Abstract
Background
A subset of patients with diffuse gastric cancer harbor deleterious cancer-causing germline mutations in the E-cadherin (CDH1) gene, which predisposes to the autosomal dominantly inherited hereditary diffuse gastric cancer (HDGC) syndrome. These mutations are associated with a 70% life-time risk for diffuse gastric cancer (DGC) and, in addition, a 40% risk for lobular breast cancer in females. Management options for unaffected mutation carriers include prophylactic total gastrectomy.
Methods
Four HDGC pedigrees from a cohort of 56 CDH1 mutation-positive families have been subjected to further analysis. Cancer diagnoses, whenever possible, were verified with pathology reports and/or slides/tissue block review. Genetic counseling for family members covered HDGC’s natural history, the pros and cons of mutation testing, the lack of effective screening procedures available to CDH1 mutation-positive individuals, and the option for them of prophylactic total gastrectomy.
Results
Within these four families, carrier testing for mutations in the CDH1 gene was carried out on 52 individuals, of whom 25 were mutation positive. Prophylactic gastrectomies were performed on a total of 17 individuals from 3 of the families, including 11 first cousins from one of the families. Occult DGC was diagnosed in gastrectomy specimens from 13/17 (76.5%) individuals. During follow-up questioning, each of the mentioned 11 cousins stated that the decision for the prophylactic procedure was the “right one” for them.
Conclusions
Unaffected mutation carriers from HDGC families face difficult decisions and can best be assisted through education and the interactions with counseling by an informed multidisciplinary team.
Introduction
Gastric cancer is the second most common cause of cancer mortality worldwide.1 The two major categories are the intestinal type, which is more commonly associated with environmental perturbations, and diffuse gastric cancer (DGC) which is more often ascribed etiologically to host factor effects.2 The intestinal type has declined in incidence, while DGC has remained stable and may even be increasing.3 Hereditary diffuse gastric cancer (HDGC) is an autosomal dominantly inherited disorder caused by mutations of the E-cadherin gene (CDH1, epithelial cadherin, OMIM#19209). The decreased expression of this gene in DGCs appears to account for the pathological differences between intestinal and diffuse variants of gastric cancer in a subset of families.4–6 CDH1 germline mutations have been identified in approximately 40–50% of well-defined HDGC families from low-incident populations.5,7,8 There is also an excess of lobular carcinoma of the breast in women from those HDGC families with CDH1 mutations.6,9,10 The clinical advantage of identifying a CDH1 mutation carrier is its level of certainty of disease expression, limited only by its penetrance, which in DGC is estimated to be in the range of 70%.5,11 Unfortunately, it is not possible to determine which CDH1 mutation carriers will develop DGC. Pathology findings show the signet cell cancer phenotype to be submucosal, which severely limits the ability of currently available screening procedures to detect early gastric cancer. Mutation carriers can be offered the option of prophylactic total gastrectomy for reduction of cancer morbidity and mortality.7,12
Our experience with four extended HDGC families who are part of a cohort of 56 CDH1 mutation-positive families, will be discussed.
Material and Methods
This study was approved by the authors’ Institutional Review Boards. Informed consent was obtained from the subjects investigated. Detailed cancer family histories that included the assessment of cancer of all anatomic sites, with particular attention to pathology verification whenever possible, were ascertained at the respective centers. High-risk family members were identified, counseled, and, if appropriate, offered genetic testing for the cancer predisposing CDH1 mutation. Genetic counseling was again provided at the time results were revealed.
The proband in family 1 (Figure 1) contacted us because she was concerned about her cancer family history following the death of her brother due to DGC. She had been aware that her mother and four of her mother’s siblings had also died from DGC. A working pedigree was initiated through the compilation of a genealogic history in concert with medical and pathology documentation. Once permission was granted, this search was extended to as many of the proband’s relatives as possible. The family was geographically dispersed, with residences primarily in communities throughout the Midwest and far western United States.
Figure 1.
