Abstract
Dry eye disease (DED), an inflammatory autoimmune disorder affecting the ocular surface, degrades visual performance and the quality of life of more than 10 million people in the United States alone. The primary limitation in the effective treatment of DED is an incomplete understanding of its specific cellular and molecular pathogenic elements. Using a validated mouse model of DED, herein we functionally characterize the different T cell subsets, including regulatory T cells (Tregs) and pathogenic effector T cells, and determine their contribution to the pathogenesis of DED. Our data demonstrate the presence of dysfunctional Tregs and the resistance of pathogenic T cells, particularly Th17 cells, to Treg suppression in DED. In addition, we clearly show that in vivo blockade of IL-17 significantly reduces the severity and progression of disease, which is paralleled by a reduction in the expansion of Th17 cells and restoration of Treg function. Our findings elucidate involvement of a previously unknown pathogenic T cell subset (Th17) in DED which is associated specifically with Treg dysfunction and disease pathogenesis, and suggest a new target for dry eye therapy.
Keywords: Regulatory T cell, Th17 cell, Autoimmunity, Cornea, Dry eye
INTRODUCTION
Dry eye disease (DED) is a complex multifactorial autoimmune disorder with a largely unknown pathogenesis. The disease is characterized by an immune and inflammatory process that affects the ocular surface, and in severe cases may lead to blindness due to corneal ulceration or scarring (1-3). It has been estimated that over 9 million Americans suffer from DED, and tens of millions more have less severe symptoms that are notable during exposure to adverse predisposing factors such as dry air (4-6).
The ocular surface inflammation in DED is sustained by ongoing activation and infiltration of pathogenic immune cells, primarily of the CD4+ T cell compartment (1-3). The contribution of T cells to the immunopathogenesis of DED is supported by many observations, including: (i) increased T cell infiltration of the conjunctiva in both clinical and experimental dry eye (7,8); (ii) presence of IFNγ+ T cells in the conjunctiva of dry eye mice; and (iii) induction of disease in nude mice by transfer of CD4+ T cells from dry eye mice (9). In addition, topical application of T cell immunosuppressants, such as cyclosporine (10) or agents inhibiting the pathogenic T cell recruitment to the ocular surface such as α4β1-integrin antagonist (11), lead to disease remission.
Despite this strong evidence for T cell involvement in the pathogenesis of dry eye, the majority of studies are focused primarily on the effector-T cell responses. The concept that inefficient functioning of regulatory-T cells may cause unrestrained generation of pathogenic T cells in DED has not yet been systematically investigated. A functional balance between regulatory- and effector-T cells is important for maintaining efficient immune responses while avoiding autoimmunity. Foxp3 expressing CD4+CD25+ T regulatory cells (Tregs) are a naturally occurring suppressor T-cell population that inhibits the activity of self-reactive T helper (Th)-1 (IFNγ+), Th-2 (IL-4+) and Th-17 (IL-17+, also known as IL-17A) cells which potentially induce inflammation and tissue destruction (12-15). Here we explored a well characterized mouse model of DED (16) and tested the hypothesis that defective Treg homeostasis leads to a dysregulated activation of self-reactive T cells. Our data demonstrate the presence of dysfunctional Tregs and the resistance of pathogenic T cells, particularly Th17 cells, to Treg suppression in DED. In addition, we investigated whether in vivo blockade of IL-17 would ameliorate the disease and prevent both Th17 cell differentiation and loss of Treg function.
MATERIALS AND METHODS
Mice
Six to 8 week-old female C57Bl/6 mice (Taconic Farms, and Charles River Lab) were used in these experiments. The protocol was approved by the Institutional Animal Care and Use Committee, and all animals were treated according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
Induction of dry eye
Dry eye disease (DED) was induced by exposure of mice to the Controlled-Environment Chamber (CEC) for nine days during which mice also received administration of scopolamine to maximize ocular dryness as described previously (16,17). Briefly, 0.5mg/0.2ml scopolamine hydrobromide (Sigma-Aldrich) was injected subcutaneously in the dorsal skin of mice four times daily. The mice placed in the CEC were continuously exposed to a relative humidity below 30%, airflow of 15 l/min and a constant temperature (21-23°C). Age and sex matched mice raised in the standard environment of the vivarium were used as normal controls. Induction of dry eye was confirmed by measuring changes in the corneal integrity using corneal fluorescein staining. One μl of 5% fluorescein was applied to the lateral conjunctival sac of the mice and eyes were examined for fluorescein staining with a slit lamp biomicroscope under cobalt blue light. Punctate staining was recorded in a masked fashion using the standard National Eye Institute grading system giving a score from 0 to 3 for each of the five areas of the cornea (18).
Cell isolation
Draining submandibular and cervical lymph nodes (LN) were harvested from normal and DED mice, and single-cell suspensions were prepared. Total CD4+ T cells and CD4+CD25+ Tregs were isolated by magnetic separation using CD4 T cell and regulatory T cell isolation kits (Miltenyi Biotec). Purity of sorted cells was > 95%, and 97% of CD4+CD25+ Tregs were Foxp3+ in all the groups as confirmed by FACS analysis.
