Abstract
Globally, agriculture accounts for 80–90% of all freshwater used by humans, and most of that is in crop production. In many areas, this water use is unsustainable; water supplies are also under pressure from other users and are being affected by climate change. Much effort is being made to reduce water use by crops and produce ‘more crop per drop’. This paper examines water use by crops, taking particularly a physiological viewpoint, examining the underlying relationships between carbon uptake, growth and water loss. Key examples of recent progress in both assessing and improving crop water productivity are described. It is clear that improvements in both agronomic and physiological understanding have led to recent increases in water productivity in some crops. We believe that there is substantial potential for further improvements owing to the progress in understanding the physiological responses of plants to water supply, and there is considerable promise within the latest molecular genetic approaches, if linked to the appropriate environmental physiology. We conclude that the interactions between plant and environment require a team approach looking across the disciplines from genes to plants to crops in their particular environments to deliver improved water productivity and contribute to sustainability.
Keywords: water, crop productivity, water use efficiency, irrigation, crop physiology, drought
1. Introduction
The supply of some freshwater is an absolute essential for all forms of agriculture, although the amount of water required varies greatly between different agricultural types and climatic regions. Given the scale of agricultural activity (1.54 bn ha of arable and permanent crops alone; FAOSTAT 2006), this means that agricultural activity dominates the use of freshwater and accounts for some 70% of withdrawals from water resources globally (FAO 2002; WRI 2005). Such large abstraction of water has led to large reductions in river flow, indeed, to long and repeated periods of zero flow for several major river systems (e.g. China's Yellow River; FAO 2003b; Ma et al. 2003), with consequent huge changes in estuarine and coastal ecosystems. In addition, abstraction has overexploited many major regional aquifers. As most of the water withdrawn by agriculture is lost in evaporation and transpiration, in marked contrast to domestic and industrial withdrawals, agriculture accounts for 80–90% of freshwater used by humans (FAO 2002; Shiklomanov & Rodda 2003; Rijsberman 2004). Clearly, making agriculture sustainable requires a major reduction in water use in many regions.
Shortage of water leads to drought with obvious agricultural and societal impacts. Periods of repeated droughts have a particularly severe effect on societies, and there are many historical examples (Pereira et al. 2007). For example, the fall of the Roman Empire has been attributed to a drying climate, both in Europe and in the Central Asian steppes, which may have stimulated the movement of the barbarian tribes (Lamb 1995). The decline of the Maya civilization has been attributed to droughts of increasing frequency and severity between AD 750 and 1050 (e.g. Haug et al. 2003; Hodell et al. 2005). More recently, the difficulties of British settlement in North America in the beginning of the seventeenth century may have resulted from coincidental extreme droughts (Stahle et al. 1998; Blanton 2000). In current times, we are all too familiar with droughts and consequent famine in Africa, for example. Furthermore, there is widespread agreement that increasing anthropogenic climate change will exacerbate the present shortages of water, and is likely to increase drought (IPCC 2007).
Farmers in many countries are now faced with legislative restrictions on use of water (e.g. the 2003 Water Act in the UK), which are being imposed to try to secure safe and adequate water supplies for domestic water users. Under some circumstances, restrictions on use can be substantial. For example, in China, government has set a target of a reduction of 20% in water use in agriculture by the year 2020. If food security for the region is not to be threatened, this must be achieved without a loss in production. Overuse of water in some regions of China is having serious environmental consequences. For example, the Shiyang River Basin in arid, northwest China is the largest inland river basin in the region with the largest human population density and the highest exploitation of water resources. Over the last 50 or more years, as a result of an explosion of agriculture in the region, severe water shortage has developed which not only constrains social and economic developments but is also leading to some of the worst ecological and environmental deterioration in China. Kang et al. (personal communication, 2006) have highlighted the changes in the water systems in the Minqin oasis at the end of the river basin caused by both the changing climate and intensive farming activity. With an increasing population (159% rise in 50 years), the amount of cultivated land in the basin has been expanded by more than 50%. Many reservoirs have been built by damming rivers and large-scale irrigation has been introduced. Water use due to human activities has exceeded the capacity of the water resources in the basin, which has led to a dramatic and rapid fall in the water table in the Minqin oasis (by as much as 14 m). The oasis is shrinking in area, natural vegetation that relies on underground water is disappearing and desertification is accelerating, leading to more dust storms. ‘Super’ dust storms occurred only five times in the 1950s, but in 2001 they occurred six times in the first half of the year. Now the Minqin area is one of the origins of major dust storms for China and the rest of the world. With the rapid deterioration of its water resources, increasing droughts and dust storms and with large areas of abandoned land, more and more farmers are eventually migrating out as ‘ecological refugees’. So, an ancient oasis that can be traced back 2000 years is disappearing, with local and global effects on the society and climate.
Thus how to reduce agricultural water use and make water resources more sustainable is an increasingly urgent question. It is a question that requires combined agronomic, physiological, biotechnological/genetic and engineering approaches which may be collectively described as ‘water saving agriculture’. We need to use appropriate agronomy to grow crops best suited to the environment, with least use of water, whether in irrigated or rainfed production. We need to develop crops that require less water to produce sufficient yield, through understanding the physiological mechanisms that determine growth and water loss, and plant response to reduced water availability. We need to consider fully the returns on irrigation, as part of making real improvements in sustainable water management (Parry et al. 2005). Ideally, as many commentators have pointed out, we need to reduce the use of irrigation in hot, dry environments, as it is in these environments that irrigation is most wasteful, in that it produces least yield per unit of water, owing to the high evaporation rates. Similarly, some techniques are less efficient than others. For example, in a recent survey in England, 72% of farmers irrigating field crops were using rain guns (Defra 2002), despite their inefficiency in delivering water to the crops. Ultimately, the goal must be to reduce the amount of water used per unit yield achieved. As Kofi Annan, UN Secretary General, declared in 2000 ‘we need a Blue Revolution in agriculture that focuses on increasing productivity per unit of water—‘more crop per drop’ (UNIS 2000). Perhaps it would be most beneficial if the maximum nutrition per unit of water was achieved, but economics dictate that crops grown are those that produce the maximum economic yield, so that the exact definition of sustainability becomes important. The amount of yield produced per unit of water used is referred to as ‘water use efficiency’, or ‘water productivity’, and a discussion of how this might be improved is the focus of this paper.
2. Water use in agriculture
Some figures at different scales illustrate the amount of water used in agriculture. Globally, some 2.7×103 km3 of water were used in agriculture in 2000 (WRI 2005). While significant amounts of water are used in animal production and food processing, water availability limits animal production mostly through the production of fodder crops and forage, and it is therefore crop production that we consider in this paper. Figures that are widely quoted (e.g. FAO 2002; Pimental et al. 2004) are that production of 1 kg of wheat requires 1 m3 of water and 1 kg of rice requires at least 1.2 m3 of water. However, these are low requirement figures compared to some, such as the 0.7–2 m3 required for grain yield in temperate zones and 3–5 m3 in arid regions given by Gregory (2004). Most of the water used is for the irrigation of some 276 Mha of crops (FAOSTAT 2006). The area irrigated is still increasing globally, although the rate of increase in area irrigated appears to be reducing, averaging 1.1 Mha yr−1 between 1999 and 2003, compared with 4.2 Mha yr−1 between 1990 and 1994 (data from FAOSTAT 2006). In the 15 countries of the EU in 2003, an area equivalent to 15.5% of the arable and permanent crop area was irrigated and irrigation comprised over half of the total water consumption (EEA 2003). Even within the humid, temperate climate of England, 147 kha of outdoor crops were irrigated in 2001 (approx. 3% of the cropped area) using 131×106 m3 of water (Defra 2002).
The key point about this irrigation demand is that although irrigated areas accounted for approximately 18% of the world's cropped land in 2003 (FAOSTAT 2006), they produced approximately 40–45% of the global food (Döll & Siebert 2002). Therefore, reduction in irrigated areas or productivity could have very serious impacts on global food supply. Conversely, any reduction in irrigation water used would have major benefits to freshwater resources. However, the other 82% of the global cropland which is rainfed and the 3.4 bn ha of rainfed permanent pasture is just as important as it is obviously a major part of the local catchment and regional hydrological balances, and a major contributor to local and regional food security. Even in the Mediterranean region, with large irrigated areas, 60% of the cereal production comes from rainfed cropping (Parry et al. 2005).
Clearly, at a regional scale, the major problems in water supply are in regions with low rainfall, yet high evaporative demand, and those with expanding populations, such as North Africa, Southern Africa and the Near East, where water availability is less than 2000 m3 per person per year (Wallace 2000; FAO 2003a; Gregory 2004). Population growth, urban expansion and economic development are increasing other demands for water. Therefore, there is increased competition for water between agriculture and other users, and in many regions reductions in water quality as well as quantity. Salinization and other contamination of surface and groundwater are major problems (Wild 2003; Khan et al. 2006), although outside the scope of this paper (see other papers in this issue). Agricultural water use is also a major international political issue, for example, in regions where irrigation depends on the river inflow from neighbouring countries, such as in Egypt, Iraq or Syria (Araus 2004).
In addition, most plausible predictions for the climate over the next 50 years indicate increases of annual mean temperature of 1–2.5°C (IPCC 2007), but possibly considerably higher. This will probably increase evapotranspiration rates, particularly in warmer regions, and T is transpiration rate. Precipitation amounts are also changing, and in many temperate environments there are already changes to the seasonal distribution, with further predictions of milder, wetter winters and warmer, drier summers for much of Europe, for example (IPCC 2007). Other serious changes predicted are less snowfall for key melt-water rivers that provide water for major crop growing regions, such as in India and Pakistan, and for more storms and localized flooding. In addition, there are predictions that sea-level increase and greater abstraction of water will increase salinization of coastal aquifers (e.g. Danielopol et al. 2003). All of these changes add substantially to pressures on water resources and increase the urgency of reducing water use in crop production, thus improving yields and yield stability at the same time.
3. Water use in plant growth and ‘water use efficiency’
Water is essential for crop production because plants require water for growth and tissue expansion. However, well over 90% of the water required by terrestrial plants is not ‘used’ in any biochemical way but lost through transpiration. Much that has been written about water and crop growth tries to make a distinction between ‘water-limited’ and ‘wet’ environments. However, this is unhelpful from a physical and physiological viewpoint, as all terrestrial environments are water limited for plant growth to some extent, because terrestrial plants must have a cuticle and stomata to control water loss and prevent tissue dehydration. This dehydration control therefore usually limits the uptake of CO2 and thus growth. What is usually meant is that water ‘limits’ crop productivity to below the maximum or potential production when water supply is less than the demand for water set by atmospheric conditions. This atmospheric demand is usually quantified by estimating the ‘potential evapotranspiration’ rate (ETp), which was a concept introduced by Howard Penman in 1948, although other, usually less successful, methods of estimation are sometimes used. Penman (1948) defined potential evapotranspiration as the evaporation rate of a short, actively growing grass surface completely covering the ground. Therefore, if the crop actual evaporation is less than the potential (ETa<ETp), this indicates that either crop ground cover is not complete or stomata are restricting transpiration. Thus CO2 uptake per unit ground area is likely to be restricted and growth will be ‘water limited’. However, as we discuss below, CO2 uptake may not necessarily be restricted by reductions in stomatal aperture.
The question of how much water plants require to grow is an old one. Indeed, Stanhill in a comprehensive review in 1986 traced the earliest study to John Woodward (1699) in the Philosophical Transactions of the Royal Society (Stanhill 1986). Many such reviews (most recently chapters by Passioura, Gregory & Jones in Bacon (2004)) have summarized the key information on water use and its ‘efficiency’ of use in plant production. Although Monteith (1984, 1993) and Stanhill (1986) have pointed out that the usual terms ‘water use’ and ‘water use efficiency’ are completely erroneous, unfortunately they remain in common use. Monteith's (1993) suggestions of the much more correct terms ‘water loss’ and ‘biomass water ratio’ have still not had a wide take-up. In this paper, we will use BWR to indicate the biomass water ratio (=M/T or sometimes M/ETa, where M is dry biomass but the effect will depend on any changes in humidity, wind speed, cloudiness and hence solar radiation) or biomass water use efficiency. Whatever the problems in terminology, and the many different units used, there are several key points to note. Firstly, the term water use efficiency can be used at several very spatially different scales: at the catchment, the farm, the field, the plant and down to the leaf. It can be applied to the water lost in producing just the economic yield, or the biological yield which can be all the above-ground biomass, or (more rarely) the total biomass. Furthermore, it can include or exclude the evaporation from the soil and plant surfaces directly (the latter usage being termed ‘transpiration efficiency’, TE). It can also be applied at very different time scales. At the crop or field scale, it can be used for time spans of days or months, or the entire crop growing season, or per year. At the leaf or plant scale, it can be applied when considering the diffusive fluxes of CO2 and water vapour in and out of leaves, where it is the ratio between net CO2 assimilation rate (A) and transpiration rate (T), first derived by Penman & Schofield (1951), and usually obtained over a period of seconds or minutes. This is the so-called ‘instantaneous water use efficiency’, but termed ‘assimilation transpiration ratio’ (ATR) in this paper to align with the use of BWR. It is given by
| (3.1) |
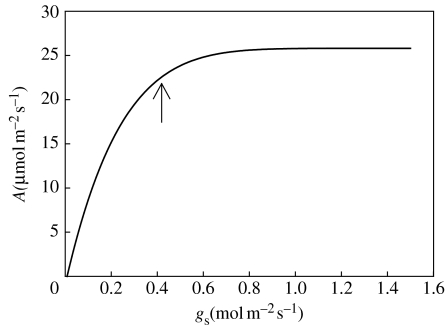
where ΔCO2 and Δe are the differences in partial pressure for CO2 (Ca−Ci) and water vapour (ei−ea), respectively, between inside (in the substomatal cavity) and outside the leaf; rs and rb are the stomatal and boundary layer resistances for diffusion through stomata and boundary layer for water vapour; and rs′ and rb′ are the same resistances for CO2. Owing to the low partial pressure of CO2 in the air (only 37 Pa) compared with water vapour (0.5–4 kPa, depending on air temperature and humidity), the partial pressure difference is approximately 100 times larger for water vapour than for CO2. When expressed as molar fluxes the ratio of CO2 taken up to water lost (ATR) in illuminated leaves is typically in the range 1–10 mmol CO2 per mol H2O. As this varies with air humidity and leaf temperature (i.e. the Δe term), several authors have used the ratio of assimilation rate to stomatal conductance (A/gs, where gs is 1/rs) because T≈Δe×gs. While it is useful to remove some of the effect of environment, confusingly this is sometimes referred to as ‘intrinsic water use efficiency’; it is usually in the range 0.2–2 mmol CO2 per mol H2O. While the A/gs ratio is conservative, owing to the close correlations of A and gs that normally occur in changing conditions, it is not a constant, as gs may vary with Δe or other factors in order to preserve leaf water status. Indeed, this is the idea on which the ideas of deficit irrigation and partial root drying (PRD) discussed later are based. At a given ambient CO2 concentration (Ca), the ratio A/gs is linearly related to the intercellular CO2 partial pressure (Ci) which reflects the balance between assimilation rate and stomatal aperture; higher Ci therefore indicates lower ATR. Thus plants with the C4 photosynthetic pathway with their typically higher assimilation rates and lower stomatal conductance usually have a lower Ci and higher ATR than plants with the C3 photosynthetic pathway. The typical relationship between A and gs is shown in figure 1; the slope of this relationship is the intrinsic water use efficiency. Achieving high yield requires (inter alia) high A, but any increases in conductance reduce efficiency, particularly at high gs. Conversely, reduction of gs when it is high usually provides gains in ATR. Note that selection for high A/gs per se would not be useful, without ensuring a reasonable value of A to produce the biomass required.
