room temperature, M7240, DAKO, Denmark) and Skp2 (1:50, 45 minute incubation at room temperature, sc-7164, Santa Cruz Biotechnology, Inc., Santa Cruz, CA, U.S.A.) were performed as described above with DAKO Autostainer. Positive and negative controls were noted to react appropriately.
Tissue Microarray Analysis
Two pathologists visually read the tissue array under the microscope and scored each core individually. Cases in which no epithelial cells were found or no core was available were excluded. Protein expression was semiquantified in regard to the intensity of cell staining graded as 0 to 3+ (“0” for negative staining if there is total absence; score of “1”, “2”, or “3” in order of ascending strength of positive staining), as well as the percentage (ranging from 0 to 100%) of lesion with positive staining of any intensity ranging from 1 to 3. Resulting staining intensity scores and percentage of positive staining were averaged between the two pathologists. Mitotic index (MI) was also assessed on routine H&E stained microarray slides as number of mitotic figures per ten (10) high power fields. An expression index (EI) for p-mTOR, p-p70S6K and EGFR, respectively was calculated by taking the product of intensity score and proportion of positively staining cells.
Statistical Analysis
Student t-test was used to determine significance of differences in staining expression between normal cervix, HSIL and SCC. Excel was used for statistical analysis and 2-tailed p values were used to assess statistical significance.
Results
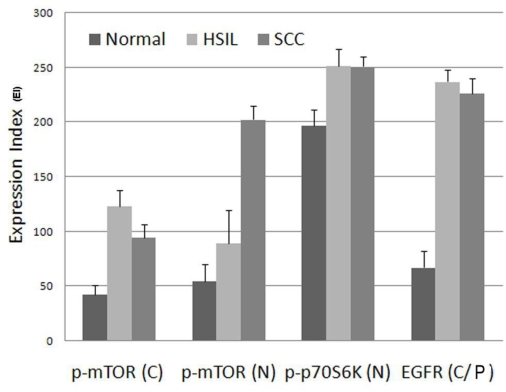
Plasmalemmal (membranous) EGFR expression was strongly positive (2-3+) in the majority (65%) of the normal cervical epithelium (NL) cases but is partial and limited to the basal/parabasal cells with an EI of 67, and more diffusely and strongly positive in all HSIL (2-3+ in 95% of cases, EI = 237) and SCC (2-3+ in 90% of cases, EI 226) (Figures 1 and 4 and Table 1).
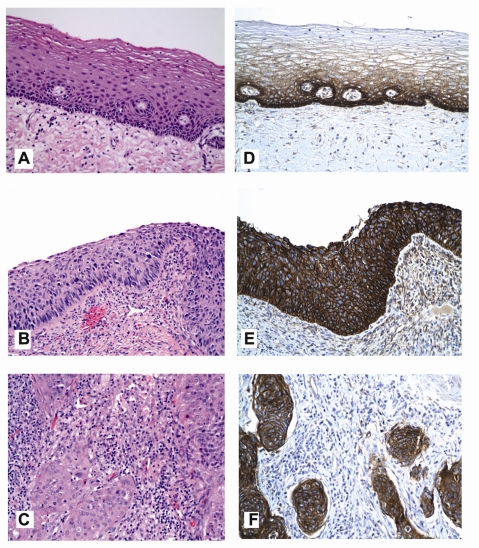
Figure 1.
Representative hematoxylin-eosin (H&E) images of uterine cervical mucosa illustrating non-neoplastic (normal) squamous epithelium (frame A), high grade squamous intraepithelial lesion (HSIL; frame B) and invasive cervical squamous cell carcinoma (SCC, frame C). Companion images depicting the level of expression of epidermal growth factor receptor (EGFR) on the plasmalemmal (cell membranous) aspect of the epithelial cells reveal: moderate to strong (2 to 3 +) DAB [brown] chromogenic signal limited to the basal and parabasal cells of normal epithelium (frame D); and overexpression of EGFR protein in HSIL and SCC evident by strong (3+) plasmalemmal expression that decorates virtually all lesional cells (frames E and F, respectively) (DAB [brown] chromogen; original magnification ×200).
Figure 4.
Graphic representation of expression index (EI) for EGFR and mTOR pathway markers, p-mTOR (Ser 2448) and p-p70S6K (Thr 389) comparing and contrasting normal (uterine cervical squamous epithelium), HSIL and invasive SCC. EI is the product of staining intensity (0-3+) and percent of positive staining cells (0-100%). C, cytoplasmic; N, nuclear; C/P, cytoplasmic/plasmalemmal (membranous).
Table 1.
