Abstract
Gradients of extracellular signaling molecules and transcription factors are used in a variety of developmental processes, including the patterning of the Drosophila embryo, the establishment of diverse neuronal cell types in the vertebrate neural tube, and the anterior–posterior patterning of vertebrate limbs. Here, we discuss how a gradient of the maternal transcription factor Dorsal produces complex patterns of gene expression across the dorsal–ventral (DV) axis of the early Drosophila embryo. The identification of 60–70 Dorsal target genes, along with the characterization of ≈35 associated regulatory DNAs, suggests that there are at least six different regulatory codes driving diverse DV expression profiles.
Keywords: Drosophila, regulatory code, embryo, morphogen
The dorsal–ventral (DV) patterning of the early Drosophila embryo is controlled by a sequence-specific transcription factor, Dorsal, which is related to mammalian NF-κB (1–3). Differential activation of the Toll receptor leads to the formation of a broad Dorsal nuclear gradient, with peak levels present in ventral regions and progressively lower levels in lateral and dorsal regions (Fig. 1) (4, 5).
Fig. 1.
The Dorsal nuclear gradient generates diverse thresholds of gene expression. The Dorsal gradient regulates 60–70 target genes in a concentration-dependent manner across the DV axis of the early embryo. The expression domains of these genes are depicted on a diagram representing a cross-section through an early embryo. Filled green circles represent high levels of nuclear Dorsal protein, and shaded green and yellow circles represent intermediate and low levels, respectively. At least six different expression patterns have been identified. High levels of Dorsal activate Type 1 genes such as snail in the presumptive mesoderm. Type 2 genes such as rho and vnd are activated by intermediate levels of Dorsal gradient in ventral regions of the presumptive neurogenic ectoderm. In addition, type 1A and 2A expression profiles, represented by sim and ind, depend on Notch and EGF signaling and high and intermediate levels of the Dorsal gradient, respectively. Two type 3 expression patterns, those represented by sog and zen, are generated by the lowest levels of the gradient. The same low levels that are sufficient to activate sog repress the expression of dpp, zen, and tolloid. Thus, the Dorsal gradient generates three basic transcription responses, and these three thresholds produce a total of six different patterns of gene expression across the DV axis of the early embryo. This figure is adapted from the review by Stathopoulos and Levine (50).
The Dorsal gradient initiates DV patterning by regulating 60–70 target genes in a concentration-dependent manner (Fig. 1) (6, 7). Altogether, the Dorsal gradient generates six discrete patterns of gene expression across the DV axis of cellularizing embryos. Most of the target genes are activated only by the highest levels of the Dorsal gradient, in the ventral third of the embryo forming the mesoderm. Intermediate levels of the gradient lead to restricted patterns of gene expression within ventral regions of the presumptive neurogenic ectoderm. The lowest levels of the gradient activate gene expression throughout the neurogenic ectoderm; these same low levels also restrict expression to the dorsal ectoderm.
In addition to these four basic patterns of gene expression, produced by high, intermediate, and low levels of the Dorsal gradient, there are at least two other expression profiles, represented by the target genes sim and ind. Insights into the regulatory codes governing their expression were obtained from the DV gene regulatory network.
A combination of genetic analysis, transgenesis, bioinformatics, and microarray assays led to the elucidation of a comprehensive gene regulatory network for the DV patterning of the early embryo (6–11). This network contains >40 genes encoding sequence-specific transcription factors or cell signaling components that impinge on the activities of these factors (12, 13). Altogether, the network comprises >200 functional interconnections among the 40 major determinants of DV patterning.
Cell–Cell Interactions Produce Additional Dorsal Gradient Readouts
The sim expression pattern depends on the prior activation of snail by high levels of the Dorsal gradient within the presumptive mesoderm (Fig. 2A). snail encodes a zinc finger repressor of tom, which encodes an inhibitor of the Notch signaling ligand, Delta (14, 15). As a result of this repression, Delta is stably expressed in the ventral mesoderm. All of the mesectoderm cells in contact with the mesoderm, the ventral-most cells of the presumptive neurogenic ectoderm, receive this Delta signal and experience Notch signaling (16, 17). Notch, in concert with the Dorsal and Twist gradients, triggers the expression of sim in the presumptive mesoderm and mesectoderm. sim expression is restricted to the mesectoderm because of repression by Snail in the presumptive mesoderm.