FACS analysis
For Treg frequencies, LN cells were stained with the anti-CD4 FITC, anti-CD25 PE and anti-FoxP3 PECy5. For intracellular cytokine detection, cells were first stimulated with PMA+ionomycin (Sigma-Aldrich) for 6 hr in the presence of Golgi-stop (BD Bioscience) and then stained with anti-IL-17A PE or anti-IFNγ PE and anti-CD4 FITC. All the antibodies with their matched isotype controls and fix-perm buffer were purchased from eBioscience Inc. Stained cells were analyzed on an EPICS XL flow cytometer (Beckman Coulter).
Suppression assay
For all the suppression assays including intracellular cytokine analysis of proliferating cells, Tregs and responder CD4+ T (Tresp) cells were isolated from the draining LN. Purified total CD4+ Tresp cells (1×105) were cocultured with CD4+CD25+ Tregs (5×104), T cell-depleted syngeneic splenocytes (1×105) and 1 μg/ml anti-CD3 antibody for 3 days. Proliferation of CD3 stimulated Tresp cells without adding Tregs was considered as control proliferation with 0% suppression. Proliferation was measured using the BrdU proliferation kit (Millipore) and percent suppression was calculated using the following formula: % suppression = [(Tresp proliferation without Tregs – Tresp proliferation with Tregs) / Tresp proliferation without Tregs] × 100.
Real-time PCR
Draining LN, cornea and conjunctiva of normal and dry eye mice were harvested. RNA was isolated with RNeasy Micro Kit (Qiagen) and reverse transcribed using Superscript III Kit (Invitrogen Life Technologies). Real-time PCR was performed using Taqman Universal PCR Mastermix and preformulated primers for FoxP3 (assay ID: Mm00475156_m1), IL-17A (Mm00439619_m1), IL-6 (Mm00446190_m1), IL-17RA (Mm00434214_m1) and GAPDH (Mm99999915_g1) (Applied Biosystems). The results were analyzed by the comparative threshold cycle method and normalized by GAPDH as an internal control.
Immunohistochemical staining
Acetone fixed cryo-sections of whole eyeballs were blocked with 2% BSA and anti-FcR antibody (eBioscience Inc.). The sections were immunostained with primary rabbit anti-mouse IL-17RA or isotype-matched control antibodies (Santa Cruz Biotechnology, Inc.) overnight at 4°C, and then with rhodamine conjugated secondary antibody (Jackson Laboratories) for 1 hr at room temperature. The stained cross-sections were mounted using Vector Shield DAPI mounting medium (Vector Laboratories) and examined under the epifluorescence microscope (Nikon).
In vivo IL-17 neutralization
Lyophilized rat anti-mouse IL-17 and isotype monoclonal antibodies (R&D Systems) were resuspended in PBS to yield a concentration of 100μg/100μl. Anti-IL-17 or isotype antibody was administered to mice by intraperitoneal injections at 1 day before (200μg/mouse) placing them in to controlled-environment chamber and then at Day 1, Day 3 and Day 5 (100μg/mouse). Corneal fluorescein staining was performed daily to score the disease. At Day 6 mice were euthanized, and the draining LN and conjunctiva were harvested for further analyses.
Statistical analysis
Student’s t test was used for comparison of mean between the groups and p-value < 0.05 was considered significant.
RESULTS
Defect in Tregs is qualitative, not quantitative
Regional draining lymph nodes (LN) were collected from dry eye (DED) and normal mice. Single cell suspensions prepared from the LN were subjected to triple-color FACS analysis for CD4+ CD25+ Foxp3+ Tregs and to immunomagnetic sorting for isolation of Tregs. We did not observe significant differences in the frequencies of Tregs either as a proportion of total CD4+ cells (range: 9 - 11%) or of the total LN cells (range: 3 - 4%) between normal and DED mice (Fig. 1a and 1b). Using an in-vitro co-culture suppression assay system, we next compared the regulatory potential of purified Tregs to suppress the proliferation of CD3-antibody stimulated naïve-T cells (isolated from the LN of normal mice) and primed-T cells (isolated from the LN of DED mice) (Fig. 1c). Tregs isolated from DED mice showed significantly reduced potential in suppressing proliferation of both naïve-T cells (p = 0.015) and primed-T cells (p = 0.008) compared to those from normal mice. In addition, Tregs of DED mice were significantly less effective in suppressing the proliferation of primed-T cells than those of naïve-T cells (p = 0.041). However, Tregs of normal-mice were found to be equally effective in suppressing the proliferation of both the naïve- and primed T cells. These results clearly suggest a defect in Treg function, not in their numbers, in mice with dry eye disease.
Figure 1. Frequencies and function of Tregs.
