Abstract
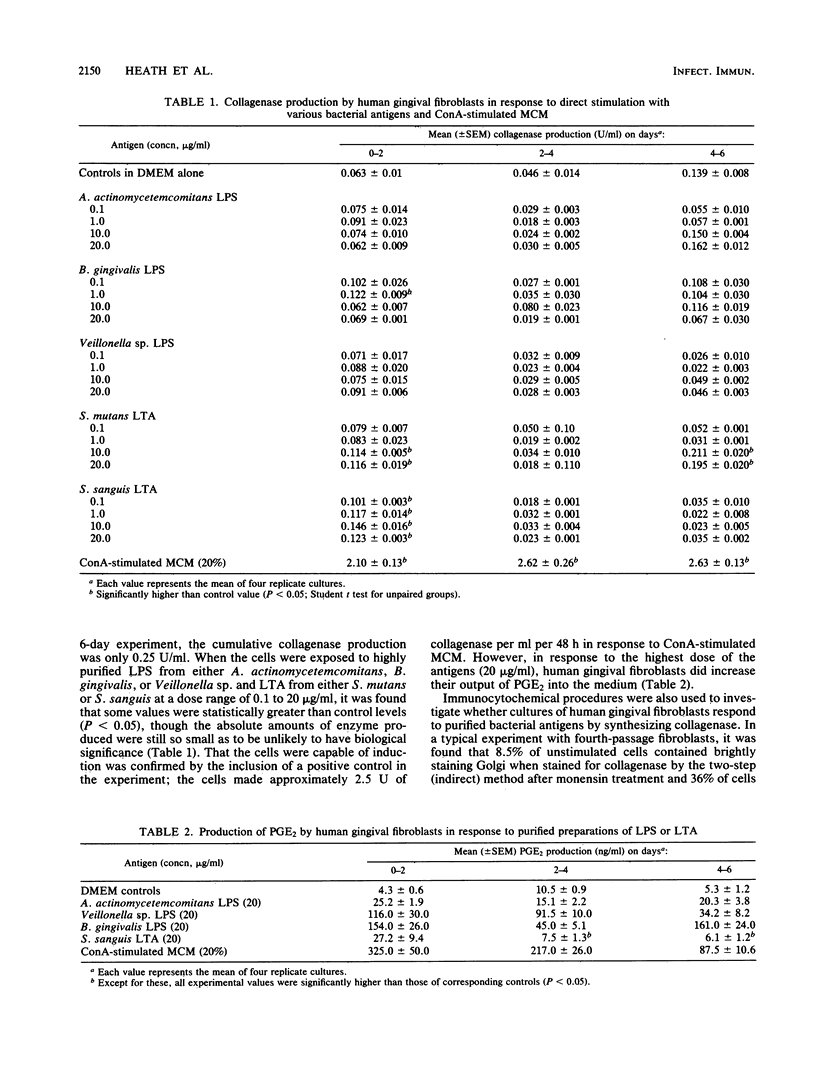
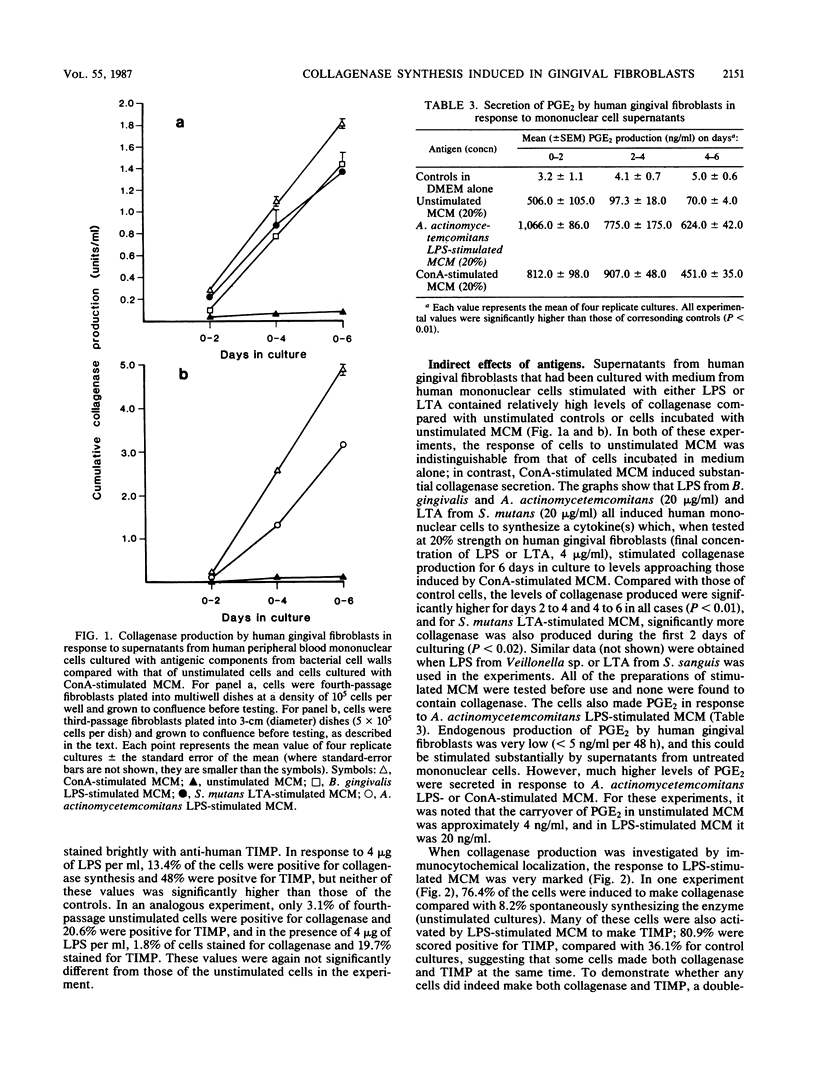
Our previous work suggests that one mechanism through which connective tissue breakdown might occur in periodontal diseases is the production of metalloproteinases, including collagenase, by gingival fibroblasts. In this study we investigated whether highly purified preparations of lipopolysaccharide (LPS) and lipoteichoic acid (LTA) from a number of putative periodontal pathogens could induce monolayer cultures of human gingival fibroblasts to synthesize collagenase and prostaglandin E2. Using both biochemical assays and immunocytochemical techniques, we found that cells synthesized only very small amounts of collagenase in direct response to LPS or LTA (0.1 to 20.0 micrograms/ml). At the highest dose of both antigens, prostaglandin E2 production was increased. We then studied whether LPS and LTA could signal collagenase production by interacting primarily with a different cell type. Our results show that LPS and LTA were each able to stimulate cultures of human blood mononuclear cells (greater than 95% monocytes) to release collagenase-inducing cytokines. By indirect immunocytochemistry, we found that a large proportion of human gingival fibroblasts was activated to produce collagenase by supernatants from LPS- and LTA-stimulated mononuclear cells, whereas gingival fibroblasts cultured with supernatants from unstimulated mononuclear cells were not. Furthermore, in a population of activated fibroblasts we demonstrated, using a double-labeling technique, that some cells made collagenase and the specific tissue inhibitor of metalloproteinases (TIMP) simultaneously. As yet, the collagenase-inducing signals remain poorly characterized but the interleukins-1 and tumor necrosis factors seem likely candidates.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arend W. P., Massoni R. J. Characteristics of bacterial lipopolysaccharide induction of interleukin 1 synthesis and secretion by human monocytes. Clin Exp Immunol. 1986 Jun;64(3):656–664. [PMC free article] [PubMed] [Google Scholar]
