Abstract
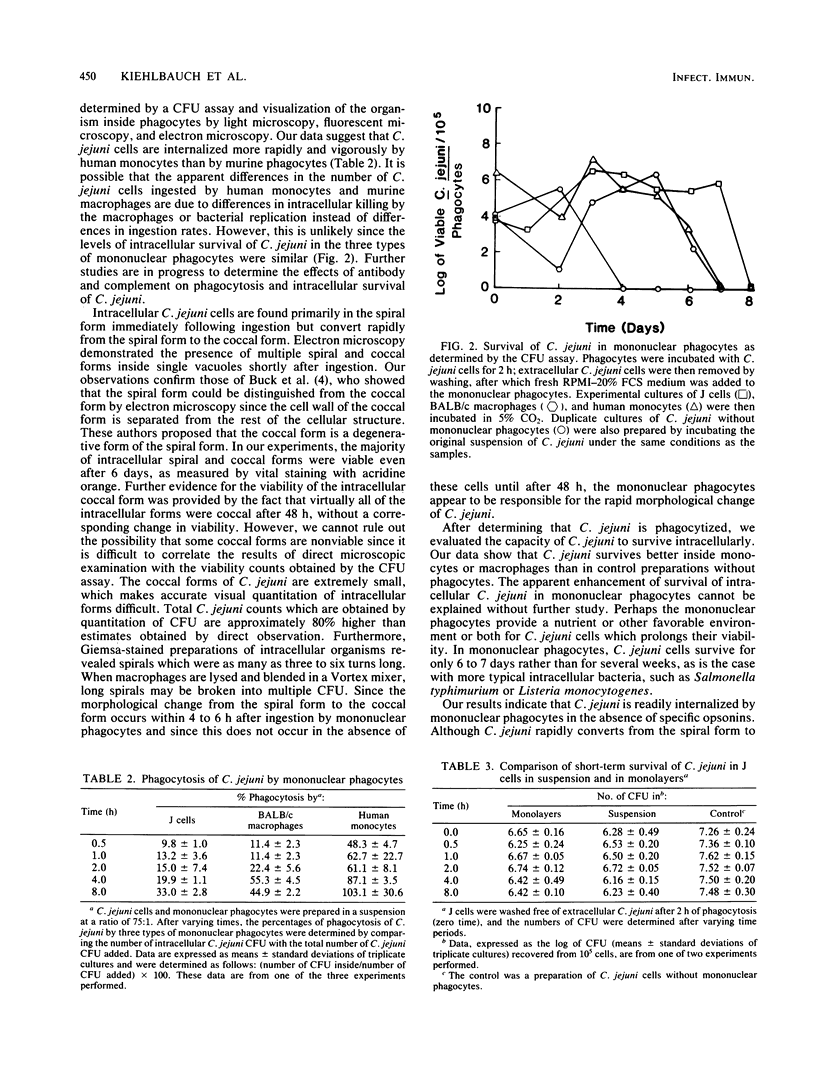
In vitro phagocytosis and intracellular survival of Campylobacter jejuni strain 2964 in mononuclear phagocytes were studied. The following three types of mononuclear phagocytes were used: a J774G8 peritoneal macrophage line derived from BALB/c mice, resident BALB/c peritoneal macrophages, and human peripheral blood monocytes. When C. jejuni and mononuclear phagocytes were combined at a ratio of 75:1, light microscopy, fluorescent microscopy, and electron microscopy all indicated that C. jejuni cells were readily phagocytized. The majority of C. jejuni cells were spirals immediately following ingestion and were rapidly converted to the coccal form within 4 to 8 h. Conversion from the spiral form to the coccal form was complete in the presence of phagocytes within 96 h. In control preparations without phagocytes, conversion began after 24 h and was complete after 48 h. The extent of phagocytosis over time was determined by observing Giemsa-stained preparations and counting the number of intracellular bacterial colony-forming units after removal of extracellular C. jejuni. Human monocytes ingested C. jejuni more rapidly and vigorously than murine macrophages. Intracellular survival of C. jejuni was examined by measuring the number of C. jejuni colony-forming units associated with phagocytes after phagocytosis for 2 h and removal of extracellular bacteria. C. jejuni survived intracellularly for up to 6 to 7 days.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blaser M. J., Duncan D. J., Warren G. H., Wang W. L. Experimental Campylobacter jejuni infection of adult mice. Infect Immun. 1983 Feb;39(2):908–916. doi: 10.1128/iai.39.2.908-916.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaser M. J., Wells J. G., Feldman R. A., Pollard R. A., Allen J. R. Campylobacter enteritis in the United States. A multicenter study. Ann Intern Med. 1983 Mar;98(3):360–365. doi: 10.7326/0003-4819-98-3-360. [DOI] [PubMed] [Google Scholar]
- Buck G. E., Parshall K. A., Davis C. P. Electron microscopy of the coccoid form of Campylobacter jejuni. J Clin Microbiol. 1983 Aug;18(2):420–421. doi: 10.1128/jcm.18.2.420-421.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böyum A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand J Clin Lab Invest Suppl. 1968;97:77–89. [PubMed] [Google Scholar]
- Caldwell M. B., Walker R. I., Stewart S. D., Rogers J. E. Simple adult rabbit model for Campylobacter jejuni enteritis. Infect Immun. 1983 Dec;42(3):1176–1182. doi: 10.1128/iai.42.3.1176-1182.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang K. P. Hamster peritoneal macrophages in vitro: substratum adhesion, spreading, phagocytosis and phagolysosome formation. In Vitro. 1978 Aug;14(8):663–674. doi: 10.1007/BF02616163. [DOI] [PubMed] [Google Scholar]