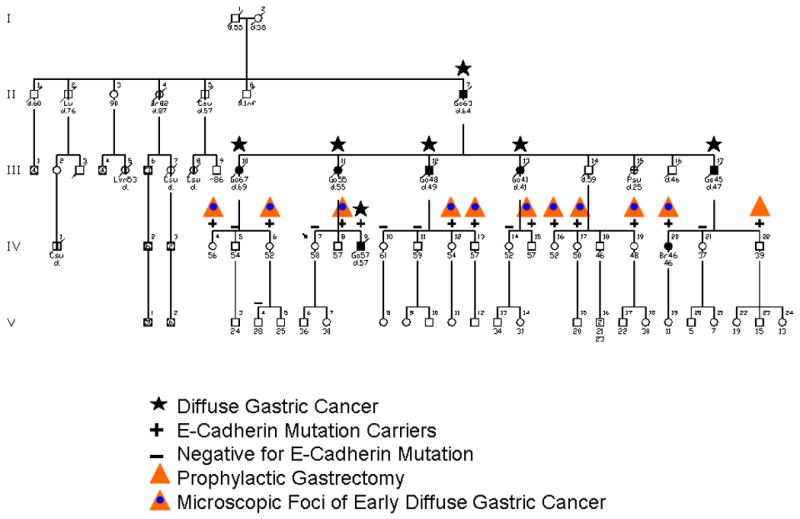
This hereditary diffuse gastric cancer (HDGC) family features 11 first cousins who underwent prophylactic total gastrectomy, 10 of whom manifested submucosal signet cell cancer at the time of surgery. All of these individuals agreed that this preventive surgery was appropriate. Particularly, they noted parents who had died of diffuse gastric cancer (DGC).
Prior to his death, a blood sample had been obtained from the proband’s affected brother and genetic testing for the CDH1 mutation was performed in the molecular genetic laboratories of one of the co-authors (DGH)6,7 and a CDH1 mutation was found. Once knowledge that a mutation in CDH1 had been identified in a family member, those relatives at high risk were advised of the availability of genetic counseling and DNA testing. Seventeen relatives were tested and results were then provided to them during genetic counseling sessions. The 11 who tested positive were advised about the limitations of DGC screening and the pros and cons of prophylactic total gastrectomy.
Eleven first cousins harbored the CDH1 mutation (C1003T in exon 7), and underwent prophylactic total gastrectomy. Six of these cousins underwent chromoendoscopy, endoscopic ultrasound, random biopsies, and PET-CT scanning immediately prior to surgery. All screening tests were negative; however, 10 of the 11 gastrectomies harbored occult carcinomas.13
Subsequently, the family held a reunion, and all of the 11 who had undergone prophylactic total gastrectomy, were in attendance. Three of the authors of this article (H.T.L., J.F.L., and D.G.H.) were invited to attend the reunion, at which time they had an opportunity to review with the attendees all of the ramifications of HDGC. Following the reunion, a questionnaire was forwarded to the 11 subjects in order to evaluate their emotional and physical wellbeing pre- and post-gastrectomy.
In the second family (Figure 2), the proband contacted us following the death of three of his siblings due to DGC over the course of only 18 months. DNA was extracted from their paraffin-embedded tumor tissue blocks and a CDH1 70G→T mutation was found.14 Once the pedigree was extended, a Family Information Service (FIS)15 was provided; family members were educated intensively about all facets of HDGC’s natural history, its screening limitations, potential sequelae of prophylactic total gastrectomy, as well as the pros and cons of DNA testing for the deleterious germline cancer-predisposing CDH1 mutation.
Figure 2.
