This study shows that ex vivo culture of hematopoietic stem cells is associated with downregulation of both α4 and CXCR4-mediated engraftment.
Keywords: stem cells, hematopoiesis, cell trafficking
Abstract
Background
Several studies indicate that ex vivo cytokine-supported expansion induces defective hematopoietic stem cell engraftment. We investigated the role of α4 integrin, α5 integrin and CXCR4 in engraftment of unmanipulated and cytokine-treated human cord blood CD34+ cells.
Design and Methods
Uncultured or expanded CD34+ cells were infused in NOD/SCID-β2microglobulin-null mice. The function of α4, and α5 integrins and CXCR4 was assessed by incubating cells with specific neutralizing antibodies, prior to transplant. The activation state of α4 integrin was further tested by adhesion and migration assays.
Results
Neutralization of either α4 integrin or CXCR4 abolished engraftment of uncultured CD34+ cells at 6 week spost-transplant, while α5 integrin neutralization had no significant effect. However, after short-term ex vivo culture, blocking α4 integrin or CXCR4 did not affect repopulating activity whereas neutralization of α5 integrin inhibited engraftment. Using soluble vascular cell adhesion molecule-1 binding assays, we observed that α4 integrin affinity in fresh CD34+ cells was low and susceptible to stimulation while in cultured CD34+ cells, it was high and insensitive to further activation. In addition, stromal cell-derived factor-1 stimulated migration across vascular cell adhesion molecule-1 in fresh CD34+ cells but not in cultured CD34+ cells.
Conclusions
Our data show that ex vivo culture of hematopoietic progenitor cells is associated with downregulation of both α4 integrin- and CXCR4-mediated engraftment. Further investigations suggest that this is caused by supraphysiological increase of α4 integrin affinity, which impairs directional migration across vascular cell adhesion molecule-1 in response to stromal cell-derived factor-1. Such changes may underlie the engraftment defect of cytokine-stimulated CD34+ cells.
Introduction
Many studies have been dedicated to designing procedures allowing quantitative ex vivo expansion of transplantable hematopoietic stem cells. Although self-renewal divisions occur under defined culture conditions,1,2 it appears that ex vivo-generated hematopoietic stem cells engraft inefficiently in recipient bone marrow, a defect that may be associated with their proliferative status. We demonstrated that hematopoietic stem cells progressing in a first cell cycle in culture have reduced repopulating ability in NOD/SCID recipients compared to their counterparts staying in G0.3 It is unlikely that this defect resulted from hematopoietic stem cell differentiation since it was observed prior to any cell division. Using synchronized murine hematopoietic stem cells, Habibian et al. found decreased repopulating capacity during the first cell cycle executed ex vivo and further showed reversibility of this defect at the end of the cycle.4 The concept of a reversible engraftment defect related to cell cycle transit was further supported by data from Takatoku et al. who showed that cultured hematopoietic stem cells recovered their repopulating capacity after transfer into non-stimulating conditions.5 After longer periods of culture, in conditions that promote self-renewal divisions of transplantable hematopoietic stem cells, residual hematopoietic activity is essentially located in cells residing in the G1 phase, while cells in S, G2 and M do not retain any functional activity.6 These variations of repopulating activity result from differences in bone marrow homing capacity. Indeed, the proportion of stem/progenitor cells recovered from the host bone marrow 24 hours after transplantation is markedly reduced, in both congenic and xenogenic models.7,8 Two studies have documented that impaired bone marrow homing of cultured progenitor cells is associated with accumulation of the cells in the lungs.8,9
We previously reported that short-term cytokine-supplemented culture induces significant alterations in the interactions of progenitor cells with bone marrow ligands.10–13 Some of these changes are strongly associated with cell cycle status: CD34+ cells and long term culture-initiating cells (LTC-IC) transiting in S/G2/M phase display increased adhesion to fibronectin and decreased adhesion to immobilized vascular cell adhesion molecule-1 (VCAM-1), compared to cells in G0/G1 harvested from the same cultures. Other differences occurring in cultured CD34+ cells and LTC-IC are not associated with a particular phase of the cell cycle: stromal cell-derived factor-1 (SDF-1)-stimulated migration across fibronectin, VCAM-1 and ICAM-1 is decreased in cultured compared to in uncultured progenitor cells. We also observed that uncultured LTC-IC preferentially use α4 integrin (α4) to adhere and migrate on fibronectin, while in culture-expanded LTC-IC, the same function is achieved through α5 integrin (α5) usage, with concomitant inactivation of α4.
Papayannopoulou et al. have documented the role of α4 interaction with VCAM-1 in bone marrow homing. Antibody neutralization of either α4 or VCAM-1 inhibits homing of murine progenitors in the bone marrow of lethally irradiated recipients.14 Neutralization of another ligand of α4, the CS-1 domain of fibronectin, does not result in significant perturbation of bone marrow lodgment.15 Human hematopoietic progenitor cells bind in vitro to the RGD motif of fibronectin through interaction with α5.16 However, contradictory data have been reported concerning the contribution of α5 in mobilization and homing in vivo. α5 neutralization does not interfere with bone marrow lodgment and retention of murine progenitor cells,15,17 but results in significant impairment of human CD34+ cell engraftment in NOD/SCID mice,18 albeit to a lesser degree than treatment with α4 antibody.19 The chemokine SDF-1 and its receptor CXCR-4 define another major pathway involved in bone marrow homing, apoptosis20,21 and proliferation.22 The contribution of SDF-1/CXCR-4 to bone marrow homing involves two mechanisms: first, SDF-1 stimulates actin polymerization and chemotaxis of primitive progenitors;23 second, SDF-1 is also implicated in mediating transition of α4 from a low to a high affinity state, a critical step allowing arrest on vascular endothelium and subsequent migration.24
In this study, we evaluated the effects of cytokine-supported culture on the respective contribution of α4, α5 and CXCR-4 in homing and engraftment of human progenitors using NOD/SCID-β2microglobulin-null (β2m-null) mice as recipients. This model was selected since proliferating progenitor cells do engraft in NOD/SCID β2m-null mice while they remain mostly undetectable in standard NOD/SCID mice.25
Design and Methods
Human cells
After obtaining informed consent, cord blood samples were collected following full-term vaginal delivery according to the guidelines established by the Ethical Committee of the University of Liège. Mononuclear low density cells were isolated by centrifugation over Ficoll-Paque (Amersham Pharmacia Biotech, Uppsala, Sweden). Cord blood CD34+ cells were purified by immunomagnetic selection using MACS CD34 isolation kits (Miltenyi Biotech, Bergisch Gladbach, Germany) according to the manufacturer’s instructions. CD34+ cell purity in the final product always exceeded 97%.
Short-term expansion cultures
Cells were plated in short-term expansion culture in serum-free medium consisting of Iscoves’ medium supplemented with 20% BIT (Stem Cell Technologies, Meylan, France), 2 mmol/L alanyl-glutamine, 1% (v/v) cholesterol-rich lipids, 1 mmol/L sodium pyruvate (all from Sigma, Bornem, Belgium), 100 U/mL penicillin, 100 μg/mL streptomycin and 5×10−2 mmol/L 2-mercap-toethanol (all from Lonza, Verviers, Belgium). Cells were stimulated for 72 hours by a combination of 100 ng/mL each of stem cell factor, thrombopoietin (both from Amgen, Brussels, Belgium) and flt-3 ligand (Peprotech, Boechout, Belgium) and 20 ng/mL of interleukin-6 and granulocyte-colony stimulating factor (both from Amgen).
Homing and repopulation assays
The NOD/SCID-β2m-null mice colony was established from breeding pairs purchased from The Jackson Laboratory (Bar Harbor, ME, USA). NOD/SCID mice were purchased from Charles River (Lyon, France). Mice were housed in microisolators under pathogen-free conditions. Animal experiments were performed in accordance with institutional guidelines and approved by the Animal Care Ethical Committee of the University of Liège.
