Abstract
Epithelial to mesenchymal transitions (EMT) have been suggested to be crucial during epithelial cancer cell invasion. However, in a three-dimensional “organotypic” invasion assay squamous cell carcinoma (SCC) cells that retain epithelial characteristics “hitch a ride” with carcinoma associated fibroblasts (CAFs) in order to collectively invade. Thus epithelial cancer cells can utilise the mesenchymal characteristics of CAFs without the need to undergo EMT themselves. This work provides new insight in cancer cell invasion and shows a new role for CAFs as a target for an anti-invasive therapy.
Key words: collective invasion, carcinoma associated fibroblast, extracellular matrix, matrix metalloproteinases, Rho
Cancer cell invasion and metastasis are the main causes of mortality in cancer patients. Understanding how cancer cells move and invade within the surrounding tissue is therefore a key issue. Stromal fibroblasts within a tumor play a crucial role in cancer cell proliferation, survival, angiogenesis as well as invasion (reviewed in ref. 1). In many cases stromal CAFs are able to produce a wide range of growth factors and cytokines that modulate tumor growth and invasion.2,3 Their influence in cancer cell invasion and metastasis can also be mediated through the production of MMP's that promote extra-cellular matrix degradation.4
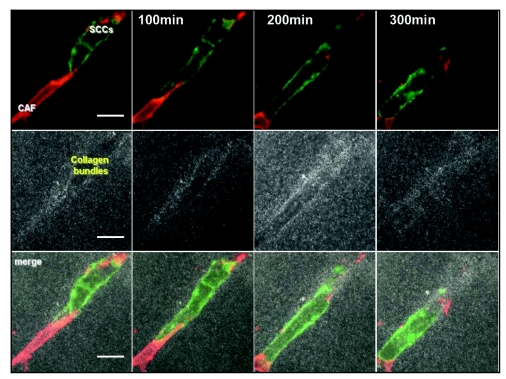
It has recently been shown that CAFs can play an unexpected role in SCC invasion.5 In a 3D ‘organotypic’ model of invasion that recreates the epidermal/dermal environment CAFs promote the collective invasion of SCC cells.6 3D time-lapse confocal microscopy imaging showed that CAFs were always the leading cell of the invading cohort with the SCC cells following behind. These cohorts closely resembled invading clusters of SCC cells observed in human cancer samples.7 CAFs promoted SCC cells collective invasion by remodelling the matrix and making a path that SCC cells can use to invade. This process is clearly shown in Figure 1: a CAF (in red) leads the invasion of a collective chain of SCC cells (green) and makes a path in the surrounding matrix, visualized in grey using confocal reflectance microscopy. Two key experiments helped to understand the role of fibroblasts in this system. Firstly, the separation of the two cell populations by a thin layer of gel without fibroblasts completely abolished SCC invasion and so ruled out the possibility of long distance chemoattractant molecules inducing SCC invasion. Secondly, SCC cells were able to invade into a gel which had previously been remodelled by CAFs that had subsequently been removed. Together these experiments showed that tracks made by the fibroblasts are essential and sufficient to promote collective carcinoma cells invasion. Heterotypic cell contact between both populations was not required, as SCC cells can invade using tracks made by the CAFs even if the CAFs have been removed.
Figure 1.
Collective invasion of carcinoma cells led by fibroblast. Confocal time-lapse imaging of carcinoma associated fibroblast (red) leading the way of an invading chain of SCC cells (green) and making path into the surrounding matrix (grey). Panel is 80 x 80 mm and spans 300 minutes, scale are 20 um.
Interestingly, inhibition of Rho/ROCK signalling to the actomyosin cytoskeleton or MMPs using small molecule inhibitors blocked SCC invasion even when only CAFs where targeted. Blocking these pathways in carcinoma cells had little or no effect on their invasion. Moreover, inhibition of Rho function specifically in CAFs did not block their invasion into matrices but prevented SCC cells from following. These experiments showed the role of Rho/ROCK and MMPs molecular pathways in track generation by the CAFs and that targeting these pathways in CAFs, but not SCC cells, is critical for preventing cancer invasion. Strikingly, blockade of protease function after CAFs had remodelled the ECM had little effect on the ability of SCC cells to invade. This could explain the relative poor results obtained using MMPs inhibitors as anti-invasive therapies.8 Rho/ROCK function was dispensable in SCC cells; however, depletion of the small GTPase Cdc42 and its effector MRCK disrupted the acto-myosin cortex of carcinoma cells and blocked their capacity to invade in response to CAFs.