Figure 1.
Generalized relationship between net CO2 assimilation rate (A) and stomatal conductance (gs). In many situations, the responses of A and gs to environmental and internal conditions are such that the rates are often near the ‘break point’ on the relationship (indicated by arrow). Reductions in gs below this point will reduce A by restricting CO2 diffusion, and increases above it will bring only small increases in A, while causing large increases in transpiration.
The relationship between BWR and ATR has been discussed by several authors (e.g. Bierhuizen & Slatyer 1965; Tanner & Sinclair 1983; Farquhar et al. 1989; Morison 1993; Jones 2004a). It is important to realize that the temporal and spatial integration of the short-term, leaf-scale ATR up to the seasonal, crop-scale BWR is not simple, as it requires estimates of crop respiration losses and water loss from soil and canopy surfaces (referred to as interception loss). Clearly, the proportion of water lost from soil or surface evaporation will vary with the time courses of leaf area development and climate conditions, soil conditions and the frequency of rainfall events. Translating the seasonal, canopy-scale BWR into a yield figure additionally requires information on the partitioning of fixed carbohydrate into the yield component. However, in practice, the analysis of Tanner (1981) and Tanner & Sinclair (1983) showed that if transpiration alone is considered, then the relationship in equation (3.1) could be simplified at the canopy and seasonal scale as BWR=kc/Δe, with kc a crop-specific parameter. Because A and gs are strongly correlated, it means that kc is actually quite conservative within the two photosynthetic physiologies of C3 and C4 species. This simplified relationship has been the basis of much analysis of crop TE and is used in some crop growth simulation models. While many measurements do appear to show that kc is quite conservative within a crop, it is clear that because gs usually declines with increasing Δe (e.g. Morison & Gifford 1983; Zhang & Nobel 1996; Xu & Hsiao 2004), kc increases. As a recent analysis of barley crop TE shows (Kemanian et al. 2005), this response acts to slightly reduce the advantage of more humid environments in improving the BWR.
In 1977, John Passioura proposed a simple model as a framework for examining the determinants of crop yield, particularly in water-limited crop production,
| (3.2) |
where Y is the yield; W is water available; and HI is the harvest index (Passioura 1977). The model has been a most useful framework, as it focused attention on three areas: how to increase the water available; how to increase the proportion of crop biomass, i.e. yield (HI); and how to improve the ratio of crop biomass produced to water lost. For the physiologist, W is usually taken to be the water available for transpiration; thus BWR becomes synonymous with TE. While water availability is thus partly determined by irrigation and precipitation, clearly all three terms in equation (3.2) have strong physiological determinants and it is these that we concentrate on in this paper. However, Gregory et al. (1997) have emphasized that for the agronomist W in equation (3.2) is not necessarily all used in productive transpiration of the crop (Tc), but that soil evaporation (Es), drainage from the root zone (D) and run-off (R) are important components. Furthermore, another agronomic aspect is that some water may be lost in weed transpiration (Tw; Turner 2004b). Thus
| (3.3) |
Therefore, increasing W does not increase yield unless Tc is increased more than (Es+Tw+R+D). Both Es and D are partly physiological aspects too. Firstly, the degree of ground cover is a key determinant of the rate of soil evaporation. There is considerable evidence that establishing high ground cover, for example by using genotypes with rapid leaf growth, using denser planting or fertilizers, can increase the proportion of water lost in transpiration (Gregory 2004). Secondly, loss of water through drainage below the root zone depends on root growth as well as on how wet the profile is; thus deeper rooting can recover more water from the profile. There is much interest in the use of deficit irrigation to enhance the growth of roots and some suggestion that many of the benefits of partial root zone drying (PRD; see below) can be attributed to enhanced water uptake from deeper in the soil profile, thereby enhancing yield (Y) through an enhancement of the water available (W) in equation (3.2) (Dry et al. 2000; Kang et al. 2002a,b; Mingo et al. 2004).
In irrigated agriculture, there are further losses of water to be considered through evaporation and leakage from reservoirs, and losses in water movement to the field. Globally, storage and transfer losses are estimated at approximately 30% (Wallace & Gregory 2002). Run-off and drainage losses may represent another 44%, which means that after accounting for soil evaporation losses probably only some 13–18% of water available for irrigation ends up in transpiration (Wallace & Gregory 2002; Gregory 2004). However, a substantial fraction of the ‘lost’ water may be available for other processes and users within the catchment, so is not a complete loss.
4. New techniques for assessing plant transpiration efficiency
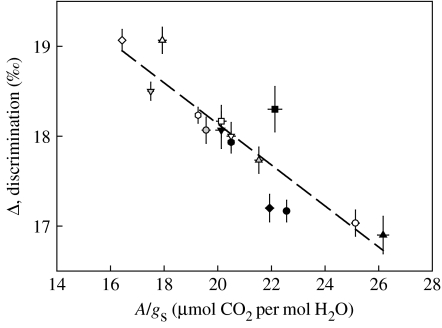
In the 1980s, Farquhar et al. (1982) developed a new method for assessing TE using the ratio of the abundance of the natural isotopes of C, 13C and 12C. During diffusion and biochemical fixation of CO2, the heavier 13C is discriminated against, so that inside leaves and in leaf tissue the ratio 13C/12C is reduced compared with the normal abundance in the atmosphere. The ratio (or δ13C, the discrimination against 13C) depends on the balance between diffusion into the leaf and demand by A, so δ13C gives a measure of TE, either at the moment (for gas measurements) or integrated over the period of fixation of leaf organic material. It is therefore related to the ratio of A/gs and to ATR. Drier conditions usually result in less discrimination (less negative δ13C values). The approach has now been used to investigate TE in many crop and tree species, ranging from cereals, through cotton and soya bean, to grapes, coffee, eucalypts and pines. A further recent example is shown in figure 2, where the discrimination in leaf material of 15 coconut genotypes was found to be very closely related to the A/gs measured by gas exchange (Nainanayake 2004). In such C3 plants where there is a strong relationship between Ci and δ13C, the technique has led directly to the selection of improved crop varieties, most notably in wheat (Condon et al. 2004; see below). However, it is not suitable for screening C4 plants because δ13C also depends on the ‘leakiness’ of the bundle sheath for CO2 (Ghannoum et al. 2002).
Figure 2.
Relationship between the mean 13C discrimination (expressed as Δ, discrimination compared to air, ‰) and the mean ratio of net CO2 assimilation rate (A) to stomatal conductance (gs) for leaves of 15 varieties and accessions of mature coconut palms growing at Pottukulama Research Station, Sri Lanka in 2003. Bars indicate s.e.m., n=3. Line is linear regression, y=0.228x+22.7, R2=0.813, p<0.001. Results from Nainanayake (2004).
Furthermore, the δ13C method cannot distinguish between changes of net assimilation rate or of stomatal conductance. More recently, the isotopic ratio of 18O to 16O has been used to indicate the transpiration conditions during C fixation (e.g. Farquhar et al. 1994; Yakir & Israeli 1995; Barbour et al. 2000). In a similar way to the C isotope discrimination, the ratio of the oxygen isotopes in leaf water or in leaf organic matter depends on diffusion and transpiration rates. The heavier isotope becomes enriched at the sites of evaporation because 16O-containing water is preferentially evaporated, resulting in a positive discrimination, but the relationship with conductance is altered by leaf temperature and air humidity which affects the gradient in vapour pressure (Δe; Barbour & Farquhar 2000; Sheshshayee et al. 2005). Therefore, when δ18O and δ13C data are combined, it can provide information on the relative contribution of stomatal conductance or CO2 assimilation rate in determining TE (e.g. Barbour et al. 2000; Keitel et al. 2003).
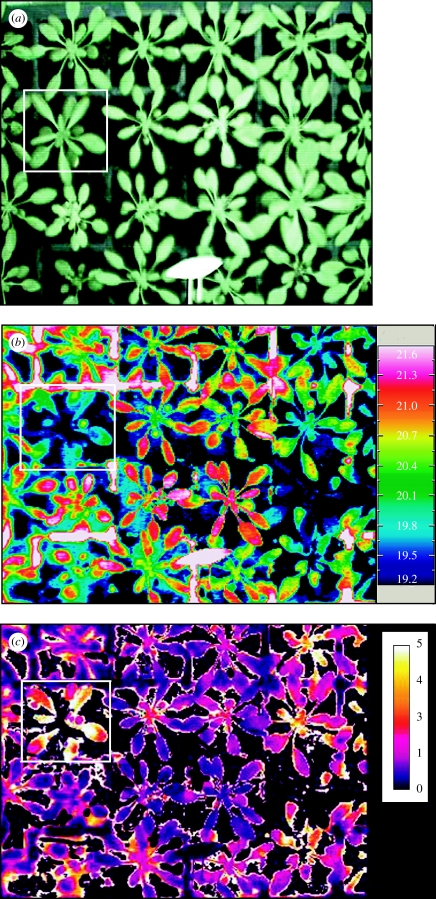
Much of the routine use of the above isotope discrimination approaches has been based on leaf or seed material. This material is dominated by the cellulose fraction and so integrates the conditions during and after organ growth, and can be rather insensitive to changes in photosynthetic rate caused, for example, by drought stress after expansion. It is possible to refine the technique to look at the dynamics of photosynthetic fixation as stresses occur, by examining just the soluble and starch carbohydrate components (e.g. Ghashghaie et al. 2001), but this is not yet routine. Clearly, techniques are required for rapidly assessing both the photosynthetic rate and stomatal conductance of leaves for the screening of genotypes and their response to drought or other environmental stresses. Chlorophyll fluorescence emissions from intact plants are easily and rapidly measured, and have been shown to be directly related to photosynthetic activity (Baker & Rosenqvist 2004). The fluorescence parameter Fq′/Fm′, which estimates the quantum yield of photosystem II photochemistry, has been shown to be directly related to the rates of linear electron transport and CO2 assimilation in leaves (Baker & Oxborough 2004). Consequently, this parameter potentially provides an effective tool for rapidly screening for differences in photosynthesis and thus A, or for differences in the response to stresses like drought. However, it is well established that photosynthetic performance across the area of individual leaves can be extremely heterogeneous, especially for stressed plants. Any attempts to screen for effects on leaf photosynthesis using fluorescence should ideally sample the whole of the leaf. Conventional commercial fluorimeters generally only measure fluorescence from a small area of leaf and are unsuitable for such screening. Imaging instruments using charge coupled device (CCD) cameras overcome this problem and allow an image of fluorescence parameters to be produced from whole leaves (Nedbal & Whitmarsh 2004; Oxborough 2004). Figure 3 shows an example of the heterogeneity in leaf photosynthetic function caused by drought.
Figure 3.
Images of the chlorophyll fluorescence parameter Fq′/Fm′ for Arabidopsis thaliana wild-type plants grown (a) well watered (control) and (b) in a slowly developing drought (droughted). Note the smaller droughted plant and the difference in photosynthetic efficiency between young and older leaves. Images courtesy of T. Lawson, University of Essex.
Another imaging technique, IR thermography (IRT), has recently been used to study stomatal responses to drought stress (Jones 2004c). IRT can be used in a simple way to look for differences in leaf temperature, and thereby infer differences in transpiration and stomatal behaviour. It has been used this way to screen for and identify mutants with altered stomatal function (e.g. the OST mutant; Merlot et al. 2002). Figure 4a shows Arabidopsis thaliana (Arabidopsis) plants in a tray, with the thermal image (figure 4b) showing that the leaves of the OST plant (circled) were 1–1.5°C cooler than those of surrounding wild-type plants. In addition, IRT can be used to estimate gs for whole leaves and canopies if suitable reference surfaces are used (Jones 1999; Jones et al. 2002; Leinonen et al. 2006). In particular, Jones et al. (2002) have shown that an index Ig that is proportional to gs can be readily determined from thermal images of leaves and is a suitable parameter for rapidly screening for genotypic variation in gs. Figure 4c illustrates this approach with the Arabidopsis example. Horie et al. (2006) have recently shown the strong potential of IRT for selecting for rice genotypes with higher conductance associated with higher yields.
Figure 4.