Average expression index (EI) of EGFR and mTOR pathway markers: p-mTOR & p-p70S6K; percent cells with positive expression of proliferation markers: Ki-67 & Skp-2; and average mitotic index per ten (10) high power fields.
| Tissue type | Cytoplasmi c p-mTOR | Nuclear p-mTOR | Nuclear p-p70S6K | Cytoplasmic/membranous EGFR | Mitotic index | Ki-67 | Skp-2 |
|---|---|---|---|---|---|---|---|
| Normal | 42 | 54 | 197 | 67* | 0 | 2%* | 4%* |
| HSIL | 123 | 89 | 251 | 237 | 13 | 70% | 25% |
| SCC | 94 | 202 | 251 | 226 | 14 | 72% | 54% |
Basal/parabasal cell staining only. Each tissue type consists of 20 specimens
The pattern of cytoplasmic p-mTOR and nuclear p-p70S6K expression was similar to that of EGFR. Cytoplasmic p-mTOR expression was intermediate positive (1-2+) in most (100% of cases) of NL cervical epithelium but is partial and limited to the basal/parabasal cells with an EI of 67, and more diffusely and strongly positive in all HSIL (2-3+ in 95% of cases, EI = 237) and SCC (2-3+ in 90% of cases, EI 226). Nuclear p-p70S6K expression was strongly positive (2-3+) in majority (80% of cases) of NL cervical epithelium but was partial and generally limited to the basal half with an EI of 197, and more diffusely and strongly positive in all HSIL (2-3+ in 85% of cases, EI = 251) and SCC (2-3+ in 80% of cases, EI = 251) (Figures 2 and 4 and Table 1). Moderately increased cytoplasmic retention of p-p70SbK was evident in the NL epithelium Vis-a-Vis that in HSIL/SCC.
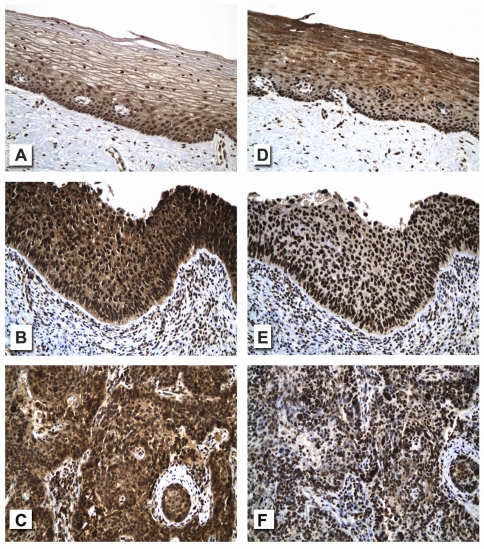
Figure 2.
Relative expression levels for mTOR pathway components, mTOR (phosphorylated on serine 2448) and p70S6K (phosphorylated on threonine 389) reveal: weak to moderate (1-2+) cytoplasmic and nuclear immunoreactivity (brown [DAB] chromogenic signal) for p-mTOR (Ser 2448) in the normal cervical squamous epithelium, particularly in the basal and parabasal cells (frame A) but with moderate to strong (2-3+) cytoplasmic and nuclear expression of this analyte in both HSIL and invasive squamous cell carcinoma involving virtually all of the lesional cells (frames B and C, respectively); and with diffuse and qualitatively similar nuclear expression levels for p-p70S6K (Thr 389) in normal cervical squamous epithelium, HSIL and invasive squamous cell carcinoma (frames D, E and F respectively; also see quantitative Expression Index [EI] for each of these analytes in Figure 4 and statistical differences in Table 2. Note the moderate cytoplasmic signal for p-p70S6K in supra basal zones of normal cervical epithelium (frame D) vis-à-vis that seen in HSIL and invasive squamous cell carcinoma (frames E and F). Subepithelial and interstitial inflammatory cell infiltrate present in HSIL and invasive squamous cell carcinoma shows variable nuclear signals for p-mTOR (frames B and C) and p-p70S6K (frames E and F) (original magnification ×200).
Nuclear p-mTOR expression was intermediate positive (1-2+) in most (95%) of NL cervical epithelium but is partial and limited to the basal/parabasal cells with an EI of 54, and more diffusely and strongly positive in all HSIL (2-3+ in 30% of cases, EI = 89) and SCC (2-3+ in 45% of cases, EI = 202) (Figures 2 and 4 and Table 1).
The proliferation markers Ki-67 and Skp-2 showed similar stepwise increase in percent of positive cells as in the average mitotic index in NL cervical epithelium (limited to the basal/parabasal layer), HSIL (lower two-thirds in majority of cases) and SCC (diffuse). Skp2 showed positive staining in a smaller percent of cells compared to Ki-67 in NL, HSIL and SCC (Figure 3 and Table 1).
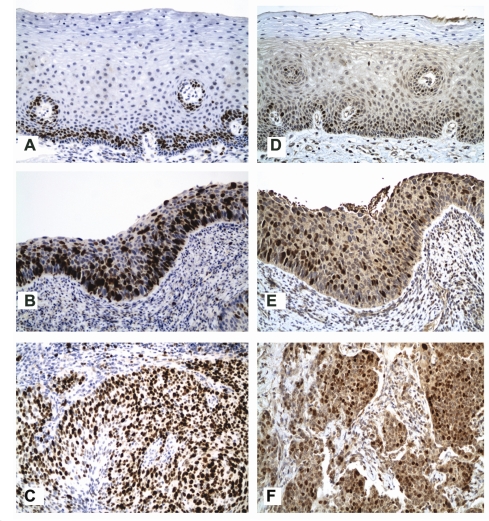
Figure 3.
