Abstract
We develop a superficial diffusing probe with a 3mm source-detector separation that can be used in combination with diffuse optical spectroscopic (DOS) methods to noninvasively determine full-spectrum optical properties of superficial in vivo skin in the wavelength range from 650 to 1000 nm. This new probe uses a highly scattering layer to diffuse photons emitted from a collimated light source and relies on a two-layer diffusion model to determine tissue absorption coefficient μa and reduced scattering coefficient . By employing the probe to measure two-layer phantoms that mimic the optical properties of skin, we demonstrate that the probe has an interrogation depth of 1 to 2mm. We carry out SSFDPM (steady state frequency-domain photon migration) measurements using this new probe on the volar forearm and palm of 15 subjects, including five subjects of African descent, five Asians, and five Caucasians. The optical properties of in vivo skin determined using the superficial diffusing probe show considerable similarity to published optical properties of carefully prepared ex vivo epidermis+dermis.
Keywords: tissue optics, optical properties, epithelial tissue, near-infrared spectroscopy, photon migration
1 Introduction
The ability to accurately quantify the optical properties of superficial skin is important for many clinical applications including the determination of light distribution in laser-induced cutaneous therapies;1 monitoring skin blood oxygenation, melanin concentration,2 and water concentration;3 and improving noninvasive optical measurements of tissue.4 Investigation of the optical properties of skin has been carried out by several researchers. Troy and Thennadil,5 Simpson et al.,6 and Baskatov et al.,7 characterized the optical properties of ex vivo skin using an integrating-sphere-based approach. Although the optical properties of skin can be assessed using such ex vivo techniques, the necessity of taking biopsies from subjects’ skin limits the clinical usefulness of these techniques. Currently, in vivo techniques that are capable of measuring optical properties of skin volumes consisting primarily of epidermis+dermis do exist, but are limited. For example, Zhang et al. determined the optical properties of in vivo skin using visible reflectance spectroscopy along with a multilayer skin model and a genetic optimization algorithm.8 A multilayer skin model and a number of fitting parameters, such as layer thickness, chromophores, and scattering properties for each layer, and their corresponding ranges must be chosen carefully in advance to avoid nonuniqueness in the solution space. Amelink et al. developed differential pathlength spectroscopy to investigate local optical properties of tissue, and this has been successfully applied to study bronchial mucosa.9 However, this technique requires that all of the chromophores contributing to the measured signals are known in advance to separate absorption and reduced scattering coefficients from the measured reflectance. For the case in which constituent chromophores cannot be determined or the chromophore absorption spectra are not accurate, the empirical mathematical model will not recover tissue optical properties correctly.
We developed a new fiber-based probe that can be used to determine the optical properties of superficial in vivo skin using diffuse optical spectroscopy (DOS) in conjunction with a two-layer diffusion model.10 This probe employs a highly diffusing Spectralon layer that enables a diffusion model for small source-detector distances that results in superficial sampling. In this paper, we briefly review the probe design and the DOS method. To verify the applicability of this probe for measuring the optical properties of skin, we fabricated two-layer phantoms with a thin top layer ranging in thickness from 1 to 8mm. The optical properties of the two-layer phantoms are designed to simulate those of the epidermis+dermis layer and the subcutaneous fat layer of skin.8 These layered phantom results illustrate that for samples with optical properties similar to skin, the superficial diffusing probe of source-detector separation 3mm has an interrogation less than 2mm. Measurements using this probe were subsequently carried out in vivo at two anatomical locations (volar forearm and palm) on 15 subjects (five subjects of African descent, five Asians, and five Caucasians). The optical properties obtained using the superficial diffusing probe correlate closely with the layer structure of skin and demonstrate similarities to those of ex vivo skin determined from integrating sphere measurement techniques6,7 (Caucasian skin μa=0.03 to 0.06/mm and =1.8 to 2.8/mm at the wavelength of 650 nm).
2 Materials and Methods
2.1 Layer Structure of Skin
Skin is a layered heterogeneous medium that consists of epidermis, dermis, and subcutaneous fat, as illustrated in Fig. 1. The epidermis is composed primarily of several cell layers of basal keratinocytes and ranges from tens to hundreds of micrometer in thickness, depending on anatomic location. In normal skin, melanocytes within the epidermis are the primary source of apparent skin color. The dermis, which may be a few hundred micrometer to several millimeters thick, underlies the epidermis and provides skin with mechanical integrity via a meshwork of structural proteins including collagen and elastin. A capillary vascular supply permeates the papillary dermis. Capillary blood flow, structural proteins, and to a lesser extent, cells (such as fibroblasts) all contribute to the absorption and scattering properties of the dermis layer. For most anatomic locations, a subcutaneous fat layer, which is composed of adipose, connective fibrils and blood vessels, underlies the dermis. The scattering and absorption properties of subcutaneous tissue are introduced by adipocytes, structural fibrils, and blood.
