Abstract
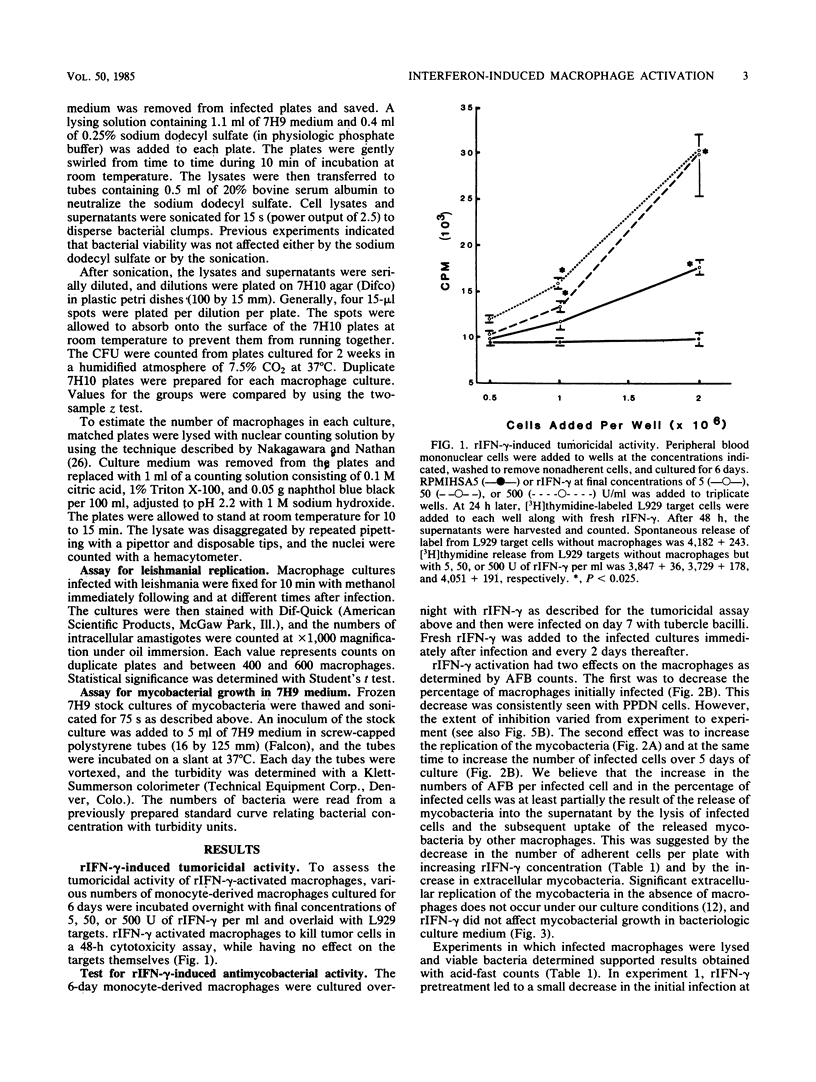
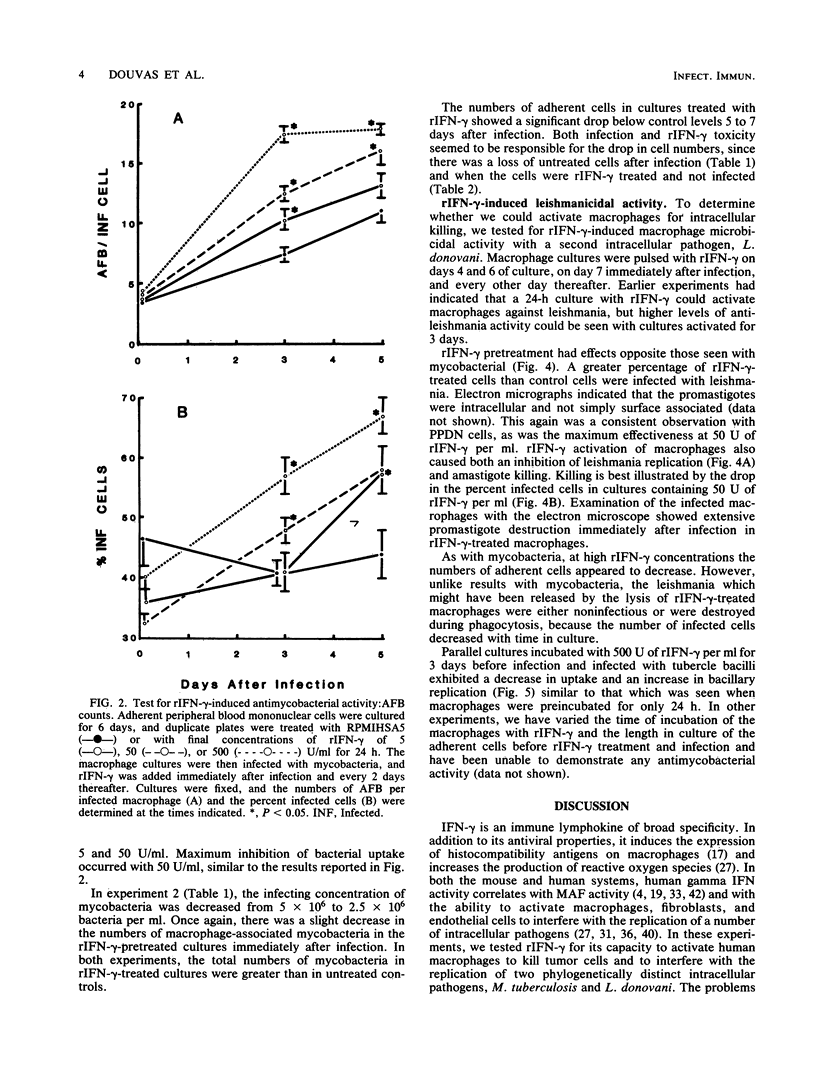
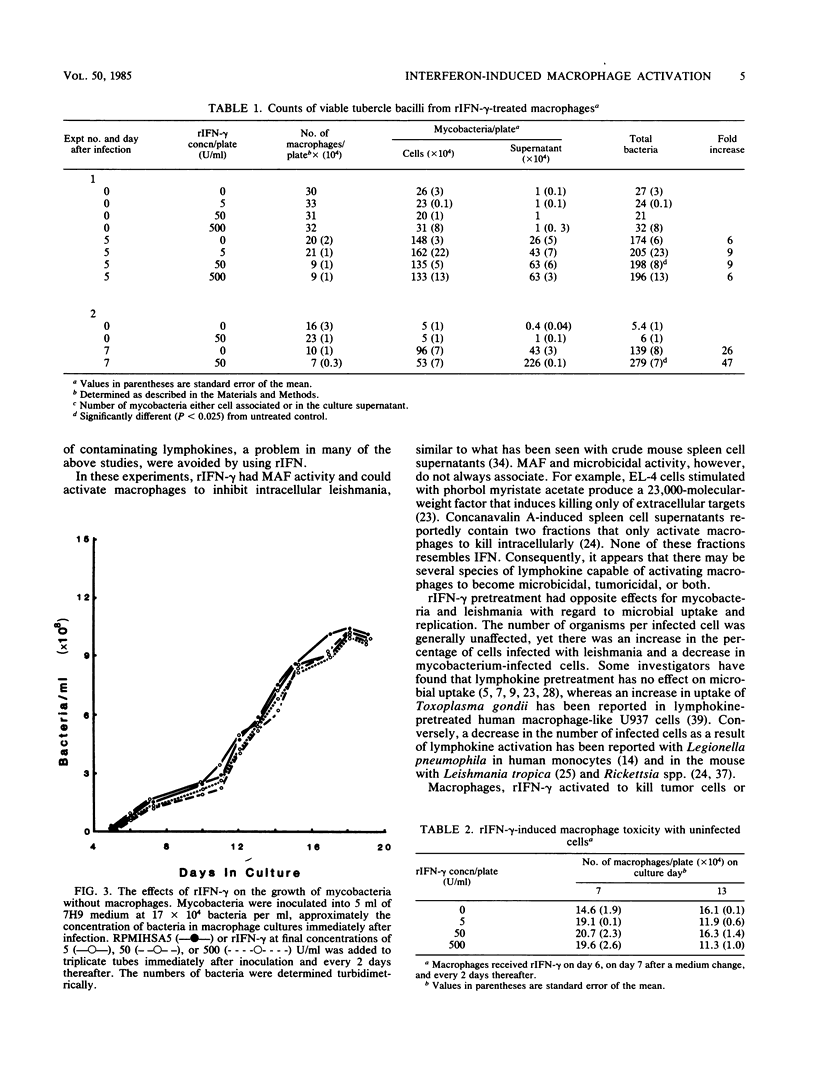
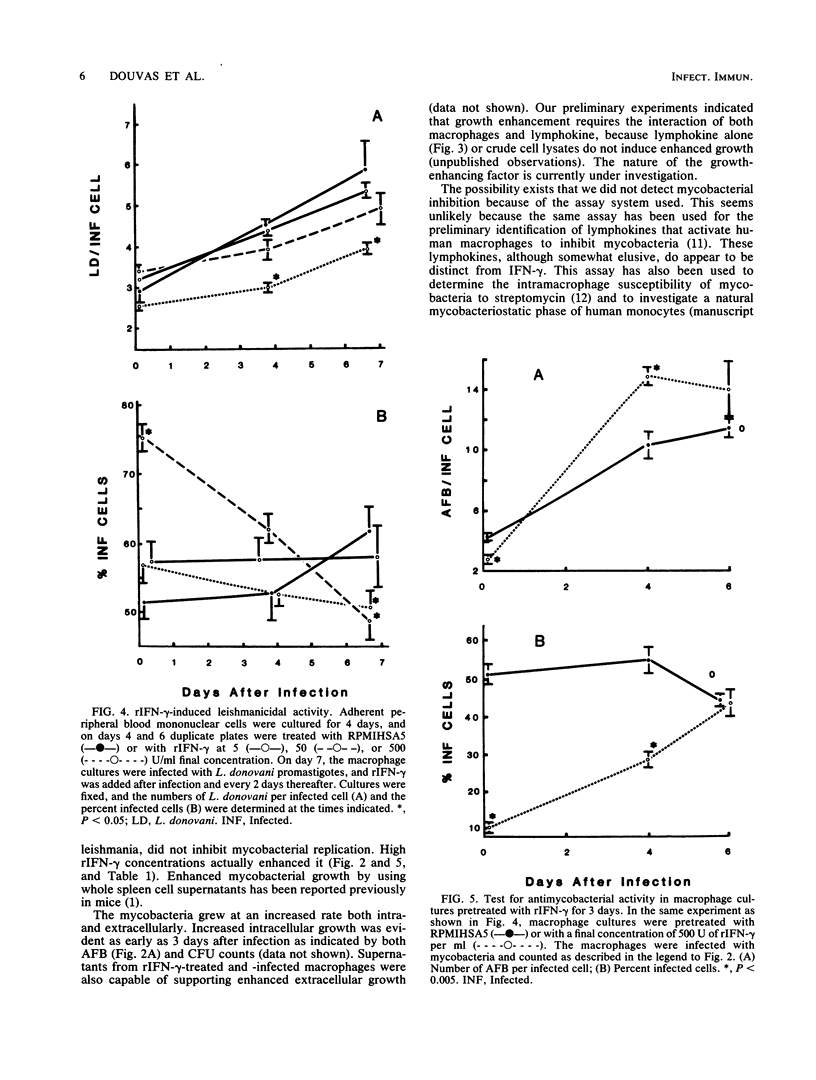
Recombinant human gamma interferon (rIFN-gamma) was examined for its ability to activate human peripheral blood monocyte-derived macrophages to kill tumor cells and to affect the replication of two phylogenetically distinct intracellular pathogens, Mycobacterium tuberculosis and Leishmania donovani. Macrophages preincubated overnight with doses of rIFN-gamma from 5 to 500 U/ml killed [3H]thymidine-labeled mouse L929 tumor targets, as measured by the release of [3H]thymidine into the supernatant after 48 h. Counts of macrophages initially infected with leishmania promastigotes showed that rIFN-gamma-pretreated macrophages could both inhibit the replication of and kill the resulting intramacrophage amastigotes over a 7-day period. However, rIFN-gamma pretreatment of macrophages actually enhanced mycobacterial replication over a 5- to 7-day period, as assessed by (i) counting acid-fast bacilli or (ii) lysing macrophages to release bacteria and determining the numbers of viable units. Mycobacterial growth was not affected by rIFN-gamma in the absence of macrophages. rIFN-gamma pretreatment had opposite effects on the uptake of mycobacteria and leishmania. As many as 80% fewer activated macrophages ingested mycobacteria compared with controls, whereas 50% more activated macrophages were infected with leishmania. These results suggest that rIFN-gamma may interfere with the immune destruction of intracellular tubercle bacilli and that the mechanisms of immunity against mycobacteria and leishmania may differ.