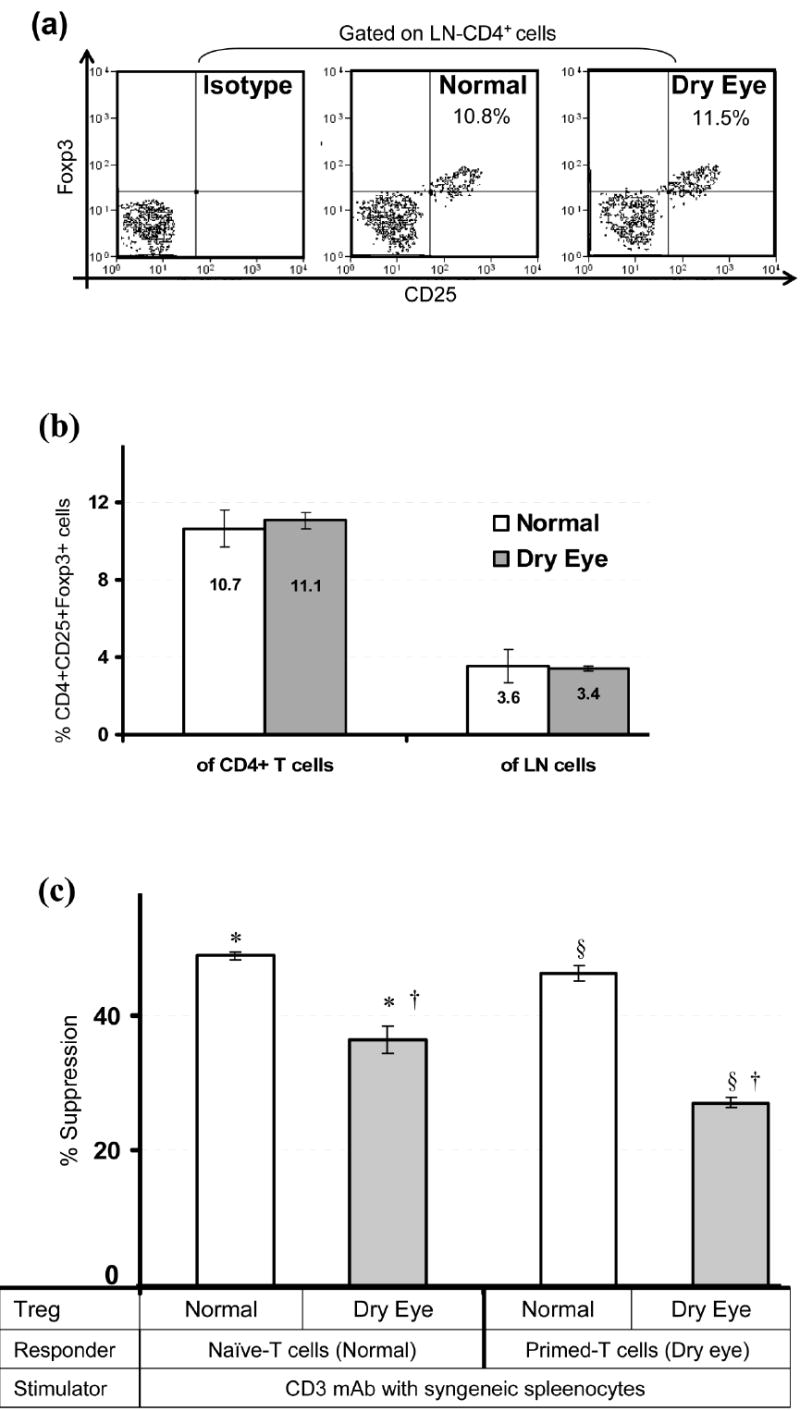
Regional draining lymph nodes (LN) were harvested from normal and dry eye mice and analyzed for CD4+CD25+Foxp3+ Treg frequency and function. (a) Representative flow cytometric dot plots showing frequencies of Tregs. (b) Frequencies of CD4+CD25+Foxp3+ Tregs either as a proportion of total CD4+ T cells or of the total LN cells as analyzed by FACS. (c) In-vitro Treg suppression assay; naïve- or primed-T cells isolated from the LN of normal and dry eye mice respectively, were stimulated with CD3-antibody for 3 days in the presence of Tregs isolated from the LN of normal or dry eye mice. The activity of Tregs is measured at Treg:Teff cell ratio of 1:2 as standardized in prior experiments. Proliferation was measured using the BrdU incorporation assay and compared with the proliferative responses of respective CD3-stimulated T cells in the absence of Tregs and the % suppression was calculated as described in Materials and Methods. Each group consists of 4-6 mice and data from a representative experiment of three performed is shown. P values are calculated using student’s t-test and error bars represent SEM. *p = 0.015; §p = 0.008; †p = 0.041.
Inefficiency of Tregs to suppress Th17 cells
To determine the subsets of primed-T cells which are resistant to Treg suppression we analyzed intracellular cytokine by FACS in proliferating subsets of primed-T cells (isolated from the LN of DED mice) co-cultured with Tregs of normal and DED mice (Fig. 2). The main proliferating subset of primed-T cells in the presence of Tregs of DED mice was found to be IL-17-producing CD4+T cells, which proliferate about 2.5 times more avidly as compared to their proliferation in the presence of Tregs of normal mice. A modest increase in IFN-γ producing CD4+T cell proliferation was also observed in the presence of Tregs of DED mice than those of normal mice. However, no significant difference was observed in the percentages of any of the subsets in proliferating naïve-T cell (isolated from the LN of normal mice) in the presence of Tregs from either normal or DED mice (data not shown).
Figure 2. Resistance of T cells to Treg suppression.