Fig. 1.
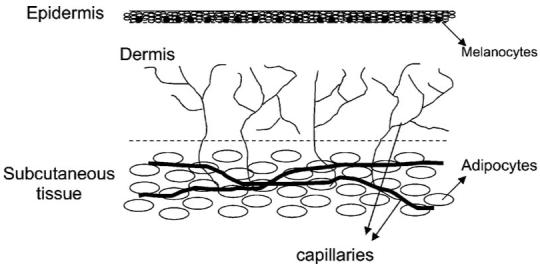
Layer structure of skin.
2.2 Instrumentation
A steady state frequency-domain photon migration (SSFDPM) instrument, which was been described in detail elsewhere11,12 and successfully applied to deep tissue optical property studies,13,14 was employed to carry out measurements in this study. In brief, the instrument consists of two subsystems: (1) a steady state light source and spectrometer and (2) a bank of frequency-modulated laser diodes and an avalanche photodiode. The steady state subsystem includes a tungsten halogen light source (Ocean Optics, Model HL2000) and spectrometer (BWTek, Model 611) to record wavelength-dependent reflectance from the sample under investigation. The frequency domain photon migration apparatus employs six laser diodes at six wavelengths—661, 681, 783, 806, 823, and 850 nm—which are sinusoidally modulated at frequencies ranging from 50 to 300 MHz. The diffuse reflectance is detected by an avalanche photodiode (Hamamatsu, Model C5658). The detected phase delay and the amplitude demodulation introduced by tissues were fit to an appropriate photon diffusion model to calculate absorption and reduced scattering coefficients for each laser wavelength. These optical properties were used to scale the reflectance obtained from the steady state subsystem. By fitting the reduced scattering coefficient to a scattering power law, the full-spectrum reduced scattering coefficients in the region from 650 to 1000 nm can be obtained.15 The absorption coefficient spectrum can subsequently be calculated for the same wavelength region using a model of light propagation, such as a photon diffusion model or a Monte Carlo model. A conventional DOS probe consisting of source and detection fibers is usually utilized along with a standard diffusion model to recover optical properties of tissue.12 DOS measurements performed using a source-detector separation less than 8mm generally do not produce consistently stable optical property results in tissue since the standard diffusion equation fails to describe photon transport accurately when the source and detector separation is only a few scattering lengths.16 In this study, we employed a superficial diffusing probe, as shown in Fig. 2, which is described in detail in the next section, along with a two-layer diffusion model to determine optical properties of in vivo human skin.
Fig. 2.
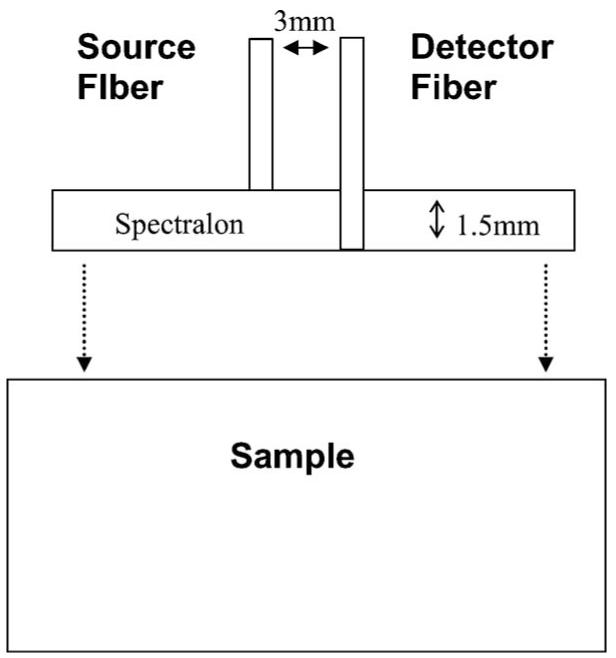
Geometry of a superficial diffusing probe.