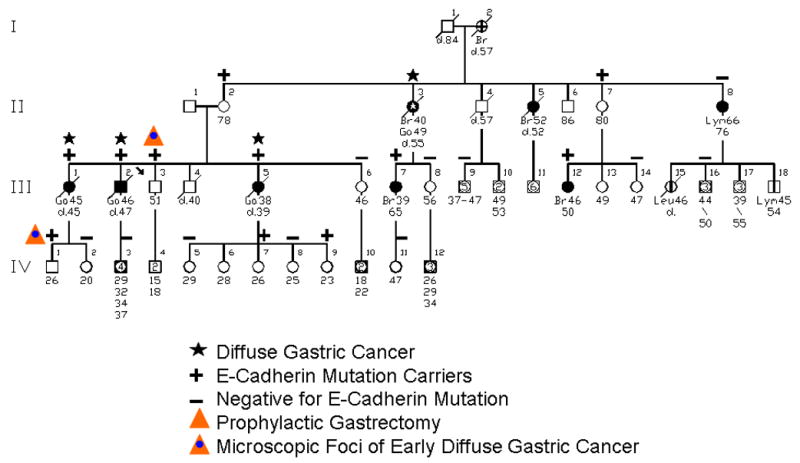
This pedigree depicts an HDGC family wherein 3 of the proband’s siblings died of DGC within an 18-month time period. The findings of CDH1 mutation in the 78-year-old mother (II-2) of the sibship and in her sister (II-7) at age 80, both symptom free, indicated reduced penetrance of this mutation.
Families 3 and 4 (Figures 3 and 4, respectively) were evaluated and underwent essentially the same methods as described for families 1 and 2. The pedigree of family 3 has been published recently.16
Figure 3.
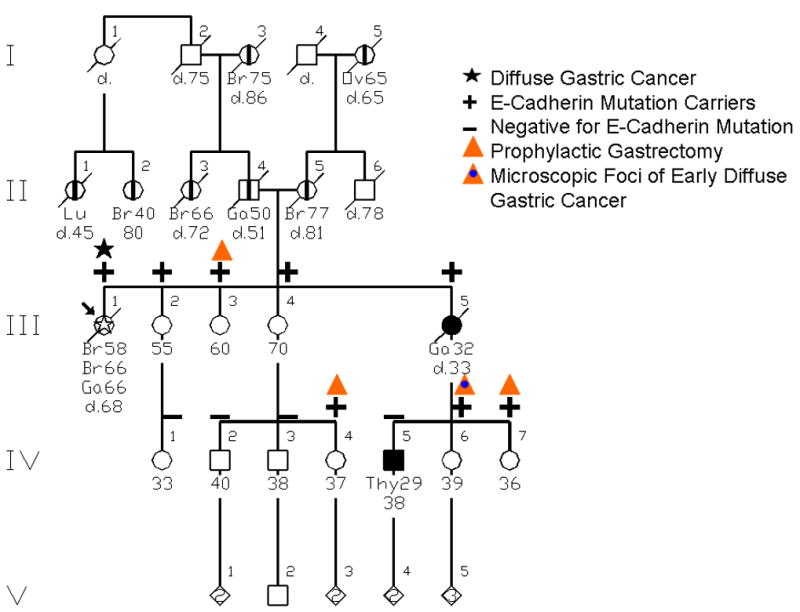
This is an HDGC pedigree which was initially thought to be a hereditary breast-ovarian cancer (HBOC) syndrome family with a BRCA1 or BRCA2 mutation, since the proband (III-1) had bilateral breast cancer and ultimately died of DGC. Importantly, her mother (II-5) also had breast cancer and her maternal grandmother (I-5) had ovarian cancer. Mutational testing identified the CDH1 germline mutation.
Figure 4.
