Abstract
OBJECTIVE—To examine the association of the metabolic syndrome, defined by World Health Organization (WHO) and Adult Treatment Panel III (ATP-III) criteria, and its components with coronary artery calcium (CAC) progression.
RESEARCH DESIGN AND METHODS—Participants were 338 older community-dwelling men and women without known heart disease who had measurements of heart disease risk factors and CAC at two clinic visits within an average interval of 4.5 years. Progression was defined as an increase in total CAC volume score ≥2.5 mm3.
RESULTS—At baseline, mean age was 67.6 years; metabolic syndrome was present in 15.1% by WHO criteria and in 11.8% by ATP-III criteria, and 5.3% met both criteria. Participants with WHO-defined metabolic syndrome had a greater change in total CAC volume score than those without (P = 0.001). There was no significant difference in CAC volume change by ATP-III–defined metabolic syndrome status (P = 0.69). Overall, 46.4% of participants were CAC progressors. In logistic regression analyses adjusted for age, sex, smoking status, and LDL cholesterol, neither WHO–nor ATP-III–defined metabolic syndrome predicted CAC progression. Among metabolic syndrome components, only hypertension was independently associated with CAC progression (odds ratio 2.11 [95% CI 1.33–3.3], P = 0.002). Fasting blood glucose (>100 mg/dl) was an independent predictor of CAC progression, but only for the 118 participants younger than age 65 years (2.3 [1.01–5.5], P = 0.04).
CONCLUSIONS—In older adults without known heart disease, blood pressure levels and fasting plasma glucose were better independent determinants of CAC progression than metabolic syndrome itself.
Coronary artery calcium (CAC) assessed by electron-beam computed tomography (CT) is a marker of atherosclerotic plaque burden (1) and predicts future cardiac events independent of traditional coronary heart disease (CHD) risk factors (2,3). Moreover, CAC progression is associated with worsening of plaque burden as assessed by angiography (4). An increase of more than 15% in the total CAC score predicts an increased risk of myocardial infarction in observational studies (1,5,6).
Population-based studies using either World Health Organization (WHO) or National Cholesterol Education Program Adult Treatment Panel III (ATP-III) definitions of metabolic syndrome have shown that mortality from CHD is higher in people with metabolic syndrome (7,8). In cross-sectional studies, metabolic syndrome has been associated with greater CAC burden (8–10), and one study found that metabolic syndrome components such as hypertension and diabetes were independent predictors of CAC progression (11), but the effect of metabolic syndrome on CAC progression has not been reported.
We examined the prevalence of metabolic syndrome defined by WHO and ATP-III criteria in older community-dwelling ambulatory adults without known CHD and the independent association of baseline metabolic syndrome or its components with CAC progression in this cohort.
RESEARCH DESIGN AND METHODS
Participants were members of the Rancho Bernardo Study, a southern California community-based study of middle–to upper-middle–class Caucasian adults established in 1972. This article examines the surviving community-dwelling participants with no history of CHD (angina pectoris, myocardial infarction, or coronary artery revascularization) who participated in a clinic visit in 1997–1999 and returned for a follow-up visit in 2005–2006 (mean interval 4.5 ± 0.5 years). At the 1997–1999 visit, 422 participants had electron-beam CT of the heart to test for coronary artery calcification; 342 returned for the follow-up evaluation. Those who did not return for follow-up (n = 84) included refusals (n = 43), deaths (n = 21), and participants who were unreachable or who had cancelled their appointment for unknown reasons (n = 20). In addition, four participants completed the second visit but refused blood draw and were excluded, leaving 338 participants for the present analyses. Compared with the participants who returned for follow-up, those who did not return were older and had higher levels of systolic blood pressure and fasting plasma glucose (FPG) at the baseline visit but did not differ in other risk factors. All participants provided written informed consent at both visits. The study protocol was approved by the Human Research Protection Program at the University of California San Diego.
Data collection
In 1997–1999, height and weight were measured in participants wearing light clothing and no shoes using a regularly calibrated scale and stadiometer. BMI was calculated as weight in kilograms divided by the square of height in meters. Waist circumference was measured in standing subjects midway between the inferior lateral margin of the ribs and the superior lateral border of the iliac crest. Hip circumference was measured as the widest hip circumference.
