Abstract
The pleiotropic cytokine interleukin 6 (IL-6) has favorable and harmful effects on survival from bacterial infections. While many innate immune cells produce IL-6, little is known about relevant sources in vivo and the nature of its contributions to host responses to severe bacterial infections. To examine these roles, we subjected mast cell-specific IL-6-deficient mice to the cecal ligation and puncture model of septic peritonitis, finding that survival in these mice is markedly worse than in controls. Following intranasal or intraperitoneal inoculation with Klebsiella pneumoniae, IL-6-/- mice are less likely to survive than wild-type controls and at the time of death have higher numbers of bacteria but not inflammatory cells in lungs and peritoneum. Similarly, mast cell-specific IL-6-deficient mice have diminished survival and higher numbers of K. pneumoniae following intraperitoneal infection. Neutrophils lacking IL-6 have greater numbers of live intracellular K. pneumonia, suggesting impaired intracellular killing contributes to reduced clearance in IL-6-/- mice. These results establish that mast cell IL-6 is a critical mediator of survival following K. pneumoniae infection and sepsis and suggest that IL-6 protects from death by augmenting neutrophil killing of bacteria.
Keywords: Bacterial killing, cytokine, innate immunity, pulmonary inflammation
Introduction
In the United States, pneumonia and sepsis remain leading causes of death despite availability of broad-spectrum antibiotics. In 2002, nearly 100,000 deaths were attributed to pneumonia and sepsis, making them the fifth leading causes of death (1). Furthermore, the aging population will likely be associated with an increase in the prevalence of lung infections and sepsis in the future. Because of this significant disease burden, it is imperative we learn more of the pathogenesis of infections of the lower respiratory tract and sepsis so we can derive treatments that will improve patient outcomes.
The innate immune system provides the initial defense against infectious microbes (2). Resident innate immune cells include dendritic cells (3), macrophages (4), gd-T cells (5), and mast cells (6). These cells rely on germline-encoded receptors to recognize conserved microbial products produced by the infecting bacteria. Stimulation of these receptors leads to release of pro-inflammatory mediators (e.g., TNF-α, IFN-γ, IL-6) that recruit or activate other innate immune cells, such as neutrophils, to the site of infection, where they kill bacteria using reactive oxygen species (7, 8) or proteases to hydrolyze cell wall proteins (9, 10).
Important cytokines that mediate host defense include TNF-α (11), IL-6 (12), IL-12 (13), and IL-18 (14) among others. IL-6 is a pleiotropic cytokine that plays disparate roles in inflammatory conditions such as the host response to bacterial infection (15). Animal models of bacterial infection suggest it protects the host from death following infection. For example, IL-6-/- mice have higher mortality when infected with Escherichia coli, Klebsiella pneumoniae, or Streptococcus pneumoniae (12, 16, 17). In contrast, treating mice with IL-6 blocking antibodies improves survival from polymicrobial septic peritonitis by reducing C5a receptor expression (18). Thus, IL-6 plays diverse and contrasting roles in the host response to bacterial infections. While these studies show that IL-6 regulates survival, they do not explain its pleiotropic effects.
We became interested in the role of IL-6 in bacterial infections while studying dipeptidyl peptidase I (DPPI)-deficient mice in the cecal ligation and puncture (CLP) model of septic peritonitis (19). In those studies, we found DPPI-deficient mice are more likely to survive septic peritonitis. This protection is due to the absence of mast cell-DPPI, which increases peritoneal levels of IL-6. In these studies, the observation that lack of IL-6 reduces survival in DPPI-deficient mice indicated the survival advantage was dependent on IL-6, but it did not identify the cellular source of the beneficial IL-6. We hypothesized that mast cell IL-6 may be a critical cellular source of IL-6 in the CLP model. To test this hypothesis in the current work, we studied mast cell-specific IL-6 deficient mice in response to CLP sepsis and K. pneumonia lung and peritonitis infection. Our results suggest that mast cell IL-6 is a major mediator of survival from these severe infections and that it protects from death by enhancing intracellular killing of bacteria by neutrophils.
Materials and Methods
Materials
All chemicals were from Sigma (St. Louis, MO) unless otherwise noted.
Experimental animals
C57BL/6 IL-6-/- and IL-6+/+ mice were purchased from Jackson Labs (Bar Harbor, ME). C57BL/6 KitW-sh/KitW-sh mice (20) were provided by Peter Besmer (Memorial Sloan-Kettering Institute, New York, NY). All experimental procedures were performed in 8-12 week-old mice and were approved by the UCSF Committee on Animal Research.
Cecal ligation and puncture
A 1-cm midline incision was made in the abdominal wall of anesthetized mice and the cecum identified. The distal 50% of exposed cecum was ligated with 3-0 silk suture and punctured once with a 25-gauge needle. The cecum was replaced into the abdomen, the incision closed with 3-0 suture and the mouse recovered with a 0.5 ml intraperitoneal injection of sterile 0.9% NaCl. Mice were monitored three times daily and survival recorded. Moribund mice were euthanized by CO2 inhalation and cervical dislocation.
Induction of K. pneumoniae infection in mice
K. pneumoniae (strain 43816, serotype 2, American Type Culture Collection, Manassas, VA) was resuspended in 5 cc of Nutrient Broth (Difco) and cultured overnight at 37°C. 100 μl of this suspension was added to 50 ml of Nutrient Broth and grown for 3-4 hours to log phase when CFU’s were determined by OD600 readings and confirmed by culture. Using this K.pneumoniae, gram-negative pneumonia was generated in mice by inoculating anesthetized mice intranasally via a sterile pipet tip with 3000 CFU of K. pneumoniae suspended in 50 μl of culture medium. K. pneumoniae septic peritonitis was generated in mice by injecting the peritoneum of mice with 150 CFU of K. pneumoniae suspended in 200 μl of culture medium. Mice were recovered from anesthesia and survival monitored three times daily. Moribund mice were euthanized by CO2 inhalation and cervical dislocation.
Mast cell culture from bone marrow
Mouse bone marrow mast cells (BMMC) were cultured in r-m-IL-3 and r-m-SCF (Peprotech, Rocky Hill, NJ) as described (21). Cells were used after 5 weeks in culture, at which time the cell populations consisted of > 95% mast cells (identified by metachromatic granules in toluidine blue-stained cells). Further, BMMC cultured from IL-6+/+ and IL-6-/- bone marrow showed similar granular morphology, levels of active tryptase, and expression of FceR-Ia and CD117, indicating they have similar maturation when cultured in the presence of IL-3 and SCF.
