Abstract
Objective
To estimate the probability of reaching the criteria for starting highly active antiretroviral therapy (HAART) in a prospective cohort of adult HIV-1 seroconverters in Abidjan, Côte d’Ivoire.
Methods
We recruited participants from HIV-positive donors at the blood bank of Abidjan for whom the delay since the estimated date of seroconversion (midpoint between last negative and first positive HIV-1 test) was ≤ 36 months. Participants were offered early trimethoprim-sulfamethoxazole (cotrimoxazole) prophylaxis, twice-yearly measurement of CD4 count and we made standardized records of morbidity. We used the Kaplan–Meier method to estimate the probability of reaching the criteria for starting HAART according to WHO 2006 guidelines.
Findings
217 adults (77 women (35%)) were followed up during 668 person-years (PY). The most frequent diseases recorded were mild bacterial diseases (6.0 per 100 PY), malaria (3.6/100 PY), herpes zoster (3.4/100 PY), severe bacterial diseases (3.1/100 PY) and tuberculosis (2.1/100 PY). The probability of reaching the WHO 2006 criteria for HAART initiation was estimated at 0.09, 0.16, 0.24, 0.36 and 0.44 at 1, 2, 3, 4 and 5 years, respectively.
Conclusion
Our data underline the incidence of the early HIV morbidity in an Ivorian adult population and provide support for HIV testing to be made more readily available and for early follow-up of HIV-infected adults in West Africa.
Résumé
Objectif
Estimer la probabilité de remplir les critères de mise en route d’un traitement antirétroviral hautement actif (HAART) dans le cadre d’une étude prospective portant sur une cohorte d’adultes ayant subi une séroconversion au VIH-1, à Abidjan (Côte d’Ivoire).
Méthodes
Nous avons recruté des sujets parmi les donneurs positifs pour le VIH d’une banque de sang d’Abidjan pour lesquels le délai écoulé depuis la date estimée de la séroconversion (date située à mi-distance entre le dernier test négatif et le premier test positif pour le VIH-1) était ≤ 36 mois. Nous avons proposé à ces sujets un traitement prophylactique précoce par le triméthoprime-sulfaméthoxazole (cotrimoxazole) et une numération deux fois par an des lymphocytes T CD4. Nous avons procédé à un enregistrement normalisé de la morbidité. Nous avons utilisé la méthode de Kaplan-Meier pour estimer la probabilité de remplir les critères de mise en route d’un traitement de type HAART selon les Directives OMS de 2006.
Résultats
217 adultes, dont 77 femmes (35%), ont été suivis sur 668 personnes-ans. Les maladies les plus fréquemment relevées chez eux étaient des affections bactériennes bénignes (6,0/100 personnes-ans), des paludismes (3,6/100 personnes-ans), des zonas (3,4/100 personnes-ans), des affections bactériennes graves (3,1/100 personnes-ans) et des tuberculoses (2,1/100 personnes-ans). La probabilité de remplir les critères 2006 de l’OMS concernant la mise en route d’un traitement HAART a été estimée à 0,09 ; 0,16 ; 0,24 ; 0,36 et 0,44 respectivement pour les années 1, 2, 3, 4 et 5.
Conclusion
Nos données font ressortir l’incidence de la morbidité précoce liée au VIH parmi une population adulte ivoirienne et confirment la nécessité de rendre plus accessible le dépistage de ce virus et de pratiquer un suivi précoce des adultes séropositifs en Afrique de l’Ouest.
Resumen
Objetivo
Estimar la probabilidad de satisfacer los criterios para comenzar la terapia antirretroviral de gran actividad (TARGA) en una cohorte prospectiva de seroconvertidos para el VIH-1 en el banco de sangre de Abidján, África Occidental.
