Abstract
Abuse of anabolic–androgenic steroids (AAS) for improving physical performance is associated with serious, sometimes fatal, adverse effects. The aim of the present work was to investigate the effects of AAS on the cardiac structure and the plasma lipoprotein profile isolated and in combination with exercise. Transgenic mice with a human lipaemic phenotype (expressing cholesteryl ester transfer protein on the LDL receptor knockout background) were used in this study. Sedentary and exercised mice (treadmill running, five times per week for 6 weeks) were treated with mesterolone (2 μg/g body weight) or vehicle (control-C) in the last 3 weeks. Four groups were compared: (i) exercise + mesterolone (Ex-M), (ii) exercise + vehicle (Ex-C), (iii) sedentary + mesterolone (Sed-M) and (iv) sedentary + vehicle (Sed-C). Arterial blood pressure and body mass increased in all groups along time, but Sed-M reached the highest values and Ex-C the lowest. Treatment with mesterolone increased total cholesterol, triglyceride, low-density lipoprotein cholesterol (LDL-c) and very LDL-c (VLDL-c) plasma levels. However, exercise blunted some of these deleterious effects by increasing high-density lipoprotein cholesterol and decreasing LDL-c, VLDL-c and triglycerides. Exercise training induced beneficial effects, such as physiological cardiomyocyte hypertrophy, increase in myocardial circulation and decrease in cardiac interstitium. However, mesterolone impaired such physiological gains and in addition increased troponin T plasma levels both in sedentary and exercised mice. Thus, while mesterolone induced pro-atherogenic lipoprotein profile and pathogenic cardiac hypertrophy, exercise counteracted these effects and modified favourably both the lipoprotein profile and the cardiac remodelling induced by mesterolone.
Keywords: cardiac interstitium, cardiomyocytes, lipid profile, mesterolone, transgenic murine model, ventricular hypertrophy
Androgenic–anabolic steroids (AASs) are synthetic derivatives of the male hormone testosterone. The therapeutic use of AASs is indicated in endocrine dysfunction of the testes, age-related and HIV-related muscle wasting (Vermeulen 2001). From clinical trials, it seems likely that appropriate prescription of AASs may have positive anabolic effects on cachexia associated with HIV, cancer, burns, renal and hepatic failure and anaemia associated with leukaemia or kidney failure, which overcome the threat of side effects (for review, see Basaria et al. 2001).
The non-therapeutic use of AASs is a common practice among athletes for improving physical performance given its putative capacity of enhancing muscle mass and strength (Hartgens & Kuipers 2004). Generally administered in supraphysiological doses, the abuse of AASs by healthy athletes has been connected with the incidence of cardiovascular disease (CVD), such as development of hypertension, cardiomyopathy, atrial fibrillation, cerebrovascular accident, myocardial infarction, disturbances in haemostatic system, ventricular thrombosis and systemic embolism and acute heart failure (Dickerman et al. 1996; Sullivan et al. 1998). In addition, it has been shown that testosterone may affect negatively the recovery of postischaemic myocardial infarction by activating pro-inflammatory cytokine production (Wang et al. 2005).
Studies have also shown that the use of AASs induces atherogenic lipoprotein profile with a decrease in high-density lipoprotein cholesterol (HDL-c) and apolipoprotein Al and an increase in low-density lipoprotein cholesterol (LDL-c) plasma levels (Glazer 1991). On the other hand, exercise training has been associated with a reduced risk of CVD, possibly because it leads to an improvement of the lipoprotein profile (LaRosa 1992).
On the other hand, experimental and clinical evidence suggests androgen deficiency as a potential risk factor for CVD (for review, see Winkler 1996). Therefore, testosterone deficiency in mice resulted in increased plasma levels of apolipoprotein B-containing lipoproteins [very LDL (VLDL) and LDL] and decreased HDL-c, but transgenic mice that express the cholesteryl ester transfer protein (CETP) had such effects significantly attenuated (Casquero et al. 2006). This transgenic mice model is interesting because its lipoprotein profile is closer to that of the humans than the wild-type mouse. Contrary to humans, wild-type mice show very low levels of LDL and high levels of HDL. These differences are attributed to the lack of expression of the CETP, which increases HDL-c, and to the high number of LDL receptors, which reduce plasma LDL-c in the wild-type mice. Thus, by introducing the expression of the CETP gene and reducing the expression of the LDL receptor (LDLr) gene, the lipaemic phenotype of these transgenic mice resembles those of humans (Cazita et al. 2003; Casquero et al. 2006).
In the present study, the effects of mesterolone (17-beta-hydroxy-1-alpha-methyl-5-alpha-androstan-3-one, C20H32O2), a synthetic steroid with anabolic and androgenic activities, were evaluated for its potency in inducing cardiac structure remodelling and altering lipoprotein profile in this transgenic murine model. In addition, we verified whether exercise training modulates such effects.