Mast cell reconstitution of KitW-sh/KitW-sh mice
We used C57BL/6 KitW-sh/KitW-sh mast cell-deficient mice (22) for these studies because they have the same genetic background as the IL-6-/- mice and have no mast cells detectable by metachromatic staining (23). To reconstitute intraperitoneal mast cells, 4×106 IL-6+/+ or IL-6-/- BMMC suspended in 500 μl of sterile PBS were injected into the peritoneum of 5-week old KitW-sh/KitW-sh mice. After allowing 5 weeks for mast cells to differentiate within the peritoneum (24, 25), reconstituted mice were used in experiments. This method selectively reconstitutes peritoneal mast cells to similar levels in the mesentery or peritoneum (3.6 × 105 ± 1.2 × 105 vs. 3.4 × 105 ± 2.3 × 105 mast cells/peritoneum respectively) using either IL-6+/+ or IL-6-/- BMMC.
To reconstitute lungs of KitW-sh/KitW-sh mice, 107 BMMC suspended in 200 μl of PBS were injected into the tail vein of 5-week-old KitW-sh/KitW-sh mice. After allowing 12 weeks for mast cells to migrate to and differentiate within lung (23, 26), the reconstituted mice were used in experiments. This method selectively reconstitutes lung mast cells to similar levels using either IL-6+/+ or IL-6-/- BMMC.
Quantification of cellular response to infection
To recover intraperitoneal inflammatory cells, anesthetized mice were euthanized by cervical dislocation and the abdominal skin cleansed with 70% ethanol. 4 ml of sterile 0.9 % NaCl was then instilled into the peritoneum. The abdomen was massaged gently for 1 min, and opened with a sterile scissors and lavage fluid reclaimed. Similarly, to recover intrapulmonary inflammatory cells, lungs of euthanized mice were lavaged 3x with 0.7 cc of sterile 0.9 % NaCl. Recovered lavage fluid was pooled. In both cases, recovered lavage fluid was centrifuged at 850 g for 4 min at 4°C and the supernatant saved for cytokine analysis. Cell pellets were resuspended in red cell lysis buffer (Sigma) for 10 min, recentrifuged, and the cell pellet resuspended in PBS. Cell numbers were counted with a hemocytometer and cell differentials determined on cytospun cells stained with Diff-Quik (American Scientific Products, McGaw Park, IL).
Quantification of bacterial colony forming units (CFU’s)
Lungs and spleens were aseptically harvested, placed in 500 μl sterile saline and homogenized. 10 μl of tissue homogenate or peritoneal lavage fluid were diluted serially in sterile 0.9 % NaCl. 10 μl of each dilution were aseptically plated and cultured on tryptose blood agar plates (for CLP experiments), or nutrient agar for non-fastidious microorganism plates (for K. pneumoniae infection experiments) at 37°C. After 24 h, the numbers of bacterial colonies counted.
Cytokine analysis
Cytokine concentrations were measured in lung and peritoneal lavage fluid using ELISA kits: MIP-2 (R & D systems, Minneapolis, MN), KC (R & D systems), TNF-a (eBioscience, San Diego, CA), IL-6 (R & D systems), and IFN-γ (eBioscience) according to the manufacturers' protocols.
Neutrophil isolation
Neutrophils were isolated from murine bone marrow by flushing front and hind legs of mice with Salt-free HBSS (Invitrogen). The bone marrow cells were then layered on an 82-62% Percoll (Sigma) gradient buffered with Salt-Free HBSS, 0.02 M HEPES, 0.5% FBS (Gibco), and 0.04 M NaHC03 (Sigma) then spun at 1200g at 18°C for 30 min. Neutrophils were collected from the interface between the percoll layers and resuspended in Krebs ringers. Neutrophil purity was 85-95% when assessed by Diff-Quik (American Scientific Products) staining.
Intracellular killing assay
Fresh cultures of K. pneumoniae were suspended in KRP at 4 × 108 CFU/ml. 50 μl of bacteria were then added to 4 × 106 neutrophils with or without a 1-h pretreatment with 100 or 500 ng/ml recombinant murine IL-6 (Peprotech), or 4 × 106 thioglycolate elicited peritoneal macrophages, suspended in 2 ml of KRP and incubated at 37°C for 1 hour. Gentamicin (100 μg/ml) was added to kill extracellular bacteria, and the cells were subsequently washed then incubated at 18°C for varying lengths of time. Cells were then washed three times in ice cold Salt-free HBSS, and lysed in 200 ml of Salt-free HBSS containing 0.1% Triton X-100 (Sigma). 20 ml of the lysate was then plated on LB-Agar plates (Sigma), cultured overnight at 37°C and colonies counted.
Mast cell Klebsiella killing
500 CFU of freshly cultured K. pneumoniae were suspended in 40 μl of RPMI 1640 containing 5000 BMMC, with or without a 1-h pretreatment with 0.5 ng/ml recombinant murine IL-6 (Peprotech), and incubated at 37°C. Media containing bacteria but no mast cells was cultured as control. After 60 min 10 μl of the mixture was plated on nutrient agar plates, cultured overnight at 37°C and colonies counted.
Statistics
Survival curves were analyzed using the two-tailed Fisher's exact test. ANOVA followed by two-tailed t testing was used to compare markers of organ dysfunction, bacterial CFU's, and mean cytokine concentrations. All calculations were performed using Statview 5.0.1 software (SAS Institute Inc., Cary, NC). Significance was assigned to P values < 0.05
Results
Mast cell-IL-6 modulates survival following polymicrobial septic peritonitis
To test the hypothesis that mast cell IL-6 modulates sepsis survival, we compared IL-6 levels in peritoneal lavage fluid from mast cell-deficient KitW-sh/KitW-sh mice selectively reconstituted with IL-6-/- bone marrow mast cells (BMMC) (pmc-IL-6-/- mice) to lavage fluid from KitW-sh/KitW-sh mice selectively reconstituted with IL-6+/+ BMMC (pmc-IL-6+/+ mice). We found that mice lacking IL-6 solely in mast cells (pmc-IL-6-/- mice) had lower levels of peritoneal IL-6 2 h (but not 12 h) after CLP (Fig. 1A). Thus, mast cells are a key intraperitoneal source of IL-6 early after the onset of polymicrobial peritoneal infection.
FIGURE 1.
Mast cell-IL-6 regulates survival from septic peritonitis. (A) Concentrations of IL-6 were measured by ELISA in peritoneal lavage fluid obtained from pmc-IL-6+/+ and pmc-IL-6-/- mice 2 and 12 h after CLP. Note that lavage fluid from pmc-IL-6-/- mice has significantly lower levels of IL-6 compared to that obtained from pmc-IL-6+/+ mice 2h but not 12h after CLP. (B) pmc-IL-6-/- mice are protected from death by septic peritonitis. pmc-IL-6+/+ (n = 24) and pmc-IL-6-/- (n = 25) mice were subjected to CLP (50% ligation and single puncture with an 25-gauge needle) and survival monitored for 8 d. The figure is representative of survival curves from three separate experiments. (C, D) Bacterial load and intraperitoneal inflammatory cell recruitment after CLP is similar in pmc-IL-6+/+ and pmc-IL-6-/- mice. Pmc-IL-6+/+ and pmc-IL-6-/- mice (n = 7-9 mice per group) were euthanized 12 and 24 h after CLP. Peritoneal lavaged fluid was cultured on blood agar plates. Total cells were counted using a hemocytometer and the cell types determined on cytospun cells stained with Diff-Quik. (E) Intraperitoneal bacteria cultured on blood agar plates are not pathogenic. Peritoneums of 10 wild-type mice were injected with 106 CFU of bacteria (suspended in 200 μl culture medium) cultured from lavage fluid of mice subjected to CLP. Survival was monitored for 8 d after inoculation. Data are representative of two separate experiments. *P = 0.01, **P < 0.01.
