Abstract
Maternally inherited female-biased sex ratios have been documented in many invertebrate species. One cause of such biased sex ratios is male killing, i.e. only males die. In most species, male killing occurs during embryonic stages (early male killing) and is associated with cytoplasmic bacteria, including Wolbachia, Spiroplasma, Rickettsia, Flavobacteria and gamma proteobacteria. However, the oriental tea tortrix, Homona magnanima, is one of the few species in which male death occurs in the larval or pupal stage, and is thus an example of late male killing. We partially purified the agent causing late male killing in H. magnanima and showed that it consists of two RNA sequences. This represents an entirely novel agent causing late male killing.
Keywords: female-biased sex ratio, late male killing, Homona magnanima, RNA sequences
1. Introduction
Maternally inherited female-biased sex ratios have been documented in many invertebrate species. One of their causes is male killing (Hurst 1993; Dunn et al. 1995). The male-killing phenomenon falls into two categories (Hurst 1991): host males are killed in the embryonic stage (early male killing) or in the larval or pupal stage (late male killing). Early male killing is known in five orders of insect (Coleoptera, Diptera, Hymenoptera, Lepidoptera and Hemiptera). To date, five clades of bacteria have been identified as male-killing agents: in Coleoptera, Rickettsia (Werren et al. 1994; Lawson et al. 2001; von der Schulenburg et al. 2001), Wolbachia (Hurst et al. 1999a; Fialho & Stevens 2000), Spiroplasma (Hurst et al. 1999b; Majerus et al. 1999) and Flavobacteria (Hurst et al. 1999c); in Diptera, Wolbachia (Hurst et al. 2000) and Spiroplasma (Hackett et al. 1985); in Hymenoptera, the gamma proteobacterium Arsenophonus nasoniae (Gherna et al. 1991); and in Lepidoptera, Wolbachia (Jiggins et al. 2000a; Fujii et al. 2001; Dyson et al. 2002) and Spiroplasma (Jiggins et al. 2000b).
Late male killing has previously been recorded only in mosquitoes (Andreadis & Hall 1979; Andreadis 1985), caused by a microsporidian parasite, in Drosophila subquinaria (Jaenike 2007), caused by Wolbachia, and in strains of the oriental tea tortrix, Homona magnanima (Morimoto et al. 2001), at Ibaraki, Japan. The causal agent in H. magnanima is transmitted both vertically and horizontally. The pathology of late male killing in H. magnanima has been reported recently (Hoshino et al. 2008).
To identify the late male-killing agent in H. magnanima, we partially purified a causal agent from the late male-killing H. magnanima strain (LMK strain) and analysed its nucleic acids. We identified two novel RNA sequences associated with late male killing in H. magnanima, i.e. MK1068 and MK1241, and discussed their characteristics.
2. Material and methods
(a) Insects
The late male-killing strain (LMK strain) of H. magnanima was collected from Ibaraki Prefecture, Japan, in 1995. The normal strain of H. magnanima was collected from Itsukaichi, Tokyo, Japan, in 1999. These strains were maintained as described previously (Morimoto et al. 2001).
(b) Purification of the late male-killing agent
Female adult abdomens from the LMK strain were homogenized in phosphate-buffered saline (PBS) and centrifuged (9240g, 60 min, 4°C). The supernatant was filtered using a 0.45 μm pore size filter. The filtrate was then centrifuged (84 900g, 2 hours, 4°C). The pellet was suspended in PBS, layered on a 10–40% (w/v) sucrose density gradient column and centrifuged again (84 900g, 2 hours, 4°C). The band fraction that formed, designated as the purified fraction, was collected and used in the following experiments.
(c) Bioassay
The purified fraction or PBS was injected into fifth-instar larvae from the normal strain of H. magnanima using a microsyringe. The injected larvae were reared individually and their mortality was checked at the adult stage. Eleven female adults were allowed to mate randomly with 19 uninjected, normal strain males in a plastic box and to lay eggs. Several egg masses were randomly collected, and all progeny were reared to adults and then sexed. The sex of fifth-instar larvae, pupae and adults was determined by abdominal morphology.
