Abstract
Summary: Throughout much of the world, Trichinella spp. are found to be the causative agents of human trichinellosis, a disease that not only is a public health hazard by affecting human patients but also represents an economic problem in porcine animal production and food safety. Due to the predominantly zoonotic importance of infection, the main efforts in many countries have focused on the control of Trichinella or the elimination of Trichinella from the food chain. The most important source of human infection worldwide is the domestic pig, but, e.g., in Europe, meats of horses and wild boars have played a significant role during outbreaks within the past 3 decades. Infection of humans occurs with the ingestion of Trichinella larvae that are encysted in muscle tissue of domestic or wild animal meat. Early clinical diagnosis of trichinellosis is rather difficult because pathognomonic signs or symptoms are lacking. Subsequent chronic forms of the disease are not easy to diagnose, irrespective of parameters including clinical findings, laboratory findings (nonspecific laboratory parameters such as eosinophilia, muscle enzymes, and serology), and epidemiological investigations. New regulations laying down rules for official controls for Trichinella in meat in order to improve food safety for consumers have recently been released in Europe. The evidence that the disease can be monitored and to some extent controlled with a rigorous reporting and testing system in place should be motivation to expand appropriate programs worldwide.
INTRODUCTION
Throughout much of the world, Trichinella spp. have been found to be the causative agents of human trichinellosis, a disease that not only is a public health hazard by affecting human patients but also represents an economic problem in porcine animal production and food safety. Due to the predominantly zoonotic importance of infection, the main efforts in many countries have focused on the control or elimination of Trichinella from the food chain. The most important source of human infection worldwide is the domestic pig, but, e.g., in Europe, meats of horses and wild boars have played a significant role during outbreaks within the past three decades. Infection of humans occurs with the ingestion of Trichinella larvae that are encysted in muscle tissue of meat from domestic or wild animals. In humans, the lowest infectious dose causing disease is not clearly defined. Dupouy-Camet and Bruschi (29) estimated that approximately 100 and 300 larvae of Trichinella spiralis start to cause disease and that an intake of 1,000 to 3,000 or more larvae causes severe disease, but this estimate was not based on scientific data, and consequently, it does not have any practical value.
Due to political and economic changes, recent increases in prevalence and incidence have been observed in many former eastern European countries (8, 19, 24). Such increases have been related mainly to a reduced efficacy of the veterinary control on susceptible production animals. This represents a serious problem for the meat trade within the European Union and for the exportation of pork outside European Union countries. Proposed solutions include the definition of regions with a negligible risk for Trichinella in fattening pigs or the certification of Trichinella-free pig production units. As a consequence of the emerging European problem, the European Union and some associated non-European Union member countries implemented a Trichinella monitoring program for pigs, horses, wild boar, and other wildlife species (32). The European Commission has implemented a new regulation, regulation no. 2075/2005, laying down specific rules for the official controls of Trichinella in meat in order to improve food safety for European consumers. In the United States, a pilot program for Trichinella-free pig production has been developed (141). The described Trichinella certification mechanism allows the establishment of a process for ensuring the Trichinella safety of swine and, ultimately, food products derived from swine at the production level. Estimation of the likely impact of trichinellosis in nonindustrialized countries with reference to health, social, and economic costs is very difficult. There is considerable uncertainty regarding the prevalence and significant underestimation of incidence because of the lack of access to standardized approaches to diagnosis and reporting of trichinellosis in animals and humans. In the same context, the effects of globalization in exacerbating the risk of spreading trichinellosis are mediated not only through the movement and travel activity of people but also by the increased movement of livestock, wildlife, and potentially infectious food products.
BIOLOGY AND EPIDEMIOLOGY
Life Cycle
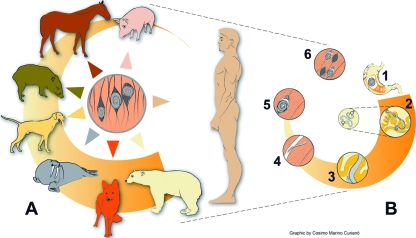
The life cycle of all species of the genus Trichinella principally comprises two generations in the same host (Fig. 1) and includes a very broad range of host species (mammals, birds, and reptiles), although only humans become clinically affected. Following delivery by the gravid female worm, which lives within the intestinal mucosa of the host, newborn larvae (NBL) migrate directly into predominantly lymphatic and blood vessels of the host. This allows them to be transported to predilection sites (highly oxygenated muscles), where they penetrate. It is likely that NBL enter in the striated muscle cells by the aid of its stylet. Penetration mechanisms involving enzymes have been suspected but have not yet been ruled out (21). In experimental infections in which NBL were injected directly into muscles, the penetration occurred as early as 10 min after injection (22). Within such muscle nurse cells, NBL develop to the infective muscle-stage larvae without molting (this L1 is 0.65 to 1.45 mm in length and 0.026 to 0.040 mm in width). This maturation terminates within approximately 15 days. In muscle nurse cells, parasite larvae can survive for years (up to 40 years in humans and over 20 years, e.g., in polar bears) (44, 77). After a period of time that is under the influence of the host species, its immune response, which can change among individuals within a given species, and the Trichinella species or genotype, calcification of the collagen capsule first and of the nurse cell and larva can occur. This hypobiotic stage is maintained until being ingested by a new host. Following such an ingestion, parasite larvae are released upon gastric digestion in the new host, and the first-stage larval parasite subsequently reaches the duodenum and, embedded in the intestinal mucosa, undergoes four molts, thus developing into the adult stage within a very short time of 2 days. Males and females copulate, and 5 to 7 days postinfection (p.i.), the females start to deliver new generations of NBL. Within several weeks, an intestinally immune-mediated host response becomes established, and immune effector mechanisms affect the viability of the female parasites, resulting in a continuous expulsion of adult worms (122).
FIG. 1.
Trichinella sp. life cycle. (A) Main sources of Trichinella sp. infections for humans (including pigs, horses, wild boars, dogs, walruses, foxes, and bears). (B) Trichinella sp. cycle in the host body. In the enteral phase, muscle tissues are digested in the stomach, and larvae are released (1); larvae penetrate the intestinal mucosa of the small intestine and reach the adult stage within 48 h p.i., and male and female mate (2); female worm releases newborn larvae in the lymphatic vessels (from the fifth day p.i. onwards; the length of newborn production, from 1 week to several weeks, is under the influence of host immunity) (3). In the parenteral phase, the newborn larvae reach the striated muscle and actively penetrate in the muscle cell (4); the larva grow to the infective stage in the nurse cell (the former muscle cell) (5); and, after a period of time (weeks, months, or years), a calcification process occurs (6). (Modified from www.iss.it/site/Trichinella/index.asp with permission of the publisher.)
Species and Taxonomy
Conversely to the older conventional scientific recognition of Trichinella spiralis as the only member of the genus Trichinella (13), more recent studies of the genetic diversity and zoogeographical and epidemiological peculiarities within this genus yielded a new Trichinella taxonomy encompassing eight species (Table 1) (66, 76, 126, 134). All 12 recognized taxa are genetically and biologically delineated into two distinct clades characterized by the presence or absence of an intramuscular collagen capsule (167). Thus, one clade is represented by all species and taxa that accordingly encapsulate in host muscle tissue of mammals only, and the other one does not encapsulate after muscle cell dedifferentiation and infects mammals, birds (one species), and even some reptiles (125). Today's identification of samples to the species level and genotyping are based primarily upon molecular means (133). Information on species and genotype distribution and host range can be downloaded from the website of the International Trichinella Reference Centre (www.iss.it/site/Trichinella/index.asp).
TABLE 1.
Main epidemiological features of Trichinella species and genotypesa
| Species or genotype | Geographical distribution | Host range | Main source of infection of humans | Resistance of larvae in frozen muscles |
|---|---|---|---|---|
| Encapsulated | ||||
| T. spiralis | Cosmopolitan | Domestic and sylvatic mammals | Domestic and sylvatic swine horses | Yes in horse muscles |
| T. nativa | Arctic and subarctic areas of America, Asia, Europe | Sylvatic carnivores | Bears, walruses | Yes in carnivore muscles |
| Trichinella genotype T6 | Canada, Alaska, Rocky Mountains, and Appalachian Mountains in the United States | Sylvatic carnivores | Carnivores | Yes in carnivore muscles |
| T. britovi | Temperate areas of Europe and Asia, Northern and Western Africa | Sylvatic mammals and seldomly domestic pigs | Wild boars, domestic pigs horses, foxes, jackals | Yes in carnivore and horse muscles |
| Trichinella T8 | South Africa and Namibia | Sylvatic carnivores | None documented | No |
| T. murrelli | United States and Southern Canada | Sylvatic carnivores | Bears, horses | No |
| Trichinella genotype T9 | Japan | Sylvatic carnivores | None documented | No |
| T. nelsoni | Eastern-Southern Africa | Sylvatic mammals | Warthogs, bush pigs | No |
| Trichinella genotype T12 | Argentina | Cougars | None documented | Unknown |
| Nonencapsulated | ||||
| T. pseudospiralis | Cosmopolitan | Sylvatic mammals and birds, domestic pigs | Domestic and wild pigs | No |
| T. papuae | Papua New Guinea, Thailand | Wild pigs, saltwater crocodiles | Wild pigs | No |
| T. zimbabwensis | Zimbabwe, Mozambique, Ethiopia, South Africa | Nile crocodiles, monitor lizards | None documented | No |
Based on data from reference 125.
Trichinella spiralis.
Trichinella spiralis is the species most adapted to domestic and wild swine but can also include synanthropic rats in its life cycle. T. spiralis exhibits a wide and global distribution (Table 1 and Fig. 2). This species is also the most important etiological agent to cause disease in humans (125). Conversely to the domestic cycle, the sylvatic cycle of T. spiralis includes a broad range of wild carnivores, which may, however, become the origin of a life cycle introduction into a domestic host population (23, 120). In the domestic cycle, pork scraps from T. spiralis-infected pigs are the main source of infection for synanthropic animals (e.g., rats, horses, stray cats, and dogs [no Trichinella infection has been detected in urban wild animals so far, such as the red fox]) (137).
FIG. 2.
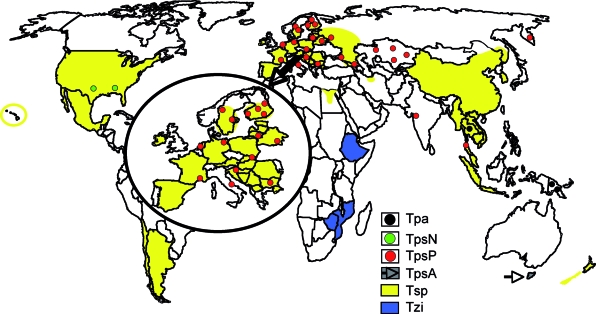
World map showing the distribution areas of Trichinella spiralis (Tsp), Trichinella pseudospiralis from north America (TpsN), T. pseudospiralis from Europe and Asia (TpsP), T. pseudospiralis from Tasmania (TpsA), Trichinella papuae (Tpa), and Trichinella zimbabwensis (Tzi). (Modified from www.iss.it/site/Trichinella/index.asp with permission of the publisher.)
Trichinella nativa and Trichinella genotype T6.
Trichinella nativa affects predominantly sylvatic carnivores living in frigid zones of Asia, northern states of North America, and Northeastern Europe, while the closely related Trichinella genotype T6 appears to be restricted to several regions of Canada (British Columbia, Ontario, Manitoba, and Nunavut) and the United States (Alaska, Montana, Idaho, and Pennsylvania) (Table 1 and Fig. 3). The main hosts are terrestrial (e.g., brown and black bears, wolverines, raccoons, lynxes, wolves, and foxes) and marine (e.g., polar bears, walruses, and seals) carnivores (23, 39, 66, 80, 131). This species developed the ability of muscle-stage larvae to survive in frozen muscles of carnivores for up to 5 years (23). Humans who are at risk for infection are meat-consuming people living in frigid zones of Canada, Greenland, and Russia (98, 147, 149) or hunters from Europe and the United States who consume raw or undercooked meat from bears hunted in arctic or subarctic regions (2).
FIG. 3.
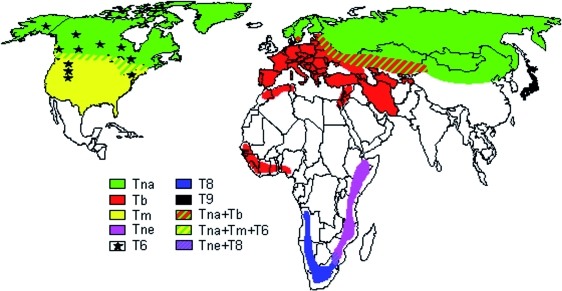
World map showing the distribution areas of Trichinella nativa (Tna), Trichinella britovi (Tb), Trichinella murrelli (Tm), Trichinella nelsoni (Tne), Trichinella genotype T6 (T6), Trichinella genotype T8 (T8), and Trichinella genotype T9 (T9). In some regions, the distribution areas of these encapsulated species and genotypes overlap between them. (Modified from www.iss.it/site/Trichinella/index.asp with permission of the publisher.)
Trichinella britovi.
Trichinella britovi is the most widely distributed species within sylvatic life cycles of Europe, Asia, and Northern and Western Africa (123, 136, 137). As is the case with T. spiralis, T. britovi can also affect domestic pig populations mainly via extensive grazing systems or feed with scraps or carrion originating from sylvatic carnivores. Zoonotically, T. britovi is the second-most common species of Trichinella that may affect human health.
Trichinella murrelli.
Trichinella murelli is spread among sylvatic carnivores across the United States and some southern regions of Canada. This species does not develop in swine. It is the causative agent of infection in humans, especially following the consumption of meat originating from hunted black bears. Valuable clinical information on this species was gained from a 1985 outbreak in France due to the consumption of horse meat imported from the United States (3).
Trichinella nelsoni.
Trichinella nelsoni has been detected in Eastern Africa, from Kenya to South Africa (88). The host range includes sylvatic carnivores and, at least occasionally, bush pigs and warthogs, some of which have been the source of infection for humans. Less than 100 human infections have been documented for this species in Kenya and Tanzania (123).
Genotypes T8, T9, and T12.
Trichinella genotype T8, very similar to T. britovi, has been identified in wild animals of South Africa and Namibia (88). No human case due to this genotype has been documented. Trichinella isolates from Japanese wildlife, originally identified as being T. britovi, are now designated a separate genotype, named Trichinella genotype T9, which is phylogenetically related to T. murrelli (125, 167). Finally, Trichinella genotype T12 is a new encapsulated genotype of Trichinella recently detected in a mountain lion (Puma concolor) from Trapalco, Patagonia, Río Negro, Argentina. The only information available is the molecular structure of two noncoding sequences and one coding sequence that are different from those of the 11 currently recognized species and/or genotypes of the genus Trichinella (76).