(a) Visible image of Arabidopsis thaliana wild-type and OST mutants growing in a controlled environment cabinet. The ‘dry’ reference surface (white area) is visible. (b) Thermal image of same plants. OST mutant plant shown within white box is cooler (mean leaf temperature 19.9°C) compared with wild-type plants (mean leaf temperature range 20.3–21.0°C). (c) Calculated index of conductance (Ig) for the image in (b), calculated from temperature of leaves and dry and wet reference surfaces, using the method of Jones (1999).
By coupling fluorescence and thermal imaging techniques it is now possible to rapidly evaluate both the photosynthetic performance and stomatal conductance of leaves (Chaerle et al. 2005, 2007). This has been done at the small scale as a tool to examine stomatal control of photosynthesis in leaves (e.g. Omasa & Takayama 2003; Messinger et al. 2006). Clearly, as the ratio of the two parameters (Fq′/Fm′)/Ig is directly proportional to A/gs, combining such images offers exciting possibilities to screen plant material for photosynthesis, water loss and ATR non-destructively and repeatedly.
5. Drought resistance and water productivity
One approach to understand plant water productivity is to study plants from dry environments which have mechanisms that confer drought resistance, traditionally divided into dehydration avoidance and dehydration tolerance mechanisms (Levitt 1972). However, it has been argued that such comparative studies are rarely useful as drought resistance is usually linked to low productivity, and is thus of limited use in agricultural production (Sinclair & Purcell 2005). In addition, severe drought is usually prolonged, so survival will not usually lead to subsequent productivity, as the water is simply not there (Passioura 2004). Also, Passioura (2002) has stated that ‘severe water deficits are rare in viable agriculture, and asking how crops respond to or survive them is unlikely to have much general impact’. This is true for commercial agriculture, but is perhaps not true of much of ‘subsistence’ farming. In these situations, it is possible that physiological mechanisms that allow maintenance of a high plant water status such as osmotic adjustment, reduced water loss through cuticle, avoidance of xylem cavitation and altered root-to-shoot ratio could be important in providing some yield in ‘resource-poor’ cropping systems (Blum 2005). Several recent reviews have pointed out that the main improvements in drought resistance of crops have come from increasing dehydration avoidance, particularly by increasing W, through changes such as earlier development, smaller leaves, and deeper roots, and not by improving dehydration tolerance. One important example of improved dehydration tolerance is in improving the response of arable crops to terminal drought by improving the mobilization of stem carbohydrate reserves for grain filling (Blum 2005). However, in many regions where drought stress occurs, rainfall is variable, and whether increased drought resistance will inevitably result in plants that will not be able to take advantage of any higher rainfall needs to be further investigated. Blum (2005) points out that there is usually a negative relation between yield potential and yield drought resistance, and argues that genotypic variations in BWR are normally due to variations in water use, and improvements are usually achieved by traits and environmental responses that reduce the yield potential. Blum therefore cautions that selection for BWR directly in order to improve drought resistance is not necessarily the best way forward. Furthermore, Parry et al. (2005) have pointed out that most biotechnological approaches using transgenics to improve performance under drought have targeted survival and reductions in water use, not improved photosynthesis under a given supply of water. Passioura (2004) has noted that improving water productivity is now seen as the way forward, rather than the search for drought resistance.
6. Improving crop water productivity: some success stories
Conventional crop breeding continues to release varieties that have improved yield in water-scarce environments, particularly in cereals. Several analyses of Mediterranean cropping systems have pointed out that about half of this is due to improvements in the crop, and half due to improved agronomy and management, and the two have combined and facilitated each other (Passioura 2002; Richards 2004; Turner 2004b; Slafer et al. 2005). Despite the generally well-developed understanding of why yield is limited under drought, there has been only limited progress through directly selecting physiological traits, apart from flowering time, and plant height (Richards 2004). Richards has noted that part of the reason for limited progress is that water-scarce environments are highly variable in the time at which drought occurs and in the amount of water that crops receive each season. Some cropping areas can experience regular droughts at one particular time in the growing season, whereas others have droughts at different stages of the crop development. In some areas, yield depends entirely on water stored in the soil profile, in others on current rainfall only, while for many areas plant yield is a result of water available from soil stores, rainfall and irrigation. These variations mean that there are many and varied targets for plant improvement under drought.
Despite these challenges, we are now in a stronger position to exploit our physiological understanding of water-limited yield (see, e.g. Chaves & Oliveira 2004; EPSO 2005). Advances in genetics and the molecular sciences and in technology can now help us exploit novel understanding of plant drought stress responses and this combination can result in a more targeted selection programme (for example, where biotechnology can be used with conventional breeding) to increase BWR in dry environments.
Variation in complex traits, such as those that contribute to improved BWR, are the basis for crop breeding programmes, especially where traits are introgressed from wild relatives into domesticated varieties (e.g. Gur & Zamir 2004). The identification of such variation can lead to the statistical association of the trait with particular polymorphic region(s) of the plant's genome. Such regions are termed quantitative trait loci (QTL; Salvi & Tuberosa 2005; Mitchell-Olds & Schmitt 2006) and are identified by observing the frequency of co-segregation of particular DNA sequence polymorphisms or markers (detected by various means) and the trait of interest (Quarrie 1996). These techniques have been used to isolate the genes responsible for QTL (see below). However, for a breeding programme targeted to a specific trait the main requirement is to obtain only as tightly linked a DNA marker as is possible given the resources of the project. Marker-assisted selection (MAS) improves the efficiency of breeding programmes especially where complex traits are involved (Quarrie 1996; see also other contributions in this issue). For example, it may be easier to follow a DNA marker through a segregating population than to carry out the necessary complex physiological tests on all the material. Pre-selection of progeny for more detailed analysis is possible, again saving time and cost. As a consequence, MAS is well established in many breeding programmes, including those selecting for improved drought resistance (e.g. Schneider et al. 1997; Subudhi et al. 2000; Foolad et al. 2003; Serraj et al. 2005; Jongdee et al. 2006; Steele et al. 2006).
One of the best examples of new approaches is the production of new lines of wheat where TE has been increased. In particular environments in Australia, lines selected for higher TE using the carbon isotope discrimination technique described above have outyielded commonly used commercial varieties (see Richards 2004). The variety Drysdale was released for southern New South Wales in 2002 and Rees for the northern Australian cropping region in 2003. In comparison with high discrimination sister lines, yield trials have shown a yield advantage between 2 and 15% of lines with low carbon isotope discrimination (high TE), but only at yield levels from 5 to 1 t ha−1, i.e. in dryland or drought-limited conditions (Rebetzke et al. 2002). Highest yield advantages were found in the most droughted environments. Trials in southern NSW demonstrated 23% yield increases in Drysdale compared with the current recommended varieties for this region.
Richards (2004) has emphasized that progress in increasing commercial crop yields in water-scarce environments has been made by selecting plants for a variety of characters which can impact on yield by influencing components of the Passioura equation (equation (3.2)). These include:
extended crop duration (in wheat), allowing crops to be grown at different times of year thereby reducing the Es component in equation (3.3) and putting more water through the plant to increase W;
increased axial resistances to water transport in wheat (Richards & Passioura 1989), which means the plant uses water more slowly, ensuring water is available for development during anthesis and subsequent grain filling (increased HI);
reduced anthesis-silking interval in maize (ASI, Edmeades et al. 1999), reducing the chances of development of severe drought stress during vital phases of reproductive development of the crop (thus leading to increased HI). If drought stress does occur at this time, then complete crop failure can result, whatever the water relations and growth of the crop during the vegetative phase;
osmotic adjustment in wheat (Morgan 2000) which also helps sustain shoot water status during key phases of plant development (increased HI), and
selection for ‘stay-green’ traits in sorghum, which has led to lines which retain more green leaves under terminal drought. The delayed leaf senescence allows further uptake of soil water and nitrogen, both of which can enhance yields (and yield per unit of water used) in drought-prone environments as a result of more carbohydrate being available for grain filling (increased HI; Borrell et al. 2000, 2001).
Richards emphasizes that although all these particular traits contribute to increased grain yield in dry environments, the maintenance of favourable plant water relations are not the main features in several of the responses. Passioura (2004) has also highlighted that their common feature is that the traits operate over the life of the crop (or at least during key developmental periods), rather than through short-term, immediate stress-tolerance responses.
7. New genes and insights from Arabidopsis: can a model species make a contribution to breeding for improved water productivity?
Whereas many genetic loci (QTLs) have been identified in crop species that contribute to particular aspects of crop performance, only a few genes responsible for such QTLs have been identified (Salvi & Tuberosa 2005). In contrast, with Arabidopsis and increasingly for other model species, it is technically more feasible for laboratories to isolate genes responsible for QTL (Salvi & Tuberosa 2005). This is an important development and is part of a ‘new synthesis to elucidate mechanisms for the adaptive evolution of complex traits’ (Mitchell-Olds & Schmitt 2006). However, will this emerging combination of disciplines have the potential to provide extra genes and concepts to improve crop water productivity? The answer is a cautious ‘yes’ because new genes may come from exploitation of natural variation in accessions (hereafter called ecotypes) that may display considerable variation in adaptation to adverse environmental factors. Arabidopsis is a species that grows in open and often rocky or disturbed sites (Mitchell-Olds & Schmitt 2006). Within its geographical range, Arabidopsis encounters a wide range of climatic conditions including regions with low rainfall during the growing season. This suggests that this species would be a good source of novel alleles at loci coding for traits that are important for adaptation to water-scarce environments (Alonso-Blanco & Koornneef 2000; Loudet et al. 2005). Furthermore, another reason for optimism is the well-organized, freely available resources for molecular genetic analysis. These include the complete genome sequence data (Arabidopsis Genome Initiative 2000), extensive web-based data for important information on markers, gene annotation, programmes to interrogate large datasets, expression data, passport data and linkage to crop-based genetic resources (e.g. The Arabidopsis Information Resource; www.arabidopsis.org), stock centres (e.g. Nottingham Arabidopsis Stock Centre; http://arabidopsis.info/) that hold large collections of mutants, accessions and recombinant inbred (RI) populations (central to QTL mapping; Alonso-Blanco & Koornneef 2000) and, finally, a large community of researchers that can make rapid progress.
Assuming that a gene responsible for a QTL has been identified, there are three reasons why this may prove useful, which are as follows:
Identification of the gene, especially if this has been achieved using unbiased approaches such as QTL analysis, very often provides a major step forward in understanding the underlying biology that gives rise to a trait and the basis for variation at that locus. For example, the role of ERECTA in regulating TE (Masle et al. 2005, see §7a).
Comparative genomics and bioinformatics would allow a determination of the gene as a candidate for a QTL of the same trait in other crop species thus providing increased possibilities for searching for variation at that locus. For example, genes and QTL involved in low-temperature tolerance in Arabidopsis and the Triticeae (Skinner et al. 2006).
The gene from Arabidopsis or its crop homologue could be transformed into crop species in an attempt to reproduce the trait.
However, some commentators are rightly sceptical of the contribution that a genetic engineering or biotechnological approach to manipulating water productivity traits can make (e.g. Passioura 2004; Parry et al. 2005). It is argued that molecular biologists have studied genes associated with immediate response or resistance to severe dehydration stress (Bray 2002), a trait of little relevance to water productivity (Passioura 2004; Sinclair & Purcell 2005), and have analysed transgenic plants with poorly designed experiments or interpreted data in a naive manner (Parry et al. 2005). A secondary reason is suggested that manipulation of a single or few genes is unlikely to have any major impact on this trait (Passioura 2004; Parry et al. 2005). In our view, the main reason for these failures has been a lack of communication and understanding between researchers in appropriate disciplines at the planning stages of a project. Thus, physiological and agronomic expertise was not brought to bear on the design of experiments that would allow the ‘right’ genes to be isolated. Recent published work suggests this situation is changing, as will be demonstrated in §7a–c. While current scepticism is justified owing to the focus of the molecular biology community on severe dehydration stress (Passioura 2004; Sinclair & Purcell 2005), there is no a priori reason why the right gene transferred from Arabidopsis cannot function and confer complex traits on crop species. While convincing examples are lacking for improvement of water productivity, experience from other areas of research, such as with Arabidopsis genes that regulate flowering and stature (Petty et al. 2003; Tadege et al. 2003; Ellul et al. 2004), suggest that a useful contribution will be made. Where progress is made and a promising gene is isolated and transferred to a crop species, the subsequent involvement of physiologists, breeders and agronomists will be crucial in realizing its potential.
In summary, provided that the appropriate disciplines collaborate effectively in planning a project and then subsequently exploiting its outputs, then in our view, current scepticism should not lead to pessimism about the future. The correct experiments will lead to the right genes (Bray 2002). Furthermore, it is only in the last decade or so that progress on manipulating regulatory genes, such as those coding for transcription factors, has been made, or where genes coding for mutant alleles or QTL can be feasibly isolated or manipulated. Thus, it is only recently that there has been any realistic prospect of molecular genetics providing any traits that might impact on water productivity, notwithstanding the hundreds of patents claiming genes that promote drought tolerance, as Passioura (2004) has pointed out. Examples in three areas that fit within the framework provided by Passioura (equation (3.2)) include the following.
(a) Identification and analysis of the effects of genes conferring high TE
In breeding for improved TE, the exploitation of the δ13C assay has proved particularly effective (see above). As a consequence, this parameter has been used to identify TE QTL in several crop species and in Arabidopsis (Martin et al. 1989; Thumma et al. 2001; Hall et al. 2005; Juenger et al. 2005 and references therein). For example, five QTLs for δ13C that show considerable interaction with each other (epistasis) have been mapped in Arabidopsis and two of them co-locate with flowering-time QTL and may indicate a functional or pleiotropic relationship between these traits (Juenger et al. 2005). This observation of a link between a TE parameter and flowering time has been made also in the ecotype Shahdara, which originates from a dry, high-altitude environment in Central Asia (Loudet et al. 2005). Using QTL mapping, a causal link was made between pre-flowering plant water content and the possession of a defective FLOWERING LOCUS C (FLC) gene, which consequently promotes precocious flowering in Shahdara (Loudet et al. 2003). The connection was made to a study which suggested that because FLC can control the circadian rhythm of leaf movement it may also impact on the regulation of stomatal transpiration (Loudet et al. 2003). Although this hypothesis remains to be tested, recent support comes from the identification of the FCA protein as an abscisic acid (ABA) receptor (Razem et al. 2006). FCA is a negative regulator of FLC expression by preventing the accumulation of its mRNA (Quesada et al. 2003) and may provide the beginnings of a mechanistic explanation of the link between flowering time and TE.