Materials and methods
Animal protocols
The experimental protocol was approved by the university’s Committee for Ethics in Animal Experimentation (CEEA/UNICAMP) in accordance with the Guide for the Care and Use of Laboratory Animal published by the US National Institutes of Health (NIH publication no. 85-23, revised 1996). The transgenic mice used in this study were cross-bred and maintained in the Department of Physiology and Biophysics (Institute of Biology, UNICAMP) for 10 years. Mice heterozygous for the human CETP transgene and for the LDL-receptor null allele (CETP+/−LDLr−/+) have been described elsewhere (Cazita et al. 2003; Casquero et al. 2006). The plasma CETP activity, expressed as % cholesteryl ester transferred from LDL to HDL, in this model is 36.6 ± 1.6 (n = 24) while wild-type mice are completely deficient of circulating CETP. The mice were housed in a temperature-controlled room (22 ± 1 °C), humidity of 55–65%, 12 h light/dark cycle and had free access to water and food standard chow (Nuvilab, Colombo, PR, Brazil).
Experimental protocol
Twenty-four adult male mice aged 2 months were divided into four groups of six animals: the control groups, sedentary (Sed-C) and exercised (Ex-C), received gum arabic [2 μg/g body mass (BM) via orogastric] during the last 3 weeks of training or sedentary period and the experimental groups, sedentary (Sed-M) and exercised (Ex-M), received the AAS mesterolone (Proviron™; Schering, Sao Paulo, SP, Brazil; 2 μg/g BM via orogastric) during the last 3 weeks of training or sedentary period. Gum arabic was used as vehicle given its non-toxic and pro-absorptive effects in small intestine (Codipilly et al. 2006). Exercised mice were allowed to adapt to treadmill running for 1 week prior to the use of the experimental protocol. This consisted of low to moderate level exercise of the treadmill running carried out daily for 5 days (15 m/min for 20 min/day). After adaptation, exercised mice were subjected to 6 weeks of intensive exercise training (treadmill running), 5 days a week, as scheduled in Table 1 (adapted from Smolka et al. 2000). Blood pressure (BP) and BM were monitored weekly prior to any experimental procedure. All animals were habituated to the BP measurement device for 7 days before the onset of the experimental period. Blood pressure was measured weekly through the non-invasive computerized tail-cuff plethysmography (RTBP 1000; Kent Scientific Co., Litchfield, CT, USA) in conscious mice.
Table 1.
Exercise (6 weeks) and anabolic–androgenic steroid (AAS) or vehicle administration (3 weeks) protocol
| Weeks | Velocity (m/min) | Duration (min) |
|---|---|---|
| 1 | 12.42 | 20 |
| 2 | 14.70 | 30 |
| 3 | 16.68 | 45 |
| 4–6* | 17.04 | 60 |
AAS or gum arabic (vehicle) administered orally by gavage at 10.00 am (Monday, Wednesday and Friday) in the last 3 weeks.
At the end of the experimental period, overnight fasted mice were deeply anaesthetized with a 1:1 mixture of ketamine chloride (Dopalen, 100 mg/kg of animal) and xylazine chloride (Anasedan, 10 mg/kg; 2 μl/mg BM, i.p.; both anaesthetics from Vetbrands, Jacarei, SP, Brazil). Blood samples were taken from the right atrium of the heart for plasma lipid analyses. Hearts were removed and the atria were separated from the ventricles and the left ventricle with the interventricular septum (LV) was weighed and then cut into two halves, put with the sectioned face down and then sectioned perpendicular to the ventral face at random. The LV fragments fixed with freshly prepared 4% (w/v) formaldehyde in 0.1 M phosphate buffer, pH 7.2 for 48 h, were then routinely processed until embedding in Histosec (Merck, Rio de Janeiro, RJ, Brazil). Sections (3 μm thick) stained with haematoxylin–eosin and Masson’s trichrome were used for stereology. The LV mass/BM ratio was used as an index of cardiac hypertrophy.
Plasma lipid and troponin T analysis
Levels of total cholesterol (TC), triglycerides (TG), HDL in plasma were determined by enzymatic assays using commercially available kits (Roche, São Paulo, SP, Brazil) according to the instructions given by the manufacturer's protocol. Very low-density lipoprotein cholesterol was estimated as TG/5 and LDL-c by the Friedewald’s formula (Friedewald et al. 1972). Troponin T (TnT) was determined by one-step enzyme immunoassay based on third-generation ECLIA assay on an Elecsys 2010 analyzer (Roche Diagnostics, Basel, Switzerland).