Analysis of nuclear expression levels for cell cycle–associated markers, Ki-67 (G1, S, G2 and M phases of cell cycle) and Skp2 (S phase kinase-associated protein) reveals limited (parabasal only) expressions for both analytes in normal cervical squamous epithelium (frames A and B, respectively) but with strong and frequent expression involving middle to full thickness epithelium in HSIL and strong and diffuse expressions in invasive squamous cell carcinoma (frames B and D, and E and F for Ki-67 and Skp2, respectively) (original magnification ×200; also see numerical data and statistical comparisons in Tables 1 and 2).
Cytoplasmic/plasmalemmal or membranous EGFR, cytoplasmic p-mTOR and nuclear pp70S6K all show significantly increased EI in HSIL/SCC compared to NL (p<0.02). The nuclear p-mTOR EI in SCC lesions was significantly increased compared to both HSIL and NL (p<0.015 and p<0.0001, respectively). Similarly, the proliferation markers Ki-67, Skp2 and mitotic index were significantly increased in HSIL and SCC compared to NL (Table 2).
Table 2.
Statistically significant differences among morphoproteomic analytes expression in normal cervical squamous epithelium (NL), HSIL and SCC
| Significant mean differences | Student’s t-test, 2 tailed p-value |
|---|---|
| Nuclear p-mTOR in HSIL vs NL | 0.049 |
| Nuclear p-mTOR in SCC vs NL | <0.0001 |
| Nuclear p-mTOR in SCC vs HSIL | 0.015 |
| Cytoplasmic p-mTOR in HSIL vs NL | 0.0001 |
| Cytoplasmic p-mTOR in SCC vs NL | 0.0026 |
| Nuclear p-p70S6K in HSIL vs NL | 0.019 |
| Nuclear p-p70S6K in SCC vs NL | 0.0042 |
| Cytoplasmic/membranous EGFR in HSIL/SCC vs NL | <0.0001 |
| Nuclear Ki-67 in HSIL/SCC vs NL | <0.0001 |
| Nuclear Skp2 in SCC vs NL | <0.0001 |
| Nuclear Skp2 in SCC vs HSIL | <0.0001 |
| Mitotic Index in HSIL/SCC vs NL | <0.0001 |
| Mitotic Index in NL vs SCC | 0.0003 |
These comparative results for normal cervical epithelium (NL), HSIL and SCC are graphically depicted in Figure 4 and Table 1, and significant mean differences are summarized in Table 2.
Discussion
This is a morphoproteomic study of the mTOR pathway in HSIL and invasive cervical SCC in relation to non-neoplastic (normal) uterine cervical epithelium. Morphoproteomic analysis utilizes phosphospecific probes directed against putative sites of activation in a given protein to characterize its expression level and subcellular translocation within a signal transduction pathway. It also incorporates expression levels of other protein analytes such as upstream transducers and downstream effectors from a signal transduction pathway to affirm the state of constitutive activation. Ultimately, comparative quantitation of signaling pathway activation between normal and neoplastic or nonneoplastic diseased tissues provides insight into the potential role of a signaling pathway in the etiopathogenesis of a disease process and/or the histogenesis of a tumor [25].
The morphoproteomic analysis in this study reveals the constitutive activation and overexpression of the mTOR pathway in HSIL and invasive cervical SCC vis-à-vis nonneoplastic (normal) uterine cervical epithelium by virtue of the following findings: the phosphorylation of mTOR at serine 2448 with nuclear translocation [25–29] and an increased EI in both HSIL and invasive SCC for cytoplasmic and nuclear p-mTOR (Ser 2448) as compared with normal cervical squamous epithelium; the correlative downstream overexpression of nuclear p-p70S6K (Thr 389) [27, 28, 30–36]; consistent cell cycle promoting effects of p-p70S6K in terms of the percent of neoplastic epithelial nuclei expressing the cell cycle protein analytes, Ki-67 and Skp2 [11, 12, 37–41], as well as increased mitotic activity compared with the normal epithelial cells; and finally, correlative overexpression of EGFR, an upstream tyrosine kinase that transduces signals through both the PI3'-K/Akt/mTOR and ras/Raf kinase/extracellular signal-regulated kinase (ERK) 1/2 pathways, thereby contributing to the constitutive activation of mTOR kinase and p70S6K in HSIL and invasive SCC (see Figure 5). Parenthetically, signaling of EGFR through both pathways is consistent with the finding by Bertelsen and colleagues that there is frequent PI3KCA amplification and Akt phosphorylation in uterine cervical neoplasia [42] and with the finding by Chen and associates of increased expression of phospho-ERK1/2 and Skp2 during tumor progression of cervical neoplasms [43].