Recent analysis of genetic variation in leaf δ13C in Arabidopsis (Masle et al. 2005) has identified ERECTA, a gene that apparently regulates TE, through both reducing stomatal conductance and controlling leaf photosynthetic capacity. ERECTA affected stomatal and mesophyll cell density through epidermal cell expansion in the plants produced by Masle et al. (2005). Importantly, ERECTA and other gene family members are known to be involved in the control of stomatal differentiation (Shpak et al. 2005). We also have some understanding of the control of stomatal patterning in response to a range of environmental perturbations (e.g. Gray et al. 2000). Thus, the ERECTA work provides a clear link between stomatal and mesophyll patterning, TE and yield, and illustrates the adaptive value of traits which are present throughout the life of the plant.
The most research effort for single-gene manipulations aimed at improving dehydration tolerance has centred on increasing or altering the pattern of expression of the so-called DREB/CBF family of transcription factors isolated from Arabidopsis plants subjected to severe dehydration stress or during acclimation to low temperatures (reviewed in Nakashima & Yamaguch-Shinozaki (2006)). The involvement of these genes in immediate ABA-regulated (e.g. ABF3) as well as ABA-independent (e.g DREB1A/CBF3) signalling pathways suggest that these genes will affect TE, although no experiments measuring this have been reported.
Such transcription factors control the expression of hundreds of other genes and define a ‘regulon’, i.e. a cohort of genes regulated by each transcription factor (Nakashima & Yamaguch-Shinozaki 2006). Overexpression of these transcription factors can lead to improved dehydration tolerance sometimes without apparent deleterious growth affects on the plant. For example, transgenic rice overexpressing CBF3/DREB1A or ABF3 showed no deleterious growth effects and enhanced dehydration stress tolerance (Oh et al. 2005). However, adverse effects are reported for constitutive expression of these Arabidopsis transcription factors in other species, mainly through stunted growth (Kasuga et al. 1999; Kang et al. 2002a,b; Nakashima & Yamaguch-Shinozaki 2006). Negative effects on plant development (at least under glasshouse conditions) have been avoided and dehydration tolerance greatly improved in transgenic tobacco and wheat by placing DREB1A expression under the control of a dehydration stress-inducible promoter (Kasuga et al. 1999; Pellegrineschi et al. 2004). As noted above, it has been argued that such manipulations are of little value in improving crop water productivity (Passioura 2004). Nevertheless, this work illustrates that expression of a single controlling gene can have profound effects on whole plant functioning, contrary to the view of Sinclair & Purcell (2005). While the examples provided here are negative, there is no reason why other regulatory genes, identified in experiments designed to analyse water productivity, cannot achieve similar but positive effects.
(b) Increasing the amount of water transpired
There are a number of examples which illustrate how the manipulation of stomatal characteristics can lead to improved plant performance under drought and associated high-temperature conditions. Zeiger and colleagues (Lu & Zeiger 1994; Radin et al. 1994; Lu et al. 1998) found that high yielding lines of Pima cotton exhibited higher stomatal conductance. This resulted in cooler leaves and canopy temperatures in these lines. In the hot environment of the southern USA, the cooler temperatures at critical flowering and fruiting stages when temperatures were supra-optimal resulted in improved yields.
One other way of increasing the amount of water transpired by the crop is to increase root growth and the plant's ability to forage for water under drought. An environmental genomics project focused on root growth responses to reduced water availability (Sharp et al. 2004) has generated considerable understanding of the network of mechanisms involved in the maintenance of growth of primary roots of maize under water deficits. The work has shown that several processes interact, including the adjustment of growth zone dimensions, turgor maintenance by osmotic adjustment and enhanced cell wall loosening. The role of the hormones ABA and ethylene in regulating root and shoot growth under water deficits is also important. Recent work shows the potential for restricting ethylene production and increasing water transpired with a positive effect on biomass production in tomatoes (Sobeih et al. 2004). The work on maize roots is now tackling transcript profiling in the growing root and holds promise for improving drought resistance through genetic and metabolic engineering (Sharp et al. 2004).
Variation in root system architecture has been exploited to map QTL controlling root growth and development in Arabidopsis and crop plants (Loudet et al. 2005; Fitz Gerald et al. 2006 and references therein). Analysis of root architecture under normal and mild osmotic stress conditions has resulted in the identification of several QTLs that are involved in both primary and lateral root growth (Loudet et al. 2005; Fitz Gerald et al. 2006). For example, two robust QTLs (termed EDG1 and EDG2) were identified which influence lateral root development under mild osmotic stress conditions (Fitz Gerald et al. 2006). Further, the introgression of both these QTLs into near isogenic lines (NILs) was achieved (Fitz Gerald et al. 2006), which is a major step towards the identification of genes subtending QTL (Salvi & Tuberosa 2005). Similar studies identified 13 QTLs for different aspects of root architecture and growth that segregated in an Arabidopsis RI population, of which two co-mapped to QTL for plant water content (Loudet et al. 2005). The prospects for identifying genes responsible for QTL controlling root growth and development were confirmed with the positional cloning of the BREVIS RADIX (BRX) gene which, as a QTL, accounts for 80% of the natural variation in cell elongation in the growth zone of the root tip in an RI population specially constructed for this study (Mouchel et al. 2004). BRX is one member of a gene family that codes for a novel class of transcription factor which was proven to exert its effect by using transformation to alter the trait in different ecotypes (Mouchel et al. 2004). Such reports substantiate the contention made above that these types of regulatory genes are capable of singly exerting extensive effects on whole organ phenotypes.
Osmotic adjustment as a trait has received attention for QTL mapping and its manipulation through genetic engineering of osmolytes. Although the values of such traits for improving water productivity have been questioned in well-watered conditions (Zhang et al. 1999; Serraj & Sinclair 2002), they can be important in maintaining yield in droughted conditions (Saranga et al. 2001; Serraj & Sinclair 2002; Blum 2005, see above). While increasing the levels of osmoprotectants such as glycinebetaine has been achieved in transgenic plants of various species, the effects on dehydration tolerance have been variable (Huang et al. 2000; Sakamoto & Murata 2002) and as a consequence, recent studies have focused more on their role in chilling and heat stress where consistent positive effects have been noted (Park et al. 2004; Quan et al. 2004; Yang et al. 2005).
(c) Increasing harvest index
Much of agriculture depends on plant reproduction for the harvested part and for the seeds for the next crop (Boyer & Westgate 2004). Plant reproductive processes are extremely sensitive to water deficit, especially during the early phases when development may cease irreversibly even though the parent remains alive and vegetatively active. Grain numbers decrease if water deficits affect key development processes, especially ovary abortion in maize (Boyer & Westgate 2004) or pollen sterility in small grains (Saini & Westgate 2000). In maize, water deficits result in a decrease in photosynthate flux to the developing organs and this appears to trigger abortion. Manipulations that increase carbohydrate supply to developing grains during drought stress not only decrease abortion but can also increase grain yield. Recent work suggests that the enzyme invertase is a limiting step for grain development during water deficit, and genes have been identified whose regulation is affected during water deficit-induced abortion in maize (Boyer & Westgate 2004). We now need to understand tissue- and developmentally specific gene expression to link metabolic changes to the decreased sugar flux which controls the development of reproductive organs. This is an important area for plant improvement which needs increased attention.
8. Crop management strategies
Equations (3.2) and (3.3) highlight that in water-scarce environments, there are a number of strategies for crop management that can improve water use efficiency. These have been reviewed in several recent papers (e.g. Gregory 2004; Passioura 2004; Turner 2004a,b; Turner & Asseng 2005). In brief, they can be grouped into the following.
Capturing more water (W) for crop transpiration through water harvesting, reducing soil evaporation, improved weed control and by deeper root growth.
Improving BWR by exchanging transpired water for CO2 more effectively and converting into biomass.
Convert more biomass into harvestable yield (HI).
Water harvesting approaches may be appropriate in some dryland regions, by harvesting water on larger areas and channelling it onto crops. This is, in effect, a form of irrigation that has been practised traditionally in some regions. However, it is questionable that this is sustainable when viewed on the wider landscape scale, as it reduces groundwater recharge and possibly seasonal river flows that may be important to catchments and natural ecosystems. Soil evaporation can be substantially reduced through minimum tillage and mulching using materials or crop residues (see Hatfield et al. 2001). Most importantly, there is considerable recent work showing that growing paddy rice without standing water and with soil mulches can provide major improvements in water use efficiency, with small or no yield penalties (e.g. Bouman et al. 2005; Qin et al. 2006; Tao et al. 2006). Traditional paddy rice cultivation has used inundation for some 5000 years, but this results in 2–5 times more water use than other cereals (Bindraban et al. 2006). Given that paddy rice cultivation now covers a huge area and is of overwhelming importance in global food supply, these are potentially major developments, although the full agronomic, environmental and socio-economic consequences of such changes are complex (Bindraban et al. 2006). Soil evaporation can also be reduced by achieving a rapid ground cover through early crop vigour and fertilizers have been shown to help (Gregory 2004). However, there is a water cost to increased ground cover: the larger crop area will transpire more and thus needs to be coupled to selection of suitable genotypes to avoid later season water shortages (see above and discussion in Gregory 2004).
Improving BWR can potentially be done by improvement of the genotype (see above). In addition, reducing yield losses from weeds, pests and diseases through mechanical and agrochemical methods can achieve improved BWR, although such practices raise other questions about sustainability. However, altered management interacts with physiological changes introduced in new genotypes. For example, much has been achieved by selection of crops or cultivars that grow earlier in climates such as Mediterranean regions where potential evaporation increases during the season, and Δe is lower earlier on (Turner 2004a). This close matching of phenological development with rainfall patterns and environment is very important, but needs to be coupled with management that ensures early and rapid sowing (Turner 2004a,b). Adoption of minimum tillage practices should also help with early and rapid sowing. As a result of such better matching of crops and environment, coupled with the introduction of cultivars that respond to increased inputs, improved disease control and other agronomic changes, yields have doubled over the last 50 years in rainfed agriculture in Australia. Consequently, water productivity has doubled and both of these improvements have occurred even though water available for agriculture has declined over this period (Passioura 2004; Turner 2004a,b).
The HI of many of the major crops has been substantially improved genetically, and it is sometimes argued that the HI of cereals is reaching an upper limit in some systems (see, e.g. Hay 1995). New improvement targets are for improved assimilate production, storage and remobilization at the optimum time for seed filling, as in the stay-green sorghum lines with delayed senescence (see above). In contrast, management techniques such as deficit irrigation (see below) can be used to promote senescence, to promote remobilization of assimilate from storage in the stem to increase grain yield in both wheat and rice (Yang & Zhang 2006). In these species, stored non-structural carbohydrates in the stem and leaf sheaths may contribute 10–40% of the final grain yield, and this may be unavailable for grain development unless senescence is promoted (e.g. Schnyder 1993; Yang & Zhang 2006). The problem may be particularly acute if there is an excess of nitrogen in the soil after anthesis (Yang & Zhang 2006).
There is substantial evidence for all these measures contributing to improved crop yield in Mediterranean-climate areas (e.g. Passioura 2004; Turner 2004a,b), and there are good examples in other climates. Recently, Zhang et al. (2005) have reported that there have been 50% yield and water use efficiency increases in the North China Plain in winter wheat and maize over the last 20 years associated with mulching and improved irrigation scheduling. In the Central Plains of the USA, no-tillage practices have made it possible to intensify cropping from the traditional wheat–fallow system and produce a 30% increase in water use efficiency, as well as increase soil C (Peterson & Westfall 2004).
(a) Deficit irrigation as a means of managing crop water loss and increasing HI
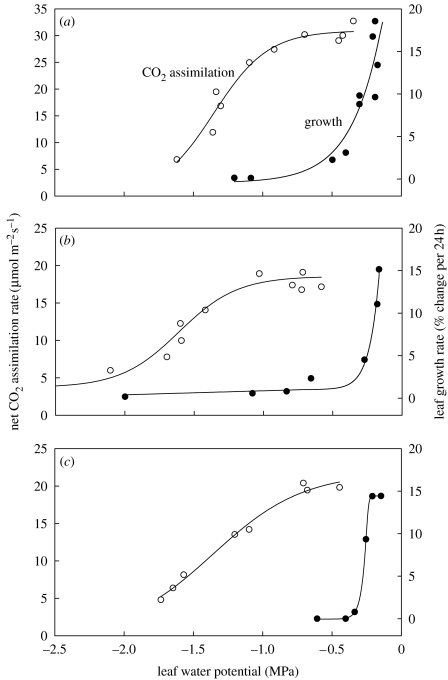
John Boyer showed in 1970 that in many cases, shoot growth is very sensitive to mild soil moisture deficits, and more sensitive than photosynthetic rate (figure 5). Owing to this shoot growth sensitivity, it is common in many production systems to supply excess irrigation water to ensure rapid and extensive canopy development and thereby maximize radiation interception and biomass accumulation. In many situations, irrigation systems are badly designed or maintained so that much of the water goes nowhere near the crop (so that the R term in equation (3.3) can be enormous). The spatial and temporal distribution of water is highly variable and growers will often leave irrigation systems running to ensure that the driest plants receive enough water to avoid the development of damaging water deficits. Thus, the wettest plants receive perhaps 3–4 times more water than they need, which itself can have damaging effects on crop growth and development.
Figure 5.
Response of net CO2 assimilation rates (open circles) and leaf growth rate (solid circles) to leaf water potential in three species: (a) maize, (b) soya bean and (c) sunflower. Redrawn from Boyer (1970).