Métodos
Se invitó a participar en la cohorte a todos los donantes de sangre en los que habían transcurrido ≤ 36 meses desde la fecha estimada de seroconversión (punto medio entre la última prueba negativa y la primera positiva para el VIH-1). El seguimiento incluyó la profilaxis temprana con cotrimoxazol, el recuento semestral de CD4 y la documentación normalizada de la morbilidad. Mediante el método de Kaplan–Meier calculamos la probabilidad de cumplir los criterios para iniciar la TARGA de acuerdo con las directrices de la OMS de 2006.
Resultados
Se siguió la evolución de 217 adultos (35% de mujeres) durante 668 años-persona. Las dolencias más frecuentes fueron enfermedades bacterianas leves (incidencia de 6,0 por 100 años-persona [/100 AP]), malaria (3,6/100 AP), herpes zóster (3,4/100 AP), enfermedades bacterianas graves (3,1/100 AP) y tuberculosis (2,1/100 AP). La probabilidad de cumplir los criterios de la OMS de 2006 para comenzar la TARGA se estimó en 0,09, 0,16, 0,24, 0,36 y 0,44 a los uno, dos, tres, cuatro y cinco años, respectivamente.
Conclusión
Estos datos destacan la incidencia de morbilidad precoz por VIH en una población adulta de África Occidental y respaldan la idea de realizar un cribado temprano de la infección por VIH y un seguimiento temprano de los adultos infectados por el virus en esa parte de África.
ملخص
الەدف
استەدفت ەذە الدراسة تقييم احتمالية الوصول إلى المعايير التي تستوجب بدء المعالجة الفعالة بمضادات الفيروسات القەقرية في مجموعة من البالغين الذين تحولوا من إيجابية المصل إلى سلبية المصل خلال سبع سنوات، في أبيدجان، كوت ديفوار.
الطريقة
قمنا بتعيين المشاركين في الدراسة من بين الإيجابيين لفيروس الإيدز، المتبرعين بالدم في بنك الدم في أبيدجان، الذين كانت مدة تأخرەم عن تاريخ التحول من الإيجابية إلى السلبية (النقطة المتوسطة بين آخر اختبار سلبي وأول اختبار إيجابي لفيروس الإيدز من النمط 1) 36 شەراً أو أقل. وأُعطي للمشاركين في الدراسة جرعات وقائية من الكوتريموكسازول، مع تعداد الخلايا اللمفية CD4 مرتين في العام، كما أعددنا سجلات موحدة للمراضة، واتبعنا طريقة كابلان ماير لتقدير احتمالية تلبية المعايير التي توجب المعالجة الفعَّالة بمضادات الفيروسات القەقرية وفقاً للدلائل الإرشادية لمنظمة الصحة العالمية لسنة 2006.
الموجودات
تمت متابعة 217 بالغاً (منەم 77 امرأة، أي بنسبة 35%) على مدى 668 شخص عام. وكانت الأمراض المسجلة الأكثر تكراراً ەي أمراض جرثومية خفيفة (6 لكل 100 شخص عام)، والملاريا (3.6 لكل 100 شخص عام)، والەربس النطاقي (3.4 لكل 100 شخص عام)، وأمراض جرثومية وخيمة (3.1 لكل 100 شخص عام)، والسل (2.1 لكل 100 شخص عام). وتم تقديـر احتمالية تلبية معايير منظمة الصحة العالمية لسنة 2006، التي تستوجب بدء المعالجة الفعّالة بمضادات الفيروسات القەقرية، على النحو التالي: 0.09 في العام الأول، 0.16 في العام الثاني، 0.24 في العام الثالث، 0.36 في العام الرابع، 0.44 في العام الخامس.
الاستنتاج
تؤكد معطياتنا وقوع إصابات مبكِّرة بفيروس الإيدز بين مجموعة من السكان البالغين في كوت ديفوار، وتدعم ەذە البيانات ضرورة القيام باختبار فيروس الإيدز وأەمية المتابعة المبكِّرة للبالغين المصابين بالفيروس في غرب أفريقيا.
