Short abstract
Advances are being made in the management of sickle cell disease for all age groups. This review discusses the progress in amelioration of symptoms, problems unique to particular age groups, and the types of drugs and treatments currently under investigation
In 1973, the average life span of a patient with sickle cell disease was 14 years. Thirty years later, the development of comprehensive care models, day hospitals, and ongoing efforts in both basic sciences and clinical research has increased life expectancy to 50 years. Advances in the management of sickle cell disease have dramatically changed the outlook for patients (box 1).
Polymerisation of sickle haemoglobin is the catalyst in the development of vaso-occlusion. Additional factors are critical to the pathophysiology of sickling (the development of sickle cells in the blood), including abnormalities in coagulation, white cells, vascular endothelium, and damage to the membranes of red cells.1 The culmination of these factors results in red cells with reduced deformability that easily adhere to vascular endothelium, increasing the potential for decreased blood flow and vascular obstruction. As a result, patients develop both haemolytic anaemia and vasculopathy.2 Organ damage is also an ongoing problem. Although the course of the disease is punctuated by episodes of pain, the organ damage is often silent until far advanced. We discuss the advances in the clinical management of sickle cell disease and new therapies that are available.
Sources and selection criteria
Our review is based on information from published research studies from the Children's Hospital and Research Center at Oakland. Electronic searches were through Medline and PubMed, and we downloaded relevant articles. We also extracted information from reviews of current work and consultations with other experts and health providers in the specialty of sickle cell disease.
Optimal management
The optimal setting for the care of patients with sickle cell disease is a comprehensive centre, with a multidisciplinary team to provide ongoing support. Ideally, patients should be identified at birth as part of a screening programme and referred to a centre for periodic evaluations. A close working relation between the primary care provider and the centre are essential for appropriate care.3-5
It is important to provide preconception genetic counselling for women of childbearing age with sickle cell disease. All contraceptive devices are suitable for patients with the exception of intrauterine devices, owing to the risk of infection. Risks during pregnancy include an increase in gestational hypertension, preterm birth, and small for gestational age infants.6 Pain crises may also increase.7 Pregnant patients therefore require close monitoring by health professionals familiar with high risk obstetrics and haematology.
Summary points
New therapy offers patients with sickle cell disease a prolonged life span, with the potential to prevent or minimise complications that impair quality of life
Multiorgan screening can now detect early injury and allow corrective intervention
Advances in transfusion therapy has resulted in improved safety
Advances in clinical therapy include prophylactic antibiotics, red cell pheresis, hydroxyurea, and angiotensin converting enzyme inhibitors to prevent renal disease
Bone marrow transplantation has become accepted therapy for severely affected patients
Paediatric patients
Parents of newborn infants with sickle cell disease receive genetic counselling and support. They are taught to assess fever, respiratory distress, splenomegaly, and jaundice.8 Giving infants prophylactic penicillin has been shown to reduce mortality from pneumococcal sepsis.9,10 The 7-valent pneumococcal conjugated vaccine is suitable for infants and toddlers and the 23-valent preparation for children over 2 years. Immunisation against influenza is also important. Common problems in children include febrile events, acute chest syndrome, sequestration crisis, and aplastic episodes due to infection with parvovirus. One of the earliest manifestations of sickling in young children is dactylitis, or “hand-foot syndrome.” An episode of painful swelling of the bones of the hand or foot may predict severe disease.7 Growth, nutrition, and educational performance require close monitoring. The management of febrile events should be reviewed with the family, including outpatient use of ceftriaxone for selected patients with fever.
Ischaemic stroke is one of the most devastating problems in children.11 Early detection and prevention is now possible. The Stroke Prevention Trial in Sickle Cell Anemia (STOP) study showed that screening by transcranial Doppler ultrasonography followed by transfusion noticeably reduced the incidence of stroke.12 Patients aged over 2 years should undergo transcranial Doppler screening annually. Similarly, patients with poor educational performance should undergo neurocognitive testing, as they may have early ischaemia of the central nervous system.
