Abstract
Background
There are reports of rigors and/or clinical deterioration following recruitment maneuvers (RMs), leading us to question whether the use of sustained high-pressure inflation could lead to release of inflammatory mediators.
Methods
Prospective cohort study of 26 patients with ARDS receiving mechanical ventilation. A single RM was performed during which the mean airway pressure was increased to 40 cm H2O and held constant for a period of 30 s. The concentration of nine cytokines (interleukin [IL]-1, IL-6, IL-8, IL-10, tumor necrosis factor [TNF]-α, Fas ligand, vascular endothelial growth factor, TNF receptor 1, TNF receptor 2) was measured longitudinally at three time points: prior to initiation of the RM, 5 min after the RM, and 60 min after the RM.
Results
RMs were tolerated well from a hemodynamic perspective. Oxygenation improved as reflected by an increased PaO2/fraction of inspired oxygen (FIO2) ratio from 140 ± 49 at baseline to 190 ± 78 (mean ± SD) at 5 min after the RM (p = 0.01). At 60 min, the increase in PaO2/FIO2 ratio, to 172 ± 76, was no longer significant (p = 0.1). There were no important changes in the levels of any of the measured cytokines at 5 min or 60 min following RM as compared with the baseline levels.
Conclusions
The results of our study demonstrate that recruitment maneuvers are well tolerated in patients with ARDS. Our data suggest no major hemodynamic or immunologic evidence of deterioration within the first hour of RM. In particular, cytokines, previously related to worsening lung injury and distal organ failure in patients with ARDS, are not elevated by use of an RM.
Keywords: ARDS, cytokines, lung injury, recruitment maneuver, ventilator
ARDS is a devastating condition with high morbidity and mortality rates. 1 There is controversy regarding how best to provide mechanical ventilation to patients with this syndrome. Steady progress has been made regarding the use of mechanical ventilation based on animal and human research. These studies have clearly established a number of findings. First, the ventilator, if misapplied, can lead to damage to the lung and potentially perpetuate underlying lung injury, a process now referred to as ventilator-induced lung injury.2,3 Second, the minimization of transpulmonary pressure at end inflation can lead to improvements in outcome as compared with higher pressures that can lead to overdistension and further injury.4–6 Third, cytokines can be released from the lung and contribute to hypercytokinemia. These cytokines have been implicated in multiple-system organ failure.7–9 Some authors10–12 have suggested that the use of protective mechanical ventilation strategies can reduce the levels of circulating cyto-kines, which in theory could reduce the development of distal organ failure. Thus, the existing data suggest that mechanical ventilation is important in acute lung injury, and that techniques to limit damage induced by the ventilator can improve outcome, perhaps by limiting systemic inflammation.13
One area of controversy is that of the recruitment maneuver (RM).14 The RM is defined as a sustained high-pressure inflation maneuver aiming to overcome the opening pressure of lung units with high surface tension. Such maneuvers may be helpful in improving gas exchange, at least transiently, and in promoting homogeneity within the lung.15 The combined approach of using RMs to open the lung and then applying high levels of positive end-expiratory pressure (PEEP) to maintain alveolar patency may be the optimal method to promote lung homogeneity.16 Prior studies16 have suggested that heterogeneous areas within the lung are sites where marked shear forces can develop and may propagate further lung injury. Thus, promoting lung homogeneity to minimize ventilator-induced lung injury has theoretical appeal. However, RMs have thus far been minimally studied from standpoint of efficacy as well as safety. We were aware of some reports of rigors and/or clinical deterioration occurring following RMs, leading us to question whether the use of sustained high-pressure inflation could have deleterious consequences on indexes of systemic inflammation. One recent report17 suggested minimal cy-tokine release following RMs in a small sample of patients receiving mechanical ventilation; however, the authors used a 7-s RM, which tells us little about the response to sustained high-pressure inflation, which is more typically used in this context. Because there are potentially both short-term and longer-term effects of RMs, we focused on a narrow time window (1 h) to assess the release of preformed cytokines, rather than longer-term changes that may reflect changes in underlying lung biology.
The objective of this study was to define the hemodynamic and immunologic effects of sustained high-pressure inflation maneuvers to promote alveolar recruitment. Our study design allows us to test the hypothesis that RMs can be safely performed without major deleterious short-term effects on BP and systemic cytokine levels.