Introduction
Experts from the World Health Organization (WHO) and the United Nations Joint Programme on AIDS (UNAIDS) have estimated that at the end of 2005, 65% of the 39 million people with HIV worldwide were living in sub-Saharan Africa.1
In Africa, only a small proportion of people who need highly active antiretroviral therapy (HAART) are currently being treated. However, the number who receive HAART has been increasing rapidly since 2004, and will continue to increase within the next few years thanks to national and international funds and initiatives.
In 2003 and 2006, WHO experts defined criteria for initiating HAART in low-resource areas based on clinical criteria and, depending on the availability of laboratory facilities, either CD4 count or total lymphocyte count. Since cohort studies of patients from the time of HIV seroconversion are rare, little is known about the natural history of HIV/AIDS in sub-Saharan Africa and, therefore, about the probability of patients reaching WHO clinical and/or biological HAART starting criteria.
Here, we describe early HIV and non-HIV morbidity, time to first WHO-stage II, III or IV clinical events, and time to reach WHO criteria for starting HAART in a cohort of HIV-infected adults recruited shortly after their estimated date of HIV-seroconversion at the national blood bank of Abidjan, Côte d’Ivoire.
Methods
Patients
Recruitment for the The Primo-CI ANRS 1220 prospective cohort started in 1997 in the HIV care clinic (FonSIDA clinic) of the Centre National de Transfusion Sanguine (CNTS), at the national blood bank of Abidjan.2 The study protocol was approved by the ethics committees of the national Ivorian program on AIDS and the institutional review board of the French Agency for Research on AIDS (ANRS).
In the Côte d’Ivoire, blood donors are adult volunteers who are not paid. Blood donations are limited to five per year in men and four per year in women. Informed consent to test for HIV, syphilis, hepatitis B and hepatitis C is obtained at each donation. Patients are informed that they can, if they wish, obtain results of these tests at the FonSIDA clinic after a post-test counselling session.
From 9 June 1997 enrolment in the Primo-CI cohort was proposed to each blood donor who: (i) was diagnosed with HIV-1 or HIV-1 and HIV-2 co-infection during a blood donation; (ii) had been HIV-seronegative at the preceding donation; (iii) had returned to the clinic for their HIV test result; and (iv) had a delay of ≤ 36 months since the estimated date of seroconversion. We used the midpoint between the last negative and the first positive HIV test to estimate the date of seroconversion.
After obtaining written informed consent to participate in this study, we asked patients to return to the clinic every six months, or at any time in the case of medical problems. All drugs, investigations, hospital stays and transport to and from the clinic were offered free of charge.
Follow-up
We used standardized forms to record baseline and follow up sociodemographic and clinical data. Patients were examined by clinicians at enrolment and every six months. Patients also had free access to services at the study clinic between the six-month visits. Symptomatic patients were managed according to pre-defined standardized algorithms, including laboratory X-ray investigations and standardized first-line treatment regimens for most frequent syndromes.3 Patients needing parenteral treatment lasting less than 12 hours were managed in the day-care hospital at the study clinic. For life-threatening diseases or for conditions necessitating overnight care, patients were referred to the Treichville University Hospital in Abidjan. All clinical events were reviewed by an event documentation committee. The diagnostic criteria were the same as those used in the Cotrame cohort, a cohort of HIV-infected adults followed up in Abidjan during the same period, under the supervision of the same Franco-Ivoirian team, and for which procedures are described elsewhere.3,4 Autopsies were not performed.
Tuberculosis prophylaxis was not offered, as it is not recommended by the Côte d’Ivoire program on AIDS and tuberculosis. Trimethoprim-sulfamethoxazole (cotrimoxazole) prophylaxis (800/160 mg daily) was systematically offered to all patients at enrolment. Before February 2001, no antiretroviral treatment was available. Since February 2001, the clinic became one of the centres of the UNAIDS/Côte d’Ivoire initiative for improving access to HIV drugs, and HAART was provided in accordance with international guidelines for resource-limited settings.5,6
Biological measurements
The diagnosis of HIV infection was based on two repeated reactive enzyme-linked immunosorbent assays (ELISA) (Murex ICE 1–0-2 (Abbott, North Chicago, IL, USA) and Vironostika HIV Uni-Form II (Organon Teknika BV, Boxtel, Netherlands)). Before enrolment, we confirmed HIV infection and differentiated between HIV-1 and HIV-2 using two ELISAs (Murex ICE 1–0-2, and Pepti-Lav 1-2 (Pasteur Diagnostics, Marnes-la-Coquette, France)).