In repopulation assays, 12 week-old NOD/SCID-β2m-null mice were sublethally irradiated with 3Gy from a Cesium137 source (GammaCell 40, Nordion, ON, Canada) and were transplanted intravenously with freshly isolated cord blood CD34+ cells or their expansion product following short-term cultures. Integrin function was assessed by incubating grafts with azide-free neutralizing antibodies, anti-α4 (clone P4C2, isotype IgG3, Invitrogen, Paisley, UK), and anti-α5 (clone P1D6, isotype IgG3, Chemicon, Temecula, CA, USA), at 1:100 dilution for 30 minutes at 4°C prior to infusion. CXCR4 activity was assessed by treating grafts with 10 μg/mL azide-free 12G5 anti-CXCR4 neutralizing antibody (iso-type IgG2a, BD Biosciences, Erembodegem, Belgium). Control cells were treated with anti-CD34 antibody (iso-type IgG1, Miltenyi Biotech). Human chimerism in recipient bone marrow was determined after 6 weeks by flow cytometric detection of human CD45+ cells. Co-expression of CD19 or CD33 was used to evaluate multilineage repopulation.
In homing assays, a fixed dose of 500×103 of uncultured or cultured CD34+ cells was infused intravenously into sublethally irradiated mice. Cells were incubated prior to injection for 30 minutes at 4°C with azide-free neutralizing antibodies anti-α4, anti-α5, anti-CXCR4 or anti-CD34 as described above. The presence of human cells co-expressing CD34 and CD45 in recipient bone marrow was assessed 20 hours after graft infusion, acquiring at least 105 bone marrow cells. All fluorochrome-conjugated antibodies were from BD Biosciences. Samples were analyzed on a FACSCalibur flow cytometer (BD Biosciences). Gates were set to exclude 99.9% of non-specifically-stained cells as detected in bone marrow harvested from mice which had not been injected with human CD34+ cells.
Determination of α4 and β1 integrin activation state
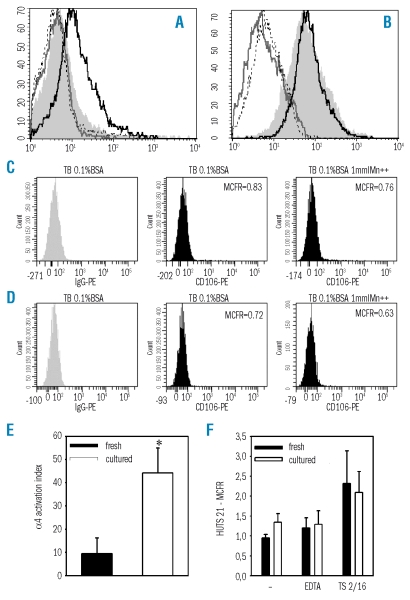
The activation state of α4 was evaluated by soluble ligand binding assays. Soluble VCAM-1 binding to fresh and expanded CD34+ cells was assessed as previously described.28 Briefly, fresh or expanded CD34+ cells were washed and resuspended in Tyrode’s buffer (Sigma) supplemented with 1% bovine serum albumin (BSA). As a negative control, integrin function was inhibited by adding 5 mmol/L EDTA. As a positive control, 1 mmol/L MnCl2 was used to induce maximal integrin activation. The specificity of the assay in measuring α4 function was confirmed by treating cells with Mn2+ and a 1:100 dilution of the α4 blocking monoclonal antibody, P4C2. Cells were then incubated with 4 μg/mL recombinant VCAM-1/Fc chimera (R&D Systems, Abingdon, UK) for 3 hours at 37°C. Next, cells were washed in their respective buffers and stained with phycoerythrin-conjugated anti-VCAM-1 antibody (anti-CD106, BD, Biosciences) for 20 minutes at 4°C. Cells were washed and fixed in 1% formaldehyde after which VCAM-1 binding was assessed on a FACSCalibur flow cytometer.
The activation index of α4 was calculated as 100 x [(Fo-Fr)/(Fmax-Fr)], where Fo is the mean fluorescence intensity of soluble VCAM-1 binding, Fr is background fluorescence in the presence of 5 mmol/L EDTA, and Fmax is the fluorescence intensity in the presence of 1 mmol/L MnCl2.
The activation state of β1 integrin was determined by staining CD34+ cells with HUTS 21-PE antibody (BD Biosciences), which is directed against an activation epitope of β1.29 Staining with HUTS 21 was done in Tyrode’s buffer 1% BSA, in 5 mmol/L EDTA as described above, or after CD34+ cell treatment with 1:100 dilution of the TS2/16 β1 activating antibody.30 Data are expressed as mean channel fluorescence ratio.
Actin polymerization assay
Fresh or culture-expanded CD34+ cells were washed and incubated with 100 ng/mL SDF-1 (R&D Systems). At defined time points, cells were fixed in phosphate-buffered saline (PBS) 3% formaldehyde for 15 minutes at room temperature. After washing in PBS, cells were resuspended in PBS 0.1M glycine (Sigma) for 15 minutes at room temperature. Cells were then permeabilized in PBS 1% BSA 0.2% Triton-X (Sigma) for 10 minutes at room temperature. Triton-X was omitted for the negative control sample. Phalloïdin-fluorescein isothio-cyanate (Molecular Probes Invitrogen, Merelbeke, Belgium) was added at a final concentration of 0.5 μM for 30 minutes at room temperature, after which cell samples were analyzed on a FACSCalibur flow cytometer (BD Biosciences) for filamentous actin content.
Adhesion assays
Adhesion assays were carried out in 24-well plates coated with 1 μg/cm2 VCAM-1 or 1% fraction V BSA in PBS at 4°C overnight. After two washes in RPMI 1640 with 25 mM HEPES, 2×105 fresh or cultured CD34+ cells were plated in 100 μL serum-free medium for 1 hour at 37°C, in the presence or absence of 100 ng/mL SDF-1. After incubation for 1 hour, non-adherent cells were harvested by two standardized washes using warm PBS 1% BSA. Adherent cells were then recovered after a 5-minute incubation in cell dissociation buffer (Invitrogen) at 37°C followed by vigorous pipetting. Adherent and non-adherent cells were counted in Fuchs-Rosenthal hemacytometers. Percent adhesion was calculated relative to the total number of cells plated. In some experiments, CD34+ cells were pre-incubated with a 1:100 dilution of P4C2 anti-α4 blocking antibody.
Migration assays
Migration assays were performed in 6.5-mm diameter 5-μm pore Transwells (Costar, Elscolab, Kruibeke, Belgium). Transwell filters were coated with 1 μg/cm2 VCAM-1 or 1% fraction V BSA in PBS at 4°C overnight. After two washes in RPMI 1640 with 25 mM HEPES, 2×105 fresh or cultured CD34+ cells were plated in 100 μL serum-free medium in the upper chamber of the Transwell. The bottom compartment was filled with 600 μL serum-free medium supplemented with 100 ng/mL SDF-1 (R&D Systems). After incubation at 37°C for 3 h, non-migrating and migrating cells were harvested from the top and bottom compartments, respectively. Percent migration was calculated as: number of migrating cells/(number of migrating cells + number of non-migrating cells).
Statistical analysis
Results are reported as mean ± SEM. The Gaussian distribution of the data was assessed with Kolmogorov-Smirnov tests (SigmaStat software; SSPS, Richmond, CA, USA). Paired Student’s t tests and non-parametric Wilcoxon signed rank tests were used for Gaussian and non-Gaussian distributions, respectively. All p values are two-sided.
