Abstract
The estrogen-related receptor α (ERRα) is an orphan member of the superfamily of nuclear hormone receptors expressed in tissues that preferentially metabolize fatty acids. Despite the molecular characterization of ERRα and identification of target genes, determination of its physiological function has been hampered by the lack of a natural ligand. To further understand the in vivo function of ERRα, we generated and analyzed Estrra-null (ERRα−/−) mutant mice. Here we show that ERRα−/− mice are viable, fertile and display no gross anatomical alterations, with the exception of reduced body weight and peripheral fat deposits. No significant changes in food consumption and energy expenditure or serum biochemistry parameters were observed in the mutant animals. However, the mutant animals are resistant to a high-fat diet-induced obesity. Importantly, DNA microarray analysis of gene expression in adipose tissue demonstrates altered regulation of several enzymes involved in lipid, eicosanoid, and steroid synthesis, suggesting that the loss of ERRα might interfere with other nuclear receptor signaling pathways. In addition, the microarray study shows alteration in the expression of genes regulating adipogenesis as well as energy metabolism. In agreement with these findings, metabolic studies showed reduced lipogenesis in adipose tissues. This study suggests that ERRα functions as a metabolic regulator and that the ERRα−/− mice provide a novel model for the investigation of metabolic regulation by nuclear receptors.
Nuclear receptors are ligand-regulated transcription factors that control key pathways required for normal development and maintenance of homeostasis throughout life (37). Nuclear receptors now comprise a family of 48 genes in mice and humans that encode structurally and functionally related proteins. However, the existence of fewer than 10 receptors had been predicted by classic physiological and biochemical studies (10). Since the discovery of many nuclear receptors had not been anticipated and thus is not linked to recognized natural ligands, these new gene products were referred to as orphan nuclear receptors. During the last decade, extensive study of this gene family revealed that orphan nuclear receptors control essential developmental and metabolic functions in response to natural ligands as diverse as steroid hormones, retinoic acids, leukotrienes, bile acids, cholesterol metabolites, and long-chain fatty acids (reviewed in references 5, 26, and 48). In addition, orphan nuclear receptors have been shown to react to the presence of exogenous ligands such as pesticides (15, 70), phenobarbital (53, 61), and a wide variety of xenobiotic agents and drugs (2, 24, 39, 55, 66, 68, 69). Gene deletion analyses in mice have been particularly useful to uncover biological functions of orphan nuclear receptors. Nuclear orphan receptors have been shown to participate in the development and/or maintenance of the placenta, somitogenesis, brain, heart, hypothalamus-pituitary axis, immune system, and pathways controlling steroidogenesis and cholesterol metabolism.
The estrogen-related receptor α (ERRα) (NR3B1) was the first orphan nuclear receptor identified more than a decade ago on the basis of its close homology to the classic estrogen receptor α (ERα) (NR3A1) (14). ERRα was also identified as a repressor of the simian virus 40 major late promoter, revealing that ERRα had the ability to regulate gene expression in the absence of exogenous stimuli (67). Two other orphan nuclear receptors, ERRβ (NR3B2) (6, 14) and ERRγ (NR3B3) (9, 16, 18), also belong to the same subfamily.
ERRα expression begins at day 9.5 post coitum in extraembryonic tissues and is later detected in the heart, intestine, brain, spinal cord, brown fat, and bone (4, 50, 62). ERRα is expressed in a nearly ubiquitous fashion in adult tissues but most prominently in tissues demonstrating a high capacity for fatty acid β-oxidation or activation, suggesting that ERRα may play a role in regulating cellular energy balance (14, 21, 50). This is further supported by the observations that ERRα is a direct regulator of the medium-chain acyl coenzyme A dehydrogenase gene (MCAD) (50, 64) and that PGC-1, a coactivator regulating transcription in response to signals relaying metabolic needs, is a potent regulator of ERRα activity (19, 21, 49).
Recent experiments have also shown that ERRα is functionally closer to the ERs than originally anticipated (reviewed in reference 13). Like the ERs, the ERRs recognize the ERE as a homodimer, suggesting that these receptors may control overlapping regulatory pathways (32, 45, 73). In addition, ERRα binds to extended half-sites with the consensus sequence TCAAGGTCA referred to as an ERRE (22, 50, 64, 67, 71). ERα has also been shown to recognize a subset of ERREs and stimulate the transcriptional activity of promoters containing such sites, reinforcing the concept that the two classes of related receptors share common target genes (63). It has also been discovered that the synthetic estrogen diethylstilbestrol and the partial estrogen antagonist 4-hydroxytamoxifen can act as selective inverse agonists on the three ERR isoforms, leading to the dissociation of coactivator proteins and loss of transcriptional activation function (8, 58, 59). In addition, we have recently demonstrated that the ERR isoforms can regulate the transcriptional activity of the human breast cancer marker gene pS2 and, unexpectedly, that the full transcriptional activity of the ERs and the ERRs on the pS2 gene is dependent on the presence of a previously unidentified ERRE within its promoter (32). Taken together, these results suggest that ERRα may also influence classic endocrine estrogenic pathways. However, in spite of these studies, the exact physiological roles of ERRα remain unknown.
To explore the biological functions of ERRα in vivo, we used gene targeting technology to generate ERRα deficient mice (ERRα−/− mice). These ERRα−/− mice are viable and fertile and show no gross anatomical abnormalities. However, ERRα−/− mice display reduced fat mass and are resistant to high-fat diet-induced obesity, and their adipose tissue shows alterations in the expression of several genes directly linked to lipid metabolism and adipogenesis. ERRα−/− mice thus provide a new model to investigate nuclear receptor-regulated metabolic pathways and associated physiology and pathology.
MATERIALS AND METHODS
Targeting of ERRα and production of chimeric mice.
Three overlapping λ clones containing the mouse Estrra locus were isolated from a 129Sv genomic library (a gift from A. Joyner, Skirball Institute, New York, N.Y.) and characterized by restriction mapping and direct sequencing of the exon boundaries. The knockout construct was created by using pNT (60) and contained 6.73 kb of genomic DNA flanking the second exon of Estrra. An endfilled 4.36-kb BamHI/NotI fragment, lying upstream of the second exon, was cloned into the XhoI site of pNT, whereas a 2.37-kb HindIII fragment was cloned between the neor and thymidine kinase cassettes to provide the 3′ arm of the construct. Correct targeting of the Estrra locus replaces the second exon of the receptor, which encodes the amino-terminal region and an essential component of its DNA-binding domain, with a neo cassette. The linearized construct was electroporated into R1 embryonic stem (ES) cells (41), which were selected with G418 (150 μg/ml) and ganciclovir (2 μM). Two ES cell clones were isolated and injected into C57BL/6 blastocysts to generate chimeras, and three chimeras transmitted the mutation to their offspring. Heterozygous mice, generated by mating the chimeric animals with 129SvJ mice were mated with C57BL/6 animals to generate hybrid F1 animals: physiologic studies were performed by using the F2-null mutant and wild-type offspring obtained by mating the F1 hybrid heterozygotes, with the exception of the study on high-fat diet-induced obesity, which was performed with ERRα-null mice in the C57BL/6 background. Complete disruption of the Estrra allele was verified by performing Northern and Western blots with RNA and proteins obtained from kidney and intestine, respectively, as previously described (50).
Studies of Estrra−/− mice.
Mice were housed in an specific-pathogen-free facility with a daily 12-h light cycle (7:00 a.m. to 7:00 p.m.) and with free access to food and water. Between two and four mice were housed in each cage. Growth curves were obtained by weighing mice of defined ages between 10:00 and 12:00 a.m. Fasting serum and biochemical studies were performed between 10:00 and 12:00 a.m. with animals that had been deprived of food for 18 h. Body composition was determined by desiccating mouse carcasses from which the intestines had been removed (29). After desiccation, the carcass was homogenized and a 1-g aliquot was saponified by using potassium hydroxide and extracted with petroleum ether. After complete evaporation of the ether, the residue was weighed to determine the fat content. Statistical comparisons of the body composition data was performed by using the Mann-Whitney U test. Baseline biochemical studies were performed with serum samples obtained from tail bleeds of restrained animals at between 20 and 28 weeks of age. Enzymatic assays were used to determine serum triglycerides (GPO-PAP; Boehringer Mannheim) and glycerol (TC Glycerin; Boehringer Mannheim), glucose (Glucose Oxidase-Trinder; Sigma), free fatty acids (GPO-PAP Half Micro Test; Boehringer Mannheim), and β-hydroxybutyrate (TC β-hydroxybutyrate; Boehringer Mannheim). Energy balance and body composition measurements were performed as previously described (46). For high-fat feeding, a diet containing 45% (wt/wt) fat content (D12451; Research Diets, Inc., New Brunswick, N.J.) was used.