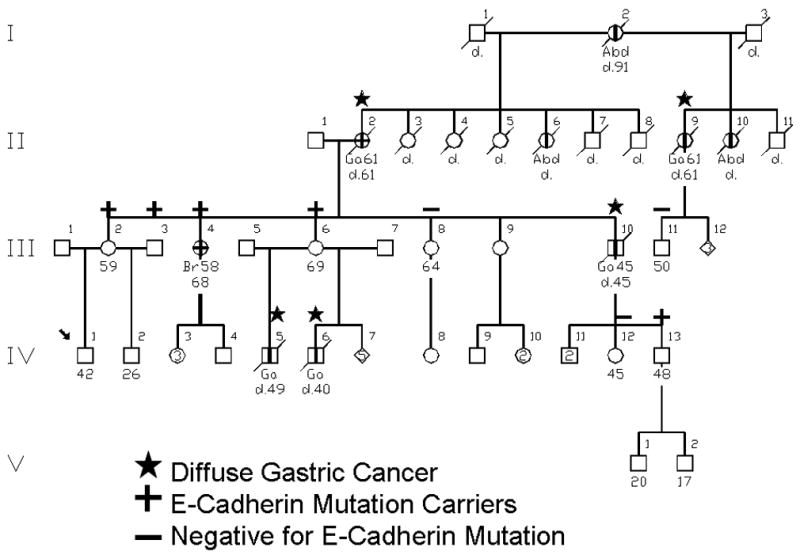
This is an HDGC family wherein initial testing was done on III-6, found to be positive for a CDH1 mutation. She was selected because through two marriages she had sons (IV-5, IV-6) with DGC, and her mother (II-2) died of gastric cancer. Thus, III-6 was considered an obligate germline mutation carrier, which was confirmed by the presence of the deleterious CDH1 mutation.
Results
Family 1
The 11 first cousins in Family 1 (Figure 1) who had undergone prophylactic total gastrectomy, reported in their questionnaires that they were satisfied with their decision to have this surgery, especially when 10 of the 11 cousins learned that their post-gastrectomy pathology specimens showed microscopic evidence of DGC.
The age range when first informed about a member of their family being markedly prone to HDGC was between 45–55 years, with an average age of 48.5 years. Eight of the 11 were told about the importance of genetic testing by a cousin, one by a sister-in-law, one by a sibling, and only one by a physician. Four of the 11 decided immediately to be tested, while 7 indicated that it took them as long as 6 months to make that decision.
Five of the cousins reported that prior to their surgery they had felt frustrated sometimes or often about the fact that there were no cancer screening modalities of proven efficacy available to them. Six thought about how the test results might affect their work or family life, and 6 were concerned about their health insurance status. Ten of the 11 never had difficulty discussing their test results with family members, and they believed that their family had been supportive.
One of these cousins, who had learned at our FIS that mutation carriers also have a high risk of developing lobular carcinoma of the breast, went for breast cancer screening. This resulted in a diagnosis of lobular breast cancer. She stated that “...this [diagnosis] did not come as a big shock. We were looking for it.”
Family 2
The proband (III-3, Figure 2) contacted the Creighton center because three of his siblings (III-1, III-2, III-5) had died of stomach cancer within an 18-month timeframe.14 Family history and pathology reports were gathered. Tumor tissue blocks were retrieved and all three of the siblings were positive for the CDH1 70G→T mutation. We decided that it would be appropriate to have an FIS for the family, and many family members attended. Those high-risk patients who consented were tested for the CDH1 mutation.
The proband initially was reluctant to consider prophylactic total gastrectomy, even though he was positive for the germline mutation. Part of this reluctance was attributed to the fact that his 78-year-old mother (II-2) was mutation positive but had never manifested DGC. Furthermore, he was undergoing periodic upper gastric endoscopy with biopsies and felt confident that this would be protective, even though we had repeatedly discussed the fact that this screening was ineffective. We kept in contact with him for three years with annual telephone calls providing him with information about screening limitations. He finally agreed that his best prospect would be prophylactic gastrectomy. He underwent that procedure and was found to have microscopic foci of early DGC. He became a spokesman for getting better education for the rest of his family. As a consequence, we held a second FIS. This accounts, in part, for IV-1 being tested and found to harbor the CDH1 mutation. He chose to undergo prophylactic gastrectomy, and two foci of intramucosal signet cell carcinoma were identified in the gastric body mucosa.
Also noteworthy in the pedigree is II-3, who had lobular carcinoma of the breast at age 40 and DGC at age 49, and who died at age 55. Her daughter (III-7) also had lobular carcinoma of the breast. Another case of interest is II-7, who is completely asymptomatic at 80 years of age, and is CDH1 mutation positive; she has not undergone gastric screening. Her daughter is mutation positive and has had breast cancer (pathology unavailable).
