Abstract
Purpose
Angiogenesis plays an important role in the growth, progression, and metastasis of tumors. Vascular endothelial growth factor (VEGF) overexpression has been associated with advanced stage and poor survival in several cancers. We investigated the present case-control study to determine whether there is an association between the VEGF 936C > T polymorphism and stomach cancer.
Patients and Methods
The association of functional single nucleotide polymorphisms (SNPs) of the VEGF gene with stomach cancer development was evaluated in a case-control study of 154 Korean stomach cancer patients. Genotypes were determined by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) analysis.
Results
Our results revealed significant association of T allele-bearing genotypes with increased risk for stomach cancer development. Genotype frequencies of the VEGF 936C > T polymorphisms were significantly different between patient and control groups (CT, AOR: 2.007, 95% CI: 1.277 - 3.156, TT, AOR: 4.790, 95% CI: 1.174 - 19.539, CT + TT, AOR: 2.147, 95% CI: 1.382 - 3.337). When stratified by gender and age, genotype frequencies were significantly different for stomach cancer in women and in patients younger than 55 years (in women, CT, OR: 3.049, 95% CI: 1.568 - 5.930, CT+TT, OR: 3.132, 95% CI: 1.638 - 5.990; in < 55 years, CT, OR: 3.306, 95% CI: 1.413 - 7.732, CT + TT, OR: 3.967, 95% CI: 1.729 - 9.104). In addition, this association partially remained in cases with intestinal and diffuse-type stomach cancer.
Conclusion
Our present study suggests that the VEGF 936C > T polymorphism is a susceptibility factor for stomach cancer, at least in Korean.
Keywords: Stomach cancer, polymorphism, vascular endothelial growth factor, Korean
INTRODUCTION
As one of the most common human malignant tumors, stomach cancer ranks as the first leading cause of gastrointestinal cancer-related mortality worldwide.1 Over the past few years, many attempts have been made to better define the biological profile of stomach cancer in order to improve early diagnosis and prognostic stratification, and to find an eventual cure.2-4
Angiogenesis is an essential step for tumor growth, playing a critical role in invasion and metastasis.5 It is regulated by various growth factors, among which VEGF plays a central role. A number of studies have confirmed that VEGF expression is closely associated with the extent of vascularization and prognosis in many solid tumors, and is predictive of resistance to radiotherapy, chemotherapy, and endocrine therapy.6-8 Furthermore, in vitro and in vivo experiments have shown that increased VEGF expression is associated with tumor growth and metastasis, whereas inhibition of VEGF expression results in suppression of tumor growth and tumor-induced neoangiogenesis.9 Clinical observations have revealed that high levels of VEGF expression and increased microvessel density in tumors are associated with advanced-stage disease and worse prognosis for various types of tumors, including stomach cancer.6,7,10 Many studies have found a correlation between VEGF expression and several malignancies, such as stomach cancer and breast, gastrointestinal, urinary tract, and ovarian tumors.11-15 In stomach cancer, VEGF has been correlated with vascular involvement, lymph node and liver metastasis.16
Several SNPs in the VEGF gene have been shown to affect the expression of the gene.17 One of these, the 936C > T polymorphism in the 3-untranslated region (UTR) of the VEGF gene, was shown to affect VEGF plasma levels, and carriers of the VEGF 936T allele had significantly reduced VEGF plasma levels.18 Although some association studies have been performed, the effect of VEGF polymorphisms on the risk of stomach cancer has not yet been evaluated.
Since VEGF is significant in the angiogenesis of various types of tumors, it is reasonable to hypothesize that VEGF is a good candidate for determining the risk of developing stomach cancer. To test this hypothesis, we investigated genetic variations at the 936C > T polymorphic site in the 3'-UTR of the VEGF gene in a Korean population who have had stomach cancer.