(d) RNA extraction, reverse transcription (RT) and modified random amplified polymorphic DNA method
Total RNA was extracted from the purified fraction using ISOGEN (Nippon gene, Japan) and was reverse transcribed with AMV Reverse Transcriptase XL (TaKaRa, Japan) using random 9-mer primers. Synthesized cDNA was used as the template; PCRs were performed using 96 arbitrary 10-mer primers and Taq polymerase (TaKaRa) under conditions described previously (Abe et al. 1998). Amplified bands were cloned and sequenced.
(e) Specific RT-PCR
Total RNA from neonates, fifth-instar larvae and adults was reverse transcribed as described above. PCRs were performed using specific primers according to the sequence information and Taq polymerase under the following conditions: 1 cycle at 94°C for 2 min and 45 cycles at 94°C for 30 s, 50°C for 30 s and 72°C for 30 s, followed by 1 cycle at 72°C for 3 min.
(f) 5′- and 3′-RACE
Total RNA from the purified fraction was reverse transcribed as described above. Experiments were performed using the BD SMART RACE cDNA Amplification Kit (Clontech, Japan) according to the manufacturer's protocol. The amplified cDNA fragments were subcloned, and the nucleotide sequences of both strands were determined.
(g) Northern blotting
Total RNA extracted from adult females and the purified fraction was subjected to 1% denaturing agarose gel electrophoresis. RNA fragments were transferred to Hybond N membranes (Amersham Biosciences, Japan). A 445 bp fragment of the MK1068 cDNA (263–707) and a 409 bp of the MK1241 cDNA (233–641) were used as probes. The probes were random primers labelled with the Gene Images random labelling module (Amersham Biosciences). Hybridization and detection were performed using the Gene Images CDP-Star detection module (Amersham Biosciences) according to the manufacturer's protocol.
3. Results
(a) Purification of the male-killing agent
Late male killing in H. magnanima is transmitted vertically only from mothers (Morimoto et al. 2001). The causal agent of late male killing in H. magnanima is not Wolbachia, Spiroplasma, Rickettsia, Flavobacteria or the gamma proteobacteria, which were reported to be early male-killing agents, or the protozoa reported to be the late male-killing agent in mosquitoes. A preliminary study showed that inoculation of an ultracentrifuge fraction of homogenized H. magnanima larvae from the LMK strain showed specific male-killing activity to H. magnanima larvae (data not shown), suggesting that the agent consisted of a particle with the size of a small virus. We hypothesized that the late male-killing agent was a virus and used a virus purification protocol to isolate the agent.
To test whether the purified fraction contained the late male-killing agent, we injected the purified fraction into fifth-instar larvae from the normal strain. The number of adults emerging from pupae that had been injected with the purified fraction or PBS at the fifth instar is shown in table 1. Almost all PBS-injected larvae and the purified fraction-injected female larvae developed to adulthood. By contrast, in the purified fraction-injected male larvae, only 4 out of 16 larvae survived to adulthood. The survival rate of the purified fraction-injected male larvae was significantly lower than that of PBS-injected male larvae (Χ2-test, p<0.01), and the survival rate of the purified fraction-injected female larvae (Χ2-test, p<0.05). We examined 10 egg masses laid by the purified fraction-injected females and found that seven egg masses showed significant female-biased sex ratios, from 0.65 to 1.00 (p<0.05; table 2). By contrast, no egg masses laid by PBS-injected females showed a significant female-biased sex ratio (p>0.05; table 2). These experiments demonstrated that the purified fraction caused the female-biased sex ratio.
Table 1.