FPG and HDL and LDL cholesterol levels were measured in a lipid research clinic laboratory using standard enzymatic methods in blood samples collected after an overnight, usually 12-h, fast. Systolic and diastolic blood pressure was measured twice in seated resting subjects by certified staff according to a standard protocol (12). Hypertension was defined as systolic blood pressure ≥140 and/or diastolic ≥90 mmHg or use of antihypertensive medication. Other cardiovascular risk factors including current cigarette smoking, alcohol intake (at least three times a week), and physical activity (exercise at least three times a week) were assessed using standard questionnaires. Baseline medication use was validated by a trained nurse who examined pills and prescriptions brought to the clinic for that purpose; medication use at the time of the second CAC evaluation was obtained by questionnaire.
Metabolic syndrome was defined in two ways, in accord with WHO criteria (13) or ATP-III guidelines (14). Metabolic syndrome defined by WHO criteria required type 2 diabetes, impaired fasting glucose (fasting glucose 110–125 mg/dl), or impaired glucose tolerance (postchallenge glucose 140–199 mg/dl) plus any two of the following: hypertension (≥140/90 mmHg or use of antihypertensive drugs), high triglycerides (≥150 mg/dl), low HDL cholesterol (<35 mg/dl for men and <39 mg/dl for women), obesity (BMI >30 kg/m2 and/or waist-to-hip ratio >0.9 for men and >0.85 for women), urinary albumin excretion rate ≥20 μg/min, or albumin-to-creatinine ratio ≥30 mg/g. Metabolic syndrome defined by ATP-III criteria required three or more of the following: FPG ≥110 mg/dl, abdominal obesity (waist circumference >102 for men cm and >88 cm for women), high triglycerides (≥150 mg/dl), low HDL cholesterol (<40 mg/dl for men and <50 mg/dl for women), or hypertension (≥130/85 mmHg or use of antihypertensive drugs).
CT imaging protocol
CAC scores were assessed using an electron-beam CT (Imatron C-150 scanner; Imatron, San Francisco, CA). Heart images were obtained with 100-ms scan time using 3-mm slices starting at the level of the carina and proceeding to the level of the diaphragm; ∼40–45 slices were obtained. Tomographic imaging was electrocardiographically triggered at 40 or 65% of the RR interval, depending on the participant's heart rate. CAC was defined as a plaque of at least two pixels (area 0.67 mm2) with a density ≥130 Hounsfield units (HU). Quantitative calcium scores were calculated using the method of Agatston et al. (15) and by volumetric scores (acquired by multiplying the pixel area by the section thickness of the region of interest). The total volume scores were derived by the sum of all lesion volumes in cubic millimeters.
Statistical analyses
All analyses were performed using SPSS (version 13.1; SPSS, Chicago, IL). In univariate analyses, clinical characteristics and metabolic syndrome components were compared according to the presence of metabolic syndrome by ATP-III or WHO criteria using Student's t tests. P < 0.05 was considered significant. Variables with normal distribution were presented as means ± SD, and those with non-normal distribution are presented as median (interquartile range). Skewed variables were log transformed for statistical analyses.
CAC scores were categorized according to the criteria of Rumberger et al. (16), which define CAC scores of 0–10 HU as none/minimal, 11–99 HU as mild, 100–399 HU as moderate, and ≥400 HU as severe. CAC progression was analyzed as a continuous outcome (CAC volume scores change = CAC volume score on second visit − CAC volume score at baseline visit) and as a categorical outcome (CAC progression yes/no). We defined progression as a difference between baseline CAC and follow-up square-root–transformed total CAC volume score ≥2.5 mm3 (17). Because interscan variability and error depend on CAC absolute value (CAC variability and score error are larger in patients with increased baseline CAC scores than in those with lower scores), this definition provides an estimate of change that is unbiased with respect to baseline CAC. In the present study, this definition of CAC progression is between the mean and −1 SD of square-root–transformed CAC change, which is approximately equivalent to an annual increase of 30% in CAC absolute values and concordant with previous studies that observed an annual 24% CAC increase in untreated adults (18).
The association between the presence of metabolic syndrome or its individual components and total CAC volume score change was examined using linear regression before and after adjustment for potential non–metabolic syndrome confounding variables (age, sex, smoking status, and LDL cholesterol). These covariates were chosen based on established associations with CAC and atherosclerosis; highly correlated variables such as total cholesterol and LDL cholesterol were not used in the same model. The Hosmer and Lemeshow test was applied to evaluate whether the estimates of the model fit the data at an acceptable level (P > 0.05). Individual risk factors were examined as predictors of categorical CAC progression in logistic regression models before and after adjustment for the same confounders. CAC progression analyses were repeated stratifying by age <65 and ≥65 years.