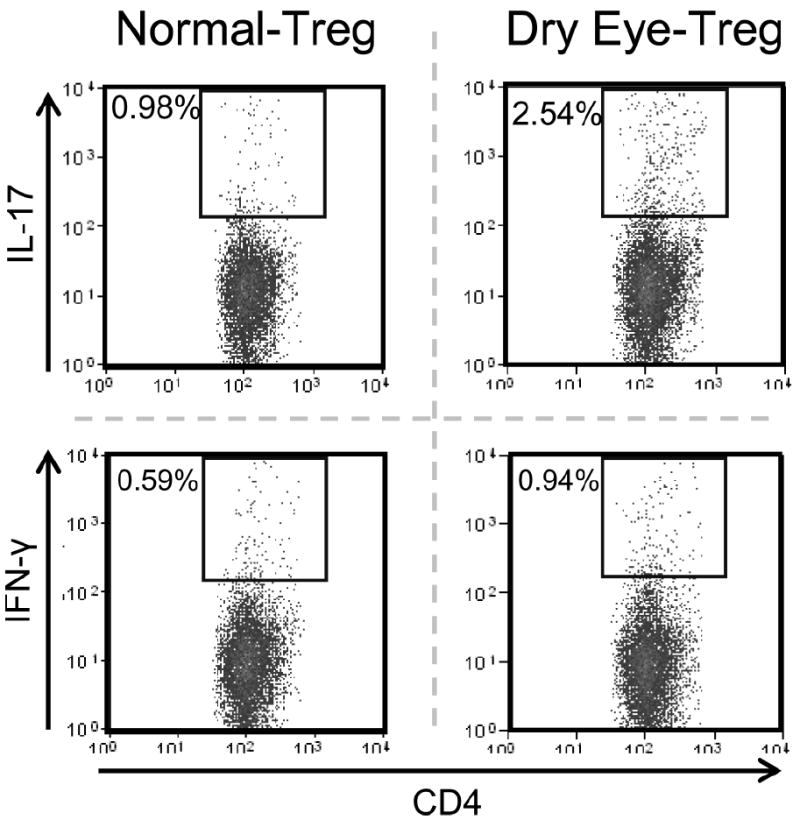
Primed-T cells isolated from the draining LN of dry eye mice were stimulated with CD3-antibody in the presence of Tregs isolated from the draining LN of normal or dry eye mice. After 3 days of co-culture, cells were washed and activated with PMA+ionomycin for 6 hr in the presence of golgi-plug. The proliferating subsets of primed-T cells were analyzed for intracellular IL-17A and IFN-γ by FACS. Data from a representative experiment of three performed is shown and each group consists of 4-6 mice.
Presence of Th17 profile in dry eye disease
To further confirm the in vivo presence of Th17 cells in DED mice, we examined both the LN and conjunctiva for IL-17 and its receptor (IL-17RA) along with IL-6 and Foxp3 expression using FACS and real-time PCR. The LN of DED mice showed a significant increase (4-5 fold) in the frequency of IL-17+CD4+ T cells compared to those of normal mice (p = 0.026) (Fig. 3a). Similarly, real time-PCR analysis confirmed an increase in the expression levels of IL-17 (2-3 fold) and IL-6 (1.2-1.5 fold) mRNA in the LN of DED mice compared to normal mice. However, a minor decrease in the Foxp3 mRNA levels was observed in the LN of DED mice compared to those of normal mice (Fig. 3b). As observed in the LN, a similar trend was found in the mRNA expression levels of IL-17, IL-6 and Foxp3 in the conjunctiva of DED mice (Fig. 3c). In addition, we investigated the presence of IL-17 receptors on the ocular surface by immunostaining and real-time PCR. Immunostaining performed on cross sections of eye using anti-IL-17RA-antibody showed that corneal and conjunctival epithelia of both normal and DED mice express IL-17RA (Fig. 3d). Real-time-PCR analysis for IL-17RA mRNA expression in cornea and conjunctiva showed no significant difference in the expression levels of IL-17 receptors in both the groups (Fig. 3e).
Figure 3. Detection of Th17 cells and IL-17 receptor.
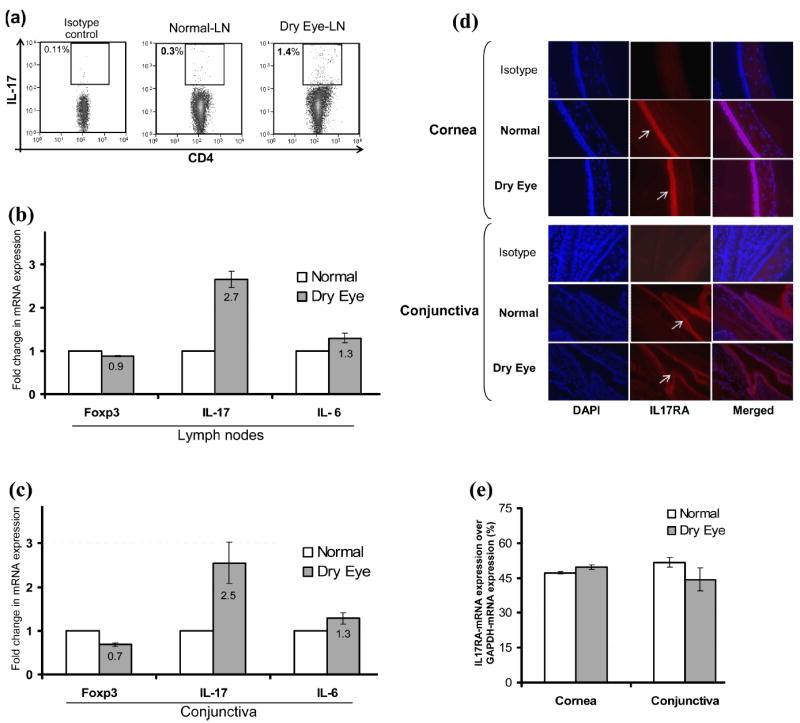
(a) Draining LN cells of normal and dry eye mice were stimulated with PMA+ionomycin for 6 hr and analyzed for intracellular IL-17A in CD4+ T cell population by flow cytometry (p = 0.026). (b) Real-time PCR analysis of the draining LN and (c) conjunctiva of normal and dry eye mice showing expression levels of IL-17A, IL-6 and Foxp3 mRNAs. (d) Representative micrograph of immunostaining of cross sections of eye showing expression of IL-17 receptor (IL-17RA) by the corneal and conjunctival epithelia of normal and dry eye mice. (e) Real-time PCR analysis showing IL-17RA mRNA expression levels in the cornea and conjunctiva of normal and dry eye mice.