Stereology
A video microscopic system composed of a Leica DMRBE microscope, Kappa videocamera (Gleichen, Germany) and Sony Trinitron monitor (Pencoed, UK) was used. For stereology, the myocardium of the LV consisting of cardiomyocytes (cmy) plus cardiac interstitium (int, the latter consisting of connective tissue and intramyocardial vessels, ve) was analysed. Ten random histological sections were observed from the various LV fragments of each animal (n = 5 mice/group) and 10 random microscopic fields per animal were analysed blindly with a planachromatic objective 40× (Leica, Wetzlar, Germany) by moving the stage of the microscope. The stereological analysis used a test system with 36 test points and a known area. The volume densities of the structures were estimated as: Vv [structure] = Pp [structure]/PT, where Pp is the number of points that hit the structure and PT is the total number of test points (36 in the present study) contained in the frame. The mean cross-sectional area of the cardiomyocyte A(cmy) was estimated as: A(cmy) = Vv(cmy)/2·QA(cmy), where Vv is the volume density of the cardiomyocytes and QA is the number of cardiomyocyte nuclei within the frame whose area is known respecting the ‘forbidden line’ (Gundersen 1977). The LV intramyocardial vascularization was analysed by the Vv(ve)/Vv(cmy) ratio (Mandarim-de-Lacerda 2003).
Statistical analysis
The data were tested for deviations from Gaussian distribution using the Kolmogorov–Smirnov test (graph pad prism version 5.0; GraphPad Software, San Diego, CA, USA) and expressed as mean ± SEM. The statistical comparison among the control and treated groups was determined using one-way anova (pair-wise) followed by the post hoc test of Tukey. In addition, two-way anova was used where appropriate to determine how mesterolone treatment or exercise training affected the results, and whether there was an interaction between these two conditions. A P-value of 0.05 indicated statistical significance.
Results
Blood pressure and body mass
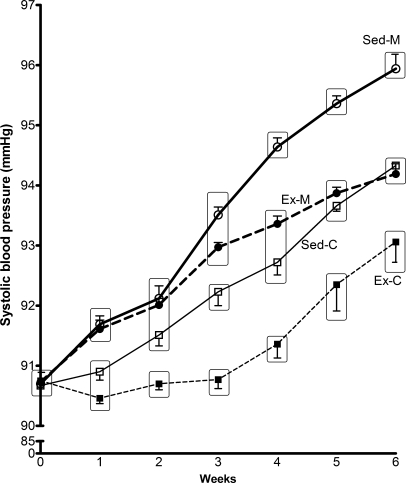
The weekly variation in BP and BM during the 6-week period of experimentation is shown in Figures 1 and 2 respectively. Blood pressure tended to increase along the trial period, particularly from the third week onwards in the mesterolone groups (highest BP values in both the Sed-M and Ex-M groups). The BP values at the sixth week were significantly different from the initial values in the same group (P < 0.05). It is noteworthy that exercise training delayed the rhythm of increase of BP in both the mesterolone-treated and vehicle-treated groups. Despite the significant difference in BP between Ex-C and Sed-M (P < 0.05), the little variation was not relevant from the physiological point of view.
Figure 1.
Caudal blood pressure (systolic-like) evolution in control groups sedentary (Sed-C) and exercised (Ex-C) treated with gum arabic (vehicle), and the experimental groups sedentary (Sed-M) and exercised (Ex-M) treated with mesterolone of CETP+/−LDLr−/+ transgenic mice. When the groups were put together (into a rectangle), they did not show statistically significant difference, but among the rectangles the difference was significant (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
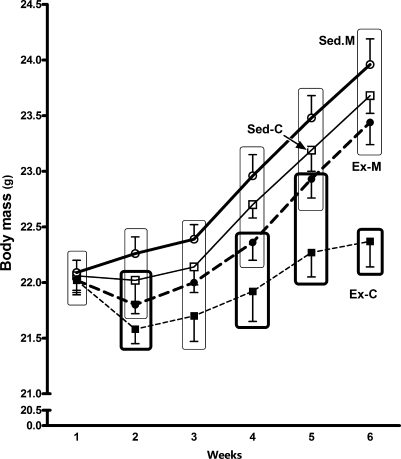
Figure 2.
Body mass (BM in grams) evolution during the 6 week period in the gum arabic- (vehicle) and mesterolone-treated sedentary (Sed-C and Sed-M) or exercised (Ex-C and Ex-M) CETP+/−LDL−/+ transgenic mice. Sed-M exhibited the highest weight gain whereas Ex-C exhibited the lowest (Sed-M = Sed-C = Ex-M > Ex-C). Groups inside rectangles not inter-sectioned to each other present a week-by-week significant difference as follows: Ex-C ≠ Sed-M (in all weeks, except in the third); Ex-C ≠ Sed-C (fourth, fifth and sixth); Ex-C ≠ Ex-M (sixth) (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
Figure 2 shows that BM gain was lower in exercised than in sedentary mice. Sed-M showed better week-by-week BM increase and the highest BM gain at the end of the sixth week. Because in Ex-C, BM was maintained practically at a constant level since the beginning of the experimental period, a slight, but statistically significant difference was found comparing Sed-M vs. Ex-C (P < 0.05). However, the effect of exercise in maintaining low BM gain was not enough to abolish the gain promoted by mesterolone and hence there is no significant difference between Sed-M and Ex-M.