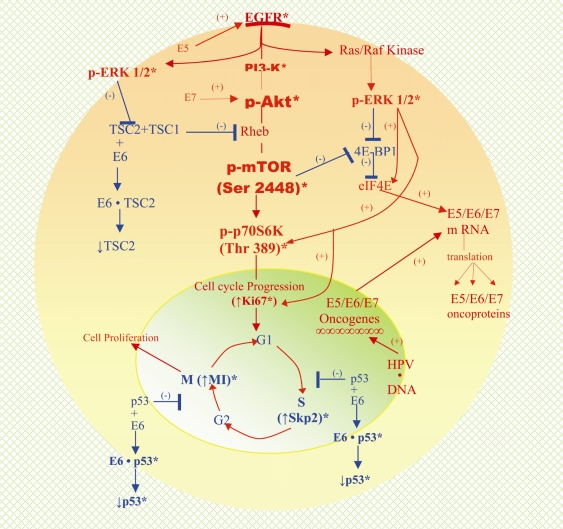
Figure 5.
A schematic composite of the complex protein circuitries identified by the authors and others utilizing immunohistochemistry in cases of HSIL and/or invasive squamous cell carcinoma of the uterine cervix [42, 43, 48] and depicting established interrelationships with both HPV-encoded oncoproteins - E5, E6, and E7 [2–5, 7, 45, 46] and with the mTOR pathway. HPV infection of cervical squamous epithelium leads to the incorporation of HPVoncogenes, E5, E6 and E7 into the genome resulting in the production of the corresponding oncoproteins. E5 activates EGFR and delays its degradation resulting in increased EGFR expression and then its signaling through the PI3'-K/Akt and ras/Raf kinase/ERK pathway, thereby contributing to phosphorylative activation of Akt and ERK1/2, respectively. This accounts for the expression of p-Akt and p-ERK1/2 in HSIL and invasive squamous cell carcinoma. Downstream signaling by p-Akt results in the constitutive activation and expression of p-mTOR (Ser 2448). Interaction of E6 oncoprotein with TSC2 (tuberin) contributes to signaling through the mTOR and probably ERK pathways [43, 45, 49, 50]. The former accounts for increased expression of p-p70S6K (Thr 389) and its downstream effector p-pS6 kinase and the latter collaborates with p-mTOR and contributes to activation of eIF4E through the phosphorylative inactivation of 4E-BP1. Additionally, p-ERK1/2 could contribute to mTOR pathway signaling by also contributing to the inactivation of TSC2 [51]. Translational synthesis of proteins including E7 oncoprotein results from the activation of eIF4E. E7 oncoprotein has been linked to growth and survival (immortalization) of HPV-associated, transformed cancer cells and to the signaling via the Akt/mTOR pathway by upregulating Akt activity. Cell cycle progression associated with mTOR and ERK pathway downstream signaling and aided by destabilization of wild type p53 by E6 oncoprotein is consistent with the increased expressions of Ki-67 and S phase kinase-associated protein (Skp) 2 and mitotic indices in HSIL and invasive squamous cell carcinoma. * indicates established immunohistochemical findings with respect to protein analytes; HSIL: high grade squamous intraepithelial lesion; HPV, human papilloma virus; mTOR, mammalian target of rapamycin; EGFR, epidermal growth factor receptor; PI3’-K, phosphatidylinositol 3-kinase; ERK, extracellular signal-regulated kinase; eIF4E, eukaryotic initiation factor 4E; 4E-BP1, 4E-binding protein-1.
Moreover, these morphoproteomic findings coincide with the effects of HPV, the principal etiologic agent associated with HSIL and invasive SCC of the cervix, in providing an etiopathogenic sequence that enables the renewal and proliferation of the dysplastic process and the maintenance of growth of the invasive SCC, when it develops. Specifically, HPV has been implicated in high risk cervical cancers as well as its precursor lesion, HSIL, reportedly accounting for some 80% of cervical cancers [1, 44]. Furthermore, as noted previously, the HPV-encoded oncoproteins, E5, E6 and especially, E7 confer replicative and immortalizing activities in cervical neoplasms. Such effects are achieved, in part, by virtue of their interactions with or impact upon EGFR, TSC2, p53 and Rb proteins, respectively [2–5, 7, 45, 46]. E5 activates EGFR and delays its degradation [7] and E6 interacts with and degrades the mTOR pathway inhibitor, tuberous sclerosis complex (TSC) 2 [45]. These effects, in turn, would lead to increased signaling through the PI3'-K/Akt/mTOR and ras/Raf kinase/ERK 1/2 pathways [25], eventuating in cellular proliferation. The binding of E6 with p53 has been shown to destabilize wild type p53 [5], which should also facilitate cell cycle progression (see Figure 5). Similarly, the binding of viral oncoprotein, E7 to Rb has been linked to its transforming capacity [5], possibly related in part to its upregulation of Akt activity [46]. This sequence accords with our morphoproteomic findings of increased plasmalemmal expression of EGFR in HSIL and invasive SCC of the uterine cervix and with the correspondingly increased cell cycle activity, as described above. In addition, the constitutive activation and overexpression of the mTOR pathway in HSIL and invasive SCC in this study should result in the phosphorylative inactivation of 4E-BP1 and thereby, the release of eIF-4E to effect translational synthesis of proteins to include viral oncoprotein E7 from their corresponding messenger RNA [6]. As noted earlier, E7 oncoprotein has been directly linked to the growth and survival (immortalization) of the HPV-associated cancer cells [2–5]. A schematic depicting the role of the HPV-encoded oncoproteins and incorporating our observations and those of others from the literature regarding EGFR overexpression and the activation and overexpression of the mTOR pathway with cell cycle correlates in HSIL and invasive cervical squamous cell carcinoma is represented in Figure 5.