There is now much interest in using remote sensing techniques such as thermal imaging (see above) to assess the level of plant stress and to use such measurements to control irrigation systems. This combination should result in water being applied to the plants that need it, in just the required amounts. Such technology which uses the plant as an indicator of water requirement (rather than soil water status, Jones 2004a–c; Parry et al. 2005) has the potential to greatly reduce the use of water in commercial, highly mechanized agriculture and very significantly increase BWR.
One management option with great promise is deficit irrigation (the application of only a predetermined percentage of calculated potential plant water use), and in particular, regulated deficit irrigation (RDI). As less water is supplied, mild soil drying occurs, but stomatal aperture is reduced more than assimilation rate (owing to the nonlinearity of the ratio A/gs; figure 1) so that ATR is increased, and possibly BWR. As figure 5 shows, mild soil drying resulting from the use of deficit irrigation will almost inevitably restrict shoot growth and leaf development. This response to drying may have several benefits for growers. Firstly, many fruit crops can show excessive vegetative vigour (Loveys et al. 2004), which can lead to high rates of water loss and development of damaging plant water deficits. Secondly, leaf growth can also compete with reproductive development for assimilates. Thirdly, excessive canopy development can cause shade, predispose plants to fungal diseases and adversely affect fruit development and fruit quality (Chalmers 1986). While vigorous crops are often pruned, this can provide infection sites for diseases and can be expensive if labour costs are high. Therefore, deficit irrigation can be beneficial in fruit crops. In cereals, it may also result in beneficial redistribution of assimilates from storage to developing grains to increase HI and grain yield (Yang et al. 2002; see above). However, deficit irrigation can also reduce fruit yields with the risk of catastrophic crop failure (e.g. Matthews & Anderson 1988), so that to operate RDI effectively, the grower must monitor plant and soil moisture status and deliver precisely regulated and directed quantities of water on demand.
(b) Partial root zone drying to manipulate the plant's signalling capacity
Plant growth in drying soil is commonly limited by a combination of chemical and hydraulic influences (e.g. Davies et al. 2002). Frequently, reductions in water availability result in reduced shoot turgor which can reduce shoot growth and development (figure 5). Even when turgor of growing shoot cells is sustained, growth can be limited by chemical ‘signals’ generated as a result of interactions between the root and the drying soil and transmitted to the shoot via the transpiration stream. Gowing et al. (1990) showed that production and expansion of leaves of apple trees could be restricted by watering only half the plant root system (purtial root drying; PRD) and that this limitation occurred without any detectable influence of the treatment on shoot water status. Excision of the roots in the drying soil promoted shoot growth so that the growth limitation under PRD was attributable to delivery to the shoots of chemical inhibitors in the transpiration stream, generated as a result of soil drying.
In recent years, there has been much interest in modifying the long-distance signalling through deficit irrigation and thereby modifying plant growth to the advantage of the producer and saving water. This possibility was first mooted by Loveys (1991), who suggested reduced water application to grapevines to restrict vegetative shoot development. It was proposed that irrigation could be regulated so as to control stomatal conductance, reduce plant water loss but sustain shoot water status to ensure ‘normal’ fruit development. Figure 6 shows that fruit turgor of tomatoes can be sustained under a particular form of deficit irrigation (Mingo et al. 2003), and Dry & Loveys (1999) have described results from experiments where grapevine vigour was significantly reduced and less water used without any yield penalty. Fruit production per unit of water used was therefore greatly increased compared to the industry standard. In addition, the quality of fruit was improved significantly.
Figure 6.
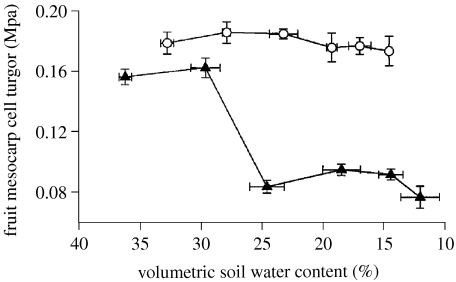
The relationship between volumetric soil water content and fruit cell turgor for tomato plants subjected to regulated deficit irrigation (closed triangles) and partial root zone drying (open circles). Points are means of 3±s.e. Adapted from Mingo et al. (2003).
In PRD, the two halves of the plant's root system are watered alternately (see Loveys et al. 2004). Roots in wet soil supply most of the water, while roots in contact with drying soil generate signals that move to the shoot to restrict shoot growth and functioning. Irrigation must be switched regularly from one side of the root to the other to keep roots in dry soil alive and fully functional and sustain the supply of root signals (figure 7). Maintenance of turgor and total water potential in the shoot can be important for maintenance of fruit growth and development although it is not entirely clear why fruit growth does not respond as sensitively as leaf growth to the increased flux of chemical signals (Davies et al. 2000). Increases in grape quality combined with reduced water use have now been reported as a result of the use of PRD on a commercial field scale (De Souza et al. 2003; Dos Santos et al. 2003).
Figure 7.
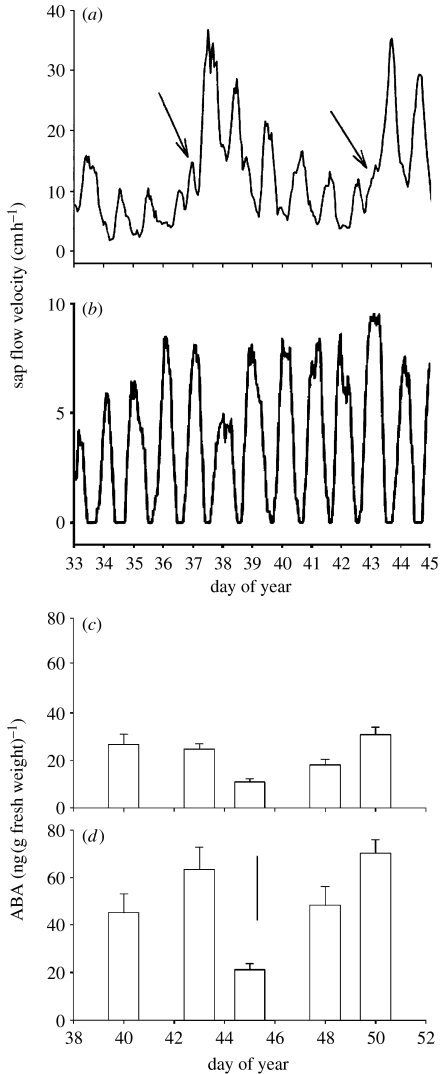
(a) Sap flow velocity in roots of Valencia orange on the (a) irrigated side of trees (arrows show times when irrigation occurred) and (b) non-irrigated side of trees. Abscisic acid (ABA) concentration in the roots (c) of control (both sides irrigated) Valencia orange trees and (d) from the non-irrigated side of PRD Valencia orange trees. Bar represents least significant difference (p=0.05), n=4. Adapted from Loveys et al. (2004) .
The application of the technique is not restricted to grapevines or other tree crops. In an extended series of studies in Australia, the UK, China and in countries around the Mediterranean (e.g. Stoll et al. 2000; Davies et al. 2002; Kang et al. 2002a,b; Kang & Zhang 2004), application of PRD to a range of other crops with different irrigation methods has resulted in substantial saving of irrigation water coupled with the maintenance of an economic yield. In these experiments, BWR has often at least doubled and in some cases increased much more. Despite some difficulties in operating PRD irrigation techniques in certain soils and some climatic conditions (Kriedemann 2003), it is clear that substantial water savings and increases in crop quality can result. Large-scale development of the technique continues (Kang & Zhang 2004).
9. Concluding comments
Water is a key driver of sustainability. Crop production in many parts of the world is seriously limited by lack of water, and water supplies for agriculture are also dwindling. Ultimately, this threatens the sustainability of agriculture in these regions, and furthermore agricultural water use has major impacts on the rest of the ecosystem. In areas where irrigation is used, there is an urgent need to increase the efficiency of supply and make much better use of the water applied. In other areas, rainfall is the only source of water for crop growth, thus in those areas any improvements in food yield per unit land area through crop management, fertilization, soil improvement, pest and disease control will usually improve water productivity. Many of the innovative local and regional schemes in developing countries where techniques have been adopted to improve agricultural system sustainability have improved yield and yield stability, and thus often improved water productivity (Pretty et al. 2006).
While major efforts are being made to improve yield per unit of water supplied, we clearly need more progress. As this review has demonstrated, many interlinked processes and factors underlie the apparent simplicity of the basic requirement for water in crop production, as encapsulated in equation (3.2). This inevitably means that to make progress requires understanding and progress in all of agronomy, hydrology, agricultural engineering, crop and plant physiology and molecular genetics. In addition, where changes are being made to fundamental aspects of a cropping system, such as changing the inundation duration of rice, the socio-economic aspects are as important. Encouragingly, there is currently exciting progress in improving crop production and water productivity through a combination of these disciplines as the examples described here show. Many of the more technologically demanding approaches are unlikely to be appropriate or available to small farmers. However, the development and release of new varieties with characteristics that will improve water productivity through combined physiological, biotechnological and agronomic research will help with the ‘Blue Revolution’ that is being called for, which could benefit all crop production.
Footnotes
One contribution of 16 to a Theme Issue ‘Sustainable agriculture I’.
References
- Alonso-Blanco C, Koornneef M. Naturally occurring variation in Arabidopsis: an underexploited resource for plant genetics. Trends Plant Sci. 2000;5:22–29. doi: 10.1016/s1360-1385(99)01510-1. doi:10.1016/S1360-1385(99)01510-1 [DOI] [PubMed] [Google Scholar]
- Araus J.L. The problems of sustainable water use in the Mediterranean and research requirements agriculture. Ann. Appl. Biol. 2004;144:259–272. doi:10.1111/j.1744-7348.2004.tb00342.x [Google Scholar]
- Bacon M.A. Water use efficiency in plant biology. Blackwell Publishing; Oxford, UK: 2004. p. 327. [Google Scholar]
- Baker N.R, Oxborough K. Chlorophyll fluorescence as a probe of photosynthetic productivity. In: Papageorgiou G.C, Govindjee , editors. Chlorophyll a fluorescence: a signature of photosynthesis. Springer; Dordrecht, The Netherelands: 2004. pp. 65–82. [Google Scholar]
- Baker N.R, Rosenqvist E. Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. J. Exp. Bot. 2004;55:1607–1621. doi: 10.1093/jxb/erh196. doi:10.1093/jxb/erh196 [DOI] [PubMed] [Google Scholar]
- Barbour M.M, Farquhar G.D. Relative humidity and ABA-induced variation in carbon and oxygen isotope ratios of cotton leaves. Plant Cell Environ. 2000;23:473–485. doi:10.1046/j.1365-3040.2000.00575.x [Google Scholar]
- Barbour M.M, Fischer R.A, Sayre K.D, Farquhar G.D. Oxygen isotope ratio of leaf and grain material correlates with stomatal conductance and grain yield in irrigated wheat. Aust. J. Plant Physiol. 2000;27:625–637. [Google Scholar]
- Bierhuizen J.F, Slatyer R.O. Effects of atmospheric concentration of water vapour and CO2 in determining transpiration–photosynthesis relationships of cotton leaves. Agric. Meteorol. 1965;2:259–270. doi:10.1016/0002-1571(65)90012-9 [Google Scholar]
- Bindraban P.S, Hengsdijk H, Cao W, Shi Q, Thiyagarajan T.M, Van der Krogt W, Wardana I.P. Transforming inundated rice cultivation. Int. J. Water Res. Dev. 2006;22:87–100. doi:10.1080/07900620500405338 [Google Scholar]
- Blanton D.B. Drought as a factor in the Jamestown colony, 1607–1612. Hist. Archaeol. 2000;34:74–81. doi: 10.1007/BF03374329. [DOI] [PubMed] [Google Scholar]
- Blum A. Drought resistance, water-use efficiency, and yield potential–are they compatible, dissonant, or mutually exclusive? Aust. J. Agric. Res. 2005;56:1159–1168. doi:10.1071/AR05069 [Google Scholar]
- Borrell A.K, Hammer G.L, Henzell R.G. Does maintaining green leaf area in sorghum improve yield under drought? 2. Dry matter production and yield. Crop Sci. 2000;40:1037–1048. [Google Scholar]
- Borrell A.K, Hammer G.L, Van Oosterom E. Stay-green: a consequence of the balance between supply and demand for nitrogen during grain filling? Ann. Appl. Biol. 2001;138:91–95. doi:10.1111/j.1744-7348.2001.tb00088.x [Google Scholar]
- Bouman B.A.M, Peng S, Castaneda A.R, Visperas R.M. Yield and water use of irrigated tropical aerobic rice systems. Agric. Water Manage. 2005;74:87–105. doi:10.1016/j.agwat.2004.11.007 [Google Scholar]
- Boyer J.S. Leaf enlargement and metabolic rates in corn, soybean, and sunflower at various leaf water potentials. Plant Physiol. 1970;46:233–235. doi: 10.1104/pp.46.2.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyer J.S, Westgate M.E. Grain yields with limited water. J. Exp. Bot. 2004;55:2385–2394. doi: 10.1093/jxb/erh219. doi:10.1093/jxb/erh219 [DOI] [PubMed] [Google Scholar]
- Bray E.A. Abscisic acid regulation of gene expression during water-deficit stress in the era of the Arabidopsis genome. Plant Cell Environ. 2002;25:153–161. doi: 10.1046/j.1365-3040.2002.00746.x. doi:10.1046/j.1365-3040.2002.00746.x [DOI] [PubMed] [Google Scholar]
- Chaerle L, Saibo N, Van Der Straeten D. Tuning the pores: towards engineering plants for improved water use efficiency. Trends Biotechnol. 2005;23:308–315. doi: 10.1016/j.tibtech.2005.04.005. doi:10.1016/j.tibtech.2005.04.005 [DOI] [PubMed] [Google Scholar]
- Chaerle L, Leinonen I, Jones H.G, Van Der Straeten D. Monitoring and screening plant populations with combined thermal and chlorophyll fluorescence imaging. J. Exp. Bot. 2007;58:773–784. doi: 10.1093/jxb/erl257. doi:10.1093/jxb/er1257 [DOI] [PubMed] [Google Scholar]
- Chalmers D.J. Research and progress in cultural systems and management in temperate fruit orchards. Acta Horticult. 1986;175:215–225. [Google Scholar]
- Chaves M.M, Oliveira M.M. Mechanisms underlying plant resilience to water deficits: prospects for water-saving agriculture. J. Exp. Bot. 2004;55:2365–2384. doi: 10.1093/jxb/erh269. doi:10.1093/jxb/erh269 [DOI] [PubMed] [Google Scholar]
- Condon A.G, Richards R.A, Rebetzke G.J, Farquhar G.D. Breeding for high water-use efficiency. J. Exp. Bot. 2004;55:2447–2460. doi: 10.1093/jxb/erh277. doi:10.1093/jxb/erh277 [DOI] [PubMed] [Google Scholar]
- Danielopol D.L, Griebler C, Gunatilaka A, Notenboom J. Present state and future prospects for groundwater ecosystems. Environ. Conserv. 2003;30:104–130. doi:10.1017/S0376892903000109 [Google Scholar]
- Davies W.J, Bacon M.A, Thompson D.S, Sobeih W, Gonzales-Rodriguez L. Regulation of leaf and fruit growth in plants growing in drying soil: exploitation of the plants' chemical signalling system and hydraulic architecture to increase the efficiency of water use in agriculture. J. Exp. Bot. 2000;51:1617–1626. doi: 10.1093/jexbot/51.350.1617. doi:10.1093/jexbot/51.350.1617 [DOI] [PubMed] [Google Scholar]
- Davies W.J, Wilkinson S, Loveys B.R. Stomatal control by chemical signalling and the exploitation of this mechanism to increase water use efficiency in agriculture. New Phytol. 2002;153:449–460. doi: 10.1046/j.0028-646X.2001.00345.x. doi:10.1046/j.0028-646X.2001.00345.x [DOI] [PubMed] [Google Scholar]
- Defra, 2002 A survey of irrigation of outdoor crops in 2001—England, (eds E. K. Wetherhead & K. Danert), pp. 4.