DNA microarrays and quantitative PCR.
Microarray analysis was performed with pooled adipose tissue obtained from two wild-type and two ERRα−/− adult male mice. The animals were fasted overnight and euthanized by using Avertin. Total RNA was prepared from homogenized adipose tissue by using Trizol reagent according to the manufacturer's instructions (Invitrogen, Carlsbad, Calif.). Probes for microarray analysis were prepared by using 10 μg of total RNA and hybridized to Affymetrix (Santa Clara, Calif.) Mu74Av2 GeneChips. Detailed protocols for the probe synthesis and hybridization reactions, as well as for the posthybridization washing and staining, have been previously described (43). The hybridized arrays were scanned and raw data extracted by using Microarray Analysis Suite 5.0 (Affymetrix). Expression profiles obtained from both knockout animals were compared individually to those obtained from each wild-type mouse by using Microarray Analysis Suite 5.0, which examines the hybridization intensities of individual probe pairs in order to estimate the significance of observed changes in gene expression: in the present study, the default range for the change P value (lower cutoff, 0.0025; upper cutoff, 0.9975) was used to identify genes whose expression changed in each comparison. Genes whose expression levels differed in three of four comparisons were considered to show significantly different levels of expression between the wild-type and knockout animals. For quantitative PCR, total RNA was extracted from white and brown fat tissues of wild-type and knockout animals by using Trizol reagent. cDNA was prepared by reverse transcription of 1 μg of total RNA with Superscript II enzyme and oligo(dT) primer. The resulting cDNAs were amplified by using the LightCycler FastStart DNA Master SYBR Green I kit and a LightCycler instrument according to the manufacturer's instructions (Roche Diagnostics, Indianapolis, Ind.). All mRNA expression data was normalized to hypoxanthine phosphoribosyltransferase expression in the corresponding sample.
Lipogenesis rate.
Mice were studied at 10:00 a.m. after free access to food overnight. The animals were conditioned by sham intraperitoneal injections of water. On the day of the experiment, the animals were injected intraperitoneally with 3H2O (0.5 mCi per 100 g of body weight) and sacrificed by cervical dislocation 30 min later. Adipose tissue samples were harvested and stored at −80°C. The tissues were homogenized and heated in ethanolic KOH. The resulting tissue extracts, which contained saponified lipids, were acidified with concentrated sulfuric acid and extracted by using petroleum ether. The extracts were dried by evaporation, and 3H incorporation in the lipid fraction was determined by scintillation counting.
RESULTS
Targeting of the murine Estrra gene.
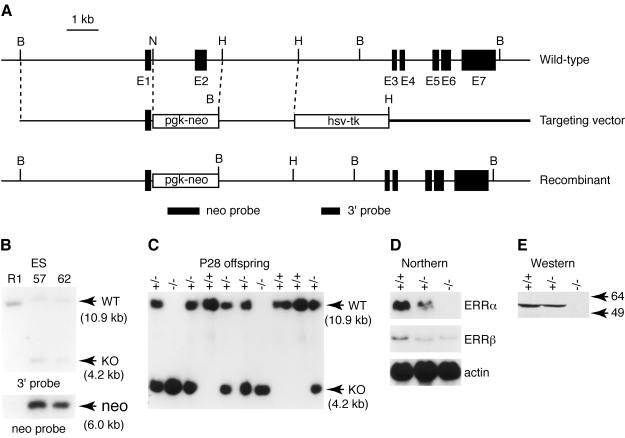
The gene targeting strategy was designed to produce mice null for Estrra by removing the exon encoding the amino-terminal domain and the first zinc finger (DNA-binding domain) and replacing it with a neor cassette (Fig. 1A). Southern blot analysis of genomic DNA with an external probe and a neo probe from two ERRα+/− ES cell lines (Fig. 1B) confirmed that the locus had been targeted as expected.
FIG. 1.
Targeted disruption of the murine Estrra gene in ES cells and mice. (A) Schematic representation of the Estrra locus and targeting vector. Black boxes in the wild-type allele schematics represent exons of the Estrra gene. The open boxes in the targeting vector schematics correspond to the pgk-neo and hsv-tk selectable marker genes. The position of the 3′ flanking and neomycin probes used in Southern blot analysis are indicated by filled boxes. (B) Homologous recombination of the targeting vector in ES cells was verified by Southern blot analysis, digesting genomic DNA with BamHI, and hybridization with a 3′-flanking probe (upper panel) and a neomycin probe (lower panel). The wild-type allele generated a 10.9-kb band, while the mutant allele produced a 4.2-kb band due to the introduction of a BamHI site in the targeting vector. The neomycin probe recognized a 6.0-kb band in the targeted locus. (C) Southern blot analysis of 4-week-old offspring from heterozygote intercrosses with the 3′-flanking probe. (D) Northern blot analysis of ERRα expression. Total RNA isolated from adult wild-type, heterozygous, and homozygous kidneys. Twenty micrograms of total RNA were used in each lane. ERRα and ERRβ transcripts were detected by using mouse cDNA probes (51). A mouse β-actin cDNA probe was used as a loading control (lower panel). (E) Western analysis of ERRα knockout mice. Intestine lysates were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The blot was probed with antibodies against mouse ERRα, which were made by using the amino-terminal domain of the ERRα protein.
ERRα−/− mice are viable and fertile.
Heterozygotes for the disrupted ERRα allele were apparently normal and were crossed to generate homozygous mutant mice. Figure 1C shows a representative Southern blot of 4-week-old offspring from crosses of heterozygous mice. Homozygous ERRα−/− mice were obtained at the expected Mendelian frequency and did not differ from their wild-type littermates in overall health, fertility (including interbreeding between ERRα−/− mice), and longevity. Histological examinations detected no obvious abnormalities in the brain, spinal cord, pituitary, heart, kidneys, adrenal, testis, ovary, intestine, and liver (data not shown). Northern blot analysis with total mRNA extracted from kidneys failed to detect ERRα transcripts in ERRα−/− mice, whereas approximately half the amount of transcripts was observed in ERRα+/− mice (Fig. 1D). Interestingly, the levels of the ERRβ transcript were found to be downregulated in both heterozygous and ERRα knockout animals, suggesting that ERRα may regulate ERRβ expression in the kidneys. Finally, no ERRα protein could be detected by Western blot analysis of proteins extracted from the intestine of knockout animals (Fig. 1E).
ERRα−/− mice have reduced fat mass.
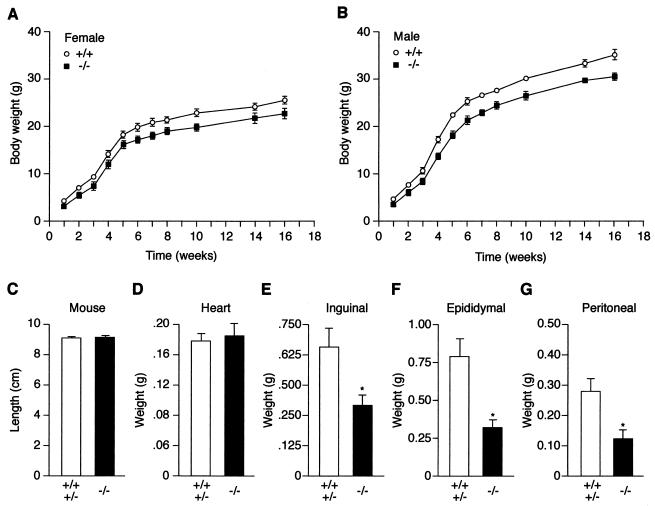
To verify the general well-being of ERRα−/− mice and explore the possibility that ERRα may play a role in maintaining homeostasis in the adult animal, their growth rates relative to those of wild-type littermate controls were measured in terms of total body weight (Fig. 2A and B). During the first postnatal week, the mean body weight of ERRα−/− mice was slightly decreased, although it was not statistically different from wild-type littermates. However, beginning at postnatal week 2, ERRα−/− mice displayed a significant decrease in body weight. At postnatal week 10, for example, both male and female ERRα−/− mice weighed ca. 15% less than their wild-type littermates (P < 0.01). No significant reduction in body weight was observed in heterozygous ERRα+/− mice (Table 1 and data not shown). The body length (nose to anus) of 10-week-old male and female mice showed that linear growth was not impaired in ERRα−/− mice (Fig. 2C). In addition, the weight of the heart did not differ in ERRα−/− mice (Fig. 2D). However, ERRα−/− had a 50 to 60% reduction in the weight of the inguinal, epididymal and peritoneal fat pads (Fig. 2E to G). Biochemical characterization of carcass composition showed that the ERRα-null mice had, in comparison with control littermates, 80.6% fat, 105% lean mass, and 106.4% water (Table 1). The histology of fat tissue shows an apparent normal number of adipocytes with decreased adipocyte size (Fig. 3).