Blood samples were collected at inclusion and every six months thereafter to measure CD4 count (FACScan, Becton Dickinson, Aalst-Erembodegem, Belgium) and plasma HIV-1 RNA level (HIV-1 Monitor, version 1.5; Roche Diagnostics, Indianapolis IN, USA).
Microbiological procedures were the same as those used in the Cotrame cohort.3,4
Statistical analysis
Baseline was the date of inclusion in the cohort. For this analysis, data were censored on 31 December 2004 for patients who were still living, or the date of death for patients who had died. For patients who were lost to follow-up before 31 December 2004, data were censored at the date of their last contact with the study team. However, we used tracing procedures for up to one year after the date of data censoring. For patients whose last contact with the study team was before 31 December 2004 but who, via the tracing system, were found to be alive or have died during 2005 were considered to be alive on 31 December 2004.
The incidence of a given event was defined as the number of patients with an event per 100 patient-years (PY) of at-risk follow-up. The at-risk period began at enrolment and continued to the date of the first event or to the end of the study period. Patients were censored on HAART initiation, first event or default. Default was the date of the last contact with the cohort team for all patients who were not receiving HAART and whose last contact was before 31 December 2004.
The analyses had three steps. First, we used the Kaplan–Meier method to estimate the probability of survival and event-free survival from enrolment to first WHO stage II, III or IV defining event, and the probability of remaining free of criteria for starting HAART. Criteria for HAART initiation were successively defined in accordance with WHO 2003–2006 recommendations for settings where measurement of CD4 count was possible (WHO stage IV; WHO stage III with CD4 < 350/mm³; or CD4 < 200/mm³, irrespective of clinical stage);6,7 and WHO 2003–2006 recommendations for settings where measurement of CD4 count was unavailable (WHO stage III or IV; or WHO stage II and total lymphocyte count < 1200/mm³).6,7
Second, in order to assess how initiating HAART at earlier stages of HIV disease would affect the probability of starting treatment, we modified WHO criteria and considered the effect of offering HAART to all patients at WHO stage III irrespective of CD4 count, and all patients at stage II with CD4 < 350/mm³. Following this option, criteria for starting HAART became: WHO stage III or stage IV; WHO stage II with CD4 < 350/mm³; or CD4 < 200/mm³ irrespective of clinical stage. Throughout the article, we refer to these parameters as “alternative early-initiation criteria”.
Third, in a second set of analyses, survival and event-free survival were also estimated from the estimated date of seroconversion (and not from the date of enrolment in the cohort). Incidence rates and survival probabilities were given with 95% confidence interval (95% CI).
Results
Patients
By the end of 2004, 217 seroconverters (216 with HIV-1 and one with HIV-1 and HIV-2 co-infection) were enrolled in the cohort. Median time between the last HIV-negative test and the first HIV-positive test was seven months (interquartile range (IQR) 3–17). Median time from the estimated date of seroconversion to enrolment in the cohort was 9 months (IQR 5–18). Other baseline and follow-up characteristics are shown in Table 1.