- Dos Santos T.P, Lopes C.M, Rodrigues M.L, de Souza C.R, Maroco J.P, Pereira J.S, Silva J.R, Chaves M.M. Partial rootzone drying: effects on growth and fruit quality of field grown grapevines. Funct. Plant Biol. 2003;30:663–671. doi: 10.1071/FP02180. doi:10.1071/FP02180 [DOI] [PubMed] [Google Scholar]
- De Souza C.R, Maroco J.P, dos Santos T.P, Rodriges M.L, Lopes C.M, Pereira J.S, Chaves M.M. Partial rootzone drying: regulation of stomatal aperture and carbon assimilation in field-grown grapevines. Funct. Plant Biol. 2003;30:653–662. doi: 10.1071/FP02115. doi:10.1071/FP02115 [DOI] [PubMed] [Google Scholar]
- Döll P, Siebert S. Global modeling of irrigation water requirements. Water Resour. Res. 2002;38:1037. doi:10.1029/2001WR000355 [Google Scholar]
- Dry P, Loveys B.R. Grapevine shoot growth and stomatal conductance are reduced when part of the root system is dried. Vitis. 1999;38:151–156. [Google Scholar]
- Dry P.R, Loveys B.R, During H. Partial drying of the root zone of grape 2. Changes in the pattern of root development. Vitis. 2000;39:9–12. [Google Scholar]
- Edmeades G.O, Bolaños J, Chapman S.C, Lafitte H.R, Banziger M. Selection improves drought tolerance in tropical maize populations. I. Gains in biomass, grain yield and harvest index. Crop Sci. 1999;39:1306–1315. [Google Scholar]
- EEA 2003 Europe's environment: the third assessment, ch. 8: Water. Copenhagen. See http://reports.eea.europa.eu/environmental_assessment_report_2003_10/en
- Ellul P, Angosto T, Garcia-Sogo B, Garcia-Hurtado N, Martin-Trillo M, Salinas M, Moreno V, Lozano R, Martinez-Zapata M. Expression of Arabidopsis APETALA1 in tomato reduces its vegetative cycle without affecting plant production. Mol. Breed. 2004;13:155–163. doi:10.1023/B:MOLB.0000018763.64585.6b [Google Scholar]
- EPSO. European plant science: a field of opportunities. J. Exp. Bot. 2005;56:1699–1709. doi: 10.1093/jxb/eri212. doi:10.1093/jxb/eri212 [DOI] [PubMed] [Google Scholar]
- Farquhar G.D, O'Leary M.H, Berry J.A. On the relationship between carbon-isotope discrimination and intercellular carbon dioxide concentration. Aust. J. Plant Physiol. 1982;9:121–137. [Google Scholar]
- Farquhar G.D, Ehleringer J.R, Hubick K.T. Carbon isotope discrimination and photosynthesis. Annu. Rev. Plant Phys. 1989;40:503–537. doi:10.1146/annurev.pp.40.060189.002443 [Google Scholar]
- Farquhar G.D, Condon A.G, Masle J. Use of carbon and oxygen isotope composition and mineral ash content in breeding for improved rice production under favourable, irrigated conditions. In: Cassman K.G, editor. Breaking the yield barrier. IRRI; Manila, The Philippines: 1994. pp. 95–101. [Google Scholar]
- FAO. Crops and drops: making the best use of water for agriculture. FAO; Rome, Italy: 2002. p. 28. [Google Scholar]
- FAO. Review of world water resources by country. FAO; Rome, Italy: 2003a. p. 123. Water reports 23. [Google Scholar]
- FAO 2003b AQUASTAT–Global information system on water and agriculture. See http://www.fao.org/ag/agl/aglw/aquastat/main/index.stm
- FAOSTAT 2006–FAO data for agriculture: statistics database. See http://faostat.fao.org/faostat/collections?version=ext&hasbulk=0&subset=agriculture
- Fitz Gerald J.N, Lehti-Shiu M.D, Ingram P.A, Deak K.I, Biesiada T, Malamy J.E. Identification of quantitative trait loci that regulate Arabidopsis root system size. Genetics. 2006;172:485–498. doi: 10.1534/genetics.105.047555. doi:10.1534/genetics.105.047555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foolad M.R, Zhang L.P, Subbiah P. Genetics of drought tolerance during seed germination in tomato: inheritance and QTL mapping. Genome. 2003;46:536–545. doi: 10.1139/g03-035. doi:10.1139/g03-035 [DOI] [PubMed] [Google Scholar]
- Ghannoum O, von Caemmerer S, Conroy J. The effect of drought on plant water use efficiency of nine NAD-ME and nine NADP-ME Australian C4 grasses. Funct. Plant Biol. 2002;29:1337–1348. doi: 10.1071/FP02056. doi:10.1071/FP02056 [DOI] [PubMed] [Google Scholar]
- Ghashghaie J, Duranceau M, Badeck F.W, Cornic G, Adeline M.T, Deleens E. δ13C of CO2 respired in the dark in relation to δ13C of leaf metabolites: comparison between Nicotiana sylvestris and Helianthus annuus under drought. Plant Cell Environ. 2001;24:505–515. doi:10.1046/j.1365-3040.2001.00699.x [Google Scholar]
- Gowing D.J.G, Davies W.J, Jones H.G. A positive root-sourced signal as an indicator of soil drying in apple, Malus x domestica Borkh. J. Exp. Bot. 1990;41:1535–1540. doi:10.1093/jxb/41.12.1535 [Google Scholar]
- Gray J.E, Holroyd G.H, van der Lee F, Bahrami A.R, Sijmons P.C, Woodward F.I, Schuch W, Hetherington A.M. The HIC signalling pathway links CO2 perception to stomatal development. Nature. 2000;408:713–716. doi: 10.1038/35047071. doi:10.1038/35042663 [DOI] [PubMed] [Google Scholar]
- Gregory P.J. Agronomic approaches to increasing water use efficiency. In: Bacon M.A, editor. Water use efficiency in plant biology. Blackwell Publishing; Oxford, UK: 2004. pp. 142–170. ch. 6. [Google Scholar]
- Gregory P.J, Simmonds L.P, Warren G.P. Interactions between plant nutrients, water and carbon dioxide as factors limiting crop yield. Phil. Trans. R. Soc. B. 1997;352:987–996. doi:10.1098/rstb.1997.0077 [Google Scholar]
- Gur A, Zamir D. Unused natural variation can lift breeding barriers in plant breeding. PLoS Biol. 2004;2:1610–1615. doi: 10.1371/journal.pbio.0020245. doi:10.1371/journal.pbio.0020245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall N.M, Griffiths H, Corlett J.A, Jones H.G, Lynn J, King G.J. Relationships between water-use traits and photosynthesis in Brassica oleracea resolved by quantitative genetic analysis. Plant Breed. 2005;124:557–564. doi:10.1111/j.1439-0523.2005.01164.x [Google Scholar]
- Hatfield J.L, Sauer T.J, Prueger J.H. Managing soils to achieve greater water use efficiency: a review. Agron. J. 2001;93:271–280. [Google Scholar]
- Haug G.H, Gunther D, Peterson L.C, Sigman D.M, Hughen K.A, Aeschlimann B. Climate and the collapse of Maya civilization. Science. 2003;299:1731–1735. doi: 10.1126/science.1080444. doi:10.1126/science.1080444 [DOI] [PubMed] [Google Scholar]
- Hay R.K.M. Harvest index—a review of its use in plant-breeding and crop physiology. Ann. Appl. Biol. 1995;126:197–216. [Google Scholar]
- Hodell D.A, Brenner M, Curtis J.H. Terminal classic drought in the northern Maya lowlands inferred from multiple sediment cores in Lake Chichancanab (Mexico) Quat. Sci. Rev. 2005;24:1413–1427. doi:10.1016/j.quascirev.2004.10.013 [Google Scholar]
- Horie T, Matsuura S, Takai T, Kuwasaki K, Ohsumi A, Shiraiwa T. Genotypic difference in canopy diffusive conductance measured by a new remote-sensing method and its association with the difference in rice yield potential. Plant Cell Environ. 2006;29:653–660. doi: 10.1111/j.1365-3040.2005.01445.x. doi:10.1111/j.1365-3040.2005.01445.x [DOI] [PubMed] [Google Scholar]
- Huang J, Hirji R, Adam L, Rozwadowski K.L, Hammerlindl J.K, Keller W.A, Selvaraj G. Genetic engineering of glycinebetaine production toward enhancing stress tolerance in plants: metabolic limitations. Plant Physiol. 2000;122:747–756. doi: 10.1104/pp.122.3.747. doi:10.1104/pp.122.3.747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- IPCC, 2007 Climate change 2007: the physical science basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (eds S. D. Solomon, M. Qin, M. Manning, Z. Chen, M. Marquis, K. B. Averyt, M. Tignor & H. L. Miller). Cambridge, UK and New York, USA: Cambridge University Press.
- Jones H.G. Use of thermography for quantitative studies of spatial and temporal variation of stomatal conductance over leaf surfaces. Plant Cell Environ. 1999;22:1043–1055. doi:10.1046/j.1365-3040.1999.00468.x [Google Scholar]
- Jones H.G. What is water use efficiency? In: Bacon M.A, editor. Water use efficiency in plant biology. Blackwell Publishing; Oxford, UK: 2004a. pp. 27–41. ch. 3. [Google Scholar]
- Jones H.G. Irrigation scheduling: advantages and pitfalls of plant-based methods. J. Exp. Bot. 2004b;55:2427–2436. doi: 10.1093/jxb/erh213. doi:10.1093/jxb/erh213 [DOI] [PubMed] [Google Scholar]
- Jones H.G. Application of thermal imaging and infrared sensing in plant physiology and ecophysiology. Adv. Bot. Res. 2004c;41:107–163. [Google Scholar]
- Jones H.G, Stoll M, Santos T, de Sousa C, Chaves M.M, Grant O.M. Use of infrared thermography for monitoring stomatal closure in the field: application to grapevine. J. Exp. Bot. 2002;53:2249–2260. doi: 10.1093/jxb/erf083. doi:10.1093/jxb/erf083 [DOI] [PubMed] [Google Scholar]
- Jongdee B, Pantuwan G, Fukai S, Fischer K. Improving drought tolerance in rainfed lowland rice: an example from Thailand. Agric. Water Manage. 2006;80:225–240. doi:10.1016/j.agwat.2005.07.015 [Google Scholar]
- Juenger T.E, McKay J.K, Hausmann N, Keurentjes J.J.B, Sen S, Stowe K.A, Dawson T.E, Simms E.L, Richards J.H. Identification and characterization of QTL underlying whole-plant physiology in Arabidopsis thaliana: δ13C, stomatal conductance and transpiration efficiency. Plant Cell Environ. 2005;28:697–708. doi:10.1111/j.1365-3040.2004.01313.x [Google Scholar]
- Kang S, Zhang J. Controlled alternate partial root-zone irrigation: its physiological consequences and impact on water use efficiency. J. Exp. Bot. 2004;55:2437–2446. doi: 10.1093/jxb/erh249. doi:10.1093/jxb/erh249 [DOI] [PubMed] [Google Scholar]
- Kang J.-Y, Choi H.-I, Im M.-Y, Kim S.Y. Arabidopsis basic leucine zipper proteins that mediate stress-responsive abscisic acid signaling. Plant Cell. 2002a;14:343–357. doi: 10.1105/tpc.010362. doi:10.1105/tpc.010362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang S, Hu X, Li Z, Jerie P. Soil water distribution, water use and yield response to partial rootzone drying under shallow groundwater conditions in a pear orchard. Scientia Horticulturae. 2002b;92:277–291. doi:10.1016/S0304-4238(01)00300-4 [Google Scholar]
- Kasuga M, Liu Q, Miura S, Yamaguchi-Shinozaki K, Shinozaki K. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat. Biotech. 1999;17:287–291. doi: 10.1038/7036. doi:10.1038/7036 [DOI] [PubMed] [Google Scholar]
- Keitel C, Adams M.A, Holst T, Matzarakis A, Mayer H, Rennenberg H, Gessler A. Carbon and oxygen isotope composition of organic compounds in the phloem sap provides a short-term measure for stomatal conductance of European beech (Fagus sylvatica L.) Plant Cell Environ. 2003;26:1157–1168. doi:10.1046/j.1365-3040.2003.01040.x [Google Scholar]
- Kemanian A.R, Stöckle C.O, Higgins D.R. Transpiration-use efficiency of barley. Agric. Forest Meteorol. 2005;130:1–11. doi:10.1016/j.agrformet.2005.01.003 [Google Scholar]
- Khan S, Tariq R, Cui Y.L, Blackwell J. Can irrigation be sustainable? Agric. Water Manage. 2006;80:87–99. doi:10.1016/j.agwat.2005.07.006 [Google Scholar]
- Kriedemann, P. W. 2003 Regulated deficit irrigation and partial root drying. An occasional paper commissioned by Land and Water Australia.