RESULTS
Participants included 156 men and 182 women aged 67.6 ± 7.6 years. Mean follow-up was 4.5 ± 0.5 years. The average BMI ± SD was 26.2 ± 3.8 kg/m2, and the mean total cholesterol ± SD was 208.1 ± 34 mg/dl; 43.5% were hypertensive, and 7.2% had diabetes. Half the cohort had never smoked, and 5.3% were current smokers. Prevalence of metabolic syndrome at baseline was 15.1% (44 of 292) by WHO criteria and 11.8% (40 of 338) by ATP-III criteria. (The sample size was smaller for participants classified by WHO criteria because only 292 of the 338 participants were evaluated for baseline albuminuria.) Only 18 of 292 subjects (5.3%) met both definitions of metabolic syndrome. Table 1 shows prevalence of metabolic syndrome components for participants with and without WHO–and ATP-III–defined metabolic syndrome. By definition, participants with metabolic syndrome by either criteria had increased conventional risk factors (blood pressure, fasting glucose, triglycerides, and low HDL cholesterol) as well as adverse anthropometric characteristics compared with those without. Total and LDL cholesterol were not associated with metabolic syndrome by either definition.
Table 1.
Baseline prevalence of metabolic syndrome defined by WHO and ATP-III criteria and components in older adults without known CHD
| Yes | No | |
|---|---|---|
| Metabolic syndrome defined by WHO criteria | ||
| n | 44 | 248 |
| Prevalence of WHO components (%) | ||
| Impaired glucose metabolism (type 2 diabetes, impaired fasting glucose [110–125 mg/dl] or impaired glucose tolerance [140–199 mg/dl]) | 100.0 | 5.2 |
| Obesity (BMI >30 kg/m2 and/or waist-to-hip ratio >0.9 for men and >0.85 for women) | 97.7 | 55.2 |
| High triglycerides (≥150 mg/dl) | 56.8 | 25.8 |
| Low HDL cholesterol (<35 mg/dl for men and <39 mg/dl for women) | 11.4 | 1.2 |
| Hypertension (≥140/90 mmHg or use of antihypertensive drugs) | 84.1 | 37.1 |
| Albuminuria (urinary albumin excretion rate ≥20 μg/min or albumin-to-creatinine ratio ≥30 mg/g) | 9.1 | 2.8 |
| Metabolic syndrome defined by ATP-III criteria | ||
| n | 40 | 298 |
| Prevalence of ATP-III components (%) | ||
| Fasting plasma glucose >110 mg/dl | 47.5 | 14.8 |
| Abdominal obesity (waist circumference >102 cm for men and >88 cm for women) | 72.5 | 14.1 |
| High triglycerides (≥150 mg/dl) | 80.0 | 22.5 |
| Low HDL cholesterol (<40 mg/dl for men and <50 mg/dl for women) | 80.0 | 7.4 |
| Hypertension (≥130/85 mmHg or use of antihypertensive drugs) | 90.0 | 51.7 |
Participants with WHO-defined metabolic syndrome at baseline were more likely to be using a diuretic (25 vs. 9.7%, P = 0.04), cholesterol-lowering medication (29.5 vs. 14.9%, P = 0.01), or calcium channel blocker (18.2 vs. 6.9%, P = 0.01) than those without metabolic syndrome, and estrogen therapy was less common among women with metabolic syndrome (13.2 vs. 41.9%, P = 0.03). There were no differences in the use of aspirin, β-blockers, or ACE inhibitors. More individuals with metabolic syndrome by ATP-III criteria were using diuretics (27.5 vs. 9.7%, P < 0.001) or β-blockers (27.5 vs. 6.7%, P < 0.001) than those without metabolic syndrome, but use of cholesterol-lowering medications, calcium channel blockers, ACE inhibitors, aspirin, or estrogen therapy did not differ significantly.