PATIENTS AND METHODS
Subjects
A total of 154 patients (mean age ± SD, 58.05 ± 12.74 years) with stomach cancer diagnosed at Bundang CHA Hospital, Pochon CHA University, from July 1999 to June 2004, were enrolled in this study. The diagnosis and staging of stomach cancer were assessed according to the Lauren classification.19 Among stomach cancer patients, there were 91 men (age 57.76 ± 12.31 years; range, 30 - 88 years) and 63 women (age 58.46 ± 13.42 years; range, 28 - 81 years). Ninety-nine patients (age 59.85 ± 11.45 years; range, 28 - 88 years) had intestinal subtype stomach cancer and 55 patients (age 54.80 ± 14.32 years; range, 29 - 81 years) had diffuse-subtype stomach cancer. The control group consisted of 229 individuals (age 59.57 ± 11.80 years; range, 31 - 91 years) who were randomly selected through health screening to exclude those with a history of thrombotic diseases or cancer. This study was approved by the Institutional Review Board of Pochon CHA University, Korea.
VEGF genotyping
Genomic DNA was extracted from peripheral blood lymphocytes by proteinase K digestion and phenol/chloroform extraction. The VEGF 936C > T (rs3025039) genotype polymorphisms were determined using the PCR-RFLP method. The PCR primers used to detect the VEGF 936C > T polymorphism were 5'-AGG AAG AGG GAC TCT GCG CAG AGC-3' (forward) and 5'-TAA ATG TAT GTA TGT GGG TGG GTG TGT CTA CAG G-3' (reverse). The PCR product was digested overnight with NlaIII, the appropriate restriction enzyme (New England BioLabs, Beverly, MA, USA) for VEGF 936C > T genotyping. The VEGF 936T allele was cut into 2 fragments of 122 and 86 base pairs, whereas the VEGF 936C allele remained uncut with a length of 208 base pairs.
Data analysis
Genotype frequencies of patients and healthy control subjects were in Hardy-Weinberg equilibrium, as tested by Χ2 test. Allele and genotype frequencies between the case and control groups were compared using Χ2 test and Fisher's exact test. Odds ratios (ORs) and 95% confidence intervals (95% CIs) were used as the measure of the strength of association between the VEGF genotypes and stomach cancer. Stratification analysis was used to study subgroups by age and gender. StatsDirect Statistical Software Version 2.4.4 (StatsDirect Ltd., Altrincham, UK) was used to calculate the adjusted odds ratio (AOR) and 95% CI.
RESULTS
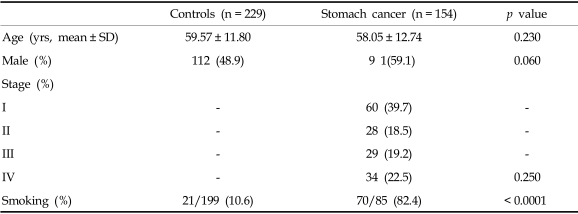
The median patient age was 60 years (range, 28-88 years), and 91 patients (59.1%) were male. There was no statistically significant difference in age (p = 0.230) or gender distribution (p = 0.060) between patients and controls (Table 1). The histopathological classification after surgical resection or biopsy included at least 2 main subtypes: intestinal type (n = 99, 64.3%) and diffuse type (n = 55, 35.7%).
Table 1.