Plasma lipids
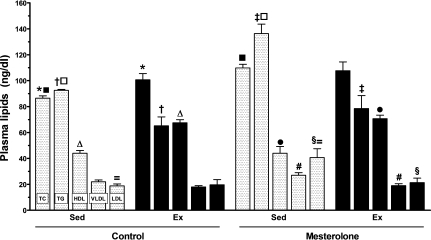
The plasma levels of TC, TG and cholesterol distribution in lipoprotein fractions (HDL and LDL) are shown in Figure 3. Considering TC plasma levels in the Sed-C group as reference (86.5 ± 1.8), there was a significant increase in Sed-M (P = 0.00004) and Ex-C (P = 0.02). Compared with the Sed-C group (92.5 ± 0.7 mg/dl), the TG plasma levels in the Sed-M group were increased by 38% (P < 0.0001), in the Ex-M group decreased by 15% (P < 0.0001) and in the Ex-C group decreased by 30% (P = 0.003). These findings indicated that a beneficial effect of the exercise training on the plasma lipids was sufficient to counteract the adverse mesterolone effect.
Figure 3.
Plasmatic lipid and lipoprotein levels in the CETP+/−LDLr−/+ transgenic mice in control groups treated with gum arabic (vehicle), sedentary (Sed-C) or exercised (Ex-C) and matched groups treated with mesterolone (Sed-M and Ex-M). The same symbol over the bar indicates significant differences among groups (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
The plasma levels of HDL-c were not different between Sed-M and Sed-C, but were elevated in Ex-C (P = 0.0002) and Ex-M (P < 0.001), indicating the predominant effect of the exercise training in controlling HDL-c levels in mice administered mesterolone.
The plasma levels of LDL-c were increased in Sed-M as compared with Sed-C (P < 0.01) and in Sed-M as compared with Ex-M (P = 0.03), indicating the side effect of mesterolone and the benefit of exercise.
The levels of VLDL-c were lower in Ex-C (approximately 40% reduced) and Ex-M (approximately 30% reduced) compared with Sed-C group (P < 0.005), and were decreased approximately by 40% in Ex-M compared with Sed-M (P < 0.01). Very low-density lipoprotein cholesterol did not differ between exercised groups.
Cardiac hypertrophy
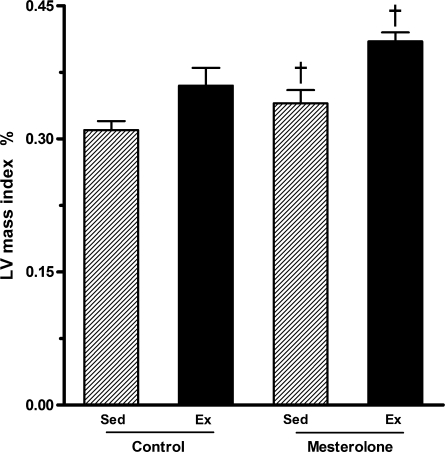
Exercise alone (Ex-C) or mesterolone alone (Sed-M) promoted a trend for increasing left ventricle (LV) mass index in comparison with Sed-C but the differences were in the borderline of statistical significance (P = 0.054). In contrast, the LV mass index increased by 25% in Ex-M compared with Sed-M (P = 0.03). These results show that exercise associated with mesterolone had an additive effect on LV mass index (Figure 4).
Figure 4.
Left ventricular mass index in exercised and sedentary CETP+/−LDLr−/+ transgenic mice submitted to mesterolone or gum arabic (vehicle) orogastric administration. There was significant difference between Sed-M vs. Ex-M (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
Stereology
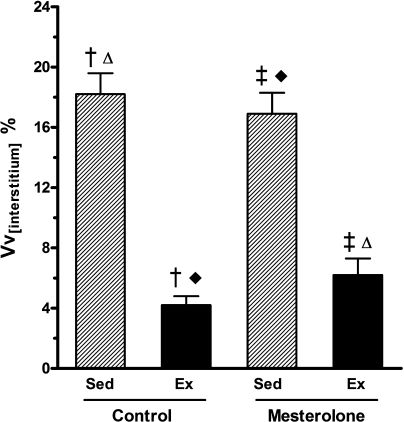
The volume density of the interstitium, Vv(int), decreased by 65% in Ex-M and 80% in Ex-C when compared with their sedentary counterparts (P < 0.05). Exercise training per se decreased the Vv(int) in control mice treated with vehicle whereas increased in the sedentary mice treated with mesterolone (Figure 5). These findings show that exercise training is able to reduce the cardiac interstitium (collagen fibre increment).