Cross-sectional CAC score results and metabolic syndrome defined by WHO and ATP-III
At the baseline visit, 37% of participants had none/minimal, 21.6% had mild, 19.5% had moderate, and 21.9% had severe CAC according to Rumberger category. Individuals with WHO-defined metabolic syndrome were more likely to be in a more severe Rumberger category of CAC at the baseline visit than those without metabolic syndrome (none/minimal, 6.8 vs. 44%; mild, 15.9 vs. 22.6%; moderate, 31.8 vs. 16.5%; severe, 45.5 vs. 16.9%; P for trend <0.001). In contrast, the presence of metabolic syndrome defined by ATP-III criteria was not associated with CAC category at baseline (none/minimal, 30 vs. 37.9%; mild, 25 vs. 21.1%; moderate, 20 vs. 19.5%; severe, 25 vs. 21.5%; P for trend 0.73). When prevalence of metabolic syndrome components by both criteria and CAC categories were compared, only hypertension, impaired glucose metabolism, and obesity increased with CAC severity (Table 2).
Table 2.
Prevalence of metabolic syndrome defined by WHO and ATP-III criteria and components by CAC score category at baseline
| CAC score category |
P | ||||
|---|---|---|---|---|---|
| None/minimal | Mild | Moderate | Severe | ||
| n | 125 | 73 | 66 | 74 | |
| Prevalence of WHO components (%) | |||||
| Impaired glucose metabolism (type 2 diabetes, impaired fasting glucose [110–125 mg/dl] or impaired glucose tolerance [140–199 mg/dl]) | 7.2 | 17.8 | 28.8 | 31.1 | <0.001 |
| Obesity (BMI >30 kg/m2 and/or waist-to-hip ratio >0.9 for men and >0.85 for women) | 40.0 | 68.5 | 71.2 | 82.4 | <0.001 |
| High triglycerides (≥150 mg/dl) | 28.0 | 28.8 | 30.3 | 31.1 | 0.61 |
| Low HDL cholesterol (<35 mg/dl for men and <39 mg/dl for women) | 0.8 | 2.7 | 6.1 | 4.1 | 0.08 |
| Hypertension (≥140/90 mmHg or use of antihypertensive drugs) | 32.8 | 44.3 | 53 | 55.4 | 0.001 |
| Albuminuria (urinary albumin excretion rate ≥20 μg/min or albumin-to-creatinine ratio ≥30 mg/g)* | 4.5 | 1.6 | 3.6 | 4.8 | 0.90 |
| Prevalence of ATP-III components (%) | |||||
| Fasting plasma glucose >110 mg/dl | 7.2 | 17.8 | 27.3 | 31.1 | <0.001 |
| Abdominal obesity (waist circumference >102 cm for men and >88 cm for women) | 17.6 | 20.5 | 19.7 | 28.4 | 0.10 |
| High triglycerides (≥150 mg/dl) | 28.0 | 28.8 | 30.3 | 31.1 | 0.61 |
| Low HDL cholesterol (<40 mg/dl for men and <50 mg/dl for women) | 15.2 | 17.8 | 18.2 | 13.1 | 0.87 |
| Hypertension (≥130/85 mmHg or use of antihypertensive drugs) | 40.8 | 61.6 | 66.7 | 67.6 | <0.001 |
N = 292: none/minimal CAC, n = 112; mild CAC, n = 63; moderate CAC, n = 55; and severe CAC, n = 62.
The CAC volume score at baseline (median [interquartile range]) was significantly higher among participants with metabolic syndrome by WHO criteria (231.4 mm3 [430]) compared with participants without metabolic syndrome by WHO criteria (25.3 mm3 [155], P < 0.001). In similar comparisons between those with and without metabolic syndrome defined by ATP-III criteria, there were no differences in CAC volume scores at baseline (71 mm3 [409] vs. 34 mm3 [201], P = 0.34).
Changes in CAC volume score and metabolic syndrome defined by WHO and ATP-III
Overall, 157 (46.4%) participants had significant CAC progression (change in square-root volume score ≥2.5 mm3) over the 4.5 years of follow-up. The increase in CAC volume score (median [interquartile range]) was higher in participants with metabolic syndrome by WHO criteria (102 mm3 [227]) than in those without (22.9 mm3 [85], P = 0.001); however, the proportion of CAC progressors did not differ (54.5 vs. 44%, respectively, P = 0.19). Comparing CAC changes between those with and without metabolic syndrome by ATP-III criteria, neither the absolute change (21 mm3 [118] vs. 31 mm3 [110], P = 0.69) nor the proportion of CAC progressors (45 vs. 46.6%, P = 0.84) were significantly different.