FIG. 2.
Comparison of total weight, length, and fat pad weight of wild-type and ERRα-null mice. (A) Growth of female ERRα-null mice. ERRα−/− F2 mice (▪; n = 15) and littermate wild-type controls (○; n = 16) were weighed, and the data were binned by using 1- to 4-week intervals. The data are means ± the standard error of the mean (sem). (B) Growth of male ERRα-null mice. ERRα−/− F2 mice (▪; n = 13) and littermate wild-type controls (○; n = 10) were weighed, and the data were binned by using 1- to 4-week intervals. The data are mean ± the SEM. (C to F) Anatomical characteristics of wild-type plus heterozygote (n = 9) and ERRα-null (n = 7) mice at 26 weeks of age. The P values (✽) were as follows: inguinal, 0.008; epididymal, 0.002; and peritoneal, 0.005.
TABLE 1.
Body composition of ERRα knockout mice and control littermatesa
| Parameter | Mean value ± SEM for mouse genotype:
|
Ratio (−/− vs +/+) (%) |
P
|
|||
|---|---|---|---|---|---|---|
| +/+ | +/− | −/− | −/− vs +/+ | −/− vs +/+ and +/− | ||
| Wt (g) | 32.9 ± 1.3 | 32.5 ± 1.8 | 27.3 ± 1.0 | 83.0 | 0.0014 | 0.00034 |
| Fat mass (g) | 6.9 ± 1.0 | 6.9 ± 1.2 | 4.6 ± 0.5 | 67.4 | 0.039 | 0.014 |
| Lean mass (g) | 6.9 ± 0.2 | 6.8 ± 0.3 | 6.0 ± 0.2 | 87.4 | 0.0669 | 0.0016 |
| Water content (g) | 14.9 ± 0.3 | 14.6 ± 0.4 | 13.1 ± 0.4 | 88.0 | 0.0054 | 0.0011 |
| Fat mass (%) | 23.5 ± 2.2 | 23.6 ± 2.7 | 19.0 ± 1.6 | 80.7 | 0.053 | 0.029 |
| Lean mass (%) | 24.2 ± 0.6 | 24.2 ± 0.8 | 25.4 ± 0.4 | 105.0 | 0.061 | 0.036 |
| Water content (%) | 52.2 ± 1.8 | 52.2 ± 2.0 | 55.6 ± 1.3 | 106.4 | 0.12 | 0.071 |
The results are shown for wild-type (n = 7), heterozygous (n = 6), and knockout mice (n = 15). The mean body weight of the ERRα knockout mice was significantly decreased and was accompanied by decreased total fat mass and compensatory increases in lean mass and water content.
FIG. 3.
Histology of adipose tissue. Hematoxylin-and-eosin-stained wild-type (WT) and ERRα−/− epididymal WATs are shown. Magnification, ×150.
Serum biochemistry of ERRα−/− mice.
To further investigate the biological basis of this phenotype, a series of physiological tests were performed on the ERRα−/− mice. Fasting glucose levels, as well as serum free fatty acids and triglyceride levels, were not significantly affected by the absence of ERRα (data not shown).
Food consumption and energy expenditure.
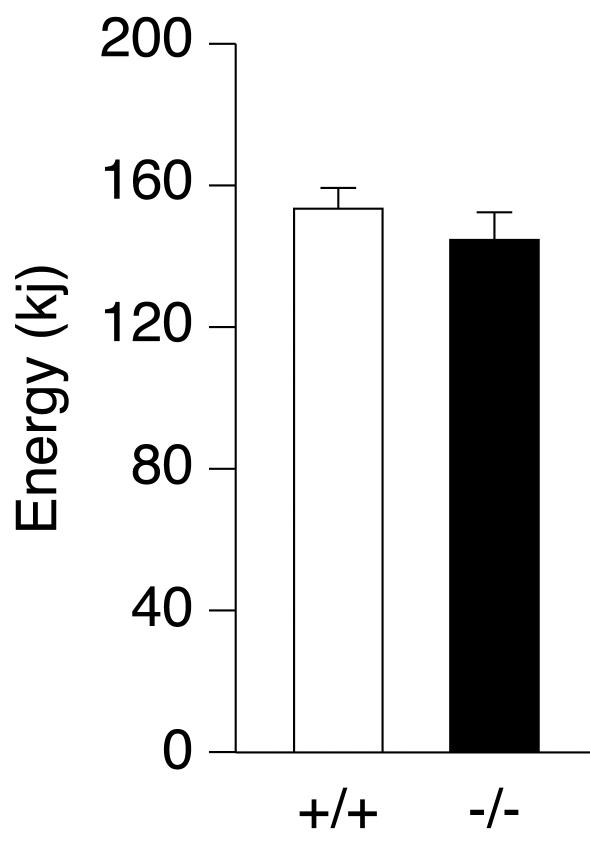
To examine whether the reduction in fat mass observed in ERRα−/− mice was caused by decreased food intake or increased energy expenditure, both parameters were measured over a 4-day period. We observed no statistically significant change in either parameter (Fig. 4 and data not shown).
FIG. 4.
Energy consumption in wild-type (n = 4) and ERRα−/− (n = 8) male mice over a 4-day period.
ERRα−/− mice are resistant to high-fat diet-induced obesity.
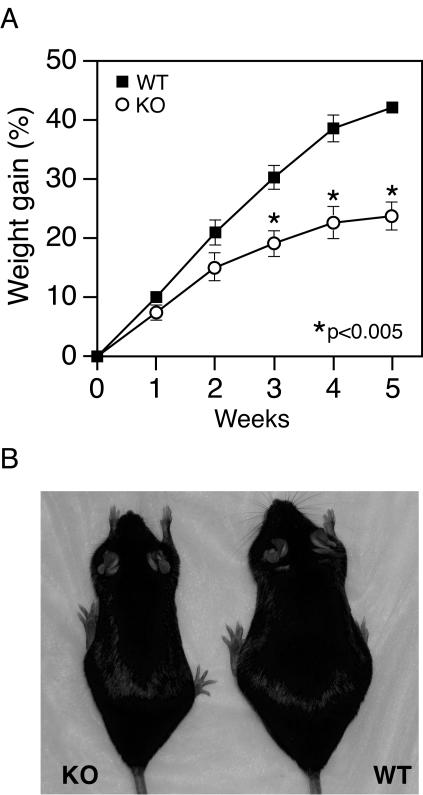
We next challenged 10-week-old mice with a high-fat diet containing 45% (wt/wt) fat for 5 weeks to stimulate weight gain. As shown in Fig. 5A, weight gain was significantly reduced in the ERRα−/− mice relative to the control littermates. A statistical difference between the two groups was observed in the third week of feeding. By the end of the experiment, the control littermates had become mildly obese (Fig. 5B), whereas the ERRα−/− mice weighed only slightly more than wild-type mice fed a normal diet.
FIG. 5.
ERRα−/− mice are resistant to high-fat diet-induced obesity. (A) Ten-week-old male mice (n = 7 for each group) were fed a high-fat diet. The increases in body weights were calculated based on the initial body weight at day 0 of high-fat feeding. The average initial body weight was 10% smaller (P < 0.05) for the ERRα−/− mice compared to the control littermates. (B) Representative male ERRα−/− mouse and control littermate after 5 weeks of feeding with a high fat diet.
