Abstract
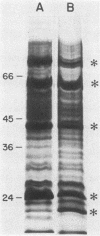
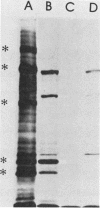
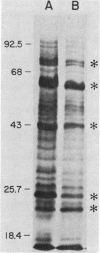
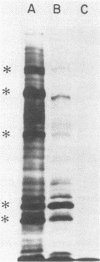
Growth of Legionella pneumophila serogroup 1 in yeast extract broth was standardized, and protein profiles of detergent-solubilized cells were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Over 30 protein bands were identified, 6 of which were more prominent both in Coomassie brilliant blue-stained profiles and in fluorograms with intrinsically radiolabeled bacteria. These major proteins were 22,000 dalton (22K), 24K, 43K, 63K, 76K, and 78K. We found that the 24K and 63K major proteins were antigenic, as demonstrated both by radioimmunoprecipitation (RIP) of [35S]methionine-labeled organisms and by immunoblotting with rabbit hyperimmune sera. In addition, both techniques detected antigens migrating at 58K, 72K, 76K, and 78K. The major 22K and 43K major proteins and antigens migrating at 25.5K, 29K, and 46K were only detected by radioimmunoprecipitation, whereas antigens of 19K, 48K, 53K, and 68K were detected only by immunoblotting. Cell-surface localization of the proteins was determined by a modified radioimmunoprecipitation assay designed to react specifically with surface antigens and by the use of hyperimmune sera that had been extensively preabsorbed with intact cells to deplete the sera of antibodies directed against surface components. The 22K, 24K, 43K, 63K, and 78K major proteins and several minor proteins were found to be located on the surface of L. pneumophila cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alderete J. F. Antigen analysis of several pathogenic strains of Trichomonas vaginalis. Infect Immun. 1983 Mar;39(3):1041–1047. doi: 10.1128/iai.39.3.1041-1047.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amano K., Williams J. C. Peptidoglycan of Legionella pneumophila: apparent resistance to lysozyme hydrolysis correlates with a high degree of peptide cross-linking. J Bacteriol. 1983 Jan;153(1):520–526. doi: 10.1128/jb.153.1.520-526.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baseman J. B., Hayes E. C. Molecular characterization of receptor binding proteins and immunogens of virulent Treponema pallidum. J Exp Med. 1980 Mar 1;151(3):573–586. doi: 10.1084/jem.151.3.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins M. T., Cho S. N., Høiby N., Espersen F., Baek L., Reif J. S. Crossed immunoelectrophoretic analysis of Legionella pneumophila serogroup 1 antigens. Infect Immun. 1983 Mar;39(3):1428–1440. doi: 10.1128/iai.39.3.1428-1440.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis G. S., Winn W. C., Jr, Gump D. W., Beaty H. N. The kinetics of early inflammatory events during experimental pneumonia due to Legionella pneumophila in guinea pigs. J Infect Dis. 1983 Nov;148(5):823–835. doi: 10.1093/infdis/148.5.823. [DOI] [PubMed] [Google Scholar]
- Edelstein P. H. Comparative study of selective media for isolation of Legionella pneumophila from potable water. J Clin Microbiol. 1982 Oct;16(4):697–699. doi: 10.1128/jcm.16.4.697-699.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehret W., Ruckdeschel G. Species specific membrane proteins of Legionellaceae. Zentralbl Bakteriol Mikrobiol Hyg A. 1983 Jul;255(1):33–38. [PubMed] [Google Scholar]
- Elliott J. A., Johnson W. Immunological and biochemical relationships among flagella isolated from Legionella pneumophila serogroups 1, 2, and 3. Infect Immun. 1981 Aug;33(2):602–610. doi: 10.1128/iai.33.2.602-610.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engleberg N. C., Drutz D. J., Eisenstein B. I. Cloning and expression of Legionella pneumophila antigens in Escherichia coli. Infect Immun. 1984 May;44(2):222–227. doi: 10.1128/iai.44.2.222-227.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engleberg N. C., Pearlman E., Eisenstein B. I. Legionella pneumophila surface antigens cloned and expressed in Escherichia coli are translocated to the host cell surface and interact with specific anti-Legionella antibodies. J Bacteriol. 