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander J., Smith C. C. Growth of Mycobacterium lepraemurium in nonstimulated and stimulated mouse peritoneal-derived and bone marrrow-derived macrophages in vitro. Infect Immun. 1978 Dec;22(3):631–636. doi: 10.1128/iai.22.3.631-636.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaman L., Benjamini E., Pappagianis D. Role of lymphocytes in macrophage-induced killing of Coccidioides immitis in vitro. Infect Immun. 1981 Nov;34(2):347–353. doi: 10.1128/iai.34.2.347-353.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berens R. L., Brun R., Krassner S. M. A simple monophasic medium for axenic culture of hemoflagellates. J Parasitol. 1976 Jun;62(3):360–365. [PubMed] [Google Scholar]
- Biondi A., Roach J. A., Schlossman S. F., Todd R. F., 3rd Phenotypic characterization of human T lymphocyte populations producing macrophage-activating factor (MAF) lymphokines. J Immunol. 1984 Jul;133(1):281–285. [PubMed] [Google Scholar]
- Biroum-Noerjasin Listericidal activity of non-stimulated and stimulated human macrophages in vitro. Clin Exp Immunol. 1977 Apr;28(1):138–145. [PMC free article] [PubMed] [Google Scholar]
- Byrne G. I., Faubion C. L. Lymphokine-mediated microbistatic mechanisms restrict Chlamydia psittaci growth in macrophages. J Immunol. 1982 Jan;128(1):469–474. [PubMed] [Google Scholar]
- Cole P. Activation of mouse peritoneal cells to kill Listeria monocytogenes by T-lymphocyte products. Infect Immun. 1975 Jul;12(1):36–41. doi: 10.1128/iai.12.1.36-41.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowle A. J., May M. Preliminary demonstration of human tuberculoimmunity in vitro. Infect Immun. 1981 Jan;31(1):453–464. doi: 10.1128/iai.31.1.453-464.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowle A. J., Sbarbaro J. A., Judson F. N., Douvas G. S., May M. H. Inhibition by streptomycin of tubercle bacilli within cultured human macrophages. Am Rev Respir Dis. 1984 Nov;130(5):839–844. doi: 10.1164/arrd.1984.130.5.839. [DOI] [PubMed] [Google Scholar]