Diminished severity and progression of disease by anti-IL-17 therapy
We next studied the effects of in-vivo neutralization of IL-17 by anti-IL-17 antibody on the clinical signs of dry eye disease. A schematic diagram of the experimental design is shown in Fig. 4a. Corneal fluorescein staining scores, a readout for the principal clinical sign of dry eye inflammation, was significantly lower in anti-IL-17 antibody-treated mice compared to isotype antibody-treated and untreated mice during both the induction (day 2) and the progression phase of the disease (Fig. 4b).
Figure 4. Effects of in-vivo blockade of IL-17 on severity and progression of disease.
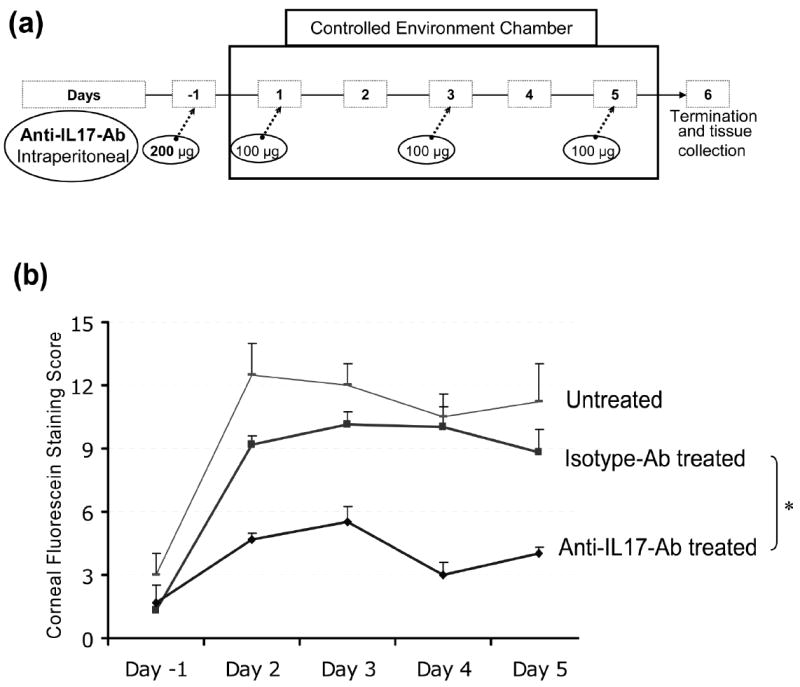
(a) Schematic diagram of experimental design to study the effects of in-vivo IL-17 blockade using intraperitoneal injections of anti-IL17-antibody or isotype-antibody on clinical signs and pathogenesis of dry eye disease in mice. (b) Corneal fluorescein staining scores, a read out for clinical signs of dry eye inflammation, in anti-IL-17 antibody-treated, isotype antibody-treated and untreated groups (n=5/group) from Day -1 (base line) to Day 5. P values are calculated using student’s t-test and error bars represent SEM. (* p-values between anti-IL17 antibody-treated versus isotype antibody-treated groups at different time points; day -1, 0.842; day 2, 0.001; day 3, 0.008; day 4, 0.002; day 5, 0.021).
Reversal of Th17 cell frequency and Treg function by anti-IL-17 therapy
In addition to the clinical signs of DED, we analyzed the effects of IL-17 blockade on Th17 cell frequency and Treg function. The LN of anti-IL-17 antibody-treated mice showed a more than three-fold decrease in the frequency of IL-17+CD4+ T cells compared to that of isotype antibody-treated mice (Fig. 5a). Similarly, an approximately two-fold decrease in the expression levels of IL-17 mRNA in the conjunctiva of anti-IL-17 antibody-treated mice was observed compared to those of isotype antibody-treated mice (Fig. 5b). The in vitro Treg suppression assay using CD3 stimulated primed-T cells (isolated from the LN of dry eye mice) and Tregs (isolated from the LN of mice treated with anti-IL-17 or isotype antibodies) showed a significant recovery in the suppressor potential of Tregs only in mice treated with anti-IL-17 antibody compared to those isolated from the isotype antibody treated groups (p = 0.029) (Fig. 5c). These results clearly suggest an inverse relationship between Th17 cells and Treg function in DED.
Figure 5. Effects of in-vivo blockade of IL-17 on Th17 and Tregs.