In order to invade and metastasise, carcinoma cells can switch from an epithelial state to a more mesenchymal phenotype.9 This process, called EMT, allows epithelial cancer cells to adapt their behaviour and confers the capacity to remodel the ECM on the cancer cells.10 However, in patient tissue samples, it has been observed that carcinoma cells can invade without undergoing an EMT, these cancer cells do not upregulate mesenchymal markers and retain cell to cell contact during their invasion.11 This work explains how carcinoma cells that have not undergone EMT could invade a 3D matrix. These cells use the mesenchymal characteristics of the stromal fibroblasts to remodel the ECM and consequently follow behind invading fibroblasts. In tumours of mesenchymal origin CAFs are not required for invasion; work from Friedl and colleagues, clearly shows that HT1080 fibrosarcoma cells could lead collectively invading chains of cancer cells The authors showed how the leading cell of the collective chain remodels collagen fibres into tracks as it invades through the action of MT1-MMP (MMP14).12
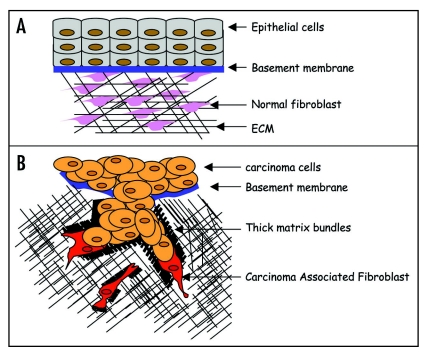
In normal conditions, epithelial cells and dermal fibroblasts are in complete homeostasis and separated by a basement membrane (Fig. 2A). In addition, normal dermal fibroblasts are unable to promote SCC invasion. Understanding how CAFs are activated will be an important step forward. A desmoplastic response is observed in many tumours indicating a change in behaviour of fibroblasts.13 During wound healing or fibrosis, fibroblasts are in an active state that has been suggested to be similar to cancer activation.14 TGFβ has been shown to be a key player in fibroblasts activation and could support cancer progression.15 However, TGFβ was not responsible for SCC cells invasion since a TGFβ inhibitor had no effect in carcinoma cells collective invasion induced by the CAFs in the 3D invasion assay (Cedric Gaggioli and Steven Hooper, unpublished data). Interestingly, a probe that binds only to the active form of the small GTPase Rho showed that the activity of this protein was increased in CAFs compared to normal fibroblasts in tissue samples. Elevated expression of α5 integrin was also present in these cells and this has been implicated in Rho activation in a number of systems.16–18 Consistent with this observation, depletion of integrin a5 in CAFs reduced their ability to promote the invasion of SCC cells. Alternatively, CAFs could also be derived from endothelial cells through a process called endothelial to mesenchymal transition19 (EndMT), or from cancer cells through EMT.20 These processes could be responsible for CAFs generation in the tumor stroma resulting in matrix remodelling and tracks generation in order for the carcinoma cells to collectively invade the surrounding tissue and metastasize (Fig. 2B).
Figure 2.
Model of carcinoma cells collective invasion. (A) Schematic representation of a normal epithelium. Epithelial cells (light blue) and normal fibroblasts (pink) are separated by a basal membrane and are in a perfect homeostasis. Cross talk between both cell types occurs through adhesion and chemokine secretion. (B) Schematic representation of carcinoma cells collective invasion. CAFs (red) take the lead of a collective invading chain of SCC cells (brown). Invasion of CAFs is MMPs dependent but Rho/ROCK independent. However, track generation by CAFs is Rho/ROCK/MLC dependent. SCC cells require the small GTPase Cdc42 and its effector MRCK in order to collectively invade trough those tracks (black).
This study opens a new field of investigation for collective cancer cell invasion. This work highlights carcinoma associated fibroblasts as new therapeutic targets which will be a new direction in cancer cell invasion and metastasis therapy.
Acknowledgements
Many thanks to S. Hooper, C. Hidalgo-Carcedo, S. Giamperi, S. Chaudhry, S. Pinner and E. Sahai for critical reading of this manuscript. This work was supported by Cancer Research UK and additional funds from Bettencourt-Schueller Fondation.