In summary, our morphoproteomic analysis of high grade squamous intraepithelial lesions and invasive squamous cell carcinoma of the uterine cervix strongly suggests that a constitutively activated and overexpressed mTOR pathway, acting in concert with overexpressed EGFR and in collaboration with viral oncoproteins, plays a key role in both neoplastic processes. Furthermore in those cases associated with HPV infection, which represent the historical majority, the mTOR pathway may play a role in the translational synthesis of the encoded viral oncogene protein products, leading to the perpetuation of HSIL and the development of invasive SCC in some cases. As shown in previous studies, HSIL has a greater risk of persistent/recurrent disease after excision, and is the precursor lesion to invasive SCC of the uterine cervix [1, 44, 47]. Therefore, targeting the mTOR pathway and EGFR signaling with small molecule inhibitors to include a rapamycin and a tyrosine kinase inhibitor such as erlotinib [17–25] in patients with persistent or recurrent HSIL may be a reasonable therapeutic strategy. This combinatorial approach could obviate recurrence and reduce the risk of developing invasive SCC. Such an approach is consistent with certain stated objectives of the U.S. National Cancer Institute, namely to apply treatments or interventions at the individual level that keep cancers from starting or progressing.
Acknowledgments
The authors would like to thank Mr. Richard A. Breckenridge, HT (ASCP) and Ms. Pamela K. Johnston, HT(ASCP) for their technical assistance and expertise and Ms. Bheravi Patel for her secretarial and graphic design expertise.
References
- 1.Rosai J, editor. ROSAI AND ACKERMAN’S Surgical Pathology. 9th edition. Philadelphia, USA (Printed in China): Mosby, An Affiliate of Elsevier Inc; 2004. pp. 1532–1539. [Google Scholar]
- 2.Hall AH, Alexander KA. RNA interference of human papillomavirus type 18 E6 and E7 induces senescence in HeLa cells. J Virol. 2003;77:6066–6069. doi: 10.1128/JVI.77.10.6066-6069.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jiang M, Milner J. Selective silencing of viral gene expression in HPV-positive human cervical carcinoma cells treated with siRNA, a primer of RNA interference. Oncogene. 2002;21:6041–6048. doi: 10.1038/sj.onc.1205878. [DOI] [PubMed] [Google Scholar]
- 4.Jiang M, Milner J. Selective silencing of viral gene E6 and E7 expression in HPV-positive human cervical carcinoma cells using small interfering RNAs. Methods Mol Biol. 2005;292:401–420. doi: 10.1385/1-59259-848-x:401. [DOI] [PubMed] [Google Scholar]
- 5.von Knebel Doeberitz M, Rittmüller C, Aengeneyndt F, Jansen-Dürr P, Spitkovsky D. Reversible repression of papillomavirus oncogene expression in cervical cells: consequences for the phenotype and E6-p53 and E7-pRB interactions. J Virol. 1994;68:2811–2821. doi: 10.1128/jvi.68.5.2811-2821.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Oh KJ, Kalinina A, Park NH, Bagchi S. Deregulation of eIF4E: 4E-BP1 in differentiated human papillomavirus-containing cells leads to high levels of expression of the E7 oncoprotein. J Virol. 2006;80:7079–7088. doi: 10.1128/JVI.02380-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Straight SW, Hinkle PM, Jewers RJ, McCance DJ. The E5 oncoprotein of human papillomavirus type 16 transforms fibroblasts and effects the downregulation of the epidermal growth factor receptor in keratinocytes. J Virol. 1993;67:4521–4532. doi: 10.1128/jvi.67.8.4521-4532.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Atkins MB, Hidalgo M, Stadler WM, Logan TF, Dutcher JP, Hudes GR, Park Y, Liou SH, Marshall B, Boni JP, Dukart G, Sherman ML. Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J Clin Oncol. 2004;22:909–918. doi: 10.1200/JCO.2004.08.185. [DOI] [PubMed] [Google Scholar]
- 9.Hudes A. phase III, randomized, 3 arm study of temsirolinus (TEMSR) or interferon-alpha (IFN) or the combination of TEMSR + IFN in the treatment of first-line, poor risk patients with advanced renal cell carcinoma. Proc Am Soc Clin Oncol. 2006;24:2S. [Google Scholar]