Multivariate analysis
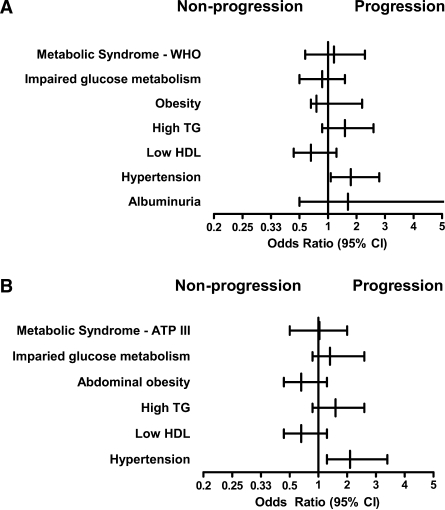
Multivariate linear regression was used to assess the association between CAC volume score change and individual components of metabolic syndrome as continuous variables adjusting for age, sex, LDL cholesterol, and smoking status. Only blood pressure (standardized β-coefficient: systolic blood pressure 0.19, P = 0.001; diastolic blood pressure 0.14, P = 0.007) and FPG (standardized β-coefficient 0.16, P = 0.02) were independently associated with CAC progression (Table 3). The same multivariable model was used to assess the association between metabolic syndrome by WHO or ATP-III criteria with categorical (yes/no) progression of CAC. Metabolic syndrome neither by WHO nor by ATP-III was associated with CAC progression in models adjusted for age, sex, LDL cholesterol, and smoking status (Fig. 1). Of the individual components, only hypertension was associated with CAC progression (odds ratio [OR] 2.11 [95% CI 1.33–3.3], P = 0.002) (Fig. 1). Moreover, CAC volume changes were greater for higher categories of systolic blood pressure (CAC volume change [square root]: systolic blood pressure <120 mmHg, 1.8 ± 2.2; 120–140 mmHg, 2.4 ± 2.8; 140–160 mmHg, 3.1 ± 3.2; >160 mmHg, 4.1 ± 3.0; P = 0.003 for linear trend) in models adjusting for the same covariates.
Table 3.
Standardized β-coefficients for multivariate linear regressions of individual metabolic syndrome components and change in total CAC volume score (square-root)
| Metabolic syndrome components | Standardized β-coefficient* | P |
|---|---|---|
| Fasting blood glucose (mg/dl) | 0.16 | 0.02 |
| Waist circumference (cm) | 0.10 | 0.11 |
| Waist-to-hip ratio | 0.16 | 0.07 |
| BMI (kg/m2) | 0.04 | 0.43 |
| Triglycerides (mg/dl) | 0.04 | 0.36 |
| HDL cholesterol (mg/dl) | 0.06 | 0.29 |
| Systolic blood pressure (mmHg) | 0.19 | 0.001 |
| Diastolic blood pressure (mmHg) | 0.14 | 0.007 |
| Albuminuria (mg/l) | 0.05 | 0.38 |
Adjusted for sex, age, current smoking status, and LDL cholesterol.
Figure 1.
Odds ratios for metabolic syndrome and metabolic syndrome components as predictors for CAC progression adjusted for age, sex, LDL cholesterol, and smoking habit. A: Metabolic syndrome by WHO criteria and components. B: Metabolic syndrome by ATP-III criteria and components.
In a post hoc analysis stratified by age-group, fasting hyperglycemia (>100 mg/dl) was an independent predictor of CAC progression (OR 2.3 [95% CI 1.01–5.5], P = 0.04) for participants younger than 65 years of age (n = 118) but not for the older group. Age-stratified analyses did not differ for other metabolic syndrome components or for the presence of the metabolic syndrome. Further adjustment for medication use did not materially change any of the results.
CONCLUSIONS
The prevalence of metabolic syndrome in this older Caucasian population without known CHD was 11–13% depending on metabolic syndrome definition; metabolic syndrome by either definition was not an independent predictor of CAC progression. Among the metabolic syndrome components, only hypertension was independently associated with CAC change overall, while hyperglycemia independently predicted CAC progression only in individuals younger than age 65 years.
The prevalence of the metabolic syndrome in adult Caucasians in the U.S. is reported to be 20–25% (19). The lower prevalence in our cohort likely reflects the fact that individuals with known CHD were excluded. This might have had the effect of reducing associations by exclusion of those with more risk factors who already had heart disease. This fits with the age-stratified analyses, where results were more pronounced in participants younger than age 65 years.