Fig. 2.
sim and ind expression patterns are variants of type 1 and type 2 threshold responses. (A) Colocalization of snail (green) and sim (red) mRNAs in a pregastrular embryo. Sharp boundaries of snail expression delineate dorsal limits of the presumptive mesoderm that directly contact the ventral-most cells of the presumptive neurogenic ectoderm, the mesectoderm, where sim is expressed. The Snail repressor leads to stable presentation of the Notch signaling ligand, Delta, in the presumptive mesoderm by inhibiting Tom expression. The adjacent mesectoderm cells receive this Delta signal and experience Notch signaling. Notch, in conjunction with the Dorsal and Twist gradients, triggers the expression of sim. Thus, the sim expression pattern arises from the type 1 snail pattern, which produces a localized source of Notch signaling. (G) Expression of ind depends on EGF signaling generated by the products of two type 2 Dorsal target genes, vein and rho. (D) vein encodes a secreted EGF signaling ligand, whereas rho encodes a membrane-bound protease that releases the Spitz EGF signaling ligand from ventral regions of the neurogenic ectoderm. This notion is consistent with two experimental observations. (B and C) ind expression (B) is lost in rho;vn double mutants (C). (E and F) Furthermore, misexpression of rho via the eve stripe2 enhancer (E) induces ectopic expression of ind in the presumptive neurogenic ectoderm (F). Ectopic staining is restricted to lateral regions where there are limiting amounts of the Dorsal.
Cell signaling also works in concert with the Dorsal gradient to establish the ind expression pattern (Fig. 2B). ind expression depends on EGF signaling, which is produced by vein and rho, two target genes activated by intermediate levels of the Dorsal gradient (Fig. 1). vein encodes a secreted EGF signaling molecule (18, 19), whereas rho encodes a membrane protease that releases the Spitz EGF ligand from ventral regions of the neurogenic ectoderm (20, 21). These EGF signals activate MAP kinase in both ventral and lateral regions of the neurogenic ectoderm (22). In principle, ind can be activated in both regions by the combination of low levels of the Dorsal gradient and EGF signaling, but expression is kept off in the ventral neurogenic ectoderm by Vnd, a sequence-specific transcriptional repressor that is activated by intermediate levels of Dorsal (Fig. 1) (23–25). As a result of this repression, ind expression is restricted to lateral regions of the neurogenic ectoderm. Thus, just as the sim expression profile is defined by the combination of Dorsal and Notch signaling (Fig. 3 C and D), the ind expression pattern is regulated by the combination of Dorsal and EGF signaling (Figs. 2G and 3 G and H).
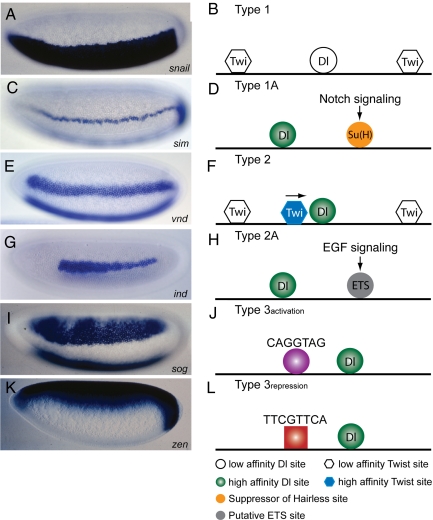
Fig. 3.