Next, we examined whether this early production of mast cell IL-6 regulates survival following septic peritonitis by comparing survival of pmc-IL-6-/- and pmc-IL-6+/+ mice subjected to CLP. In so doing, we found that survival in mast cell-specific IL-6-deficient pmc-IL-6-/- mice is markedly worse than in pmc-IL-6+/+ control mice (37% vs. 12%, Fig. 1B) 8 days after onset of peritonitis. These results indicate mast cell IL-6 is a major regulator of survival following septic peritonitis.
To understand how mast cell IL-6 modulates survival, we examined endpoints commonly explaining differences in survival in this model, including cellular indices of inflammation, cytokine levels, and bacterial load at intervals after CLP. Surprisingly, we found no differences (Fig. 1C and D, and data not shown).
Because IL-6 reportedly enhances bacterial clearance and the pathogenic bacteria in the CLP model are unknown, we suspected we were not culturing the pathogenic bacteria using standard blood agar plates and therefore underestimating differences in bacterial load of the true pathogenic bacteria. To test this possibility, we inoculated peritoneums of ten C57BL/6 wild-type (WT) mice with 106 CFU of bacteria recovered on the blood agar plates and found that none of the infected mice died 8 days after inoculation (Fig. 1E). Thus, the bacteria recovered on blood agar plates were not pathogenic, leaving unresolved the possibility that clearance of pathogenic bacteria is impaired in pmc-IL-6-/- mice.
Mast cell IL-6 regulates survival and bacterial clearance following peritoneal K. pneumoniae infection
Our lack of a biological explanation for the survival advantage of pmc-IL-6-/- mice following CLP and the unanswered question of whether bacterial load of the pathogenic bacteria was different in pmc-IL-6-/- mice prompted us to study whether IL-6 regulated survival from K. pneumoniae septic peritonitis. This model allowed us to reliably quantify the load of infectious bacteria. First, IL-6+/+ and IL-6-/- mice were infected intraperitoneally with K. pneumoniae. We found that IL-6-/- mice have significantly worse survival and impaired clearance of K. pneumoniae from the peritoneum 6, or 24 hours after inoculation compared to IL-6+/+ controls (Fig. 2A and B). In contrast, there was no difference in the number or type of inflammatory cells in the peritoneum 6, 12 or 24 hours after inoculation in IL-6+/+or IL-6-/-mice (Fig. 2C). Next, to determine whether mast cell IL-6 regulates K. pneumoniae peritonitis, we infected pmc-IL-6-/- and pmc-IL-6+/+ mice, finding that survival is worse and peritoneal clearance of K. pneumoniae is impaired in pmc-IL-6-/- mice (Fig. 3A and B). Further, there were no significant differences in the number or type of inflammatory cells in the peritoneum of pmc-IL-6+/+or pmc-IL-6-/- mice 6 hours after inoculation (Fig. 3C). These data indicate that mast cell IL-6 modulates survival from K. pneumoniae infections by enhancing bacterial clearance.
FIGURE 2.
Mast cell-IL-6 regulates survival following K. pneumoniae peritonitis. (A) IL-6+/+ and IL-6-/- mice (n = 25 mice per group) were inoculated with 150 CFU of K. pneumoniae (strain 43816, serotype 2) suspended in 200 μl PBS and survival monitored. Note that IL-6-/-mice have significantly worse survival than controls. (B) IL-6-/- mice have impaired clearance of K. pneumonia from the peritoneum. IL-6+/+ and IL-6-/- mice inoculated intraperitoneally with 150 CFU of K. pneumonia were euthanized 6 h or 24 h after infection, their peritoneums lavaged and bacterial CFU's quantified by culturing dilutions of lavage fluid on nutrient agar plates. (C) Intraperitoneal inflammatory cell recruitment is normal in IL-6-/- mice. IL-6+/+ and IL-6-/- mice inoculated intraperitoneally with 150 CFU of K. pneumonia were euthanized 6 h, 12 h and 24 h after infection, their peritoneums lavaged and numbers of inflammatory cells recovered counted. n = 6-9 mice per group. The survival curve is representative of two separate experiments, and CFU quantification from three separate experiments. *P <0.003, **P < 0.05.
FIGURE 3.
Mast cell-IL-6 regulates survival following K. pneumoniae peritonitis. (A) pmc-IL-6+/+ (n = 30) and pmc-IL-6-/- (n = 29) mice were inoculated with 150 CFU of K. pneumoniae suspended in 200 μl PBS and survival monitored. Note that pmc-IL-6-/- mice have significantly worse survival than controls. (B) pmc-IL-6-/- mice have impaired clearance of K. pneumonia from the peritoneum. pmc-IL-6+/+ and pmc-IL-6-/- mice inoculated intraperitoneally with 150 CFU of K. pneumonia were euthanized 24 h after infection, their peritoneums lavaged and bacterial CFU's quantified by culturing dilutions of lavage fluid on nutrient agar plates. n = 6-9 mice per group. (C) Intraperitoneal inflammatory cell recruitment is normal in pmc-IL-6-/- mice. Pmc-IL-6+/+ and pmc-IL-6-/- mice inoculated intraperitoneally with 150 CFU of K. pneumonia were euthanized 6 h after infection, their peritoneums lavaged and numbers of inflammatory cells recovered counted. n = 9 mice per group. The survival curve is representative of two separate experiments, and CFU quantification from three separate experiments. *P = 0.02.