All species and/or genotypes described so far are characterized by one common biological feature: they all induce the development of a thick collagen capsule, which can be detected by light microscopy, during the muscle phase of infection. Conversely, three other species produce only a thin capsule detectable by electron microscopy only.
Trichinella pseudospiralis.
Trichinella pseudospiralis exhibits a cosmopolitan distribution and infects both mammals and birds. Three genetically distinct populations can be distinguished, each referring to a specific geographical origin: Palaearctic, Nearctic, and Australian (Tasmania) origins (79, 165). T. pseudospiralis has been found in 14 mammalian host species including domestic and sylvatic swine and 13 avian species (121), where the number of reports for mammals is much higher than that for birds. Cases of trichinellosis in humans with some deaths have been documented in Kamchatka, Thailand, and France (125).
Trichinella papuae.
Trichinella papuae circulates in both mammals and reptiles (domestic sows, wild pigs, and farmed saltwater crocodiles) of Papua New Guinea and Thailand (122, 135). Infections in humans have been documented (113).
Trichinella zimbabwensis.
Trichinella zimbabwensis, very similar to T. papuae, has been detected only in wild and farmed reptiles of Africa (Zimbabwe, Mozambique, South Africa, and Ethiopia), although experimentally, it is able to infect mammals (129, 130). Human infections are not known so far.
Epidemiology in Animals
Parasites of the genus Trichinella are present on all continents except Antarctica, where no report or investigation of these parasites has been released or carried out so far (123). Most of the species, with the exception of T. spiralis, parasitize predominantly wild animals. A switch from wild animals to domestic animals can occur when there is an improper management in segregating husbandry and wildlife. Domestic cycles and the sylvatic cycle can function either independently from each other or interactively (123).
The term “domestic cycle” refers to the transmission pattern where the focus is on a swine herd being fed, e.g., uncooked pork scraps, carrion, garbage (i.e., garbage-fed pigs), or the pigs can feed on carcasses that are not promptly removed from the farm; transmission can also become domestic via synanthropic animals living near the swine herd (e.g., rats and mustelides). Horses fattened with pork scraps or with carcasses of fur animals became infected with Trichinella. Similarly, infections in sled dogs fed with carcasses of other dogs or of game from the arctic, including carcasses of slaughtered fur animals, were reported. The use of meat of slaughtered crocodiles to feed other farmed crocodiles has been reported as well (135). With regard to the geographic distribution of the domestic cycle of Trichinella, since World War II, there have been no reports of infections on industrialized farms in Canada, the United States, and western Europe; the domestic cycle has recently been reported only among small swine herds of Southern Finland and certain regions of Spain where control measures were not adopted (119). In several countries of Central-Eastern Europe, the transient breakdown of governmental veterinary services and state farms accompanied by economic problems and war have resulted in sharp increases in the incidence of Trichinella infection among domestic pig herds, with prevalence rates reaching 50% in some villages in the 1990s (94). In Canada, the United States, and most European Union countries, Trichinella infection in domestic animals has virtually disappeared, although sporadic foci do occur (6, 127). In South and Central America, Trichinella infection is still endemic in Argentina, Chile, and Mexico in both humans and pigs (111, 144, 148). In East Asia, the domestic cycle occurs in China (158). Foci of Trichinella infection involving swine and humans are also widespread in Thailand, Indonesia, Laos, Malaysia, and Myanmar (123). Occasionally, T. britovi can be transmitted within the domestic cycle when humans feed pigs with game meat scraps or when “pasture” pigs had access to dumps containing offal of sylvatic animals (120). T. pseudospiralis has also been transmitted to domestic pigs and rats on farms in Croatia, Kamchatka, Russia, and the Slovak Republic (61).
The “sylvatic cycle” oscillates between wildlife hosts and also includes all Trichinella species and genotypes, including T. spiralis for mammals, T. pseudospiralis for mammals and birds, and T. papuae and T. zimbabwensis for mammals and reptiles. Peroral infection occurs either after ingestion of muscle tissue from an infectious prey animal or by consumption of infectious tissue from a carrion of a homologous (the former actually representing “cannibalism”) or a heterologous species. Natural Trichinella infections have been reported for more than 100 species of mammals, seven avian species, and three reptile species (121). Despite the potential broad host spectrum for Trichinella spp., the predominant biotic potential concerns carnivores (13, 23) and porcine omnivores (mainly domestic pigs, different races of wild pigs, wild boars, bush pigs, and warthogs) (13, 121). One of the most important biological factors promoting transmission is the physiological ability of muscle-stage larvae to survive in decaying carcasses/carrion. Thus, even nonencapsulated larvae of T. papuae retained their infectivity in decaying tissues of a pig exposed at 35°C for 9 days (114). Encapsulated larvae of T. spiralis have been found to be infective for laboratory animals up to 4 months in extremely rotten meat (85). Encapsulated larvae of T. britovi and T. nelsoni in mouse carcasses packed in plastic vials have been found to be infective for laboratory animals up to 45 days at room temperature even if the muscle tissues were completely liquefied (E. Pozio, unpublished data). The importance of this well-established environmental adaptation is underscored by survival even at low freezing temperatures, as, e.g., T. britovi can survive in frozen carrion for up to 1 year, and T. nativa and Trichinella genotype T6 can survive for up to several years, maintaining infectivity for future hosts (23, 132). The anaerobic metabolism favoring survival in putrefying flesh along with the ability of larvae of some species to survive freezing are two separate mechanisms that strongly increase the survival of the parasite in nature. It is important to stress that the survival of muscle larvae after freezing occurs mainly when these larvae parasitize striated muscles of carnivores (bears, wolves, and foxes, etc.), whereas the survival time after freezing is strongly reduced to a few days or weeks, when muscle larvae of the same strain parasitize other mammalian hosts such as swine and rodents.
DIAGNOSIS OF ANIMALS
Direct Methods
Meat inspection for the detection of Trichinella larvae is designed to prevent clinical trichinellosis in humans but not to prevent infection. The identification of Trichinella larvae in muscle samples from pigs and other animal species intended for human consumption (e.g., horses, wild boars, and bears) is limited to postmortem inspection of carcasses (49). Direct detection is also applied in wildlife monitoring, where indicator animals (e.g., foxes or raccoon dogs) are examined to assess the prevalence of Trichinella infection among the wildlife reservoir and the risk of introduction into domestic animals. Methods to detect Trichinella larvae in muscle samples need to be highly sensitive, and performance is greatly influenced by the sample size, the muscle type selected for sampling, and the specific method used (103).
Host animals ingesting even high numbers of Trichinella larvae from infectious meat will not develop clinical symptoms such as those observed in human patients. Therefore, “Trichinella infection” rather than “trichinellosis” should be used for animals.
In order to identify predilection sites and, in particular, animal species that optimal for diagnostic investigations, several experimental studies using doses that mimic natural infections have been performed. Thus, in domestic swine, the three main predilection sites for T. spiralis are the diaphragm crus, the tongue, and the masseter muscle (41, 49), and analogous results were observed in experimental T. britovi and T. pseudospiralis infection in this host species (104). Some of the sampling sites recommended by the International Commission on Trichinellosis (http://monsite.wanadoo.fr/intcomtrichinellosis/) for different domestic and wild animals subjected to meat inspection or epidemiological studies are summarized in Table 2.
TABLE 2.
Predilection sites for Trichinella larvae in different animal speciesa
| Animal species | Predilection sites | Aim of examination |
|---|---|---|
| Domestic swine | Diaphragm, tongue, masseter | Meat inspection (domestic animals) |
| Horse | Tongue, masseter | Meat inspection (domestic animals) |
| Wild boar | Forearm, diaphragm, tongue | Meat inspection (game) |
| Bear | Diaphragm, masseter, tongue | Meat inspection (game) |
| Walrus seal | Tongue, diaphragm, flippers, masseter | Meat inspection (game) |
| Fox | Diaphragm, forearm muscles, tongue | Epidemiological studies (reservoir animals) |
| Raccoon dog | Diaphragm, forearm muscles, tongue | Epidemiological studies (reservoir animals) |
Besides the choice of the diagnostically optimal muscle, an adequate sample size needs to yield an acceptable level of sensitivity for the detection of Trichinella larvae. For the systematic meat inspection of each animal intended for human consumption, a methodical sensitivity of at least 1 to 3 larvae per gram (lpg) of tissue is considered to be the level above which infection constitutes a food safety issue (100). For the routine slaughter inspection of pig carcasses, using the pooled sample digestion method, a minimum of a 1-g sample of tissue from a predilection site should be examined. To reach a higher diagnostic sensitivity, for horse and game meat, 5 and 10 g of predilection muscle should be tested by artificial digestion, respectively (49). The use of the conventional trichinoscopy for testing of a single pig carcass (examination of 28 oat kernel-size pieces of diaphragm muscle corresponding to a 0.5-g sample) is not recommended anymore due to a lack in methodical sensitivity and the fact that larvae of nonencapsulating species such as T. pseudospiralis are very difficult to detect by this approach (68, 100, 108). If the muscles from prescribed predilection sites are not available for inspection, carcasses should be tested using larger amounts of other muscles in order to provide adequate sensitivity. For epidemiological studies of reservoir animals (wildlife), the sample size is recommended to be adjusted to a sensitivity of less than 1 lpg, as the mean infection intensity in wild carnivores is typically very low (49).
Principally, the artificial digestion technique allows the examination of a pool of muscle samples from up to 100 carcasses (103). Although the artificial digestion technique requires more technical equipment, it meets all the requirements for efficiency, cost-effectiveness, and sensitivity, and nonencapsulated Trichinella larvae can be detected by microscopic examination of the digestive fluid very easily. Muscle larvae are released after digestion of the muscle tissue by means of artificial digestion fluid composed of 1% pepsin (1:10,000; U.S. National Formulary) and 1% hydrochloric acid, followed by selective screening, filtration, or sedimentation procedures and a final microscopic examination for the presence of larvae (108). There are various published procedures for the pooled digestion technique for the detection of Trichinella in meat (47, 49), whereas the magnetic stirrer method is considered to be the “gold standard” because it is a method specifically designed for pooled samples, and it has been subjected to validation studies (70). In a validation trial of the magnetic stirrer method, a larval burden of more than 3 lpg was consistently detected with a currently accepted sample size of 1 g from the individual pig carcass, whereas larval burdens of 1.0 to 1.9 lpg required a sample size of 3 to 5 g (41). The magnetic stirrer method has become the method of choice for routine slaughter inspection in most industrialized countries (160). Because one cannot include positive and negative controls during the routine performance of the magnetic stirrer approach, it is compulsory to validate the test prior to using it at the slaughterhouse.
Molecular Techniques
For epidemiological studies and improvement of knowledge on the occurrence and spread of Trichinella spp. in the domestic and sylvatic cycles, all isolates should be identified to the species or genotype level. Since there are no morphological features to specify larvae, molecular diagnosis is used to yield the species or genotype diagnostically recovered. For this purpose, a multiplex PCR has been developed for the simple and unequivocal differentiation of Trichinella species and genotypes. Partial DNA sequence data were generated from the internal transcribed spacers ITS1 and ITS2 and from the expansion segment V region of the rRNA repeat from different Trichinella species and genotypes (166). This multiplex PCR is a sensitive, inexpensive, and rapid molecular approach that can unequivocally identify a single larva at the species and genotype levels (133).
The identification of Trichinella isolates at the species and genotype levels and data collection are provided by the International Trichinella Reference Centre (http://www.iss.it/site/Trichinella/index.asp).
Serology
Animals can be tested for the presence of anti-Trichinella antibodies in the serum or in the meat juice either upon antemortem or upon postmortem examination (108). According to the International Commission on Trichinellosis, indirect methods such as the detection of anti-Trichinella antibodies in domestic and wild animals are not recommended as a substitute for meat inspection of individual carcasses (49). However, Trichinella serology is considered to be suitable for the surveillance and epidemiological investigations of domestic animals and wildlife (51).
The time of seroconversion after a primary Trichinella infection is dependent upon the infection dose and the larval burden in the muscle, as has been demonstrated by comprehensive experimental studies with different Trichinella species in various host animals such as pigs, horses, wild boars, and foxes. Following ingestion of high numbers of T. spiralis larvae, anti-Trichinella immunoglobulin G (IgG) can be detected in animals about 2 to 3 weeks p.i. Conversely, there is a delayed antibody response for several weeks if animals get a low infection dose (Table 3). Analogous results are available for other Trichinella species from experimental studies of pigs where an increasing infection dose of 100, 1,000, and 20,000 larvae of T. pseudospiralis and T. britovi in Yorkshire and Iberian pigs was correlated with a shorter time of seroconversion (104).
TABLE 3.
Relationship between time of seroconversion and infection dose (T. spiralis) in pig, horse, wild boar, and red foxes
| Animal species | Infection dose (no. of larvae/animal) | No. of lpg | Time of seroconversion p.i. (wk) |
|---|---|---|---|
| Swine | 100 | 1.62-6.50b | 5-7 |
| 500 | 18.4-48.6b | 4-5 | |
| 1,000 | 26.3-90.6e | 4-6 | |
| 2,500 | 87.6-99.5b | 4 | |
| 8,000 | 12.1-81.4c | 3 | |
| 20,000 | 699.2-1103.5e | 3-4 | |
| 64,000 | 221.4-466.6c | 2.5-3 | |
| Horse | 1,000 | 0.10-0.26b | 3-4 |
| 4,000 | 0.39-7.8b | 3-7 | |
| 5,000 | 0.02-8.9e | 2-4.5 | |
| 10,000 | 6.6-60.0b | 3-4 | |
| 40,000 | 484-1060d | 2-3 | |
| Wild boar | 10,000 | 43-100e | 3-4 |
| Red fox | 10,000 | 7.7-202.7 | 3 |
a Data for pig based on references 46, 104, and 152; data for horse based on references 47 and 157; data for wild boar based on reference 67; and data for red foxes based on reference 91.
Mean of tongue.
Mean of tongue, masseter, diaphragm, intercostal, psoas, and rectus abdominis.
Mean of masseter.
Mean of diaphragm.
Longitudinal studies revealed that anti-Trichinella antibodies may persist in pigs for a long and presumably indefinite time (106), assuming that in slaughter pigs, it is unlikely that a false-negative result will be obtained at a later stage of infection because of putatively declining antibody levels (103). In an experimental study carried out with wild boars, the antibody level against excretory/secretory (E/S) antigens remained stable in animals infected with T. spiralis, T. britovi, and T. nelsoni, but the decline of antibodies directed against T. nativa, T. murrelli, and Trichinella genotype T6 was associated with the rapid disappearance of larvae in the muscle sample (67) due to the lower susceptibility of wild boar to these species and/or genotypes. Experimental and field studies of horses have demonstrated that the serological response to Trichinella infection in this species is less consistent than that observed in pigs (51). For instance, in naturally infected horses, specific antibodies were not detected in spite of the presence of a high larval burden in muscles (128, 139). Therefore, serological methods cannot be recommended for the examination of single animals or for monitoring horses for Trichinella infection.