DNA microarrays.
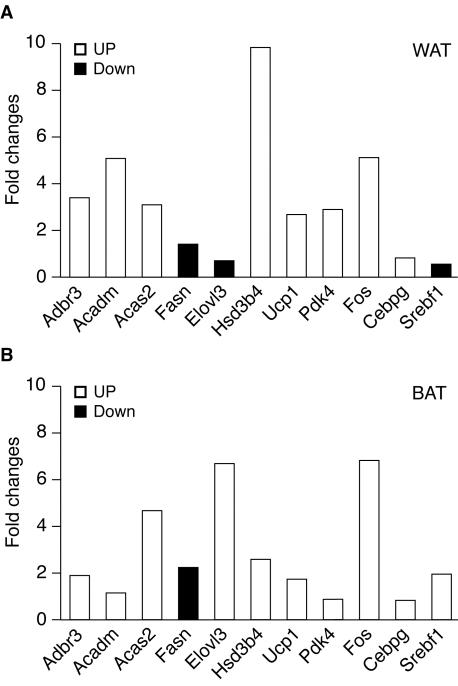
Microarray studies were performed to determine whether ERRα ablation causes changes in gene expression in adipose tissue (Table 2). Pooled adipose tissue was harvested from fasted adult male mice and studied by using Affymetrix GeneChips. The expression studies show downregulation of ERRα and changes in the expression of two known ERRα target genes: lactotransferrin and MCAD. The expression studies also demonstrate altered regulation of several enzymes involved in lipid metabolism (fatty acid synthase [gene Fasn], stearoyl-coenzyme A desaturase 2 [Scd2], fatty acid coenzyme A ligase long-chain 5, [Facl5], acetyl-coenzyme A dehydrogenase medium chain [Acadm], and acetyl-coenzyme A dehydrogenase long chain [Acadl]) and energy metabolism (cytochrome c somatic [Cycs], carnitine acetyltransferase [Crat], creatine kinase mitochondrial 2 [Ckmt2], and uncoupling protein 1 [Ucp1]). These changes create an imbalance between enzymes involved in fat synthesis and fat catabolism that may result in decreased levels of de novo triglyceride synthesis in the knockout animals. The alterations in transcript expression observed in the microarray studies may include genes that are direct ERRα targets, as well as genes that are regulated by other transcription factors or cell signaling pathways. In particular, the microarray studies demonstrate alterations in sterol regulatory element binding factor 1 (Srebf1) and CCAAT/enhancer binding protein gamma (Cebpg) expression, transcription factors that have been implicated in the regulation of adipogenesis (reviewed in reference 11). In addition, decreased expression of prostaglandin D2 synthase (Ptgds) and hydroxysteroid dehydrogenase-4 δ〈5〉-3-β (Hsd3b4) and hydroxysteroid dehydrogenase-6 δ〈5〉-3-β (Hsd3b6) isoforms may alter peroxisome proliferator-activated receptor (PPAR) activity by regulating levels of their putative endogenous ligands, which in turn may explain prostaglandin-endoperoxide synthase 2 (Ptgs2) upregulation (25, 72). Finally, adipose tissue from ERRα knockout animals also expresses increased levels of adrenergic receptor β3, a known regulator of murine adiposity (54). The quality of the array data was further validated by quantitative PCR analysis of 15 selected genes indicated in bold in Table 1. Of these genes, changes in the expression of 11 of 15 (74%) were validated by quantitative PCR, including Esrra, whose expression could not be detected as expected. The levels of expression for two genes, Ckmt21 and Hdac6, in white adipose tissue (WAT) were also too low to obtain accurate measurements. For two other genes, Ptgds and Ucp1, we observed upregulation (∼30- and 2.7-fold, respectively) by quantitative PCR, while the microarray data indicated that the expression of these genes was downregulated. Thus, we are confident that, for more than 75% of genes identified as differentially expressed, the results of our microarray gene screening method is accurate. The changes in the expression of the validated genes in the WAT of ERRα−/− mice compared to wild-type mice, as obtained by quantitative PCR, are shown in Fig. 6A. Of note, true fold changes are generally greater than the averaged microarray data, leading us to include genes whose level of expression was as low as 1.2-fold (Table 2). Since brown fat expressed high levels of ERRα, we also measured changes in expression levels of the selected genes in this tissue (Fig. 6B). In most cases, changes in gene expression observed in brown fat parallel those seen in white fat, with the exception of Elovl3 and Srebf1.
TABLE 2.
Differentially expressed genes in the adipose tissue of ERRα knockout micea
| Function | Probe set | Gene | Fold change | Unigene no. | LocusLink no. | Cytoband no. (centimorgans) | Description |
|---|---|---|---|---|---|---|---|
| Fat metabolism | 92536_at | Adrb3 | 1.7 | Mm.4369 | 11556 | 8 (10.0) | Adrenergic receptor, beta 3 |
| 92581_at | Acadm | 1.5 | Mm.10530 | 11364 | 3 (73.6) | Acetyl coenzyme A dehydrogenase, medium chain | |
| 160921_at | Acas2 | 1.5 | Mm.7044 | 68738 | Acetyl coenzyme A synthetase 2 | ||
| 95425_at | Acad1 | 1.4 | Mm.198049 | 98243 | 1 | Acetyl coenzyme A dehydrogenase, long chain | |
| 93754_at | Ech1 | 1.3 | Mm.2112 | 51798 | Enoyl coenzyme A hydratase 1, peroxisomal | ||
| 103646_at | Crat | 1.3 | Mm.20396 | 12908 | 2 (18.0) | Carnitine acetyltransferase | |
| 97456_at | Facl5 | −1.3 | Mm.30263 | 71879 | Fatty acid coenzyme A ligase, long chain 5 | ||
| 95758_at | Scd2 | −1.5 | Mm.193096 | 20250 | 19 (43.0) | Stearoyl coenzyme A desaturase 2 | |
| 98575_at | Fasn | −1.7 | Mm.3760 | 14104 | 11 (72.0) | Fatty acid synthase | |
| 103469_at | Elovl3 | −7.6 | Mm.21806 | 12686 | 19 (47.0) | Elongation of very long chain fatty acids (FEN1/Elo2, SUR4/Elo3, yeast)-like 3 | |
| Eicosanoid metabolism | 104647_at | Ptgs2 | 1.5 | Mm.3137 | 19225 | 1 (76.2) | Prostaglandin-endoperoxide synthase 2 |
| 92545_f_at | Ptgds | −3.6 | Mm.1008 | 19215 | 2 (12.9) | Prostaglandin D2 synthase (21 kDa, brain) | |
| Sterol or steroid metabolism | 92869_at | Hsd3b4 | 4.8 | Mm.14309 | 15495 | 3 (49.1) | Hydroxysteroid dehydrogenase-4,delta5-3-beta |
| 102729_f_at | Hsd3b6 | 2.6 | Mm.14435 | 15497 | Hydroxysteroid dehydrogenase-6,delta5-3-beta | ||
| 104509_at | Ch25h | −2.7 | Mm.30824 | 12642 | Cholesterol 25-hydroxylase | ||
| Electron transport | 98132_at | Cycs | −1.4 | Mm.35389 | 13063 | 6 (23.0) | Cytochrome c, somatic |
| 99507_at | Ucp1 | −2.6 | Mm.4177 | 22227 | 8 (38.0) | Uncoupling protein 1, mitochondrial | |
| Other metabolism | 102049_at | Pdk4 | 1.8 | Mm.10283 | 27273 | 6 (0.63) | Pyruvate dehydrogenase kinase 4 |
| 97950_at | Xdh | 1.3 | Mm.11223 | 22436 | 17 (45.3) | Xanthine dehydrogenase | |
| 94966_at | G6pdx | −1.3 | Mm.27210 | 14381 | X (30.02) | Glucose-6-phosphate dehydrogenase X linked | |
| 101044_at | Alad | −1.4 | Mm.6988 | 17025 | 4 (30.6) | Aminolevulinate, delta-, dehydratase | |
| 104172_at | Folr2 | −1.5 | Mm.2724 | 14276 | 7 | Folate receptor 2 (fetal) | |
| 101408_at | Gamt | −1.5 | Mm.7329 | 14431 | 10 (43.0) | Guanidinoacetate methyltransferase | |