Box 1: Clinical advances in treatment of sickle cell disease
Screening of newborn infants
Family education
Counselling
Comprehensive care
Infection
Prophylactic penicillin
Pneumococcal vaccine
Prevention
Brain injury
Screening
Transcranial Doppler ultrasonography (now recommended for detection of brain injury and subsequent transfusion of high risk patients)
Magnetic resonance imaging
Neurocognitive testing
Lung injury
Incentive spirometry
Antibiotics, including macrolides
Transfusion
Prevention with prophylactic hydroxyurea
Screening for pulmonary hypertension
Transfusion safety and prevention of iron overload
Phenotypically matched red cells (routine transfusion with C, E, and Kell red cells minimises alloimmunisation)
Red cell pheresis (minimises iron overload)
Safety during surgery and anaesthesia
Preoperative transfusion
Avascular necrosis of hip
Decompression coring procedures (may prevent progression of disease; randomised controlled trial in process)
Priapism
Adrenergic agonist
Antiandrogen therapy
Pain
Multidisciplinary management (decreases number of admissions to hospital)
Prevention with prophylactic hydroxyurea
Patient controlled analgesic devices
New non-steroidal anti-inflammatory drugs
Day unit
Renal system
Angiotensin converting enzyme inhibitors for proteinuria (may prevent renal disease)
Improved renal transplantation
Gallbladder disease
Laparoscopic cholecystectomy (decreases perioperative morbidity)
Severe disease
Allogenic bone marrow transplantation (patients aged < 16 years) Chronic transfusions or hydroxyurea
Recurrent acute chest syndrome, pain crises, or central nervous system disease indicate eligibility for transplantation
Adolescent patients
Adolescents with sickle cell disease present with unique needs, including delayed sexual development, avascular necrosis of the hip, gallstones, priapism, proteinuria, pulmonary hypertension, and the onset of retinopathy. Ongoing counselling is required for issues of sexuality, drug use, birth control, and educational performance. A plan should also be put in place for transition from paediatric to adult care.5
Adult patients
The care of adult patients requires a complex team approach. Organ damage is a major problem in these patients so pulmonary, renal, and cardiac function needs to be monitored closely. Yearly ophthalmological examinations are important to assess for retinopathy.
Many adult patients may be overloaded with iron as a result of multiple transfusions and therefore require chelation therapy. Pain management may be an issue, as some patients develop chronic pain syndromes.13 Psychosocial support can help patients to cope with the economic and long term social consequences of a chronic illness.
Chronic organ damage
Spleen
Autosplenectomy in sickle cell disease results in immune dysfunction and the propensity for overwhelming infections with encapsulated organisms such as Pneumococcus and Haemophilus influenzae. Prophylactic penicillin has reduced the incidence of mortality in children under 2 years.9 Splenic sequestration, caused by red blood cells trapped in the splenic circulation, causes precipitous decreases in haemoglobin concentration and rapid enlargement of the spleen, and may be life threatening.5 Immediate treatment includes transfusion. Recurrence is about 50%, and splenectomy is often needed.
Central nervous system
Vascular damage to large cerebral arteries can cause brain injury in children. Prompt imaging and evaluation should be undertaken in children with neurological deficits.5 Transfusion must be instituted as soon as possible. Exchange transfusion is preferred, since this does not raise the viscosity of blood.5,14 Smaller “watershed” infarcts may be clinically silent but produce cognitive defects shown by neuropsychiatric testing.15,16 Patients may also present with cerebral haemorrhage secondary to berry aneurysms and “moya-moya” vascular abnormalities, more commonly seen in adults. Most patients with brain injury require long term transfusion therapy. Children should be considered for bone marrow transplantation.