Regulatory codes for diverse DV patterns. (A) The highest levels of Dorsal activate type 1 genes, such as snail, thereby restricting the expression of the associated target genes to the presumptive mesoderm. (B) Most of type 1 enhancers contain a disordered series of low-affinity Dorsal and/or Twist binding sites. (C) Expression of the type 1A gene, sim, in the mesectoderm depends on Notch signaling, which is produced by the Snail-dependent inhibition of Tom. (D) The sim enhancer contains a high-affinity Dorsal binding site and Suppressor of Hairless Su(H) sites, which mediate Notch signaling. (E) Type 2 genes, such as vnd, are activated by intermediate levels of the Dorsal gradient, and low levels of Twist. (F) All five of the known type 2 enhancers contain a fixed arrangement of Dorsal and Twist binding sites. An optimal Dorsal site is closely linked to an asymmetric Twist site, which is positioned in a convergent orientation relative to the Dorsal site. This arrangement plays a critical role for cooperative DNA binding interactions between Dorsal and Twist/Da heterodimers. These cooperative interactions generate a stable pattern of type 2 expression in ventral regions of the neurogenic ectoderm. (G) Intermediate levels of the Dorsal gradient induce EGF signaling, which is important for the activation of the Type 2A gene, ind, in the neurogenic ectoderm. (H) The ind enhancer contains a high-affinity Dorsal binding site and ETS sites, which mediate EGF signaling. (I and K) Two different classes of type 3 enhancers are activated (type 3activation) and repressed (type 3repression) by the same low levels of the Dorsal gradient. (I and J) Optimal Dorsal binding sites are closely linked to CAGGTAG motifs in the intronic enhancer that regulates the type 3activation gene, sog. This CAGGTAG motif is probably recognized by a ubiquitous activator distributed throughout the early embryo. It is conceivable that cooperative DNA binding interactions between CAGGTAG binding factor and Dorsal would permit both intermediate and low levels of the Dorsal protein to activate sog expression throughout the neurogenic ectoderm. (K) zen is repressed by the same low levels of the Dorsal gradient that activate sog. (L) The zen enhancer contains optimal Dorsal binding sites and a closely linked motif, WTCKTTCAT, that binds one or more ubiquitous factors that convert Dorsal into a silencer. Dorsal–corepressor interactions appear to induce the exposure of a cryptic peptide motif in Dorsal that recruits the Groucho corepressor (42).
Two lines of evidence support the view that ind expression requires EGF signaling. First, the endogenous ind expression pattern is completely lost in rho;vn double mutants (Fig. 2 B and C) (23). Second, misexpression of rho in transgenic embryos using the eve stripe 2 enhancer induces ectopic activation of ind in the presumptive neurogenic ectoderm. However, this ectopic expression is restricted to a pyramid pattern, suggesting that EGF signaling is not sufficient for induction, but is limited by diminishing levels of the Dorsal gradient (Fig. 2 D–F).
Whole-Genome Identification of Dorsal Target Enhancers
We have argued that four of the DV expression profiles, those represented by snail, sim, vnd, and ind, are produced by high and intermediate levels of the Dorsal gradient. High levels of Dorsal activate snail, which leads to the localized sim pattern via Notch signaling, whereas intermediate levels activate vnd, vn, and rho, which delineate the ind pattern. The final two expression patterns, those represented by sog and zen, are generated by the lowest levels of the gradient (Fig. 1). The same low levels that are sufficient to activate sog repress the expression of dpp, zen, and tolloid (26). Thus, the Dorsal gradient generates a total of six different patterns of gene expression across the DV axis of the early embryo (Fig. 3).
Of course, Dorsal does not work alone in regulating these differential patterns of gene expression. Many target genes are regulated by a combination of Dorsal, Twist, and/or Snail. High levels of Dorsal activate twist, and the encoded Twist basic helix–loop–helix (bHLH) activator is distributed in a steeper gradient than the input Dorsal gradient. Dorsal and Twist jointly activate more than half of all Dorsal target genes (27, 28). High levels of both activators are required for induction and maintenance of snail expression, which exhibits a tight pattern of expression within the presumptive mesoderm (29). The encoded zinc finger Snail repressor establishes a sharp boundary between the presumptive mesoderm and neurogenic ectoderm (e.g., Fig. 3A) (30, 31). Several genes are activated by low levels of Dorsal and Twist in lateral regions of early embryos (e.g., Fig. 3E). The Snail repressor keeps these genes off in the mesoderm, thereby restricting their expression to the neurogenic ectoderm where they are important for the differentiation of the CNS.
To understand the regulatory basis for the six DV expression patterns, a large number of Dorsal target enhancers have been identified and characterized. Several methods have been used, including conventional gene fusion assays (e.g., refs. 27–29), bioinformatics methods (9, 10), and most recently, ChIP-chip assays (7). The latter method proved to be the most effective means for the genomewide identification of Dorsal target enhancers.