2.3 Modified Two-Layer Geometry
The superficial diffusing probe with the modified two-layer geometry has been validated as a quantitative tool for accurately deducing optical properties in homogeneous turbid phantoms.10 In the phantom study, we demonstrated that the recovered optical properties have a maximal 8% deviation from benchmark values. The superficial diffusing probe contains a high-scattering, low-absorption layer with known optical properties that is placed on the surface of the sample under investigation. In practice, the superficial diffusing probe is composed of a Spectralon slab (LabSphere) and two optical fibers [3M Inc., numerical aperture (NA)=0.37, core diameter=600 μm], as illustrated in Fig. 3. The Spectralon slab has a thickness of 1.5 mm, an index of refraction of 1.35, and μa and of 10−5 /mm and 50/mm at a wavelength of 660 nm, respectively. Our studies thus far have shown that for measurements on skin, a Spectralon slab of 1.5 mm thickness provides us with adequate signal and is also thick and rigid enough to prevent a curved probe surface. The source fiber is mounted flush with the top surface of Spectralon and the detection fiber penetrates through the Spectralon and is flush with its lower surface. Source and detection fibers are attached to the Spectralon with epoxy and their separation is 3mm. In this geometry, all detected photons are constrained to travel through the Spectralon scattering layer before passing through the sample of interest and arriving at the detector. The measured reflectance from both frequency-domain and steady state subsystems is described by a modified two-layer diffusion model. The modified two-layer diffusion model is an extension of a general two-layer diffusion model proposed by Kienle et al.17 In the two-layer model, there are two boundaries: the air/layer-1 (high-scattering layer) boundary and the layer-l/layer-2 (sample layer) boundary. To apply the model to the superficial diffusing probe, the source is modeled as a point source pulse located beneath the air/layer-1 boundary at a distance of , and the detector is located at the layer- l/layer-2 boundary at a distance from the source. Extrapolated boundary conditions are applied and the fluence rate and z component of the flux of different layers are continuous at the layer-l/layer-2 boundary.
Fig. 3.
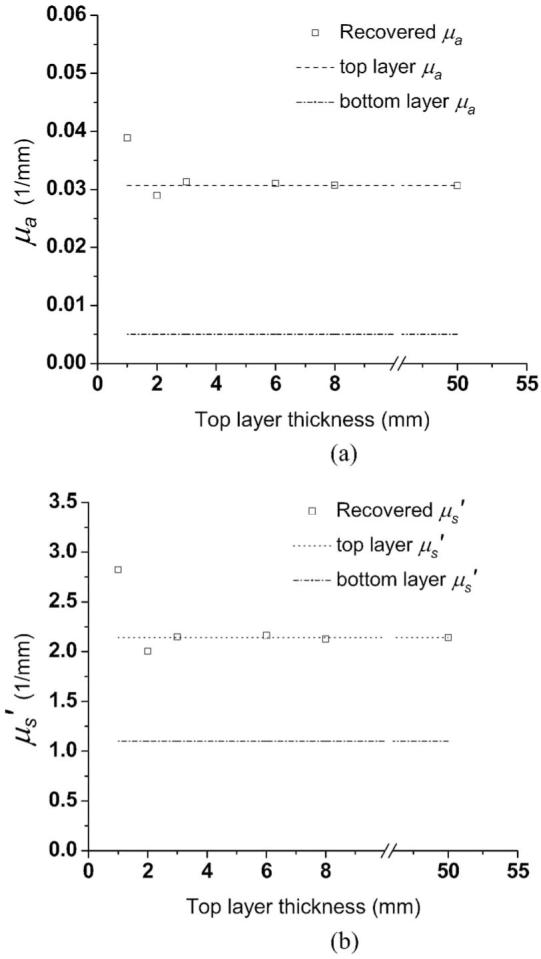
Optical properties (a) μa and (b) recovered from two-layer phantoms having top layer thicknesses from 1 to 8 mm. The dot-dotted and dash-dotted lines represent benchmark optical properties of the top layers and the substrate of two-layer phantoms, respectively.
For the purpose of modeling, the high-scattering layer is considered homogeneous and extends infinitely in lateral directions, and the sample is assumed homogeneous and semiinfinite in lateral extent in the two-layer diffusion-based model. The actual diameter of the Spectralon slab is 10 mm, and we have demonstrated that the modified two-layer diffusion model can be used to recover the accurate optical properties of semiinfinite homogeneous phantoms.10 However, because tissues are inhomogeneous, the next step in characterizing this method is to test the probe and model using two-layer phantoms. These phantoms have top layers of various thicknesses as described in the following. This enables us to methodically characterize the performance of the probe and its applicability for measuring in vivo skin.
3 Validation of the Applicability of the Superficial Diffusing Probe for Measuring In Vivo Skin Using Two-Layer Phantoms
To investigate the interrogation depth and the accuracy of recovered optical properties of our probe as it is applied to measure in vivo skin, we designed the optical properties of two-layer phantoms according to Simpson’s ex vivo measurement results.6 Five two-layer and two homogeneous silicone phantoms were made by mixing polydimethylsiloxane (Eager Plastics, Illinoes), catalyst, titanium dioxide (scatterer), and India ink (absorber) in disposable polystyrene beakers. The substrates of phantoms were fabricated first by mixing 440 mg of titanium dioxide, 500 μl of India ink, and 500 ml of polydimethylsiloxane. The substrates were designed to have optical properties similar to subcutaneous fat.17 Five “bulk” substrates having thickness larger than 50 mm were poured at the same time so as to have same optical properties. One set of “top layer” phantoms, having optical properties that differ from those of the substrate, were then prepared. We added 440 mg of titanium dioxide and 3ml of India ink into 500 ml polydimethylsiloxane to make the optical properties of the top layer phantoms similar to those of light skin epidermis-dermis. We designed the optical properties of the top layer phantoms to mimic those of light skin epidermis-dermis.17 Once the bulk substrate material cured, the top layers of different thicknesses were poured and allowed to cure. This resulted in five two-layer phantoms having different top layer thicknesses, ranging from about 1 to 8mm. The thicknesses of the top layers were well controlled since the polydimethylsiloxane was in liquid form before cured, and they were punctiliously measured with a digital caliper. A single bulk substrate was left without a top layer as a homogeneous semiinfinite control. In addition, a 50-mm-thick phantom of the top layer material was left alone to cure in the absence of an underlying substrate. The optical properties of bulk substrate and top layer material at a wavelength of 785 nm are listed in Table 1.