Results
Changes in α4 and α5 integrin-mediated repopulation in ex vivo expanded CD34+ cells
In a first set of experiments, we determined the role of α4 and α5 by 6-week repopulation assays in NOD/SCID β2m-null recipient mice. After transplantation of 150–200×103 freshly isolated cord blood CD34+ cells, average chimerism was 37.6±11.4% CD45+ cells. Neutralization of α4 by P4C2 monoclonal antibody induced a marked decrease in the repopulating activity such that 6-week human chimerism was 1.6±0.9% (p<0.05). Conversely, inhibition of α5 function by P1D6 monoclonal antibody did not significantly reduce the ability of native cord blood CD34+ cells to repopulate the bone marrow NOD/SCID β2m-null mice, as the percentage human chimerism was 22.9±9.9 (p>0.05) (Figure 1). The reconstituting ability of cord blood CD34+ cells was, therefore, primarily dependent on α4 while only a modest contribution of α5 in bone marrow repopulation could be detected.
Figure 1.
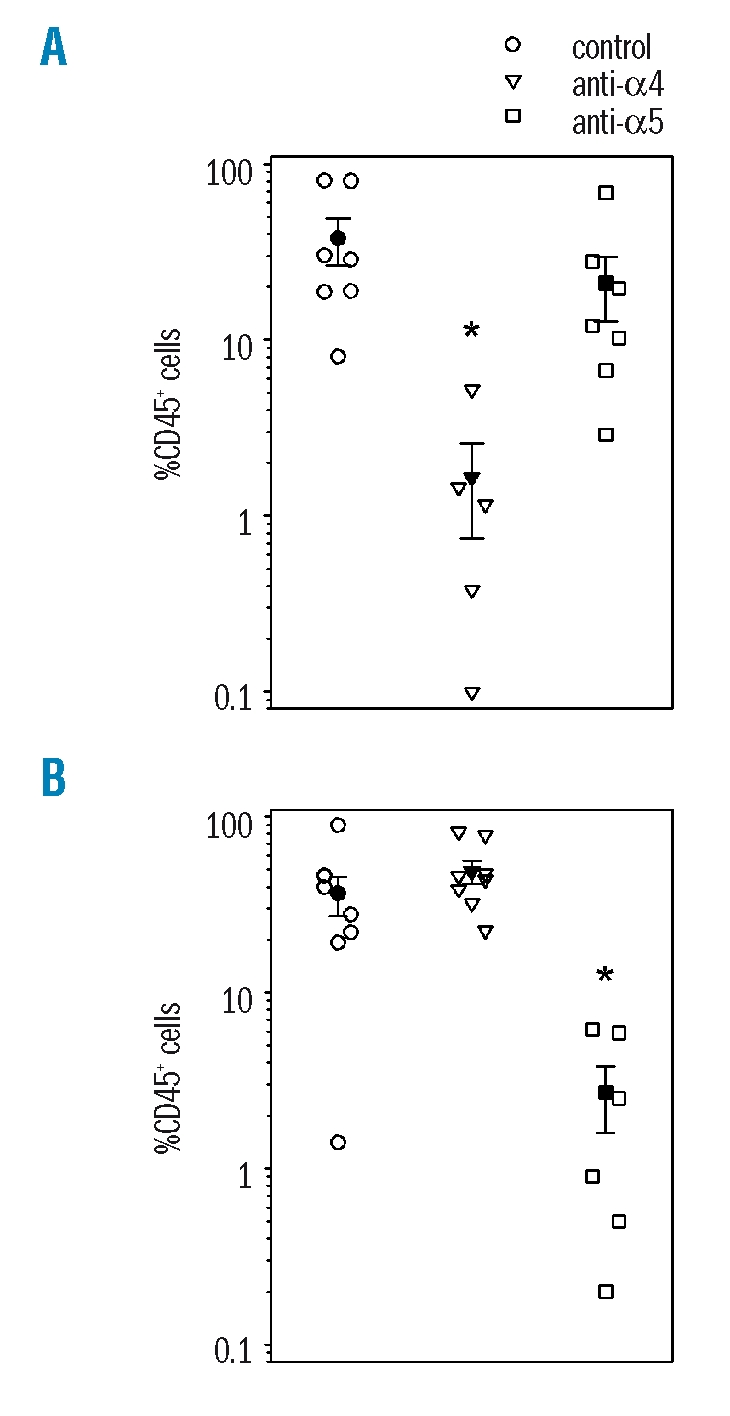
α4 and α5 function in freshly isolated or ex vivo expanded SRC. The percentage of human CD45+ cells was measured in bone marrow cells of recipient mice transplanted 6 weeks previously with 100–150×103 uncultured cord blood CD34+ cells (A) or their expansion product (B) after short-term culture in stem cell factor, thrombopoietin, Fit-3 ligand, interleukin-6 and granulocyte colony-stimulating factor. The contribution of α4 and α5 integrins to bone marrow repopulation was determined by incubating cells with neutralizing monoclonal antibodies P4C2 (anti-α4, triangles) or P1D6 (anti-α5, squares) prior to transplantation. Control cells were incubated with anti-CD34 (circles). Solid symbols indicate the mean ± SEM engraftment levels in each experimental condition (n=3 independent experiments of one to three mice per group). *p<0.05 compared to transplantation of control cells.
A similar series of experiments was carried out with cord blood CD34+ cells that had been previously stimulated in short-term (72 hours) culture. In these conditions, the proportion of cells in S/G2+M phases of the cell cycle, which was less than 2% in uncultured cells, increased to more than 40% in all experiments. Amplification of total CD34+ cell numbers was 2.4 fold on average. To avoid dilution of repopulating cells by cell proliferation, recipient mice were infused with the total expansion product of 150–200×103 fresh CD34+ cells, which corresponded to 300–480×103 cultured CD34+ cells. No increase in repopulating activity was observed following ex vivo culture and average chimerism was 36.3±9.3% CD45+ cells. Strikingly, prior incubation of expanded cells with anti-α4 monoclonal antibody did not affect SRC activity (48.9±7.4% chimerism, p>0.05 vs. control), whereas α5 neutralization inhibited engraftment down to 2.7±1.1% CD45+ cells after 6 weeks (p<0.05). Engraftment of cultured CD34+ cells was thus mediated by α5, while the function of α4 was down-modulated (Figure 1).
It has been reported that NOD/SCID β2m-null mice may be reconstituted with both long-term lympho-myeloid stem cells and short-term myeloid-restricted progenitor cells.25 This prompted us to evaluate whether differential usage of α4 and α5 by unmanipulated and cultured cells resulted from the accumulation of myeloid progenitors in the expansion products. We determined co-expression of myeloid and B lymphoid markers by engrafted human CD45+ cells recovered from the bone marrow of recipient mice. All NOD/SCID β2m-null recipients receiving either fresh or expanded grafts were reconstituted with both B lymphoid (CD19+) and myeloid (CD33+) cells (data not shown). Integrin inhibition did not change the ratio between myeloid and lymphoid cell reconstitution. This suggests that α4 and α5 blocking antibodies did not target distinct populations of committed progenitor cells but rather inhibited engraftment by multilineage reconstituting cells.