Family 3
The proband (Figure 3, III-1) was diagnosed with infiltrating lobular carcinoma of the breast at age 58, followed by contralateral infiltrating ductal carcinoma at age 66. Based on her personal history of breast cancer, coupled with breast and ovarian cancer confirmed by death certificate in her family, she was tested for BRCA1 and BRCA2, and found to have a previously unreported BRCA2 variant of uncertain significance (E2175Q). While being treated for her breast cancer, she began to complain of epigastric pain. Gastric duodenoscopy identified a stage IV DGC. During palliative surgery for the DGC, she was found to have a metastatic pancreatic islet cell tumor. CDH1 genetic testing was performed on a research protocol; a CDH1 mutation (2195G>A) was revealed, and subsequently confirmed in a clinical laboratory.8 Although this mutation was originally reported as a missense mutation (R372Q), its likely pathogenic effect is through the activation of a cryptic splice site in exon 14 leading to a truncated protein.8,17
The CDH1 mutation discovery in the family led several of the proband’s high-risk relatives to undergo genetic testing, many of whom were troubled with the decision of whether to pursue prophylactic total gastrectomy. The varied age of onset of DGC in the family and the three-long-lived unaffected mutation-positive women (III-2, III-3, III-4) were reasons given to delay or decline prophylactic surgery. Because of her concern for her children, the 39-year-old daughter (IV-6) of a woman who had died from gastric cancer at age 33 (III-5) elected to have surgery. Her clinical course and personal sentiments are documented in a case report.16 A 2mm focus of signet cell cancer was found in her stomach; a Brenner tumor of the ovary was also discovered and removed. The finding of the occult gastric cancer reinforced her belief that she made the right surgical decision; in addition, it sparked three other women in the family to stop endoscopic screening and pursue preventive surgery. Her sister (IV-7) had a fundic gland polyp removed from her stomach during a screening endoscopy prior to prophylactic gastrectomy; no cancer was identified in her surgical specimen.
Family 4
In Family 4 (Figure 4), the proband (IV-1) is a 42-year-old unaffected male who consulted with a gastroenterologist regarding a 6-month history of abdominal pain plus a striking family history of gastric cancer. Esophageal gastroduodenoscopy (EGD) and CT scan of the abdomen were performed with normal findings. A consultation to the genetics clinic was initiated by his gastroenterologist, who recognized the likely hereditary nature of gastric cancer in this family. This referral enabled our initial contact with the family.
It was recognized that individual III-6, an unaffected 69-year-old female, would be the most informative family member for genetic testing since her mother and two sons (IV-5, IV-6) from different partners died of DGC. Genetic testing for the CDH1 mutation was performed in the molecular genetic laboratories of one of the co-authors (DGH) using previously described methods, and the CDH1 1792 C→T mutation was found.8 Individual III-6 has been cooperative and has participated actively in this research as a way to help her family. Even though doctors have told her that gastric cancer is not hereditary, she has always believed that it is in her family and was not surprised to learn of the CDH1 mutation. She does not undergo regular cancer screening but has recently become interested in gastric cancer screening. She is aware that members of other families with HDGC have had prophylactic total gastrectomy, but for her this seems too extreme.
The proband’s mother (III-2), age 59, had been having yearly EGD screening for approximately 20 years and was recently diagnosed with Barrett’s esophagus. She had been seriously considering prophylactic total gastrectomy for the past 2–3 years. She chose to have predictive genetic testing and was found to have the CDH1 gene mutation. She stated that she will undergo total gastrectomy in the very near future. Because she has early satiety and epigastric pain, she believes that she may have DGC already and hopes that it will be caught early. Now that his mother’s results are known, the proband would like to have predictive genetic testing, and he is getting his insurance in place prior to testing.
Individual III-4 is a 68-year-old sister of the proband’s mother. She was diagnosed with lobular breast cancer at 58 years of age. Two of 29 lymph nodes were positive. She declined chemotherapy and has not undergone regular mammograms since being diagnosed with breast cancer. She was initially reluctant to participate in a family meeting but decided to do so when her family physician said it was a good thing to do. She tested positive for the CDH1 mutation. She also feels that prophylactic total gastrectomy is too extreme. Prior to an FIS, none of the family members knew of the association of lobular breast cancer and HDGC.