The experiments were done by using Toll10b mutant embryos (32), which possess high levels of constitutive Toll signaling across the entire DV axis. As a result, there are high levels of Dorsal, Twist, and Snail throughout the embryo in place of the normal gradients. High levels of Dorsal and Twist activate mesoderm genes throughout the embryos; the Snail repressor keeps ectodermal genes off. As a result, all of the cells of Toll10b embryos form mesoderm at the expense of ectodermal derivatives.
Toll10b embryos were cross-linked at 2–4 h after fertilization because this is the time when the Dorsal gradient regulates its various target genes. Chromosomal DNA was isolated from the mutants, sonicated into small pieces, and then immunoprecipitated with antibodies directed against Dorsal, Twist, and Snail. There are at least 200–300 significant clusters of Dorsal, Twist, and Snail binding sites scattered throughout the Drosophila genome (7). These clusters identified 22 of the 24 known Dorsal target enhancers (7) and another 10 enhancers that had not been previously identified (WntD, kni, mes5, pnr, tup, and shadow enhancers for vnd, miR-1, brk and sog; see refs. 7 and 33). Altogether, the 34 experimentally confirmed Dorsal target enhancers regulate 29 of the 60–70 known Dorsal target genes (6, 11). The ChIP-chip assays also provide clear predictions for at least another ≈20 putative DV enhancers, including those associated with Mes4 (5′), Neu3 (intron), SoxN (3′), CadN (3′), and Doc 1 (5′).
Regulatory Codes for DV Expression Patterns
The highest levels of the Dorsal gradient activate gene expression in the presumptive mesoderm. Most of the associated enhancers (16 in total) contain a disordered series of low affinity Dorsal and/or Twist binding sites that function in an additive fashion to activate gene expression (Fig. 3 A and B) (6, 10). A very different scenario is observed for those enhancers that mediate gene expression in ventral regions of the neurogenic ectoderm. These enhancers are activated by intermediate levels of the Dorsal gradient and low levels of Twist (e.g., Fig. 3E).
All five of the known type 2 enhancers contain a fixed arrangement of Dorsal and Twist binding sites (Fig. 3F) (34–36). An optimal Dorsal site is closely linked to an asymmetric Twist site, which is positioned in a convergent orientation relative to the Dorsal site. This organization is conserved in all 12 of the sequenced drosophilids (36). Inverting the Twist site causes a severe reduction in the activities of an otherwise normal vein-lacZ fusion gene (35). These enhancers also contain Snail binding sites, which inhibit their activities in the ventral mesoderm. Thus, there are two differences in the regulatory codes of enhancers mediating expression in the mesoderm and ventral neurogenic ectoderm. The latter enhancers contain a fixed arrangement of Dorsal and Twist binding sites and also contain Snail repressor sites. Enhancers mediating expression in the mesoderm lack an intrinsic organization, or grammar, and contain low-affinity Dorsal and/or Twist sites.
The fixed arrangement of Dorsal and Twist sites seen in enhancers active in the ventral neurogenic ectoderm is probably essential for cooperative DNA binding interactions between Dorsal and the Daughterless (Da) subunit of Da/Twist heterodimers bound to the asymmetric Twist site (28). Da is a ubiquitous bHLH protein that is expressed throughout the early embryo and related to the mammalian E12/E47 protein (38, 39). In general, Da does not function as a homodimer, but forms heterodimers with tissue-specific transcription factors such as Twist (mesoderm) and Achaete (neurogenic ectoderm). Dl-Da/Twi cooperative binding interactions produce a stable pattern of type 2 expression in ventral regions of the neurogenic ectoderm, even though there is a 100-fold reduction in the levels of the Twist gradient (35, 36, 40).
A similar principle, cooperative DNA binding interactions to a fixed arrangement of sites, might also apply to those enhancers responding to the lowest levels of the Dorsal gradient. The sog enhancer contains a series of optimal Dorsal binding sites and linked CAGGTAG sequences (Fig. 3 I and J). This latter motif has been identified in a number of genes expressed in the early, precellular Drosophila embryo (41). The CAGGTAG motif is probably recognized by a ubiquitous, maternal activator distributed throughout the early embryo. Linked Dorsal and CAGGTAG sites are conserved in the sog intronic enhancer among all 12 drosophilids. It is possible that the binding of the activator to CAGGTAG facilitates the binding of Dorsal to linked sites via cooperative DNA binding interactions, similar to those seen for Dorsal and Twist. This cooperative interaction would permit both intermediate and low levels of the Dorsal protein to activate sog expression throughout the neurogenic ectoderm.