Table 1.
Optical properties of bulk substrate and bulk top layer materials at a wavelength of 785 nm.
| Substrate | Top Layer | |
|---|---|---|
| μa (1/mm) | 0.005 | 0.031 |
| μ’s (1/mm) | 1.10 | 2.14 |
Figure 3 illustrates the recovered μa and , values versus top layer thickness of the two-layer phantoms. Each symbol in the plot represents the average of three measurements, and deviations are all within 0.1%. Optical properties of samples having a 50-mm-thick top layer are actually results obtained from measurements performed on the homogeneous phantom. We consider the results from homogeneous phantoms (top layer and substrate) as the benchmark optical properties.
In Fig. 3 for top layer thicknesses larger than 2mm, the deviation of recovered μa and and values is within 5% of the benchmark values. As the top layer thickness decreases to 1 mm, the deviation increases to 28 and 32% relative to the benchmark values for μa, and , respectively. These results indicate that the maximum penetration depth of the majority of detected photons does not exceed 2 mm. Thus, the top layer is virtually semiinfinite and homogeneous to the diffusing probe when the top layer is thicker than 2 mm. We estimate that the average interrogation depth of our superficial diffusing probe will be less than 2 mm when it is applied to in vivo skin, given that these phantoms were designed to mimic the average properties of epidermis+dermis. In the study carried out by Simpson et al.,6 the thickness of the epidermis+dermis of the samples ranged from 1.5 to 2 mm.
As illustrated in Fig. 3, when the two probes are applied to the two-layer phantoms with 1-mm top layer thicknesses, the recovered optical property values do not fall between the benchmark optical properties of the top layer and the substrate optical properties. This is a consequence of the limitations of the diffusion model, which assumes the sample is homogenous and semiinfinite, whereas the sample is actually layered. This phenomenon was previously reported by Farrell et al.18 Steady state reflectance measurements by this group using a probe having source-detector separations ranging between 1 and 10 mm resulted in recovered optical properties for layered tissue samples that were not simply weighted averages of those of individual layers of tissues.
Our results19 support their observations when the top layer thickness of our phantoms is reduced to less than 2 mm, given a probe with source-detector separation of 3 mm. The modified two-layer diffusion model we employed for our diffusing probe assumes that samples are semiinfinite and homogeneous. Based on our measurement results, this assumption does not seem to be valid when the top layer of our phantoms is reduced in thickness to less than 2 mm. In the next section, we present in vivo measurement results obtained from volar forearm and palm skin. Based on these results, we conclude that the interrogation region of our probe is limited to the epidermis and dermis of the two measurement sites. For sites for which the epidermis+dermis thickness is within the range from 1 to 2 mm, such as the cheek skin of some subjects, the interrogation depth of the diffusing probe must be decreased to ensure that the interrogation region is in the epidermis and dermis. Otherwise, the recovered absorption coefficients will be dependent on the absorption properties of the epidermis, the dermis, and the subcutaneous layer. One possible way to further reduce the interrogation depth of the diffusing probe is to shorten the separation between source and detection fibers. The detailed characterization of the interrogation depth of the diffusing probe is discussed elsewhere.19
4 Results of In Vivo Skin Measurements
Measurements using the superficial diffusing probe were taken on the volar forearm and the palm of 15 subjects. The current in vivo measurements were approved by the Institutional Review Board (IRB) of the University of California, Irvine. Volar forearm skin usually appears to be more pigmented than the palm of the hand.20 Because melanin is localized in the epidermis, we expect to observe differences in absorption properties acquired from these two anatomical locations when the superficial diffusing probe is employed. Subjects were five subjects of African descent, five Asians, and five Caucasians. In the process of collecting data, the probe was gently placed against the skin at each measurement site. Three measurements were made at each site to assess both random and systematic errors. In every case, the probe was physically removed from the measurement site and replaced to perform measurement again. Variations in the recovered optical properties of different measurements at a same site are within 5%.