| 100046_at | Mthfd2 | −1.7 | Mm.443 | 17768 | 6 (35.15) | Methylenetetrahydrofolate dehydrogenase (NAD+ dependent) | |
| 162015_f_at | Ckmt2 | −1.9 | Mm.20240 | 76722 | Creatine kinase, mitochondrial 2 | ||
| 97089_at | Folh1 | −1.9 | Mm.7522 | 53320 | 7 (D1-D2) | Folate hydrolase | |
| Gene transcription | 160901_at | Fos | 2.3 | Mm.5043 | 14281 | 12 (40.0) | FBJ osteosarcoma oncogene |
| 99532_at | Tob1 | 1.5 | Mm.4218 | 22057 | Transducer of ErbB-2.1 | ||
| 160526_s_at | Crem | 1.3 | Mm.5244 | 12916 | 18 A | Cyclic cAMP responsive element modulator | |
| 92195_at | Cebpg | 1.3 | Mm.3402 | 12611 | CCAAT/enhancer binding protein (C/EBP), gamma | ||
| 94812_at | Gtf2h1 | 1.3 | Mm.22700 | 14884 | General transcription factor IIH, polypeptide 1 (62-kDa subunit) | ||
| 100984_at | Atf1 | 1.2 | Mm.676 | 11908 | Activating transcription factor 1 | ||
| 100094_at | Supt5h | 1.2 | Mm.460 | 20924 | (710.0) | Suppressor of Ty5 homolog (Saccharomyces cerevisiae) | |
| 96633_s_at | Mrgx-pendi | −1.2 | Mm.27218 | 56397 | MORF-related gene X | ||
| 94288_at | H1f2 | −1.4 | Mm.193539 | 50708 | H1 histone family, member 2 | ||
| 103634_at | Isgf3g | −1.4 | Mm.2032 | 16391 | 14 (21.5) | Interferon-dependent positive acting transcription factor 3 gamma | |
| 93264_at | Srebf1 | −1.5 | Mm.214958 | 20787 | 8 (33.0) | Sterol regulatory element binding factor 1 | |
| 92502_at | Zac1 | −1.6 | Mm.42200 | 22634 | 10 (15.0) | Zinc finger protein regulator of apoptosis and cell cycle arrest | |
| 99059_at | Elf3 | −1.6 | Mm.3963 | 13710 | E74-like factor 3 | ||
| 104471_at | Hdac6 | −1.9 | Mm.29854 | 15185 | X (2.0) | Histone deacetylase 6 | |
| 101913_at | Hcy1 | −2.0 | Mm.29581 | 15213 | 3 (2.4) | Hairy/enhancer-of-split related with YRPW motif 1 | |
| 103964_at | Esrra | −2.0 | Mm.2563 | 26379 | 19 (3.0) | ERRα | |
| 93915_at | Pou2af1 | −2.1 | Mm.897 | 18985 | POU domain, class 2, associating factor 1 |
The results of microarray analysis performed on the WAT of male ERRα knockout mice are shown for replicated studies performed with two wild-type and two ERRα−/− mice. The fold change represents an average of the values obtained by a pairwise comparison of the animals. A total of 305 probe sets were differentially regulated in the ERRα−/− mice: those implicated in lipid metabolism or its regulation, as well as transcriptional regulation, are shown here. Genes in boldface were tested by quantitative PCR analysis (Fig. 6 and data not shown).
FIG. 6.
The absence of ERRα−/− influences the expression of genes involved in lipid metabolism in both white (A) and brown (B) adipose tissues. Quantitative PCR data shows the difference in expression of both upregulated (□) and downregulated (▪) genes in these tissues. The data displayed represent the means of at least two experiments.
Lipogenesis.
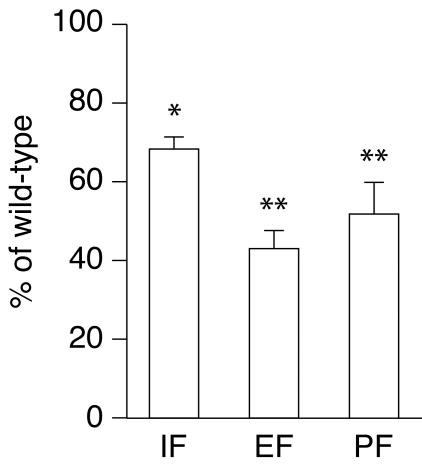
Given the observation of altered expression of key enzymes involved in lipid metabolism, we next investigated whether de novo synthesis of triglyceride is affected in the knockout animals. Lipogenesis was assessed by treating ERRα−/− mice with 3H2O: the amount of radioactive label incorporated into triacylglycerol can be measured by saponification and ether extraction of adipose tissues and other organs (12). ERRα−/− mice demonstrated significantly decreased lipogenesis in comparison to littermate controls: knockout animals show a 30 to 55% decrease in 3H incorporation into adipose tissue lipids (Fig. 7). This finding demonstrates that adipose tissues of knockout mice possess a defect in triglyceride synthesis, which may result from decreased adipocyte glycolytic activity, decreased fatty acid synthesis, or decreased esterification.
FIG. 7.
ERRα−/− mice demonstrate decreased lipogenesis in comparison to littermate controls. After intraperitoneal injection of with 3H2O, ERRα−/− mice incorporate 30 to 55% less 3H into adipose tissue lipids. IF, inguinal fat; EF, epididymal fat; PF, perirenal fat. ✽, P < 0.05; ✽✽, P < 0.01.
DISCUSSION
We have generated ERRα-deficient mice through homologous recombination in ES cells. No ERRα expression was detectable in these mice by Northern and Western blot analyses and quantitative PCR. The mice were viable and fertile and have a normal life span. Although ERRα-deficient mice weighed less compared to their wild-type littermates, histological analysis of adult tissues failed to show morphological abnormalities. Therefore, we conclude that there is no absolute requirement for ERRα function in development, sexual maturation, and homeostasis. This finding is in sharp contrast with the phenotype observed with the ERRβ-null mice who die in utero due to the complete absence of the diploid trophoblast layers of the placenta (36). This finding is also surprising in view of the very high level of ERRα expression in brown adipose tissue (50) and the proposed role for ERRα in bone formation (3, 4).
Although ERRα deficiency by itself may be largely asymptomatic, if present with other genetic defects such as null mutations in other members of the ERR or ER families, it could conceivably give rise to an overtly abnormal developmental, metabolic, or endocrine phenotype. In fact, deletion of genes encoding nuclear receptors does not always produce an apparent phenotype. Perhaps the most dramatic example is that of the genes encoding the three retinoic acid receptors (RARα, -β, and -γ). Mice devoid of either RARα (30, 34), RARβ (35, 40), or RARγ (31) display only subtle developmental phenotypes. An extensive functional redundancy between RARs accounts for this observation as severe developmental defects results from the lack any combinations of two RAR genes (reviewed in reference 38). Disruption of other nuclear receptor genes produce a phenotype only when animals are physiologically or pharmacologically challenged, such as with a high cholesterol diet in LXRα mutant mice (44) or xenobiotic agents in PXR/SXR-null mice (52, 68, 69). Therefore, a more evident phenotype may be observed only in ERR double mutants or in response to a specific physiological stress.