Individual IV-13 is a 48 year-old unaffected male. He has never had EGD. He tested positive for the CDH1 mutation. He received some information about screening and prevention of gastric cancer at the FIS and met with one of the co-authors (WG) for more detailed information about these options. He felt that prophylactic total gastrectomy would negatively impact his employment. He will undergo screening for gastric cancer and hopes that having a positive attitude about cancer and eating a healthy diet will make a positive impact.
Identification of the CDH1 mutation confirmed many family members’ beliefs about the hereditary nature of gastric cancer in their family. Six of 8 (75%) family members in direct contact with one of the co-authors (SW) had already had some form of gastric cancer screening. Six of 7 (86%) family members who attended the FIS chose to have predictive genetic testing and they all valued the opportunity to participate in CDH1 research.
Discussion
Several challenges are presented when counseling and clinically managing family members who carry a deleterious CDH1 mutation. For example, the age of onset of DGC in HDGC families with a CDH1 mutation may be extremely variable, even within families. Thus, while the average age of onset of DGC in HDGC is approximately 38 years, it can range from 16 to 82 years.8 The marked intrafamilial variability in age of onset in family 3 was a factor in the family’s general reluctance to undergo prophylactic gastrectomy. This wide variability in age of onset also poses a serious challenge for clinical geneticists and genetic counselors when discussing predictive genetic testing for family members who are in their teens. Given the young ages of onset that have been seen, it is not unreasonable to offer genetic testing to individuals in their late teens. It is important to realize that predictive testing in these at risk teens will raise complex medical, ethical, psychosocial and legal implications.18 It is therefore imperative that sensitive counseling be offered over several sessions by genetic counselors and clinical geneticists dealing with the family.
There remain a large number of progeny of the mutation positive and/or HDGC cancer affected parents (Figures 1–4) who are at increased cancer risk and who clearly need special attention. Our primary concern is to continue this work in counseling and educating those individuals who are at inordinately high risk for DGC so that they can make their own decisions about testing and, should they be positive for a CDH1 mutation, they may then consider the option of prophylactic total gastrectomy. Some who tested positive for the CDH1 mutation indicated that they may elect to delay prophylactic surgery, considering the disease’s incomplete penetrance, and hope that a diagnostic measure will be discovered that is of acceptable sensitivity and specificity so that their decision about surgical prophylaxis can be reconciled. Those who test negative can be relieved that the life-threatening burden of DGC has been reduced to a population-based risk of much less than 1%.
The choices for unaffected mutation carriers are screening programs which lack acceptable sensitivity vs. the option of prophylactic total gastrectomy.14 Decision for gastric surgical prophylaxis in a CDH1 mutation carrier must be made relatively early in life, since the mentioned average age of onset of DGC in HDGC is approximately 38 years.5,11 This decision may be heightened by patients’ knowledge that diagnostic procedures capable of providing early diagnosis are limited, given the submucosal presence of DGC. Further support in the decision-making process is the extremely poor prognosis when there is DGC with regional or distal spread.14 The high fatality rate among members of HDGC families provides a further stimulus to preventive surgery.
Decision Concerns
A patient’s decision to undergo prophylactic total gastrectomy is often an extremely difficult one, since the entire stomach must be sacrificed. For some, it may be equally difficult to accept that one can live a reasonably normal life in the absence of this organ. However, the surgical decision was eased in a major way for the mentioned 11 first cousins (Figure 1) because the majority had witnessed a parent die as a result of the progressive course of DGC. When told that there was an approximate 30% chance that they would not develop DGC, due to its reduced penetrance, this clearly factored into some of their decision making. Knowing, however, that they had the deleterious mutation, they had to resolve the cancer probabilities, which they clearly knew favored development of DGC. They knew through our educational program that available DGC screening procedures were wholly inadequate.14 Perhaps foremost on the positive side of their decision-making process was the psychological support provided to each other in the sense of a “group therapy” family commitment to help each other, as was so strongly evidenced in family 1.15 There was very strong solidarity among these cousins about the importance of prophylactic gastrectomy, and this influence of camaraderie was pervasive throughout our genetic counseling sessions dealing with these high-risk individuals.