The same low levels of the Dorsal gradient that activate sog (e.g., Fig. 3I) also repress target genes such as zen (e.g., Fig. 3K) (26, 42). The zen enhancer contains a series of optimal Dorsal binding sites, similar to those seen in the sog enhancer, which is activated by Dorsal. However, unlike sog, the zen enhancer contains linked binding sites that convert Dorsal into a transcriptional repressor (Fig. 3L).
Dorsal contains a cryptic peptide motif that can interact with the Groucho corepressor (42, 43). The exposure of this motif depends on protein–protein interactions between Dorsal and neighboring corepressor proteins. One of these proteins recognizes a highly conserved sequence motif, WTCKTTCAT. Previous studies in Drosophila melanogaster demonstrated that Dorsal–corepressor interactions depend on helical phasing of Dorsal and the linked corepressor binding site (44). Helical phasing is classical evidence for direct protein–protein interactions on the DNA template (45–47), and this phasing is highly conserved among the zen enhancers in all 12 sequenced drosophilids. Thus, the Dorsal-CAGGTAG linkage seen in the sog enhancer is replaced by Dorsal-WTCKTTCAT linkage in zen (44). These distinct regulatory codes determine whether the target genes are activated or repressed by the same low levels of the Dorsal gradient.
Conclusions and Implications
A combination of classical genetic screens, computational methods, conventional gene fusion assays, along with the advent of more recent postgenome technologies such as whole-genome tiling arrays and ChIP-chip assays, has permitted the systematic identification of Dorsal target genes and their associated regulatory DNAs (e.g., refs. 7 and 11; reviewed in refs. 8 and 13). This information was used to create an extensive gene regulatory network for the DV patterning of the Drosophila embryo (12, 13). The network helped define six regulatory codes underlying diverse DV patterns of gene activity. The recent determination of 12 different drosophilid whole-genome assemblies helped confirm the notion that stable patterns of gene expression (e.g., in the ventral neurogenic ectoderm) in response to sharply diminishing levels of the Dorsal and Twist gradients depend on a fixed arrangement of linked activator sites. Target enhancers that respond to peak levels of the Dorsal and/or Twist gradients lack an intrinsic organization, whereas enhancers responding to low levels exhibit a “grammar” that is conserved among divergent drosophilids (7, 35, 36). This organization probably fosters cooperative occupancy of Dorsal binding sites.
We propose that other regulatory gradients function like Dorsal to produce complex patterns of gene expression. For example, the floorplate organizer specifies at least four different neuronal cell types in the developing vertebrate neural tube (48). As seen for the Dorsal gradient, they might arise from the combination of Gli activator thresholds and cell–cell interactions similar to the role of Notch and EGF signaling in producing the sim and ind expression patterns, respectively (Fig. 3 C, D, G, and H). Similar rules might also apply to the zone of polarizing activity (ZPA) and anterior–posterior compartment organizers that pattern vertebrate and insect limbs (49).
Acknowledgments.
This study was funded by grants from the Moore Foundation and the National Institutes of Health.
Footnotes
This paper results from the Arthur M. Sackler Colloquium of the National Academy of Sciences, “Gene Networks in Animal Development and Evolution,” held February 15–16, 2008, at the Arnold and Mabel Beckman Center of the National Academies of Sciences and Engineering in Irvine, CA. The complete program and audio files of most presentations are available on the NAS web site at http://www.nasonline.org/SACKLER_Gene_Networks.