Survival rate of larvae injected with the purified fraction or PBS. (The purified fraction or PBS was injected into fifth-instar larvae from the normal strain of H. magnanima. The injected larvae were reared individually and their mortality was checked at the adult stage. The significance of differences among rates of emergence was analysed by the Χ2-test. n.s., comparison between the purified fraction-injected and PBS-injected females; no significant difference.)
| sex of larvae | inoculum | no. of larvae injected | no. of adults emerged | the rate of emergence (%) | significant difference |
|---|---|---|---|---|---|
| female | the purified fraction | 14 | 11 | 78.6 | n.s.a |
| female | PBS | 14 | 14 | 100 | n.s. |
| male | the purified fraction | 16 | 4 | 25.0 | a,b |
| male | PBS | 14 | 13 | 92.9 | b |
Comparison between the purified fraction-injected female and male. Significant difference, p<0.05.
Comparison between the purified fraction-injected and PBS-injected males. Significant difference, p<0.01.
Table 2.
Sex ratios in the second-generation offspring from females of the normal strain of H. magnanima that was injected with the purified fraction or PBS during the fifth instar. (Fifth-instar larvae of the normal strain were injected with the purified fraction or PBS. The injected larvae were reared to adults. Female adults were allowed to mate with uninjected normal strain males and to lay eggs. Ten egg masses were randomly collected, and the progeny of each were reared to adults and then sexed.)
| the purified fraction | PBS | ||||
|---|---|---|---|---|---|
| egg mass no. | no. of progeny | sex ratio (proportion female) | egg mass no. | no. of progeny | sex ratio (proportion female) |
| 1 | 98 | 0.39 | 1 | 71 | 0.45 |
| 2 | 55 | 0.85a | 2 | 88 | 0.44 |
| 3 | 26 | 1.00a | 3 | 48 | 0.58 |
| 4 | 60 | 0.47 | 4 | 113 | 0.42 |
| 5 | 31 | 0.89a | 5 | 79 | 0.48 |
| 6 | 14 | 0.78a | 6 | 172 | 0.41 |
| 7 | 54 | 0.65a | 7 | 109 | 0.58 |
| 8 | 24 | 0.88a | 8 | 89 | 0.53 |
| 9 | 43 | 0.95a | 9 | 87 | 0.35a |
| 10 | 67 | 0.54 | 10 | 53 | 0.44 |
Sex ratios were significantly different from 0.5 (5% level, binominal test).
(b) Identification of RNA sequences specific to the LMK strain
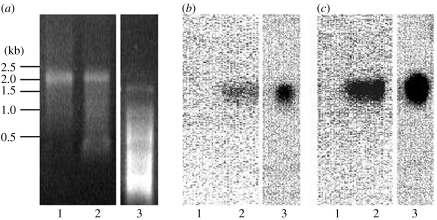
Nucleic acids were extracted from the purified fraction and then treated with DNase I or RNase A. Nucleic acids in the purified fraction were degraded by RNase A, but not by DNase I (data not shown). Thus, the nucleic acids in the purified fraction were RNA, not DNA. RNA was extracted from the purified fraction and the length was determined, using denaturing agarose gel electrophoresis, to be approximately 1500 bases (figure 1a). We used a modified random amplified polymorphic DNA method to determine the nucleic acid sequences. Each of the amplified 40 cDNA fragments was cloned and sequenced. Using specific primers according to sequence information, cDNA-specific RT-PCR was performed. As a result, two cDNA fragments were amplified from the LMK strain-derived RNA, but not from the normal strain. Moreover, two fragments were not amplified on DNA from both the LMK and the normal strain.
Figure 1.
Northern blots of MK1068 and MK1241. Northern blotting was performed using total RNA from the normal strain adult female (12 μg; lane 1), the LMK strain adult female (12 μg; lane 2) and the purified fraction (5 μg; lane 3), using partial MK1068 and MK1241 cDNA as probes. (a) Ethidium bromide stained, (b) MK1068 probe and (c) MK1241 probe.
To confirm the LMK-specific presence of the two sequences, RT-PCR analyses were performed with total RNA extracted from neonates, fifth-instar larvae and adults of both the LMK and the normal strain. Both sequences were detected specifically in the LMK strain, but not in the normal strain (table 3), although the control actin mRNA was detected almost equally among all RNA samples obtained from the two strains. These results suggest that both sequences exist in all stages of the LMK strain, but not in the normal strain.