Baseline Characteristics in Patients with Stomach Cancer and Controls
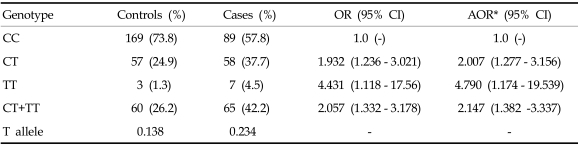
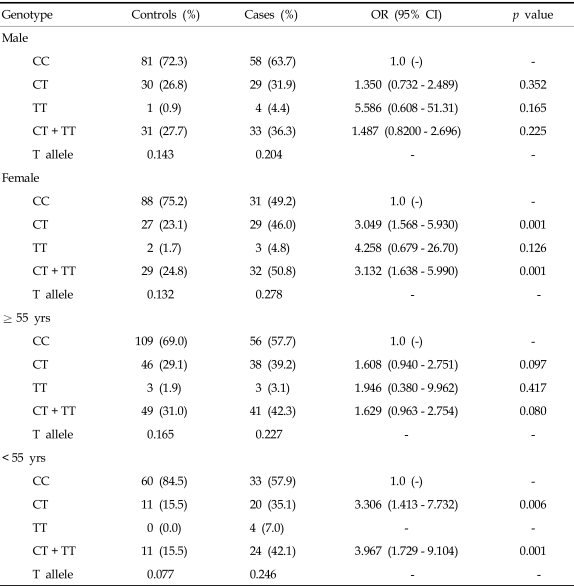
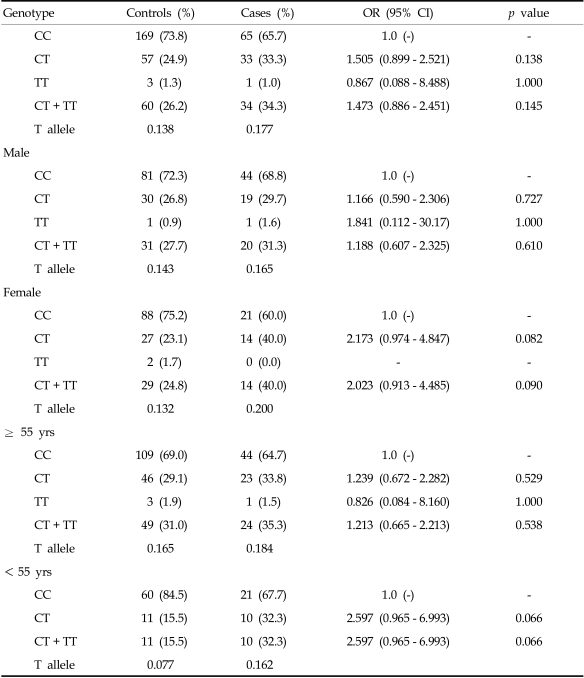
Tables 2 and 3 compare genotype frequencies of the VEGF 936C > T polymorphism according to the study group as a whole or by gender and age between case and control groups. Genotypes were in Hardy-Weinberg equilibrium in both the study and control groups (p = 0.241). The distribution of genotypes with the T allele at the VEGF 936C > T polymorphism was significantly different between the control and case groups (936CT, AOR, 2.007; 95% CI, 1.277 - 3.156; 936TT, AOR, 4.790; 95% CI, 1.174 - 19.539; 936CT + TT, AOR, 2.147; 95% CI, 1.382 - 3.337; Table 2). Frequencies of the 936CT and 936CT+TT genotypes in patients were associated with increased risk for stomach cancer in females (OR, 3.049; 95% CI, 1.568 - 5.930; p = 0.001 and OR, 3.132; 95% CI, 1.638 - 5.990; p = 0.001; Table 3) when stratified by gender. When the data were stratified by age, the association remained in patients who were less than 55 years old (936CT, OR, 3.306; 95% CI, 1.413 - 7.732; p = 0.006, 936CT + TT, OR, 3.967; 95% CI, 1.729 - 9.104; p = 0.001).
Table 2.
Comparison of Genotype Frequencies of the Vascular Endothelial Growth Factor 936C > T Polymorphism in Patients with Stomach Cancer and Controls
OR, odds ratio; AOR, adjusted odds ratio.
*Adjusted by age and gender.
Table 3.
Comparison of Subtype Frequencies of the Vascular Endothelial Growth Factor 936C > T Polymorphism in Patients with Stomach Cancer and Controls
OR, odds ratio.