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
References
- 1.Rushlow CA, Han K, Manley JL, Levine M. The graded distribution of the dorsal morphogen is initiated by selective nuclear transport in Drosophila. Cell. 1989;59:1165–1177. doi: 10.1016/0092-8674(89)90772-1. [DOI] [PubMed] [Google Scholar]
- 2.Roth S, Stein D, Nüsslein-Volhard C. A gradient of nuclear localization of the dorsal protein determines dorsoventral pattern in the Drosophila embryo. Cell. 1989;59:1189–1202. doi: 10.1016/0092-8674(89)90774-5. [DOI] [PubMed] [Google Scholar]
- 3.Ip YT, Kraut R, Levine M, Rushlow CA. The dorsal morphogen is a sequence-specific DNA-binding protein that interacts with a long-range repression element in Drosophila. Cell. 1991;64:439–446. doi: 10.1016/0092-8674(91)90651-e. [DOI] [PubMed] [Google Scholar]
- 4.Anderson KV, Bokla L, Nüsslein-Volhard C. Establishment of dorsal-ventral polarity in the Drosophila embryo: The induction of polarity by the Toll gene product. Cell. 1985;42:791–798. doi: 10.1016/0092-8674(85)90275-2. [DOI] [PubMed] [Google Scholar]
- 5.Anderson KV, Jürgens G, Nüsslein-Volhard C. Establishment of dorsal-ventral polarity in the Drosophila embryo: Genetic studies on the role of the Toll gene product. Cell. 1985;42:779–789. doi: 10.1016/0092-8674(85)90274-0. [DOI] [PubMed] [Google Scholar]
- 6.Stathopoulos A, Van Drenth M, Erives A, Markstein M, Levine M. Whole-genome analysis of dorsal-ventral patterning in the Drosophila embryo. Cell. 2002;111:687–701. doi: 10.1016/s0092-8674(02)01087-5. [DOI] [PubMed] [Google Scholar]
- 7.Zeitlinger J, et al. Whole-genome ChIP-chip analysis of Dorsal, Twist, and Snail suggests integration of diverse patterning processes in the Drosophila embryo. Genes Dev. 2007;21:385–390. doi: 10.1101/gad.1509607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stathopoulos A, Levine M. Dorsal gradient networks in the Drosophila embryo. Dev Biol. 2002;246:57–67. doi: 10.1006/dbio.2002.0652. [DOI] [PubMed] [Google Scholar]
- 9.Markstein M, Markstein P, Markstein V, Levine MS. Genomewide analysis of clustered Dorsal binding sites identifies putative target genes in the Drosophila embryo. Proc Natl Acad Sci USA. 2002;99:763–768. doi: 10.1073/pnas.012591199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Papatsenko D, Levine M. Quantitative analysis of binding motifs mediating diverse spatial readouts of the Dorsal gradient in the Drosophila embryo. Proc Natl Acad Sci USA. 2005;102:4966–4971. doi: 10.1073/pnas.0409414102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Biemar F, et al. Comprehensive identification of Drosophila dorsal-ventral patterning genes using a whole-genome tiling array. Proc Natl Acad Sci USA. 2006;103:12763–12768. doi: 10.1073/pnas.0604484103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Levine M, Davidson E. Gene regulatory networks for development. Proc Ntal Acad Sci USA. 2005;102:4936–4942. doi: 10.1073/pnas.0408031102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stathopoulos A, Levine M. Genomic regulatory networks and animal development. Dev Cell. 2005;9:449–462. doi: 10.1016/j.devcel.2005.09.005. [DOI] [PubMed] [Google Scholar]
- 14.Bardin AJ, Schweisguth F. Bearded family members inhibit Neuralized-mediated endocytosis and signaling activity of Delta in Drosophila. Dev Cell. 2006;10:245–255. doi: 10.1016/j.devcel.2005.12.017. [DOI] [PubMed] [Google Scholar]
- 15.De Renzis S, Yu J, Zinzen R, Wieschaus E. Dorsal-ventral pattern of Delta trafficking is established by a Snail-Tom-Neuralized pathway. Dev Cell. 2006;10:257–264. doi: 10.1016/j.devcel.2006.01.011. [DOI] [PubMed] [Google Scholar]
- 16.Cowden J, Levine M. The Snail repressor positions Notch signaling in the Drosophila embryo. Development. 2002;129:1785–1793. doi: 10.1242/dev.129.7.1785. [DOI] [PubMed] [Google Scholar]