IL-6-/- mice have impaired bacterial clearance and worse survival following K. pneumonia lung infection
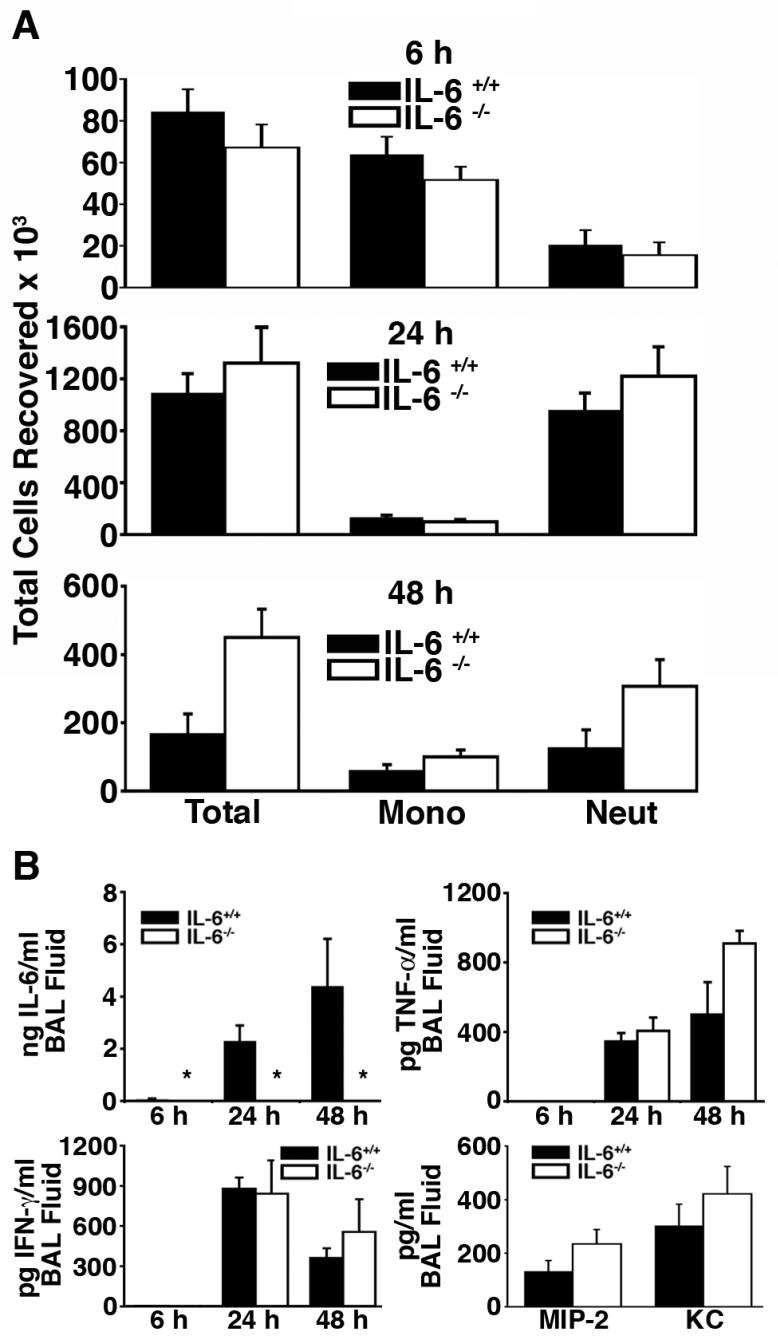
Next, we sought to extend our understanding of the role of IL-6 to K. pneumoniae lung infections. Similar to peritoneal infection, we found that following intranasal inoculation with K. pneumoniae IL-6-/- mice have significantly worse survival than IL-6+/+ controls (0% vs. 32%) 8 d after inoculation (Fig. 4A). At the time they succumb to infection (24- 48 hours after inoculation), IL-6-/- mice have significantly higher numbers of bacteria in their lungs and spleens (Fig. 4B) and qualitatively, 8/8 IL-6-/- mice had a heavy growth of bacteria cultured from their mediastinum compared to only 3/8 IL-6+/+ mice (Fig. 4C). IL-6-/- mice recruited inflammatory cells normally as levels of lung cellular inflammation were similar in IL-6+/+ and-/- mice 6, 24 and 48 h after infection (Fig. 5A). Interestingly, with the exception of IL-6, the IL-6-/- mice did not express differences in TNF-a, IFN-g, MIP-2, or KC compared to IL-6+/+ controls at various time-points after infection (Fig. 5B). These data indicate that IL-6 regulates survival from lung infection and that the higher mortality rate of IL-6-/- mice correlates with impaired bacterial clearance.
FIGURE 4.
IL-6 is required for survival from K. pneumonia lung infection. (A) IL-6+/+ (n = 30) and IL-6-/- (n = 27) mice were intranasally inoculated with 3000 CFU of K. pneumonia and survival monitored for 8d. Note that IL-6-/- mice have significantly worse survival than IL-6+/+ mice 8 d after infection. The figure is representative of three separate experiments. (B) IL-6-/- mice have impaired clearance of K. pneumonia from the lung. IL-6+/+ and IL-6-/-mice (n = 7-9 mice per group) inoculated with 3000 CFU of K. pneumonia were euthanized 24 or 48 h after infection, their lungs and spleens aseptically harvested and homogenized in 500 μl sterile saline and bacterial CFU's quantified by culturing dilutions of homogenates on nutrient agar plates. Note there are significantly more bacteria in the lung and spleen homogenates obtained from IL-6-/- mice. Similar results were found in two separate experiments. (C) IL-6-/- mice have a greater burden of mediastinal bacteria than IL-6+/+ controls. 48 hours after intranasal inoculation with 3000 cfu of K. pneumonia, the mediastinums of euthanized mice were swiped once with a sterile swab and cultured on nutrient agar plate. Note that 7/8 of the swabs from IL-6-/- mice had large bacterial growth compared to only 3/8 from IL-6+/+ mice. *P = 0.05, **P = 0.01.
FIGURE 5.
IL-6-deficient mice have normal recruitment of inflammatory cells to the lung. (A) IL-6+/+ and IL-6-/- mice (n = 8-10 mice per group) were euthanized 6h, 24 h and 48h after intranasal inoculation of 3000 CFU of K. pneumonia. Lungs were lavaged and the total cells in lavage fluid counted using a hemocytometer. Cell differentials determined on cytospun cells stained with Diff-Quik. (B) Cytokine measurements. Concentrations of TNF-a, IL-6, and IFN-g were measured by ELISA in bronchoalveolar lavage fluid obtained from IL-6+/+ and IL-6-/- mice 6, 24 and 48 h and MIP-2 and KC 6 h after intranasal inoculation of 3000 CFU of K. pneumonia. n = 8-10 mice/group. *P < 0.001.
Next we tested whether mast cell IL-6 modulates survival following K. pneumonia lung infection. First, to test whether mast cells are important in this model, we compared survival of WT and KitW-sh/KitW-sh mice infected intranasally with K. pneumoniae, finding that mast cell-deficient KitW-sh/KitW-sh mice have worse survival 4 d after inoculation (Fig. 6A). To test whether mast cell IL-6 regulates survival from K. pneumonia lung infection, we first measured survival of KitW-sh/KitW-sh mice pretreated with IL-6 and found that KitW-sh/KitW-sh mice pretreated with IL-6 have better survival 5 d after inoculation with K. pneumoniae than saline treated controls (Fig. 6C). This result indicates that exogenous IL-6 administration improves survival of KitW-sh/KitW-sh mice and suggests an absence of mast cell IL-6 is partially responsible for the worse survival of KitW-sh/KitW-sh mice. To further investigate whether the reduced survival of KitW-sh/KitW-sh mice reflects the absence of mast cells and to test whether mast cell IL-6 influences survival, we also compared the survival of KitW-sh/KitW-sh mice and KitW-sh/KitW-sh mice reconstituted with BMMC cultured from IL-6+/+ and IL-6-/- mice. Surprisingly, we found no difference in survival of the three groups of mice (Fig. 6B).
FIGURE 6.