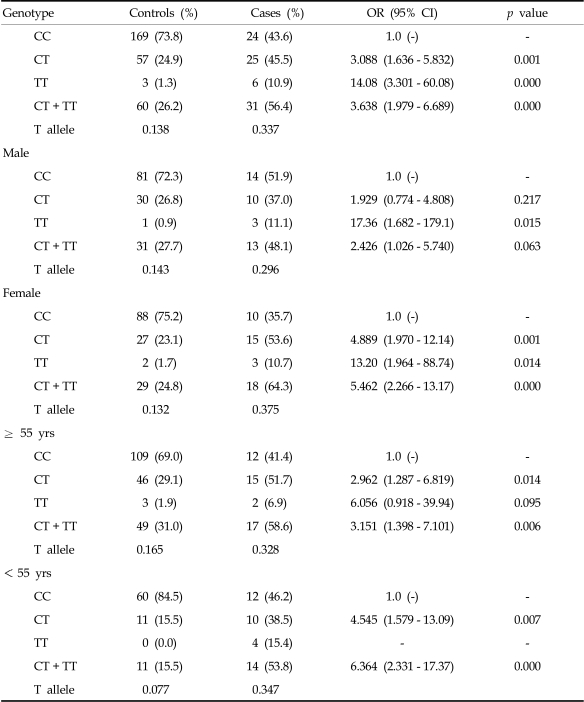
Moreover, in females with diffuse-subtype stomach cancer, the 936CT and 936CT + TT genotypes were associated with increased risk for stomach cancer (936CT, OR, 4.889; 95% CI, 1.970 - 12.14; p = 0.001, 936CT + TT, OR, 5.462; 95% CI, 2.266 - 13.17; p = 0.000) (Table 5). For intestinal-subtype stomach cancer, however, there was no significant difference in the genotype frequencies between the 2 groups (Table 4).
Table 5.
Comparison of Genotype Frequencies of the Vascular Endothelial Growth Factor 936C > T Polymorphism in Patients with Diffuse-type Stomach Cancer and Controls
OR, odds ratio.
Table 4.
Comparison of Genotype Frequencies of the Vascular Endothelial Growth Factor 936C > T Polymorphism in Patients with Intestinal-type Stomach Cancer and Controls
OR, odds ratio.
DISCUSSION
Angiogenesis is essential for tumor growth and plays a critical role in the invasion and metastasis of tumor cells. It is regulated by many growth factors, among which VEGF plays a central role and serves as an important prognostic factor in a variety of tumors, including stomach cancer. The role of genetic polymorphisms, which are important determinants of endogenous causes of cancer, in the risk of stomach cancer has attracted increasing interest due to advances in DNA analysis technologies and knowledge of the human genome. VEGF and its family play a critical role in tumor-related angiogenesis, and several functional polymorphisms in the VEGF gene have already been reported to be associated with VEGF gene expression or an increased risk of solid tumors, making them potential predictive markers for clinical outcome.7,20-24 For example, the less common T allele of the 936C > T polymorphism in the 3'-untranslated region of the VEGF gene is correlated with lower VEGF levels.20 The contribution of this polymorphism to oncogenesis has been investigated in several types of cancer but not in stomach cancer until now to the our best knowledge.6,10,20,25-28
Based on the involvement of VEGF in the risk of advanced-stage cancer through tumor growth and metastasis of several types of cancer, including stomach cancer, we evaluated the relationship between the VEGF 936C > T polymorphism and stomach cancer in a Korean patient case-control study. In the present study, the VEGF 936T allele was found to be associated with increased risk for stomach cancer not only overall but also in female subjects and subjects less than 55 years old when stratified by gender and age.
Several attempts to classify stomach cancer have been made in the past decades. The most successful and widely used classification is by Lauren, who distinguished 2 main cancer pathogeneses, diffuse and intestinal subtypes, by microscopic morphology alone. These 2 subtypes clearly appear as clinical and epidemiological by dissimilar entities. Here, we analyzed samples from each patient according to the main differences in epidemiology, histopathology, and molecular pathology of the 2 main subtypes of stomach cancer based on the Lauren classification.29 There was no significant difference in the genotype frequencies with intestinal-subtype stomach cancer between the control and patient groups. However, despite the relatively small number of individuals in this study, highly significant differences were noted with diffuse-subtype stomach cancer between patients and controls.