In conclusion, from the diagnostic point of view, the early stage of Trichinella infection in the host is characterized by a “diagnostic window” at which false-negative results may occur compared with direct (larval detection) tests (108). During this early stage of infection, however, Trichinella larvae may not yet have reached maturity in the muscle cell (approximately 20 days after infection) to allow infection of the next appropriate host (103).
Blood serum is conventionally the preferred sample matrix for conducting serological tests for Trichinella. However, serum samples of poor quality due to extensive hemolysis or microbial contamination, especially in samples obtained from wild animals, may significantly influence the sensitivity and specificity of the test (108). Other body fluids that can be used for serological testing include plasma, whole blood, and tissue fluids. Results from experimental studies indicate that tissue fluids such as meat juice from slaughtered pigs or from other animals (e.g., wild boars and foxes) may be suitable for serological examinations (46, 71, 91). Comparable correlations and dilution ratios between blood serum and muscle juice were observed in a comparative study addressing pigs, wild boars, and foxes (91).
Enzyme-linked immunosorbent assay (ELISA) is the most commonly used method for the detection of Trichinella infection due mainly to the methodical sensitivity that allows the detection of as low as 1 larva per 100 g of muscle tissue (108). Thus, a large series of experimental and/or field studies has been carried out using pig serum and meat juice samples (46, 47, 62, 95, 102, 106, 152, 155). The specificity of ELISA became greatly improved by utilizing metabolic E/S antigens released from Trichinella muscle larvae and generated upon in vitro maintenance. These E/S antigens consist of a group of structurally related glycoproteins (48, 52). The predominant Trichinella epitopes inducing humoral immunity are localized on the so-called TSL-1 antigen, which is found in the stichocyte cells and on the surface of the parasite's cuticle and which is secreted by first-stage larvae in the muscle (5, 112). TSL-1 antigen epitopes are highly conserved, and a high cross-reactivity between Trichinella antigens derived from different species and genotypes was revealed, allowing serology to detect infection with all of these Trichinella species (5, 51, 69, 86, 104). Tyvelose has been identified as being a major carbohydrate epitope of the TSL-1 antigen, and a synthetic variant of tyvelose has been developed for use in ELISA (143, 161). This synthetic carbohydrate antigen offers the advantages of stability and standardization and shows a higher test specificity in many host species (20). Unfortunately, the tyvelose antigen suffers from sensitivity problems in some instances (51). Results from studies of Trichinella seroprevalence in sylvatic and domestic animals demonstrated a less sensitive but very specific antibody response against synthetic carbohydrate compared to E/S antigen. Therefore, tyvelose ELSIA can be used for confirmatory testing (91).
Multiple experimental and field studies have been carried out using swine to evaluate ELISA using E/S antigens. The infection status of pigs was determined by the digestion of diaphragm muscle samples. The sensitivity of ELISA ranged from between 93.1 and 99.2%, whereas the specificity varied from 90.6 to 99.4% after examination of serum samples of pigs which originated from Trichinella-free farms (95, 110, 155). In a validation study for testing of pigs using tyvelose antigen ELISA, the sensitivity and specificity were 94.3 and 96.7%, respectively, compared with the E/S ELISA, which had specificity and sensitivity of 84.9 and 96.0%, respectively (42).
Epidemiological Investigation
Outbreaks of trichinellosis can cause severe harm to the affected group of persons. By definition, a trichinellosis outbreak requires at least one case that must be laboratory confirmed (http://www.med.unipi.it/ict/outbr1.htm). Associated cases should be reported as confirmed if the patient shared an epidemiologically implicated meal or ate an epidemiologically implicated meat product and has either a positive serological test for trichinellosis or a clinically compatible illness (http://www.cdc.gov/epo/dphsi/casedef/trichinosis_current.htm). If two or more persons in the same household or a number of persons in the same community have high fever, periorbital or facial edema, and myalgia, trichinellosis can be suspected. When cases are sporadic or the clinical course is atypical, it is less likely that the infection will be suspected. Thus, the diagnosis can be more complex, e.g., for people who have consumed very small amounts of infected meat over a period of several days. The clinical course will then be atypical, and the clinical diagnosis will be arduous.
Once infection is suspected, information on the consumption of raw or undercooked meat or meat products, including the place and time of purchase (or receipt) and consumption, should be collected (29).
Reviewing past trichinellosis outbreaks provides evidence of the close relationship between clinical findings, diagnostic results, and epidemiological uncoverings conducted by the local public health and veterinary public health authorities. In this context, the fast and correct identification of the infection source and tracing it back to the animal farm are very important issues to assess the maximal number of people who might have been exposed to food contaminated with infective Trichinella larvae (105). Usually, such outbreak studies are conducted as case-control studies on the basis of a close connection between the time (appearance of clinical signs) and location of trichinellosis cases.
TRICHINELLOSIS IN HUMANS
Epidemiology of Trichinellosis in Humans
In approximately 20% of countries around the globe, including predominantly small islands or city-states, where Trichinella sp. infections cannot develop for the lack of potential reservoirs, case numbers are very low, as infections of humans only accidentally occur upon the (legal and illegal) importation of Trichinella-infected meat from abroad. Following reports from the main 55 countries where trichinellosis occurs autochthonously, the yearly total number of clinical trichinellosis was estimated to be 10,000 cases, with a death rate of 0.2% (123).
Trichinella infection in humans is strongly associated with the consumption of raw or undercooked meat; thus, cultural factors such as traditional dishes based on raw or undercooked meat or meat-derived products play an important role in the epidemiology of the disease. Conversely, when a population uniquely consumes well-cooked meat, trichinellosis cases are lacking or very scarce despite persistent wildlife transmission (123). Overall, domestic pork and related products remain the most important source of Trichinella infection in humans, especially when pigs are raised under free-ranging or backyard production conditions. Another important source of infection related to local meat consumption habit is found in France, where, for the past two decades, most trichinellosis cases have been due to the consumption of raw horse meat, a peculiarity related to the French culture (9). In Italy, human infections due to the consumption of horse meat have been documented in only two areas (Emilia Romagna and Lombardy regions in northern Italy and the Apulia region in southern Italy), where the French habit to consume raw horse meat was introduced some centuries ago (120). In China and the Slovak Republic, dog meat was the source of infection for several foci (25, 81). In Romania, the highest prevalence of trichinellosis in humans occurred in the Transylvanian region, where the local ethnic group maintains the food habit of raw meat consumption (8). In Israel, Lebanon, and Syria, where the Judaic and Muslim religions forbid the consumption of pork, human outbreaks of trichinellosis have been documented only following the consumption of pork from wild boars among the Christian Arab population and immigrants from Thailand (33, 54, 57, 89, 109). In Algeria and Senegal, since the majority of the human population is Muslim, trichinellosis has been documented only in Europeans (99, 123). However, the Muslim population is not exempt from acquiring trichinellosis, as shown by the occurrence of a large outbreak of trichinellosis for the consumption of minced beef illegally mixed with pork of unknown origin in Turkey (1). Hunters, their relatives, and their friends are at risk of trichinellosis infection when raw meat from game animals (e.g., bears, cougars, foxes, walruses, and wild pigs) is not tested for Trichinella before consumption (2, 17, 31, 39, 72, 87, 92, 115, 147, 158, 164).
The migratory flow of humans with their own food practices including the consumption of raw meat, the illegal importation of not-controlled meat from endemic to nonendemic countries, and new food practices and dishes including raw meat resulted in outbreaks in Denmark, Germany, Italy, Spain, and the United Kingdom (45, 101, 124, 154). The increasing number of international travelers has resulted in many reports of tourists who acquired Trichinella infections while traveling or hunting in areas of endemicity and subsequently developed disease after their return to their home countries. In most instances, diagnosis was difficult because infections appeared as isolated cases (28, 90, 96, 151).
Clinical Trichinellosis
Principally, a Trichinella infection in the human host can be divided into two phases: an intestinal (or enteral) phase and a muscular (or parenteral or systemic) phase. Infections with low intensities can remain asymptomatic, but parasite burdens higher than a few hundred larvae can initially cause gastroenteritis associated with diarrhea and abdominal pain approximately 2 days p.i. (intestinal acute phase of disease). The pathology is caused by the larvae released into the intestinal mucosa, which subsequently migrate to the blood vessels, by means of which they spread throughout the body until reaching their final location (i.e., the cells of the striated skeletal muscles).
Migrating Trichinella larvae and their metabolites provoke an immediate reaction, which causes immunological, pathological, and metabolic disturbances and the various clinical phenomena observed during the acute stage of the infection (14, 73, 93). The immunological reaction is characterized by infiltrating inflammatory cells (i.e., mast cells, eosinophils, monocytes, and lymphocytes) and a rather Th2-oriented cytokine profile. In this context, eosinophilia is a common characteristic in most cases of trichinellosis. Eosinophils may contribute to immunopathology in trichinellosis due to the release of enzymes such as histaminase and aryl sulfatase. However, eosinophils also induce damage to Trichinella larvae upon activity of the major basic protein, eosinophil cationic protein, and eosinophil peroxidase. The intensity and dynamics of the eosiniphilic phenomenon are dependent on the dose of larvae, the species of Trichinella involved, susceptibility of the host to infection, as well as the time at which the treatment (in particular, the treatment with anthelmintics) has been started (73). The release of histamine, serotonin, a slow-reacting substance of anaphylaxis, bradykinin, and prostaglandins (PGE2, PGD2, and PGJ2) results in an augmented permeability of capillaries and a leakage of fluids, electrolytes, albumins, and cell elements into the surrounding tissue (73). Thus, tissue edema, mainly around the eyes, will be found. Furthermore, another consequence of these inflammatory processes will be vasculitis and fine intravascular thrombi, which represent the principal pathology in the acute stage of trichinellosis.
The production of IgE is another Th2-oriented characteristic; however, an increase in total IgE levels is not a consistent phenomenon found in trichinellosis, and thus, it is not possible to exclude trichinellosis on the basis of its absence. Clinical observations suggest that Trichinella-specific IgE is responsible for the allergic manifestations typical of the clinical picture of trichinellosis, such as cutaneous rash or edemas (38, 159).
Dupouy-Camet and Bruschi (29) described the following the three major cell modifications occurring during the acute stage of infection, the penetration, and, finally, the residence of larvae in the striated skeletal muscle cells: (i) the host cell transformation into a new phenotype called “nurse cell,” accompanied by the disappearance of sarcomere myofibrils; (ii) the encapsulation of the larvae (in the case of encapsulated species); and (iii) the development of a capillary network surrounding the infected cell (15).
In addition to these three major modifications, the sarcoplasm becomes basophilic, the cell nucleus is displaced to the center of the cell, and the nucleoli increase in both number and size. The cell becomes more permeable, resulting in an increased release of muscle enzymes (29). In humans, the calcification of encapsulated T. spiralis larvae may take place after 6 months, but this process is not synchronized and will not simultaneously include all parasites. The time point of calcification appears to depend on the anatomical localization of the parasites as well as other unknown reasons. Calcification of the collagen capsule occurs first, followed by the nurse cell and the larva. The whole process may lead to the death of the larvae, but not all larvae, as some larvae may survive for years in the same host.
The severity of clinical disease upon infection with T. spiralis (and also other species) is strongly dependent on and directly correlated with the number of infective larvae ingested by the person or patient. Thus, infection may result in a large spectrum of clinical forms ranging from asymptomatic to fatality. An overview of such different forms is presented in Table 4.
TABLE 4.
Classification of different clinical forms of trichinellosis, in dependence of the severity of signs and larval densitya
| Clinical form/outcome of infectionb | Serology | Presence of eosinophils (>500 eosinophils per mm3) | Presence of main clinical signs (fever, edema, myalgia) | Recovery after infection | Hypoalbuminemia | Complications | Estimated no. of larvae/g muscle | Hospitalization | Putative fatality |
|---|---|---|---|---|---|---|---|---|---|
| Asymptomatic | + | Transient | − | − | − | − | <10 | − | − |
| Abortive | + | + | Transient (1-2 days) | − | − | − | > | − | − |
| Mild | + | + | + | 3 wk | − | − | > | − | − |
| Pronounced | + | + | ++ | 6 wk | +/− | Rare | > | +/− | − |
| Severe | + | Sometimes absent | +++ | >6 mo | + | Frequent | >100 | + | +/− |
Modified from reference 73 with permission of the publisher, with additional data from reference 153.
The asymptomatic form of trichinellosis relates to a history of exposure, but signs and/or symptoms are lacking. The diagnosis of asymptomatic cases is usually based upon serological findings. In the abortive form, the clinical signs and symptoms are weakly expressed and last up to a few days; diagnosis should also be confirmed by serological testing. The mild form exhibits a low intensity of signs and/or symptoms. No complications are encountered, and serological testing is indispensable in establishing a diagnosis. The pronounced form is characterized by the appearance of the complete syndrome of significant intensity, but complications are rare, and if present, they are benign and vanish soon. The severe form is characterized by the development of the full syndrome of highly pronounced signs and symptoms with metabolic disturbances accompanied by circulatory and/or neurological complications.
Acute-stage trichinellosis.
A Trichinella infection that becomes clinically manifest in a human host starts with nonspecific signs such as uneasiness, headache, fever, fever-associated chills, and, occasionally, gastrointestinal disorders. The fever usually persists for 1 to 3 weeks, depending on the infection dose and severity of disease. Clinical features of this acute stage of infection and disease include pyrexia, eyelid or facial edema, and myalgia as the principal syndrome, occasionally complicated by myocarditis, thromboembolic disease, and encephalitis. One week p.i. (or up to 20 days p.i. in the case of mild infections), but still during the acute stage, high fever (as mentioned above), shivering, myalgia, and symmetrical periorbital or facial edema (invasive or myopathic phase) are characteristic. Some of the patients will present urticarial and maculopapular exanthema and subungual bleedings. These are caused by vasculitis, the leading pathological process of trichinellosis. Other signs are conjunctivitis including subconjunctival bleedings, headache, dry cough, petechial bleedings, and painful movement disorder of the eye muscles. This parenteral or muscular phase is associated with periparasitic inflammatory and allergic responses. Thus, the muscle cell damage stimulates the infiltration of inflammatory cells, primarily eosinophils. A correlation between eosinophil levels and serum muscle enzymes such as lactate dehydrogenase and creatine phosphokinase has been observed in trichinellosis patients, suggesting that muscle damage may be mediated indirectly by these activated granulocytes (36). Subsequently, the regression of eosinophilia occurs with a resulting long-term low but still elevated level for a period of weeks to months.