Table 1. Baseline and follow-up characteristics of 217 HIV-1 seroconverters from the ANRS 1220 cohort study, Abidjan, 1997–2004.
| Baseline characteristica | ||
|---|---|---|
| Male sex | 140 | (65%) |
| Age in years (median (range)) | 29 | (19 – 53) |
| Heterosexual transmission | 215 | (99%) |
| Estimated time from seroconversion in months (median (IQR)) | 8.9 | (4.9 –18.0) |
| Schooling | ||
| Illiterate | 13 | (6%) |
| ≤ Primary school level | 48 | (22%) |
| ≥ Secondary school level or above | 156 | (72%) |
| Unemployed | 93 | (43%) |
| Marital status | ||
| Single | 134 | (62%) |
| Free union or married | 74 | (34%) |
| Divorced or widowed | 9 | (4%) |
| CD4-cell count ≤ 350/mm³ | 73 | (34%) |
| HIV-1 RNA level in copies/mL (median (IQR)) | 4.7 | (4.1–5.2) |
| CD4-cell count in per mm3 (median (IQR)) | 461 | (333–650) |
| Haemoglobin in g/L (median (IQR)) | 12.7 | (11.6–14.0) |
| Hepatitis B surface antigen positive serum | 9 | (4%) |
| Body mass index in kg/m2 (median (IQR)) | 22.2 | (20.5–24.7) |
| Status at study termination | ||
| HAART non initiated | 159 | (73%) |
| Dead | 12 | (6%) |
| Lost to follow-upc | 21 | (10%) |
| Alive | 126 | (58%) |
| HAART initiated | 58 | (27%) |
| Dead | 2 | (1%) |
| Lost to follow-up | 0 | (0%) |
| Alive | 56 | (26%) |
| Cumulative follow-up (person-years) | 668 | – |
| Follow-up in months (median (IQR)) | 33.5 | (18.0, 54.1) |
IQR, interquartile range a Data are number (%) unless otherwise stated. b Last contact with study team before 31 December 2004 and no further information on vital status at 31 December 2005.
At inclusion, 146 (67%) patients reported symptoms compatible with an acute retroviral infection syndrome in the preceding months, including acute fever (n = 125), rash (n = 17), pharyngitis (n = 31), acute diarrhoea (n = 44) and herpes zoster (n = 5). At baseline examination, 17 patients had polylymphadenopathy. By the end of the study period, 58 patients had started HAART. Of these patients on treatment, none was lost to follow-up and all but one patient (a woman who continued in active follow-up but refused to continue antiretroviral drugs) were still receiving HAART at study termination.
Survival
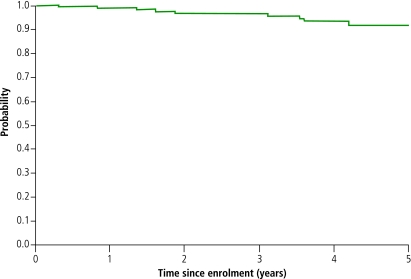
Twelve patients died during follow-up, including eight (67% of deaths) at Centres for Disease Control and Prevention (CDC) clinical stage C. At the time of death, the median last available CD4 count was 94/mm³ (IQR 19–372) and the median last plasma HIV-1 RNA 5.1 log10 copies/mm³ (range: 4.8–6.1). Causes of death included cerebral toxoplasmosis (n = 1, last CD4 = 9/mm³); tuberculosis (n = 4, last CD4 = 4/mm³, 27/mm³, 22/mm³ and 374/mm³); and pneumococcus meningitis (n = 1, last CD4 = 666/mm³). Cause of death was unknown in the remaining patients (n = 6, last CD4 = 5/mm³, 26/mm³, 161/mm³, 372/mm³, 288/mm³ and 572/mm³). Of the seven patients who had a CD4 count less than 200/mm³ at the time of death, five died before HAART was available in Abidjan and two died after the Côte d’Ivoire HAART programme had begun, but they refused treatment. The probability of survival at 1, 2, 3, 4 and 5 years after enrolment was estimated at 0.99 (95% CI 0.97–1.00), 0.96 (95% CI 0.93–0.99), 0.96 (95% CI 0.93–0.99), 0.93 (95% CI 0.88–0.98) and 0.92 (95% CI 0.86–0.98), respectively. The probability of survival at 1, 2, 3, 4 and 5 years after the estimated date of seroconversion was 1.00 (95% CI 0.98–1.00), 0.99 (95%CI 0.98––1.00), 0.98 (95% CI 0.95–1.00), 0.96 (95% CI 0.93–1.00) and 0.94 (95% CI 0.89–0.99), respectively (Fig. 1).