Bones and joints
Orthopaedic problems begin in infancy. Bones and joints are often the site of vaso-occlusive episodes, and chronic infarcts may result. Abnormalities of the vertebrae (“fish mouthing”) are characteristic of sickle cell disease. Hyperplasia of the bone marrow may cause growth disturbances and osteopenia. Avascular necrosis of the weightbearing joints (hip and shoulders) causes chronic pain and may require surgical intervention. Adults are particularly susceptible to this complication. Plain x ray films may not detect early disease, and magnetic resonance imaging may be necessary. Early disease may improve with coring and osteotomy.17 Late disease requires joint replacement. Patients with sickle cell disease have an increased incidence of infection and failure of prosthesis. Osteomyelitis is also more common and may be difficult to distinguish from infarction. Radionucleotide imaging and bone aspiration are often necessary to diagnose bone infection.
Eye
Ophthalmological problems tend to occur in patients with increased concentrations of haemoglobin; patients with milder variants, especially sickle cell disease, are at higher risk. Symptomatic disease usually occurs in adults. Occlusion of small retinal vessels with neovascularisation is asymptomatic until haemorrhage occurs within the vitreous. Detachment of the retina may occur in late disease.5 Prevention is the most effective treatment. All patients must be evaluated yearly by an ophthalmologist. Fluorescein angiography followed by laser photocoagulation is an effective and safe treatment for retinal detachment. Occlusion of the central retinal artery, which presents as acute loss of vision, is an emergency, and patients must receive a transfusion immediately and be evaluated by a specialist.
Genitourinary system
Genitourinary problems are common in patients with sickle cell disease.18 The earliest lesion is hyposthenuria, which may become evident in childhood as enuresis and dehydration. Other effects of sickling on the kidney include damage to the distal renal tubule, producing type IV renal acidosis and haematuria secondary to papillary necrosis. Recently, renal medullary carcinoma has been reported in patients with sickle cell trait and young patients with sickle cell anaemia. Therefore, all patients with haematuria must be carefully investigated. Glomerular sclerosis, manifested by proteinuria, progresses as patients age. Proteinuria is a potential harbinger of chronic renal failure but can be ameliorated by angiotensin converting enzyme inhibitors. Chronic renal failure occurs in up to 5% of patients with sickle cell anaemia, and there is no proved treatment.19,12 This complication is often heralded by a worsening anaemia. Patients may require treatment with high doses of erythropoietin.
Another important genitourinary problem is priapism: a painful, persistent erection due to vaso-occlusive obstruction of the venous drainage of the penis. By adulthood, about 89% of males with sickle cell anaemia will have had a least one episode of priapism. This may be brief (less than two hours) and occur frequently (“stuttering”) or be prolonged. Both stuttering and prolonged episodes may result in impotence. Treatment for severe cases includes prompt drainage, injection of α adrenergic drugs such as phenylephrine, and transfusions.5,20 Prevention utilises hormonal treatments (lupron and diethylsilbesterol) and vasoconstrictors (terbutaline and pseudoephedrine at bedtime).21
Pulmonary system
Acute chest syndrome is the second most common cause of admission to hospital and a leading cause of mortality in all age groups with sickle cell disease.22 A multicentre study showed that fat emboli, from infarction of long bone, was the cause in a significant number of cases (table 1). An infectious cause, including atypical bacteria, occurs in all ages.23 Asthma may accompany acute chest syndrome and may persist after recovery, suggesting that airway hyper-reactivity may be a marker for lung injury in patients. Patients with acute chest syndrome improve rapidly with transfusion, a modality that should be considered early in the course of the illness.14 Patients should be given adequate pain control, incentive spirometry to prevent further infiltrates, and broad antibiotic coverage, which should also act against atypical bacteria.24 Hydroxyurea may prevent recurrence of acute chest syndrome.
Table 1.