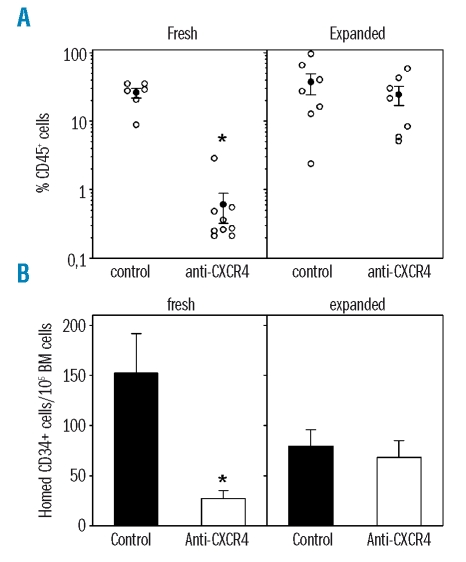
α4-mediated homing in NOD/SCID β2m-null mice is downregulated after ex vivo culture
Integrin neutralization may interfere with direct bone marrow seeding after transplantation. Alternatively, integrin neutralization may mediate lodgment of repopulating cells in non-functional and unproductive bone marrow niches, thus preventing efficient post-homing proliferation and differentiation. To distinguish between these two possibilities, we determined the effect of α4 and α5 blocking monoclonal antibodies on the homing capacity of CD34+ cells. Homing assays were carried out by intravenously injecting a fixed number of 500×103 uncultured or ex vivo-cultured CD34+ cells into NOD/SCID β2m-null mice. Homing efficiency was determined as the proportion of human CD34+ CD45+ cells in the bone marrow of the recipient mice, 20 hours after transplantation. Homing was slightly decreased in cultured cells compared to in uncultured cells, but the difference was not statistically significant. Homing of uncultured CD34+ cells was significantly inhibited by α4 neutralization while α5 neutralization had no effect (Figure 2). In contrast, homing of expanded CD34+ cells was not affected by blocking α4 but was markedly reduced after incubation with α5 blocking monoclonal antibody (Figure 2). Overall, these data indicate that changes in integrin functional state after ex vivo culture directly affected the bone marrow homing efficiency of CD34+ cells.
Figure 2.
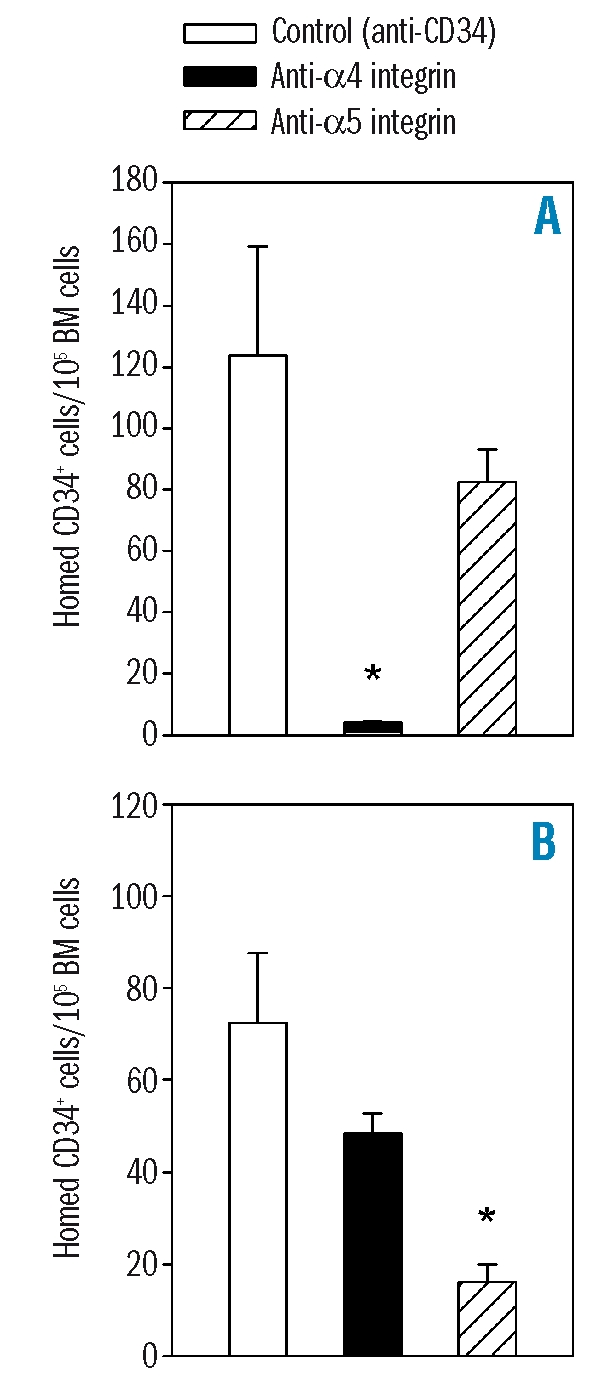
Role of α4 and α5 in bone marrow homing of uncultured or ex vivo-expanded cord blood CD34+ cells. The presence of human CD34+ cells was determined in bone marrow (BM) harvested from NOD/SCID β2m-null mice transplanted 16–24 hours previously with 500×103 cord blood CD34+ cells before (A) or after (B) short-term culture. The function of α4 and α5 was assessed by incubating cells with P4C2 and P1D6 monoclonal prior to infusion in recipient mice antibodies, respectively. Control cells were treated with anti-CD34. p<0.05 compared to transplantation of control cells (n=3 experiments of two or three mice per group).
Modulation of α4 affinity in ex vivo expanded CD34+ cells
It has been reported by several investigators that expression of adhesion molecules by CD34+ cells is maintained, and even increased, after ex vivo expansion.32–34 We observed that before expansion, α4 and α5 were uniformly expressed on the surface of cord blood CD34+ cells. Expression of α4 and α5 was increased on cultured CD34+ cells (data not shown), ruling out that differences in expression could account for the changes in integrin function observed after ex vivo culture. Integrin function can be dynamically regulated through changes in affinity state without modification in integrin expression, a process called inside-out signaling.35 We thus carried out a functional analysis of α4 affinity in native and expanded CD34+ cells.
The binding capacity of α4 for a soluble form of VCAM-1 was assessed by flow cytometry.28,36 Native or cultured CD34+ cells were suspended in Tyrode’s buffer to provide the baseline condition. As a negative control, Tyrode’s buffer was supplemented with 5 mmol/L EDTA in order to inactivate α4 integrin. As a positive control, 1 mmol/L Mn2+ was added to obtain maximal α4 activation. Soluble VCAM-1 was added and cells were then incubated for 3 hours at 37°C. The extent of soluble VCAM-1 binding was determined by flow cytometry using anti-VCAM-1 (anti-CD106) fluorescent antibody. We observed that in uncultured CD34+ cells, soluble VCAM-1 binding was not modified by EDTA but was upregulated by Mn2+ (Figure 3A). This indicates that α4 was present in a low affinity state in native CD34+ cells and was susceptible to external stimuli. Conversely, in expanded CD34+ cells, soluble VCAM-1 binding could be inhibited by EDTA but was not upregulated by Mn2+, indicating maximal α4 affinity (Figure 3B). The specificity of the assay for measuring α4 affinity was confirmed by showing that pre-treatment of CD34+ cells with anti-α4 blocking antibody completely abolished Mn2+-enhanced soluble VCAM-1 binding in both fresh and cultured CD34+ cells (Figure 3A–B). Further control experiments showed that, as compared to irrelevant IgG, binding of anti-CD106 was undetectable in fresh or cultured CD34+ cells which had not been previously exposed to soluble VCAM-1, either in Tyrode’s buffer or in Tyrode’s buffer supplemented with 1 mmol/L MnCl2 (Figure 3C–D). The activation index of α4, computed as described in the Methods section, was increased 4-fold in expanded CD34+ cells (p<0.05) (Figure 3E). Thus, after expansion, α4 was present in a maximally activated state which prevented further response to activating stimuli.
Figure 3.