Once armed with facts about the natural history of DGC, many told us that, in essence, the only decision left was when the prophylactic gastrectomy should be performed. Knowing that their prognosis would become extremely grave once DGC symptoms became manifest, all 11 first cousins in family 1 decided to undergo prophylactic surgery, most within a couple of years of receiving their CDH1 mutation results.
Prophylactic Gastrectomy and Reconstruction
Preoperative multidisciplinary evaluation includes genetic counseling, gastroenterology, dietary consultation, and surgical consultation. In our protocol established for a group of families with a recently described founder CDH1 mutation, patients are evaluated by the surgeon on at least three separate occasions in order that they understand the potential short- and long-term complications of prophylactic gastrectomy, the estimated predicted weight loss, and potential metabolic consequences.8 They are advised of the possible need for re-operation, anastomotic leak, and potential death. They are familiarized with a post-gastrectomy diet and informed that it may take up to one year for weight stabilization and accommodation to dietary modifications to take place. They are also advised that the estimated lifetime risk of gastric cancer may decline somewhat over time as more families are evaluated and that medical-genetic therapy may eventually become available for this condition.
Endoscopic evaluation is undertaken by the surgeon who will perform the surgery, with at least 15 random gastric mucosal biopsies provided for pathology. Delineation of the gastroesophageal junction and presence-absence of hiatal herniation is noted. If the latter is present, discussion of possible thoraco-abdominal approach to gastrectomy is undertaken. Colonoscopy is performed and abdominal CT is used to delineate possible extra-gastric extension or nodal involvement. Although a variety of approaches to prophylactic gastrectomy have been described including laparoscopic gastrectomy and the provision of a Hunt Lawrence pouch reconstruction, we have successfully performed gastrectomy with end-to-side esophago-jejunal reconstruction and feeding jejunostomy successfully in 17 patients with the 2398delC founder mutation (DW unpublished data). We are currently in the process of evaluation, including quality-of-life assessment, of a cohort of patients undergoing this procedure.
Limitation
An important limitation of our study was due to the lack of formal psychological evaluation. This might have provided a more full assessment of the emotional concerns and how these might have been ameliorated through psychological/psychiatric consultation. This limitation becomes of particular importance when the at-risk patient receives a positive CDH1 mutation result, knows the gravity of DGC, and must wrestle with the decision to undergo prophylactic total gastrectomy. It will be important to conduct formal behavioral studies in HDGC families to better understand the determinants of their medical decision making and to help them cope with the arduous decisions at hand.
Acknowledgments
Trudy Shaw, M.A., provided technical assistance throughout the development of this manuscript. We gratefully acknowledge the dedication, help, and inspiration provided by the HDGC family members who participated in this study.
Financial disclosures and funding considerations: This study was supported by revenue from Nebraska cigarette taxes awarded to Creighton University by the Nebraska Department of Health and Human Services. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the State of Nebraska or the Nebraska Department of Health and Human Services. Support was also given by the National Institutes of Health through grant #1U01 CA 86389. Dr. Henry Lynch’s work is partially funded through the Charles F. and Mary C. Heider Chair in Cancer Research, which he holds at Creighton University.