Materials and Methods
Research Protocol
Institutional review board approval was obtained for this study. Twenty-six patients with a diagnosis of ARDS by American-European Consensus Conference criteria were enrolled in the study.18 All patients were within 24 h of the onset of ARDS criteria. A baseline blood sample was drawn from each patient. Patients then underwent an RM during which mean airway pressure was increased to 40 cm H2O and held constant for a period of 30 s. Patients were then returned to ventilator settings consistent with the ARDSnet trial.4 Blood was drawn again at 5 min and 60 min after recruitment. All blood samples were centrifuged and stored at – 20°C until laboratory analysis.
Laboratory Analysis
In each subject, the concentrations of nine cytokines (interleu-kin [IL]-1, IL-6, IL-8, IL-10, tumor necrosis factor [TNF]-α, Fas ligand, vascular endothelial growth factor, TNF receptor 1, and TNF receptor 2) were measured longitudinally at three time points: immediately prior to initiation of the RM, 5 min after the RM, and 60 min after the RM. Levels of cytokines in serum samples were measured using proteomic arrays (SearchLight; Pierce Biotechnology; Woburn, MA). This proteome array is a quantitative, multiplexed, sandwich enzyme-linked immunosor-bent assay. Each antibody captures specific cytokines/chemo-kines present in the standards and samples added to the plate. The bound proteins are then detected with a biotinylated detection antibody, followed by the addition of streptavidin-horseradish per-oxidase, and lastly chemiluminescent substrate (SuperSignal ELISA Femto Chemiluminescent Substrate; Pierce Biotechnol-ogy). The luminescent signal produced from the streptavidin-horseradish peroxidase-catalyzed oxidation of the substrate is measured by imaging the plate using a cooled charge-coupled device camera. The data are then analyzed using customized software. The amount of luminescent signal produced is proportional to the amount of each protein present in the original standard or sample. Concentrations are extrapolated off a standard curve.
Statistical Analysis
Summary statistics are reported as means with their SDs. Two-way t tests were performed to assess the significance of changes in continuous data such as the hemodynamic and oxygenation responses to RM. The distributions of cytokine concentrations at each time point were positively skewed. In order to render them normally distributed, a natural log (x + 1) transformation was applied to all concentrations. This resulted in mean log (concentration + 1) values being similar to median log (concentration + 1) values at each time point. At each time point after baseline, each subject’s log (concentration + 1) value at baseline was subtracted from the later log (concentration + 1) value. For brevity, we will refer to them as differences from baseline at each time point. Analysis was then performed using analysis of variance for repeated measures by examining the time trend.
Results
Patient Characteristics
Twenty-six patients with either acute lung injury or ARDS admitted to the surgical and medical ICUs were enrolled between June 2004 and June 2005. The characteristics of the individual patients enrolled in the study are summarized in Table 1, and the summary of these data appears in Table 2. Mean APACHE (acute physiology and chronic health evaluation) II score of the 26 patients was high, 27 ± 7 (mean ± SD). The 28-day mortality rate was 42%, consistent with the severity of illness. On enrollment in the study, the average PaO2/fraction of inspired oxygen (FIO2) ratio was 142 ± 50 and the oxygen index was 16 ± 9, indicating significant hypoxemia.
Table 1.
Individual Patient Characteristics
| Patient No. |
Gender | Age, yr |
Primary Diagnosis |
APACHEII Score |
28-d Mortality |
Ventilator Days, No. |
PEEP, cm H2O | Mean Airway Pressure, mm Hg |
Inspiratory Plateau Pressure, mm Hg |
Tidal Volume, mL |
PaO2/FIO2 Ratio | Oxygenation Index |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Female | 40 | Aspiration pneumonitis | 19 | No | 8 | 10 | 16 | 26 | 550 | 135 | 12 |