Causes of acute chest syndrome in patients with sickle cell disease
|
Age at episode of acute chest syndrome
|
||||
|---|---|---|---|---|
| Cause | All episodes (n=670) | 0-9 years (n=329) | 10-19 years (n=188) | ≥20 years (n=153) |
| Fat embolism, with or without infection* | 59 (8.8) | 24 | 16 | 19 |
| Chlamydia† | 48 (7.2) | 19 | 15 | 14 |
| Mycoplasma‡ | 44 (6.6) | 29 | 7 | 8 |
| Viruses | 43 (6.4) | 36 | 5 | 2 |
| Bacteria | 30 (4.5) | 13 | 5 | 12 |
| Mixed infections | 25 (3.7) | 16 | 6 | 3 |
| Legionella | 4 (0.6) | 3 | 0 | 1 |
| Miscellaneous infections§ | 3 (0.4) | 0 | 3 | 0 |
| Infarction¶ | 108 (16.1) | 50 | 43 | 15 |
| Unknown** | 306 (45.7) | 139 | 88 | 79 |
Data on one episode excluded because patient's date of birth was not known.
19 episodes were associated with infectious pathogens.
Episodes with only chlamydia and not with mixed infections or pulmonary fat embolism.
Episodes with only Mycoplasma pneumoniae or Mycoplasma hominis, but not with mixed infections, Mycobacterium tuberculosis, or pulmonary fat embolism.
Includes two cases of tuberculosis and one case of Mycobacterium avium complex infection.
Presumed to have occurred during episodes in which results of analysis for pulmonary fat embolism, bacterial studies, viral isolation studies, and serological tests were complete and all negative.
Cause of episodes for which some or all of diagnostic data were incomplete and no causal agent was identified.
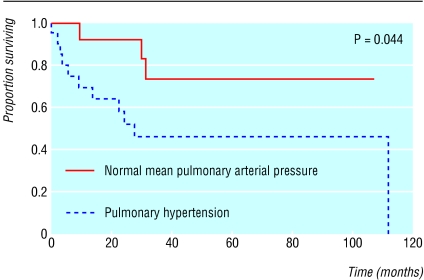
Patients with a history of pulmonary disease should undergo pulmonary function tests yearly to assess lung injury. Chronic lung damage results in worsening hypoxia and fibrosis, as shown by a decreased diffusion capacity. Over one third of adult patients will develop pulmonary hypertension, defined as a pulmonary artery systolic pressure greater than 25 as determined by echocardiography.25 Such patients may present with worsening hypoxia and chest pain. Sudden death is common, and mortality is high (fig 1). At present, the ideal therapy for pulmonary hypertension is unclear. Transfusion has proved a beneficial therapy in patients. Other potential therapies, used in primary pulmonary hypertension, include vasodilation and anti-coagulation. Newer agents that may aid patients include arginine and inhaled nitric oxide.20,26,27
Fig 1.
Kaplan-Meier survival plot of patients with sickle cell disease with and without pulmonary hypertension
Transfusion therapy
Transfusion therapy is a key component of the comprehensive management of patients with sickle cell disease.14,28 It is effective treatment for many of the serious complications (box 2). Transfusion should be considered in consultation with a haematologist. Therapy increases the oxygen carrying capacity of blood by increasing haemoglobin concentration and decreasing the percentage of sickle haemoglobin. Increasing the haemoglobin concentrations to over 11 g/dl may cause complications from increased viscosity.29 Straight transfusion, when the patient is given additional units of blood without removal of sickle blood, is best used when the haemoglobin concentration is lower than 8 or 9 g/dl. Exchange transfusion is better when the haemoglobin concentration is high. This procedure, which may be done manually or by automated red cell exchange, results in little net iron gain and decreases the amount of sickle haemoglobin while keeping the whole blood viscosity unchanged.14
It is important that patients undergo transfusion only when clearly indicated. Steady state anaemia, uncomplicated painful episodes, and minor surgery should not be treated with transfusions.5 Clear indications for therapy are acute chest syndrome, heart failure, multiorgan failure syndrome, stroke, splenic sequestration, and aplastic crisis.14
Box 2: Efficacy of chronic treatment for sickle cell disease
Prevention of organ damage
Brain injury
Acute chest syndrome
Pulmonary hypertension
Growth and puberty
Skin ulcers
Surgical complications
Bed wetting
Spleen function or sequestration
Quality of life
School attendance
IQ
Energy level
Wellbeing
Exercise tolerance
Mood
All transfused blood should be matched for the minor blood group antigens Kell and Rh (C and e, as well as D antigens). If patients develop antibodies despite this, more extensive matching is necessary. Other important transfusion practices include administering blood that is negative for sickle cells by sickle-dex, a routine sickle cell screening test that ensures that patients are not being transfused with blood containing sickle cells (to aid in the analysis of the percentage of sickle haemoglobin present after transfusion), and leukoreduction (to decrease viral transmission and prevent febrile transfusion reactions).