Table 3.
Detection of MK1068 and MK1241 from the LMK and normal strains of H. magnanima. (Total RNA was extracted from neonates, fifth-instar larvae and adults of the LMK or normal strain and subjected to specific RT-PCR to detect MK1068 and MK1241.)
| strain | stage | sex | no. of samples examined | no. of samples having | |
|---|---|---|---|---|---|
| MK1068 | MK1241 | ||||
| LMK | neonate | unknown | 5 | 5 | 5 |
| fifth instar | female | 5 | 5 | 5 | |
| male | 5 | 5 | 5 | ||
| adult | female | 5 | 5 | 5 | |
| male | 5 | 5 | 5 | ||
| normal | neonate | unknown | 5 | 0 | 0 |
| fifth instar | female | 5 | 0 | 0 | |
| male | 5 | 0 | 0 | ||
| adult | female | 5 | 0 | 0 | |
| male | 5 | 0 | 0 | ||
(c) Full-length nucleotide sequences of MK1068 and MK1241 RNA
Northern blotting showed that both of the RNA sequences were approximately 1500 bases long (figure 1b,c). Using the 5′- and 3′-RACE methods, we sequenced the full length of the two cDNA fragments and designated them as MK1068 (1068 bases) and MK1241 (1241 bases); they had poly(A) tails. Open reading frame (ORF) finder research revealed that there were six putative ORFs in MK1068 and five putative ORFs in MK1241. But we do not know whether their ORFs are transcribed or not. A BLAST research revealed that their nucleotide and deduced amino acid sequences did not match any known sequence. Moreover, there were not any other predicted amino acid motifs in both MK1068 and MK1241 from MOTIF search.
The nucleotide sequences can be accessed at DDBJ/GenBank under the accession nos. of AB280943 for MK1068 and AB280944 for MK1241.
(d) MK1068 and MK1241 associated with late male killing
To examine whether MK1068 and MK1241 existed in adults from larvae of the normal strain that were injected with the purified fraction or PBS, specific RT-PCR was performed (table 4a). All adults from the purified fraction-injected female and male larvae showed amplification of MK1068 and MK1241. By contrast, no adult from PBS-injected larvae showed amplification of MK1068 or MK1241. These results indicate that both MK1068 and MK1241 were retained in H. magnanima after injection of the purified fraction.
Table 4.
The presence of MK1068 and MK1241 in the normal strain of H. magnanima that was injected with the purified fraction or PBS during the fifth instar. (Fifth-instar larvae of the normal strain were injected with the purified fraction or PBS. The injected larvae were reared to adults. Female adults were allowed to mate with uninjected normal strain males and to lay eggs. Ten egg masses were randomly collected, and the progeny of each were reared to adults. Total RNA was extracted from adults and subjected to gene-specific RT-PCR to detect MK1068 and MK1241. n.t., not tested.)
| (a) the adults from larvae that were injected with the purified fraction or PBS | ||||||
|---|---|---|---|---|---|---|
| inoculum | sex | no. of samples examined | no. of samples having | |||
| MK1068 | MK1241 | |||||
| the purified fraction | female | 5 | 5 | 5 | ||
| male | 4 | 4 | 4 | |||
| PBS | female | 5 | 0 | 0 | ||
| male | 5 | 0 | 0 | |||
| (b) the second-generation adults | ||||||
|---|---|---|---|---|---|---|
| egg mass no. | no. of progeny | sex ratio (proportion female) | MK1068 amplificationa | MK1241 amplificationa | ||
| female | male | female | male | |||
| 1 | 98 | 0.39 | 0/5 | 0/5 | 0/5 | 0/5 |
| 2 | 55 | 0.85b | 5/5 | 3/5 | 5/5 | 2/5 |
| 3 | 26 | 1.00b | 4/4 | n.t. | 4/4 | n.t. |
| 4 | 60 | 0.47 | 0/4 | 0/5 | 0/4 | 0/5 |
| 5 | 31 | 0.89b | 5/5 | 4/4 | 5/5 | 4/4 |
| 6 | 14 | 0.78b | 5/5 | 4/4 | 5/5 | 4/4 |
| 7 | 54 | 0.65b | 1/5 | 1/5 | 1/5 | 3/5 |
| 8 | 24 | 0.88b | 5/5 | 3/3 | 5/5 | 3/3 |
| 9 | 43 | 0.95b | 4/5 | n.t. | 5/5 | n.t. |
| 10 | 67 | 0.54 | 0/5 | 0/5 | 0/5 | 0/5 |
Number of amplified samples/number of tested samples.
