Abstract
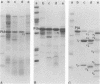
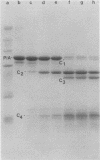
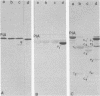
Whole bacteria, isolated outer membranes, and purified protein I (PI) from one transparent (O-) and two different opaque (O+) phenotype gonococcal strains (serogroups I, II, and III; PI serotypes 1, 5, and 9b) were each treated with tolylsulfonyl phenylalanyl chloromethyl ketone-trypsin, alpha-chymotrypsin, and proteinase K. Protein IA (PIA) of strain 7122 (O-, serotype 1, serogroup I) was resistant to proteolysis by tolysulfonyl phenylalanyl chloromethyl ketone-trypsin and alpha-chymotrypsin and only slightly affected by proteinase K, as long as it was associated with intact bacteria or isolated outer membranes. Purified PIA however was cleaved by these enzymes, resulting in two to five fragments. In contrast, all preparations of strains 5766 opaque phenotype (O+, serotype 7, serogroup II) and 1955 (O+, serotype 9b, serogroup III) were accessible to proteolysis, resulting in cleavage fragments of PIB compatible to those described previously by O. Barrera and J. Swanson (Infect. Immun. 44:565-568, 1984), M. S. Blake et al. (Infect. Immun. 33:212-222, 1981), and Blake (in G. K. Schoolnik, ed., The Pathogenic Neisseriae, 1985). Our data indicated that the purified PIB fraction was more accessible to proteases than the PIBs of whole bacteria or outer membranes. The fragmentation pattern of PIA cleavage products were quite different from PIB fragments, consistent with the different structure of these two groups of PI molecules. Time-dependent cleavage experiments with proteases, i.e., alpha-chymotrypsin, indicated that PIA was subsequently cleaved into smaller fragments. Highly reactive monoclonal antibodies, each specific for a surface-exposed epitope of PIA of strain 7122 or PIB of strains 5766 and 1955, as assessed by coagglutination, Western blot, and immunofluorescence, were reacted with PIA and PIB cleavage fragments in Western blot experiments. All cleavage fragments of the purified PIA and PIB preparations with molecular weights of greater than or equal to 14,200 showed immune reaction in Western blotting, whereas whole cell and outer membrane PIB fragments were less reactive with the specific monoclonal antibodies.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barrera O., Swanson J. Proteins IA and IB exhibit different surface exposures and orientations in the outer membranes of Neisseria gonorrhoeae. Infect Immun. 1984 Jun;44(3):565–568. doi: 10.1128/iai.44.3.565-568.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blake M. S., Gotschlich E. C., Swanson J. Effects of proteolytic enzymes on the outer membrane proteins of Neisseria gonorrhoeae. Infect Immun. 1981 Jul;33(1):212–222. doi: 10.1128/iai.33.1.212-222.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan T. M., Arko R. J. Immunity to gonococcal infection induced by vaccination with isolated outer membranes of Neisseria gonorrhoeae in guinea pigs. J Infect Dis. 1977 Jun;135(6):879–887. doi: 10.1093/infdis/135.6.879. [DOI] [PubMed] [Google Scholar]
- Buchanan T. M., Eschenbach D. A., Knapp J. S., Holmes K. K. Gonococcal salpingitis is less likely to recur with Neisseria gonorrhoeae of the same principal outer membrane protein antigenic type. Am J Obstet Gynecol. 1980 Dec 1;138(7 Pt 2):978–980. doi: 10.1016/0002-9378(80)91091-1. [DOI] [PubMed] [Google Scholar]