Surprisingly, stomach cancer in women is associated with some genotypes of the VEGF 936C > T polymorphism in this study. These trends remained in women with diffuse-subtype stomach cancer, even when the data were stratified by the Lauren classification. That is, the association was gender-specific, suggesting that VEGF production according to VEGF 936C > T genotype might differ between men and women. In addition, the association was also specific for the histological subtype of the stomach tumor, indicating that VEGF production depends on the histological subtype of the stomach tumor.
Our results strongly indicate that the T allele is linked to an increased risk for stomach cancer, despite being associated with lower circulating levels of VEGF. It may, therefore, be assumed that high VEGF levels are not a prerequisite for stomach cancer susceptibility. Similarly, in gliomas, low-expression VEGF genotypes coexist with high VEGF levels in patients but not in healthy controls.30 High VEGF expression is attributed to independent cancer and tumor stroma production.30
The findings of this study clearly demonstrate that the low-expression T allele is associated with an increased stomach cancer risk, however, the underlying mechanism might not involve angiogenesis but rather other VEGF-related functions such as thrombosis. As previously mentioned, in accordance with our results, a significant increase in the T allele frequency has been found in cancer patients with thrombotic complications compared to healthy controls and non-thrombotic cancer patients.31 This association has been reported in many cases, while the 936TT genotype is associated with larger tumors and presence of metastases.24 This study clearly implicates the low-expression T allele with an increased risk of stomach cancer.
In cancer patients, additional risk factors for thrombotic complications raise close relationships between tumors and activation of blood coagulation. Tumor and tumor-associated endothelial cells are able to produce and release some molecules,32 and one of the molecules is VEGF, which is a the well-characterized regulator of angiogenesis. VEGF also affects hemostasis of endothelial cells. VEGF has a prothrombotic effect on endothelial cells, thus encouraging tissue factor expression and additionally platelet activation.33 Polymorphisms of the VEGF gene have been shown to correlate with variation in VEGF protein production.34 Other oral and colon cancer studies have reported that low VEGF production of the 936C > T polymorphism is strongly associated with increased cancer risk.35,36
We were interested in studying VEGF 936C > T polymorphisms in the Korean population because it has a relatively homogeneous ethnic origin in contrast to the more heterogeneous characteristics of ethnic groups examined in previous studies.36 The literature survey indicates that the frequency of VEGF 936T allele among healthy controls was 0.138 in Koreans and 0.157 in Japanese, indicating that the allele ratio was similar among Asian populations.36,37 This study revealed that allele frequencies of the VEGF 936C > T polymorphism in our study control group were not significantly different from other Asian populations. On the other hand, there was a racial difference in the frequency of the T allele: 0.167 in Canadians, 0.161 in Polish (only female group), 0.140 in Americans, and 0.091 in Germans.6,26,38,39 Earlier differences in the literature might have been results may be due to different genetic backgrounds.
Functional polymorphisms, which affect the regulation of gene expression, can contribute to differences in susceptibility to and severity of a disease between individuals. The effect may be seen with polymorphism alone or in combination with other polymorphisms. These functional polymorphisms may result in altered transcription factor recognition sites, which subsequently affect transcriptional activation and alter protein production.37 Haplotype analysis, which is currently the focus of intense genetic research efforts, will make risk estimates more specific than single locus analyses.40 Identification of associations between candidate genes and disease will be one of the main objectives in the development of personalized and interactive medicine.
In conclusion, our present findings indicate that the VEGF 936C > T polymorphism may be a useful indicator of susceptibility to stomach cancer. Large-scale genetic studies including haplotype analyses may be needed to improve statistical power. In addition, studies of other VEGF sequence variants and their biological functions are also needed to determine the risk of stomach cancer. Since genetic polymorphisms often vary between ethnic groups, further studies are needed to clarify the association between VEGF polymorphisms and stomach cancer in diverse ethnic populations.