Sex ratios were significantly different from 0.5 (5% level, binominal test).
To confirm that MK1068 and MK1241 were associated with late male killing, we examined the relationship between the female-biased sex ratio and the presence of MK1068 and MK1280 in the progeny of purified fraction-injected females (table 4b). All progeny that showed female-biased sex ratios exhibited amplification of both MK1068 and MK1241. By contrast, all three progeny that had a normal sex ratio (egg mass nos. 1, 4, 10) showed no amplification of MK1068 or MK1241. These results indicate that MK1068 and MK1241 were associated with late male killing in H. magnanima. Moreover, the amplification of MK1068 and MK1241 was not seen in some progenies of the egg mass nos. 2 and 7. This result suggests that the transmission rate of MK1068 and MK1241 from mother to the progeny may be variable.
4. Discussion
We determined that novel MK1068 and MK1241 RNA sequences are contained within the causal agent of late male killing in H. magnanima. These two sequences were identified as being precisely correlated with the late male-killing trait from the following observations. First, the purified fraction from adult females of the LMK strain showed an exact association with the late male-killing trait (tables 1 and 2). MK1068 and MK1241 were present in the purified fraction (figure 1b,c). MK1068 and MK1241 were retained in H. magnanima that had been injected with the purified fraction (table 4a) and subsequently produced female-biased progeny (table 4b). MK1068 and MK1241 were present in all stages of the LMK strain, but not in the normal strain (table 3). These findings satisfy Koch's postulates.
Morimoto et al. (2001) reported that the late male-killing agent in H. magnanima was not any of the micro-organisms previously reported as causing early male killing: the LMK strain of H. magnanima was infected with Wolbachia, but the female-biased sex ratio persisted after antibiotic treatment. Here, the purified fraction was filtered using a 0.45 μm pore size filter in the purification process. Thus, it is unlikely that the late male-killing agent in H. magnanima is a bacterium. The position of the purified fraction band obtained from sucrose density gradient centrifugation was the same as that of small RNA viruses. The purified fraction has not yet been examined using electron microscopy. However, the evidence presented here suggests that the late male-killing agent in H. magnanima is a novel RNA virus. Some known and sequenced RNA viruses are pathogenic in insects. However, the nucleotide sequences of MK1068 and MK1241 did not match any known sequence. To date, we do not have any information to identify this late male-killing candidate RNA virus.
It is unclear how the late male-killing candidate virus has lethal effects on males, but not on females. The mechanisms by which early male-killing bacteria and late male-killing microsporidia kill only males are also unknown. Further analyses are needed to identify this novel late male-killing agent in H. magnanima.
Acknowledgments
We thank Dr Hiroaki Abe for providing technical advice and 96 arbitrary 10-mer primers. We also thank Dr Max Bergoin for providing valuable advice, and Drs Michael E. N. Majerus and Steve Sait for their critical review of the manuscript. This work was partially supported by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (no. 17208006).