1984 Oct;160(1):199–203. doi: 10.1128/jb.160.1.199-203.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulig P. A., McCracken G. H., Jr, Frisch C. F., Johnston K. H., Hansen E. J. Antibody response of infants to cell surface-exposed outer membrane proteins of Haemophilus influenzae type b after systemic Haemophilus disease. Infect Immun. 1982 Jul;37(1):82–88. doi: 10.1128/iai.37.1.82-88.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen E. J., Frisch C. F., Johnston K. H. Detection of antibody-accessible proteins on the cell surface of Haemophilus influenzae type b. Infect Immun. 1981 Sep;33(3):950–953. doi: 10.1128/iai.33.3.950-953.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman P. S., Pine L., Bell S. Production of superoxide and hydrogen peroxide in medium used to culture Legionella pneumophila: catalytic decomposition by charcoal. Appl Environ Microbiol. 1983 Mar;45(3):784–791. doi: 10.1128/aem.45.3.784-791.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A. Cell-mediated immunity in Legionnaires' disease. J Clin Invest. 1983 Jun;71(6):1686–1697. doi: 10.1172/JCI110923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A., Silverstein S. C. Activated human monocytes inhibit the intracellular multiplication of Legionnaires' disease bacteria. J Exp Med. 1981 Nov 1;154(5):1618–1635. doi: 10.1084/jem.154.5.1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A., Silverstein S. C. Interaction of the Legionnaires' disease bacterium (Legionella pneumophila) with human phagocytes. I. L. pneumophila resists killing by polymorphonuclear leukocytes, antibody, and complement. J Exp Med. 1981 Feb 1;153(2):386–397. doi: 10.1084/jem.153.2.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A., Silverstein S. C. Interaction of the legionnaires' disease bacterium (Legionella pneumophila) with human phagocytes. II. Antibody promotes binding of L. pneumophila to monocytes but does not inhibit intracellular multiplication. J Exp Med. 1981 Feb 1;153(2):398–406. doi: 10.1084/jem.153.2.398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz M. A. The Legionnaires' disease bacterium (Legionella pneumophila) inhibits phagosome-lysosome fusion in human monocytes. J Exp Med. 1983 Dec 1;158(6):2108–2126. doi: 10.1084/jem.158.6.2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leith D. K., Trevino L. B., Tully J. G., Senterfit L. B., Baseman J. B. Host discrimination of Mycoplasma pneumoniae proteinaceous immunogens. J Exp Med. 1983 Feb 1;157(2):502–514. doi: 10.1084/jem.157.2.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDade J. E., Shepard C. C., Fraser D. W., Tsai T. R., Redus M. A., Dowdle W. R. Legionnaires' disease: isolation of a bacterium and demonstration of its role in other respiratory disease. N Engl J Med. 1977 Dec 1;297(22):1197–1203. doi: 10.1056/NEJM197712012972202. [DOI] [PubMed] [Google Scholar]
- Norrby E., Enders-Ruckle G., Meulen V. Differences in the appearance of antibodies to structural components of measles virus after immunization with inactivated and live virus. J Infect Dis. 1975 Sep;132(3):262–269. doi: 10.1093/infdis/132.3.262. [DOI] [PubMed] [Google Scholar]
- Pine L., George J. R., Reeves M. W., Harrell W. K. Development of a chemically defined liquid medium for growth of Legionella pneumophila. J Clin Microbiol. 1979 May;9(5):615–626. doi: 10.1128/jcm.9.5.615-626.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ristroph J. D., Hedlund K. W., Allen R. G. Liquid medium for growth of Legionella pneumophila. J Clin Microbiol. 1980 Jan;11(1):19–21. doi: 10.1128/jcm.11.1.19-21.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolstad B., Berdal B. P. Immune defenses against Legionella pneumophila in rats. Infect Immun. 1981 May;32(2):805–812. doi: 10.1128/iai.32.2.805-812.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]