Footnotes
This work was supported by a grant from the Korea Research Foundation of the Korean government (MOEHRD) (KRF-2005-041-E00360).
References
- 1.Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. doi: 10.3322/canjclin.55.2.74. [DOI] [PubMed] [Google Scholar]
- 2.Kong U, Koo J, Choi K, Park J, Chang H. The expression of GAGE gene can predict aggressive biologic behavior of intestinal type of stomach cancer. Hepatogastroenterology. 2004;51:1519–1523. [PubMed] [Google Scholar]
- 3.Yasui W, Ito H, Yoshida K, Tahara E. Biological malignancy in human gastric carcinoma--molecular aspects. Gan To Kagaku Ryoho. 1991;18:927–933. [PubMed] [Google Scholar]
- 4.Tokunaga A, Onda M, Okuda T, Teramoto T, Fujita I, Mizutani T, et al. Clinical significance of epidermal growth factor (EGF), EGF receptor, and c-erbB-2 in human gastric cancer. Cancer. 1995;75(6 Suppl):1418–1425. doi: 10.1002/1097-0142(19950315)75:6+<1418::aid-cncr2820751505>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 5.Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 6.Jin Q, Hemminki K, Enquist K, Lenner P, Grzybowska E, Klaes R, et al. Vascular endothelial growth factor polymorphisms in relation to breast cancer development and prognosis. Clin Cancer Res. 2005;11:3647–3653. doi: 10.1158/1078-0432.CCR-04-1803. [DOI] [PubMed] [Google Scholar]
- 7.Koukourakis MI, Papazoglou D, Giatromanolaki A, Bougioukas G, Maltezos E, Sivridis E. VEGF gene sequence variation defines VEGF gene expression status and angiogenic activity in non-small cell lung cancer. Lung Cancer. 2004;46:293–298. doi: 10.1016/j.lungcan.2004.04.037. [DOI] [PubMed] [Google Scholar]
- 8.Ferrara N. Vascular endothelial growth factors as a target for anticancer therapy. Oncologist. 2004;9(Suppl 1):2–10. doi: 10.1634/theoncologist.9-suppl_1-2. [DOI] [PubMed] [Google Scholar]
- 9.Ferrara N. VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer. 2002;2:795–803. doi: 10.1038/nrc909. [DOI] [PubMed] [Google Scholar]
- 10.Lu H, Shu XO, Cui Y, Kataoka N, Wen W, Cai Q, et al. Association of genetic polymorphisms in the VEGF gene with breast cancer survival. Cancer Res. 2005;65:5015–5019. doi: 10.1158/0008-5472.CAN-04-2786. [DOI] [PubMed] [Google Scholar]
- 11.Takahashi Y, Kitadai Y, Bucana CD, Cleary KR, Ellis LM. Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res. 1995;55:3964–3968. [PubMed] [Google Scholar]
- 12.Toi M, Hoshina S, Takayanagi T, Tominaga T. Association of vascular endothelial growth factor expression with tumor angiogenesis and with early relapse in primary breast cancer. Jpn J Cancer Res. 1994;85:1045–1049. doi: 10.1111/j.1349-7006.1994.tb02904.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brown LF, Berse B, Jackman RW, Tognazzi K, Manseau EJ, Senger DR, et al. Expression of vascular permeability factor (vascular endothelial growth factor) and its receptors in adenocarcinomas of the gastrointestinal tract. Cancer Res. 1993;53:4727–4735. [PubMed] [Google Scholar]
- 14.Brown LF, Berse B, Jackman RW, Tognazzi K, Manseau EJ, Dvorak HF, et al. Increased expression of vascular permeability factor (vascular endothelial growth factor) and its receptors in kidney and bladder carcinomas. Am J Pathol. 1993;143:1255–1262. [PMC free article] [PubMed] [Google Scholar]