Figure 5.
Volume density of left ventricle interstitium (Vv(int)) from exercised and sedentary CETP+/−LDLr−/+ transgenic mice treated orally with mesterolone and gum arabic (vehicle). There were significant differences among groups as follows: Sed-C vs. Ex-C and Sed-M vs. Ex-M (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
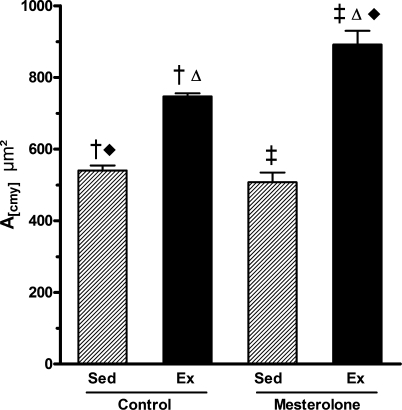
Figures 6 and 7 show the cardiomyocyte area measurements and morphology. Compared with Sed-C mice, the largest cardiomyocytes were observed in the Ex-M group (A(cmy) where there was a 65% area increase (P < 0.01), followed by the Ex-C group (with 40% increase, P < 0.02) [differences were significant between Sed-C vs. Ex-C; Sed-C vs. Ex-M; Sed-M vs. Ex-M and Ex-C vs. Ex-M (P < 0.05) (Figure 6)]. These findings indicated that exercise training per se causes cardiomyocyte hypertrophy and exercise training combined with administration of mesterolone enhances this hypertrophy. This was confirmed by the two-way anova (P = 0.03; Figure 6) and histological observations (Figure 7).
Figure 6.
Left ventricle cardiomyocyte cross-sectional area (A(cmy)) from exercised and sedentary CETP+/−LDLr−/+ transgenic mice treated with mesterolone or gum arabic (vehicle). There were significant differences among groups as follows: Sed-C < Ex-C, Ex-C < Ex-M and Ex-M > Sed-M (P < 0.05). One-way and two-way anova followed by Tukey test. Data are expressed as mean ± SEM.
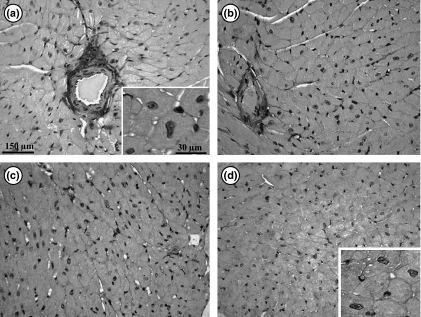
Figure 7.
Photomicrographs showing the left ventricle myocardium in transgenic mice. (a) Ex-M: exercised mice treated with mesterolone; (b) Ex-C: exercised animals treated with gum arabic (vehicle); (c) Sed-M: sedentary animals treated with mesterolone; (d) Sed-C sedentary mice treated with vehicle. Note that qualitatively cardiomyocytes are higher in size in exercised (a, b) and smaller in sedentary (d, c) groups. The insets depict a high magnification of cardiomyocytes of Ex-M (a) and Se-C (d).
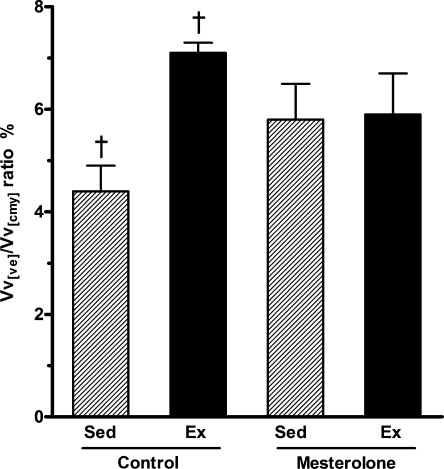
The intramyocardial vascularization (Figure 8) was significantly greater in the Ex-C group compared with the Sed-C group (by 40%, P < 0.05). However, no statistical differences were observed in intramyocardial blood vessels of Ex-M and Sed-M groups (Figure 8). Thus, mesterolone treatment blunted the benefit gained by exercise.
Figure 8.
Myocardial vascularization in the left ventricle from exercised and sedentary CETP+/−LDLr−/+ transgenic mice treated with mesterolone and gum arabic (vehicle). There was significant difference between Sed-C vs. Ex-C (P < 0.05). One-way and two-way anova followed by Tukey test. Data are expressed as mean ± SEM.