- Hoover D. L., Berger M., Nacy C. A., Hockmeyer W. T., Meltzer M. S. Killing of Leishmania tropica amastigotes by factors in normal human serum. J Immunol. 1984 Feb;132(2):893–897. [PubMed] [Google Scholar]
- Horwitz M. A., Silverstein S. C. Activated human monocytes inhibit the intracellular multiplication of Legionnaires' disease bacteria. J Exp Med. 1981 Nov 1;154(5):1618–1635. doi: 10.1084/jem.154.5.1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann S. H. Effective antibacterial protection induced by a Listeria monocytogenes-specific T cell clone and its lymphokines. Infect Immun. 1983 Mar;39(3):1265–1270. doi: 10.1128/iai.39.3.1265-1270.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann S. H., Hahn H. Biological functions of t cell lines with specificity for the intracellular bacterium Listeria monocytogenes in vitro and in vivo. J Exp Med. 1982 Jun 1;155(6):1754–1765. doi: 10.1084/jem.155.6.1754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelley V. E., Fiers W., Strom T. B. Cloned human interferon-gamma, but not interferon-beta or -alpha, induces expression of HLA-DR determinants by fetal monocytes and myeloid leukemic cell lines. J Immunol. 1984 Jan;132(1):240–245. [PubMed] [Google Scholar]
- Kerckhaert J. A., Hofhuis F. M., Willers J. M. Influence of cyclophosphamide on delayed hypersensitivity and acquired cellular resistance to Listeria monocytogenes in the mouse. Immunology. 1977 Jun;32(6):1027–1032. [PMC free article] [PubMed] [Google Scholar]