References
- 1.Alberts SR, Cervantes A, van De Velde CJ. Gastric cancer: epidemiology, pathology and treatment. Ann Oncol. 2003;14(Suppl 2):ii31–ii36. doi: 10.1093/annonc/mdg726. [DOI] [PubMed] [Google Scholar]
- 2.Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma: An attempt at a histoclinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49. doi: 10.1111/apm.1965.64.1.31. [DOI] [PubMed] [Google Scholar]
- 3.Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12:354–362. doi: 10.3748/wjg.v12.i3.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guilford P, Hopkins J, Harraway J, et al. E-cadherin germline mutations in familial gastric cancer. Nature. 1998;392:402–405. doi: 10.1038/32918. [DOI] [PubMed] [Google Scholar]
- 5.Lynch HT, Grady W, Suriano G, Huntsman D. Gastric cancer: New genetic developments. J Surg Oncol. 2005;90:114–133. doi: 10.1002/jso.20214. [DOI] [PubMed] [Google Scholar]
- 6.Suriano G, Yew S, Ferreira P, et al. Characterization of a recurrent germ line mutation of the E-cadherin gene: implications for genetic testing and clinical management. Clin Cancer Res. 2005;11:5401–5409. doi: 10.1158/1078-0432.CCR-05-0247. [DOI] [PubMed] [Google Scholar]
- 7.Suriano G, Oliveira C, Ferreira P, et al. Identification of CDH1 germline missense mutations associated with functional inactivation of the E-cadherin protein in young gastric cancer probands. Hum Mol Genet. 2003;12:575–582. doi: 10.1093/hmg/ddg048. [DOI] [PubMed] [Google Scholar]
- 8.Kaurah P, MacMillan A, Boyd N, et al. Founder and recurrent CDH1 mutations in families with hereditary diffuse gastric cancer. JAMA. 2007;297:2360–2372. doi: 10.1001/jama.297.21.2360. [DOI] [PubMed] [Google Scholar]
- 9.Keller G, Vogelsang H, Becker I, et al. Diffuse type gastric and lobular breast carcinoma in a familial gastric cancer patient with an E-cadherin germline mutation. Am J Pathol. 1999;155:337–342. doi: 10.1016/S0002-9440(10)65129-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Oliveira C, Seruca R, Caldas C. Genetic screening for hereditary diffuse gastric cancer. Expert Rev Mol Diagn. 2003;3:201–215. doi: 10.1586/14737159.3.2.201. [DOI] [PubMed] [Google Scholar]
- 11.Caldas C, Carneiro F, Lynch HT, et al. Familial gastric cancer: overview and guidelines for management. J Med Genet. 1999;36:873–880. [PMC free article] [PubMed] [Google Scholar]
- 12.Bacani JT, Soares M, Zwingerman R, et al. CDH1/E-cadherin germline mutations in early onset gastric cancer. J Med Genet. 2006;43:867–872. doi: 10.1136/jmg.2006.043133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Norton JA, Ham CM, Van Dam J, et al. CDH1 truncating mutations in the E-cadherin gene: an indication for total gastrectomy to treat hereditary diffuse gastric cancer. Ann Surg. 2007;245:873–879. doi: 10.1097/01.sla.0000254370.29893.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lynch HT, Grady W, Lynch JF, Tsuchiya KD, Wiesner G, Markowitz SD. E-cadherin mutation-based genetic counseling and hereditary diffuse gastric carcinoma. Cancer Genet Cytogenet. 2000;122:1–6. doi: 10.1016/s0165-4608(00)00273-9. [DOI] [PubMed] [Google Scholar]
- 15.Lynch HT. Family Information Service and hereditary cancer. Cancer. 2001;91:625–628. doi: 10.1002/1097-0142(20010215)91:4<625::aid-cncr1044>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 16.Chung DC, Yoon SS, Lauwers GY, Patel D. Case 22–2007: A woman with a family history of gastric and breast cancer. N Engl J Med. 2007;357:283–291. doi: 10.1056/NEJMcpc079016. [DOI] [PubMed] [Google Scholar]
- 17.Brooks-Wilson AR, Kaurah P, Suriano G, et al. Germline E-cadherin mutations in hereditary diffuse gastric cancer: assessment of 42 new families and review of genetic screening criteria. J Med Genet. 2004;41:508–517. doi: 10.1136/jmg.2004.018275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.MacDonald DJ, Lessick M. Hereditary cancers in children and ethical and psychosocial implications. J Pediatr Nurs. 2000;15:217–225. doi: 10.1053/jpdn.2000.8044. [DOI] [PubMed] [Google Scholar]