- 10.Vega F, Medeiros LJ, Leventaki V, Atwell C, Cho-Vega JH, Tian L, Claret FX, Rassidakis GZ. Activation of mammalian target of rapamycin signaling pathway contributes to tumor cell survival in anaplastic lymphoma kinase-positive anaplastic large cell lymphoma. Cancer Res. 2006;66:6589–6597. doi: 10.1158/0008-5472.CAN-05-3018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gao N, Zhang Z, Jiang BH, Shi X. Role of PI3K/AKT/mTOR signaling in the cell cycle progression of human prostate cancer. Biochem Biophys Res Commun. 2003;310:1124–1132. doi: 10.1016/j.bbrc.2003.09.132. [DOI] [PubMed] [Google Scholar]
- 12.Gao N, Flynn DC, Zhang Z, Zhong XS, Walker V, Liu KJ, Shi X, Jiang BH. G1 cell cycle progression and the expression of G1 cyclins are regulated by PI3K/AKT/mTOR/p70S6K1 signaling in human ovarian cancer cells. Am J Physiol Cell Physiol. 2004;287:C281–291. doi: 10.1152/ajpcell.00422.2003. [DOI] [PubMed] [Google Scholar]
- 13.Faried LS, Faried A, Kanuma T, Sano T, Nakazato T, Tamura T, Kuwano H, Minegishi T. Predictive and prognostic role of activated mammalian target of rapamycin in cervical cancer treated with cisplatin-based neoadjuvant chemotherapy. Oncol Rep. 2006;16:57–63. [PubMed] [Google Scholar]
- 14.Chinnaiyan P, Harari PM. Clinical advancement of EGFR inhibitors in cancer therapy. Methods Mol Biol. 2006;327:189–202. doi: 10.1385/1-59745-012-X:189. [DOI] [PubMed] [Google Scholar]
- 15.Ciardiello F, Tortora G. Epidermal growth factor receptor (EGFR) as a target in cancer therapy: understanding the role of receptor expression and other molecular determinants that could influence the response to anti-EGFR drugs. Eur J Cancer. 2003;39:1348–1354. doi: 10.1016/s0959-8049(03)00235-1. [DOI] [PubMed] [Google Scholar]
- 16.Aissat N, Le Tourneau C, Ghoul A, Serova M, Bieche I, Lokiec F, Raymond E, Faivre S. Antiproliferative effects of rapamycin as a single agent and in combination with carboplatin and paclitaxel in head and neck cancer cell lines. Cancer Chemother Pharmacol. 2008;62:305–313. doi: 10.1007/s00280-007-0609-2. [DOI] [PubMed] [Google Scholar]
- 17.Azzariti A, Porcelli L, Gatti G, Nicolin A, Paradiso A. Synergic antiproliferative and antiangiogenic effects of EGFR and mTor inhibitors on pancreatic cancer cells. Biochem Pharmacol. 2008;75:1035–1044. doi: 10.1016/j.bcp.2007.11.018. [DOI] [PubMed] [Google Scholar]
- 18.Buck E, Eyzaguirre A, Brown E, Petti F, McCormack S, Haley JD, Iwata KK, Gibson NW, Griffin G. Rapamycin synergizes with the epidermal growth factor receptor inhibitor erlotinib in non-small-cell lung, pancreatic, colon, and breast tumors. Mol Cancer Ther. 2006;5:2676–2684. doi: 10.1158/1535-7163.MCT-06-0166. [DOI] [PubMed] [Google Scholar]
- 19.Conde E, Angulo B, Tang M, Morente M, TorresLanzas J, Lopez-Encuentra A, Lopez-Rios F, Sanchez-Cespedes M. Molecular context of the EGFR mutations: evidence for the activation of mTOR/S6K signaling. Clin Cancer Res. 2006;12:710–711. doi: 10.1158/1078-0432.CCR-05-1362. [DOI] [PubMed] [Google Scholar]
- 20.Frolov A, Schuller K, Tzeng CW, Cannon EE, Ku BC, Howard JH, Vickers SM, Heslin MJ, Buchsbaum DJ, Arnoletti JP. ErbB3 expression and dimerization with EGFR influence pancreatic cancer cell sensitivity to erlotinib. Cancer Biol Ther. 2007;6:548–554. doi: 10.4161/cbt.6.4.3849. [DOI] [PubMed] [Google Scholar]
- 21.Jimeno A, Kulesza P, Wheelhouse J, Chan A, Zhang X, Kincaid E, Chen R, Clark DP, Forastiere A, Hidalgo M. Dual EGFR and mTOR targeting in squamous cell carcinoma models, and development of early markers of efficacy. Br J Cancer. 2007;96:952–959. doi: 10.1038/sj.bjc.6603656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pham NA, Schwock J, Iakovlev V, Pond G, Hedley DW, Tsao MS. Immunohistochemical analysis of changes in signaling pathway activation downstream of growth factor receptors in pancreatic duct cell carcinogenesis. BMC Cancer. 2008;8:43. doi: 10.1186/1471-2407-8-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sos ML, Zander T, Thomas RK, Staratschek-Jox A, Claasen J, Wolf J. Expression of signaling mediators downstream of EGF-receptor predict sensitivity to small molecule inhibitors directed against the EGF-receptor pathway. J Thorac Oncol. 2008;3:170–173. doi: 10.1097/JTO.0b013e3181622c05. [DOI] [PubMed] [Google Scholar]