Iron overload leads to endocrine failure, cirrhosis, and cardiomyopathy. Serum ferritins are not accurate for monitoring iron overload. It is reliably detected by assessing dry weight of iron in liver biopsy samples. Patients with over 7 mg/g dry weight need chelation. Cardiac complications are common with iron levels above 15 mg/g.30 Non-invasive superconducting quantum interference devices (SQUID) accurately measure iron in liver and should be used if available. Desferal therapy, a method of chelation that patients who undergo repeated transfusions receive to remove excess iron from their bodies, is effective in treating haemosiderosis, but requires attention to compliance. Oral iron chelators are currently being tested, but seem less superior to desferal therapy.14
Pain management
Treatment of pain in sickle cell disease is one of the most daunting tasks of the disease's management. It requires a comprehensive team strategy. The most common form of pain occurs during vaso-occlusive episodes, which may start as early as 6 months of age and continue in an unpredictable manner throughout the patient's life. Pain rates are inversely proportional to the concentrations of haemoglobin and fetal haemoglobin.31 The inability of providers to adequately treat pain, due to misperceptions about the cause of the pain and fear of opioid addiction, are some of the most major barriers to successful pain control.
The cause of acute pain includes vaso-occlusive episodes, acute chest syndrome, and other serious complications. Chronic pain often results from orthopaedic problems, such as avascular necrosis, vertebral collapse, or chronic arthritis. Acute pain, which is not treatable at home, requires parenteral opioids and hydration. Algorithms for treating pain are available.4,5
Day units, where patients can be seen quickly in hospital and avoid the emergency room, decrease the admission rates for pain and provide excellent resources for patients.4 The use of patient controlled analgesia devices is effective for improved narcotic delivery.32 Non-steroidal analgesia can be an excellent adjunct to narcotics. Hydroxyurea has been shown to be effective in the prevention of pain and other complications of sickling.33,34
New therapies
Of all the new therapies for treating sickle cell disease, hydroxyurea, a ribonuclease reductase inhibitor, is the most promising. Several paediatric and adult trials have reported decreases in pain, the incidence of acute chest syndrome, and overall mortality (table 2). Hydroxyurea was initially developed because of its ability to increase fetal haemoglobin production (a known inhibitor of sickle polymerisation), but it has other beneficial effects.35 These include a decrease in white blood counts, increased red cell deformability, and altered adhesive receptors on sickle reticulocytes. Toxicity is mainly related to cytopenias, and patients must be carefully monitored, especially early in therapy. This drug should not be used to treat acute pain.
Table 2.