Activation state of α4 and β1 integrins on fresh or expanded CD34+ cells. (A) Fresh or (B) expanded CD34+ cells were resuspended in Tyrode’s buffer 1% BSA (shaded histogram) or Tyrode’s buffer 1% BSA supplemented with 5 mmol/L EDTA (gray line), with 1 mmol/L MnCl2 (black line), or with 1 mmol/L MnCl2 together with function-blocking anti-α4 monoclonal antibody P4C2 (dotted line). Soluble VCAM-1 was added and cells were incubated for 3 hours at 37°C. Binding was measured by cell staining with phycoerythrin (PE)-conjugated anti-VCAM-1 (anti-CD106) monoclonal antibody. Results of one representative experiment out of four are shown. As compared to IgG-PE, binding of CD106 was undetectable in fresh (C) or cultured CD34+ cells (D) which had not been previously exposed to soluble VCAM-1, either in Tyrode’s buffer (TB) 0.1% BSA or in TB supplemented with 1 mmol/L MnCL2. (E) The activation index of α4 integrin on fresh and expanded CD34+ cells was calculated as 100 × [(Fo−Fr)/(Fmax−Fr)], where Fo is the mean fluorescence intensity of soluble VCAM-1 binding in Tyrode’s buffer 1% BSA, Fr is background fluorescence in the presence of 5 mmol/L EDTA, and Fmax is the fluorescence intensity in the presence of 1 mmol/L MnCl2. *p<0.05, n=4. (F) Fresh or expanded CD34+ cells were stained with HUTS-21 monoclonal antibody which is directed against an activation epitope of β1 integrin. HUTS-21 expression was measured in cells incubated in Tyrode’s buffer (−), in Tyrode’s buffer supplemented with 5 mmol/L EDTA or with β1 activating monoclonal antibody TS2/16 (n=3).
The expression of the HUTS-21 epitope by native and cultured CD34+ cells was assessed as a marker of β1 integrin activation state.37 As a negative control, HUTS-21 expression was measured after treating CD34+ cells with EDTA. As a positive control, the response to the β1 activating antibody TS2/1630 was measured. We observed that HUTS-21 was not expressed in either uncultured, or cultured CD34+ cells, with a mean fluorescence ratio close to 1. CD34+ cell stimulation with TS2/16 induced a slight increase in HUTS-21 expression, of the same magnitude in uncultured and in cultured CD34+ cells (Figure 3F). CD34+ cell labeling with an anti-β1 antibody not directed to an activation epitope yielded mean channel fluorescence values of 5.45±0.20 and 10.64±2.09 for uncultured and cultured CD34+ cells, respectively. β1 was thus present on fresh and expanded CD34+ cells but did not present the activation epitope. Overall, this suggests that ex vivo culture was not associated with changes in the β1 subunit of α 4β1 integrin.
Downmodulation of CXCR4-mediated homing and repopulation in NOD/SCID β2m-null mice by ex vivo cultured CD34+ cells
A critical function of the SDF-1/CXCR4 axis is to induce the transition of α4 from low to high affinity.38 We thus reasoned that if α4 was locked in a maximally activated state in expanded cells, homing and engraftment would then become CXCR4-independent. We first compared the contribution of CXCR4 in engraftment of native and expanded CD34+ cells. Cells were exposed to function-blocking CXCR4 antibody, infused into NOD/SCID β2m-null mice, and assessed in 20-hour homing assays and in 6-week repopulation experiments. Consistently with previous reports,39 both homing and repopulation of fresh CD34+ cells were strongly inhibited by CXCR4 neutralization. However, after infusion of expanded cells, anti-CXCR4 antibody induced only a slight and non-significant reduction in bone marrow repopulation (Figure 4A) and did not impair direct bone marrow lodgment in NOD/SCID β2m-null recipient mice (Figure 4B). CXCR4 intensity was slightly down-modulated after culture but still expressed on the majority of CD34+ cells (not shown).
Figure 4.
Role of CXCR-4 in repopulating activity and homing of fresh or expanded CD34+ cells in NOD/SCID β2m-null mice. (A) Mice were transplanted with 100–150×103 CD34+ cells or their expansion equivalent after a 3-day expansion culture. Prior to infusion, cells were incubated with function-blocking anti-CXCR4 12G5 monoclonal antibody or anti-CD34 as control. Bone marrow chimerism was determined after 6 weeks by staining with human-specific anti-CD45. Solid symbols indicate the mean ± SEM engraftment levels in each experimental condition. (B) Mice were infused with 500×103 fresh or 500×103 expanded CD34+ cells. Homed CD34+ cells were measured in bone marrow of recipient mice 16–20 hours later. *p<0.05 compared to transplantation of control cells (n=3 experiments with two to four mice in each condition).
SDF-1 induces actin polymerization but not VCAM-1-supported transmigration in cultured CD34+ cells
SDF-1/CXCR-4 signaling controls homing of hematopoietic progenitor cells through two mechanisms. First, CXCR-4 mediates reorganization of the actin cytoskeleton, which is required for cell motility. Second, CXCR-4 induces transition of α4β1 from a low to a high affinity state, which allows firm adhesion of rolling cells onto VCAM-1 presented at the endothelial cell surface, and finally transmigration. Localized secretion of SDF-1 in the bone marrow ensures selectivity of the homing process. The apparent desensitization of cultured CD34+ cells to CXCR-4 inhibition prompted us to assess which of these two mechanisms was impaired.
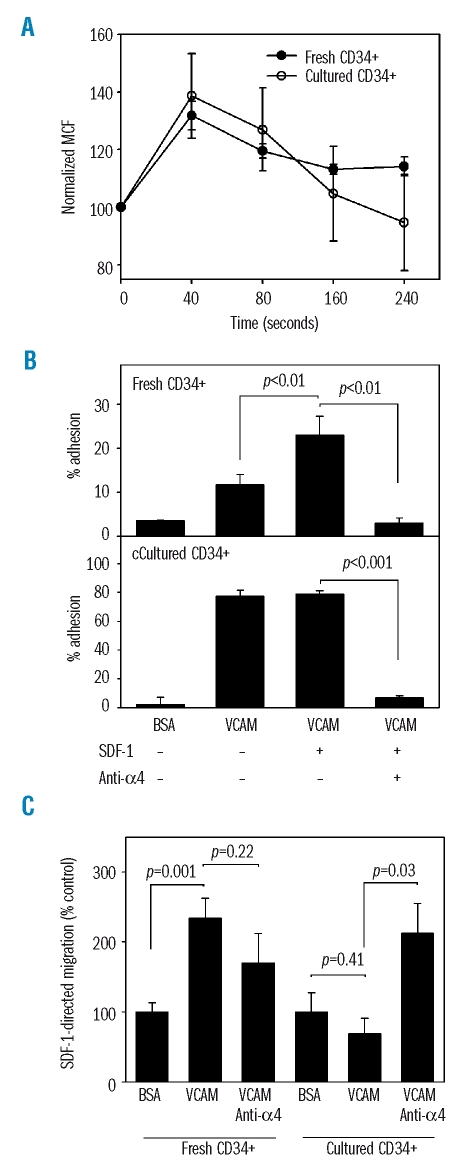
The evaluation of CXCR-4 function in actin polymerization was studied in native and expanded CD34+ cells. Cells were stimulated with 100 ng/mL SDF-1 and stained with phalloïdin-fluorescein isothiocyanate, which binds to filamentous but not globular actin. In native CD34+ cells, SDF-1 mediated a 32±5% increase in filamentousactin content in 40 seconds. The stimulation lasted up to 160 seconds. The increase in filamentousactin content at 40 seconds was similar in cultured CD34+ cells (38±14%, n=3, p>0.05), indicating a conservation of the effect of SDF-1 on cytoskeletal organization (Figure 5A).
Figure 5.
Effect of SDF-1 in mediating actin polymerization, adhesion to VCAM and VCAM-supported transmigration of fresh and cultured CD34+ cells. (A) Fresh or cultured CD34+ cells were stimulated with 100 ng/mL SDF-1 for the indicated times and stained for filamentous actin with phalloidin-fluorescein isothiocyanate. Mean channel fluorescence (MCF) is shown as a function of time (n=3). (B) Adhesion assays were carried out on BSA- and VCAM-coated plates. The effect of SDF-1 and anti-α4 antibody was studied. Adhesion of fresh and cultured CD34+ cells is shown (n=3). (C) Transmigration assays were set up in response to 100 ng/mL SDF-1 across VCAM-coated Transwells or across BSA as a control. The effect of α4 neutralization is depicted. Migration of fresh and cultured CD34+ cells is shown (n=4).