- Le J., Prensky W., Yip Y. K., Chang Z., Hoffman T., Stevenson H. C., Balazs I., Sadlik J. R., Vilcek J. Activation of human monocyte cytotoxicity by natural and recombinant immune interferon. J Immunol. 1983 Dec;131(6):2821–2826. [PubMed] [Google Scholar]
- Liew F. Y., Howard J. G., Hale C. Prophylactic immunization against experimental leishmaniasis. III. Protection against fatal Leishmania tropica infection induced by irradiated promastigotes involves Lyt-1+2- T cells that do not mediate cutaneous DTH. J Immunol. 1984 Jan;132(1):456–461. [PubMed] [Google Scholar]
- Murray H. W., Cartelli D. M. Killing of intracellular Leishmania donovani by human mononuclear phagocytes. Evidence for oxygen-dependent and -independent leishmanicidal activity. J Clin Invest. 1983 Jul;72(1):32–44. doi: 10.1172/JCI110972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nacy C. A., James S. L., Benjamin W. R., Farrar J. J., Hockmeyer W. T., Meltzer M. S. Activation of macrophages for microbicidal and tumoricidal effector functions by soluble factors from EL-4, a continuous T cell line. Infect Immun. 1983 May;40(2):820–824. doi: 10.1128/iai.40.2.820-824.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nacy C. A., Leonard E. J., Meltzer M. S. Macrophages in resistance to rickettsial infections: characterization of lymphokines that induce rickettsiacidal activity in macrophages. J Immunol. 1981 Jan;126(1):204–207. [PubMed] [Google Scholar]
- Nacy C. A., Meltzer M. S., Leonard E. J., Wyler D. J. Intracellular replication and lymphokine-induced destruction of Leishmania tropica in C3H/HeN mouse macrophages. J Immunol. 1981 Dec;127(6):2381–2386. [PubMed] [Google Scholar]
- Nakagawara A., Nathan C. F. A simple method for counting adherent cells: application to cultured human monocytes, macrophages and multinucleated giant cells. J Immunol Methods. 1983 Jan 28;56(2):261–268. doi: 10.1016/0022-1759(83)90418-0. [DOI] [PubMed] [Google Scholar]