Although Trichinella larvae do not mature or become encapsulated in heart muscle tissue, their transitory passage can lead to morphological alterations, which consist of focal cellular infiltrates of eosinophils and mononuclear cells. Thus, myocarditis may result as a severe complication in approximately 5 to 20% of cases. Symptomatically, it includes pericardial pain, tachycardia, and electrocardiogram abnormalities such as nonspecific ventricular repolarization disturbances, followed by bundle-branch conduction disturbances, and sinus tachycardia (29). Compensation of the potassium deficit in affected persons provides normalization of the electrocardiogram. Myocarditis should be screened systematically even when specific symptoms are missing; the dosage of serum troponin is a simple and reliable means for such screening and yields positive findings even in asymptomatic myocarditis (78). Hypoalbuminemia, as a result of cardiovascular complications, can lead to edema formation, predominantly in the lower limbs. Echography can identify pericardial effusion or a transitory intracavitary thrombus (2). During the acute stage of disease, ocular affection may result from impaired microcirculation. Clinically, this results in edema and vascular lesion formation within the conjunctiva, the uvea, and the retina. An intense invasion of muscles of the ocular bulb provokes pain when moving the eyeballs, muscle paralysis, diplopia, or a disturbed accommodation (29, 36).
Chronic-stage trichinellosis.
Other complications include encephalitis and secondary infections (bronchopneumonia and sepsis), which usually occur later during infection, i.e., in the transitional phase to the chronic stage of disease (i.e., between weeks 3 and 4 p.i.) or later. Neurological complications, which are rather rare in trichinellosis patients, show multiple small subacute cortical infarcts upon magnetic resonance imaging with gadolinium diethylenetriamine penta-acetic acid (Gd-DTPA) enhancement (37). The electroencephalogram shows a total deceleration of the cortical electric activity without critical aspect. Patients exhibiting such complications can show consciousness disorders or excessive excitement and, frequently, somnolence and apathy. Anisocoria, facial nerve paresis, and Babinsky's reflexes have also been observed in such severe cases. Most brain abnormalities visualized by computed tomography or magnetic resonance imaging as well as the clinical signs and symptoms disappear in 4 to 8 weeks p.i. As mentioned above, pneumonia and pleuritis of bacterial etiology may appear at the late stage of disease (116). Following glucocorticosteroid treatment, the respiratory disturbances regress within a few days. Months or even years after the acute stage, chronic trichinellosis may yield persistent formication, numbness, and excessive sweating (118) as well as impaired muscle strength and conjunctivitis, which may persist up to 10 years p.i. (56) in persons who had not been treated early in the acute phase of infection. Electromyographic disturbances as well as a persistence of inflammatory cells in the muscles can be observed for several years (75).
Although clinical differences among persons infected with different species of Trichinella have been observed (11), it has not been possible to attribute these differences to the species of the pathogen because the number of infecting larvae ingested by each person was generally unknown. However, T. spiralis infections could be more severe than those caused by T. britovi, and this could be due to the fact that T. britovi females are less prolific (140). T. murrelli seems to be more likely to provoke skin reactions and less likely to cause facial edema (27). T. pseudospiralis, which is nonencapsulated, seems to provoke signs and symptoms that last longer (64, 142). Nevertheless, these differences should be considered with caution due to the obvious lack of knowledge on the infecting dose for each patient.
DIAGNOSIS OF HUMANS
In general, the early clinical diagnosis of trichinellosis is rather difficult because pathognomonic signs or symptoms are lacking (29), and also, later chronic forms of the disease are not easy to diagnose. Furthermore, physicians practicing in nonendemic countries are usually unfamiliar with the disease and may thus experience problems in diagnosing trichinellosis. This problem becomes obvious when reviewing the extensive diagnostic delay found in cohort studies, indicating that the diagnosis was usually made at the late phase of the disease (63). This is of concern for the patients, because a delay in diagnosis and treatment favors the establishment of larvae in muscle tissue and the development of a collagen capsule, which leaves the larvae resistant to drugs (26, 138).
Differential diagnosis of trichinellosis is especially difficult for isolated cases and atypical clinical courses. Therefore, trichinellosis should be differentiated from various other diseases for which similar clinical findings may occur (Table 5).
TABLE 5.
Differential diagnosis of trichinellosisa
| Clinical finding | Disease to be differentiated |
|---|---|
| Protracted diarrhea | Salmonellosis, shigellosis, and other viral, bacterial, or parasitic infections of the gastrointestinal tract |
| High fever and myalgia | Influenza virus infection |
| Periorbital or facial edema with fever | Glomerulonephritis, serum sickness, toxic-allergic reactions to drugs or allergens, polymyositis, periarteritis nodosa, dermatomyositis |
| High fever and neurological symptoms without | |
| periorbital edema | Typhoid fever |
| Intense headaches, fever, nuchal pseudorigidity with blurred consciousness and drowsiness, irritability, | |
| and neurological symptoms | Cerebrospinal meningitis, encephalitis, neuroinfections |
| Intraconjunctival hemorrhages, intradermal petechiae, fever | Leptospirosis, bacterial endocarditis, and typhus exanthematicus |
| Eosinophilia combined with myalgia and an inflammatory | |
| response | Eosinophilia-myalgia syndromes (e.g., toxic oil syndrome, trytophan intake, and eosinophilic fasciitis) |
| Eosinophilia combined with fever | Fasciolasis, toxocarosis, and invasive schistosomosis |
The diagnosis of trichinellosis should be based on three main criteria (73): (i) clinical findings (recognition of the signs and symptoms of trichinellosis); (ii) laboratory findings (nonspecific laboratory parameters [eosinophilia and muscle enzymes], antibody detection, and/or detection of larvae in a muscle biopsy); and (iii) epidemiological investigation (identification of source and origin of infection and outbreak studies).
The European Center for Disease Control has issued a case definition to be used when a human trichinellosis case or outbreak is suspected (Table 6). An algorithm for the diagnosis of acute trichinellosis and for defining very unlikely, suspected, probable, highly probable, and confirmed cases is shown in Table 7 (see reference 29).
TABLE 6.
Case definition for human trichinellosis according to the European Center for Disease Controla
| Criterion group | Prerequisites and case classificationb |
|---|---|
| Clinical | At least three of the following six: fever, muscle soreness and pain, gastrointestinal symptoms, facial edema, eosinophilia, and subconjunctival, subungual, and retinal hemorrhages |
| Laboratory | At least one of the following two laboratory tests: demonstration of Trichinella larvae in tissue obtained by muscle biopsy and demonstration of Trichinella-specific antibody response by indirect immunofluorescence, ELISA, or Western blot (i.e., seroconversion) |
| Epidemiological | At least one of the following three: consumption of laboratory-confirmed parasitized meat, consumption of potentially parasitized products from a laboratory-confirmed infected animal, epidemiological link to a laboratory-confirmed human case by exposure to the same common source |
Modified from reference 29 with permission of the publisher.
Case classification is as follows: possible case, not applicable; probable case, any person meeting the clinical criteria and with an epidemiological link; confirmed case, any person meeting the laboratory criteria and with clinical criteria within the past 2 months (to be reported to the European Union level).
TABLE 7.
Algorithm for diagnosing acute trichinellosis in humansa
| Group | Symptom |
|---|---|
| A | Fever, eyelid and/or facial edema, myalgia |
| B | Diarrhea, neurological signs, cardiological signs, conjunctivitis, subungual hemorrhages, cutaneous rash |
| C | Eosinophilia (>1,000 eosinophils/ml) and/or increased total IgE levels, increased levels of muscular enzymes |
| D | Positive serology (with a highly specific test), seroconversion, positive muscular biopsy |
Modified from reference 29 with permission of the publisher. The diagnosis is very unlikely with one symptom from group A or one from group B or C, suspected with one symptom from group A or two from group B and one from group C, probable with three symptoms from group A and one from group C, highly probable with three symptoms from group A and two from group C, and confirmed with three symptoms from group A, two from group C, and one from group D or any of group A or B, one from group C, and one from group D.
Laboratory Findings
If persons suffer from clinically manifest trichinellosis, leukocytosis accompanied with eosinophilia and increased levels of muscle enzymes will usually occur as nonspecific laboratory signs. Eosinophilia appears early prior the development of clinical signs and symptoms, between the second and the fifth week of infection in practically every case of trichinellosis, with only few exceptions (26). Several studies have shown that eosinophilia is correlated with the degree of myalgia (29) and is significantly higher in persons with neurological complications (43). Besides moderately elevated white blood cell counts, eosinophilia appears already early after infection and increases between the second and the fifth week p.i. in various degrees: low (<1,000 white blood cells/mm3), moderate (1,000 white blood cells/mm3 to 3,000 white blood cells/mm3), and high (>3,000 white blood cells/mm3). During the acute stage of infection, a massive decrease in eosinophil levels in persons with severe trichinellosis can be considered to be a predictor for a severe outcome. Eosinophilia regresses slowly and remains at lower levels for periods of 10, 9, and 7 weeks in 50, 30, and 20% of patients, respectively (116).
Elevated levels of muscle enzymes (e.g., creatinine phosphokinase, lactate dehydrogenase, and aldolase) in blood, which indicate myositis, are found in 75 to 90% of infected persons between the second and fifth weeks of infection (15) and may persist for up to 4 months (64).
The detection of specific anti-Trichinella antibodies in blood serum is of great diagnostic value. After primary infection with Trichinella larvae, antibody detection in the human patient depends on seroconversion time, whereas differences between the different immunoglobulin classes exist (82, 156). During the acute stage of infection, an early increase in IgE levels can be observed in most cases, suggesting that this antibody class is related to allergic reactions such as cutaneous rash or edema. However, trichinellosis cannot be excluded if this antibody class is absent. Therefore, IgE is not considered to exhibit diagnostic relevance for routine diagnosis (12). Also, IgA and IgM presented no advantage to conventional IgG detection (74, 84, 93, 105). First, IgG antibodies can generally be demonstrated 12 to 60 days after infection. The time point of seroconversion is dependent on several factors such as the number of ingested larvae, the Trichinella species involved, and the individual immune response (12, 140).
Out of conventional serodiagnostic methods, ELISA is the most commonly used approach for the detection of Trichinella infection in humans, while the indirect fluorescent antibody test may be used as well. For the indirect fluorescent antibody test, cryostat sections of infected rodent muscle, frozen sections of free muscle larvae, or formalin-fixed whole larvae are employed to detect anti-Trichinella antibodies in serum samples (18). This test is more time-consuming and expensive than ELISA, and cross-reactions with antibodies directed against filariae (Onchocerca spp.) and Schistosoma mansoni have been observed in blood samples of Trichinella-negative patients (145). For the ELISA, and for any serological test, independent validation should be performed using sera from infected and Trichinella-free humans representative of the local population where the test is being used. The cutoff value of the ELISA, for example, can change based on host genotype and exposure to antigens in the local environment and in food (51). An important aspect of ELISA and other serological tests is the choice of the best antigens to be used. The validation process of an ELISA to detect anti-Trichinella IgG in human sera has revealed a large number of cross-reactions with sera from persons affected by other diseases, mainly if these persons originate from developing countries where a high number of parasitic diseases occur (53).
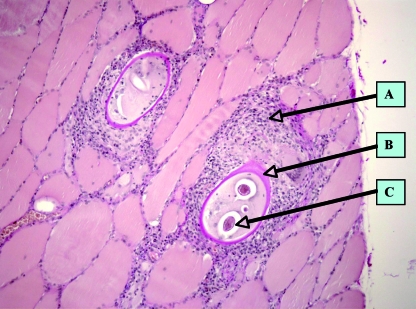
Immunoblotting showed promise for confirming positive cases or excluding false-positive serological results. Using E/S antigens of muscle larvae of T. spiralis, immunoblotting proved to be more sensitive in screening a population for trichinellosis than conventional ELISA (107). This technique, however, may be used as confirmatory rather than screening tests due to the high expenditure of labor and time and intensive costs. The direct detection of muscle-stage larvae based upon appropriate examination of muscle biopsies etiologically proves the diagnosis. Furthermore, isolated larvae allow the molecular identification of the Trichinella species or genotype (see “Molecular Techniques”), a procedure that is not possible by serology. The disadvantage of this method is that it requires a significant surgical intervention in the affected person and that the sensitivity of the diagnosis depends on the amount of muscle sample tested (26). Usually, 0.2 to 0.5 g of muscle tissue without fat and skin is collected from the deltoid or another skeletal muscle. Examination of a muscle biopsy can be performed through artificial digestion (identically to that described above for animals) or histological analysis upon hematoxylin-eosin staining (Fig. 4). The basophilic transformation of muscle cells represents a valuable diagnostic criterion of Trichinella invasion even when no larvae have been detected. This method is more sensitive than trichinoscopy at an early stage of muscle invasion, when larvae are very small and cannot easily be differentiated from muscle fibers (163).
FIG. 4.
Histological section (hematoxylin-eosin staining) of a muscle biopsy from a patient involved in a trichinellosis outbreak (100). (A) Cellular infiltrates; (B) collagen capsule of a “nurse cell”; (C) intersected muscle larva. (Photograph courtesy of Dietrich-Bonhoeffer-Klinikum, Neubrandenburg, Germany.)
TREATMENT OF TRICHINELLOSIS IN HUMANS
Upon appropriate diagnosis, therapy has to be initiated as early as possible (Table 8). Drugs administered in trichinellosis patients include anthelmintics, glucocorticosteroids, and preparations that compensate for protein and electrolyte deficits. Anthelmintics are the principal drugs for the treatment of trichinellosis. They include primarily albendazole (Zentel; Smith-Kline Beecham) and mebendazole (Vermox; Janssen Pharmaceutica, Beerse, Belgium) (4). As experienced with the treatment of echinococcosis (58), albendazole has the slight advantage that most patients reach requested plasma levels and thus do not require monitoring, while mebendazole plasma levels can vary considerably among different patients and might require individual monitoring and dosing (65). Secondarily, pyrantel (Combantrin; Pfizer) can also be used (29).
TABLE 8.
Practical recommendations to handle clinical trichinellosis casesa
| Severity code | Recommendation for treatment |
|---|---|
| Severe and moderately | |
| severe diseases | Hospitalization is compulsory for severe forms and debatable for moderately severe forms |
| Administration of anthelmintics (albendazole or mebendazole) | |
| Monitoring of the pharmacokinetics of anthelmintics (if possible) | |
| Administration of glucocorticosteroids (e.g., prednisolone), always with anthelmintics | |
| Compensation of fluid and electrolyte deficits | |
| Administration of pain killers | |
| Benign, abortive, and | |
| asymptomatic diseases | Administration of anthelmintics (albendazole or mebendazole) |
| Administration of nonsteroidal anti-inflammatory drugs if necessary |
Modified from reference 29 with permission of the publisher.