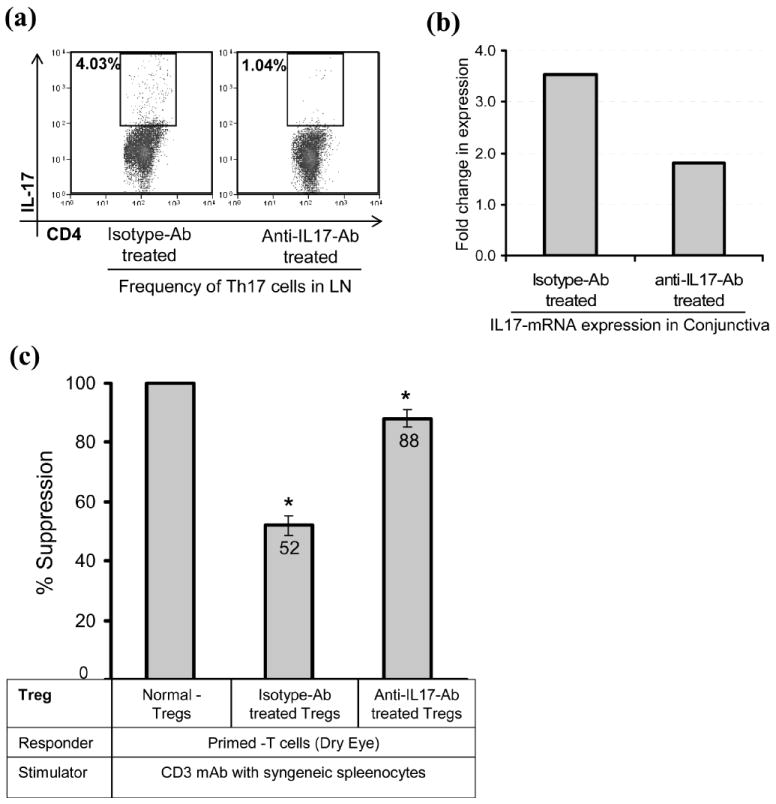
(a) Frequencies of IL-17 producing CD4+ T cells in the draining LN of anti-IL17 and isotype antibody-treated mice were analyzed by FACS after stimulation of LN cells with PMA+Ionomycin for 6 hr. (b) Real-time PCR analysis showing IL-17 mRNA expression in the conjunctiva of isotype and anti-IL17 antibody-treated mice. (c) Suppressor potential of Tregs isolated from the draining LN of isotype and anti-IL17 antibody-treated groups to inhibit the proliferation of CD3 antibody stimulated primed-T cells isolated from the LN of dry eye mice (*p = 0.029). The suppressor potential of Tregs isolated from different groups is calculated in relation to the suppression potential of Tregs of normal mice, considered as 100%. Each experiment consists of 5 mice per group; P values are calculated using student’s t-test and error bars represent SEM.
DISCUSSION
The demonstration of impaired Treg function and pathogenic Th17 cells in DED are novel findings which extend our understanding of immunopathogenesis of the disease. Specifically, we show herein that CD4+CD25+Foxp3+ Tregs are functionally incompetent in downregulating the activation of pathogenic T cells in DED, and in vivo blockade of IL-17 significantly reduces the severity and progression of disease as well as markedly prevents the loss of Treg function.
Tregs play a central role in maintaining peripheral tolerance and controlling organ-specific autoimmunity by suppressing pathogenic autoreactive T cells (12,13). A defective Treg homeostasis has previously been associated with the pathogenesis of several autoimmune diseases such as experimental autoimmune encephalomyelitis, multiple sclerosis, and rheumatoid arthritis (19-21). However, data on the role of Tregs in DED are very limited, and their homeostasis in disease condition has not been investigated. There is only one report in which authors showed that adoptive transfer of CD4+T cells from DED mice induces more severe disease in Treg deficient-mice compared to normal mice (9). Thus, until now, it has remained unclear whether the exacerbation of the disease is merely due to the absence of normal Tregs, or a functional Treg defect. In the present study, we have analyzed Tregs both quantitatively and qualitatively. Our data demonstrate that Treg frequencies remain unchanged, but their potential to suppress the activation of effector T cells is markedly decreased in DED.
Previously, involvement of Th1 cells has been demonstrated in the pathogenesis of DED (9). In the current study, we show a second major Th subset (Th17) which is associated specifically with the Treg dysfunction and pathogenesis of DED. Many of the classic Th1-mediated autoimmune diseases are now also being associated with Th17 cells, suggesting that autoimmune disorders are not necessarily restricted to just one type of immune response (14-15). Our data demonstrate that DED-Tregs are relatively more inefficient in suppressing the activation of DED-T effectors than that of naïve-T cells. These findings prompted us to analyze the proliferating subsets of T effectors. Interestingly, the main proliferating subset of DED-T effectors in the presence of Tregs is found to be IL-17 secreting CD4+ T cells, confirming previous reports which suggest that in contrast to Th1, Th17 cells may not be suppressed by Tregs (22-24).
IL-17 is known to induce epithelial, stromal, and immune cells to secrete proinflammatory cytokines such as IL-6, TNF-α, IL-1 and IL-8 (15). Indeed, these cytokines have also been shown to be increased in the DED (25,26), and along with our demonstration of Th17 cells and expression of IL-17R by the ocular surface epithelia, point toward the importance of an IL-17-mediated cytokine cascade in the immunopathogenesis of DED. The constitutive expression of IL-17 receptors by corneal and conjunctival epithelia makes ocular surface more vulnerable to IL-17 mediated inflammation and tissue damage. The presence of proinflammatory cytokines in the milieu may actually favor the generation of autoreactive T cells. For instance, IL-6 in the presence of TGF-β leads to the differentiation of autoreactive Th17 cells which can restrain Treg function and favor autoreactivity (27). Recently, it has also been reported that Tregs can be self induced to become Th17 cells in the presence of IL-6 (28). However, our data do not demonstrate any decrease in Treg frequency in DED suggesting that generation of Th17 cells in DED mice is primarily not due to the self conversion of Tregs into Th17 cells.
