INTRODUCTION
Fascia corresponds to the continuous connective tissue network throughout the body that is localized beneath the skin and surrounds muscle, bone, and nerve. Much of body work practice is accomplished by physical manipulation aimed at reorganizing fascia to improve its integration and functional characteristics.
Type 1 collagen is the major protein found in fascia, and fibroblasts are the cell type primarily responsible for collagen biosynthesis and organization. Research in my laboratory focuses on understanding the structural, functional and mechanical features of fibroblasts (early passage human foreskin fibroblasts in our work) interacting with three dimensional collagen matrices. Unlike conventional 2D surfaces, with which most research on fibroblast-extracellular matrix interactions has been carried out, 3D collagen matrices offer unique, tissue-like features.
Cells can penetrate into the matrix.
Cell adhesions are limited to matrix fibrils rather than continuous fields of adsorbed matrix proteins.
Cells can remodel matrix fibrils into stably reorganized structures.
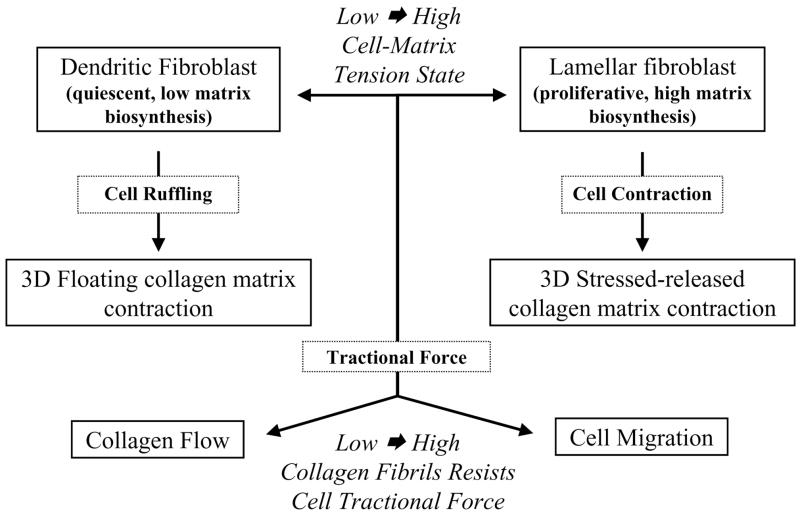
In what follows, I will summarize several key points from my presentation at the First International Fascia Research Congress held in Boston, 2007. Figure 1 outlines the main ideas to be discussed. Readers interested in additional details including microscopic images and videos should consult our recent publications (Grinnell 2003; Rhee & Grinnell 2007; Rhee et al 2007; Jiang et al 2008; Miron-Mendoza et al 2008). I will not cite the large and growing literature in this field but simply comment that 3D matrices increasingly are being used to model different aspects of normal and pathological tissue function (Yamada & Cukierman 2007).
Figure 1.
Fibroblast mechanics in three dimensional collagen matrices
CELL-MATRIX TENSION STATE
As indicated by Figure 1, fibroblast interactions with collagen matrices are determined at least in part by the cell-matrix tension state. Diverse parameters control cell-matrix tension including collagen density, matrix restraint, and growth factor environment. Experimentally, we assess tension state empirically as follows. At high cell-matrix tension, fibroblasts exhibit stress fibers, focal adhesions, and activation of focal adhesion signaling such as phosphorylation of focal adhesion kinase. At low cell-matrix tension, fibroblasts lack these features.
Fibroblasts at low cell-matrix tension state in collagen matrices exhibit dendritic morphology. They form a cell network throughout the matrix interconnected by gap junctions. Platelet-derived growth factor (PDGF), which activates the small G protein Rac, causes increased formation of cellular dendritic extensions. Lysophosphatidic acid (LPA), sphingosine-1-phosphate (S1P) and serum activate the small G protein Rho and cause dendritic extensions to retract by a myosin II-dependent process. Dendritic fibroblasts are quiescent and exhibit low matrix biosynthetic activity. These cells probably correspond to the resting tissue fibroblast. An attractive hypothesis is that an interconnected network of dendritic fibroblasts in soft tissue can function as a mechanosensing system analogous to mechanosensing by osteocytes in bone.
Fibroblasts at high cell-matrix tension state appear lamellar and resemble cells interacting with collagen-coated culture surfaces. Lamellar fibroblasts are proliferative and exhibit high matrix biosynthetic activity. These cells correspond to the wound fibroblasts responsible for connective tissue biosynthesis and contraction during wound repair. Lamellar fibroblasts can differentiate into myofibroblasts.
Fibroblasts at low and high cell-matrix tension states regulate their morphologies using different cytoskeletal mechanisms. Cells at a low tension state require microtubules for formation of dendritic extensions. Cells at a high tension state require microtubules for polarization but not for cell spreading. However, if fibroblasts are unable to develop a high cell-matrix tension state -- for instance, if they lack myosin II function -- then they will remain dendritic and not become lamellar.