The finding that individuals with metabolic syndrome by WHO but not by ATP-III criteria had higher values of CAC at the baseline visit and more progression during follow-up is concordant with a small study (100 men aged 30–39 years) in which metabolic syndrome defined by WHO, but not ATP-III, was independently associated with CAC >10 HU (20). This study did not report CAC progression.
The main CHD risk factors reported to be associated with CAC progression are age, male sex, baseline CAC scores, diabetes, hypertension, and use of cholesterol-lowering medication (5,6,21). In the large (5,756 participants) Multi-Ethnic Study of Atherosclerosis (MESA), which evaluated risk factors for CAC progression over 2.4 years, many cardiovascular risk factors (age, male sex, hypertension, BMI, diabetes, and family history of heart attack) were associated with both the risk of developing incident CAC and increases in existing calcification (11). Compared with the present study, MESA was much larger, had a shorter interval between scans, and defined CAC progression as any newly detectable CAC, which may not allow a fair comparison. Moreover, MESA did not evaluate metabolic syndrome as a predictor of CAC progression.
Based on new evidence, the usefulness of the metabolic syndrome definition has been questioned. After the present analysis was completed, in a study based on a large cohort of 7,258 adults aged 65 years or older and free of CHD, metabolic syndrome by either ATP-III or WHO definitions did not predict CHD mortality, but two components (blood pressure and glucose) did predict CHD mortality (22). These results parallel those in the present study, in which, after adjusting for confounders, blood pressure and glucose but not metabolic syndrome were useful in predicting those that will have progression of subclinical atherosclerosis. In our study, the presence of hypertension was associated with a 110% increased likelihood of CAC progression, and its presence was a better predictor than the presence of metabolic syndrome by WHO or ATP-III criteria.
In summary, metabolic syndrome defined by WHO was superior to that defined by ATP-III in predicting subclinical atherosclerosis by CAC scores. When other classical heart disease risk factors were included, the presence of the metabolic syndrome itself did not predict CAC progression, but two of its components, systolic blood pressure and FPG, were independently associated with CAC changes. These results highlight the importance of these reversible risk factors in older adults.
Acknowledgments
The Rancho Bernardo Study was funded by the National Institutes of Health/National Institute on Aging (grants AG07181 and AG028507) and the National Institute of Diabetes and Digestive and Kidney Diseases (grant DK31801).
No potential conflicts of interest relevant to this article were reported.
Published ahead of print at http://care.diabetesjournals.org on 13 October 2008.
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C Section 1734 solely to indicate this fact.
References
- 1.Greenland P, Bonow RO, Brundage BH, Budoff MJ, Eisenberg MJ, Grundy SM, Lauer MS, Post WS, Raggi P, Redberg RF, Rodgers GP, Shaw LJ, Taylor AJ, Weintraub WS: ACCF/AHA 2007 clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain: a report of the American College of Cardiology Foundation Clinical Expert Consensus Task Force (ACCF/AHA Writing Committee to Update the 2000 Expert Consensus Document on Electron Beam Computed Tomography) developed in collaboration with the Society of Atherosclerosis Imaging and Prevention and the Society of Cardiovascular Computed Tomography. J Am Coll Cardiol 49:378–402, 2007 [DOI] [PubMed] [Google Scholar]
- 2.Kondos GT, Hoff JA, Sevrukov A, Daviglus ML, Garside DB, Devries SS, Chomka EV, Liu K: Electron-beam tomography coronary artery calcium and cardiac events: a 37-month follow-up of 5635 initially asymptomatic low- to intermediate-risk adults. Circulation 107:2571–2576, 2003 [DOI] [PubMed] [Google Scholar]
- 3.Bellasi A, Lacey C, Taylor AJ, Raggi P, Wilson PW, Budoff MJ, Vaccarino V, Shaw LJ: Comparison of prognostic usefulness of coronary artery calcium in men versus women (results from a meta- and pooled analysis estimating all-cause mortality and coronary heart disease death or myocardial infarction). Am J Cardiol 100:409–414, 2007 [DOI] [PubMed] [Google Scholar]