The recommended dose for albendazole is 400 mg twice daily for 8 to 14 days; for mebendazole, it is 200 to 400 mg three times a day for 3 days, followed by 400 to 500 mg three times a day for 10 days (4). Both treatment schemes are suitable for adult and pediatric dosages; however, they are contraindicated during pregnancy and not recommended in children aged <2 years (60). Steroids, e.g., prednisone, administered at a dose of 30 mg/day to 60 mg/day for 10 to 15 days for severe symptoms are the standard choice of chemotherapy. Prednisone was shown to be safe and to alleviate symptoms due to active tissue larvae (150). Pyrantel (Combantrin) is given in a single dose of 10 to 20 mg/kg of body weight, repeated for 2 to 3 days, and may be used by pregnant women and children, but it is active only against worms in the gut, and it has no effect against newborn and muscle larvae (29). The application of anthelmintics at the stage of intestinal invasion aims primarily at the elimination of intestinal forms of Trichinella sp. from the lumen of the gastrointestinal tract. Such treatment is of basic importance for an early and effective therapy, particularly in the first 3 days following the infection (73). If applied, such therapy prevents subsequent muscular invasion and the development of disease. However, in many cases, this option cannot be achieved anymore. The effect of anthelmintic therapy on the course of a more advanced stage of disease, on lethality, and on already encysted larvae is poorly elucidated so far. Prognosis for severe cases including cardiac or cerebral complications is not good. Despite therapy, lethality in cases with high infection intensity was up to 5%. In milder cases, prognosis is good, and most patients exhibit a disappearance of symptoms within 2 to 6 months. Occasionally, chronic myalgia and rheumatalgia will remain in otherwise successfully treated patients.
Principally, the effectiveness of chemotherapy using, e.g., albendazole or mebendazole is strongly dependent upon the time of administration; application only at the early stage of infection yields good effectiveness, although no sound case-control efficacy studies have been conducted to date. Unfortunately, most infected people are diagnosed only several weeks after the infection, when the larvae have already established themselves in the muscles.
As a general rule, the later a treatment is prescribed, the higher the probability that the infected person will already harbor viable larvae in their muscles, which can then survive for years despite treatment, with possible persistent myalgia (138). As a consequence, in advanced stages of infection and disease, medication should be administered for longer periods of time. Nonetheless, anthelmintics may be useless against long-term sequelae and chronic trichinellosis. Attempts to increase the bioavailability of anthelmintics have been successfully approved in a mouse model by adding 2-hydroxypropyl-β-cyclodextrin (16). Cymetidine has yielded similar effects on human cystic echinococcosis (83), but experience is lacking for human trichinellosis.
Astonishingly, beside benzimidazoles, other groups of compounds have not been tested so far or did not reach the stage of clinical trials in human trichinellosis. Since the middle of the 1970s, the new class of the macrocyclic lactones has revolutionized the animal health market. They can be divided into two subclasses: the milbemycins and the avermectins. Ivermectin, abamectin, eprinomectin, doramectin, milbemycin, moxidectin, and selamectin belong to these classes. In human medicine, only ivermectin has been used against some nematodes. Macrocyclic lactones may thus exhibit good potential to act against Trichinella spp. at different stages. Thus, Ros-Moreno et al. (146) showed that the gamma-aminobutyric acid receptor of Trichinella as a target for ivermectin's mode of action displayed sufficient binding activity in vitro to suggest further investigation. El-Azzouni (34) demonstrated some effect of ivermectin against experimental T. spiralis infection in Swiss albino mice at an early stage of infection. In veterinary medicine, studies were undertaken to determine the efficacy of milbemycin oxime against T. spiralis in experimentally infected dogs and cats (10). One of the most recent classes of compounds, that of cyclooctadepsipeptides, has entered the scene of anthelmintic research in the early 1990s. Anthelmintically active molecules were isolated as a natural compound from the fungus Mycelia sterilia (55). The semisynthetic drug emodepside has been shown to be active against T. spiralis larvae in muscles. Electrophysiological studies revealed that emodepside inhibits pharyngeal pumping of the nematodes (55). Future investigations and clinical trials will have to show if such new compounds provide a better efficacy against maturated and encapsulated Trichinella muscle larvae.
CONTROL AND PREVENTION
General Considerations
In many European countries, trichinellosis has been declared a disease that must be monitored, and for European Union Member States, this is obligatory according to the European Union directive on zoonoses, 2003/99/EC. Cases of trichinellosis must be reported, but further actions are not necessarily taken. However, the European Commission (EC) has edited legislation laying down rules for the official controls for Trichinella in meat in order to improve food safety for European consumers (35). According to regulation (EC) no. 2075/2005, meat inspections are required for domestic pigs, horses, wild boars, and other farmed and wild animal species that are susceptible to Trichinella infection and that are slaughtered for consumption. It foresees the possibility of a derogation from the testing program for herds, categories of herds, or regions where the risk of Trichinella infection in domestic swine is officially recognized as being negligible. Trichinella-free herds or categories of herds will have to meet several requirements. Most requirements are related to general hygiene and rodent control. Additionally, fattening pigs are not allowed to have access to outdoor facilities as of their fourth week and only if strict conditions are met during the first 4 weeks. Additional requirements apply to categories of herds that may be recognized as Trichinella free, including the need for a 10-year national surveillance program that would have detected a prevalence exceeding 0.0001%. In comparison to the EC, the Office International des Epizooties recognizes the possibility that a country or zone might be considered to be free from trichinellosis caused by T. spiralis in domestic swine. This may occur when trichinellosis is notifiable in the country and an effective disease reporting system is in place and has been shown to be capable of capturing the occurrences of cases. Additionally, a surveillance program that must be in place was described previously (35). For surveillance of the slaughter sow population, FAO/WHO/OIE guidelines (100) prescribe serological test methods, but the test method to be used for surveillance in slaughter pigs is not specified. Conclusively, the most appropriate and economically reasonable approach that could be developed will be a risk-based assessment, surveillance, and management system.
Prevention of Trichinella Infections in Humans
The prevention of trichinellosis in humans is based on three main approaches: (i) education of the consumer about the risk of consumption of raw or semiraw meat and meat products from both domestic (e.g., pigs, horses, and dogs) and sylvatic (e.g., wild boars, bears, walruses, cougars, badgers, foxes, jackals, armadillos, crocodiles, and monitor lizards) animals that can be carriers of Trichinella parasites if they are not properly tested for Trichinella larvae upon meat inspection; (ii) farming of pigs (the most important source of Trichinella infection for humans) in modern, industrialized, indoor pigsties under strict veterinary control and use of certified feedstuff (see below); and (iii) control of all susceptible animals (both domestic and sylvatic) by a standardized artificial digestion method at slaughtering or after hunting.
All meat from animals which might contain Trichinella larvae but cannot be tested by an appropriate laboratory method should be treated by a procedure that has been proven to inactivate Trichinella prior to distribution for human consumption (49, 50). This applies to both commercial and noncommercial sources of meat. Three methods have been shown to reliably inactivate Trichinella larvae in meat: (i) cooking to reach a core temperature of not less than 71°C (159.8°F) for at least 1 min (i.e., the meat must change the color from pink to gray, and muscle fibers are easily separated from each other), (ii) freezing (see below), and (iii) irradiation (see below).
Methods for the preparation of meat and meat products that are not considered safe include (i) cooking by using microwave ovens and (ii) curing, drying, or smoking. Education of hunters for the proper preparation of game meat should follow the same guidelines issued to consumers. Particular caution should be given to the putative presence of freeze-resistant Trichinella species or genotypes in game meats.
Freezing to inactivate Trichinella larvae in meat.
In the absence of proper temperature and time control and monitoring systems, processors and consumers of meat should ensure that cuts or pieces of meat up to 15 cm in thickness are frozen solid (at least −15°C [5°F]) for no less than 3 weeks, and cuts and pieces of meat up to 50 cm in thickness should be frozen solid for no less than 4 weeks. The requirements for freezing are restricted to pork infected with T. spiralis only (49). Indeed, T. britovi larvae in pork have survived up to 3 weeks at −20°C (−4°F) (132). Since T. spiralis larvae in horse meat frozen at −18°C (−0.4°F) can survive up to 4 weeks (59), and game meat often harbors freeze-resistant species of Trichinella, freezing principally represents a public health risk even after months or years (up to 5 years in bear meat) of treatment (23). While freeze-resistant Trichinella species have a low infectivity for pigs, such infections cannot be ignored in other host species in areas where these parasite species are endemic (e.g., northern latitudes).
Irradiation to inactivate Trichinella larvae in meat.
Irradiation at levels proven to inactivate Trichinella (0.3 kGy) can be an acceptable method for rendering meat safe for human consumption in those countries where irradiation of food is permitted. Irradiation is recommended for sealed packaged food only (49).
Curing to inactivate Trichinella larvae in meat.
Curing and smoking processes are not recommended to inactivate Trichinella larvae in pork, horse, or game meat (49, 50). Although individual validation studies have shown that various combinations of salt, temperature, and drying time will inactivate Trichinella larvae, curing and smoking are methods that are difficult to reliably monitor and control. Curing should be used only after successful extensive validation studies upon the use of strict process controls and protocols (49, 50).
Control of Trichinella Infection in Pigs
The category of pigs at highest risk for Trichinella infection are those raised on small holdings with minimal confinement. These so-called “backyard pigs” are often fed food scraps or other forms of meat-containing waste and have ready access to rodents and wildlife (49, 50). To compound the problems, pigs raised in this manner are generally not sold through retail marketing channels and therefore are not subjected to reliable methods of veterinary inspection. In addition, free-ranging pigs are also at risk for this zoonosis. The degree of risk to pigs raised outdoors depends in great part on the infection levels in local wildlife, and this degree of risk is of substantial importance for “organic” or “green” pig producers, who provide products to consumers seeking meat from animals raised under natural conditions (49, 50).
While this scenario historically might be more typical for developing countries, the situation increases in those countries of the world (including the European Union, the United States, and New Zealand) where organic food production and animal welfare efforts have increased the number of free-ranging pigs or pigs on pastures (49, 50). Knowledge about the modes of transmission of Trichinella to domestic pigs allows pig farmers and producers to design management systems that prevent or drastically reduce the risk of exposure. By following a series of good management practices, combined with documentation of these practices and regular official veterinary control to verify that these practices are effective, it is possible to certify the safety of pork without subsequent slaughter inspection or further processing (49, 50). In both the European Union and the United States, owners of pig holdings can apply to reach a production system with a negligible risk for Trichinella if pigs are bred in certified indoor farms, which greatly reduces or precludes exposure to Trichinella infection (100). The main key points are (i) architectural and environmental barriers, (ii) feed and feed storage, (iii) rodent control, (iv) farm hygiene including proper disposal of dead animals, and (v) piglets from farms with controlled housing conditions.
Programs that allow the certification of pigs as being free from Trichinella parasites based on good management practices that eliminate the risk of exposure should be administratively organized to allow a proper documentation of certified herds. This administration should perform the following functions: (i) develop a system of documentation of good production practices, which addresses all the points raised above; (ii) issue certifications and maintain records of certified farms; (iii) periodically conduct spot audits of certified producers to ensure the integrity of the system; and (iv) conduct periodic serology testing of pigs originating from certified farms to verify the absence of infection.
Economic impact of Trichinella infection in domestic animals.
In 1998, the global cost of detecting Trichinella infection in pigs was roughly estimated to be $3.00 (€2.10) per pig, which means that the control of Trichinella infection in the 190 million domestic pigs slaughtered at that time in the former 15 countries of the European Union had an economic impact of about $570 million (€388 million)/year (119). However, the true cost of Trichinella inspection by digestion ranges from $0.18 to $3.70 (€0.12 to €2.5) according to the size of the slaughterhouse. For example, in large Danish slaughterhouses, where about 10,000 pigs are slaughtered per day, the cost estimate for inspection of pooled sample digestion is $0.22 (€0.15) per pig (68). In the United States, the herd certification program for Trichinella-free farms (141) estimates a cost of $0.25 to $0.70 (€0.16 to €0.48) per pig, which roughly is similar to that of traditional inspection by digestion (68).
Control of Trichinella Infection in Wildlife
The importance of wildlife to provide reservoir hosts for all species of Trichinella is underscored by the parasite's biomass, which is greater in wild animals than in domestic animals; consequently, the only measures that can be implemented to reduce the prevalence of infection among wildlife is to instruct hunters to avoid leaving animal carcasses in the field after skinning or removing and discarding the entrails, increasing the probability of transmission to new hosts. A very close relationship between the conventional hunter's practice to leave animal carcasses in the field after skinning and the prevalence of trichinellosis among wildlife has been documented in arctic and subarctic regions, northern Kazakhstan, New Hampshire, Extremadura (Spain), and northwest European Russia (7, 85, 117, 120, 131, 162). When humans fail to implement a proper management of domestic animals and wildlife, Trichinella (especially T. spiralis but also T. britovi and T. pseudospiralis) infection is transmitted from the sylvatic environment into the domestic one, sometimes through synanthropic (intermediary between domestic and sylvatic) animals (125). In addition, some species can transfer in a reversible path from domestic animals to wildlife when carrion of domestic animals is not properly disposed of or carcasses of dead free-ranging pigs are not appropriately removed from the field.
Conclusively, there is ample evidence that trichinellosis in humans can be monitored and controlled to some extent with a rigorous reporting and testing system, an option that requires a good interaction between the public health sector and the corresponding veterinary sector. It is to be hoped that most affected countries will in time be able to join in the campaign to minimize human infection from this zoonotic and costly disease.
REFERENCES
- 1.Akkoc, N., Z. Kuruuzum, S. Akar, A. Yuce, F. Onen, N. Yapar, O. Ozgenc, M. Turk, D. Ozdemir, M. Avci, Y. Guruz, A. M. Oral, and E. Pozio. 2008. A large scale outbreak of trichinellosis due to Trichinella britovi in Turkey. Zoonoses Public Health. doi: 10.1111/j.1863-2378.2008.01158.x. [DOI] [PubMed]
- 2.Ancelle, T., A. E. De Bruyne, and J. Dupouy-Camet. 2005. Outbreak of trichinellosis due to consumption of bear meat from Canada, France, September 2005. Euro Surveill. 10:EO51013.3. [DOI] [PubMed] [Google Scholar]
- 3.Ancelle, T. 1998. History of trichinellosis outbreaks linked to horse meat consumption 1975-1998. Euro Surveill. 3:86-89. [DOI] [PubMed] [Google Scholar]
- 4.Anonymous. 2004. Drugs for parasitic infections. Med. Lett. 2004:1-12. [Google Scholar]
- 5.Appleton, J. A., R. G. Bell, W. Homan, and F. Van Knapen. 1991. Consensus on Trichinella spiralis antigens and antibodies. Parasitol. Today 7:190-192. [Google Scholar]
- 6.Appleyard, G. D., and A. A. Gajadhar. 2000. A review of trichinellosis in people and wildlife in Canada. Can. J. Public Health 91:293-297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Batkaev, A. I., and V. G. Vakker. 1992. The role of corsac in circulation of trichinelles in the Middle Irtysh territory, p. 24-26. In Proceedings of the 6th Scientific Conference on Trichinellosis. Kirov, Moscow, Russia.