- Auron P. E., Webb A. C., Rosenwasser L. J., Mucci S. F., Rich A., Wolff S. M., Dinarello C. A. Nucleotide sequence of human monocyte interleukin 1 precursor cDNA. Proc Natl Acad Sci U S A. 1984 Dec;81(24):7907–7911. doi: 10.1073/pnas.81.24.7907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolini D. R., Nedwin G. E., Bringman T. S., Smith D. D., Mundy G. R. Stimulation of bone resorption and inhibition of bone formation in vitro by human tumour necrosis factors. Nature. 1986 Feb 6;319(6053):516–518. doi: 10.1038/319516a0. [DOI] [PubMed] [Google Scholar]
- Cawston T. E., Murphy G., Mercer E., Galloway W. A., Hazleman B. L., Reynolds J. J. The interaction of purified rabbit bone collagenase with purified rabbit bone metalloproteinase inhibitor. Biochem J. 1983 May 1;211(2):313–318. doi: 10.1042/bj2110313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W. T., Olden K., Bernard B. A., Chu F. F. Expression of transformation-associated protease(s) that degrade fibronectin at cell contact sites. J Cell Biol. 1984 Apr;98(4):1546–1555. doi: 10.1083/jcb.98.4.1546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dayer J. M., Beutler B., Cerami A. Cachectin/tumor necrosis factor stimulates collagenase and prostaglandin E2 production by human synovial cells and dermal fibroblasts. J Exp Med. 1985 Dec 1;162(6):2163–2168. doi: 10.1084/jem.162.6.2163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dayer J. M., de Rochemonteix B., Burrus B., Demczuk S., Dinarello C. A. Human recombinant interleukin 1 stimulates collagenase and prostaglandin E2 production by human synovial cells. J Clin Invest. 1986 Feb;77(2):645–648. doi: 10.1172/JCI112350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinarello C. A. Interleukin-1. Rev Infect Dis. 1984 Jan-Feb;6(1):51–95. doi: 10.1093/clinids/6.1.51. [DOI] [PubMed] [Google Scholar]
- Duncan S. J., Reynolds J. J. The effects of razoxane (ICRF 159) on the production of collagenase and inhibitor (TIMP) by stimulated rabbit articular chondrocytes. Biochem Pharmacol. 1983 Dec 15;32(24):3853–3858. doi: 10.1016/0006-2952(83)90160-0. [DOI] [PubMed] [Google Scholar]
- Galloway W. A., Murphy G., Sandy J. D., Gavrilovic J., Cawston T. E., Reynolds J. J. Purification and characterization of a rabbit bone metalloproteinase that degrades proteoglycan and other connective-tissue components. Biochem J. 1983 Mar 1;209(3):741–752. doi: 10.1042/bj2090741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gavrilovíc J., Reynolds J. J., Murphy G. Inhibition of type I collagen film degradation by tumour cells using a specific antibody to collagenase and the specific tissue inhibitor of metalloproteinases (TIMP). Cell Biol Int Rep. 1985 Dec;9(12):1097–1107. doi: 10.1016/s0309-1651(85)80007-2. [DOI] [PubMed] [Google Scholar]