The optical properties of volar forearm skin of one subject of African descent, one Asian, and one Caucasian measured with the superficial diffusing probe are plotted in Fig. 4. Lines and discrete data points in Fig. 4 represent steady state and frequency-domain recovered optical properties, respectively. In Fig. 4(a), the absorption spectrum of the subject of African descent and those of the Asian and the Caucasian subjects have distinct values and trend in the wavelength range from 650 to 850 nm. The monotonic decrease of the subject of African descent’s absorption spectrum in the 650 to 850 nm range corresponds to melanin absorption.21 The absorption features of oxyhemoglobin and deoxyhemoglobin are weakly demonstrated in the Asian’s and the Caucasian’s absorption spectra, but they are not apparent in the subject of African descent’s absorption spectrum. This is likely due to the reduced interrogation depth of the diffusing probe when applied to dark skin relative to depths probed in light skin, and the fact that the absorption of melanin is much stronger than that of hemoglobin in the 650- to 850-nm window. The lipid absorption feature at 930 nm cannot be observed for all absorption spectra. We therefore estimate that the interrogation region of the diffusing probe is limited to epidermis and dermis when it is applied to volar forearm skin. Figure 4(b) illustrates the reduced scattering coefficient spectra obtained from the volar forearms of the three subjects. The reduced scattering coefficient reflects the structure and density of scatterers in the skin such as collagen fibrils, cells, and intracellular structures. Reduced scattering coefficients of tissue in the near IR range usually follow a power law , where λ is the wavelength, and a and b characterize scatter density and average size, respectively.22 Similar scatter size and different density is found in African descent and Asian data, while the Caucasian seems to have different size and density than other categories. The differences in the reduced scattering coefficient magnitude between subjects is likely a result of modulation of interrogation depths by the absorption properties of each skin type. Because the absorption in light skin is relatively small compared to that in dark skin, the diffusing probe will collect more photons that have passed through greater volumes of collagen and elastin in the dermis than when applied to dark skin. Thus, the recovered scattering coefficients of the Asian and the Caucasian are higher than that of the subject of African descent.
Fig. 4.
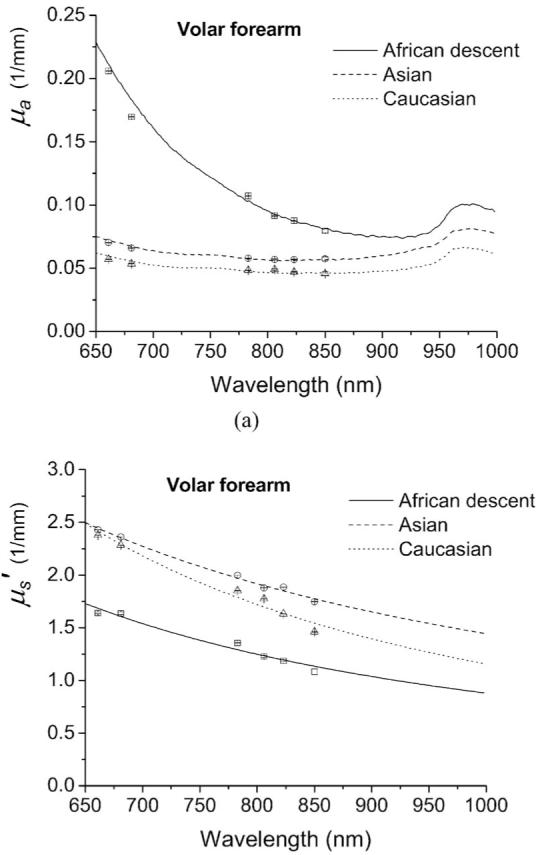
Optical properties (a) μa and (b) of in vivo volar forearm skin of one subject of African descent, an Asian, and a Caucasian measured with a superficial diffusing probe. Discrete data points are recovered from frequency-domain photon migration (FDPM) measurements and lines represent broadband optical property spectra of the three subjects.
Figure 5 demonstrates the absorption and scattering coefficients from the skin of five subjects of African descent, five Asians, and five Caucasians. The lines and error bars represent the average and standard deviation of the three ethnic groups. As shown in Fig. 5(a), the skin of each ethnic group has a dissimilar slope in the absorption coefficient in the 650-to 850-nm window, which can be attributed to the different melanin concentration contained in the epidermis. The superficial diffusing probe is sensitive to the differences in melanin concentration that exists in the epidermis, and the results demonstrate, for this particular set of measurements, the ratio of the average absorption coefficients of the subjects African descent’s skin and the Caucasian skin at the wavelength of 650 nm is about 5. In the 900- to 1000-nm window, the absorption coefficient demonstrates12 a water peak at 980 nm. The magnitude of absorption coefficient of the three groups at 980 nm is very similar (within 2%), indicating that water concentration of superficial skin of the three ethnic groups is similar, despite the color of the skin. The average scattering coefficient for the three groups is shown in Fig. 5(b). The data acquired from the group of African descent still exhibits the lowest scattering magnitude among the three groups, which is consistent with the trend observed in Fig. 4(b). The average optical properties of volar forearm obtained using this probe compare favorably to properties of ex-vivo epidermis+dermis determined by Simpson et al.6 and Bashkatov et al.7 using integrating sphere techniques.