- 17.Morel V, Le Borgne R, Schweisguth F. Snail is required for Delta endocytosis and Notch-dependent activation of single-minded expression. Dev Genes Evol. 2003;213:65–72. doi: 10.1007/s00427-003-0296-x. [DOI] [PubMed] [Google Scholar]
- 18.Schnepp B, Grumbling G, Donaldson T, Simcox A. Vein is a novel component in the Drosophila epidermal growth factor receptor pathway with similarity to the neuregulins. Genes Dev. 1996;10:2302–2313. doi: 10.1101/gad.10.18.2302. [DOI] [PubMed] [Google Scholar]
- 19.Yarnitzky T, Min L, Volk T. The Drosophila neuregulin homolog Vein mediates inductive interactions between myotubes and their epidermal attachment cells. Genes Dev. 1997;11:2691–2700. doi: 10.1101/gad.11.20.2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sturtevant MA, Roark M, Bier E. The Drosophila rhomboid gene mediates the localized formation of wing veins and interacts genetically with components of the EGF-R signaling pathway. Genes Dev. 1993;7:961–973. doi: 10.1101/gad.7.6.961. [DOI] [PubMed] [Google Scholar]
- 21.Urban S, Lee JR, Freeman M. Drosophila rhomboid-1 defines a family of putative intramembrane serine proteases. Cell. 2001;107:173–182. doi: 10.1016/s0092-8674(01)00525-6. [DOI] [PubMed] [Google Scholar]
- 22.Gabay L, Seger R, Shilo BZ. In situ activation pattern of Drosophila EGF receptor pathway during development. Science. 1997;277:1103–1106. doi: 10.1126/science.277.5329.1103. [DOI] [PubMed] [Google Scholar]
- 23.Skeath JB. The Drosophila EGF receptor controls the formation and specification of neuroblasts along the dorsal-ventral axis of the Drosophila embryo. Development. 1998;125:3301–3312. doi: 10.1242/dev.125.17.3301. [DOI] [PubMed] [Google Scholar]
- 24.Koizumi K, et al. Mutations that affect the ability of the vnd/NK-2 homeoprotein to regulate gene expression: Transgenic alterations and tertiary structure. Proc Natl Acad Sci USA. 2003;100:3119–3124. doi: 10.1073/pnas.0438043100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cowden J, Levine M. Ventral dominance governs sequential patterns of gene expression across the dorsal-ventral axis of the neuroectoderm in the Drosophila embryo. Dev Biol. 2003;262:335–349. doi: 10.1016/s0012-1606(03)00395-6. [DOI] [PubMed] [Google Scholar]
- 26.Jiang J, Rushlow CA, Zhou Q, Small S, Levine M. Individual dorsal morphogen binding sites mediate activation and repression in the Drosophila embryo. EMBO J. 1992;11:3147–3154. doi: 10.1002/j.1460-2075.1992.tb05387.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jiang J, Kosman D, Ip YT, Levine M. The dorsal morphogen gradient regulates the mesoderm determinant twist in early Drosophila embryos. Genes Dev. 1991;5:1881–1891. doi: 10.1101/gad.5.10.1881. [DOI] [PubMed] [Google Scholar]
- 28.Jiang J, Levine M. Binding affinities and cooperative interactions with bHLH activators delimit threshold responses to the dorsal gradient morphogen. Cell. 1993;72:741–752. doi: 10.1016/0092-8674(93)90402-c. [DOI] [PubMed] [Google Scholar]
- 29.Ip YT, Park RE, Kosman D, Yazdanbakhsh K, Levine M. dorsal-twist interactions establish snail expression in the presumptive mesoderm of the Drosophila embryo. Genes Dev. 1992;6:1518–1530. doi: 10.1101/gad.6.8.1518. [DOI] [PubMed] [Google Scholar]
- 30.Kosman D, Ip YT, Levine M, Arora K. Establishment of the mesoderm-neuroectoderm boundary in the Drosophila embryo. Science. 1991;254:118–122. doi: 10.1126/science.1925551. [DOI] [PubMed] [Google Scholar]
- 31.Leptin M. twist and snail as positive and negative regulators during Drosophila mesoderm development. Genes Dev. 1991;5:1568–1576. doi: 10.1101/gad.5.9.1568. [DOI] [PubMed] [Google Scholar]
- 32.Schneider DS, Hudson KL, Lin TY, Anderson KV. Dominant and recessive mutations define functional domains of Toll, a transmembrane protein required for dorsal-ventral polarity in the Drosophila embryo. Genes Dev. 1991;5:797–807. doi: 10.1101/gad.5.5.797. [DOI] [PubMed] [Google Scholar]