- Nogueira N., Cohn Z. A. Trypanosoma cruzi: in vitro induction of macrophage microbicidal activity. J Exp Med. 1978 Jul 1;148(1):288–300. doi: 10.1084/jem.148.1.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orme I. M., Collins F. M. Adoptive protection of the Mycobacterium tuberculosis-infected lung. Dissociation between cells that passively transfer protective immunity and those that transfer delayed-type hypersensitivity to tuberculin. Cell Immunol. 1984 Mar;84(1):113–120. doi: 10.1016/0008-8749(84)90082-0. [DOI] [PubMed] [Google Scholar]
- Roberts W. K., Vasil A. A convenient and sensitive cytotoxicity assay for macrophage activating factor. J Immunol Methods. 1982 Nov 12;54(3):371–377. doi: 10.1016/0022-1759(82)90321-0. [DOI] [PubMed] [Google Scholar]
- Rothermel C. D., Rubin B. Y., Murray H. W. Gamma-interferon is the factor in lymphokine that activates human macrophages to inhibit intracellular Chlamydia psittaci replication. J Immunol. 1983 Nov;131(5):2542–2544. [PubMed] [Google Scholar]
- Sacks D. L., Perkins P. V. Identification of an infective stage of Leishmania promastigotes. Science. 1984 Mar 30;223(4643):1417–1419. doi: 10.1126/science.6701528. [DOI] [PubMed] [Google Scholar]
- Schreiber R. D., Pace J. L., Russell S. W., Altman A., Katz D. H. Macrophage-activating factor produced by a T cell hybridoma: physiochemical and biosynthetic resemblance to gamma-interferon. J Immunol. 1983 Aug;131(2):826–832. [PubMed] [Google Scholar]
- Scott P., Sacks D., Sher A. Resistance to macrophage-mediated killing as a factor influencing the pathogenesis of chronic cutaneous leishmaniasis. J Immunol. 1983 Aug;131(2):966–971. [PubMed] [Google Scholar]