- 15.Olson TA, Mohanraj D, Carson LF, Ramakrishnan S. Vascular permeability factor gene expression in normal and neoplastic human ovaries. Cancer Res. 1994;54:276–280. [PubMed] [Google Scholar]
- 16.Maeda K, Chung YS, Ogawa Y, Takatsuka S, Kang SM, Ogawa M, et al. Prognostic value of vascular endothelial growth factor expression in gastric carcinoma. Cancer. 1996;77:858–863. doi: 10.1002/(sici)1097-0142(19960301)77:5<858::aid-cncr8>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 17.Watson CJ, Webb NJ, Bottomley MJ, Brenchley PE. Identification of polymorphisms within the vascular endothelial growth factor (VEGF) gene: correlation with variation in VEGF protein production. Cytokine. 2000;12:1232–1235. doi: 10.1006/cyto.2000.0692. [DOI] [PubMed] [Google Scholar]
- 18.Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183:S1–S22. [PubMed] [Google Scholar]
- 19.Vauhkonen M, Vauhkonen H, Sipponen P. Pathology and molecular biology of gastric cancer. Best Pract Res Clin Gastroenterol. 2006;20:651–674. doi: 10.1016/j.bpg.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 20.Krippl P, Langsenlehner U, Renner W, Yazdani-Biuki B, Wolf G, Wascher TC, et al. A common 936 C/T gene polymorphism of vascular endothelial growth factor is associated with decreased breast cancer risk. Int J Cancer. 2003;106:468–471. doi: 10.1002/ijc.11238. [DOI] [PubMed] [Google Scholar]
- 21.Lambrechts D, Storkebaum E, Morimoto M, Del-Favero J, Desmet F, Marklund SL, et al. VEGF is a modifier of amyotrophic lateral sclerosis in mice and humans and protects motoneurons against ischemic death. Nat Genet. 2003;34:383–394. doi: 10.1038/ng1211. [DOI] [PubMed] [Google Scholar]
- 22.McCarron SL, Edwards S, Evans PR, Gibbs R, Dearnaley DP, Dowe A, et al. Influence of cytokine gene polymorphisms on the development of prostate cancer. Cancer Res. 2002;62:3369–3372. [PubMed] [Google Scholar]
- 23.Shahbazi M, Fryer AA, Pravica V, Brogan IJ, Ramsay HM, Hutchinson IV, et al. Vascular endothelial growth factor gene polymorphisms are associated with acute renal allograft rejection. J Am Soc Nephrol. 2002;13:260–264. doi: 10.1681/ASN.V131260. [DOI] [PubMed] [Google Scholar]
- 24.Tzanakis N, Gazouli M, Rallis G, Giannopoulos G, Papaconstantinou I, Theodoropoulos G, et al. Vascular endothelial growth factor polymorphisms in gastric cancer development, prognosis, and survival. J Surg Oncol. 2006;94:624–630. doi: 10.1002/jso.20619. [DOI] [PubMed] [Google Scholar]
- 25.Kataoka N, Cai Q, Wen W, Shu XO, Jin F, Gao YT, et al. Population-based case-control study of VEGF gene polymorphisms and breast cancer risk among Chinese women. Cancer Epidemiol Biomarkers Prev. 2006;15:1148–1152. doi: 10.1158/1055-9965.EPI-05-0871. [DOI] [PubMed] [Google Scholar]
- 26.Jacobs EJ, Feigelson HS, Bain EB, Brady KA, Rodriguez C, Stevens VL, et al. Polymorphisms in the vascular endothelial growth factor gene and breast cancer in the Cancer Prevention Study II cohort. Breast Cancer Res. 2006;8:R22. doi: 10.1186/bcr1400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sfar S, Hassen E, Saad H, Mosbah F, Chouchane L. Association of VEGF genetic polymorphisms with prostate carcinoma risk and clinical outcome. Cytokine. 2006;35:21–28. doi: 10.1016/j.cyto.2006.07.003. [DOI] [PubMed] [Google Scholar]