- 4.Tani T, Yamakami S, Matsushita T, Okamoto M, Toyama J, Fukutomi T, Itoh M: Comparison of coronary artery calcium progression by electron beam computed tomography and angiographically defined progression. Am J Cardiol 91:865–867, 2003 [DOI] [PubMed] [Google Scholar]
- 5.Raggi P, Cooil B, Ratti C, Callister TQ, Budoff M: Progression of coronary artery calcium and occurrence of myocardial infarction in patients with and without diabetes mellitus. Hypertension 46:238–243, 2005 [DOI] [PubMed] [Google Scholar]
- 6.Budoff MJ, Raggi P: Coronary artery disease progression assessed by electron-beam computed tomography. Am J Cardiol 88:46E–50E, 2001 [DOI] [PubMed] [Google Scholar]
- 7.Malik S, Wong ND, Franklin SS, Kamath TV, L'Italien GJ, Pio JR, Williams GR: Impact of the metabolic syndrome on mortality from coronary heart disease, cardiovascular disease, and all causes in United States adults. Circulation 110:1245–1250, 2004 [DOI] [PubMed] [Google Scholar]
- 8.Isomaa B, Almgren P, Tuomi T, Forsen B, Lahti K, Nissen M, Taskinen MR, Groop L: Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 24:683–689, 2001 [DOI] [PubMed] [Google Scholar]
- 9.Santos RD, Nasir K, Tufail K, Meneghelo RS, Carvalho JA, Blumenthal RS: Metabolic syndrome is associated with coronary artery calcium in asymptomatic white Brazilian men considered low-risk by Framingham risk score. Prev Cardiol 10:141–146, 2007 [DOI] [PubMed] [Google Scholar]
- 10.Kullo IJ, Cassidy AE, Peyser PA, Turner ST, Sheedy PF 2nd, Bielak LF: Association between metabolic syndrome and subclinical coronary atherosclerosis in asymptomatic adults. Am J Cardiol 94:1554–1558, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Kronmal RA, McClelland RL, Detrano R, Shea S, Lima JA, Cushman M, Bild DE, Burke GL: Risk factors for the progression of coronary artery calcification in asymptomatic subjects: results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 115:2722–2730, 2007 [DOI] [PubMed] [Google Scholar]
- 12.The hypertension detection and follow-up program: Hypertension detection and follow-up program cooperative group. Prev Med 5:207–215, 1976 [DOI] [PubMed] [Google Scholar]
- 13.Alberti KG, Zimmet PZ: Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1. Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 15:539–553, 1998 [DOI] [PubMed] [Google Scholar]
- 14.National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults: Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 285:2486–2497, 2001 [DOI] [PubMed] [Google Scholar]
- 15.Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R: Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15:827–832, 1990 [DOI] [PubMed] [Google Scholar]
- 16.Rumberger JA, Sheedy PF 2nd, Breen JF, Fitzpatrick LA, Schwartz RS: Electron beam computed tomography and coronary artery disease: scanning for coronary artery calcification. Mayo Clin Proc 71:369–377, 1996 [DOI] [PubMed] [Google Scholar]
- 17.Hokanson JE, MacKenzie T, Kinney G, Snell-Bergeon JK, Dabelea D, Ehrlich J, Eckel RH, Rewers M: Evaluating changes in coronary artery calcium: an analytic method that accounts for interscan variability. AJR Am J Roentgenol 182:1327–1332, 2004 [DOI] [PubMed] [Google Scholar]
- 18.Maher JE, Bielak LF, Raz JA, Sheedy PF, 2nd, Schwartz RS, Peyser PA: Progression of coronary artery calcification: a pilot study. Mayo Clin Proc 74:347–355, 1999 [DOI] [PubMed] [Google Scholar]
- 19.Ford ES, Giles WH, Dietz WH: Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA 287:356–359, 2002 [DOI] [PubMed] [Google Scholar]
- 20.Takamiya T, Zaky WR, Edmundowics D, Kadowaki T, Ueshima H, Kuller LH, Sekikawa A: World Health Organization–defined metabolic syndrome is a better predictor of coronary calcium than the Adult Treatment Panel III criteria in American men aged 40–49 years. Diabetes Care 27:2977–2979, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Callister TQ, Raggi P, Cooil B, Lippolis NJ, Russo DJ: Effect of HMG-CoA reductase inhibitors on coronary artery disease as assessed by electron-beam computed tomography. N Engl J Med 339:1972–1978, 1998 [DOI] [PubMed] [Google Scholar]
- 22.Mozaffarian D, Kamineni A, Prineas RJ, Siscovick DS: Metabolic syndrome and mortality in older adults: the Cardiovascular Health Study. Arch Intern Med 168:969–978, 2008 [DOI] [PubMed] [Google Scholar]