- 8.Blaga, R., B. Durand, S. Antoniu, C. Gherman, C. M. Cretu, V. Cozma, and P. A. Boireau. 2007. Dramatic increase in the incidence of human trichinellosis in Romania over the past 25 years: impact of political changes and regional food habits. Am. J. Trop. Med. Hyg. 76:983-986. [PubMed] [Google Scholar]
- 9.Boireau, P., I. Vallee, T. Roman, C. Perret, L. Mingyuan, H. R. Gamble, and A. Gajadhar. 2000. Trichinella in horses: a low frequency infection with high human risk. Vet. Parasitol. 93:309-320. [DOI] [PubMed] [Google Scholar]
- 10.Bowman, D. D., R. A. Darigrand, M. K. Frongillo, S. C. Barr, J. A. Flanders, and L. G. Carbone. 1993. Treatment of experimentally induced trichinosis in dogs and cats. Am. J. Vet. Res. 54:1303-1305. [PubMed] [Google Scholar]
- 11.Bruschi, F., and K. D. Murrell. 2002. New aspects of human trichinellosis: the impact of new Trichinella species. Postgrad. Med. J. 78:15-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bruschi, F., C. Tassi, and E. Pozio. 1990. Parasite-specific antibody response in Trichinella sp. 3 human infection: a one year follow-up. Am. J. Trop. Med. Hyg. 43:186-193. [DOI] [PubMed] [Google Scholar]
- 13.Campbell, W. C. 1983. Modes of transmission, p. 425-444. In W. C. Campbell (ed.), Trichinella and trichinosis. Plenum Press, London, United Kingdom.
- 14.Capó, V., and D. D. Despommier. 1996. Clinical aspects of infection with Trichinella spp. Clin. Microbiol. Rev. 9:47-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Capo, V., D. Despommier, and R. I. Polvere. 1998. Trichinella spiralis: vascular endothelial growth factor is up-regulated within the nurse cell during the early phase of its formation. J. Parasitol. 84:209-214. [PubMed] [Google Scholar]
- 16.Casulli, A., M. A. Gomez Morales, B. Gallinella, L. Turchetto, and E. Pozio. 2006. HP-β-cyclodextrin improves the effectiveness of albendazole against encapsulated larvae of Trichinella spiralis in a murine model. J. Antimicrob. Chemother. 58:886-890. [DOI] [PubMed] [Google Scholar]
- 17.CDC. 2004. Trichinellosis associated with bear meat—New York and Tennessee, 2003. MMWR Morb. Mortal. Wkly. Rep. 53:606-610. [PubMed] [Google Scholar]
- 18.Cui, J., Z. Q. Wang, X. X. Jin, F. R. Mao, and F. Wu. 1995. Studies on indirect fluorescent antibody test (IFAT) for immunodiagnosis of human trichinosis. Chin. J. Parasitic Dis. Control 8:137-139. [Google Scholar]
- 19.Cuperlovic, K., M. Djordjevic, and S. Pavlovic. 2005. Re-emergence of trichinellosis in southeastern Europe due to political and economic changes. Vet. Parasitol. 132:159-166. [DOI] [PubMed] [Google Scholar]
- 20.Dea-Ayuela, M. A., F. Romaris, F. M. Uberira, S. Rama-Iniguez, A. Martinez-Fernandez, and F. Bolas. 2001. Possible presence of common tyvelose-containing glycans in Trichinella L1 larvae and embryonated eggs of several helminths. Parasite 8:120-122. [DOI] [PubMed] [Google Scholar]
- 21.Despommier, D. D. 1983. Biology, p. 75-151. In W. C. Campbell (ed.), Trichinella and trichinosis. Plenum Press, London, United Kingdom.
- 22.Despommier, D. D., L. Aron, and R. Thorson. 1975. Trichinella spiralis: growth of the intracellular (muscle) larva. Exp. Parasitol. 37:108-116. [DOI] [PubMed] [Google Scholar]
- 23.Dick, T. A., and E. Pozio. 2001. Trichinella spp. and trichinellosis, p. 380-396. In W. M. Samuel, M. J. Pybus, and A. A. Kocan (ed.), Parasitic diseases of wild mammals, 2nd ed. Iowa State University Press, Ames, IA.
- 24.Djordjevic, M., M. Bacic, M. Petricevic, K. Cuperlovic, A. Malakauskas, C. M. Kapel, and K. D. Murrell. 2003. Social, political, and economic factors responsible for the reemergence of trichinellosis in Serbia: a case study. J. Parasitol. 89:226-231. [DOI] [PubMed] [Google Scholar]
- 25.Dubinsky, P., A. Stefancikova, J. Kincekova, F. Ondriska, K. Reiterova, and M. Medvedova. 2001. Trichinellosis in the Slovak Republic. Parasite 8(Suppl. 2):100-102. [DOI] [PubMed] [Google Scholar]
- 26.Dupouy-Camet, J., W. Kociecka, F. Bruschi, F. Bolas-Fernandez, and E. Pozio. 2002. Opinion on the diagnosis and treatment of human trichinellosis. Expert Opin. Pharmacother. 3:1117-1130. [DOI] [PubMed] [Google Scholar]
- 27.Dupouy-Camet, J., A. Paugam, G. De Pinieux, V. Lavarde, and A. Vieillefond. 2001. Trichinella murrelli: pathological features in human muscles at different delays after infection. Parasite 8(Suppl. 2):176-179. [DOI] [PubMed] [Google Scholar]
- 28.Dupouy-Camet, J., S. Allegretti, and T. P. Truong. 1998. Enquete sur l'incidence de la trichinellose en France (1994-1995). Bull. Epidemiol. Hebd. 28:122-123. [Google Scholar]
- 29.Dupouy-Camet, J., and F. Bruschi. 2007. Management and diagnosis of human trichinellosis, p. 37-68. In J. Dupouy-Camet and K. D. Murrell (ed.), FAO/WHO/OIE guidelines for the surveillance, management, prevention and control of trichinellosis. World Organisation for Animal Health Press, Paris, France.
- 30.Reference deleted.
- 31.Dworkin, M. S., H. R. Gamble, D. S. Zarlenga, and P. O. Tennican. 1996. Outbreak of trichinellosis associated with eating cougar jerky. J. Infect. Dis. 174:663-666. [DOI] [PubMed] [Google Scholar]
- 32.EFSA. 2007. The Community Summary Report on trends and sources of zoonoses, zoonotic agents, antimicrobial resistance and foodborne outbreaks in the European Union in 2006. EFSA J. 94:167-174. [Google Scholar]
- 33.Eisenman, A., and R. Einat. 1992. A family outbreak of trichinosis acquired in Israel. Harefuah 122:702-704. (In Hebrew.) [PubMed] [Google Scholar]
- 34.El-Azzouni, M. Z. 1997. Effect of ivermectin on experimental trichinosis. J. Egypt. Soc. Parasitol. 27:331-340. [PubMed] [Google Scholar]
- 35.European Community. 2005. Regulation (EC) no. 2075/2005 of the European Parliament and of the Council of 5 December 2005 laying down specific rules on official controls for Trichinella in meat. Off. J. Eur. Commun. 338:60-82. [Google Scholar]
- 36.Ferraccioli, G. F., M. Mercadanti, F. Salaffi, F. Bruschi, M. Melissari, and E. Pozio. 1988. Prospective rheumatological study of muscle and joint symptoms during Trichinella nelsoni infection. Q. J. Med. 69:973-984. [PubMed] [Google Scholar]
- 37.Feydy, A., E. Touze, Y. Miaux, F. Bolgert, N. Martin-Duverneuil, D. Laplane, and J. Chiras. 1996. MRI in a case of neurotrichinosis. Neuroradiol. 38(Suppl. 1):S80-S82. [DOI] [PubMed] [Google Scholar]
- 38.Finkelman, F. D., T. Shea-Donohue, S. C. Morris, L. Gildea, R. Strait, K. B. Madden, L. Schopf, and J. F. Urban, Jr. 2004. Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol. Rev. 201:139-155. [DOI] [PubMed] [Google Scholar]
- 39.Forbes, L. B. 2000. The occurrence and ecology of Trichinella in marine mammals. Vet. Parasitol. 93:321-334. [DOI] [PubMed] [Google Scholar]
- 40.Reference deleted.
- 41.Forbes, L. B., and A. A. Gajadhar. 1999. A validated Trichinella digestion assay and an associated sampling and quality assurance system for use in testing pork and horse meat. J. Food Prot. 62:1308-1313. [DOI] [PubMed] [Google Scholar]
- 42.Forbes, L. B., G. D. Appleyard, and A. Gajadhar. 2004. Comparison of synthetic tyvelose antigen with excretory-secretory antigen for the detection of trichinellosis in swine using enzyme-linked immunosorbent assay. J. Parasitol. 90:835-840. [DOI] [PubMed] [Google Scholar]
- 43.Fourestié, V., H. Douceron, P. Brugieres, T. Ancelle, J. L. Lejonc, and R. K. Gherardi. 1993. Neurotrichinosis. A cerebrovascular disease associated with myocardial injury and hypereosinophilia. Brain 116:603-616. [DOI] [PubMed] [Google Scholar]
- 44.Fröscher, W., F. Gullotta, M. Saathoff, and W. Tackmann. 1988. Chronic trichinosis. Clinical, bioptic, serological and electromyographic observations. Eur. Neurol. 28:221-226. [DOI] [PubMed] [Google Scholar]
- 45.Gallardo, M. T., L. Mateos, J. Artieda, L. Wesslen, C. Ruiz, M. A. Garcia, A. Galmes-Truyols, A. Martin, G. Hernandez-Pezzi, Y. Andersson, T. Garate, and D. Christensson. 2007. Outbreak of trichinellosis in Spain and Sweden due to consumption of wild boar meat contaminated with Trichinella britovi. Euro Surveill. 12:E070315.1. [DOI] [PubMed] [Google Scholar]
- 46.Gamble, H. R., and I. V. Patrascu. 1996. Whole blood, serum and tissue fluids in an EIA for swine trichinellosis. J. Food Prot. 59:1213-1217. [DOI] [PubMed] [Google Scholar]
- 47.Gamble, H. R., A. A. Gajadhar, and M. B. Solomon. 1996. Methods for the detection of trichinellosis in horses. J. Food Prot. 59:420-425. [DOI] [PubMed] [Google Scholar]
- 48.Gamble, H. R., and C. E. Graham. 1984. Monoclonal antibody-purified antigen for the immunodiagnosis of trichinosis. Am. J. Vet. Res. 45:67-74.6703457 [Google Scholar]
- 49.Gamble, H. R., A. S. Bessonov, K. Cuperlovic, A. A. Gajadhar, F. van Knapen, K. Noeckler, H. Schenone, and X. Zhu. 2000. International Commission on Trichinellosis: recommendations on methods for the control of Trichinella in domestic and wild animals intended for human consumption. Vet. Parasitol. 93:393-408. [DOI] [PubMed] [Google Scholar]
- 50.Gamble, H. R., P. Boireau, K. Nöckler, and C. M. O. Kapel. 2007. Prevention of Trichinella infection in the domestic pig, p. 99-108. In J. Dupouy-Camet and K. D. Murrell (ed.), FAO/WHO/OIE guidelines for the surveillance, management, prevention and control of trichinellosis. World Organisation for Animal Health Press, Paris, France.
- 51.Gamble, H. R., E. Pozio, F. Bruschi, K. Nöckler, C. M. O. Kapel, and A. A. Gajadhar. 2004. International Commission on Trichinellosis: recommendations on the use of serological tests for the detection of Trichinella infection in animals and man. Parasite 11:3-13. [DOI] [PubMed] [Google Scholar]
- 52.Gamble, H. R., W. R. Anderson, C. E. Graham, and K. D. Murrell. 1983. Serodiagnosis of swine trichinosis using an excretory-secretory antigen. Vet. Parasitol. 13:349-361. [DOI] [PubMed] [Google Scholar]
- 53.Gómez-Morales, M. A., A. Ludovisi, M. Amati, S. Cherchi, P. Pezzotti, and E. Pozio. 2008. Validation of an enzyme-linked immunosorbent assay for the diagnosis of human trichinellosis. Clin. Vaccine Immunol. 15:1723-1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Haim, M., M. Efrat, M. Wilson, P. M. Schantz, D. Cohen, and J. Shemer. 1997. An outbreak of Trichinella spiralis infection in southern Lebanon. Epidemiol. Infect. 119:357-362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Harder, A., H. P. Schmitt-Wrede, J. Krücken, P. Marinovski, F. Wunderlich, J. Wilson, K. Amliwala, L. Holden-Dye, and R. Walker. 2003. Cyclooctapeptides—an anthelminthically active class of compounds exhibiting a novel mode of action. Int. J. Antimicrob. Agents 22:318-331. [DOI] [PubMed] [Google Scholar]
- 56.Harms, G., P. Binz, H. Feldmeier, K. Zwingenberger, D. Schleehauf, W. Dewes, I. Kress-Hermesdorf, C. Klindworth, and U. Bienzle. 1993. Trichinosis: a prospective controlled study of patients ten years after acute infection. Clin. Infect. Dis. 17:637-643. [DOI] [PubMed] [Google Scholar]
- 57.Hefer, E., S. Rishpon, and I. Volovik. 2004. Trichinosis outbreak among Thai immigrant workers in the Hadera sub-district. Harefuah 143:656-660. (In Hebrew.) [PubMed] [Google Scholar]
- 58.Hemphill, A., M. Spicher, B. Stadelmann, J. Mueller, A. Naguleswaran, B. Gottstein, and M. Walker. 2007. Innovative chemotherapeutical treatment options for alveolar and cystic echinococcosis. Parasitology 134:1657-1670. [DOI] [PubMed] [Google Scholar]
- 59.Hill, D. E., L. Forbes, A. A. Gajadhar, and H. R. Gamble. 2007. Viability and infectivity of Trichinella spiralis muscle larvae in frozen horse tissue. Vet. Parasitol. 146:102-106. [DOI] [PubMed] [Google Scholar]
- 60.Horton, J. 1993. The use of antiprotozoan and anthelmintic drugs during pregnancy and contraindications. J. Infect. 26:104-105. [DOI] [PubMed] [Google Scholar]
- 61.Hurníková, Z., V. Šnábel, E. Pozio, K. Reiterová, G. Hrčková, D. Halásová, and P. Dubinský. 2005. First record of Trichinella pseudospiralis in the Slovak Republic. Vet. Parasitol. 128:91-98. [DOI] [PubMed] [Google Scholar]
- 62.Jakob, H. P., J. Eckert, T. Jemmi, and B. Gottstein. 1994. Investigations on trichinellosis in slaughter animals and game in Switzerland with a digestion method and a serological approach (E/S-ELISA). Schweiz. Arch. Tierheilk. 136:298-308. (In German.) [PubMed] [Google Scholar]
- 63.Jansen, A., I. Schöneberg, K. Stark, and K. Nöckler. 2008. Epidemiology of trichinellosis in Germany, 1996-2006. Vector-Borne Zoon. Dis. 8:189-196. [DOI] [PubMed] [Google Scholar]
- 64.Jongwutiwes, S., N. Chantachum, P. Kraivichian, P. Siriyasatien, C. Putaporntip, A. Tamburini, G. La Rosa, C. Sreesunpasirikul, P. Yingyourd, and E. Pozio. 1998. First outbreak of human trichinellosis caused by Trichinella pseudospiralis. Clin. Infect. Dis. 26:111-115. [DOI] [PubMed] [Google Scholar]
- 65.Kadry, Z., E. C. Renner, L. M. Bachmann, N. Attigah, E. L. Renner, R. W. Ammann, and P. A. Clavien. 2005. Evaluation of treatment and long-term follow-up in patients with hepatic alveolar echinococcosis. Br. J. Surg. 92:1110-1116. [DOI] [PubMed] [Google Scholar]
- 66.Kapel, C. M. O. 2000. Host diversity and biological characteristics of the Trichinella genotypes and their effect on transmission. Vet. Parasitol. 93:263-278. [DOI] [PubMed] [Google Scholar]
- 67.Kapel, C. M. O. 2001. Sylvatic and domestic Trichinella spp. in wild boars; infectivity, muscle larvae distribution, and antibody response. J. Parasitol. 87:309-314. [DOI] [PubMed] [Google Scholar]
- 68.Kapel, C. M. O. 2005. Changes in the EU legislation on Trichinella inspection—new challenges in the epidemiology. Vet. Parasitol. 132:189-194. [DOI] [PubMed] [Google Scholar]
- 69.Kapel, C. M. O., and H. R. Gamble. 2000. Infectivity, persistence, and antibody response to domestic and sylvatic Trichinella spp. in experimentally infected pigs. Int. J. Parasitol. 30:215-221. [DOI] [PubMed] [Google Scholar]
- 70.Kapel, C. M. O., P. Webster, and R. Gamble. 2005. Muscle distribution of sylvatic and domestic Trichinella larvae in production animals and wildlife. Vet. Parasitol. 132:101-105. [DOI] [PubMed] [Google Scholar]
- 71.Kapel, C. M. O., P. Webster, P. Lind, E. Pozio, S. A. Henriksen, K. D. Murrell, and P. Nansen. 1998. Trichinella spiralis, T. britovi, and T. nativa: infectivity, larval distribution in muscle, and antibody response after experimental infection of pigs. Parasitol. Res. 84:264-271. [DOI] [PubMed] [Google Scholar]
- 72.Khamboonruang, C. 1991. The present status of trichinellosis in Thailand. Southeast Asian J. Trop. Med. Public Health 22:312-315. [PubMed] [Google Scholar]
- 73.Kociecka, W. 2000. Trichinellosis: human disease, diagnosis and treatment. Vet. Parasitol. 93:365-383. [DOI] [PubMed] [Google Scholar]
- 74.Kociecka, W., B. Mrozewicz, L. Gustowska, and D. Jakuszewska. 1997. Late sequele of trichinellosis: clinical and pathomorphological findings, p. 611-621. In G. Ortega-Pierres, H. R. Gamble, F. van Knappen, and D. Wakelin (ed.), Trichinellosis. Centro de Investigacion y Estudios Avanzados del Instituto Polytecnico Nacional Mexico, D.F., Mexico City, Mexico.