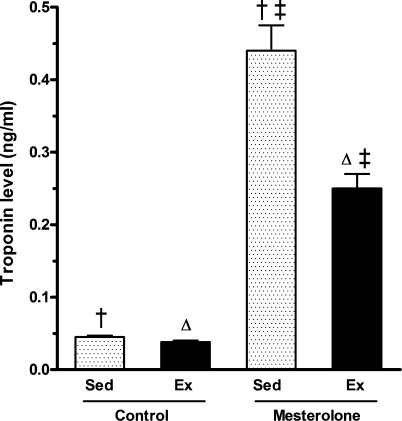
The plasma levels of TnT were lowest in the Ex-C group with no significant difference with that of the Sed-C (Figure 9). The highest value was achieved in the Sed-M group (0.44 ng/ml). In Ex-M mice, the TnT value was 0.25 ng/ml, indicating a 40% reduction in TnT release from cardiomyocytes promoted by treadmill running (P < 0.001).
Figure 9.
Troponin T (TnT) plasmatic level in the CETP+/−LDLr−/+ transgenic mice in control groups (Sed-C) and exercised (Ex-C) treated with gum arabic (vehicle) and the experimental groups sedentary (Sed-M) and exercised (Ex-M) treated with mesterolone. The same symbol over the bar indicates significant differences among groups (P < 0.05). One-way anova followed by Tukey test. Data are expressed as mean ± SEM.
Discussion
In this study, transgenic mice with a human lipaemic phenotype (CETP+/−LDLr−/+) were used to evaluate the effects of mesterolone treatment combined with or without an aerobic exercise training protocol, on the cardiovascular system, specifically on BP, plasma lipoprotein profile and cardiac remodelling. The use of testosterone or AASs is a well-recognized risk factor for hypertension, disturbances in lipid profile and accelerated coronary artery disease (Alen & Rahkila 1984; Ganten et al. 1989; Hurley et al. 1984; Rockhold 1993). In the present study, compared with vehicle, mesterolone-treated sedentary mice showed significant elevation of the TC, TG, LDL-c and VLDL-c plasma levels. On the other hand, the exercise training programme applied to these mice increased the levels of HDL-c and decreased VLDL-c (the precursor of LDL-c) and TG significantly, both in mice treated with vehicle or mesterolone. The significant difference in the BM gain and BP produced both by mesterolone and exercise per se taking as reference Sed-C is likely not relevant from the physiological point of view.
Regular and moderate exercise training have been associated with a reduced risk of CVD, partially because of an improvement in the lipoprotein profile (LaRosa 1992). However, the amount of exercise training required to obtain benefits is debatable (Blair et al. 2004). In general, only high-intensity exercise training or long-term endurance exercise programme are able to improve overall lipoprotein profile, including elevation of HDL-c and reduction in TG levels in humans (Seals et al. 1984; Kraus et al. 2002; Halverstadt et al. 2007). On the other hand, studies on the exercise-mediated improvement of lipoprotein profile in mice and rats should be observed with prudence if the aim is to compare with humans, mainly because differences exist in the expression of proteins involved in transport and tissue uptake of lipoproteins. In this regard, the animal model studied here presented a lipid profile akin to the humans and so lipoprotein response to exercise training is expected to be more feasible in comparison with that in humans. The exercise programme (6 weeks, 5 times/week and 60 min/day) with velocity of 17.4 m/min in the last 3 weeks is considered high aerobic to animals and comparable to long-term and high-endurance exercise in humans, considering the life span of both.
Clinical reports of misuse of AASs by athletes and studies in experimental animals have shown the occurrence of deleterious structural myocardial alterations and heart hypertrophy (Bauman et al. 1988; Ganten et al. 1989; Nieminen et al. 1996) (for review see Hartgens & Kuipers 2004), including irreversible changes in the myocardium, such as concentric LV hypertrophy, even after discontinuing the AAS intake (Urhausen et al. 2004). Treatment of rats with supraphysiological doses of AASs induced pathological myocardial hypertrophy, and when combined with exercise, these steroids reduced the beneficial effects of exercise on LV hypertrophy and cardiac circulation (Tagarakis et al. 2000a,b; Woodiwiss et al. 2000).
In the current study, mesterolone alone (Sed-M) promoted only slight changes in the cardiac structure, i.e. a borderline trend towards LV hypertrophy (P = 0.054). On the other hand, the exercise training induced favourable cardiac remodelling either in mice treated with mesterolone or vehicle. This is evidenced by a marked reduction in the cardiac interstitium (more prominent in Ex-C) and enlargement of cardiomyocyte size. Increased levels of circulating TnT, a marker of myocardium lesion, confirm the deleterious effect of mesterolone in sedentary mice and exercise training attenuated this adverse effect of mesterolone on the cardiac integrity. In addition, the exercise training enhanced significantly the cardiac vascularization in the Ex-C and, to a lesser extent, in Ex-M groups (compared with Sed-C). This probably represents a response to the increased myocardial oxygen demand imposed by physical activity. Similar effects of exercise training were observed in a previous study in ovariectomized rats (Marques et al. 2006). This increase in cardiac vascularization may be of importance in the case of ischaemic events. Mesterolone treatment of exercised mice suppressed the amelioration of the benefits of physiologic hypertrophy, the improvement of the myocardial circulation and part of the reduction in interstitium induced by the exercise training programme.