Mast cells regulate survival following K. pneumonia lung infections. (A) Kit+/Kit+ (n = 15) and KitW-sh/KitW-sh mice (n = 23) were intranasally inoculated with 3000 CFU of K. pneumonia and survival monitored for 4 d. (B) KitW-sh/KitW-sh mice and KitW-sh/KitW-sh mice reconstituted with IL-6+/+ (pmc-IL-6+/+) or IL-6-/-( pmc-IL-6-/-) BMMC (n = 25 mice per group) were intranasally inoculated with 3000 CFU of K. pneumonia and survival monitored for 4 d. (C) KitW-sh/KitW-sh mice pretreated 30 min before infection with 100 μl saline, 50 ng rm-IL-6, or 1000 ng rm-IL-6 were intranasally inoculated with 3000 CFU of K. pneumonia and survival monitored for 5 d (n=20 mice/group). The figure is representative of survival curves from three separate experiments. *P < 0.001.
IL-6 directly activates neutrophils to enhance intracellular killing of K. pneumoniae
Bacterial killing by macrophages, mast cells, and neutrophils are major contributors to bacterial clearance (10, 27, 28). To investigate whether a defect in intracellular Klebsiella killing by macrophages explained the higher bacterial load in IL-6-/- mice, we quantified live intracellular bacteria in macrophages isolated from IL-6+/+ and IL-6-/- mice after in vitro inoculation and found similar numbers of intracellular bacteria in IL-6 deficient macrophages at various time-points after inoculation (Fig. 7A). Next, to test whether killing of K. pneumoniae is defective in IL-6-/- mast cells, we compared total Klebsiella killing by IL-6+/+and IL-6-/- mast cells. Again finding no difference (Fig 7B). These observations indicate a defect in Klebsiella killing by macrophages or mast cells do not explain the higher bacterial load detected in IL-6-/- mice.
FIGURE 7.
IL-6 activation of neutrophils enhances intracellular killing of K. pneumoniae. (A) IL-6+/+ and IL-6-/- macrophages were incubated in the presence of K. pneumoniae. 1 h after incubation, macrophages were washed in PBS containing gentamicin, and at various time-points after washing, lysed and live intracellular K. pneumoniae identified by culture. (B) IL-6+/+ and IL-6-/- BMMC treated with and without IL-6 were incubated in the presence of K. pneumoniae. 60 min after incubation, aliquots of the mixture were removed and the number of live K. pneumoniae identified by culture. Media containing bacteria but no mast cells (No MC) was cultured as control. (C) IL-6+/+ and IL-6-/- mice were euthanized 24 h after intranasal inoculation of 3000 CFU of K. pneumonia. Their lungs were lavaged with PBS containing gentamicin, neutrophils isolated, lysed and number of live intracellular K. pneumoniae identified by culture. (D) IL-6+/+ and IL-6-/- neutrophils or (E) IL-6-/- neutrophils treated with and without IL-6 were incubated in the presence of K. pneumoniae. 1 h after incubation, neutrophils were washed in PBS containing gentamicin. 30 - 90 min (60 min, panel E) after washing, neutrophils were lysed and live intracellular K. pneumoniae identified by culture. Experiments were performed in triplicate and are representative of at least two independent assays. *P ≤ 0.03, **P = 0.06, ***P = 0.05.
To test whether neutrophil killing of K. pneumoniae is defective in IL-6 deficient mice in vivo, we quantified the number of intracellular bacteria in neutrophils isolated from lungs of IL-6w+/+ and IL-6-/- mice 24 h after intranasal K. pneumoniae inoculation, finding higher numbers of Klebsiella in neutrophils from IL-6 deficient mice (Fig. 7C). Because bacterial clearance is impaired in IL-6-/- mice, the differences in intracellular bacteria was possibly due to the higher bacterial load, rather than a defect in intracellular killing by IL-6 deficient neutrophils. To differentiate these possibilities, we next quantified live intracellular bacteria in neutrophils isolated from IL-6+/+ and IL-6-/- mice after in vitro inoculation, finding more intracellular bacteria in IL-6 deficient neutrophils (Fig. 7D). Because phagocytosis in leukocytes isolated from IL-6-/- mice is normal (data not shown), these data indicate the impaired clearance is due, at least in part, to defective intracellular killing by neutrophils. Finally, to determine whether IL-6 acts directly on neutrophils to enhance killing, we compared intracellular killing of K. pneumoniae by IL-6 deficient neutrophils in the presence and absence of IL-6, finding that addition of IL-6 enhances neutrophil killing (Fig. 7E). These data show that IL-6 directly activates neutrophils to enhance intracellular killing of K. pneumoniae.
Discussion
This work shows for the first time that mast cell IL-6 regulates survival from pneumonia and sepsis by activating neutrophils and enhancing killing of intracellular bacteria. In addition, the pleiotropic behavior of IL-6 during sepsis may be explained, in part, by our finding that mast cell sources of IL-6 are beneficial while others may be detrimental to the host during sepsis. This dependence of IL-6 activity on its cellular source predicts that attempts to achieve systemic blockade of IL-6 during sepsis may fail to be therapeutic because they simultaneously block detrimental and beneficial sources.
Rat, mouse and human mast cells have all been reported to make IL-6 in culture (29-34) or in specific disease states in humans (35). Depending on conditions, mast cells make low levels of IL-6 constitutively (29, 33, 36), and increase its production in response to various stimuli such as SCF (37), PGE2 (38, 39), and IgE (30), TLR2 (40), or TLR 4 (40, 41) receptor activation in rodent mast cells and IgE receptor activation (34, 42) and IL-1 (43) in human mast cells. Unlike TNF-α (44) and IL-15 (45) mast cells do not store IL-6 in their secretory granules. Instead, they traffic it via a separate vesicular compartment targeted for immediate release (43). While mast cells have long been recognized to make IL-6, only recently have experiments using mast cell specific IL-6 deficient mice been used to establish that mast cells sources of IL-6 play critical roles in pathologic processes such as development of abdominal aortic aneurysms and atherosclerosis (46, 47). This report is unique because it is the first to demonstrate a protective role for mast cell IL-6 following severe bacterial infections in mice. Whether mast cell-IL-6 similarly improves survival following severe bacterial infections in humans remains to be established.
Mast cells are sentinel cells, which are present in most tissues where they serve to monitor the local microenvironment for invading microorganisms and bacteria (48). When infectious bacteria are present, molecules unique to the bacteria activate receptors on the mast cell, triggering release of mediators (e.g. tryptase and TNF-α) that recruit neutrophils to the site of infection (6, 24, 49, 50). The current work extends these findings by showing that mast cells also release IL-6, which activates the recruited neutrophil to enhance killing of invading bacteria. Mast cell IL-6 seems to be required only early after infection because by 12 hours other sources compensate for the mast cell contribution (Fig. 1).