- 28.Abe A, Sato K, Habuchi T, Wang L, Li Z, Tsuchiya N, et al. Single nucleotide polymorphisms in the 3' untranslated region of vascular endothelial growth factor gene in Japanese population with or without renal cell carcinoma. Tohoku J Exp Med. 2002;198:181–190. doi: 10.1620/tjem.198.181. [DOI] [PubMed] [Google Scholar]
- 29.Vauhkonen M, Vauhkonen H, Sipponen P. Pathology and molecular biology of gastric cancer. Best Pract Res Clin Gastroenterol. 2006;20:651–674. doi: 10.1016/j.bpg.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 30.Sciacca FL, Ciusani E, Silvani A, Corsini E, Frigerio S, Pogliani S, et al. Genetic and plasma markers of venous thromboembolism in patients with high grade glioma. Clin Cancer Res. 2004;10:1312–1317. doi: 10.1158/1078-0432.ccr-03-0198. [DOI] [PubMed] [Google Scholar]
- 31.Eroglu A, Gulec S, Akar N. Vascular endothelial growth factor C936T polymorphism in cancer patients with thrombosis. Am J Hematol. 2006;82:174. doi: 10.1002/ajh.20719. [DOI] [PubMed] [Google Scholar]
- 32.Pinedo HM. The role of VEGF in oncology: effects on hemostasis and thrombosis. Pathophysiol Haemost Thromb. 2003;33(Suppl 1):11–12. doi: 10.1159/000073280. [DOI] [PubMed] [Google Scholar]
- 33.Verheul HM, Jorna AS, Hoekman K, Broxterman HJ, Gebbink MF, Pinedo HM. Vascular endothelial growth factor-stimulated endothelial cells promote adhesion and activation of platelets. Blood. 2000;96:4216–4221. [PubMed] [Google Scholar]
- 34.Brogan IJ, Khan N, Isaac K, Hutchinson JA, Pravica V, Hutchinson IV. Novel polymorphism in the promoter and 5' UTR regions of the human vascular endothelial growth factor gene. Hum Immunol. 1999;60:1245–1249. doi: 10.1016/s0198-8859(99)00132-9. [DOI] [PubMed] [Google Scholar]
- 35.Yapijakis C, Vairaktaris E, Vassiliou S, Vylliotis A, Nkenke E, Nixon AM, et al. The low VEGF production allele of the +936C/T polymorphism is strongly associated with increased risk for oral cancer. J Cancer Res Clin Oncol. 2007;133:787–791. doi: 10.1007/s00432-007-0240-y. [DOI] [PubMed] [Google Scholar]
- 36.Bae SJ, Kim JW, Kang H, Hwang SG, Oh D, Kim NK. Gender-specific association between polymorphism of vascular endothelial growth factor (VEGF 936C > T) gene and colon cancer in Korea. Anticancer Res. 2008;28:1271–1276. [PubMed] [Google Scholar]
- 37.Sakao S, Tatsumi K, Hashimoto T, Igari H, Shino Y, Shirasawa H, et al. Vascular endothelial growth factor and the risk of smoking-related COPD. Chest. 2003;124:323–327. doi: 10.1378/chest.124.1.323. [DOI] [PubMed] [Google Scholar]
- 38.Butt C, Lim S, Greenwood C, Rahman P. VEGF, FGF1, FGF2 and EGF gene polymorphisms and psoriatic arthritis. BMC Musculoskelet Disord. 2007;8:1. doi: 10.1186/1471-2474-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Günesacar R, Opelz G, Erken E, Pelzl S, Döhler B, Ruhenstroth A, et al. VEGF 936 C/T gene polymorphism in renal transplant recipients: association of the T allele with good graft outcome. Hum Immunol. 2007;68:599–602. doi: 10.1016/j.humimm.2007.03.015. [DOI] [PubMed] [Google Scholar]
- 40.Clark AG. The role of haplotypes in candidate gene studies. Genet Epidemiol. 2004;27:321–333. doi: 10.1002/gepi.20025. [DOI] [PubMed] [Google Scholar]