Although the ERRα−/− mice appear normal, they display reduced body weight and diminished fat mass (Fig. 2). We were unable to detect statistically significant changes in eating behavior or energy expenditure between the wild-type and ERRα-null littermates. One possible explanation for these results is that the putative increase in energy expenditure is small and significant only over a long period of time. How the absence of ERRα leads to this phenotype at the molecular level is currently unknown. However, gene expression profiling experiments in adipose tissue of wild-type versus knockout mice are providing interesting avenues of investigation. First, the expression studies confirmed a role of ERRα in MCAD (Acadm) gene regulation, predominantly as a repressor, since MCAD expression is upregulated in the adipose tissue of the knockout mice. This result indicates that ERRα can indeed act as a direct regulator of metabolic genes. Second, the loss of ERRα function leads to alterations in the expression of a wide variety of genes encoding transcriptional regulators, two of them (Srebf1 and Cebpg) directly implicated in the regulation of adipogenesis. The absence of ERRα might therefore induce significant changes in a regulatory cascade that normally controls the expression of genes involved in fat and sterol metabolism. We have also noted significant alterations in the expression of genes involved in sterol, steroid, and prostaglandin synthesis, suggesting that the loss of ERRα might indirectly influence the activity of other nuclear receptor-based signaling pathways, many of which are known to regulate lipid metabolism an adipogenesis (1, 11, 33, 47). Third, the gene microarray analysis detected changes in the expression of metabolic enzymes directly involved in lipid synthesis, an observation correlated with a marked decrease in lipogenesis rate in white fat tissues. Finally, the observed upregulation of the gene encoding the β3 adrenergic receptor suggests that lipolysis in white adipocytes could also be affected (7), although more specific studies will be required to validate this hypothesis. It is, however, interesting that the Adrb3 promoter has been shown to encode a putative binding site for ERRα (28). Although the physiological significance of the alteration in expression of each specific gene remains to be fully investigated, this ensemble is in agreement with the observed reduced fat mass in the ERRα−/− mice.
As introduced above, ERRα has been shown to possess the ability to interfere with estrogen signaling (reviewed in reference 13). Estrogen is known to play an important role in WAT regulation: in rodents, ovariectomy increases the amount of WAT, whereas estrogen replacement decreases the amount of WAT (65). Similar observations were made in postmenopausal women subjected to hormone replacement therapy (56). More recently, increased adipose tissue in male and female ERα-null mice was observed, demonstrating conclusively a role for estrogen and ERα in WAT functions (17). The ERRα-null mice displays the opposite phenotype, an observation consistent with the fact that ERRα can act as a potent transcriptional repressor in specific biological settings and compete with the ERs for binding sites and coactivators (27, 50, 57, 67, 73). The upregulation of Fos, a direct target of ERα signaling (20), in the white and brown adipose tissues of the ERRα-null mice is in agreement with the proposed cross talk between the two nuclear receptor-based regulatory pathways. In addition, the inhibition of estrogen production in aromatase-deficient mice results in increased adipose tissue mass and hepatic steatosis (23), which is associated with reduced rates of hepatic β-oxidation and decreased expression of AOX, VLACS, and MCAD (42). It will be interesting to test whether the combined loss of ERα and ERRα in mice “resets” their metabolism toward a more wild-type-like state.
In light of the well-known caveat that mouse development and life under controlled laboratory conditions do not reproduce the environment that mice find themselves in the wild, it is now evident that ERRα is not absolutely required for normal mouse development and growth. However, it is also evident that the ERRα-null mice display metabolic abnormalities that warrant further investigation. The development of ERRα specific ligands and the current availability of high-throughput gene expression profiling technology coupled with future ERRα−/−-based mouse models will provide further insight into the role played by ERRα in metabolic control and other biological processes.
Acknowledgments
We thank A. Joyner for the ES cell line; Annie Matthyssen, Savita Amin, Majid Ghahremani, and Geneviève Deblois for technical assistance; Rogerio Rabelo and members of the Giguère laboratory for helpful discussions; and Michel Tremblay for critical reading of the manuscript.
V.G. is a Senior Scientist of the Canadian Institutes of Health Research (CIHR). This work was supported by operating grants from the CIHR to V.G.
REFERENCES
- 1.Barak, Y., M. C. Nelson, E. S. Ong, Y. Z. Jones, P. Ruiz-Lozano, K. R. Chien, A. Koder, and R. M. Evans. 1999. PPARγ is required for placental, cardiac, and adipose tissue development. Mol. Cell 4:585-595. [DOI] [PubMed] [Google Scholar]
- 2.Blumberg, B., W. Sabbagh, Jr., H. Juguilon, J. Bolado, Jr., C. M. van Meter, E. S. Ong, and R. M. Evans. 1998. SXR, a novel steroid and xenobiotic-sensing nuclear receptor. Genes Dev. 12:3195-3205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bonnelye, E., L. Merdad, V. Kung, and J. E. Aubin. 2001. The orphan nuclear estrogen receptor-related receptor α (ERRα) is expressed throughout osteoblast differentiation and regulates bone formation in vitro. J. Cell Biol. 153:971-984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bonnelye, E., J. M. Vanacker, T. Dittmar, A. Begue, X. Desbiens, D. T. Denhardt, J. E. Aubin, V. Laudet, and B. Fournier. 1997. The ERR-1 orphan receptor is a transcriptional activator expressed during bone development. Mol. Endocrinol. 11:905-916. [DOI] [PubMed] [Google Scholar]
- 5.Chawla, A., J. J. Repa, R. M. Evans, and D. J. Mangelsdorf. 2001. Nuclear receptors and lipid physiology: opening the X-files. Science 294:1866-1870. [DOI] [PubMed] [Google Scholar]
- 6.Chen, F., Q. Zhang, T. McDonald, M. J. Davidoff, W. Bailey, C. Bai, Q. Liu, and C. T. Caskey. 1999. Identification of two hERR2-related novel nuclear receptors utilizing bioinformatics and inverse PCR. Gene 228:101-109. [DOI] [PubMed] [Google Scholar]
- 7.Collins, S., and R. S. Surwit. 2001. The β-adrenergic receptors and the control of adipose tissue metabolism and thermogenesis. Rec. Prog. Horm. Res. 56:309-328. [DOI] [PubMed] [Google Scholar]
- 8.Coward, P., D. Lee, M. V. Hull, and J. M. Lehmann. 2001. 4-Hydroxytamoxifen binds to and deactivates the estrogen-related receptor γ. Proc. Natl. Acad. Sci. USA 98:8880-8884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eudy, J. D., S. Yao, M. D. Weston, M. Ma-Edmonds, C. B. Talmage, J. J. Cheng, W. J. Kimberling, and J. Sumegi. 1998. Isolation of a gene encoding a novel member of the nuclear receptor superfamily from the critical region of Usher syndrome type IIa at 1q41. Genomics 50:382-384. [DOI] [PubMed] [Google Scholar]