Mast cell IL-6 profoundly impacts survival despite being required only in the early hours after infection. Our data suggest it regulates survival by enhancing killing by early-arriving neutrophils, thereby lowering the initial burden of bacteria. Conversely, in the absence of mast cell IL-6, greater numbers of the initial bacterial inoculum are able to survive the early mast cell dependent responses to the infection. This higher bacterial load then multiplies and overwhelms subsequent defenses to the infection and in the case of K. pneumoniae lung infection, the bacteria penetrate outside the lung parenchyma into the circulation and mediastinum with resultant mortality from overwhelming infection. Thus, host survival depends critically on mast cells and their ability to modulate bacterial load in the first hours after infection.
The poor survival of mast cell-deficient mice after intranasal inoculation with K. pneumoniae suggests that mast cells regulate host responses to lung and airway infection. However, mast cell reconstitution studies did not differentiate whether this survival difference is due to the absence of mast cells or a general defect in Kit receptor signaling in KitW-sh/KitW-sh mice. In contrast, experiments showing mast cell IL-6 modulates K. pneumoniae peritoneal infection (Fig. 3), indicate that mast cells do play a role in controlling K. pneumoniae infections and suggest the reconstitution studies did not detect a role for mast cells in the lung infection model because the mast cells reside in a different compartment of reconstituted lung (23).
Because intravenous injection of mast cells reconstitutes the lung parenchyma rather than their normal location in the trachea and large airways, we believe the reconstitution studies provide further evidence supporting the idea that mast cells are essential for very early responses to infection, but dispensable in the later stages of infection. In wild-type mice, mast cells normally are present in greatest numbers in the trachea where they are poised to readily detect and respond to the initial bacterial inoculum. In contrast, in the reconstituted mouse, mast cells are in the distal lung parenchyma where they encounter bacteria only after the initial inoculum has multiplied within the proximal airways and extended deep into the parenchyma. By this stage in the infection, the bacterial load is sufficiently high that the mast cell responses are inconsequential compared to other innate immune cells (macrophages and neutrophils), which are present in greater numbers in the lung parenchyma.
IL-6 is a pleiotropic cytokine (15) that, depending on the infection model, has been reported to have a beneficial (12, 16, 51, 52), detrimental (18), or neutral (19, 53) effect on survival of bacterial infection. This manuscript provides evidence that the pleiotropic effects of IL-6 can be explained by the idea that some (mast cell) cellular sources of IL-6 have a favorable, and others a detrimental, effect on survival. For example, when wild-type and IL-6 deficient mice are subjected to CLP, the survival of both groups is identical 8 days later (19, 53), suggesting that IL-6 does not regulate survival from septic peritonitis. However, in this report, we show that mice lacking IL-6 exclusively in mast cells are less likely to survive than mice reconstituted with wild-type mast cells 8 d after CLP. These results indicate that mast cell-derived IL-6 protects from death from septic peritonitis and, by extension, that non-mast cell sources of IL-6 likely promote death during septic peritonitis. Identification of these detrimental cellular sources of IL-6 and determination of whether, in addition to mast cells, other sources of IL-6 benefit the host requires future study.
In summary, the data presented here establish that mast cell-IL-6 is a major mediator of survival following K. pneumoniae infection and sepsis and that it protects the host from death by augmenting intracellular neutrophil killing of K. pneumoniae. While mast cell-IL-6 appears to be critical only in the first hours after infection, it has profound effects on survival days after onset of the infection, emphasizing the major impact early mast cell responses have on survival.
Acknowledgements
The authors thank Guillaume Ly for his technical assistance and George Caughey for helpful discussions and critical reading of the manuscript.
This work was supported by NIH grant HL075026.
Abbreviations
- CLP
cecal ligation and puncture
- DPPI
dipeptidyl peptidase I
- BMMC
bone marrow mast cell
Footnotes
Disclosures The authors have no conflict of interest.
References
- 1.Anderson RN, Smith BL. Deaths: leading causes for 2002. Natl Vital Stat Rep. 2005;53:1–89. [PubMed] [Google Scholar]
- 2.Janeway CA, Jr., Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- 3.Lambrecht BN, Prins JB, Hoogsteden HC. Lung dendritic cells and host immunity to infection. Eur Respir J. 2001;18:692–704. [PubMed] [Google Scholar]
- 4.Gordon SB, Read RC. Macrophage defences against respiratory tract infections. Br Med Bull. 2002;61:45–61. doi: 10.1093/bmb/61.1.45. [DOI] [PubMed] [Google Scholar]
- 5.Moore TA, Moore BB, Newstead MW, Standiford TJ. Gamma delta-T cells are critical for survival and early proinflammatory cytokine gene expression during murine Klebsiella pneumonia. J Immunol. 2000;165:2643–2650. doi: 10.4049/jimmunol.165.5.2643. [DOI] [PubMed] [Google Scholar]
- 6.Malaviya R, Ikeda T, Ross E, Abraham SN. Mast cell modulation of neutrophil influx and bacterial clearance at sites of infection through TNF-alpha. Nature. 1996;381:77–80. doi: 10.1038/381077a0. [DOI] [PubMed] [Google Scholar]
- 7.Hirche TO, Gaut JP, Heinecke JW, Belaaouaj A. Myeloperoxidase plays critical roles in killing Klebsiella pneumoniae and inactivating neutrophil elastase: effects on host defense. J Immunol. 2005;174:1557–1565. doi: 10.4049/jimmunol.174.3.1557. [DOI] [PubMed] [Google Scholar]
- 8.Reeves EP, Lu H, Jacobs HL, Messina CG, Bolsover S, Gabella G, Potma EO, Warley A, Roes J, Segal AW. Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature. 2002;416:291–297. doi: 10.1038/416291a. [DOI] [PubMed] [Google Scholar]