References
- Abe H, Kanehara M, Terada T, Ohbayashi F, Shimada T, Kawai S, Suzuki M, Sugasaki T, Oshiki T. Identification of novel random amplified polymorphic DNAs (RAPDs) on the W chromosome of the domesticated silkworm, Bombyx mori, and the wild silkworm, B. mandarina, and their retrotransposable element-related nucleotide sequences. Genes Genet. Syst. 1998;73:243–254. doi: 10.1266/ggs.73.243. doi:10.1266/ggs.73.243 [DOI] [PubMed] [Google Scholar]
- Andreadis T.G. Life cycle, epizootiology, and horizontal transmission of Amblyospora (Microspora: Amblyosporidae) in a univoltine Mosquito, Aedes stimulans. J. Invertebr. Pathol. 1985;46:31–46. doi:10.1016/0022-2011(85)90127-2 [Google Scholar]
- Andreadis T.G, Hall D.W. Significance of transovarial infections of Amblyospora sp. (Microspora: Thelohaniidae) in relation to parasite maintenance in the mosquito Culex salinarius. J. Invertebr. Pathol. 1979;34:152–157. doi: 10.1016/0022-2011(79)90095-8. doi:10.1016/0022-2011(79)90095-8 [DOI] [PubMed] [Google Scholar]
- Dunn A.M, Hatcher M.J, Terry R.S, Tofts C. Evolutionary ecology of vertically transmitted parasites: transovarial transmission of a microsporidian sex ratio distorter in Gammarus duebeni. Parasitology. 1995;111:S91–S109. [Google Scholar]
- Dyson E.A, Kamath M.K, Hurst G.D.D. Wolbachia infection associated with all-female broods in Hypolimnas bolina (Lepidoptera: Nymphalidae): evidence for horizontal transmission of a butterfly male killer. Heredity. 2002;88:166–171. doi: 10.1038/sj.hdy.6800021. doi:10.1038/sj/hdy/6800021 [DOI] [PubMed] [Google Scholar]
- Fialho R.F, Stevens L. Male-killing Wolbachia in a flour beetle. Proc. R. Soc. B. 2000;267:1469–1474. doi: 10.1098/rspb.2000.1166. doi:10.1098/rspb.2000.1166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii Y, Kageyama D, Hoshizaki S, Ishikawa H, Sasaki T. Transfection of Wolbachia in Lepidoptera: the feminizer of the azuki bean borer Ostrinia scapulalis causes male killing in the Mediterranean flour moth Ephestia kuehniella. Proc. R. Soc. B. 2001;268:855–859. doi: 10.1098/rspb.2001.1593. doi:10.1098/rspb.2001.1593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gherna R.L, Werren J.H, Weisburg W, Cote R, Woese C.R, Mandelco L, Brenner D.J. Arsenophonus nasoniae gen. nov., sp. nov., the causative agent of the son-killer trait in the parasitic wasp Nasonia vitripennis. Int. J. Syst. Bacteriol. 1991;41:563–565. [Google Scholar]
- Hackett K.J, Lynn D.E, Williamson D.L, Ginsberg A.S, Whitcomb R.F. Cultivation of the Drosophila sex-ratio Spiroplasma. Science. 1985;232:1253–1255. doi: 10.1126/science.232.4755.1253. doi:10.1126/science.232.4755.1253 [DOI] [PubMed] [Google Scholar]
- Hoshino M, Nakanishi K, Nakai M, Kunimi Y. Gross morphology and histopathology of male-killing strain larvae in the oriental tea tortrix Homona magnanima (Lepidoptera: Tortricidae) Appl. Entomol. Zool. 2008;43:119–125. [Google Scholar]
- Hurst G.D.D, et al. Male-killing Wolbachia in two species of insect. Proc. R. Soc. B. 1999a;266:735–740. doi:10.1098/rspb.1999.0698 [Google Scholar]
- Hurst G.D.D, von der Schulenburg J.H.G, Majerus T.M.O, Bertrand D, Zakharov I.A, Baungaard J, Völkl W, Stouthamer R, Majerus M.E.N. Invasion of one insect species, Adalia bipunctata, by two different male-killing bacteria. Insect Mol. Biol. 1999b;8:133–139. doi: 10.1046/j.1365-2583.1999.810133.x. doi:10.1046/j.1365-2583.1999.810133.x [DOI] [PubMed] [Google Scholar]