- Hamilton J. A., Lingelbach S. R., Partridge N. C., Martin T. J. Stimulation of plasminogen activator in osteoblast-like cells by bone-resorbing hormones. Biochem Biophys Res Commun. 1984 Jul 18;122(1):230–236. doi: 10.1016/0006-291x(84)90464-9. [DOI] [PubMed] [Google Scholar]
- Heath J. K., Atkinson S. J., Meikle M. C., Reynolds J. J. Mouse osteoblasts synthesize collagenase in response to bone resorbing agents. Biochim Biophys Acta. 1984 Nov 6;802(1):151–154. doi: 10.1016/0304-4165(84)90046-1. [DOI] [PubMed] [Google Scholar]
- Heath J. K., Gowen M., Meikle M. C., Reynolds J. J. Human gingival tissues in culture synthesize three metalloproteinases and a metalloproteinase inhibitor. J Periodontal Res. 1982 Mar;17(2):183–190. doi: 10.1111/j.1600-0765.1982.tb01143.x. [DOI] [PubMed] [Google Scholar]
- Hembry R. M., Murphy G., Cawston T. E., Dingle J. T., Reynolds J. J. Characterization of a specific antiserum for mammalian collagenase from several species: immunolocalization of collagenase in rabbit chondrocytes and uterus. J Cell Sci. 1986 Mar;81:105–123. doi: 10.1242/jcs.81.1.105. [DOI] [PubMed] [Google Scholar]
- Hembry R. M., Murphy G., Reynolds J. J. Immunolocalization of tissue inhibitor of metalloproteinases (TIMP) in human cells. Characterization and use of a specific antiserum. J Cell Sci. 1985 Feb;73:105–119. doi: 10.1242/jcs.73.1.105. [DOI] [PubMed] [Google Scholar]
- Huff E. Lipoteichoic acid, a major amphiphile of Gram-positive bacteria that is not readily extractable. J Bacteriol. 1982 Jan;149(1):399–402. doi: 10.1128/jb.149.1.399-402.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiley P., Holt S. C. Characterization of the lipopolysaccharide from Actinobacillus actinomycetemcomitans Y4 and N27. Infect Immun. 1980 Dec;30(3):862–873. doi: 10.1128/iai.30.3.862-873.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- March C. J., Mosley B., Larsen A., Cerretti D. P., Braedt G., Price V., Gillis S., Henney C. S., Kronheim S. R., Grabstein K. Cloning, sequence and expression of two distinct human interleukin-1 complementary DNAs. Nature. 1985 Jun 20;315(6021):641–647. doi: 10.1038/315641a0. [DOI] [PubMed] [Google Scholar]