The functional relevance of Th17 cells in the immunopathogenesis of DED is fundamentally confirmed by the finding that in vivo neutralization of IL-17 results in a markedly attenuated induction and severity of disease, which is paralleled by a reduction in the expansion of Th17 cells. Remission of clinical disease by IL-17 blockade has also been reported in other Th17 associated autoimmune disorders (29,30) but this is the first in vivo demonstration that blockade of IL-17 not only inhibits the generation of Th17 cells but importantly also notably prevents the loss of Treg function. Previous studies have demonstrated that anti-IL-17 treatment can directly inhibit the T cell expansion and IL-6 production by IL-17-target (epithelial, stromal and immune cells) cells (31). IL-6, which controls a reciprocal relationship in the development of Tregs and Th17 cells, inhibits Treg function and facilitates Th17 cell expansion (27). Together, these findings suggest that Th17 cell secreted IL-17 antagonizes the Treg function which in turn may further unleash Th17 and Th1 cells to expand, release inflammatory cytokines and cause pathological damage.
In summary, these findings provide new insights on the role of both regulatory and effector subsets of T cells in the pathogenesis of DED. The induction of Th17 cells and the lack of Treg control over pathogenic T-effectors, in combination, lead to the induction of autoimmunity and sustained inflammation in DED. The demonstration that IL-17 blockade leads to diminished disease severity suggests potential novel approaches for the treatment of dry eye disease. Strategies designed to target IL-17 or inhibit Th17 cell function, and thereby favor Treg functionality, may be viable approaches for the treatment of dry eye.
Footnotes
This work was supported in part by NIH (NEI RO1-12963).
References
- 1.Barabino S, Dana MR. Dry eye syndromes. Chem Immunol Allergy. 2007;92:176–184. doi: 10.1159/000099268. [DOI] [PubMed] [Google Scholar]
- 2.Dana MR, Hamrah P. Role of immunity and inflammation in corneal and ocular surface disease associated with dry eye. Adv Exp Med Biol. 2002;506(Pt B):729–738. doi: 10.1007/978-1-4615-0717-8_103. [DOI] [PubMed] [Google Scholar]
- 3.Hemady R, Chu W, Foster CS. Keratoconjunctivitis sicca and corneal ulcers. Cornea. 1990;9:170–173. [PubMed] [Google Scholar]
- 4.Pflugfelder SC. Antiinflammatory therapy for dry eye. Am J Ophthalmol. 2004;137:337–342. doi: 10.1016/j.ajo.2003.10.036. [DOI] [PubMed] [Google Scholar]
- 5.The epidemiology of dry eye disease: Report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007) Ocul Surf. 2007;5:93–107. doi: 10.1016/s1542-0124(12)70082-4. [DOI] [PubMed] [Google Scholar]
- 6.Miljanović B, Dana R, Sullivan DA, Schaumberg DA. Impact of dry eye syndrome on vision-related quality of life. Am J Ophthalmol. 2007;143:409–415. doi: 10.1016/j.ajo.2006.11.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stern ME, Gao J, Schwalb TA, Ngo M, Tieu DD, Chan CC, Reis BL, Whitcup SM, Thompson D, Smith JA. Conjunctival T-cell subpopulations in Sjögren’s and non-Sjögren’s patients with dry eye. Invest Ophthalmol Vis Sci. 2002;43:2609–2614. [PubMed] [Google Scholar]
- 8.De Paiva CS, Villarreal AL, Corrales RM, Rahman HT, Chang VY, Farley WJ, Stern ME, Niederkorn JY, Li DQ, Pflugfelder SC. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Invest Ophthalmol Vis Sci. 2007;48:2553–2560. doi: 10.1167/iovs.07-0069. [DOI] [PubMed] [Google Scholar]
- 9.Niederkorn JY, Stern ME, Pflugfelder SC, De Paiva CS, Corrales RM, Gao J, Siemasko K. Desiccating stress induces T cell-mediated Sjögren’s Syndrome-like lacrimal keratoconjunctivitis. J Immunol. 2006;176:3950–3957. doi: 10.4049/jimmunol.176.7.3950. [DOI] [PubMed] [Google Scholar]
- 10.Stevenson D, Tauber J, Reis BL. Efficacy and safety of cyclosporin A ophthalmic emulsion in the treatment of moderate-to-severe dry eye disease: a dose-ranging, randomized trial. The Cyclosporin A Phase 2 Study Group. Ophthalmology. 2000;107:967–974. doi: 10.1016/s0161-6420(00)00035-x. [DOI] [PubMed] [Google Scholar]
- 11.Ecoiffier T, El Annan J, Rashid S, Schaumberg D, Dana R. Modulation of Integrin Alpha-4 Beta-1 (VLA-4) in Dry Eye Disease. Arch Ophthalmol. 2008 doi: 10.1001/archopht.126.12.1695. In Press. [DOI] [PubMed] [Google Scholar]
- 12.Sakaguchi S. Naturally arising Foxp3-expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat Immunol. 2005;6:345–352. doi: 10.1038/ni1178. [DOI] [PubMed] [Google Scholar]