- 10.Evans, R. M. 1988. The steroid and thyroid hormone receptor superfamily. Science 240:889-895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fajas, L., J. C. Fruchart, and J. Auwerx. 1998. Transcriptional control of adipogenesis. Curr. Opin. Cell Biol. 10:165-173. [DOI] [PubMed] [Google Scholar]
- 12.Freake, H. C., H. L. Schwartz, and J. H. Oppenheimer. 1989. The regulation of lipogenesis by thyroid hormone and its contribution to thermogenesis. Endocrinology 125:2868-2874. [DOI] [PubMed] [Google Scholar]
- 13.Giguère, V. 2002. To ERR in the estrogen pathway. Trends Endocrinol. Metab. 13:220-225. [DOI] [PubMed] [Google Scholar]
- 14.Giguère, V., N. Yang, P. Segui, and R. M. Evans. 1988. Identification of a new class of steroid hormone receptors. Nature 331:91-94. [DOI] [PubMed] [Google Scholar]
- 15.Harmon, M. A., M. F. Boehm, R. A. Heyman, and D. J. Mangelsdorf. 1995. Activation of mammalian retinoid X receptors by the insect growth regulator methoprene. Proc. Natl. Acad. Sci. USA 92:6157-6160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heard, D. J., P. L. Norby, J. Holloway, and H. Vissing. 2000. Human ERRγ, a third member of the estrogen receptor-related receptor (ERR) subfamily of orphan nuclear receptors: tissue-specific isoforms are expressed during development and in the adult. Mol. Endocrinol. 14:382-392. [DOI] [PubMed] [Google Scholar]
- 17.Heine, P. A., J. A. Taylor, G. A. Iwamoto, D. B. Lubahn, and P. S. Cooke. 2000. Increased adipose tissue in male and female estrogen receptor-α knockout mice. Proc. Natl. Acad. Sci. USA 97:12729-12734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hong, H., L. Yang, and M. R. Stallcup. 1999. Hormone-independent transcriptional activation and coactivator binding by novel orphan nuclear receptor ERR3. J. Biol. Chem. 274:22618-22626. [DOI] [PubMed] [Google Scholar]
- 19.Huss, J. M., R. P. Kopp, and D. P. Kelly. 2002. PGC-1α coactivates the cardiac-enriched nuclear receptor ERR α and γ via novel leucine-rich interaction interfaces. J. Biol. Chem. 277:40265-40274. [DOI] [PubMed] [Google Scholar]
- 20.Hyder, S. M., G. M. Stancel, and D. S. Loose-Mitchell. 1991. Presence of an estradiol response region in the mouse c-fos oncogene. Steroids 56:498-504. [DOI] [PubMed] [Google Scholar]
- 21.Ichida, M., S. Nemoto, and T. Finkel. 2002. Identification of a specific molecular repressor of the peroxisome proliferator-activated peceptor γ coactivator-1α (PGC-α). J. Biol. Chem. 277:50991-50995. [DOI] [PubMed] [Google Scholar]
- 22.Johnston, S. D., X. Liu, F. Zuo, T. L. Eisenbraun, S. R. Wiley, R. J. Kraus, and J. E. Mertz. 1997. Estrogen-related receptor α1 functionally binds as a monomer to extended half-site sequences including ones contained within estrogen-response elements. Mol. Endocrinol. 11:342-352. [DOI] [PubMed] [Google Scholar]
- 23.Jones, M. E., A. W. Thorburn, K. L. Britt, K. N. Hewitt, N. G. Wreford, J. Proietto, O. K. Oz, B. J. Leury, K. M. Robertson, S. Yao, and E. R. Simpson. 2000. Aromatase-deficient (ArKO) mice have a phenotype of increased adiposity. Proc. Natl. Acad. Sci. USA 97:12735-12740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jones, S. A., L. B. Moore, J. L. Shenk, G. B. Wisely, G. A. Hamilton, D. D. McKee, N. C. Tomkinson, E. L. LeCluyse, M. H. Lambert, T. M. Willson, S. A. Kliewer, and J. T. Moore. 2000. The pregnane X receptor: a promiscuous xenobiotic receptor that has diverged during evolution. Mol. Endocrinol. 14:27-39. [DOI] [PubMed] [Google Scholar]
- 25.Kim, J. B., H. M. Wright, M. Wright, and B. M. Spiegelman. 1998. ADD1/SREBP1 activates PPARγ through the production of endogenous ligand. Proc. Natl. Acad. Sci. USA 95:4333-4337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kliewer, S. A., J. M. Lehmann, and T. M. Willson. 1999. Orphan nuclear receptors: shifting endocrinology into reverse. Science 284:757-760. [DOI] [PubMed] [Google Scholar]
- 27.Kraus, R. J., E. A. Ariazi, M. L. Farrell, and J. E. Mertz. 2002. Estrogen-related receptor α 1 actively antagonizes estrogen receptor-regulated transcription in MCF-7 mammary cells. J. Biol. Chem. 277:24826-24834. [DOI] [PubMed] [Google Scholar]
- 28.Kutoh, E., N. Ongenae, A. Claeskens, W. Verheyen, P. Cheyns, J. Neefs, and P. Kaijen. 2000. A putative white adipose tissue specific nuclear orphan receptor that interacts with the cAMP-response element of the human β3-adrenergic receptor gene. Mol. Cell. Endocrinol. 165:85-95. [DOI] [PubMed] [Google Scholar]
- 29.Leshner, A. I., V. A. Litwin, and R. L. Squibb. 1972. A simple method for carcass analysis. Physiol. Behav. 9:281-282. [DOI] [PubMed] [Google Scholar]
- 30.Li, E., H. M. Sucov, K.-H. Lee, R. M. Evans, and R. Jaenisch. 1993. Normal development and growth of mice carrying a targeted disruption of the α1 retinoic acid receptor gene. Proc. Natl. Acad. Sci. USA 90:1590-1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lohnes, D., P. Kastner, A. Dierich, M. Mark, M. LeMeur, and P. Chambon. 1993. Function of retinoic acid receptor γ in the mouse. Cell 73:643-658. [DOI] [PubMed] [Google Scholar]
- 32.Lu, D., Y. Kiriyama, K. Y. Lee, and V. Giguère. 2001. Transcriptional regulation of the estrogen-inducible pS2 beast cancer marker gene by the ERR family of orphan nuclear receptors. Cancer Res. 61:6755-6761. [PubMed] [Google Scholar]
- 33.Lu, T. T., J. J. Repa, and D. J. Mangelsdorf. 2001. Orphan nuclear receptors as eLiXiRs and FiXeRs of sterol metabolism. J. Biol. Chem. 276:37735-37738. [DOI] [PubMed] [Google Scholar]
- 34.Lufkin, T., D. Lohnes, M. Mark, A. Dierich, P. Gorrey, M.-P. Gaub, M. LeMeur, and P. Chambon. 1993. High postnatal lethality and testis degeneration in retinoic acid receptor α mutant mice. Proc. Natl. Acad. Sci. USA 90:7225-7229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Luo, J., P. Pasceri, R. A. Conlon, J. Rossant, and V. Giguère. 1995. Mice lacking all isoforms of retinoic acid receptor β develop normally and are susceptible to the teratogenic effects of retinoic acid. Mech. Dev. 53:61-71. [DOI] [PubMed] [Google Scholar]
- 36.Luo, J., R. Sladek, J.-A. Bader, J. Rossant, and V. Giguère. 1997. Placental abnormalities in mouse embryos lacking orphan nuclear receptor ERRβ. Nature 388:778-782. [DOI] [PubMed] [Google Scholar]
- 37.Mangelsdorf, D. J., C. Thummel, M. Beato, P. Herrlich, G. Schütz, K. Umesono, B. Blumberg, P. Kastner, M. Mark, P. Chambon, and R. M. Evans. 1995. The nuclear receptor superfamily: the second decade. Cell 83:835-839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mark, M., N. B. Ghyselinck, O. Wendling, V. Dupe, B. Mascrez, P. Kastner, and P. Chambon. 1999. A genetic dissection of the retinoid signalling pathway in the mouse. Proc. Nutr. Soc. 58:609-613. [DOI] [PubMed] [Google Scholar]
- 39.Masuyama, H., Y. Hiramatsu, M. Kunitomi, T. Kudo, and P. N. MacDonald. 2000. Endocrine disrupting chemicals, phthalic acid and nonylphenol, activate pregnane X receptor-mediated transcription. Mol. Endocrinol. 14:421-428. [DOI] [PubMed] [Google Scholar]
- 40.Mendelsohn, C., M. Mark, P. Dollé, A. Dierich, M.-P. Gaub, A. Krust, C. Lampron, and P. Chambon. 1994. Retinoic acid receptor β2 (RARβ2) null mutant mice appear normal. Dev. Biol. 166:246-258. [DOI] [PubMed] [Google Scholar]
- 41.Nagy, A., and J. Rossant. 1993. Production of completely ES cell-derived fetuses, p. 147-169. In A. L. Joyner (ed.), Gene targeting: a practical approach. Oxford University Press, Oxford, England.