Clinical effect and laboratory changes induced by hydroxyurea in 17 children with sickle cell anaemia. Values are means (SDs) unless stated otherwise
| Before hydroxyurea | After hydroxyurea | P value | |
|---|---|---|---|
| Clinical effect | |||
| Annual rate: | |||
| Vaso-occlusion | 3.1 (0.5) | 1.2 (0.4) | 0.0105 |
| Acute chest syndrome | 1.5 (0.5) | 0.2 (0.1) | 0.0417 |
| Blood transfusion | 1.8 (0.5) | 0.4 (0.2) | 0.0168 |
| Days in hospital | 29.1 (4.8) | 8.1 (2.8) | 0.0017 |
| Laboratory changes | |||
| Haemoglobin concentration (g/l) | 80.9 (40.3) | 100.2 (30.6) | 0.0001 |
| Reticulocyte count (10−3/μl) | 382.0 (38.0) | 232.0 (26.0) | 0.0053 |
| Fetal haemoglobin (% total haemoglobin) | 7.7 (1.6) | 16.7 (1.8) | 0.0001 |
| Mean cell volume (μm3) | 87.0 (7.0) | 104.0 (3.0) | 0.0001 |
| Absolute neutrophil count (10−3/μl) | 6.8 (0.7) | 4.5 (0.5) | 0.0087 |
| Platelet count (10−3/μl) | 502 (45) | 355 (41) | 0.0145 |
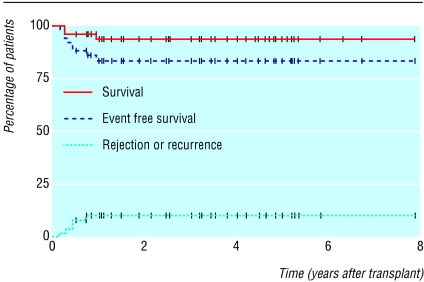
Other new therapies being evaluated include drugs to increase fetal haemoglobin concentration, including butyrate and decitabine.20,26 Drugs to prevent dehydration of the sickle red blood cells are also being studied, including clotrimazole, magnesium, and ICA-17043 “quintiles.” Nitric oxide, a potent vasodilator with anti-sickling properties, is being studied.35 Other treatments being pursued include antiadhesion and antioxidative therapies.20 Bone marrow transplantation of children has shown cure in some patients (fig 2).36
Fig 2.
Survival and recurrence rate for patients with sickle cell disease after bone marrow transplantation
Additional educational resources
HELP!: a guide to sickle cell disease programs and services. Los Angeles, CA: National Association for Sickle Cell Disease. Resources, referrals, and information for patients with sickle cell disease in the United States, Bahamas, Puerto Rico, and the Virgin Islands.
The Emory University School of Medicine (www.scinfo.org)—a comprehensive website that provides education, news, research updates, and resources for patients with sickle cell disease and professionals.
Information for patients
Earles A, Lessing S, Vichinsky E. A parents' handbook for sickle cell disease, part I and part II. California Department of Health Services, Genetic Disease Branch: Sacramento, CA. Part I published in 1991, part II published in 1993. A user friendly resource for patients with sickle cell disease and their families.
Funding: None.
Contributors: Both authors extracted the information and wrote the paper. They will act as guarantors for the paper.
Competing interests: None declared.
References
- 1.Stuart MJ, Setty BN. Acute chest syndrome of sickle cell disease: new light on an old problem. Curr Opin Hematol 2001;8(2): 111-22. [DOI] [PubMed] [Google Scholar]
- 2.Hagar RW, Vichinsky EP. Major changes in sickle cell disease. Adv Pediatr 2000;47: 249-72. [PubMed] [Google Scholar]
- 3.Vichinsky E, Hurst D, Earles A, Kleman K, Lubin B. Newborn screening for sickle cell disease: effect on mortality. Pediatrics 1988;81: 749-55. [PubMed] [Google Scholar]
- 4.Benjamin LJ, Swinson GI, Nagel RL. Sickle cell anemia day hospital: an approach for the management of uncomplicated painful crises. Blood 2000;95: 1130-6. [PubMed] [Google Scholar]
- 5.National Institutes of Health, National Heart, Lung, and Blood Institute, and Division of Blood Diseases and Resources, The management of sickle cell disease. Bethesda, MD: NIH. (NIH Publication No 02-2117.)