Fig. 5.
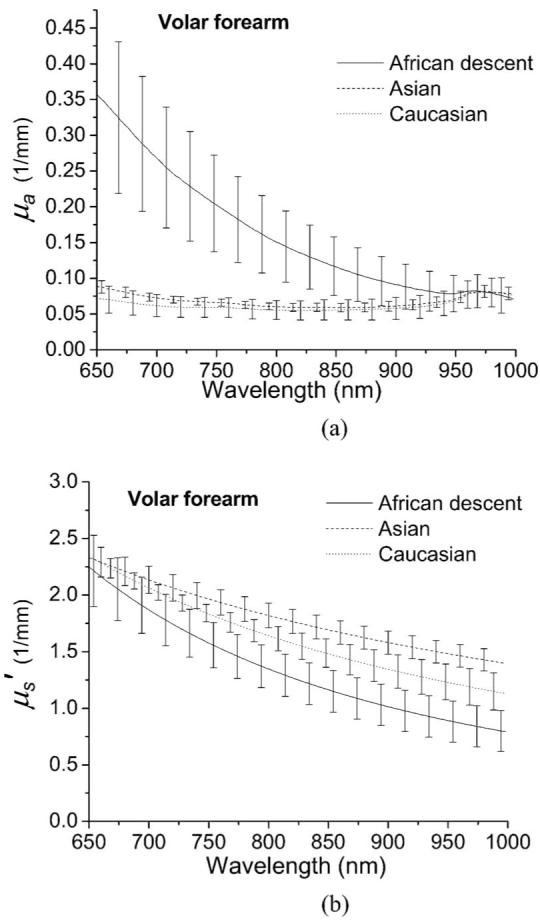
Broadband optical properties (a) μa and (b) of in vivo volar forearm skin of five subjects of African descent (solid line), five Asians (dashed line), and five Caucasians (dotted line) measured with a superficial diffusing probe. The error bars represent the standard deviation of optical properties in each group.
The average optical properties of the heel of the palm measured with a superficial diffusing probe are plotted in Fig. 6. Surprisingly, in Fig. 6(a), the average absorption coefficients of the Asian and the Caucasian groups are almost identical in the wavelength window from 650 to 1000 nm. Comparing Fig. 5(a) and Fig. 6(a), we observe that variation in absorption spectra between subjects is smaller in Fig. 6(a), which is likely a consequence of low melanin concentration in the palm of the hand regardless of skin color compared to volar forearm.20 Moreover, the deoxyhemoglobin absorption peak at 760 nm can be discerned for the three groups. The average reduced scattering coefficient shown in Fig. 6(b) for the three groups is similar in magnitude and slope, which means that the skin of the palm of the three ethnic groups have similar composition in terms of average scatterer size and density. Note that the absorption coefficients of palm skin of the three groups are more tightly clustered than for volar forearm skin. This suggests that the modulation in the interrogation depth by chromophores in the palm skin is smaller than that for the volar forearm skin. Hence, the recovered reduced scattering coefficient of palm skin does not vary as strongly between individuals relative to reduced scattering coefficient measured in volar forearm skin.
Fig. 6.
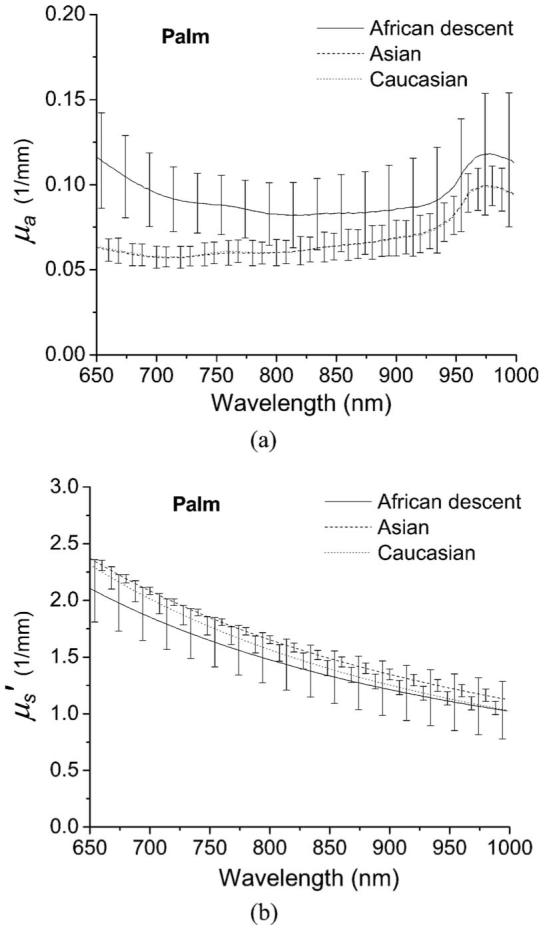
Broadband optical properties (a) μa and (b) of in vivo palm skin of five subjects of African descent (solid line), five Asians (dashed line), and five Caucasians (dotted line) measured with a superficial diffusing probe. The error bars represent the standard deviation of optical properties in each group.