Fig. 1.
Survival in the ANRS 1220 cohort study of HIV-1 seroconverters, Abidjan, 1997–2004
Morbidity
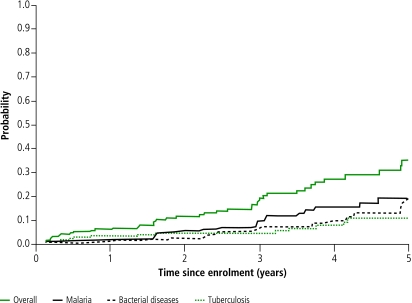
During follow-up, 119 events included in the WHO staging system occurred in 62 patients, including 55 stage II defining events (38 patients), 44 stage III defining events (30 patients), 20 stage IV defining events (15 patients). We recorded 316 events (77 patients) that are not included in the WHO staging system. The incidence of WHO stage II, III and IV defining diseases was 6.1/100 PY, 4.7/100 PY and 2.2/100 PY, respectively. Diagnoses are detailed in Table 2. Overall, the most frequent diseases were mild non-classifying bacterial diseases (incidence 6.0/100 PY), malaria (3.6/100 PY), herpes zoster (3.4/100 PY), tuberculosis (2.1/100 PY) and severe WHO stage III or IV classifying bacterial diseases (3.1/100 PY). The probabilities of developing malaria, tuberculosis, and/or severe bacterial diseases within the first 5 years of inclusion in the cohort are shown in Fig. 2. Of the 14 episodes of tuberculosis, four led to death and the remaining 10 patients were considered cured according to international criteria. Only one of the 23 of episodes of severe bacterial diseases, and none of the 25 malaria episodes, were considered as a cause of death.
Table 2. Causes of morbidity in the ANRS 1220 cohort study of HIV-1 seroconverters, Abidjan, 1997 – 2004.
| Classifying diseases | Patients (n) | Events (n) |
|---|---|---|
| WHO stage II | 38 | 55 |
| Herpes zoster | 22 | 23 |
| Fungal nail infections | 7 | 8 |
| Angular stomatitis | 9 | 11 |
| Prurigo | 12 | 12 |
| Sinusitis | 1 | 1 |
| WHO stage III | 30 | 44 |
| Oral candidiasis | 10 | 13 |
| Pulmonary tuberculosis | 10 | 10 |
| Severe bacterial diseasesa | 14 | 18 |
| Vaginal candidiasis | 1 | 3 |
| WHO stage IV | 15 | 20 |
| Severe bacterial diseasesb | 7 | 8 |
| HIV wasting syndrome | 2 | 2 |
| Extra-pulmonary tuberculosis | 4 | 4 |
| Oesophageal candidiasis | 2 | 2 |
| Cryptococcal meningitis | 1 | 1 |
| HIV encephalopathy | 1 | 1 |
| Kaposi’s sarcoma | 1 | 1 |
| Cerebral toxoplasmosis | 1 | 1 |
| Non-classifying diseases | 77 | 316 |
| Malaria | 22 | 25 |
| Non-classifying bacterial diseasesc | 37 | 59 |
a Including pneumonia with bacteraemia (n = 3), pneumococcus meningitis (n = 1), bacterial enteritis (n = 11), sinusitis with bacteraemia (n = 1). b All Salmonella typhi bactaeramia. c Including sinusitis (n = 4), otitis (n = 12), enteritis (n = 4), cystitis (n = 5), uretritis (n = 5), skin abscess (n = 18), amygdalitis (n = 5), boils (n = 6), all with no WHO stage III or IV classifying diseases criteria.
Fig. 2.