- 9.Belaaouaj A, Kim KS, Shapiro SD. Degradation of outer membrane protein A in Escherichia coli killing by neutrophil elastase. Science. 2000;289:1185–1188. doi: 10.1126/science.289.5482.1185. [DOI] [PubMed] [Google Scholar]
- 10.Belaaouaj A, McCarthy R, Baumann M, Gao Z, Ley TJ, Abraham SN, Shapiro SD. Mice lacking neutrophil elastase reveal impaired host defense against gram negative bacterial sepsis. Nat Med. 1998;4:615–618. doi: 10.1038/nm0598-615. [DOI] [PubMed] [Google Scholar]
- 11.Laichalk LL, Kunkel SL, Strieter RM, Danforth JM, Bailie MB, Standiford TJ. Tumor necrosis factor mediates lung antibacterial host defense in murine Klebsiella pneumonia. Infect Immun. 1996;64:5211–5218. doi: 10.1128/iai.64.12.5211-5218.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.van der Poll T, Keogh CV, Guirao X, Buurman WA, Kopf M, Lowry SF. Interleukin-6 gene-deficient mice show impaired defense against pneumococcal pneumonia. J Infect Dis. 1997;176:439–444. doi: 10.1086/514062. [DOI] [PubMed] [Google Scholar]
- 13.Yamamoto N, Kawakami K, Kinjo Y, Miyagi K, Kinjo T, Uezu K, Nakasone C, Nakamatsu M, Saito A. Essential role for the p40 subunit of interleukin-12 in neutrophil-mediated early host defense against pulmonary infection with Streptococcus pneumoniae: involvement of interferon-gamma. Microbes Infect. 2004;6:1241–1249. doi: 10.1016/j.micinf.2004.08.007. [DOI] [PubMed] [Google Scholar]
- 14.Lauw FN, Branger J, Florquin S, Speelman P, van Deventer SJ, Akira S, van der Poll T. IL-18 improves the early antimicrobial host response to pneumococcal pneumonia. J Immunol. 2002;168:372–378. doi: 10.4049/jimmunol.168.1.372. [DOI] [PubMed] [Google Scholar]
- 15.Akira S, Taga T, Kishimoto T. Interleukin-6 in biology and medicine. Adv Immunol. 1993;54:1–78. doi: 10.1016/s0065-2776(08)60532-5. [DOI] [PubMed] [Google Scholar]
- 16.Dalrymple SA, Slattery R, Aud DM, Krishna M, Lucian LA, Murray R. Interleukin-6 is required for a protective immune response to systemic Escherichia coli infection. Infect Immun. 1996;64:3231–3235. doi: 10.1128/iai.64.8.3231-3235.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van Enckevort FH, Sweep CG, Span PN, Netea MG, Hermus AR, Kullberg BJ. Reduced adrenal response and increased mortality after systemic Klebsiella pneumoniae infection in interleukin-6-deficient mice. Eur Cytokine Netw. 2001;12:581–586. [PubMed] [Google Scholar]
- 18.Riedemann NC, Neff TA, Guo RF, Bernacki KD, Laudes IJ, Sarma JV, Lambris JD, Ward PA. Protective effects of IL-6 blockade in sepsis are linked to reduced c5a receptor expression. J Immunol. 2003;170:503–507. doi: 10.4049/jimmunol.170.1.503. [DOI] [PubMed] [Google Scholar]
- 19.Mallen-St. Clair J, Pham CTN, Villalta SA, Caughey GH, Wolters PJ. Mast Cell Dipeptidyl Peptidase I Mediates Survival from Sepsis. J Clin Invest. 2004;113:628–634. doi: 10.1172/JCI19062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Duttlinger R, Manova K, Berrozpe G, Chu TY, DeLeon V, Timokhina I, Chaganti RS, Zelenetz AD, Bachvarova RF, Besmer P. The Wsh and Ph mutations affect the c-kit expression profile: c-kit misexpression in embryogenesis impairs melanogenesis in Wsh and Ph mutant mice. Proc Natl Acad Sci U S A. 1995;92:3754–3758. doi: 10.1073/pnas.92.9.3754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Razin E, Ihle JN, Seldin D, Mencia-Huerta JM, Katz HR, LeBlanc PA, Hein A, Caulfield JP, Austen KF, Stevens RL. Interleukin 3: A differentiation and growth factor for the mouse mast cell that contains chondroitin sulfate E proteoglycan. J Immunol. 1984;132:1479–1486. [PubMed] [Google Scholar]
- 22.Tono T, Tsujimura T, Koshimizu U, Kasugai T, Adachi S, Isozaki K, Nishikawa S, Morimoto M, Nishimune Y, Nomura S, et al. c-kit Gene was not transcribed in cultured mast cells of mast cell- deficient Wsh/Wsh mice that have a normal number of erythrocytes and a normal c-kit coding region. Blood. 1992;80:1448–1453. [PubMed] [Google Scholar]
- 23.Wolters PJ, Mallen-St Clair J, Lewis CC, Villalta SA, Baluk P, Erle DJ, Caughey GH. Tissue-selective mast cell reconstitution and differential lung gene expression in mast cell-deficient Kit(W-sh)/Kit(W-sh) sash mice. Clin Exp Allergy. 2005;35:82–88. doi: 10.1111/j.1365-2222.2005.02136.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Echtenacher B, Mannel DN, Hultner L. Critical protective role of mast cells in a model of acute septic peritonitis. Nature. 1996;381:75–77. doi: 10.1038/381075a0. [DOI] [PubMed] [Google Scholar]
- 25.Nakano T, Sonoda T, Hayashi C, Yamatodani A, Kanayama Y, Yamamura T, Asai H, Yonezawa T, Kitamura Y, Galli SJ. Fate of bone marrow-derived cultured mast cells after intracutaneous, intraperitoneal, and intravenous transfer into genetically mast cell- deficient W/Wv mice. Evidence that cultured mast cells can give rise to both connective tissue type and mucosal mast cells. J Exp Med. 1985;162:1025–1043. doi: 10.1084/jem.162.3.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grimbaldeston MA, Chen CC, Piliponsky AM, Tsai M, Tam SY, Galli SJ. Mast Cell-Deficient W-sash c-kit Mutant KitW-sh/W-sh Mice as a Model for Investigating Mast Cell Biology in Vivo. Am J Pathol. 2005;167:835–848. doi: 10.1016/S0002-9440(10)62055-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Broug-Holub E, Toews GB, van Iwaarden JF, Strieter RM, Kunkel SL, Paine R, 3rd, Standiford TJ. Alveolar macrophages are required for protective pulmonary defenses in murine Klebsiella pneumonia: elimination of alveolar macrophages increases neutrophil recruitment but decreases bacterial clearance and survival. Infect Immun. 1997;65:1139–1146. doi: 10.1128/iai.65.4.1139-1146.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Malaviya R, Ross EA, MacGregor JI, Ikeda T, Little JR, Jakschik BA, Abraham SN. Mast cell phagocytosis of FimH-expressing enterobacteria. J Immunol. 1994;152:1907–1914. [PubMed] [Google Scholar]
- 29.Bradding P, Okayama Y, Howarth PH, Church MK, Holgate ST. Heterogeneity of human mast cells based on cytokine content. J Immunol. 1995;155:297–307. [PubMed] [Google Scholar]