Therefore, our findings show the strong adverse effects of mesterolone on plasma lipid and lipoprotein metabolism, which were prevented or attenuated by exercise training in this murine model. On the other hand, our data also show that exercise per se induces beneficial changes in cardiac remodelling and myocardial vascular supply, all of which are totally or partially impaired by the mesterolone use.
Acknowledgments
This study was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) (Proc. 04/13767-9) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Proc. 522131/95-6). K.F. is a PhD student at the Departamento de Farmacologia, Faculdade de Ciências Médicas, Universidade Estadual de Campinas (UNICAMP) granted with a scholarship from FAPESP (Proc. 04/13768-5). The authors thank the Departamento de Anatomia for the use of its facility, Mr. Lécio D. Teixeira from the Departamento de Fisiologia e Biofísica for animal care and Dr Marta Krieger from the Departamento de Fisiologia e Biofísica for laboratory facility.
References
- Alen M, Rahkila P. Reduced high-density lipoprotein-cholesterol in power athletes: use of male sex hormone derivates, an atherogenic factor. Int. J. Sports Med. 1984;5:341–342. doi: 10.1055/s-2008-1025929. [DOI] [PubMed] [Google Scholar]
- Basaria S, Wahlstrom JT, Dobs AS. Clinical review 138: anabolic–androgenic steroid therapy in the treatment of chronic diseases. J. Clin. Endocrinol. Metab. 2001;86:5108–5117. doi: 10.1210/jcem.86.11.7983. [DOI] [PubMed] [Google Scholar]
- Bauman DH, Richerson JT, Britt AL. A comparison of body and organ weights, physiologic parameters, and pathologic changes in target organs of rats given combinations of exercise, anabolic hormone, and protein supplementation. Am. J. Sports Med. 1988;16:397–402. doi: 10.1177/036354658801600416. [DOI] [PubMed] [Google Scholar]
- Blair SN, LaMonte MJ, Nichaman MZ. The evolution of physical activity recommendations: how much is enough? Am. J. Clin. Nutr. 2004;79:913S–920S. doi: 10.1093/ajcn/79.5.913S. [DOI] [PubMed] [Google Scholar]
- Casquero AC, Berti JA, Salerno AG, et al. Atherosclerosis is enhanced by testosterone deficiency and attenuated by CETP expression in transgenic mice. J. Lipid Res. 2006;47:1526–1534. doi: 10.1194/jlr.M600135-JLR200. [DOI] [PubMed] [Google Scholar]
- Cazita PM, Berti JA, Aoki C, et al. Cholesteryl ester transfer protein expression attenuates atherosclerosis in ovariectomized mice. J. Lipid Res. 2003;44:33–40. doi: 10.1194/jlr.m100440-jlr200. [DOI] [PubMed] [Google Scholar]
- Codipilly CN, Teichberg S, Wapnir RA. Enhancement of absorption by gum arabic in a model of gastrointestinal dysfunction. J. Am. Coll. Nutr. 2006;25:307–312. doi: 10.1080/07315724.2006.10719540. [DOI] [PubMed] [Google Scholar]
- Dickerman RD, McConathy WJ, Schaller F, et al. Cardiovascular complications and anabolic steroids. Eur. Heart J. 1996;17:1912. doi: 10.1093/oxfordjournals.eurheartj.a014812. [DOI] [PubMed] [Google Scholar]
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- Ganten U, Schroder G, Witt M, et al. Sexual dimorphism of blood pressure in spontaneously hypertensive rats: effects of anti-androgen treatment. J. Hypertens. 1989;7:721–726. [PubMed] [Google Scholar]
- Glazer G. Atherogenic effects of anabolic steroids on serum lipid levels. A literature review. Arch. Intern. Med. 1991;151:1925–1933. [PubMed] [Google Scholar]
- Gundersen HJ. Notes on the estimation of the numerical density of arbitrary profiles: the edge effect. J. Microsc. 1977;111:219–227. [Google Scholar]
- Halverstadt A, Phares DA, Wilund KR, et al. Endurance exercise training raises high-density lipoprotein cholesterol and lowers small low-density lipoprotein and very low-density lipoprotein independent of body fat phenotypes in older men and women. Metabolism. 2007;56:444–450. doi: 10.1016/j.metabol.2006.10.019. [DOI] [PubMed] [Google Scholar]