- 6.Smith J, Espeland M, Bellevue R, Bonds D, Brown AK, Koshy M. Pregnancy in sickle cell disease: experience of the Cooperative Study of Sickle Cell Disease. Obstet Gynecol 1996;87: 199-204. [DOI] [PubMed] [Google Scholar]
- 7.Miller S, Sleeper LA, Pegelow CH, Enos LE, Wang WC, Weiner SJ, et al. Prediction of adverse outcomes in children with sickle cell disease. N Engl J Med 2000;342: 1612-3. [DOI] [PubMed] [Google Scholar]
- 8.Section on Hematology/Oncology Committee on Genetics; American Academy of Pediatrics. Health supervision for children sickle cell disease. Pediatrics 2002;109: 526-35. [DOI] [PubMed] [Google Scholar]
- 9.Falletta JM, Woods GM, Verter JI, Buchanan GR, Pegelow CH, Iyer RV, et al. Discontinuing penicillin prophylaxis in children with sickle cell anemia. Prophylactic Penicillin Study II. J Pediatr 1995;127: 685-90. [DOI] [PubMed] [Google Scholar]
- 10.Hord J, Byrd R, Stowe L. Streptococcus pneumoniae sepsis and meningitis during the penicillin prophylaxis era in children with sickle cell disease. J Pediatr Hematol Oncol 2002;24: 470-2. [DOI] [PubMed] [Google Scholar]
- 11.Ohene-Frempong K. Stroke in sickle cell disease: demographic, clinical, and therapeutic considerations. Semin Hematol 1991;28: 213-9. [PubMed] [Google Scholar]
- 12.Adams RJ, Mckie VC, Bramilla D, Carl E, Gallagher D, Nichols FT, et al. Stroke prevention trial in sickle cell anemia. Control Clin Trials 1998;19: 110-29. [DOI] [PubMed] [Google Scholar]
- 13.Ballas S. Ethical issues in the management of sickle cell pain. Am J Hematol 2001;68: 127-32. [DOI] [PubMed] [Google Scholar]
- 14.Vichinsky E, ed. Transfusion-related iron overload in sickle cell anemia. Seminars in hematology, ed. NS Young and P Beris. Vol 38(No 1, Suppl 1): 1-5, 14-23. Philadelphia, PA: Harcourt Health Sciences, 2001. [Google Scholar]
- 15.Miller ST, Macklin EA, Pegelow CH, Kinney TR, Sleeper LA, Bello JA, et al. Silent infarction as a risk factor for overt stroke in children with sickle cell anemia: a report from the Cooperative Study of Sickle Cell Disease. J Pediatr 2001;39: 385-90. [DOI] [PubMed] [Google Scholar]
- 16.Moser FG, Miller ST, Bello JA, Pegelow CH, Zimmerman RA, Wang WC, et al. The spectrum of brain MR abnormalities in sickle-cell disease: a report from the Cooperative Study of Sickle Cell Disease. Am J Neuroradiol 1996;17: 965-72. [PMC free article] [PubMed] [Google Scholar]
- 17.Styles LA, Vichinsky EP. Core decompression in avascular necrosis of the hip in sickle-cell disease. Am J Hematol 1996;52: 103-7. [DOI] [PubMed] [Google Scholar]
- 18.Ataga KI, Orringer E. Renal abnormalities in sickle cell disease. Am J Hematol 2000;63: 205-11. [DOI] [PubMed] [Google Scholar]
- 19.Abbott KC, Hypolite IO, Agodoa LY. Sickle cell nephropathy at end-stage renal disease in the United States: patient characteristics and survival. Clin Nephrol 2002;58: 9-15. [DOI] [PubMed] [Google Scholar]
- 20.Vichinsky EP. New therapies in sickle cell disease. Lancet 2002;360: 629-31. [DOI] [PubMed] [Google Scholar]
- 21.Okpala I, Westerdale N, Jegede T, Cheung B. Etilefrine for the prevention of priapism in adult sickle cell disease. Br J Haematol 2002;118: 918-21. [DOI] [PubMed] [Google Scholar]