- Meikle M. C., Heath J. K., Reynolds J. J. Advances in understanding cell interactions in tissue resorption. Relevance to the pathogenesis of periodontal diseases and a new hypothesis. J Oral Pathol. 1986 May;15(5):239–250. doi: 10.1111/j.1600-0714.1986.tb00616.x. [DOI] [PubMed] [Google Scholar]
- Murphy G., McAlpine C. G., Poll C. T., Reynolds J. J. Purification and characterization of a bone metalloproteinase that degrades gelatin and types IV and V collagen. Biochim Biophys Acta. 1985 Sep 20;831(1):49–58. doi: 10.1016/0167-4838(85)90148-7. [DOI] [PubMed] [Google Scholar]
- Nouri A. M., Panayi G. S., Goodman S. M. Cytokines and the chronic inflammation of rheumatic disease. I. The presence of interleukin-1 in synovial fluids. Clin Exp Immunol. 1984 Feb;55(2):295–302. [PMC free article] [PubMed] [Google Scholar]
- Saklatvala J., Pilsworth L. M., Sarsfield S. J., Gavrilovic J., Heath J. K. Pig catabolin is a form of interleukin 1. Cartilage and bone resorb, fibroblasts make prostaglandin and collagenase, and thymocyte proliferation is augmented in response to one protein. Biochem J. 1984 Dec 1;224(2):461–466. doi: 10.1042/bj2240461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saklatvala J. Tumour necrosis factor alpha stimulates resorption and inhibits synthesis of proteoglycan in cartilage. Nature. 1986 Aug 7;322(6079):547–549. doi: 10.1038/322547a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sellers A., Reynolds J. J. Identification and partial characterization of an inhibitor of collagenase from rabbit bone. Biochem J. 1977 Nov 1;167(2):353–360. doi: 10.1042/bj1670353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The T. H., Feltkamp T. E. Conjugation of fluorescein isothiocyanate to antibodies. II. A reproducible method. Immunology. 1970 Jun;18(6):875–881. [PMC free article] [PubMed] [Google Scholar]
- Trechsel U., Dew G., Murphy G., Reynolds J. J. Effects of products from macrophages, blood mononuclear cells and or retinol on collagenase secretion and collagen synthesis in chondrocyte culture. Biochim Biophys Acta. 1982 Jul 22;720(4):364–370. doi: 10.1016/0167-4889(82)90113-6. [DOI] [PubMed] [Google Scholar]
- Werb Z., Mainardi C. L., Vater C. A., Harris E. D., Jr Endogenous activiation of latent collagenase by rheumatoid synovial cells. Evidence for a role of plasminogen activator. N Engl J Med. 1977 May 5;296(18):1017–1023. doi: 10.1056/NEJM197705052961801. [DOI] [PubMed] [Google Scholar]