| 2 | Male | 20 | Aspiration pneumonitis | 29 | No | 3 | 8 | 13 | 20 | 650 | 109 | 12 |
| 3 | Female | 23 | Acetaminophen overdose | 39 | Yes | 6 | 15 | 23 | 29 | 325 | 74 | 31 |
| 4 | Male | 31 | Crohn disease | 33 | Yes | 2 | 30 | 40 | 50 | 450 | 89 | 45 |
| 5 | Male | 37 | Multiple trauma | 36 | No | 14 | 5 | 9 | 13 | 450 | 130 | 7 |
| 6 | Female | 72 | Pneumonia | 29 | Yes | 15 | 5 | 17 | 30 | 440 | 186 | 9 |
| 7 | Male | 30 | Sepsis | 33 | Yes | 17 | 15 | 22 | 36 | 686 | 166 | 13 |
| 8 | Male | 79 | Perforated viscous | 30 | Yes | 31 | 15 | 22 | 36 | 679 | 177 | 12 |
| 9 | Male | 71 | Sepsis | 28 | No | 31 | 5 | 16 | 26 | 650 | 125 | 13 |
| 10 | Male | 37 | Multiple trauma | 19 | No | 2 | 12 | 18 | 25 | 600 | 156 | 12 |
| 11 | Female | 74 | Large-bowel obstruction | 29 | No | 21 | 10 | 14 | 26 | 368 | 236 | 6 |
| 12 | Male | 67 | Pneumonia | 27 | No | 7 | 13 | 18 | 24 | 400 | 286 | 6 |
| 13 | Male | 51 | Intraventricular hemorrhage | 29 | Yes | 14 | 10 | 20 | 30 | 700 | 87 | 23 |
| 14 | Female | 41 | End-stage liver disease | 36 | Yes | 10 | 16 | 25 | 33 | 500 | 141 | 18 |
| 15 | Female | 90 | Colectomy | 23 | No | 7 | 12 | 15 | 23 | 400 | 97 | 16 |
| 16 | Male | 34 | Traumatic brain injury | 25 | No | 25 | 15 | 22 | 26 | 600 | 143 | 15 |
| 17 | Male | 41 | Multiple trauma | 10 | No | 11 | 15 | 22 | 28 | 550 | 131 | 17 |
| 18 | Female | 38 | Cholecystitis | 23 | No | 8 | 10 | 16 | 26 | 450 | 144 | 11 |
| 19 | Male | 60 | Smoke inhalation | 24 | No | 16 | 12 | 19 | 28 | 500 | 206 | 9 |
| 20 | Male | 79 | Pneumonia | 33 | Yes | 21 | 5 | 12 | 21 | 560 | 190 | 6 |
| 21 | Male | 20 | Multiple trauma | 11 | No | 17 | 10 | 16 | 24 | 500 | 128 | 12 |
| 22 | Female | 60 | Multiple trauma | 14 | No | 20 | 14 | 20 | 32 | 400 | 63 | 32 |
| 23 | Male | 78 | Perforated colon | 32 | Yes | 6 | 10 | 20 | 29 | 500 | 147 | 14 |
| 24 | Female | 58 | Pneumococcal pneumonia | 26 | Yes | 9 | 20 | 28 | 39 | 350 | 125 | 22 |
| 25 | Female | 68 | Small-bowel perforation | 24 | No | 17 | 14 | 18 | 30 | 450 | 107 | 17 |
| 26 | Female | 83 | Pneumonia | 31 | Yes | 15 | 14 | 22 | 42 | 350 | 118 | 19 |
Table 2.
Average Patient Characteristics*
| Characteristics | Data |
|---|---|
| Age, yr | 53 ± 22 |
| Male gender, No. (%) | 15 (58) |
| APACHE II score | 27 ± 7 |
| PaO2/FIO2 ratio | 142 ± 50 |
| Oxygenation index | 16 ± 9 |
| Ventilator days, No. | 14 ± 8 |
| 28-d mortality, % | 42 |
Data are presented as mean ± SD unless otherwise indicated.
Clinical Response
Clinical responses to the RMs are summarized in Table 3. The RMs were tolerated well from a hemodynamic perspective, and only two patients required early termination of the RM because of a decrease in systolic BP to < 80 mm Hg. Both of these patients had immediate return of BP to baseline and no significant effects at 5 min or 60 min. All other patients with less severe decreases in BP had a return of baseline heart rate and BP by the 5-min time point. PEEP was significantly elevated (12 ± 5 cm H2O vs 14 ± 5 cm H2O) at 5 min (p = 0.04) and 60 min (p = 0.03) after performance of the RM. This elevation reflected our clinical practice of increased PEEP after an RM to retain recruited lung. Oxygenation improved as reflected by an increase in the PaO2/FIO2 ratio from 140 ± 49 at baseline to 190 ± 78 at 5 min after the RM (p = 0.01). At 60 min after the RM, there was still an improvement in the PaO2/FIO2 ratio to 172 ± 76, although this was no longer significant (p = 0.1). While the oxygenation index improved at both 5 min after the RM (12 ± 4) and 60 min after the RM (14 ± 8) compared to before recruitment (16 ± 9), this improvement was not statistically significant. Compliance of the respiratory system was not improved at either 5 min or 60 min after the RM. There was however a significant increase in the PaCO2 from 41 ± 8 mm Hg at baseline to 45 ± 11 mm Hg (p = 0.02) at 5 min after the RM. This difference had disappeared at 60 min after the RM.