- 24.Wang MY, Lu KV, Zhu S, Dia EQ, Vivanco I, Shackleford GM, Cavenee WK, Mellinghoff IK, Cloughesy TF, Sawyers CL, Mischel PS. Mammalian target of rapamycin inhibition promotes response to epidermal growth factor receptor kinase inhibitors in PTEN-deficient and PTEN-intact glioblastoma cells. Cancer Res. 2006;66:7864–7869. doi: 10.1158/0008-5472.CAN-04-4392. [DOI] [PubMed] [Google Scholar]
- 25.Brown RE. Morphoproteomics: exposing protein circuitries in tumors to identify potential therapeutic targets in cancer patients. Expert Rev Proteomics. 2005;2:337–348. doi: 10.1586/14789450.2.3.337. [DOI] [PubMed] [Google Scholar]
- 26.Bachmann RA, Kim JH, Wu AL, Park IH, Chen J. A nuclear transport signal in mammalian target of rapamycin is critical for its cytoplasmic signaling to S6 kinase 1. J Biol Chem. 2006;281:7357–7363. doi: 10.1074/jbc.M512218200. [DOI] [PubMed] [Google Scholar]
- 27.Lin F, Zhang PL, Yang XJ, Prichard JW, Lun M, Brown RE. Morphoproteomic and molecular concomitants of an overexpressed and activated mTOR pathway in renal cell carcinomas. Ann Clin Lab Sci. 2006;36:283–293. [PubMed] [Google Scholar]
- 28.Cao X, Kambe F, Moeller LC, Refetoff S, Seo H. Thyroid hormone induces rapid activation of Akt/protein kinase B-mammalian target of rapamycin-p70S6K cascade through phosphatidylinositol 3-kinase in human fibroblasts. Mol Endocrinol. 2005;19:102–112. doi: 10.1210/me.2004-0093. [DOI] [PubMed] [Google Scholar]
- 29.Navé BT, Ouwens M, Withers DJ, Alessi DR, Shepherd PR. Mammalian target of rapamycin is a direct target for protein kinase B: identification of a convergence point for opposing effects of insulin and amino-acid deficiency on protein translation. Biochem J. 1999;344:427–431. [PMC free article] [PubMed] [Google Scholar]
- 30.Xu G, Zhang W, Bertram P, Zheng XF, McLeod H. Pharmacogenomic profiling of the PI3K/PTEN-AKT-mTOR pathway in common human tumors. Int J Oncol. 2004;24:893–900. [PubMed] [Google Scholar]
- 31.Pullen N, Dennis PB, Andjelkovic M, Dufner A, Kozma SC, Hemmings BA, Thomas G. Phosphorylation and activation of p70s6k by PDK1. Science. 1998;279:707–710. doi: 10.1126/science.279.5351.707. [DOI] [PubMed] [Google Scholar]
- 32.Dennis PB, Pullen N, Kozma SC, Thomas G. The principal rapamycin-sensitive p70 (s6k) phosphorylation sites, T-229 and T-389, are differentially regulated by rapamycin-insensitive kinase kinases. Mol Cell Biol. 1996;16:6242–6251. doi: 10.1128/mcb.16.11.6242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Valentinis B, Navarro M, Zanocco-Marani T, Edmonds P, McCormick J, Morrione A, Sacchi A, Romano G, Reiss K, Baserga R. Insulin receptor substrate-1, p70S6K, and cell size in transformation and differentiation of hemopoietic cells. J Biol Chem. 2000;275:25451–25459. doi: 10.1074/jbc.M002271200. [DOI] [PubMed] [Google Scholar]
- 34.Ali SM, Sabatini DM. Structure of S6 kinase 1 determines whether raptor-mTOR or rictormTOR phosphorylates its hydrophobic motif site. J Biol Chem. 2005;280:19445–19448. doi: 10.1074/jbc.C500125200. [DOI] [PubMed] [Google Scholar]
- 35.rown RE, Zhang PL, Lun M, Zhu S, Pellitteri PK, Riefkohl W, Law A, Wood GC, Kennedy TL. Morphoproteomic and pharmacoproteomic rationale for mTOR effectors as therapeutic targets in head and neck squamous cell carcinoma. Ann Clin Lab Sci. 2006;36:273–282. [PubMed] [Google Scholar]
- 36.Weng QP, Kozlowski M, Belham C, Zhang A, Comb MJ, Avruch J. Regulation of the p70 S6 kinase by phosphorylation in vivo. Analysis using site-specific anti-phosphopeptide antibodies. J Biol Chem. 1998;273:16621–16629. doi: 10.1074/jbc.273.26.16621. [DOI] [PubMed] [Google Scholar]