5 Conclusion
we developed a new DOS probe and verified its applicability to determine the optical properties of in vivo human skin consisting of epidermis+dermis using two-layer phantoms. In the phantom study, the interrogation depth of our superficial diffusing probe having a 3-mm source-detector separation was estimated to be less than 2 mm. We applied the probe to measure optical properties of in vivo skin of volar forearm and palm for 15 subjects, including five subjects of African descent, five Asians, and five Caucasians. Our measurement results agree favorably with those obtained from ex vivo techniques.6,7 The absorption spectra of the volar forearm depicted in Fig. 4(a) imply that the probe is sensitive to the melanin that exists in the epidermis. The absorption spectra of the palm determined using our probe show less difference between light- and dark-skinned subjects than the absorption spectra measured on the volar forearm. This is likely a consequence of low melanin concentration in the palm.20 The magnitudes of the reduced scattering coefficient obtained for volar forearm skin differ considerably across the three groups and may be a consequence of the modulation of interrogation depth of the superficial diffusing probe induced by the skin absorption properties. The reduced scattering coefficients of the palm of the three skin color groups are very similar, which suggests that the interrogation regions, the average scatterer size, and the average scatterer, density of the palm skin of all 15 subjects are similar.
The diffusing probe geometry provides a means for rapidly determining superficial in vivo skin optical properties over the 650- to 1000-nm range. Such an advance has the potential to impact light-based diagnostics and therapy. We have begun to work toward the application of this technology to clinical studies such as skin cancer, oral cancer, and port wine stain therapy. We are also improving our instrument so that the absorption and scattering properties of tissue at wavelengths shorter than 650 nm can be acquired, with the hope that access to more prominent hemoglobin spectral features will enable us to accurately quantify melanin and oxy- and deoxyhemoglobin concentrations.
Acknowledgments
This work was supported by the National Institutes of Health/National Center for Research Resources (NCRR) under Grant No. P41-RR01192 (Laser Microbeam and Medical Program: LAMMP), the U.S. Air Force Office of Scientific Research, the Medical Free-Electron Laser Program (F49620-00-2-0371 and FA9550-04-1-0101), and the Beckman Foundation.
References
- 1.Kelly KM, Choi B, McFarlane S, Motosue A, Jung BJ, Khan MH, Ramirez-San-Juan JC, Nelson JS. Description and analysis of treatments for port-wine stain birthmarks. Arch. Fac. Plas. Surg. 2005;7(5):287–294. doi: 10.1001/archfaci.7.5.287. [DOI] [PubMed] [Google Scholar]
- 2.Nishidate I, Aizu Y, Mishina H. Estimation of melanin and hemoglobin in skin tissue using multiple regression analysis aided by Monte Carlo simulation. J. Biomed. Opt. 2004;9(4):700–710. doi: 10.1117/1.1756918. [DOI] [PubMed] [Google Scholar]
- 3.Arimoto H, Egawa M, Yamada Y. Depth profile of diffuse reflectance near-infrared spectroscopy for measurement of water content in skin. Skin Res. Technol. 2005;11(1):27–35. doi: 10.1111/j.1600-0846.2005.00093.x. [DOI] [PubMed] [Google Scholar]
- 4.Kienle A, Glanzmann T. In vivo determination of the optical properties of muscle with time-resolved reflectance using a layered model. Phys. Med. Biol. 1999;44(11):2689–2702. doi: 10.1088/0031-9155/44/11/301. [DOI] [PubMed] [Google Scholar]
- 5.Troy TL, Thennadil SN. Optical properties of human skin in the near infrared wavelength range of 1000 to 2200 nm. J. Biomed. Opt. 2001;6(2):167–176. doi: 10.1117/1.1344191. [DOI] [PubMed] [Google Scholar]
- 6.Simpson CR, Kohl M, Essenpreis M, Cope M. Near-infrared optical properties of ex vivo human skin and subcutaneous tissues measured using the Monte Carlo inversion technique. Phys. Med. Biol. 1998;43(9):2465–2478. doi: 10.1088/0031-9155/43/9/003. [DOI] [PubMed] [Google Scholar]