- Buchanan T. M., Hildebrandt J. F. Antigen-specific serotyping of Neisseria gonorrhoeae: characterization based upon principal outer membrane protein. Infect Immun. 1981 Jun;32(3):985–994. doi: 10.1128/iai.32.3.985-994.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan T. M., Pearce W. A., Schoolnik G. K., Arko R. J. Protection against infection with Neisseria gonorrhoeae by immunization with outer membrane protein complex and purified pili. J Infect Dis. 1977 Aug;136 (Suppl):S132–S137. doi: 10.1093/infdis/136.supplement.s132. [DOI] [PubMed] [Google Scholar]
- Gillis T. P., Buchanan T. M. Production and partial characterization of monoclonal antibodies to Mycobacterium leprae. Infect Immun. 1982 Jul;37(1):172–178. doi: 10.1128/iai.37.1.172-178.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston K. H., Holmes K. K., Gotschlich E. C. The serological classification of Neisseria gonorrhoeae. I. Isolation of the outer membrane complex responsible for serotypic specificity. J Exp Med. 1976 Apr 1;143(4):741–758. doi: 10.1084/jem.143.4.741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KELLOGG D. S., Jr, PEACOCK W. L., Jr, DEACON W. E., BROWN L., PIRKLE D. I. NEISSERIA GONORRHOEAE. I. VIRULENCE GENETICALLY LINKED TO CLONAL VARIATION. J Bacteriol. 1963 Jun;85:1274–1279. doi: 10.1128/jb.85.6.1274-1279.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte J., Rodriguez H. A discontinuous electrophoretic system for separating peptides on polyacrylamide gels. Anal Biochem. 1983 Sep;133(2):515–522. doi: 10.1016/0003-2697(83)90118-5. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McDade R. L., Jr, Johnston K. H. Characterization of serologically dominant outer membrane proteins of Neisseria gonorrhoeae. J Bacteriol. 1980 Mar;141(3):1183–1191. doi: 10.1128/jb.141.3.1183-1191.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OSBORN M. J. STUDIES ON THE GRAM-NEGATIVE CELL WALL. I. EVIDENCE FOR THE ROLE OF 2-KETO- 3-DEOXYOCTONATE IN THE LIPOPOLYSACCHARIDE OF SALMONELLA TYPHIMURIUM. Proc Natl Acad Sci U S A. 1963 Sep;50:499–506. doi: 10.1073/pnas.50.3.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poolman J. T., Buchanan T. M. Monoclonal antibody activity against native and denatured forms of gonococcal outer membrane proteins as detected within ultrathin, longitudinal slices of polyacrylamide gels. J Immunol Methods. 1984 Dec 31;75(2):265–274. doi: 10.1016/0022-1759(84)90110-8. [DOI] [PubMed] [Google Scholar]
- Sandstrom E. G., Chen K. C., Buchanan T. M. Serology of Neisseria gonorrhoeae: coagglutination serogroups WI and WII/III correspond to different outer membrane protein I molecules. Infect Immun. 1982 Nov;38(2):462–470. doi: 10.1128/iai.38.2.462-470.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandström E., Danielsson D. Serology of Neisseria gonorrhoeae. Classification by co-agglutination. Acta Pathol Microbiol Scand B. 1980 Feb;88(1):27–38. doi: 10.1111/j.1699-0463.1980.tb02600.x. [DOI] [PubMed] [Google Scholar]
- Swanson J. Surface-exposed protein antigens of the gonococcal outer membrane. Infect Immun. 1981 Dec;34(3):804–816. doi: 10.1128/iai.34.3.804-816.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tam M. R., Buchanan T. M., Sandström E. G., Holmes K. K., Knapp J. S., Siadak A. W., Nowinski R. C. Serological classification of Neisseria gonorrhoeae with monoclonal antibodies. Infect Immun. 1982 Jun;36(3):1042–1053. doi: 10.1128/iai.36.3.1042-1053.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHITE L. A., KELLOGG D. S., Jr NEISSERIA GONORRHOEAE IDENTIFICATION IN DIRECT SMEARS BY A FLUORESCENT ANTIBODY-COUNTERSTAIN METHOD. Appl Microbiol. 1965 Mar;13:171–174. doi: 10.1128/am.13.2.171-174.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S. P., Holmes K. K., Knapp J. S., Ott S., Kyzer D. D. Immunologic classification of Neisseria gonorrhoeae with micro-immunofluorescence. J Immunol. 1977 Sep;119(3):795–803. [PubMed] [Google Scholar]