- 37.Gao N, Flynn DC, Zhang Z, Zhong XS, Walker V, Liu KJ, Shi X, Jiang BH. G1 cell cycle progression and the expression of G1 cyclins are regulated by PI3K/AKT/mTOR/p70S6K1 signaling in human ovarian cancer cells. Am J Physiol-Cell Physiol. 2004;287:C281–291. doi: 10.1152/ajpcell.00422.2003. [DOI] [PubMed] [Google Scholar]
- 38.Giles FJ, Albitar M. Mammalian target of rapamycin as a therapeutic target in leukemia. Curr Mol Med. 2005;5:653–661. doi: 10.2174/156652405774641034. [DOI] [PubMed] [Google Scholar]
- 39.Shaw RJ, Cantley LC. Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature. 2006;441:424–430. doi: 10.1038/nature04869. [DOI] [PubMed] [Google Scholar]
- 40.Shapira M, Kakiashvili E, Rosenberg T, Hershko DD. The mTOR inhibitor rapamycin down-regulates the expression of the ubiquitin ligase subunit Skp2 in breast cancer cells. Breast Cancer Res. 2006;8:R46. doi: 10.1186/bcr1533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chiarle R, Fan Y, Piva R, Boggino H, Skolnik J, Novero D, Palestro G, De Wolf-Peeters C, Chilosi M, Pagano M, Inghirami G. S-phase kinase-associated protein 2 expression in nonHodgkin's lymphoma inversely correlates with p27 expression and defines cells in S phase. Am J Pathol. 2002;160:1457–1466. doi: 10.1016/S0002-9440(10)62571-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bertelsen BI, Steine SJ, Sandvei R, Molven A, Laerum OD. Molecular analysis of the PI3K-AKT pathway in uterine cervical neoplasia: frequent PIK3CA amplification and AKT phosphorylation. Int J Cancer. 2006;118:1877–1883. doi: 10.1002/ijc.21461. [DOI] [PubMed] [Google Scholar]
- 43.Chen TP, Chen CM, Chang HW, Wang JS, Chang WC, Hsu SI, Cho CL. Increased expression of SKP2 and phospho- MAPK/ERK1/2 and decreased expression of p27 during tumor progression of cervical neoplasms. Gynecol Oncol. 2007;104:516–523. doi: 10.1016/j.ygyno.2006.09.015. [DOI] [PubMed] [Google Scholar]
- 44.Roden RB, Monie A, Wu TC. Opportunities to improve the prevention and treatment of cervical cancer. Curr Mol Med. 2007;7:490–503. doi: 10.2174/156652407781387127. [DOI] [PubMed] [Google Scholar]
- 45.Lu Z, Hu X, Li Y, Zheng L, Zhou Y, Jiang H, Ning T, Basang Z, Zhang C, Ke Y. Human papillomavirus 16 E6 oncoprotein interferences with insulin signaling pathway by binding to tuberin. J Biol Chem. 2004;279:35664–35670. doi: 10.1074/jbc.M403385200. [DOI] [PubMed] [Google Scholar]
- 46.Menges CW, Baglia LA, Lapoint R, McCance DJ. Human papillomavirus type 16 E7 up-regulates AKT activity through the retinoblastoma protein. Cancer Res. 2006;66:5555–5559. doi: 10.1158/0008-5472.CAN-06-0499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ramchandani SM, Houck KL, Hernandez E, Gaughan JP. Predicting persistent/recurrent disease in the cervix after excisional biopsy. Med Gen Med. 2007;9:24. [PMC free article] [PubMed] [Google Scholar]
- 48.Yao TT, Dai YZ, Li SZ. [Expression and clinical significance of phosphatidylinositol 3-kinase and protein kinase B in cervical carcinoma.] Ai Zheng. 2008;27:525–530. [PubMed] [Google Scholar]
- 49.Yoon HS, Monks TJ, Everitt JI, Walker CL, Lau SS. Cell proliferation is insufficient, but loss of tuberin is necessary, for chemically induced nephrocarcinogenicity. Am J Physiol Renal Physiol. 2002;283:F262–270. doi: 10.1152/ajprenal.00261.2001. [DOI] [PubMed] [Google Scholar]
- 50.Finlay GA, Hunter DS, Walker CL, Paulson KE, Fanburg BL. Regulation of PDGF production and ERK activation by estrogen is associated with TSC2 gene expression. Am J Physiol Cell Physiol. 2003;285:C409–418. doi: 10.1152/ajpcell.00482.2002. [DOI] [PubMed] [Google Scholar]
- 51.Ma L, Teruya-Feldstein J, Bonner P, Bernardi R, Franz DN, Witte D, Cordon-Cardo C, Pandolfi PP. Identification of S664 TSC2 phosphorylation as a marker for extracellular signal-regulated kinase mediated mTOR activation in tuberous sclerosis and human cancer. Cancer Res. 2007;67:7106–7112. doi: 10.1158/0008-5472.CAN-06-4798. [DOI] [PubMed] [Google Scholar]