- Lamb H.H. 2nd edn. Routledge; London, UK: 1995. Climate, history and the modern world. pp. 433. [Google Scholar]
- Leinonen I, Grant O.M, Tagliavia C.P.P, Chaves M.M, Jones H.G. Estimating stomatal conductance with thermal imagery. Plant Cell Environ. 2006;29:1508–1518. doi: 10.1111/j.1365-3040.2006.01528.x. doi:10.1111/j.1365-3040.2006.01528.x [DOI] [PubMed] [Google Scholar]
- Levitt J. Responses of plants to environmental stresses. Academic Press; New York, NY: 1972. [Google Scholar]
- Loudet O, Chaillou S, Krapp A, Daniel-Vedele F. Quantitative trait loci analysis of water and anion contents in interaction with nitrogen availability in Arabidopsis thaliana. Genetics. 2003;163:711–722. doi: 10.1093/genetics/163.2.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loudet O, Gaudon V, Trubuil A, Daniel-Vedele F. Quantitative trait loci controlling root growth and architecture in Arabidopsis thaliana confirmed by heterogeneous inbred family. Theor. Appl. Genet. 2005;110:742–753. doi: 10.1007/s00122-004-1900-9. doi:10.1007/s00122-004-1900-9 [DOI] [PubMed] [Google Scholar]
- Loveys B.R. How useful is a knowledge of ABA physiology for crop improvement? In: Davies W.J, Jones H.G, editors. Abscisic acid: physiology and biochemistry. BIOS Scientific Publishers; Oxford, UK: 1991. pp. 245–260. ch. 17. [Google Scholar]
- Loveys B.R, Stoll M, Davies W.J. Physiological approaches to enhance water use efficiency in agriculture: exploiting plant signalling in novel irrigation practice. In: Bacon M.A, editor. Water use efficiency in plant biology. ch. 5. Blackwell Publishing; Oxford, UK: 2004. pp. 113–141. [Google Scholar]
- Lu Z.M, Zeiger E. Selection for higher yields and heat-resistance in pima cotton has caused genetically-determined changes in stomatal conductances. Physiol. Plantarum. 1994;92:273–278. doi:10.1111/j.1399-3054.1994.tb05337.x [Google Scholar]
- Lu Z.M, Percy R.G, Qualset C.O, Zeiger E. Stomatal conductance predicts yields in irrigated Pima cotton and bread wheat grown at high temperatures. J. Exp. Bot. 1998;49:453–460. doi:10.1093/jexbot/49.suppl_1.453 [Google Scholar]
- Ma X, Fukushima Y, Liu C, Wu X. A hydrological model application to the small tributary basis of the Yellow River. Geophys. Res. Abstr. 2003;5:08024. [Google Scholar]
- Martin B, Nienhuis J, King G, Schaefer A. Restriction fragment length polymorphisms associated with water-use efficiency in tomato. Science. 1989;243:1725–1728. doi: 10.1126/science.243.4899.1725. doi:10.1126/science.243.4899.1725 [DOI] [PubMed] [Google Scholar]
- Masle J, Gilmour S.R, Farquhar G.D. The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature. 2005;436:866–870. doi: 10.1038/nature03835. doi:10.1038/nature03835 [DOI] [PubMed] [Google Scholar]
- Matthews M.A, Anderson M.M. Fruit ripening in Vitis vinifera L. Responses to seasonal water deficits. Am. J. Enol. Vitic. 1988;40:52–60. [Google Scholar]
- Merlot S, Mustilli A.C, Genty B, North H, Lefebvre V, Sotta B, Vavasseur A, Giraudat J. Use of infrared thermal imaging to isolate Arabidopsis mutants defective in stomatal regulation. Plant J. 2002;30:601–609. doi: 10.1046/j.1365-313x.2002.01322.x. doi:10.1046/j.1365-313X.2002.01322.x [DOI] [PubMed] [Google Scholar]
- Messinger S.M, Buckley T.N, Mott K.A. Evidence for involvement of photosynthetic processes in the stomatal response to CO2. Plant Physiol. 2006;140:771–778. doi: 10.1104/pp.105.073676. doi:10.1104/pp.105.073676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mingo D.M, Bacon M.A, Davies W.J. Non-hydraulic regulation of fruit growth in tomato plants growing in drying soil. J. Exp. Bot. 2003;54:1205–1212. doi: 10.1093/jxb/erg120. doi:10.1093/jxb/erg120 [DOI] [PubMed] [Google Scholar]
- Mingo D.M, Theobald J.C, Bacon M.A, Davies W.J, Dodd I.C. Biomass allocation in tomato (Lycopersicon esculentum) plants grown under partial rootzone drying: enhancement of root growth. Funct. Plant Biol. 2004;31:971–978. doi: 10.1071/FP04020. doi:10.1071/FP04020 [DOI] [PubMed] [Google Scholar]
- Mitchell-Olds T, Schmitt J. Genetic mechanisms and evolutionary significance of natural variation in Arabidopsis. Nature. 2006;441:947–952. doi: 10.1038/nature04878. doi:10.1038/nature04878 [DOI] [PubMed] [Google Scholar]
- Monteith J.L. Consistency and convenience in the choice of units for agricultural science. Exp. Agric. 1984;20:105–117. [Google Scholar]
- Monteith J.L. The exchange of water and carbon by crops in a Mediterranean climate. Irrig. Sci. 1993;14:85–91. doi:10.1007/BF00208401 [Google Scholar]
- Morgan J.M. Increases in grain yield of wheat by breeding for an osmoregulation gene: relationship to water supply and evaporative demand. Aust. J. Agric. Res. 2000;51:971–978. doi:10.1071/AR00062 [Google Scholar]
- Morison J.I.L. Response of plants to CO2 under water limited conditions. Vegetatio. 1993;104:193–209. doi:10.1007/BF00048153 [Google Scholar]
- Morison J.I.L, Gifford R.M. Stomatal sensitivity to carbon dioxide and humidity: a comparison of two C3 and two C4 species. Plant Physiol. 1983;71:789–796. doi: 10.1104/pp.71.4.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouchel C.F, Briggs G.C, Hardtke S.C. Natural genetic variation in Arabidopsis identifies BREVIS RADIX, a novel regulator of cell proliferation and elongation in the root. Genes Dev. 2004;18:700–714. doi: 10.1101/gad.1187704. doi:10.1101/gad.1187704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nainanayake, A. D. 2004 Impact of drought on coconut (Cocos nucifera L.): screening germplasm for photosynthetic tolerance in the field. PhD thesis, University of Essex, pp. 215.
- Nakashima K, Yamaguch-Shinozaki K. Regulons involved in osmotic stress-responsive and cold stress-responsive gene expression in plants. Physiol. Plant. 2006;126:62–71. doi:10.1111/j.1399-3054.2005.00592.x [Google Scholar]
- Nedbal L, Whitmarsh J. Chlorophyll fluorescence imaging of leaves and fruits. In: Papageorgiou G.C, Govindjee S, editors. Chlorophyll a fluorescence: a signature of photosynthesis. Springer; Dordrecht, The Netherlands: 2004. pp. 389–407. [Google Scholar]
- Oh S.-J, Song S.I, Kim Y.S, Jang H.-J, Kim S.Y, Kim M, Kim Y.-K, Nahm B.H, Kim J.-K. Arabidopsis CBF3/DREB1A and ABF3 in transgenic rice increased tolerance to abiotic stress without stunting growth. Plant Physiol. 2005;138:341–351. doi: 10.1104/pp.104.059147. doi:10.1104/pp.104.059147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omasa K, Takayama K. Simultaneous measurement of stomatal conductance, non-photochemical quenching, and photochemical yield of photosystem II in intact leaves by thermal and chlorophyll fluorescence imaging. Plant Cell Physiol. 2003;44:1290–1300. doi: 10.1093/pcp/pcg165. doi:10.1093/pcp/pcg165 [DOI] [PubMed] [Google Scholar]
- Oxborough K. Using chlorophyll a fluorescence imaging to monitor photosynthetic performance. In: Papageorgiou G.C, Govindjee S, editors. Chlorophyll a fluorescence: a signature of photosynthesis. Springer; Dordrecht, The Netherlands: 2004. pp. 409–428. [Google Scholar]
- Park E.J, Jeknic Z, Sakamoto A, DeNoma J, Yuwansiri R, Murata N, Chen T.H.H. Genetic engineering of glycine betaine biosynthesis in tomato protects seeds, plants and flowers from chilling damage. Plant J. 2004;40:474–487. doi: 10.1111/j.1365-313X.2004.02237.x. doi:10.1111/j.1365-313X.2004.02237.x [DOI] [PubMed] [Google Scholar]
- Parry M.A.J, Flexas J, Medrano H. Prospects for crop production under drought: research priorities and future directions. Ann. Appl. Biol. 2005;147:211–226. doi:10.1111/j.1744-7348.2005.00032.x [Google Scholar]
- Passioura J.B. Grain yield, harvest index, and water use of wheat. J. Aust. Inst. Agric. Sci. 1977;43:117–121. [Google Scholar]
- Passioura J.B. Environmental biology and crop improvement. Funct. Plant Biol. 2002;29:537–546. doi: 10.1071/FP02020. doi:10.1071/FP02020 [DOI] [PubMed] [Google Scholar]
- Passioura, J. B. 2004 Increasing crop productivity when water is scarce–from breeding to field management. In New Directions for a Diverse Planet. Proc. 4th Int. Crop Science Congress, Brisbane, Australia, 26 September–1 October, 2004, (eds R. A. Fischer, N. Turner, J. Angus, L. McIntyre, M. Robertson, A. Borrell & D. Lloyd). See www.cropscience.org.au/icsc2004
- Pellegrineschi A, Reynolds M, Pacheco M, Brito R.M, Almeraya R, Yamaguchi-Shinozaki K, Hoisington D. Stress-induced expression in wheat of the Arabidopsis thaliana DREB1A gene delays water stress symptoms under greenhouse conditions. Genome. 2004;47:493–500. doi: 10.1139/g03-140. doi:10.1139/g03-140 [DOI] [PubMed] [Google Scholar]
- Penman H.L. Natural evaporation from open water bare soil and grass. Proc. R. Soc. A. 1948;193:120–145. doi: 10.1098/rspa.1948.0037. doi:10.1098/rspa.1948.0037 [DOI] [PubMed] [Google Scholar]
- Penman H.L, Schofield R.K. Some physical aspects of assimilation and transpiration. Symp. Soc. Exp. Biol. 1951;5:115–129. [Google Scholar]
- Pereira, J. S, Chaves, M. M., Caldeira, M. C. & Correia, A. V. 2007 Water availability and productivity, ch. 6. In Plant growth & climate change (eds J. I. L. Morison & M. D. Morecroft), pp. 118–145. Oxford, UK: Blackwell Publishing.