COLLAGEN MATRIX CONTRACTION
Fibroblasts interacting with collagen matrices bind to and remodel nearby collagen fibrils. If enough cells are present, then local remodeling will be propagated throughout the matrix and result in what appears as global matrix contraction. As a result of contraction, the collagen density of experimental matrices can increase within hours from 1.5 mg/ml to 20 mg/ml, the latter similar to the density of collagen in tissue. Figure 1 summarizes the two types of fibroblast-collagen matrix contraction that occur depending on whether the cells interacting with collagen are at a low or high cell-matrix tension state. In floating matrix contraction, newly polymerized matrices are released and allowed to float in culture medium (low cell-matrix tension). In stressed-released matrix contraction, polymerized matrices containing fibroblasts are incubated restrained on culture dishes for sufficient time to allow cells to develop high cell-matrix tension. Then, the restrained matrices are released and allowed to float in culture medium. During stress-released matrix contraction, ~3 nm pores transiently open in the fibroblast plasma membrane. The physiological significance of transient pore opening is unknown.
Different cellular mechanisms are required for floating and stressed-released matrix contraction. Floating matrix contraction depends on cell ruffling and can be stimulated by PDGF, LPA, S1P or serum. Stressed-released matrix contraction, on the other hand, depends on cell contraction and can be stimulated by LPA, S1P or serum but not by PDGF. In addition to the differences in growth factor specificity, signaling mechanisms involved in matrix contraction change depending on cell-matrix tension state. LPA stimulation of floating matrix contraction requires the Rho effector mDia1; whereas LPA stimulation of stress-released matrix contraction requires the Rho effector Rho kinase.
CELL MIGRATION AND COLLAGEN FLOW
Beside for biosynthesis and remodeling of collagen, fibroblasts also move through collagen matrices. Migration has been studied using nested matrices, which are composed of dermal equivalents (cell-contracted floating collagen matrices) surrounded by an outer cell-free collagen matrices. As suggested by Figure 1, tractional force exerted by cells can couple differentially to collagen flow or cell migration depending on the ability of collagen fibrils to resist the force of cell traction. Collagen flow depends on the same cell motile machinery that results in cell migration and is capable of producing large scale tissue translocation as shown by closure of a ~1 mm gap between paired dermal equivalents in floating, nested collagen matrices. Therefore, depending on surrounding tissue mobility, fibroblasts activated to exert tractional force could have long distance as well as local effects on tissue organization.
Unlike collagen matrix contraction that can be stimulated by several growth factors and serum, PDGF appears to be unique in its capacity to promote cell migration. In addition, although S1P promotes collagen matrix contraction by fibroblasts, it inhibits cell migration. Since serum contains both PDGF and S1P, the balance between these factors likely will be a key determinant of fibroblast promigratory activity in serum. In general, depending upon which growth factors are released locally, the tissue environment for fibroblasts could differentially display promigratory or procontractile characteristics.
RELATION TO BODYWORK PRACTICE
Physical manipulation of fascia has the potential to change the cell-matrix tension state and also may influence localized release of cellular growth factors. As demonstrated by our research on fibroblast-collagen matrix interactions, such changes could lead to profound and rapid modulation of structural, functional and mechanical interactions between fibroblasts and the extracellular matrix and, as a result, contribute to the reorganization of fascia that results from bodywork practice.
Acknowledgments
The author’s research is supported by a grant from the National Institutes of Health, GM31321.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Grinnell F. Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol. 2003;13:264–9. doi: 10.1016/s0962-8924(03)00057-6. [DOI] [PubMed] [Google Scholar]
- Jiang H, Rhee S, Ho CH, Grinnell F. Distinguishing fibroblast promigratory and procontractile growth factor environments in 3-D collagen matrices. FASEB J. 2008 doi: 10.1096/fj.07-097014. In Press. [DOI] [PubMed] [Google Scholar]
- Miron-Mendoza M, Seemann J, Grinnell F. Collagen Fibril Flow and Tissue Translocation Coupled to Fibroblast Migration in 3D Collagen Matrices. Mol Biol Cell. 2008 doi: 10.1091/mbc.E07-09-0930. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhee S, Grinnell F. Fibroblast mechanics in 3D collagen matrices. Adv Drug Deliv Rev. 2007;59:1299–305. doi: 10.1016/j.addr.2007.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhee S, Jiang H, Ho CH, Grinnell F. Microtubule function in fibroblast spreading is modulated according to the tension state of cell-matrix interactions. Proc Natl Acad Sci USA. 2007;104:5425–30. doi: 10.1073/pnas.0608030104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada KM, Cukierman E. Modeling tissue morphogenesis and cancer in 3D. Cell. 2007;130:601–10. doi: 10.1016/j.cell.2007.08.006. [DOI] [PubMed] [Google Scholar]