Table 3.
Hemodynamic and Ventilatory Variables
| Variables | Baseline | 5 min After RM | p Value* | 60 min After RM | p Value† |
|---|---|---|---|---|---|
| Heart rate, beats/min | 97 ± 22 | 95 ± 23 | 0.846 | 92 ± 22 | 0.211 |
| Mean arterial pressure, mm Hg | 74 ± 13 | 77 ± 16 | 0.375 | 76 ± 13 | 0.838 |
| PaCO2, mm Hg | 41 ± 8 | 45 ± 11 | 0.021 | 42 ± 10 | 0.462 |
| PEEP, cm H2O | 12 ± 5 | 14 ± 5 | 0.040 | 14 ± 5 | 0.030 |
| Compliance of respiratory system, mL/cm H2O | 33 ± 11 | 31 ± 9 | 0.262 | 35 ± 11 | 0.081 |
| PaO2/FIO2 | 142 ± 49 | 190 ± 78 | 0.011 | 172 ± 76 | 0.109 |
| Oxygenation index | 16 ± 9 | 12 ± 4 | 0.102 | 14 ± 8 | 0.708 |
Level of significance for comparison of baseline vs 5 min after RM using matched-pair t test.
Level of significance for comparison of baseline vs 60 min after RM using matched-pair t test.
Cytokine Response
As can be seen in the Figure 1, there were no important changes in the levels of any of the measured cytokines at 5 min or 60 min following the RM as compared with the baseline levels.
Figure 1.
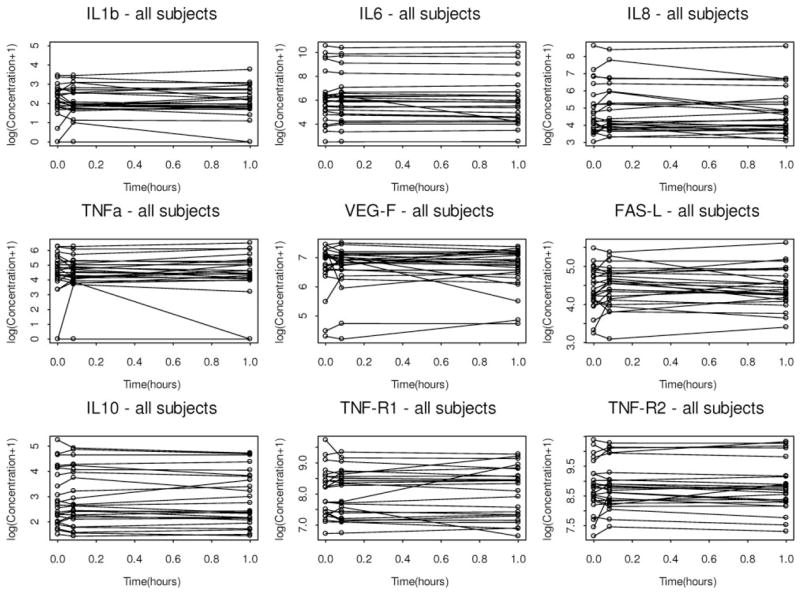
Serum cytokine levels in 26 subjects at baseline and at 5 min and 60 min after RMs. Cytokine levels are presented as log concentrations. FAS-l = Fas ligand; VEGF = vascular endothelial growth factor; TNF-R1 = TNF receptor 1; TNF-R2 = TNF receptor 2.
Discussion
The results of our study demonstrate that RMs are well tolerated in patients with ARDS, at least in the short term. Our data suggest no major hemodynamic or immunologic evidence of deterioration within the first hour of the RM. In particular, cytokines that have been previously related to worsening lung injury and distal organ failure in patients with ARDS are not elevated by use of an RM. We conclude that mechanical ventilator strategies using this technique are safe, and encourage further study in this area.
Although there are a number of reports of potential deterioration in clinical status following RMs in patients with ARDS, there are few systematic studies.17 Our patients tolerated the RM well from a hemodynamic perspective, and no patients required increased use of pressors or fluid boluses immediately following the RM. We would speculate that in patients who are underresuscitated, performance of an RM might lead to more significant hypotention. Our usual practice includes the liberal administration of fluids, as with early goal-directed therapy.19 However, in light of the more recent results from the Fluid and Catheter Treatment Trial,20 suggesting potential benefit to a “dry” strategy, we would be concerned that RMs may not be as well tolerated. Thus, further study is clearly required to determine the safety of recruitment from a hemodynamic standpoint.