- 7.Bashkatov AN, Genina EA, Kochubey VI, Tuchin VV. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D. 2005;38(15):2543–2555. [Google Scholar]
- 8.Zhang R, Verkruysse W, Choi B, Viator JA, Jung R, Svaasand LO, Aguilar G, Nelson JS. Determination of human skin optical properties from spectrophotometric measurements based on optimization by genetic algorithms. J. Biomed. Opt. 2005;10(2):024030. doi: 10.1117/1.1891147. [DOI] [PubMed] [Google Scholar]
- 9.Amelink A, Sterenborg HJCM, Bard MPL, Burgers SA. In vivo measurement of the local optical properties of tissue by use of differential path-length spectroscopy. Opt. Lett. 2004;29(10):1087–1089. doi: 10.1364/ol.29.001087. [DOI] [PubMed] [Google Scholar]
- 10.Tseng SH, Hayakawa C, Tromberg BJ, Spanier J, Durkin AJ. Quantitative spectroscopy of superficial turbid media. Opt. Lett. 2005;30(23):3165–3167. doi: 10.1364/ol.30.003165. [DOI] [PubMed] [Google Scholar]
- 11.Pham TH, Coquoz O, Fishkin JB, Anderson E, Tromberg BJ. Broad bandwidth frequency domain instrument for quantitative tissue optical spectroscopy. Rev. Sci. Instrum. 2000;71(6):2500–2513. [Google Scholar]
- 12.Bevilacqua F, Berger AJ, Cerussi AE, Jakubowski D, Tromberg BJ. Broadband absorption spectroscopy in turbid media by combined frequency-domain and steady-state methods. Appl. Opt. 2000;39(34):6498–6507. doi: 10.1364/ao.39.006498. [DOI] [PubMed] [Google Scholar]
- 13.Cerussi AE, Jakubowski D, Shah N, Bevilacqua F, Lanning R, Berger AJ, Hsiang D, Butler J, Holcombe RF, Tromberg BJ. Spectroscopy enhances the information content of optical mammography. J. Biomed. Opt. 2002;7(1):60–71. doi: 10.1117/1.1427050. [DOI] [PubMed] [Google Scholar]
- 14.Shah N, Cerussi AE, Jakubowski D, Hsiang D, Butler J, Tromberg BJ. Spatial variations in optical and physiological properties of healthy breast tissue. J. Biomed. Opt. 2004;9(3):534–540. doi: 10.1117/1.1695560. [DOI] [PubMed] [Google Scholar]
- 15.Schmitt JM, Kumar G. Optical scattering properties of soft tissue: a discrete particle model. Appl. Opt. 1998;37(13):2788–2797. doi: 10.1364/ao.37.002788. [DOI] [PubMed] [Google Scholar]
- 16.Haskell RC, Svaasand LO, Tsay TT, Feng TC, Mcadams MS. Boundary-Conditions for the diffusion equation in radiative-transfer. J. Opt. Soc. Am. A. 1994;11(10):2727–2741. doi: 10.1364/josaa.11.002727. [DOI] [PubMed] [Google Scholar]
- 17.Kienle A, Patterson MS, Dognitz N, Bays R, Wagnieres G, van den Bergh H. Noninvasive determination of the optical properties of two-layered turbid media. Appl. Opt. 1998;37(4):779–791. doi: 10.1364/ao.37.000779. [DOI] [PubMed] [Google Scholar]
- 18.Farrell TJ, Patterson MS, Essenpreis M. Influence of layered tissue architecture on estimates of tissue optical properties obtained from spatially resolved diffuse reflectometry. Appl. Opt. 1998;37(10):1958–1972. doi: 10.1364/ao.37.001958. [DOI] [PubMed] [Google Scholar]
- 19.Tseng SH, Hayakawa C, Spanier J, Durkin AJ. Determination of optical properties of superficial volumes of layered tissue phantoms. IEEE Trans. Biomed. Eng. 1998;55(1):335–339. doi: 10.1109/TBME.2007.910685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yamaguchi Y, Hearing VJ, Itami S, Yoshikawa K, Katayama I. Mesenchymal-epithelial interactions in the skin: aiming for site-specific tissue regeneration. J. Dermatol. Sci. 2005;40(1):1–9. doi: 10.1016/j.jdermsci.2005.04.006. [DOI] [PubMed] [Google Scholar]
- 21.Jacques SL, Mcauliffe DJ. The melanosome-threshold temperature for explosive vaporization and internal absorption-coefficient during pulsed laser irradiation. Photochem. Photobiol. 1991;53(6):769–775. doi: 10.1111/j.1751-1097.1991.tb09891.x. [DOI] [PubMed] [Google Scholar]
- 22.Mourant JR, Fuselier T, Boyer J, Johnson TM, Bigio IJ. Predictions and measurements of scattering and absorption over broad wavelength ranges in tissue phantoms. Appl. Opt. 1997;36(4):949–957. doi: 10.1364/ao.36.000949. [DOI] [PubMed] [Google Scholar]