- 33.Hong JW, Hendrix DA, Levine M. Shadow enhancers as a source of evolutionary novelty. Science. 2008;321:1314. doi: 10.1126/science.1160631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Markstein M, et al. A regulatory code for neurogenic gene expression in the Drosophila embryo. Development. 2004;131:2387–2394. doi: 10.1242/dev.01124. [DOI] [PubMed] [Google Scholar]
- 35.Zinzen RP, Senger K, Levine M, Papatsenko D. Computational models for neurogenic gene expression in the Drosophila embryo. Curr Biol. 2006;16:1358–1365. doi: 10.1016/j.cub.2006.05.044. [DOI] [PubMed] [Google Scholar]
- 36.Papatsenko D, Levine M. A rationale for the enhanceosome and other evolutionarily constrained enhancers. Curr Biol. 2007;17:R955–R957. doi: 10.1016/j.cub.2007.09.035. [DOI] [PubMed] [Google Scholar]
- 37.Clark AG, et al. Evolution of genes and genomes on the Drosophila phylogeny. Nature. 2007;450:203–218. doi: 10.1038/nature06341. [DOI] [PubMed] [Google Scholar]
- 38.Murre C. Helix–loop–helix proteins and lymphocyte development. Nat Immunol. 2005;6:1079–1086. doi: 10.1038/ni1260. [DOI] [PubMed] [Google Scholar]
- 39.Campos-Ortega JA, Knust E. Molecular analysis of a cellular decision during embryonic development of Drosophila melanogaster: Epidermogenesis or neurogenesis. Eur J Biochem. 1990;190:1–10. doi: 10.1111/j.1432-1033.1990.tb15538.x. [DOI] [PubMed] [Google Scholar]
- 40.Zinzen RP, Papatsenko D. Enhancer responses to similarly distributed antagonistic gradients in development. PLoS Comput Biol. 2007;3:e84. doi: 10.1371/journal.pcbi.0030084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.ten Bosch JR, Benavides JA, Cline TW. The TAGteam DNA motif controls the timing of Drosophila preblastoderm transcription. Development. 2006;133:1967–1977. doi: 10.1242/dev.02373. [DOI] [PubMed] [Google Scholar]
- 42.Dubnicoff T, et al. Conversion of dorsal from an activator to a repressor by the global corepressor Groucho. Genes Dev. 1997;11:2952–2957. doi: 10.1101/gad.11.22.2952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ratnaparkhi GS, Jia S, Courey AJ. Uncoupling dorsal-mediated activation from dorsal-mediated repression in the Drosophila embryo. Development. 2006;133:4409–4414. doi: 10.1242/dev.02643. [DOI] [PubMed] [Google Scholar]
- 44.Cai HN, Arnosti DN, Levine M. Long-range repression in the Drosophila embryo. Proc Natl Acad Sci USA. 1996;93:9309–9314. doi: 10.1073/pnas.93.18.9309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thanos D, Maniatis T. The high mobility group protein HMG I(Y) is required for NF-κB-dependent virus induction of the human IFN-β gene. Cell. 1992;71:777–789. doi: 10.1016/0092-8674(92)90554-p. [DOI] [PubMed] [Google Scholar]
- 46.Vershon AK, Johnson AD. A short, disordered protein region mediates interactions between the homeodomain of the yeast α2 protein and the MCM1 protein. Cell. 1993;72:105–112. doi: 10.1016/0092-8674(93)90054-t. [DOI] [PubMed] [Google Scholar]
- 47.Natesan S, Gilman MZ. DNA bending and orientation-dependent function of YY1 in the c-fos promoter. Genes Dev. 1993;7:2497–2509. doi: 10.1101/gad.7.12b.2497. [DOI] [PubMed] [Google Scholar]
- 48.Ericson J, Briscoe J, Rashbass P, van Heyningen V, Jessell TM. Graded sonic hedgehog signaling and the specification of cell fate in the ventral neural tube. Cold Spring Harb Symp Quant Biol. 1997;62:451–466. [PubMed] [Google Scholar]
- 49.Riddle RD, Johnson RL, Laufer E, Tabin C. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell. 1993;75:1401–1416. doi: 10.1016/0092-8674(93)90626-2. [DOI] [PubMed] [Google Scholar]
- 50.Stathopoulos A, Levine M. Whole-genome analysis of Drosophila gastrulation. Curr Opin Genet Dev. 2004;14:477–484. doi: 10.1016/j.gde.2004.07.004. [DOI] [PubMed] [Google Scholar]