- Peterson G.A, Westfall D.G. Managing precipitation use in sustainable dryland agroecosystems. Ann. Appl. Biol. 2004;144:127–138. doi:10.1111/j.1744-7348.2004.tb00326.x [Google Scholar]
- Petty L.M, Harberd N.P, Carre I.A, Thomas B, Jackson S.D. Expression of the Arabidopsis gai gene under its own promoter causes a reduction in plant height in chrysanthemum by attenuation of the gibberellin response. Plant Sci. 2003;164:175–182. doi:10.1016/S0168-9452(02)00380-1 [Google Scholar]
- Pimental D, et al. Water resources: agricultural and environmental issues. Bioscience. 2004;54:909–918. doi:10.1641/0006-3568(2004)054[0909:WRAAEI]2.0.CO;2 [Google Scholar]
- Pretty J, Noble A.D, Bossio D, Dixon J, Hine R.E, Penning de Vries F.W.T, Morison J.I.L. Resource-conserving agriculture increases yields in developing countries. Environ. Sci. Technol. 2006;40:1114–1119. doi: 10.1021/es051670d. doi:10.1021/es051670d [DOI] [PubMed] [Google Scholar]
- Qin J.T, Hu F, Zhang B, Wei Z.G, Li H.X. Role of straw mulching in non-continuously flooded rice cultivation. Agric. Water Manage. 2006;83:252–260. doi:10.1016/j.agwat.2006.01.001 [Google Scholar]
- Quan R.D, Shang M, Zhang H, Zhao Y.X, Zhang J.R. Improved chilling tolerance by transformation with beta gene for enhancement of glycinebetaine synthesis in maize. Plant Sci. 2004;166:141–149. doi:10.1016/j.plantsci.2003.08.018 [Google Scholar]
- Quarrie S.A. New molecular tools to improve the efficiency of breeding for increased drought resistance. Plant Growth Regul. 1996;20:167–178. doi:10.1007/BF00024013 [Google Scholar]
- Quesada V, MacKnight R, Dean C, Simpson G.G. Autoregulation of FCA pre-mRNA processing controls Arabidopsis flowering time. EMBO J. 2003;22:3142–3152. doi: 10.1093/emboj/cdg305. doi:10.1093/emboj/cdg305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radin J.W, Lu Z.M, Percy R.G, Zeiger E. Genetic-variability for stomatal conductance in pima cotton and its relation to improvements of heat adaptation. Proc. Natl Acad. Sci. USA. 1994;91:7217–7221. doi: 10.1073/pnas.91.15.7217. doi:10.1073/pnas.91.15.7217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Razem F.A, El-Kereamy A, Abrams S.R, Hill R.D. The RNA-binding protein FCA is an abscisic acid receptor. Nature. 2006;439:290–294. doi: 10.1038/nature04373. doi:10.1038/nature04373 [DOI] [PubMed] [Google Scholar]
- Rebetzke G.J, Condon A.G, Richards R.A, Farquhar G.D. Selection for reduced carbon isotope discrimination increases aerial biomass and grain yield of rainfed bread wheat. Crop Sci. 2002;42:739–745. [Google Scholar]
- Richards, R. A. 2004 Physiological traits used in the breeding of new cultivars for water-scarce environments. In New Directions for a Diverse Planet. Proc. 4th Int. Crop Science Congress, Brisbane, Australia, 26 September–1 October, 2004 (eds R. A. Fischer, N. Turner, J. Angus, L. McIntyre, M. Robertson, A. Borrell & D. Lloyd). See www.cropscience.org.au/icsc2004
- Richards R.A, Passioura J.B. A breeding program to reduce the diameter of the major xylem vessel in the seminal roots of wheat and its effect on grain yield in rain-fed environments. Austr. J. Agric. Res. 1989;40:943–950. doi:10.1071/AR9890943 [Google Scholar]
- Rijsberman, F. 2004 Water scarcity–fact or fiction? In New Directions for a Diverse Planet. Proc. 4th Int. Crop Science Congress, Brisbane, Australia, 26 September–1 October, 2004 (eds R. A. Fischer, N. Turner, J. Angus, L. McIntyre, M. Robertson, A. Borrell & D. Lloyd). See www.cropscience.org.au/icsc2004
- Saini H.S, Westgate M.E. Reproductive development in grain crops during drought. Adv. Agron. 2000;68:59–96. [Google Scholar]
- Sakamoto A, Murata N. The role of glycine betaine in the protection of plants from stress: clues from transgenic plants. Plant Cell Environ. 2002;25:163–171. doi: 10.1046/j.0016-8025.2001.00790.x. doi:10.1046/j.0016-8025.2001.00790.x [DOI] [PubMed] [Google Scholar]
- Salvi S, Tuberosa R. To clone or not to clone plant QTLs: present and future challenges. Trends Plant Sci. 2005;10:297–304. doi: 10.1016/j.tplants.2005.04.008. doi:10.1016/j.tplants.2005.04.008 [DOI] [PubMed] [Google Scholar]
- Saranga Y, Menz M, Jiang C.-X, Wright R.J, Yakir D, Paterson A.H. Genomic dissection of genotype x environment interactions conferring adaptation of cotton to arid conditions. Genome Res. 2001;11:1988–1995. doi: 10.1101/gr.157201. doi:10.1101/gr.157201 [DOI] [PubMed] [Google Scholar]
- Schneider K, Brothers M.E, Kelly J.D. Marker-assisted selection to improve drought resistance in common bean. Crop Sci. 1997;37:51–60. [Google Scholar]
- Schnyder H. The role of carbohydrate storage and redistribution in the source–sink relations of wheat and barley during grain filling—a review. New Phytol. 1993;123:233–245. doi:10.1111/j.1469-8137.1993.tb03731.x [Google Scholar]
- Serraj R, Sinclair T.R. Osmolyte accumulation: can it really help increase crop yield under drought conditions? Plant Cell Environ. 2002;25:333–341. doi: 10.1046/j.1365-3040.2002.00754.x. doi:10.1046/j.1365-3040.2002.00754.x [DOI] [PubMed] [Google Scholar]
- Serraj R, Hash C.T, Rizvi S.M.H, Sharma A, Yadav R.S, Bidinger F.R. Recent advances in marker-assisted selection for drought tolerance in pearl millet. Plant Prod. Sci. 2005;8:334–337. doi:10.1626/pps.8.334 [Google Scholar]
- Sharp R.E, Poroyko V, Hejlek L.G, Spollen W.G, Springer G.K, Bohnert H.J, Nguyen H.T. Root growth maintenance during water deficits: physiology to functional genomics. J. Exp. Bot. 2004;55:2343–2351. doi: 10.1093/jxb/erh276. doi:10.1093/jxb/erh276 [DOI] [PubMed] [Google Scholar]
- Sheshshayee M.S, Bindumadhava1 H, Ramesh R, Prasad T.G, Lakshminarayana M.R, Udayakumar R. Oxygen isotope enrichment (δ18O) as a measure of time-averaged transpiration rate. J. Exp. Bot. 2005;56:3033–3039. doi: 10.1093/jxb/eri300. doi:10.1093/jxb/eri300 [DOI] [PubMed] [Google Scholar]
- Shiklomanov I.A, Rodda J.C. International hydrology series. Cambridge University Press; Cambridge, UK: 2003. World water resources at the beginning of the 21st Century. pp. 450. [Google Scholar]
- Shpak E.D, McAbee J.M, Pillitteri L.J, Torii K.U. Stomatal patterning and differentiation by synergistic interactions of receptor kinases. Science. 2005;309:290–293. doi: 10.1126/science.1109710. doi:10.1126/science.1109710 [DOI] [PubMed] [Google Scholar]
- Sinclair T.R, Purcell L.C. Is a physiological perspective relevant in a ‘genocentric’ age? J. Exp. Bot. 2005;56:2777–2782. doi: 10.1093/jxb/eri297. doi:10.1093/jxb/eri297 [DOI] [PubMed] [Google Scholar]
- Skinner J, Szucs P, von Zitzewitz J, Marquez-Cardillo L, Filichkin T, Stockinger E.J, Thomashow M.F, Chen T.T.H, Hayes P.M. Mapping of barley homologs to genes that regulate low temperature tolerance in Arabidopsis. Theor. Appl. Genet. 2006;112:832–842. doi: 10.1007/s00122-005-0185-y. doi:10.1007/s00122-005-0185-y [DOI] [PubMed] [Google Scholar]
- Slafer G.A, Araus J.L, Royo C, Del Moral L.F.G. Promising eco-physiological traits for genetic improvement of cereal yields in Mediterranean environments. Ann. Appl. Biol. 2005;146:61–70. doi:10.1111/j.1744-7348.2005.04048.x [Google Scholar]
- Sobeih W.Y, Dodd I.C, Bacon M.A, Grierson D, Davies W.J. Long-distance signals regulating stomatal conductance and leaf growth in tomato (Lycopersicon esculentum) plants subjected to partial root-zone drying. J. Exp. Bot. 2004;55:2353–2363. doi: 10.1093/jxb/erh204. doi:10.1093/jxb/erh204 [DOI] [PubMed] [Google Scholar]
- Stahle D.W, Cleaveland M.K, Blanton D.B, Therrell M.D, Gay D.A. The lost colony and Jamestown droughts. Science. 1998;280:564–567. doi: 10.1126/science.280.5363.564. doi:10.1126/science.280.5363.564 [DOI] [PubMed] [Google Scholar]
- Stanhill G. Water use efficiency. Adv. Agron. 1986;39:53–85. [Google Scholar]
- Steele K, Price A.H, Shashidhar H.E, Witcombe J.R. Marker-assisted selection to introgress rice QTLs controlling root traits into an Indian upland rice variety. Theor. Appl. Genet. 2006;112:208–221. doi: 10.1007/s00122-005-0110-4. doi:10.1007/s00122-005-0110-4 [DOI] [PubMed] [Google Scholar]
- Stoll M, Loveys B, Dry P. Hormonal changes induced by partial root zone drying of irrigated grapevine. J. Exp. Bot. 2000;51:1627–1634. doi: 10.1093/jexbot/51.350.1627. doi:10.1093/jexbot/51.350.1627 [DOI] [PubMed] [Google Scholar]
- Subudhi P.K, Rosenow D.T, Nguyen H.T. Quantitative trait loci for the stay green trait in sorghum (Sorghum bicolor L. Moench): consistency across genetic backgrounds and environments. Theor. Appl. Genet. 2000;101:733–741. doi:10.1007/s001220051538 [Google Scholar]
- Tadege M, Sheldon C.C, Helliwell C.A, Upadhyaya N.M, Dennis E.S, Peacock W.J. Reciprocal control of flowering time by OsSOC1 in transgenic Arabidopsis and by FLC in transgenic rice. Plant Biotech. J. 2003;1:361–369. doi: 10.1046/j.1467-7652.2003.00034.x. doi:10.1046/j.1467-7652.2003.00034.x [DOI] [PubMed] [Google Scholar]
- Tanner C.B. Transpiration efficiency of potato. Agron. J. 1981;73:59–64. [Google Scholar]
- Tanner C.B, Sinclair T.R. Efficient water use in crop production: research or re-search? In: Taylor H.M, Jordan W.R, Sinclair T.R, editors. Limitations to efficient water use in crop production. American Society of Agronomy; Madison, WI: 1983. pp. 1–27. [Google Scholar]
- Tao H.B, Brueck H, Dittert K, Kreye C, Lin S, Sattelmacher B. Growth and yield formation of rice (Oryza sativa L.) in the water-saving ground cover rice production system (GCRPS). Field Crops Research. 2006;95:1–12. [Google Scholar]
- The Arabidopsis Genome Initiative. Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature. 2000;408:796–815. doi: 10.1038/35048692. doi:10.1038/35048692 [DOI] [PubMed] [Google Scholar]
- Thumma B.R, Naidu B.P, Chandra A, Cameron D.F, Bahnisch L.M, Liu C. Identification of causal relationships among traits related to drought resistance in Stylosanthes scabra using QTL analysis. J. Exp. Bot. 2001;52:203–214. doi:10.1093/jexbot/52.355.203 [PubMed] [Google Scholar]
- Turner N.C. Sustainable production of crops and pastures under drought in a Mediterranean environment. Ann. Appl. Biol. 2004a;144:139–147. doi:10.1111/j.1744-7348.2004.tb00327.x [Google Scholar]
- Turner N.C. Agronomic options for improving rainfall-use efficiency of crops in dryland farming systems. J. Exp. Bot. 2004b;55:2413–2425. doi: 10.1093/jxb/erh154. doi:10.1093/jxb/erh154 [DOI] [PubMed] [Google Scholar]
- Turner N.C, Asseng S. Productivity, sustainability, and rainfall-use efficiency in Australian rainfed Mediterranean agricultural systems. Aust. J. Agric. Res. 2005;56:1123–1136. doi:10.1071/AR05076 [Google Scholar]
- UNIS 2000 Secretary General address to Developing Countries ‘South Summit’, UN Information Service Press Release, 13 April 2000. (See www.unis/unvienna.org/unis/pressrels/2000/sg2543.html).
- Wallace J.S. Increasing agricultural water use efficiency to meet future food production. Agric. Ecosyst. Environ. 2000;82:105–119. doi:10.1016/S0167-8809(00)00220-6 [Google Scholar]
- Wallace J.S, Gregory P.J. Water resources and their use in food production systems. Aquat. Sci. 2002;64:363–375. doi:10.1007/PL00012592 [Google Scholar]
- Wild A. Cambridge University Press; Cambridge, UK: 2003. Soils, land and food: managing the land during the twenty-first century. [Google Scholar]
- Woodward J. Some thoughts and experiments concerning vegetation. Phil. Trans. R. Soc. 1699;24:193–227. [Google Scholar]
- WRI 2005 World Resources Institute: Freshwater resources 2005. See http://earthtrends.wri.org/pdf_library/data_tables/wat2_2005.pdf
- Xu L.K, Hsiao T.C. Predicted versus measured photosynthetic water-use efficiency of crop stands under dynamically changing field environments. J. Exp. Bot. 2004;55:2395–2411. doi: 10.1093/jxb/erh271. doi:10.1093/jxb/erh271 [DOI] [PubMed] [Google Scholar]
- Yakir D, Israeli Y. Reduced solar irradiance effects on net primary productivity (NPP) and the d13C and d18O values in plantations of Musa sp. Musaceae. Geochim. Cosmochim. Acta. 1995;59:2149–2151. doi:10.1016/S0016-7037(99)80010-6 [Google Scholar]
- Yang J.C, Zhang J.H. Grain filling of cereals under soil drying. New Phytol. 2006;169:223–236. doi: 10.1111/j.1469-8137.2005.01597.x. doi:10.1111/j.1469-8137.2005.01597.x [DOI] [PubMed] [Google Scholar]
- Yang J.C, Zhang J.H, Liu L.J, Wang Z.Q, Zhu Q.S. Carbon remobilization and grain filling in Japonica/Indica hybrid rice subjected to postanthesis water deficit. Agron. J. 2002;94:102–109. [Google Scholar]
- Yang X, Liang Z, Lu C. Genetic engineering of the biosynthesis of glycinebetaine enhances photosynthesis against high temperature stress in transgenic tobacco plants. Plant Physiol. 2005;138:2299–2309. doi: 10.1104/pp.105.063164. doi:10.1104/pp.105.063164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Nobel P.S. Dependency of ci/ca and leaf transpiration efficiency on the vapour pressure deficit. Aust. J. Plant Physiol. 1996;23:561–568. [Google Scholar]
- Zhang J, Nguyen H.T, Blum A. Genetic analysis of osmotic adjustment in crop plants. J. Exp. Bot. 1999;50:291–302. doi:10.1093/jexbot/50.332.291 [Google Scholar]
- Zhang X.Y, Chen S.Y, Liu M.Y, Pei D, Sun H.Y. Improved water use efficiency associated with cultivars and agronomic management in the North China Plain. Agron. J. 2005;97:783–790. doi:10.2134/agronj2004.0194 [Google Scholar]