The control of α4β1 activation state by SDF-1 on fresh and expanded CD34+ cells was examined by adhesion and migration experiments. Background adhesion of fresh CD34+ cells onto BSA-coated plates was 3.6±0.05%. VCAM-1-supported adhesion was 11.7±1.4%. When cells were incubated with SDF-1 during the adhesion assay, binding to VCAM-1 rose to 23.0±2.5%. Adhesion was completely inhibited by pre-treating the cells with a neutralizing anti-α4 antibody. In cultured CD34+ cells, adhesion to VCAM-1 was increased to 78.9±1.2% in the absence of SDF-1 and, interestingly, no further increase of adhesion was noted in the presence of SDF-1 (Figure 5B). Thus, expanded CD34+ cells lose the ability to upregulate VCAM-1 binding upon SDF-1 stimulation. This may represent a loss of selectivity in the bone marrow homing process.
Transmigration supported by SDF-1 was assayed in fresh and cultured CD34+ cells (Figure 5C). CD34+ cells were placed in the upper compartment of Transwells® coated with VCAM-1 or BSA as a negative control. The lower chamber was filled with serum-free medium supplemented with 100 ng/mL SDF-1.
In fresh CD34+ cells, as compared to baseline transmigration across BSA which was 12.5±1.3%, SDF-1 supported significant migration across VCAM (p=0.001). Neutralization of α4 did not change the migration rate across VCAM-1. This is consistent with a distribution of α4 molecules between forms of low and high affinity when exposed to SDF-1. Inhibition of low affinity α4 by the blocking antibody prevents cell attachment to the Transwell and subsequent migration. Conversely, inhibition of high affinity α4 releases cells from firm adhesion to the filter and allows progression across the Transwell. The net effect of α4 neutralization may, therefore, be negligible.
In contrast, in cultured CD34+ cells, as compared to baseline transmigration across BSA, which was 11.7±1.2%, the rate of migration was not enhanced across VCAM-1 (p=0.41). Addition of α4 neutralizing antibody significantly enhanced the migration rate and restored SDF-1-supported migration (p=0.03). This is consistent with the hypothesis that maximal α4 affinity impairs the chemotactic response to SDF-1 once CD34+ cells have been activated ex vivo.
Discussion
Various studies have shown that in vitro exposure of stem/progenitor cells to hematopoietic growth factors results in reduced repopulating activity. The involvement of specific ligand-receptor interactions in this defect is not well documented. Previous studies from our group showed changes in α4 and α5 functional state in hematopoietic progenitor cells harvested from expansion culture, with a shift from dominant α4-mediated interactions to preferential α5-mediated interactions with immobilized fibronectin in vitro.12 Variations in integrin activation states result in changes not only in binding and migration behavior, but also in response to growth-inhibitory signals.13 In the present work, we demonstrate for the first time that the changes in the activation state of α4 and α5 apply to NOD/SCID β2m-null mice in vivo repopulating cells such that repopulation by uncultured CD34+ cells is dependent on α4 while repopulation by ex vivo-expanded CD34+ cells is mediated by α5.
The prominent role of α4 in mediating hematopoietic reconstitution by unmanipulated hemapoietic progenitor cells has been well documented and lies in its interaction with VCAM-1.14 Neutralization of the CS-1 domain of fibronectin, another potential ligand of α4, does not appear to interfere with hematopoietic progenitor cell trafficking.15 As for α5, it has been reported that its inhibition prevents bone marrow repopulation by freshly isolated human repopulating cells,19 although to a lesser degree than α4 inhibition. In the present study, α5 inhibition induced a slight, but not statistically significant, reduction in engraftment of uncultured CD34+ cells. This discrepancy may stem from the use of different clones with varying potency in neutralizing α5. It is, however, clear from our data that engraftment inhibition with anti-α5 antibody is more pronounced with cultured cells than with native cells.
It is intriguing that expanded CD34+ cells are able to engraft NOD/SCID β2m-null mice independently of α4 integrin. In vivo, α4 contributes not only to tethering and rolling on endothelial VCAM-138 but also, together with α5, to directional migration across basal lamina fibronectin and ultimately across stromal cells.18 It is likely that interactions with other endothelial ligands, such as E-selectin and ICAM-1, compensate for the loss of α4/VCAM-1 function to mediate endothelial rolling and transmigration.40 As for transmembrane and transstromal migration, our results are consistent with a cooperative role of α5, which is uncovered once α4 is inactivated. Short-term engraftment of α4-deficient hematopoietic progenitor cells has recently been reported in conditional α4 knockouts, although a progressive decline was noted long term, suggesting abnormal homing and/or self-renewal in post-homing hematopoiesis.41 This suggests that α4-deficient stem/progenitor cells produced by cytokine-driven expansion may be competitively disadvantaged against residual host counterparts.
α4β1 integrin may contribute to various steps during SRC recruitment depending on its activation state. Affinity modulation is regulated by changes in integrin conformation which may be reproduced experimentally by activating antibodies such as anti-β1 8A242 and TS2/1630 clones. Independently of affinity modulation, cell binding may be induced by increased avidity through clustering and immobilization of the integrin in the cell membrane.43 In mature leukocytes, α4 integrin first directs primary adhesion i.e., short-term interactions with VCAM-1 which slow down leukocytes in contact with endothelial cells. These rolling interactions are mediated by short-lived reversible clustering of low affinity α4. In secondary adhesion, leukocytes are immobilized onto VCAM-1 by long-lasting interactions with high affinity α4 integrins. Both steps are under the control of endothelial chemokines.38 In stem and progenitor cells, SDF-1 expressed on bone marrow endothelial cells is pivotal in mediating the transition of α4 from low to high affinity and converting primary rolling to secondary firm adhesion to VCAM-1.44 Selective bone marrow homing of stem/progenitor cells is thus determined by localized α4 activation in contact with SDF-1-expressing endothelial cells.
Measurements of α4 binding to soluble VCAM-1 were used to assess α4 activation state.28 In baseline hematopoietic progenitor cells, α4 was present in a low affinity state, which could be upregulated by stimuli such as Mn++ ions. Conversely, in expanded CD34+ cells, soluble VCAM-1 binding to α4 was maximal and could not be further increased by activating stimuli. In static adhesion assays, we observed that VCAM-1 binding could not be upregulated by SDF-1 in expanded CD34+ cells in contrast to fresh CD34+ cells. This suggests that binding of expanded hematopoietic progenitor cells to endothelial VCAM-1 may no longer be responsive to localized SDF-1 secretion in vivo, which may result in the loss of selective bone marrow homing. Indeed, several investigators have reported increased extramedullary homing of ex vivo-cultured hematopoietic progenitor cells.8,45
Given the prominent role of SDF-1 in mediating α4 activation, we reasoned that constitutive α4 activation in expanded cells would lead to SDF-1 desensitization. Indeed, treatment of expanded CD34+ cells with CXCR4 blocking antibody 12G5 did not inhibit homing or 6-week repopulation in NOD/SCID β2m-null mice. In vitro, the observation that SDF-1-mediated actin polymerization was maintained in cultured cells argues against a direct defect in cell motility induced by ex vivo culture. In migration assays across VCAM-1-coated Transwells, expanded CD34+ cells were not responsive to SDF-1 unless α4 was neutralized. Thus, expanded cells express α4 in a high affinity state which prevents response to SDF-1.
The 6-week repopulating activity of freshly isolated CD34+ cells and that of their expansion product were very similar. This may indicate that despite a 2.4 fold increase in total nucleated cells, there was no net multiplication of SRC. Alternatively, SRC did expand in short-term culture but their amplification was masked by inefficient homing due to α4 inactivation. To explore this possibility would require the restoration of the initial activation state of very late antigen-4 in expanded SRC. One strategy would be to reinduce mitotic quiescence of cultured cells prior to transplantation. Future studies should examine whether restoration of very late antigen-4-directed homing in expanded hematopoietic stem cells would increase their repopulating capacity.