- 42.Nemoto, Y., K. M. Toda, K. Ono, T. Fujikawa-Adachi, S. Saibara, H. Onishi, T. Enzan, Y. Okada, and Y. Shizuta. 2000. Altered expression of fatty acid-metabolizing enzymes in aromatase-deficient mice. J. Clin. Investig. 105:1819-1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Novak, J. P., R. Sladek, and T. J. Hudson. 2002. Characterization of variability in large-scale gene expression data: implications for study design. Genomics 79:104-113. [DOI] [PubMed] [Google Scholar]
- 44.Peet, D. J., S. D. Turley, A. Ma, B. A. Janowski, J.-M. A. Lobaccaro, R. E. Hammer, and D. J. Mangelsdorf. 1998. Cholesterol and bile acid metabolism are impaired in mice lacking the nuclear oxysterol receptor LXRα. Cell 93:693-704. [DOI] [PubMed] [Google Scholar]
- 45.Pettersson, K., K. Svensson, R. Mattsson, B. Carlsson, R. Ohlsson, and A. Berkenstam. 1996. Expression of a novel member of estrogen response element-binding nuclear receptors is restricted to the early stages of chorion formation during mouse embryogenesis. Mech. Dev. 54:211-223. [DOI] [PubMed] [Google Scholar]
- 46.Richard, D., F. Picard, C. Lemieux, J. Lalonde, P. Samson, and Y. Deshaies. 2002. The effects of topiramate and sex hormones on energy balance of male and female rats. Int. J. Obes. Relat. Metab. Disord. 26:344-353. [DOI] [PubMed] [Google Scholar]
- 47.Rosen, E. D., P. Sarraf, A. E. Troy, G. Bradwin, K. Moore, D. S. Milstone, B. M. Spiegelman, and R. M. Mortensen. 1999. PPARγ is required for the differentiation of adipose tissue in vivo and in vitro. Mol. Cell 4:611-617. [DOI] [PubMed] [Google Scholar]
- 48.Russell, D. W. 1999. Nuclear orphan receptors control cholesterol metabolism. Cell 97:539-542. [DOI] [PubMed] [Google Scholar]
- 49.Schreiber, S. N., D. Knutti, K. Brogli, T. Uhlmann, and A. Kralli. 2003. The transcriptional coactivator PGC-1 regulates the expression and activity of the orphan nuclear receptor ERRα. J. Biol. Chem. 278:9013-9018. [DOI] [PubMed] [Google Scholar]
- 50.Sladek, R., J.-A. Bader, and V. Giguère. 1997. The orphan nuclear receptor estrogen-related receptor α is a transcriptional regulator of the human medium-chain acyl coenzyme A dehydrogenase gene. Mol. Cell. Biol. 17:5400-5409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sladek, R., B. Beatty, J. Squire, N. G. Copeland, D. J. Gilbert, N. A. Jenkins, and V. Giguère. 1997. Chromosomal mapping of the human and murine orphan nuclear receptor ERRα (ESRRA) and ERRβ (ESRRB) and identification of a novel human ERRα-related pseudogene. Genomics 45:320-326. [DOI] [PubMed] [Google Scholar]
- 52.Staudinger, J. L., B. Goodwin, S. A. Jones, D. Hawkins-Brown, K. I. MacKenzie, A. LaTour, Y. Liu, C. D. Klaassen, K. K. Brown, J. Reinhard, T. M. Willson, B. H. Koller, and S. A. Kliewer. 2001. The nuclear receptor PXR is a lithocholic acid sensor that protects against liver toxicity. Proc. Natl. Acad. Sci. USA 98:3369-3374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sueyoshi, T., T. Kawamoto, I. Zelko, P. Honkakoski, and M. Negishi. 1999. The repressed nuclear receptor CAR responds to phenobarbital in activating the human CYP2B6 gene. J. Biol. Chem. 274:6043-6046. [DOI] [PubMed] [Google Scholar]
- 54.Susulic, V. S., R. C. Frederich, J. Lawitts, E. Tozzo, B. B. Kahn, M. E. Harper, J. Himms-Hagen, J. S. Flier, and B. B. Lowell. 1995. Targeted disruption of the β3-adrenergic receptor gene. J. Biol. Chem. 270:29483-29492. [DOI] [PubMed] [Google Scholar]
- 55.Synold, T. W., I. Dussault, and B. M. Forman. 2001. The orphan nuclear receptor SXR coordinately regulates drug metabolism and efflux. Nat. Med. 7:584-590. [DOI] [PubMed] [Google Scholar]
- 56.Tchernof, A., J. Calles-Escandon, C. K. Sites, and E. T. Poehlman. 1998. Menopause, central body fatness, and insulin resistance: effects of hormone-replacement therapy. Coron. Artery Dis. 9:503-511. [DOI] [PubMed] [Google Scholar]
- 57.Trapp, T., and F. Holsboer. 1996. Nuclear orphan receptor as a repressor of glucocorticoid receptor transcriptional activity. J. Biol. Chem. 271:9879-9882. [DOI] [PubMed] [Google Scholar]
- 58.Tremblay, G. B., D. Bergeron, and V. Giguère. 2001. 4-hydroxytamoxifen is an isoform-specific inhibitor of orphan estrogen-receptor-related (ERR) nuclear receptors β and γ. Endocrinology 142:4572-4575. [DOI] [PubMed] [Google Scholar]
- 59.Tremblay, G. B., T. Kunath, D. Bergeron, L. Lapointe, C. Champigny, J.-A. Bader, J. Rossant, and V. Giguère. 2001. Diethylstilbestrol regulates trophoblast stem cell differentiation as a ligand of orphan nuclear receptor ERRβ. Genes Dev. 15:833-838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tybulewicz, V. L. J., C. E. Crawford, P. K. Jackson, R. T. Bronson, and R. C. Mulligan. 1991. Neonatal lethality and lymphopoenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65:1153-1163. [DOI] [PubMed] [Google Scholar]
- 61.Tzameli, I., P. Pissios, E. G. Schuetz, and D. D. Moore. 2000. The xenobiotic compound 1,4-bis[2-(3,5-dichloropyridyloxy)]benzene is an agonist ligand for the nuclear receptor CAR. Mol. Cell. Biol. 20:2951-2958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vanacker, J. M., E. Bonnelye, C. Delmarre, and V. Laudet. 1998. Activation of the thyroid receptor α gene promoter by the orphan nuclear receptor ERRα. Oncogene 17:2429-2435. [DOI] [PubMed] [Google Scholar]
- 63.Vanacker, J. M., K. Pettersson, J. Gustafsson, and V. Laudet. 1999. Transcriptional targets shared by estrogen receptor-related receptors (ERRs) and estrogen receptor (ER) α, but not by ERβ. EMBO J. 18:4270-4279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vega, R. B., and D. P. Kelly. 1997. A role for estrogen-related receptor α in the control of mitochondrial fatty acid β-oxidation during brown adipocyte differentiation. J. Biol. Chem. 272:31693-31699. [DOI] [PubMed] [Google Scholar]
- 65.Wade, G. N., and J. M. Gray. 1979. Gonadal effects on food intake and adiposity: a metabolic hypothesis. Physiol. Behav. 22:583-593. [DOI] [PubMed] [Google Scholar]
- 66.Wei, P., J. Zhang, M. Egan-Hafley, S. Liang, and D. D. Moore. 2000. The nuclear receptor CAR mediates specific xenobiotic induction of drug metabolism. Nature 407:920-923. [DOI] [PubMed] [Google Scholar]
- 67.Wiley, S. R., R. J. Kraus, F. Zuo, E. E. Murray, K. Loritz, and J. E. Mertz. 1993. SV40 early-to-late switch involves titration of cellular transcriptional repressors. Genes Dev. 7:2206-2219. [DOI] [PubMed] [Google Scholar]
- 68.Xie, W., J. L. Barwick, M. Downes, B. Blumberg, C. M. Simon, M. C. Nelson, B. A. Neuschwander-Tetri, E. M. Brunt, P. S. Guzelian, and R. M. Evans. 2000. Humanized xenobiotic response in mice expressing nuclear receptor SXR. Nature 406:435-439. [DOI] [PubMed] [Google Scholar]
- 69.Xie, W., A. Radominska-Pandya, Y. Shi, C. M. Simon, M. C. Nelson, E. S. Ong, D. J. Waxman, and R. M. Evans. 2001. An essential role for nuclear receptors SXR/PXR in detoxification of cholestatic bile acids. Proc. Natl. Acad. Sci. USA 98:3375-3380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yang, C., and S. Chen. 1999. Two organochlorine pesticides, toxaphene and chlordane, are antagonists for estrogen-related receptor α-1 orphan receptor. Cancer Res. 59:4519-4524. [PubMed] [Google Scholar]
- 71.Yang, N., H. Shigeta, H. P. Shi, and C. T. Teng. 1996. Estrogen-related receptor, hERR1, modulates estrogen receptor-mediated response of human lactoferrin gene promoter. J. Biol. Chem. 271:5795-5804. [DOI] [PubMed] [Google Scholar]
- 72.Yu, K., W. Bayona, C. B. Kallen, H. P. Harding, C. P. Ravera, G. McMahon, M. Brown, and M. A. Lazar. 1995. Differential activation of peroxisome proliferator-activated receptors by eicosanoids. J. Biol. Chem. 270:23975-23983. [DOI] [PubMed] [Google Scholar]
- 73.Zhang, Z., and C. T. Teng. 2001. Estrogen receptor α and estrogen receptor-related receptor α1 compete for binding and coactivator. Mol. Cell. Endocrinol. 172:223-233. [DOI] [PubMed] [Google Scholar]