- Corbeil L. B., Corbeil R. R., Winter A. J. Bovine venereal vibriosis: activity of inflammatory cells in protective immunity. Am J Vet Res. 1975 Apr;36(4 PT1):403–406. [PubMed] [Google Scholar]
- Duffy M. C., Benson J. B., Rubin S. J. Mucosal invasion in campylobacter enteritis. Am J Clin Pathol. 1980 May;73(5):706–708. doi: 10.1093/ajcp/73.5.706. [DOI] [PubMed] [Google Scholar]
- Field L. H., Underwood J. L., Pope L. M., Berry L. J. Intestinal colonization of neonatal animals by Campylobacter fetus subsp. jejuni. Infect Immun. 1981 Sep;33(3):884–892. doi: 10.1128/iai.33.3.884-892.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hébert G. A., Hollis D. G., Weaver R. E., Lambert M. A., Blaser M. J., Moss C. W. 30 years of campylobacters: biochemical characteristics and a biotyping proposal for Campylobacter jejuni. J Clin Microbiol. 1982 Jun;15(6):1065–1073. doi: 10.1128/jcm.15.6.1065-1073.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keeler R. F., Ritchie A. E., Bryner J. H., Elmore J. The preparation and characterization of cell walls and the preparation of flagella of Vibrio fetus. J Gen Microbiol. 1966 Jun;43(3):439–454. doi: 10.1099/00221287-43-3-439. [DOI] [PubMed] [Google Scholar]
- LUFT J. H. Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol. 1961 Feb;9:409–414. doi: 10.1083/jcb.9.2.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazdins J. K., Koech D. K., Karnovsky M. L. Oxidation of glucose by mouse peritoneal macrophages: a comparison of suspensions and monolayers. J Cell Physiol. 1980 Nov;105(2):191–196. doi: 10.1002/jcp.1041050202. [DOI] [PubMed] [Google Scholar]
- Longfield R., O'Donnell J., Yudt W., Lissner C., Burns T. Acute colitis and bacteremia due to Campylobacter fetus. Dig Dis Sci. 1979 Dec;24(12):950–953. doi: 10.1007/BF01311952. [DOI] [PubMed] [Google Scholar]
- McCoy E. C., Doyle D., Burda K., Corbeil L. B., Winter A. J. Superficial antigens of Campylobacter (Vibrio) fetus: characterization of antiphagocytic component. Infect Immun. 1975 Mar;11(3):517–525. doi: 10.1128/iai.11.3.517-525.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralph P., Prichard J., Cohn M. Reticulum cell sarcoma: an effector cell in antibody-dependent cell-mediated immunity. J Immunol. 1975 Feb;114(2 Pt 2):898–905. [PubMed] [Google Scholar]
- Ruiz-Palacios G. M., Escamilla E., Torres N. Experimental Campylobacter diarrhea in chickens. Infect Immun. 1981 Oct;34(1):250–255. doi: 10.1128/iai.34.1.250-255.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]