We had been concerned, based on anecdotes of rigors following RMs, that RMs may have important deleterious consequences in promoting a systemic inflammatory response. While we observed no change in serum cytokines levels at 5 min and 1 h after the RM, the release of preformed cytokines in the lung by sustained high pressure should have been detectable at 5 min, and synthesis of new cytokines in response to the RM should have led to elevation of the plasma cytokine levels by 1 h. We are aware of reports12,21 of translocation of cytokines and bacteria from the lung into the systemic circulation as manifestations of various forms of lung injury. We would speculate that physical injury to the alveolar capillary membrane in the form of stress failure could in theory allow this migration.22,23 Thus, we were reassured by the lack of elevation in cytokine levels following the RM. However, we cannot rule out the possibility that the RM induces lung stretch that leads to an inflammatory response that may occur later. We did not measure cytokines at > 1 h after recruitment, but later changes in cytokine levels could be due to the progression of the patient’s underlying disease. The existing literature in this area is quite sparse, with only one recent report17 to our knowledge assessing the inflammatory response to a 7-s RM in 16 subjects.
Based on the existing literature, we believe that heterogeneity has a major effect on the regional shear forces that develop within the lung during mechanical ventilation.16 We would therefore advocate further study of techniques that use an RM to open the collapsed lung followed by the use of high PEEP to maintain opening of lung units with high surface tension (“keep it open, leave it open”). Our data suggest that the longer-term benefits of the open-lung approach are unlikely to be offset by any short-term deterioration from an RM.
Our study has a number of limitations. First, there appear to be some patients who demonstrate robust increases in certain cytokines (Fig 1) at 5 min that recover by 60 min. Because of our modest sample size, we are unable to determine whether these “outliers” reflect true biological variability or simply variance introduced by measurement error or methodologic issues. Although we acknowledge that statistical power is an issue given that we measured nine different cytokines, we would argue that the within-subjects design of our study makes it unlikely that we missed important hypercytokinemia. Although these individuals did not show clinical deterioration, we would encourage further research on the potential biological variability, possibly on a genetic basis. Second, although our data are reassuring regarding the safety of RMs, we can say nothing about their effects on patient outcome. Our data were collected in the context of an ongoing clinical trial, and therefore we are unable to assess the isolated impact of RMs on patient outcome. Finally, we show improvement in oxygenation following RMs, but we cannot in the present study determine the impact of RMs on regional aeration.
Conclusions
RMs appear to be well tolerated in patients with ARDS. We observed no evidence of important deterioration from either hemodynamic or immunologic perspectives. The clinical efficacy of RMs either on their own or as part of an integrated ventilator-management strategy remains to be proven.
Acknowledgments
This work was supported in part by grant HL-52586 from the National Heart, Lung, and Blood Institute.
Abbreviations
- APACHE
acute physiology and chronic health evaluation
- FIO2
fraction of inspired oxygen
- IL
interleukin
- PEEP
positive end-expiratory pressure
- RM
recruitment maneuver
- TNF
tumor necrosis factor
Footnotes
The authors have no conflicts of interest to disclose.
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (www.chestjournal.org/misc/reprints.shtml).