- Hurst G.D.D, Bandi C, Sacchi L, Cochrane A.G, Bertrand D, Karaca I, Majerus M.E.N. Adonia variegata (Coleoptera: Coccinellidae) bears maternally inherited Flavobacteria that kill males only. Parasitology. 1999c;118:125–134. doi: 10.1017/s0031182098003655. doi:10.1017/S0031182098003655 [DOI] [PubMed] [Google Scholar]
- Hurst G.D.D, Johnson A.P, von der Schulenburg J.H.G, Fuyama Y. Male-killing Wolbachia in Drosophila: a temperature-sensitive trait with a threshold bacterial density. Genetics. 2000;156:699–709. doi: 10.1093/genetics/156.2.699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurst L.D. The incidences and evolution of cytoplasmic male killers. Proc. R. Soc. B. 1991;244:91–99. doi:10.1098/rspb.1991.0056 [Google Scholar]
- Hurst L.D. The incidences, mechanisms and evolution of cytoplasmic sex ratio distorters in animals. Biol. Rev. 1993;68:121–193. doi:10.1111/j.1469-185X.1993.tb00733.x [Google Scholar]
- Jaenike J. Spontaneous emergence of a new Wolbachia phenotype. Evolution. 2007;61:2244–2252. doi: 10.1111/j.1558-5646.2007.00180.x. doi:10.1111/j.1558-5646.2007.00180.x [DOI] [PubMed] [Google Scholar]
- Jiggins F.M, Hurst G.D.D, Dolman C.E, Majerus M.E.N. High-prevalence male-killing Wolbachia in the butterfly Acraea encedana. J. Evol. Biol. 2000a;13:495–501. doi:10.1046/j.1420-9101.2000.00180.x [Google Scholar]
- Jiggins F.M, Hurst G.D.D, Jiggins C.D, von der Schulenburg J.H.G, Majerus M.E.N. The butterfly Danaus chrysippus is infected by a male-killing Spiroplasma bacterium. Parasitology. 2000b;120:439–446. doi: 10.1017/s0031182099005867. doi:10.1017/S0031182099005867 [DOI] [PubMed] [Google Scholar]
- Lawson E.T, Mousseau T.A, Klaper R, Hunter M.D, Werren J.H. Rickettsia associated with male-killing in a buprestid beetle. Heredity. 2001;86:497–505. doi: 10.1046/j.1365-2540.2001.00848.x. doi:10.1046/j.1365-2540.2001.00848.x [DOI] [PubMed] [Google Scholar]
- Majerus T.M.O, von der Schulenburg J.H.G, Majerus M.E.N, Hurst G.D.D. Molecular identification of a male-killing agent in the ladybird Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) Insect Mol. Biol. 1999;8:551–555. doi: 10.1046/j.1365-2583.1999.00151.x. doi:10.1046/j.1365-2583.1999.00151.x [DOI] [PubMed] [Google Scholar]
- Morimoto S, Nakai M, Ono A, Kunimi Y. Late male-killing phenomenon found in a Japanese population of the oriental tea tortrix, Homona magnanima (Lepidoptera: Tortricidae) Heredity. 2001;87:435–440. doi: 10.1046/j.1365-2540.2001.00924.x. doi:10.1046/j.1365-2540.2001.00924.x [DOI] [PubMed] [Google Scholar]
- von der Schulenburg J.H.G, Habig M, Sloggett J.J, Webberley K.M, Bertrand D, Hurst G.D.D, Majerus M.E.N. Incidence of male-killing Rickettsia spp. (alpha-proteobacteria) in the ten-spot ladybird beetle Adalia decempunctata L. (Coleoptera: Coccinellidae) Appl. Environ. Microbiol. 2001;67:270–277. doi: 10.1128/AEM.67.1.270-277.2001. doi:10.1128/AEM.67.1.270-277.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werren J.H, Hurst G.D.D, Zhang W, Breeuwer J.A.J, Stouthamer R, Majerus M.E.N. Rickettsial relative associated with male killing in the ladybird beetle (Adalia bipunctata) J. Bacteriol. 1994;176:388–394. doi: 10.1128/jb.176.2.388-394.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]