- 75.Kociecka W., K. Bombicki, Ł. Pielok, and L. Gustowska. 2001. New aspects of clinical pathology and electrophysiological muscle disturbances in patients with history of trichinellosis. Parasite 8:173-175. [DOI] [PubMed] [Google Scholar]
- 76.Krivokapich, S. J., C. L. Prous, G. M. Gatti, V. Confalonieri, V. Molina, H. Matarasso, and E. Guarnera. 2008. Molecular evidence for a novel encapsulated genotype of Trichinella from Patagonia, Argentina. Vet. Parasitol. 156:234-240. [DOI] [PubMed] [Google Scholar]
- 77.Kumar, V., E. Pozio, J. De Borchgrave, J. Mortelmans, and W. De Meurichy. 1990. Characterization of a Trichinella isolate from polar bear. Ann. Soc. Belge Méd. Trop. 70:131-135. [PubMed] [Google Scholar]
- 78.Lachkar, S., P. Abboud, G. Gargala, M. Etienne, E. Gauliard, C. Tron, L. Favennec, I. Gueit, F. Caron, and J. Dupouy-Camet. 2007. Troponin dosage in a patient with asymptomatic myocarditis due to trichinellosis. Rev. Med. Interne 29:246-248. [DOI] [PubMed] [Google Scholar]
- 79.La Rosa, G., G. Marucci, D. S. Zarlenga, and E. Pozio. 2001. Trichinella pseudospiralis populations of the Palearctic region and their relationship with populations of the Nearctic and Australian regions. Int. J. Parasitol. 31:297-305. [DOI] [PubMed] [Google Scholar]
- 80.La Rosa, G., G. Marucci, D. S. Zarlenga, A. Casulli, R. L. Zarnke, and E. Pozio. 2003. Molecular identification of natural hybrids between Trichinella nativa and Trichinella T6 provides evidence of gene flow and ongoing genetic divergence. Int. J. Parasitol. 33:209-216. [DOI] [PubMed] [Google Scholar]
- 81.Liu, M., and P. Boireau. 2002. Trichinellosis in China: epidemiology and control. Trends Parasitol. 18:553-556. [DOI] [PubMed] [Google Scholar]
- 82.Ljungstrom, I. 1983. Immunodiagnosis in man, p. 403-424. In W. C. Campbell (ed.), Trichinella and trichinosis. Plenum Press, New York, NY.
- 83.Luder, P. J., B. Siffert, F. Witassek, F. Meister, and J. Bircher. 1986. Treatment of hydatid disease with high oral doses of mebendazole. Long-term follow-up of plasma mebendazole levels and drug interactions. Eur. J. Clin. Pharmacol. 31:443-448. [DOI] [PubMed] [Google Scholar]
- 84.Machnicka, B., D. Prokopowicz, K. Madalinski, E. Dziemian, and K. Prokopowicz. 1994. Antibodies circulating antigens and circulating immune complexes in human trichinellosis, p. 427-432. In W. C. Campbell, E. Pozio, and F. Bruschi (ed.), Trichinellosis. Istituto Superiore di Sanita Press, Rome, Italy.
- 85.Madsen, H. 1974. The principles of the epidemiology of trichinellosis with a new view on the life cycle, p. 615-638. In C. W. Kim (ed.), Trichinellosis. Intext Educational Publishers, New York, NY.
- 86.Malakauskas, A., C. M. Kapel, and P. Webster. 2001. Infectivity, persistence and serological response of nine Trichinella genotypes in rats. Parasite 8(Suppl. 2):216-222. [DOI] [PubMed] [Google Scholar]
- 87.Margolis, H. S., J. P. Middaugh, and R. D. Burgess. 1979. Arctic trichinosis: two Alaskan outbreaks from walrus meat. J. Infect. Dis. 139:102-105. [DOI] [PubMed] [Google Scholar]
- 88.Marucci, G., L. J. La Grange, G. La Rosa, and E. Pozio. 2008. Trichinella nelsoni and Trichinella T8 co-infection in a lion (Panthera leo) of the Kruger National Park (South Africa). Vet. Parasitol. doi: 10.1016/j. vetpar.2008.10.041. [DOI] [PubMed]
- 89.Marva, E., A. Markovics, M. Gdalevich, N. Asor, C. Sadik, and A. Leventhal. 2005. Trichinellosis outbreak. Emerg. Infect. Dis. 11:1979-1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.McAuley, J. B., M. K. Michelson, and P. M. Schantz. 1991. Trichinella infection in travelers. J. Infect. Dis. 164:1013-1016. [DOI] [PubMed] [Google Scholar]
- 91.Møller, L. N., E. Petersen, R. Gamble, and C. M. O. Kapel. 2005. Comparison of two antigens for demonstration of Trichinella spp. antibodies in blood and muscle fluids of foxes, pigs and wild boars. Vet. Parasitol. 132:81-84. [DOI] [PubMed] [Google Scholar]
- 92.Møller, L. N., E. Petersen, C. M. Kapel, M. Melbye, and A. Koch. 2005. Outbreak of trichinellosis associated with consumption of game meat in West Greenland. Vet. Parasitol. 132:131-136. [DOI] [PubMed] [Google Scholar]
- 93.Murrell, K. D., and F. Bruschi. 1994. Clinical trichinellosis, p. 117-150. In T. Sun (ed.), Progress in clinical parasitology. CRC Press, Boca Raton, FL. [PubMed]
- 94.Murrell, K. D., and E. Pozio. 2000. Trichinellosis: the zoonosis that won't go quietly. Int. J. Parasitol. 30:1339-1349. [DOI] [PubMed] [Google Scholar]
- 95.Murrell, K. D., W. R. Anderson, G. A. Schad, R. D. Hanbury, K. R. Kazacos, H. R. Gamble, and J. Brown. 1986. Field evaluation of the enzyme-linked immunosorbent assay for swine trichinellosis: efficacy of the excretory-secretory antigen. Am. J. Vet. Res. 47:1046-1049. [PubMed] [Google Scholar]
- 96.Nakamura, T., T. Miura, T. Nakaoka, I. Nagano, Y. Takahashi, and A. Iwamoto. 2003. A case of trichinellosis with spontaneous remission. Kansenshogaku Zasshi 77:839-843. (In Japanese.) [DOI] [PubMed] [Google Scholar]
- 97.Reference deleted.
- 98.Nelson, M., T. L. Wright, A. Pierce, and R. A. Krogwold. 2003. A common-source outbreak of trichinosis from consumption of bear meat. J. Environ. Health 65:16-19. [PubMed] [Google Scholar]
- 99.Nezri, M., J. Ruer, A. De Bruyne, R. Cohen-Valensi, E. Pozio, and J. Dupouy-Camet. 2006. Premiere observation d'un cas de humain de trichinellose a Trichinella britovi en Algerie après consummation de viande de chacal (Canis aureus). Bull. Soc. Pathol. Exot. 99:94-95. [PubMed] [Google Scholar]
- 100.Nöckler, K., and C. M. O. Kapel. 2007. Detection and surveillance for Trichinella: meat inspection and hygiene, and legislation, p. 69-97. In J. Dupouy-Camet and K. D. Murrell (ed.), FAO/WHO/OIE guidelines for the surveillance, management, prevention and control of trichinellosis. World Organisation for Animal Health Press, Paris, France.
- 101.Nöckler, K., H. Wichmann-Schauer, P. Hiller, A. Müller, and K.-H. Bogner. 2007. Trichinellosis outbreak in Bavaria caused by cured sausage from Romania, January 2007. Euro Surveill. 12:EO70823.2. [DOI] [PubMed] [Google Scholar]
- 102.Nöckler, K., A. Hamidi, R. Fries, J. Heidrich, R. Beck, and A. Marinculic. 2004. Influence of methods for Trichinella detection in pigs from endemic and non-endemic European region. J. Vet. Med. B 51:297-301. [DOI] [PubMed] [Google Scholar]
- 103.Nöckler, K., E. Pozio, W. P. Voigt, and J. Heidrich. 2000. Detection of Trichinella infection in food animals. Vet. Parasitol. 93:335-350. [DOI] [PubMed] [Google Scholar]
- 104.Nöckler, K., F. J. Serrano Aguilera, P. Boireau, C. M. O. Kapel, and E. Pozio. 2005. Experimental studies in pigs on Trichinella detection in different diagnostic matrices. Vet. Parasitol. 132:85-90. [DOI] [PubMed] [Google Scholar]
- 105.Nöckler, K., I. Reiter-Owona, J. Heidrich, D. Protz, S. Rehmet, G. Sinn, and A. Ammon. 2001. Aspects of clinical features, diagnosis, notification and tracing back in connection with two Trichinella outbreaks in North Rhine-Westphalia, Germany. Parasite 8:183-185. [DOI] [PubMed] [Google Scholar]
- 106.Nöckler, K., W. P. Voigt, D. Protz, A. Miko, and K. Ziedler. 1995. Indirect ELISA for the diagnosis of trichinellosis in living pigs. Berl. Münch. Tierärztl. Wschr. 108:167-174. (In German.) [PubMed] [Google Scholar]
- 107.Nuñez, G. G., S. L. Malmassari, S. N. Costantino, and S. M. Venturiello. 2000. Immunoelectrotransfer blot assay in acute and chronic human trichinellosis. J. Parasitol. 86:1121-1124. [DOI] [PubMed] [Google Scholar]
- 108.Office International des Epizooties. 2004. Trichinellosis, chapter 2.2.9. In Manual of standards for diagnostic tests and vaccines, 5th ed. Office International des Epizooties, Paris, France.
- 109.Olaison, L., and I. Ljungstrom. 1992. An outbreak of trichinosis in Lebanon. Trans. R. Soc. Trop. Med. Hyg. 86:658-660. [DOI] [PubMed] [Google Scholar]
- 110.Oliver, D. G., P. Singh, D. E. Allison, K. D. Murrell, and H. R. Gamble. 1989. Field evaluation of an enzyme immunoassay for detection of hogs in a high volume North Carolina abattoir, p. 439-444. In C. E. Tanner (ed.), Trichinellosis. Consejo Superior de Investigaciones Cientificas Press, Madrid, Spain.
- 111.Ortega Pierres, M. G., C. Arriaga, and L. Yepez-Mulia. 2000. Epidemiology of trichinellosis in Mexico, Central and South America. Vet. Parasitol. 93:201-225. [DOI] [PubMed] [Google Scholar]
- 112.Ortega-Pierres, M. G., L. Yepez-Mulia, W. Homan, H. R. Gamble, P. Lim, Y. Takahashi, D. L. Wassom, and J. A. Appleton. 1996. Workshop on a detailed characterization of Trichinella spiralis antigens: a platform for future studies on antigens and antibodies to this parasite. Parasite Immunol. 18:273-284. [DOI] [PubMed] [Google Scholar]
- 113.Owen, I. L., M. A. Gomez Morales, P. Pezzotti, and E. Pozio. 2005. Trichinella infection in a hunting population of Papua New Guinea suggests an ancient relationship of Trichinella with human beings. Trans. R. Soc. Trop. Med. Hyg. 99:618-624. [DOI] [PubMed] [Google Scholar]
- 114.Owen, I. L., and S. A. Reid. 2007. Survival of Trichinella papuae muscle larvae in a pig carcass maintained under simulated natural conditions in Papua New Guinea. J. Helminthol. 81:429-432. [DOI] [PubMed] [Google Scholar]
- 115.Ozeretskovskaya, N. N., L. G. Mikhailova, T. P. Sabgaida, and A. S. Dovgalev. 2005. New trends and clinical patterns of human trichinellosis in Russia at the beginning of the XXI century. Vet. Parasitol. 132:167-171. [DOI] [PubMed] [Google Scholar]
- 116.Pawlowski, Z. S. 1983. Clinical aspects in man, p. 367-402. In W. C. Campbell (ed.), Trichinella and trichinosis. Plenum Press, London, United Kingdom.