- Hartgens F, Kuipers H. Effects of androgenic–anabolic steroids in athletes. Sports Med. 2004;34:513–554. doi: 10.2165/00007256-200434080-00003. [DOI] [PubMed] [Google Scholar]
- Hurley BF, Seals DR, Hagberg JM, et al. High-density-lipoprotein cholesterol in bodybuilders v powerlifters. Negative effects of androgen use. J. Am. Med. Assoc. 1984;252:507–513. [PubMed] [Google Scholar]
- Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N. Engl. J. Med. 2002;347:1483–1492. doi: 10.1056/NEJMoa020194. [DOI] [PubMed] [Google Scholar]
- LaRosa JC. Lipids and cardiovascular disease: do the findings and therapy apply equally to men and women? Womens Health Issues. 1992;2:102–111. doi: 10.1016/s1049-3867(05)80278-6. [DOI] [PubMed] [Google Scholar]
- Mandarim-de-Lacerda CA. Stereological tools in biomedical research. Ann. Braz. Acad. Sci. 2003;75:469–486. doi: 10.1590/s0001-37652003000400006. [DOI] [PubMed] [Google Scholar]
- Marques CM, Nascimento FA, Mandarim-de-Lacerda CA. Exercise training attenuates cardiovascular adverse remodeling in adult ovariectomized spontaneously hypertensive rats. Menopause. 2006;13:87–95. doi: 10.1097/01.gme.0000191209.13115.46. [DOI] [PubMed] [Google Scholar]
- Nieminen MS, Ramo MP, Viitasalo M, et al. Serious cardiovascular side effects of large doses of anabolic steroids in weight lifters. Eur. Heart J. 1996;17:1576–1583. doi: 10.1093/oxfordjournals.eurheartj.a014724. [DOI] [PubMed] [Google Scholar]
- Rockhold RW. Cardiovascular toxicity of anabolic steroids. Annu. Rev. Pharmacol. Toxicol. 1993;33:497–520. doi: 10.1146/annurev.pa.33.040193.002433. [DOI] [PubMed] [Google Scholar]
- Seals DR, Hagberg JM, Hurley BF, et al. Effects of endurance training on glucose tolerance and plasma lipid levels in older men and women. JAMA. 1984;252:645–649. [PubMed] [Google Scholar]
- Smolka MB, Zoppi CC, Alves AA, et al. HSP72 as a complementary protection against oxidative stress induced by exercise in the soleus muscle of rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000;279:R1539–R1545. doi: 10.1152/ajpregu.2000.279.5.R1539. [DOI] [PubMed] [Google Scholar]
- Sullivan ML, Martinez CM, Gennis P, et al. The cardiac toxicity of anabolic steroids. Prog. Cardiovasc. Dis. 1998;41:1–15. doi: 10.1016/s0033-0620(98)80019-4. [DOI] [PubMed] [Google Scholar]
- Tagarakis CV, Bloch W, Hartmann G, et al. Anabolic steroids impair the exercise-induced growth of the cardiac capillary bed. Int. J. Sports Med. 2000a;21:412–418. doi: 10.1055/s-2000-3835. [DOI] [PubMed] [Google Scholar]
- Tagarakis CV, Bloch W, Hartmann G, et al. Testosterone-propionate impairs the response of the cardiac capillary bed to exercise. Med. Sci. Sports Exerc. 2000b;32:946–953. doi: 10.1097/00005768-200005000-00011. [DOI] [PubMed] [Google Scholar]
- Urhausen A, Albers T, Kindermann W. Are the cardiac effects of anabolic steroid abuse in strength athletes reversible? Heart. 2004;90:496–501. doi: 10.1136/hrt.2003.015719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vermeulen A. Androgen replacement therapy in the aging male – a critical evaluation. J. Clin. Endocrinol. Metab. 2001;86:2380–2390. doi: 10.1210/jcem.86.6.7630. [DOI] [PubMed] [Google Scholar]
- Wang M, Tsai BM, Kher A, et al. Role of endogenous testosterone in myocardial proinflammatory and proapoptotic signaling after acute ischemia–reperfusion. Am. J. Physiol. Heart Circ. Physiol. 2005;288:H221–H226. doi: 10.1152/ajpheart.00784.2004. [DOI] [PubMed] [Google Scholar]
- Winkler UH. Effects of androgens on haemostasis. Maturitas. 1996;24:147–155. doi: 10.1016/s0378-5122(96)82004-4. [DOI] [PubMed] [Google Scholar]
- Woodiwiss AJ, Trifunovic B, Philippides M, et al. Effects of an androgenic steroid on exercise-induced cardiac remodeling in rats. J. Appl. Physiol. 2000;88:409–415. doi: 10.1152/jappl.2000.88.2.409. [DOI] [PubMed] [Google Scholar]