The 6-week repopulation end-point which was used in the present study precludes a direct application of our data to true hematopoietic stem cell biology. Indeed, it has been shown that in NOD/SCID β2m-null mice, repopulation is achieved by both transient progenitors and self-renewing stem cells. Also, homing of total CD34+ cells may differ significantly from that of long-term repopulating cells. Further experiments based on longer repopulation assays and secondary transplants will be required to translate the observations of this study to the level of long-term hematopoietic stem cells.
In conclusion, our data show significant changes in α4 and CXCR4 dependence in bone marrow repopulation by ex vivo-cultured hematopoietic progenitor cells. Our results are consistent with the hypothesis that maximal α4 affinity observed in cultured CD34+ cells induces unresponsiveness to SDF-1/CXCR-4 signaling, resulting in downregulation of both α4 and CXCR4-mediated engraftment.
Acknowledgments
The authors thank Dr F. Sanchez-Madrid for the generous gift of TS/16 antibody.
Footnotes
Authorship and Disclosures
JF performed research, analyzed the data and contributed to the design of the study; IDS and OG performed research and analyzed the data; YB contributed to the design of the study and data interpretation; AG designed the research, analyzed the data and wrote the paper.
The authors reported no potential conflicts of interest.
Funding: this work was supported by grants from the Fonds de la Recherche Scientifique (F.N.R.S., Belgium), the Centre Anticancéreux près l’Université de Liège and the Fédération Belge contre le Cancer (a non-profit organization). JF was supported by a fellowship from the Fonds pour la formation à la Recherche dans l’Industrie et l’Agriculture (F.R.I.A., Belgium). YB is Research Director of the F.N.R.S.
References
- 1.Glimm H, Eaves CJ. Direct evidence for multiple self-renewal divisions of human in vivo repopulating hematopoietic cells in short-term culture. Blood. 1999;94:2161–8. [PubMed] [Google Scholar]
- 2.Dao MA, Yu XJ, Nolta JA. Clonal diversity of primitive human hematopoietic progenitors following retroviral marking and long-term engraftment in immune-deficient mice. Exp Hematol. 1997;25:1357–66. [PubMed] [Google Scholar]
- 3.Gothot A, van der Loo JCM, Clapp DW, Srour EF. Cell cycle-related changes in repopulating capacity of human mobilized peripheral blood CD34+ cells in NOD/SCID mice. Blood. 1998;92:2641–9. [PubMed] [Google Scholar]
- 4.Habibian HK, Peters SO, Hsieh CC, Wuu J, Vergilis K, Grimaldi CI, et al. The fluctuating phenotype of the lymphohematopoietic stem cell with cell cycle transit. J Exp Med. 1998;188:393–8. doi: 10.1084/jem.188.2.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Takatoku M, Sellers S, Agricola BA, Metzger ME, Kato I, Donahue RE, Dunbar CE. Avoidance of stimulation improves engraftment of cultured and retrovirally transduced hematopoietic cells in primates. J Clin Invest. 2001;108:447–55. doi: 10.1172/JCI12593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Glimm H, Oh IH, Eaves CJ. Human hematopoietic stem cells stimulated to proliferate in vitro lose engraftment potential during their S/G2/M transit and do not reenter G0. Blood. 2000;96:4185–93. [PubMed] [Google Scholar]
- 7.Szilvassy SJ, Bass MJ, Van Zant G, Grimes B. Organ-selective homing defines engraftment kinetics of murine hematopoietic stem cells and is compromised by ex vivo expansion. Blood. 1999;93:1557–66. [PubMed] [Google Scholar]
- 8.Ahmed F, Ings SJ, Pizzey AR, Blundell MP, Thrasher AJ, Ye HT, et al. Impaired bone marrow homing of cytokine-activated CD34+ cells in the NOD/SCID model. Blood. 2004;103:2079–87. doi: 10.1182/blood-2003-06-1770. [DOI] [PubMed] [Google Scholar]
- 9.Dooner M, Cerny J, Colvin G, Demers D, Pimentel J, Greer D, et al. Homing and conversion of murine hematopoietic stem cells to lung. Blood Cells Mol Dis. 2004;32:47–51. doi: 10.1016/j.bcmd.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 10.Giet O, Huygen S, Beguin Y, Gothot A. Cell cycle activation of hematopoietic progenitor cells increases very late antigen-5-mediated adhesion to fibronectin. Exp Hematol. 2001;29:515–24. doi: 10.1016/s0301-472x(00)00682-2. [DOI] [PubMed] [Google Scholar]
- 11.Giet O, Van Bockstaele DR, Di Stefano I, Huygen S, Greimers R, Beguin Y, Gothot A. Increased binding and defective migration across f ibronectin of cycling hematopoietic progenitor cells. Blood. 2002;99:2023–31. doi: 10.1182/blood.v99.6.2023. [DOI] [PubMed] [Google Scholar]
- 12.Huygen S, Giet O, Artisien V, Di Stefano I, Beguin Y, Gothot A. Adhesion of synchronized human hematopoietic progenitor cells to fibronectin and vascular cell adhesion molecule-1 fluctuates reversibly during cell cycle transit in ex vivo culture. Blood. 2002;100:2744–52. doi: 10.1182/blood.V100.8.2744. [DOI] [PubMed] [Google Scholar]
- 13.Foguenne J, Huygen S, Greimers R, Beguin Y, Gothot A. Modulation of homing properties of primitive progenitor cells generated by ex vivo expansion. Haematologica. 2005;90:445–51. [PubMed] [Google Scholar]
- 14.Papayannopoulou T, Craddock C, Nakamoto B, Priestley GV, Wolf NS. The VLA4/VCAM-1 adhesion pathway defines contrasting mechanisms of lodgement of transplanted murine hemopoietic progenitors between bone marrow and spleen. Proc Natl Acad Sci USA. 1995;92:9647–51. doi: 10.1073/pnas.92.21.9647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Craddock CF, Nakamoto B, Elices M, Papayannopoulou T. The role of CS1 moiety of fibronectin in VLA mediated haemopoietic progenitor trafficking. Br J Haematol. 1997;97:15–21. doi: 10.1046/j.1365-2141.1997.d01-2120.x. [DOI] [PubMed] [Google Scholar]
- 16.van der Loo JC, Xiao X, McMillin D, Hashino K, Kato I, Williams DA. VLA-5 is expressed by mouse and human long-term repopulating hematopoietic cells and mediates adhesion to extracellular matrix protein fibronectin. J Clin Invest. 1998;102:1051–61. doi: 10.1172/JCI3687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Papayannopoulou T, Priestley GV, Nakamoto B, Zafiropoulos V, Scott LM, Harlan JM. Synergistic mobilization of hemopoietic progenitor cells using concurrent beta1 and beta2 integrin blockade or beta2-deficient mice. Blood. 2001;97:1282–8. doi: 10.1182/blood.v97.5.1282. [DOI] [PubMed] [Google Scholar]
- 18.Peled A, Kollet O, Ponomaryov T, Petit I, Franitza S, Grabovsky V, et al. The chemokine SDF-1 activates the integrins LFA-1, VLA-4, and VLA-5 on immature human CD34+ cells: role in transendothelial/stromal migration and engraftment of NOD/SCID mice. Blood. 2000;95:3289–96. [PubMed] [Google Scholar]
- 19.Yahata T, Ando K, Sato T, Miyatake H, Nakamura Y, Muguruma Y, et al. A highly sensitive strategy for SCID-repopulating cell assay by direct injection of primitive human hematopoietic cells into NOD/SCID mice bone marrow. Blood. 2003;101:2905–13. doi: 10.1182/blood-2002-07-1995. [DOI] [PubMed] [Google Scholar]