- 30.Burd PR, Rogers HW, Gordon JR, Martin CA, Jayaraman S, Wilson SD, Dvorak AM, Galli SJ, Dorf ME. Interleukin 3-dependent and -independent mast cells stimulated with IgE and antigen express multiple cytokines. J Exp Med. 1989;170:245–257. doi: 10.1084/jem.170.1.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hultner L, Szots H, Welle M, Van Snick J, Moeller J, Dormer P. Mouse bone marrow-derived IL-3-dependent mast cells and autonomous sublines produce IL-6. Immunology. 1989;67:408–413. [PMC free article] [PubMed] [Google Scholar]
- 32.Marshall JS, Leal-Berumen I, Nielsen L, Glibetic M, Jordana M. Interleukin (IL)-10 inhibits long-term IL-6 production but not preformed mediator release from rat peritoneal mast cells. J Clin Invest. 1996;97:1122–1128. doi: 10.1172/JCI118506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Plaut M, Pierce JH, Watson CJ, Hanley-Hyde J, Nordan RP, Paul WE. Mast cell lines produce lymphokines in response to cross-linkage of Fc epsilon RI or to calcium ionophores. Nature. 1989;339:64–67. doi: 10.1038/339064a0. [DOI] [PubMed] [Google Scholar]
- 34.Zhao W, Oskeritzian CA, Pozez AL, Schwartz LB. Cytokine production by skin-derived mast cells: endogenous proteases are responsible for degradation of cytokines. J Immunol. 2005;175:2635–2642. doi: 10.4049/jimmunol.175.4.2635. [DOI] [PubMed] [Google Scholar]
- 35.Bradding P, Roberts JA, Britten KM, Montefort S, Djukanovic R, Mueller R, Heusser CH, Howarth PH, Holgate ST. Interleukin-4, -5, and -6 and tumor necrosis factor-alpha in normal and asthmatic airways: evidence for the human mast cell as a source of these cytokines. Am J Respir Cell Mol Biol. 1994;10:471–480. doi: 10.1165/ajrcmb.10.5.8179909. [DOI] [PubMed] [Google Scholar]
- 36.Okayama Y, Semper A, Holgate ST, Church MK. Multiple cytokine mRNA expression in human mast cells stimulated via Fc epsilon RI. Int Arch Allergy Immunol. 1995;107:158–159. doi: 10.1159/000236963. [DOI] [PubMed] [Google Scholar]
- 37.Gagari E, Tsai M, Lantz CS, Fox LG, Galli SJ. Differential release of mast cell interleukin-6 via c-kit. Blood. 1997;89:2654–2663. [PubMed] [Google Scholar]
- 38.Diaz BL, Fujishima H, Kanaoka Y, Urade Y, Arm JP. Regulation of prostaglandin endoperoxide synthase-2 and IL-6 expression in mouse bone marrow-derived mast cells by exogenous but not endogenous prostanoids. J Immunol. 2002;168:1397–1404. doi: 10.4049/jimmunol.168.3.1397. [DOI] [PubMed] [Google Scholar]
- 39.Gomi K, Zhu FG, Marshall JS. Prostaglandin E2 selectively enhances the IgE-mediated production of IL-6 and granulocyte-macrophage colony-stimulating factor by mast cells through an EP1/EP3-dependent mechanism. J Immunol. 2000;165:6545–6552. doi: 10.4049/jimmunol.165.11.6545. [DOI] [PubMed] [Google Scholar]
- 40.Supajatura V, Ushio H, Nakao A, Akira S, Okumura K, Ra C, Ogawa H. Differential responses of mast cell Toll-like receptors 2 and 4 in allergy and innate immunity. J Clin Invest. 2002;109:1351–1359. doi: 10.1172/JCI14704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.McCurdy JD, Lin TJ, Marshall JS. Toll-like receptor 4-mediated activation of murine mast cells. J Leukoc Biol. 2001;70:977–984. [PubMed] [Google Scholar]
- 42.Kruger-Krasagakes S, Moller A, Kolde G, Lippert U, Weber M, Henz BM. Production of interleukin-6 by human mast cells and basophilic cells. J Invest Dermatol. 1996;106:75–79. doi: 10.1111/1523-1747.ep12327815. [DOI] [PubMed] [Google Scholar]
- 43.Kandere-Grzybowska K, Letourneau R, Kempuraj D, Donelan J, Poplawski S, Boucher W, Athanassiou A, Theoharides TC. IL-1 induces vesicular secretion of IL-6 without degranulation from human mast cells. J Immunol. 2003;171:4830–4836. doi: 10.4049/jimmunol.171.9.4830. [DOI] [PubMed] [Google Scholar]
- 44.Gordon JR, Galli SJ. Mast cells as a source of both preformed and immunologically inducible TNF-alpha/cachectin. Nature. 1990;346:274–276. doi: 10.1038/346274a0. [DOI] [PubMed] [Google Scholar]
- 45.Orinska Z, Maurer M, Mirghomizadeh F, Bulanova E, Metz M, Nashkevich N, Schiemann F, Schulmistrat J, Budagian V, Giron-Michel J, Brandt E, Paus R, Bulfone-Paus S. IL-15 constrains mast cell-dependent antibacterial defenses by suppressing chymase activities. Nat Med. 2007;13:927–934. doi: 10.1038/nm1615. [DOI] [PubMed] [Google Scholar]
- 46.Sun J, Sukhova GK, Wolters PJ, Yang M, Kitamoto S, Libby P, Macfarlane LA, Clair JM, Shi GP. Mast cells promote atherosclerosis by releasing proinflammatory cytokines. Nat Med. 2007;13:719–724. doi: 10.1038/nm1601. [DOI] [PubMed] [Google Scholar]
- 47.Sun J, Sukhova GK, Yang M, Wolters PJ, MacFarlane LA, Libby P, Sun C, Zhang Y, Liu J, Ennis TL, Knispel R, Xiong W, Thompson RW, Baxter BT, Shi GP. Mast cells modulate the pathogenesis of elastase-induced abdominal aortic aneurysms in mice. J Clin Invest. 2007;117:3359–3368. doi: 10.1172/JCI31311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Galli SJ, Maurer M, Lantz CS. Mast cells as sentinels of innate immunity. Curr Opin Immunol. 1999;11:53–59. doi: 10.1016/s0952-7915(99)80010-7. [DOI] [PubMed] [Google Scholar]
- 49.Huang C, De Sanctis GT, O'Brien PJ, Mizgerd JP, Friend DS, Drazen JM, Brass LF, Stevens RL. Evaluation of the substrate specificity of human mast cell tryptase beta I and demonstration of its importance in bacterial infections of the lung. J Biol Chem. 2001;276:26276–26284. doi: 10.1074/jbc.M102356200. [DOI] [PubMed] [Google Scholar]
- 50.Maurer M, Echtenacher B, Hultner L, Kollias G, Mannel DN, Langley KE, Galli SJ. The c-kit ligand, stem cell factor, can enhance innate immunity through effects on mast cells. J Exp Med. 1998;188:2343–2348. doi: 10.1084/jem.188.12.2343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dalrymple SA, Lucian LA, Slattery R, McNeil T, Aud DM, Fuchino S, Lee F, Murray R. Interleukin-6-deficient mice are highly susceptible to Listeria monocytogenes infection: correlation with inefficient neutrophilia. Infect Immun. 1995;63:2262–2268. doi: 10.1128/iai.63.6.2262-2268.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ladel CH, Blum C, Dreher A, Reifenberg K, Kopf M, Kaufmann SH. Lethal tuberculosis in interleukin-6-deficient mutant mice. Infect Immun. 1997;65:4843–4849. doi: 10.1128/iai.65.11.4843-4849.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Leon LR, White AA, Kluger MJ. Role of IL-6 and TNF in thermoregulation and survival during sepsis in mice. Am J Physiol. 1998;275:R269–277. doi: 10.1152/ajpregu.1998.275.1.R269. [DOI] [PubMed] [Google Scholar]