References
- 1.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000;342:1334–1349. doi: 10.1056/NEJM200005043421806. [DOI] [PubMed] [Google Scholar]
- 2.Matthay MA, Bhattacharya S, Gaver D, et al. Ventilator-induced lung injury: in vivo and in vitro mechanisms. Am J Physiol Lung Cell Mol Physiol. 2002;283:L678–L682. doi: 10.1152/ajplung.00154.2002. [DOI] [PubMed] [Google Scholar]
- 3.Slutsky AS. Lung injury caused by mechanical ventilation. Chest. 1999;116(suppl):9S–15S. doi: 10.1378/chest.116.suppl_1.9s-a. [DOI] [PubMed] [Google Scholar]
- 4.the Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 5.de Chazal I, Hubmayr RD. Novel aspects of pulmonary mechanics in intensive care. Br J Anaesth. 2003;91:81–91. doi: 10.1093/bja/aeg146. [DOI] [PubMed] [Google Scholar]
- 6.Talmor D, Sarge T, O’Donnell CR, et al. Esophageal and transpulmonary pressures in acute respiratory failure. Crit Care Med. 2006;34:1389–1394. doi: 10.1097/01.CCM.0000215515.49001.A2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Parsons PE, Matthay MA, Ware LB, et al. Elevated plasma levels of soluble TNF receptors are associated with morbidity and mortality in patients with acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2005;288:L426–L431. doi: 10.1152/ajplung.00302.2004. [DOI] [PubMed] [Google Scholar]
- 8.Ranieri VM, Giunta F, Suter PM, et al. Mechanical ventilation as a mediator of multisystem organ failure in acute respiratory distress syndrome. JAMA. 2000;284:43–44. doi: 10.1001/jama.284.1.43. [DOI] [PubMed] [Google Scholar]
- 9.Ranieri VM, Suter PM, Tortorella C, et al. Effect of mechanical ventilation on inflammatory mediators in patients with acute respiratory distress syndrome: a randomized controlled trial. JAMA. 1999;282:54–61. doi: 10.1001/jama.282.1.54. [DOI] [PubMed] [Google Scholar]
- 10.Tremblay L, Valenza F, Ribeiro SP, et al. Injurious ventilatory strategies increase cytokines and c-fos m-RNA expression in an isolated rat lung model. J Clin Invest. 1997;99:944–952. doi: 10.1172/JCI119259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Imai Y, Parodo J, Kajikawa O, et al. Injurious mechanical ventilation and end-organ epithelial cell apoptosis and organ dysfunction in an experimental model of acute respiratory distress syndrome. JAMA. 2003;289:2104–2112. doi: 10.1001/jama.289.16.2104. [DOI] [PubMed] [Google Scholar]
- 12.Nahum A, Hoyt J, Schmitz L, et al. Effect of mechanical ventilation strategy on dissemination of intratracheally instilled Escherichia coli in dogs. Crit Care Med. 1997;25:1733–1743. doi: 10.1097/00003246-199710000-00026. [DOI] [PubMed] [Google Scholar]
- 13.Fan E, Needham DM, Stewart TE. Ventilatory management of acute lung injury and acute respiratory distress syndrome. JAMA. 2005;294:2889–2896. doi: 10.1001/jama.294.22.2889. [DOI] [PubMed] [Google Scholar]
- 14.Medoff BD, Harris RS, Kesselman H, et al. Use of recruitment maneuvers and high-positive end-expiratory pressure in a patient with acute respiratory distress syndrome. Crit Care Med. 2000;28:1210–1216. doi: 10.1097/00003246-200004000-00051. [DOI] [PubMed] [Google Scholar]
- 15.Brower RG, Morris A, MacIntyre N, et al. Effects of recruitment maneuvers in patients with acute lung injury and acute respiratory distress syndrome ventilated with high positive end-expiratory pressure. Crit Care Med. 2003;31:2592–2597. doi: 10.1097/01.CCM.0000090001.91640.45. [DOI] [PubMed] [Google Scholar]
- 16.Mead J, Takishima T, Leith D. Stress distribution in lungs: a model of pulmonary elasticity. J Appl Physiol. 1970;28:596–608. doi: 10.1152/jappl.1970.28.5.596. [DOI] [PubMed] [Google Scholar]
- 17.Puls A, Pollok-Kopp B, Wrigge H, et al. Effects of a single-lung recruitment maneuver on the systemic release of inflammatory mediators. Intensive Care Med. 2006;32:1080–1085. doi: 10.1007/s00134-006-0174-x. [DOI] [PubMed] [Google Scholar]
- 18.Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS: definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–824. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 19.Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–1377. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 20.Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354:2564–2575. doi: 10.1056/NEJMoa062200. [DOI] [PubMed] [Google Scholar]
- 21.Kurahashi K, Kajikawa O, Sawa T, et al. Pathogenesis of septic shock in Pseudomonas aeruginosa pneumonia. J Clin Invest. 1999;104:743–750. doi: 10.1172/JCI7124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.West JB. Thoughts on the pulmonary blood-gas barrier. Am J Physiol Lung Cell Mol Physiol. 2003;285:L501–L513. doi: 10.1152/ajplung.00117.2003. [DOI] [PubMed] [Google Scholar]
- 23.Schwartz DR, Maroo A, Malhotra A, et al. Negative pressure pulmonary hemorrhage. Chest. 1999;115:1194–1197. doi: 10.1378/chest.115.4.1194. [DOI] [PubMed] [Google Scholar]