Abbreviations
- CAF
carcinoma associated fibroblast
- SCC
squamous cell carcinoma
- ROCK
Rho kinase
- MLC
myosin light chain
- MMPs
matrix metalloproteases
- MRCK
myotonic dystrophy kinase-related Cdc42-binding protein kinase
Footnotes
Previously published online as a Cell Adhesion & Migration E-publication: http://www.landesbioscience.com/journals/celladhesion/article/5705
References
- 1.Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6:392–401. doi: 10.1038/nrc1877. [DOI] [PubMed] [Google Scholar]
- 2.Bhowmick NA, Neilson EG, Moses HL. Stromal fibroblasts in cancer initiation and progression. Nature. 2004;432:332–337. doi: 10.1038/nature03096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 2005;121:335–348. doi: 10.1016/j.cell.2005.02.034. [DOI] [PubMed] [Google Scholar]
- 4.Rodemann HP, Muller GA. Characterization of human renal fibroblasts in health and disease: II. In vitro growth, differentiation, and collagen synthesis of fibroblasts from kidneys with interstitial fibrosis. Am J Kidney Dis. 1991;17:684–686. doi: 10.1016/s0272-6386(12)80352-0. [DOI] [PubMed] [Google Scholar]
- 5.Gaggioli C, Hooper S, Hidalgo-Carcedo C, Grosse R, Marshall JF, Harrington K, Sahai E. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat Cell Biol. 2007;9:1392–1400. doi: 10.1038/ncb1658. [DOI] [PubMed] [Google Scholar]
- 6.Nystrom ML, Thomas GJ, Stone M, Mackenzie IC, Hart IR, Marshall JF. Development of a quantitative method to analyse tumour cell invasion in organotypic culture. J Pathol. 2005;205:468–475. doi: 10.1002/path.1716. [DOI] [PubMed] [Google Scholar]
- 7.Yamamoto E, Kohama G, Sunakawa H, Iwai M, Hiratsuka H. Mode of invasion, bleomycin sensitivity, and clinical course in squamous cell carcinoma of the oral cavity. Cancer. 1983;51:2175–2180. doi: 10.1002/1097-0142(19830615)51:12<2175::aid-cncr2820511205>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- 8.Overall CM, Kleifeld O. Tumour microenvironment-opinion: validating matrix metalloproteinases as drug targets and anti-targets for cancer therapy. Nat Rev Cancer. 2006;6:227–239. doi: 10.1038/nrc1821. [DOI] [PubMed] [Google Scholar]
- 9.Gotzmann J, Mikula M, Eger A, Schulte-Hermann R, Foisner R, Beug H, Mikulits W. Molecular aspects of epithelial cell plasticity: implications for local tumor invasion and metastasis. Mutat Res. 2004;566:9–20. doi: 10.1016/s1383-5742(03)00033-4. [DOI] [PubMed] [Google Scholar]
- 10.Tse JC, Kalluri R. Mechanisms of metastasis: epithelial-to-mesenchymal transition and contribution of tumor microenvironment. J Cell Biochem. 2007;101:816–829. doi: 10.1002/jcb.21215. [DOI] [PubMed] [Google Scholar]
- 11.Macpherson IR, Hooper S, Serrels A, McGarry L, Ozanne BW, Harrington K, Frame MC, Sahai E, Brunton VG. p120-catenin is required for the collective invasion of squamous cell carcinoma cells via a phosphorylation-independent mechanism. Oncogene. 2007;26:5214–5228. doi: 10.1038/sj.onc.1210334. [DOI] [PubMed] [Google Scholar]
- 12.Wolf K, Wu YI, Liu Y, Geiger J, Tam E, Overall C, Stack MS, Friedl P. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol. 2007;9:893–904. doi: 10.1038/ncb1616. [DOI] [PubMed] [Google Scholar]
- 13.Walker RA. The complexities of breast cancer desmoplasia. Breast Cancer Res. 2001;3:143–145. doi: 10.1186/bcr287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ronnov-Jessen L, Petersen OW. Induction of alpha-smooth muscle actin by transforming growth factor-beta 1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasia. Lab Invest. 1993;68:696–707. [PubMed] [Google Scholar]
- 15.Bhowmick NA, Chytil A, Plieth D, Gorska AE, Dumont N, Shappell S, Washington MK, Neilson EG, Moses HL. TGF-beta signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science. 2004;303:848–851. doi: 10.1126/science.1090922. [DOI] [PubMed] [Google Scholar]
- 16.Danen EH, van Rheenen J, Franken W, Huveneers S, Sonneveld P, Jalink K, Sonnenberg A. Integrins control motile strategy through a Rho-cofilin pathway. J Cell Biol. 2005;169:515–526. doi: 10.1083/jcb.200412081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ren XD, Kiosses WB, Schwartz MA. Regulation of the small GTP-binding protein Rho by cell adhesion and the cytoskeleton. EMBO J. 1999;18:578–585. doi: 10.1093/emboj/18.3.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.White DP, Caswell PT, Norman JC. alpha v beta3 and alpha5beta1 integrin recycling pathways dictate downstream Rho kinase signaling to regulate persistent cell migration. J Cell Biol. 2007;177:515–525. doi: 10.1083/jcb.200609004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zeisberg EM, Potenta S, Xie L, Zeisberg M, Kalluri R. Discovery of endothelial to mesenchymal transition as a source for carcinoma-associated fibroblasts. Cancer Res. 2007;67:10123–10128. doi: 10.1158/0008-5472.CAN-07-3127. [DOI] [PubMed] [Google Scholar]
- 20.Petersen OW, Nielsen HL, Gudjonsson T, Villadsen R, Rank F, Niebuhr E, Bissell MJ, Ronnov-Jessen L. Epithelial to mesenchymal transition in human breast cancer can provide a nonmalignant stroma. Am J Pathol. 2003;162:391–402. doi: 10.1016/S0002-9440(10)63834-5. [DOI] [PMC free article] [PubMed] [Google Scholar]