- 117.Pérez-Martin, J., F. J. Serrano, D. Reina, J. A. Mora, and I. Navarrete. 2000. Sylvatic trichinellosis in southwestern Spain. J. Wildl. Dis. 36:531-534. [DOI] [PubMed] [Google Scholar]
- 118.Pielok, L. 2001. Clinical analysis and evaluation of selected laboratory parameters in patients examined in distant periods after trichinellosis. Wiadom. Parazytol. 47:185-209. (In Polish.) [PubMed] [Google Scholar]
- 119.Pozio, E. 1998. Trichinellosis in the European Union: epidemiology, ecology and economic impact. Parasitol. Today 14:35-38. [DOI] [PubMed] [Google Scholar]
- 120.Pozio, E. 2001. New patterns of Trichinella infections. Vet. Parasitol. 98:133-148. [DOI] [PubMed] [Google Scholar]
- 121.Pozio, E. 2005. The broad spectrum of Trichinella hosts: from cold- to warm-blooded animals. Vet. Parasitol. 132:3-11. [DOI] [PubMed] [Google Scholar]
- 122.Pozio, E. 2007. Taxonomy, biology and epidemiology of Trichinella parasites, p. 1-35. In J. Dupouy-Camet and K. D. Murrell (ed.), FAO/WHO/OIE guidelines for the surveillance, management, prevention and control of trichinellosis. World Organisation for Animal Health Press, Paris, France.
- 123.Pozio, E. 2007. World distribution of Trichinella spp. infections in animals and humans. Vet. Parasitol. 149:3-21. [DOI] [PubMed] [Google Scholar]
- 124.Pozio, E., and G. Marucci. 2003. Trichinella-infected pork products: a dangerous gift. Trends Parasitol. 19:338. [DOI] [PubMed] [Google Scholar]
- 125.Pozio, E., and K. D. Murrell. 2006. Systematics and epidemiology of Trichinella. Adv. Parasitol. 63:367-439. [DOI] [PubMed] [Google Scholar]
- 126.Pozio, E., and D. S. Zarlenga. 2005. Recent advances on the taxonomy, systematics and epidemiology of Trichinella. Int. J. Parasitol. 35:1191-1204. [DOI] [PubMed] [Google Scholar]
- 127.Pozio, E., P. Cossu, G. Marucci, M. Amati, M. Ludovisi, M. A. Gomez Morales, G. La Rosa, and T. Firinu. 2008. The birth of a Trichinella britovi focus on the Mediterranean island of Sardinia (Italy). Vet. Parasitol. doi: 10.1016/j.vetpar.2008.10.055. [DOI] [PubMed]
- 128.Pozio, E., F. Paterlini, C. Pedarra, L. Sacchi, R. Bugarini, E. Goffredo, and P. Boni. 1999. Predilection sites of Trichinella spiralis larvae in naturally infected horses. J. Helminthol. 73:233-237. [DOI] [PubMed] [Google Scholar]
- 129.Pozio, E., C. M. Foggin, T. Gelanew, G. Marucci, A. Hailu, P. Rossi, and M. A. Gomez Morales. 2007. Trichinella zimbabwensis in wild reptiles of Zimbabwe and Mozambique and in farmed reptiles of Ethiopia. Vet. Parasitol. 143:305-310. [DOI] [PubMed] [Google Scholar]
- 130.Pozio, E., C. M. Foggin, G. Marucci, G. La Rosa, L. Sacchi, S. Corona, P. Rossi, and S. Mukaratirwa. 2002. Trichinella zimbabwensis n. sp. (Nematoda), a new non-encapsulated species from crocodiles (Crocodylus niloticus) in Zimbabwe also infecting mammals. Int. J. Parasitol. 32:1787-1799 [DOI] [PubMed] [Google Scholar]
- 131.Pozio, E., A. Casulli, V. V. Bologov, G. Marucci, and G. La Rosa. 2001. Hunting practices increase the prevalence of Trichinella infection in wolves from European Russia. J. Parasitol. 87:1498-1501. [DOI] [PubMed] [Google Scholar]
- 132.Pozio, E., C. M. O. Kapel, A. A. Gajadhar, P. Boireau, J. Dupouy-Camet, and H. R. Gamble. 2006. Trichinella in pork: current knowledge on the suitability of freezing as a public health measure. Euro Surveill. 11:E061116.1. [DOI] [PubMed] [Google Scholar]
- 133.Pozio, E., and G. La Rosa. 2003. PCR-derived methods for the identification of Trichinella parasites from animal and human samples. Methods Mol. Biol. 216:299-309. [DOI] [PubMed] [Google Scholar]
- 134.Pozio, E., G. La Rosa, K. D. Murrell, and J. R. Lichtenfels. 1992. Taxonomic revision of the genus Trichinella. J. Parasitol. 78:654-659. [PubMed] [Google Scholar]
- 135.Pozio, E., I. L. Owen, G. Marucci, and G. La Rosa. 2005. Inappropriate feeding practice favors the transmission of Trichinella papuae from wild pigs to saltwater crocodiles in Papua New Guinea. Vet. Parasitol. 127:245-251. [DOI] [PubMed] [Google Scholar]
- 136.Pozio, E., P. Pagani, G. Marucci, D. S. Zarlenga, E. P. Hoberg, D. De Meneghi, G. La Rosa, and L. Rossi. 2005. Trichinella britovi etiological agent of sylvatic trichinellosis in the Republic of Guinea (West Africa) and a re-evaluation of geographical distribution for encapsulated species in Africa. Int. J. Parasitol. 35:955-960. [DOI] [PubMed] [Google Scholar]
- 137.Pozio, E., L. Rinaldi, G. Marucci, V. Musella, F. Galati, G. Cringoli, P. Boireau, and G. La Rosa. 2008. Hosts and habitats of Trichinella spiralis and Trichinella britovi in Europe. Int. J. Parasitol. doi: 10.1016/j. ijpara.2008.06.006. [DOI] [PubMed]
- 138.Pozio, E., D. Sacchini, L. Sacchi, A. Tamburrini, and F. Alberici. 2001. Failure of mebendazole in treating Trichinella spiralis infection in humans at the stage of encapsulating larvae. Clin. Infect. Dis. 32:638-642. [DOI] [PubMed] [Google Scholar]
- 139.Pozio, E., L. Sofronic-Milosavljevic, M. A. Gomez Morales, P. Boireau, and K. Nöckler. 2002. Evaluation of ELISA and Western blot analysis using three antigens to detect anti-Trichinella IgG in horses. Vet. Parasitol. 108:163-178. [DOI] [PubMed] [Google Scholar]
- 140.Pozio, E., P. Varese, M. A. Gomez Morales, G. P. Croppo, D. Pelliccia, and F. Bruschi. 1993. Comparison of human trichinellosis caused by Trichinella spiralis and by Trichinella britovi. Am. J. Trop. Med. Hyg. 48:568-575. [DOI] [PubMed] [Google Scholar]
- 141.Pyburn, D. G., H. R. Gamble, E. A. Wagstrom, L. A. Anderson, and L. E. Miller. 2005. Trichinae certification in the United States pork industry. Vet. Parasitol. 132:179-183. [DOI] [PubMed] [Google Scholar]
- 142.Ranque, S., B. Faugère, E. Pozio, G. La Rosa, A. Tamburrini, J. F. Pellissier, and P. Brouqui. 2000. Trichinella pseudospiralis outbreak in France. Emerg. Infect. Dis. 6:543-547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Reason, A. J., L. A. Ellis, J. A. Appleton, N. Wisnewski, R. B. Grieve, M. McNeil, D. L. Wassom, H. R. Morris, and A. Dell. 1994. Novel tyvelose-containing tri- and tetra-antennary N-glycans in the immunodominant antigens of the intracellular parasite Trichinella spiralis. Glycobiology 4:593-603. [DOI] [PubMed] [Google Scholar]
- 144.Ribicich, M., H. R. Gamble, A. Rosa, J. Bolpe, and A. Franco. 2005. Trichinellosis in Argentina: an historical review. Vet. Parasitol. 132:137-142. [DOI] [PubMed] [Google Scholar]
- 145.Robert, F., B. Weil, N. Kassis, and J. Dupoy-Camet. 1996. Investigation of immunofluorescence cross-reactions against Trichinella spiralis by Western blot (immunoblot) analysis. Clin. Diagn. Lab. Immunol. 3:575-577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ros-Moreno, R. M., M. J. Moreno-Guzmán, A. Jiménez-González, and F. Rodríguez-Caabeiro. 1999. Interaction of ivermectin with gamma-aminobutyric acid receptors in Trichinella spiralis muscle larvae. Parasitol. Res. 85:320-323. [DOI] [PubMed] [Google Scholar]
- 147.Schellenberg, R. S., B. J. Tan, J. D. Irvine, D. R. Stockdale, A. A. Gajadhar, B. Serhir, J. Botha, C. A. Armstrong, S. A Woods, J. M. Blondeau, and T. L. McNab. 2003. An outbreak of trichinellosis due to consumption of bear meat infected with Trichinella nativa, in 2 northern Saskatchewan communities. J. Infect. Dis. 188:835-843. [DOI] [PubMed] [Google Scholar]
- 148.Schenone, H., A. Olea, H. Schenone, M. C. Contreras, R. Mercado, L. Sandoval, and C. Pavletic. 2002. Current epidemiological situation of trichinosis in Chile, 1991-2000. Rev. Med. Chil. 130:281-285. (In Spanish.) [PubMed] [Google Scholar]
- 149.Serhir, B., J. D. MacLean, S. Healey, and L. Forbes. 1999. Outbreak of trichinellosis associated with arctic walruses in northern Canada. Can. Commun. Dis. Rep. 27:31-36. [PubMed] [Google Scholar]
- 150.Shimoni, Z., Z. Klein, P. Weiner, M. V. Assous, and P. Froom. 2007. The use of prednisone in the treatment of trichinellosis. Isr. Med. Assoc. J. 9:537-539. [PubMed] [Google Scholar]
- 151.Shiota, T., N. Arizono, T. Yoshioka, Y. Ishikawa, J. Fujitake, H. Fujii, Y. Tatsuoka, and Y. Kim. 1999. Imported trichinellosis with severe myositis—report of a case. Kansenshogaku Zasshi 73:76-82 (In Japanese.) [DOI] [PubMed] [Google Scholar]
- 152.Smith, H. J. 1987. Evaluation of the ELISA for the serological diagnosis of trichinosis in Canadian swine. Can. J. Vet. Res. 51:194-197. [PMC free article] [PubMed] [Google Scholar]
- 153.Soulé, C., and J. Dupouy-Camet. 1991. La trichinellose: une zoonose en évolution. Office International des Epizooties, Paris, France.
- 154.Stensvold, C. R., H. V. Nielsen, and K. Molbak. 2007. A case of trichinellosis in Denmark, imported from Poland, June 2007. Euro Surveill. 12:E070809.3. [DOI] [PubMed] [Google Scholar]
- 155.Van der Leek, M. L., J. B. Dame, C. L. Adams, K. D. Gillis, and R. C. Littell. 1992. Evaluation of an enzyme-linked immunosorbent assay for diagnosis of trichinellosis in swine. Am. J. Vet. Res. 53:877-882. [PubMed] [Google Scholar]
- 156.van Knapen, F., J. H. Franchimont, A. R. Verdonk, J. Stumpf, and K. Undeutsch. 1982. Detection of specific immunoglobulins (IgG IgM, IgA, IgE) and total IgE levels in human trichinosis by means of the enzyme-linked immunosorbent assay (ELISA). Am. J. Trop. Med. Hyg. 31:973-976. [DOI] [PubMed] [Google Scholar]
- 157.Voigt, W. P., K. Nöckler, B. Freischem, S. A. Henriksen, F. van Knapen, F. Martinez-Fernandez, G. Pfeiffer, E. Pozio, G. Reuter, C. Ring, C. Soulé, and H. Weiss. 1998. Detection of low levels of Trichinella spiralis in experimentally infected horses, p. 629-634. In G. Ortega-Pierres, H. R. Gamble, F. van Knapen, and D. Wakelin (ed.), Trichinellosis. 9th International Conference on Trichinellosis. Germar Press, Nonoalco Tlateloco, Mexico.
- 158.Wang, Z. Q., J. Cui, and B. L. Xu. 2006. The epidemiology of human trichinellosis in China during 2000-2003. Acta Trop. 97:247-251. [DOI] [PubMed] [Google Scholar]
- 159.Watanabe, N., F. Bruschi, and M. Korenaga. 2005. IgE: a question of protective immunity in Trichinella spiralis infection. Trends Parasitol. 21:175-178. [DOI] [PubMed] [Google Scholar]
- 160.Webster, P., C. Maddox-Hyttel, K. Nöckler, A. Malakauskas, J. van der Giessen, E. Pozio, P. Boireau, and C. M. O. Kapel. 2006. Meat inspection for Trichinella in pork, horsemeat and game within EU: available technology and its present implementation. Euro Surveill. 11:50-55. [PubMed] [Google Scholar]
- 161.Wisnewski, N., M. McNeil, R. B. Grieve, and D. L. Wassom. 1993. Characterization of novel fucosyl- and tyvelosyl-containing glycoconjugates from Trichinella spiralis muscle stage larvae. Mol. Biochem. Parasitol. 61:25-35. [DOI] [PubMed] [Google Scholar]
- 162.Worley, D. E., F. M. Seesee, D. S. Zarlenga, and K. D. Murrell. 1994. Attempts to eradicate trichinellosis from a wild boar population in a private game park (USA), p. 611-616. In C. W. Campbell, E. Pozio, and F. Bruschi (ed.), Trichinellosis. ISS Press, Rome, Italy.
- 163.Wranicz, M. J., L. Gustowska, P. Gabryel, E. Kucharska, and W. Cabaj. 1998. Trichinella spiralis: induction of the basophilic transformation of muscle cells by synchronous newborn larvae. Parasitol. Res. 84:403-407. [DOI] [PubMed] [Google Scholar]
- 164.Yamaguchi, T. 1991. Present status of trichinellosis in Japan. Southeast Asian J. Trop. Med. Public Health 22:295-301. [PubMed] [Google Scholar]
- 165.Zarlenga, D. S., R. A. Aschenbrenner, and J. R. Lichtenfels. 1996. Variations in microsatellite sequences provide evidence for population differences and multiple ribosomal gene repeats within Trichinella pseudospiralis. J. Parasitol. 82:534-538. [PubMed] [Google Scholar]
- 166.Zarlenga, D. S., M. B. Chute, A. Martin, and C. M. Kapel. 1999. A multiplex PCR for unequivocal differentiation of all encapsulated and non-encapsulated genotypes of Trichinella. Int. J. Parasitol. 29:1859-1867. [DOI] [PubMed] [Google Scholar]
- 167.Zarlenga, D. S., B. Rosenthal, G. La Rosa, E. Pozio, and E. P. Hoberg. 2006. An old genus learns new tricks: late tertiary colonization and speciation of Trichinella nematodes among eutheria. Proc. Natl. Acad. Sci. USA 103:7354-7359. [DOI] [PMC free article] [PubMed] [Google Scholar]