- 20.Lataillade JJ, Clay D, Bourin P, Herodin F, Dupuy C, Jasmin C, Bousse-Kerdiles MC. Stromal cell-derived factor 1 regulates primitive hematopoiesis by suppressing apoptosis and by promoting G0/G1 transition in CD34+ cells: evidence for an autocrine/paracrine mechanism. Blood. 2002;99:1117–29. doi: 10.1182/blood.v99.4.1117. [DOI] [PubMed] [Google Scholar]
- 21.Lataillade JJ, Clay D, Dupuy C, Rigal S, Jasmin C, Bourin P, Bousse-Kerdiles MC. Chemokine SDF-1 enhances circulating CD34+ cell proliferation in synergy with cytokines: possible role in progenitor survival. Blood. 2000;95:756–68. [PubMed] [Google Scholar]
- 22.Cashman J, Clark-Lewis I, Eaves A, Eaves C. Stromal-derived factor 1 inhibits the cycling of very primitive human hematopoietic cells in vitro and in NOD/SCID mice. Blood. 2002;99:792–9. doi: 10.1182/blood.v99.3.792. [DOI] [PubMed] [Google Scholar]
- 23.Kim CH, Broxmeyer HE. In vitro behavior of hematopoietic progenitor cells under the influence of chemoattractants: stromal cell-derived factor-1, steel factor, and the bone marrow environment. Blood. 1998;91:100–10. [PubMed] [Google Scholar]
- 24.Chiu CP, Dragowska W, Kim NW, Vaziri H, Yui J, Thomas TE, et al. Differential expression of telom-erase activity in hematopoietic progenitors from adult human bone marrow. Stem Cells. 1996;14:239–48. doi: 10.1002/stem.140239. [DOI] [PubMed] [Google Scholar]
- 25.Glimm H, Eisterer W, Lee K, Cashman J, Holyoake TL, Nicolini F, et al. Previously undetected human hematopoietic cell populations with short-term repopulating activity selectively engraft NOD/SCID-beta2 microglobulin-null mice. J Clin Invest. 2001;107:199–206. doi: 10.1172/JCI11519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lemoine FM, Humphries RK, Abraham SD, Krystal G, Eaves CJ. Partial characterization of a novel stromal cell-derived pre-B-cell growth factor active on normal and immortalized pre-B cells. Exp Hematol. 1988;16:718–26. [PubMed] [Google Scholar]
- 27.Taswell C. Limiting dilution assays for the determination of immuno-competent cell frequencies. J Immunol. 1981;126:1614–9. [PubMed] [Google Scholar]
- 28.Rose DM, Cardarelli PM, Cobb RR, Ginsberg MH. Soluble VCAM-1 binding to alpha 4 integrins is cell-type specific and activation dependent and is disrupted during apoptosis in T cells. Blood. 2000;95:602–9. [PubMed] [Google Scholar]
- 29.Luque A, Gomez M, Puzon W, Takada Y, Sanchez-Madrid F, Cabanas C. Activated conformations of very late activation integrins detected by a group of antibodies (HUTS) specific for a novel regulatory region (355–425) of the common beta 1 chain. J Biol Chem. 1996;271:11067–75. doi: 10.1074/jbc.271.19.11067. [DOI] [PubMed] [Google Scholar]
- 30.Arroyo AG, Sanchez-Mateos P, Campanero MR, Martin-Padura I, Dejana E, Sanchez-Madrid F. Regulation of the VLA integrin-ligand interactions through the beta 1 subunit. J Cell Biol. 1992;117:659–70. doi: 10.1083/jcb.117.3.659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miyake K, Weissman IL, Green-berger JS, Kincade PW. Evidence for a role of the integrin VLA-4 in lym-phohemopoiesis. J Exp Med. 1991;173:599–607. doi: 10.1084/jem.173.3.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ramirez M, Segovia JC, Benet I, Arbona C, Guenechea G, Blaya C, et al. Ex vivo expansion of umbilical cord blood (UCB) CD34+ cells alters the expression and function of alpha 4 beta 1 and alpha 5 beta 1 integrins. Br J Haematol. 2001;115:213–21. doi: 10.1046/j.1365-2141.2001.03084.x. [DOI] [PubMed] [Google Scholar]
- 33.Chute JP, Saini AA, Kampen RL, Wells MR, Davis TA. A comparative study of the cell cycle status and primitive cell adhesion molecule profile of human CD34+ cells cultured in stroma-free versus porcine microvascular endothelial cell cultures. Exp Hematol. 1999;27:370–9. doi: 10.1016/s0301-472x(98)00004-6. [DOI] [PubMed] [Google Scholar]
- 34.Reems JA, Mielcarek M, Torok-Storb B. Differential modulation of adhesion markers with ex vivo expansion of human umbilical CD34+ progenitor cells. Biol Blood Marrow Transplant. 1997;3:133–41. [PubMed] [Google Scholar]
- 35.Springer TA. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994;76:301–14. doi: 10.1016/0092-8674(94)90337-9. [DOI] [PubMed] [Google Scholar]
- 36.Chan JR, Cybulsky MI. Detection of high-affinity alpha4-integrin upon leukocyte stimulation by chemoat-tractants or chemokines. Methods Mol Biol. 2004;239:261–8. doi: 10.1385/1-59259-435-2:261. [DOI] [PubMed] [Google Scholar]
- 37.Garcia-Gila M, Cabanas C, Garcia-Pardo A. Analysis of the activation state of alpha4beta1 integrin in human B cell lines derived from myeloma, leukemia or lymphoma. FEBS Lett. 1997;418:337–40. doi: 10.1016/s0014-5793(97)01407-5. [DOI] [PubMed] [Google Scholar]
- 38.Grabovsky V, Feigelson S, Chen C, Bleijs DA, Peled A, Cinamon G, et al. Subsecond induction of alpha4 integrin clustering by immobilized chemokines stimulates leukocyte tethering and rolling on endothelial vascular cell adhesion molecule 1 under flow conditions. J Exp Med. 2000;192:495–506. doi: 10.1084/jem.192.4.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Peled A, Petit I, Kollet O, Magid M, Ponomaryov T, Byk T, et al. Dependence of human stem cell engraftment and repopulation of NOD/SCID mice on CXCR4. Science. 1999;283:845–8. doi: 10.1126/science.283.5403.845. [DOI] [PubMed] [Google Scholar]
- 40.Frenette PS, Subbarao S, Mazo IB, von Andrian UH, Wagner DD. Endothelial selectins and vascular cell adhesion molecule-1 promote hematopoietic progenitor homing to bone marrow. Proc Natl Acad Sci USA. 1998;95:14423–8. doi: 10.1073/pnas.95.24.14423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Priestley GV, Scott LM, Ulyanova T, Papayannopoulou T. Lack of α4 integrin expression in stem cells restricts competitive function and self-renewal activity. Blood. 2006;107:2959–67. doi: 10.1182/blood-2005-07-2670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kovach NL, Carlos TM, Yee E, Harlan JM. A monoclonal antibody to beta 1 integrin (CD29) stimulates VLA-dependent adherence of leukocytes to human umbilical vein endothelial cells and matrix components. J Cell Biol. 1992;116:499–509. doi: 10.1083/jcb.116.2.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.van Kooyk Y, Figdor CG. Avidity regulation of integrins: the driving force in leukocyte adhesion. Curr Opin Cell Biol. 2000;12:542–7. doi: 10.1016/s0955-0674(00)00129-0. [DOI] [PubMed] [Google Scholar]
- 44.Peled A, Grabovsky V, Habler L, Sandbank J, Arenzana-Seisdedos F, Petit I, et al. The chemokine SDF-1 stimulates integrin-mediated arrest of CD34+ cells on vascular endothelium under shear flow. J Clin Invest. 1999;104:1199–211. doi: 10.1172/JCI7615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cerny J, Dooner M, McAuliffe C, Habibian H, Stencil K, Berrios V, et al. Homing of purified murine lymphohematopoietic stem cells: a cytokine-induced defect. J Hematother Stem Cell Res. 2002;11:913–22. doi: 10.1089/152581602321080574. [DOI] [PubMed] [Google Scholar]