- Turco J., Winkler H. H. Cloned mouse interferon-gamma inhibits the growth of Rickettsia prowazekii in cultured mouse fibroblasts. J Exp Med. 1983 Dec 1;158(6):2159–2164. doi: 10.1084/jem.158.6.2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turco J., Winkler H. H. Effect of mouse lymphokines and cloned mouse interferon-gamma on the interaction of Rickettsia prowazekii with mouse macrophage-like RAW264.7 cells. Infect Immun. 1984 Aug;45(2):303–308. doi: 10.1128/iai.45.2.303-308.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker L., Lowrie D. B. Killing of Mycobacterium microti by immunologically activated macrophages. Nature. 1981 Sep 3;293(5827):69–71. doi: 10.1038/293069a0. [DOI] [PubMed] [Google Scholar]
- Wing E. J., Koren H. S., Fischer D. G., Kelley V. Stimulation of a human macrophage-like cell line (U-937) to inhibit multiplication of an intracellular pathogen. J Reticuloendothel Soc. 1981 Apr;29(4):321–328. [PubMed] [Google Scholar]
- Wu-Hsieh B., Zlotnik A., Howard D. H. T-cell hybridoma-produced lymphokine that activates macrophages to suppress intracellular growth of Histoplasma capsulatum. Infect Immun. 1984 Jan;43(1):380–385. doi: 10.1128/iai.43.1.380-385.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlotnik A., Crowle A. J. Lymphokine-induced mycobacteriostatic activity in mouse pleural macrophages. Infect Immun. 1982 Aug;37(2):786–793. doi: 10.1128/iai.37.2.786-793.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlotnik A., Roberts W. K., Vasil A., Blumenthal E., Larosa F., Leibson H. J., Endres R. O., Graham S. D., Jr, White J., Hill J. Coordinate production by a T cell hybridoma of gamma interferon and three other lymphokine activities: multiple activities of a single lymphokine? J Immunol. 1983 Aug;131(2):794–800. [PubMed] [Google Scholar]