- 22.Vichinsky EP, Styles LA, Colangelo LH, Wright EC, Castro O, Nickerson B. Acute chest syndrome in sickle cell disease: clinical presentation and course. Cooperative Study of Sickle Cell Disease. Blood 1997;89: 1787-92. [PubMed] [Google Scholar]
- 23.Vichinsky EP, Neumayr LD, Earles AN, Williams R, Lennette CT, Dean D, et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med 2000;342: 1855-65. [DOI] [PubMed] [Google Scholar]
- 24.Dean D, Neumayr L, Kelly DM, Ballas SK, Kleman K, Roberston S, et al. Chlamydia pneumoniae and acute chest syndrome in patients with sickle cell disease. J Pediatr Hematol Oncol 2003;25: 46-55. [DOI] [PubMed] [Google Scholar]
- 25.Castro OL, Hoque M, Brown BD. Pulmonary hypertension in sickle cell disease: cardiac catheterization results and survival. Blood 2002.3: 3. [DOI] [PubMed] [Google Scholar]
- 26.Young NS, Beris P, eds. New therapies of sickle cell anemia and B-thalassemia. Seminars in hematology, ed. GP Rodgers and EA Rachmilewitz. Vol 38. Philadelphia, PA: Harcourt Health Sciences, 2001: 435.
- 27.Morris CR. Arginine therapy: a new treatment for pulmonary hypertension in sickle cell disease? Blood 2000;6(11):(Suppl, part 1) 485a. [DOI] [PubMed] [Google Scholar]
- 28.Styles LA, Vichinsky E. Effects of a long-term transfusion regimen on sickle cell-related illnesses. J Pediatr 1994;125(6 Pt 1): 909-11. [DOI] [PubMed] [Google Scholar]
- 29.Schmalzer EA, Lee JO, Brown AK, Usami S, Chion S. Viscosity of mixtures of sickle cells and normal red cells at varying hematocrit levels: implications for transfusion. Transfusion 1987;1987: 228-33. [DOI] [PubMed] [Google Scholar]
- 30.Brittenham GM, Cohen AR, McLaren CE, Martin MB, Griffith PM, Nienhuis AW, et al. Hepatic iron stores and plasma ferritin concentrationin patients with sickle cell anemia and thalassemia major. Am J Hematol 1993;42: 81-5. [DOI] [PubMed] [Google Scholar]
- 31.Platt OS, Thorington BD, Brambilla DJ, Milner PF, Rosse WF, Vichinsky EP, et al. Pain in sickle cell disease. Rates and risk factors. N Engl J Med 1991;325: 11-6. [DOI] [PubMed] [Google Scholar]
- 32.Vichinsky EP. Comprehensive care in sickle cell disease: its impact on morbidity and mortality. Semin Hematol 1991;28: 220-6. [PubMed] [Google Scholar]
- 33.Olivieri NF, Vichinsky EP. Hydroxyurea in children with sickle cell disease: impact on splenic function and compliance with therapy. J Pediatr Hematol Oncol 1998;20: 26-31. [DOI] [PubMed] [Google Scholar]
- 34.Charache S, Terrin ML, Moore RD, Dover GJ, McMahon RP, Barlow FB, et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. N Engl J Med 1995;332: 1317-22. [DOI] [PubMed] [Google Scholar]
- 35.Morris CR, Kuypers FA, Larkin S, Sweeters N, Simon J, Vichinsky EP, et al. Arginine therapy: a novel strategy to induce nitric oxide production in sickle cell disease. Br J Haematol 2000;111: 498-500. [DOI] [PubMed] [Google Scholar]
- 36.Walters MC, Storb R, Patience M, Leisenring W, Taylor T, Sanders JE, et al. Impact of bone marrow transplantation for symptomatic sickle cell disease: an interim report. Multicenter investigation of bone marrow transplantation for sickle cell disease. Blood 2000;95: 1918-24. [PubMed] [Google Scholar]