- 13.Sakaguchi S, Sakaguchi N, Shimizu J, Yamazaki S, Sakihama T, Itoh M, Kuniyasu Y, Nomura T, Toda M, Takahashi T. Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol Rev. 2001;182:18–32. doi: 10.1034/j.1600-065x.2001.1820102.x. [DOI] [PubMed] [Google Scholar]
- 14.Steinman L. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat Med. 2007;13:139–145. doi: 10.1038/nm1551. [DOI] [PubMed] [Google Scholar]
- 15.McGeachy MJ, Cua DJ. Th17 cell differentiation: the long and winding road. Immunity. 2008;28:445–453. doi: 10.1016/j.immuni.2008.03.001. [DOI] [PubMed] [Google Scholar]
- 16.Barabino S, Shen L, Chen L, Rashid S, Rolando M, Dana MR. The controlled-environment chamber: a new mouse model of dry eye. Invest Ophthalmol Vis Sci. 2005;46:2766–2771. doi: 10.1167/iovs.04-1326. [DOI] [PubMed] [Google Scholar]
- 17.Rashid S, Jin Y, Ecoiffier T, Barabino S, Schaumberg DA, Dana MR. Topical omega-3 and omega-6 fatty acids for treatment of dry eye. Arch Ophthalmol. 2008;126:219–225. doi: 10.1001/archophthalmol.2007.61. [DOI] [PubMed] [Google Scholar]
- 18.Lemp MA. Report of the National Eye Institute/Industry workshop on clinical trials in dry eyes. CLAO J. 1995;21:221–232. [PubMed] [Google Scholar]
- 19.Korn T, Anderson AC, Bettelli E, Oukka M. The dynamics of effector T cells and Foxp3+ regulatory T cells in the promotion and regulation of autoimmune encephalomyelitis. J Neuroimmunol. 2007;191:51–60. doi: 10.1016/j.jneuroim.2007.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med. 2004;199:971–979. doi: 10.1084/jem.20031579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ehrenstein MR, Evans JG, Singh A, Moore S, Warnes G, Isenberg DA, Mauri C. Compromised function of regulatory T cells in rheumatoid arthritis and reversal by anti-TNFalpha therapy. J Exp Med. 2004;200:277–285. doi: 10.1084/jem.20040165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Korn T, Reddy J, Gao W, Bettelli E, Awasthi A, Petersen TR, Bäckström BT, Sobel RA, Wucherpfennig KW, Strom TB, Oukka M, Kuchroo VK. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat Med. 2007;13:423–431. doi: 10.1038/nm1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nistala K, Moncrieffe H, Newton KR, Varsani H, Hunter P, Wedderburn LR. Interleukin-17-producing T cells are enriched in the joints of children with arthritis, but have a reciprocal relationship to regulatory T cell numbers. Arthritis Rheum. 2008;58:875–887. doi: 10.1002/art.23291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang XO, Nurieva R, Martinez GJ, Kang HS, Chung Y, Pappu BP, Shah B, Chang SH, Schluns KS, Watowich SS, Feng XH, Jetten AM, Dong C. Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity. 2008;29:44–56. doi: 10.1016/j.immuni.2008.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pflugfelder SC, Jones D, Ji Z, Afonso A, Monroy D. Altered cytokine balance in the tear fluid and conjunctiva of patients with Sjögren’s syndrome keratoconjunctivitis sicca. Curr Eye Res. 1999;19:201–211. doi: 10.1076/ceyr.19.3.201.5309. [DOI] [PubMed] [Google Scholar]
- 26.Solomon A, Dursun D, Liu Z, Xie Y, Macri A, Pflugfelder SC. Pro- and anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Invest Ophthalmol Vis Sci. 2001;42:2283–2292. [PubMed] [Google Scholar]
- 27.Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, Weiner HL, Kuchroo VK. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 28.Xu L, Kitani A, Fuss I, Strober W. Regulatory T cells induce CD4+CD25-Foxp3- T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-beta. J Immunol. 2007;178:6725–6729. doi: 10.4049/jimmunol.178.11.6725. [DOI] [PubMed] [Google Scholar]
- 29.Hofstetter HH, Ibrahim SM, Koczan D, Kruse N, Weishaupt A, Toyka KV, Gold R. Therapeutic efficacy of IL-17 neutralization in murine experimental autoimmune encephalomyelitis. Cell Immunol. 2005;237:123–130. doi: 10.1016/j.cellimm.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 30.Koenders MI, Lubberts E, Oppers-Walgreen B, van den Bersselaar L, Helsen MM, Di Padova FE, Boots AM, Gram H, Joosten LA, van den Berg WB. Blocking of interleukin-17 during reactivation of experimental arthritis prevents joint inflammation and bone erosion by decreasing RANKL and interleukin-1. Am J Pathol. 2005;167:141–149. doi: 10.1016/S0002-9440(10)62961-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yao Z, Fanslow WC, Seldin MF, Rousseau AM, Painter SL, Comeau MR, Cohen JI, Spriggs MK. Herpesvirus Saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. Immunity. 1995;3:811–821. doi: 10.1016/1074-7613(95)90070-5. [DOI] [PubMed] [Google Scholar]