Probability of developing tuberculosis, severe bacterial diseases, and/or malaria in the ANRS 1220 cohort study of HIV-1 seroconverters, Abidjan, 1997–2004
At the onset of the WHO classifying morbidity events, the median last available CD4 count was 319/mm³ for WHO stage II (IQR 214–472; range 22–994), 243/mm³ for WHO stage III (IQR 140–527, range 9–908) and 254/mm³ for WHO stage IV (IQR 146–303, range 9–576). When considering specific diseases, the median last available CD4 count was 388/mm³ for malaria (IQR 260–517), 303/mm³ for severe bacterial diseases (IQR 205–617), and 210/mm3 for tuberculosis (IQR 164–330). The median last available plasma HIV RNA was 4.9 log10 copies/mm³ for WHO stage II (IQR 4.5–5.3, range 3.6–6.0), 5.1 log10 copies/mm³ for WHO stage III (IQR 4.4–5.9, range 2.3–6.6) and 5.9 log10 copies/mm³ for WHO stage IV defining events (IQR 5.0–6.4, range 4.2–6.6).
Criteria for HAART initiation
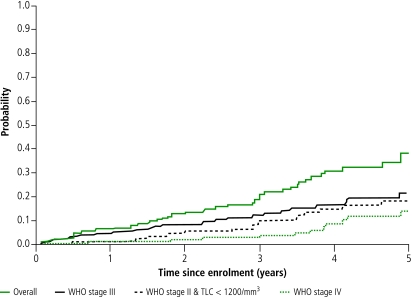
The 1-year, 2-year, 3-year, 4-year and 5-year probability of reaching criteria for starting HAART was estimated at 0.09 (95% CI 0.04–0.13), 0.16 (95% CI 0.10–0.22), 0.24 (0.17–0.31), 0.36 (0.28–0.45) and 0.44 (95% CI 0.35–0.55) following WHO 2003–2006 guidelines for settings with available CD4 count, 0.11 (95% CI 0.06–0.16), 0.23 (95% CI 0.16–0.29), 0.36 (95% CI 0.28–0.44), 0.49 (95% CI 0.39–0.58) and 0.56 (95% CI 0.46–0.66) following alternative early-initiation guidelines for settings with available CD4 count, and 0.06 (95% CI 0.03–0.10), 0.13 (95% CI 0.08–0.18), 0.19 (95% CI 0.13–0.26), 0.31 (95% CI 0.22–0.40) and 0.38 (95% CI 0.28–0.49) following WHO 2003–2006 guidelines for settings where CD4 counts were are not available, respectively. Fig. 3 shows the probability of reaching criteria for starting HAART over time from enrolment (overall and by criteria) in settings where CD4 measurement is not available, but total lymphocyte counts are possible.
Fig. 3.
Probability of reaching criteria for starting HAART using WHO 2003–2006 criteria for settings where CD4 counts is not available, ANRS 1220 cohort study of HIV-1 seroconverters, Abidjan, 1997–2004
Table 3 shows the first HAART initiation criterion that was met in our population when applying WHO 2003–2006 guidelines and alternative early-initiation criteria.
Table 3. Criteria for HAART initiation: first WHO criterion met in ANRS 1220 cohort study of HIV-1 seroconverters, Abidjan, 1997–2004.
| First criterion | CD4 count measurementsa |
|||
|---|---|---|---|---|
| Available |
Not available |
|||
| WHO 2003 & 2006 guidelines6,7 | Alternative early-initiation criteria guidelines | WHO 2003 & 2006 guidelines6,7 | ||
| CD4 < 200/mm³ | 43 (67%)b | 12 (21%)c | – | |
| CD4 < 350/mm³ & WHO stage III | 13 (21%) | – | – | |
| CD4 < 350/mm³ & WHO stage II | – | 21 (37%) | – | |
| WHO stage IV | 8 (12%) | 6 (11%) | 7 (14%) | |
| WHO stage III | – | 18 (31%) | 25 (49%) | |
| Total lymphocyte count < 1 200/mm3 & WHO stage II | – | – | 19 (37%) | |
a Data are number (%). b Including three patients who were previously at stage III with CD4 count > 350/mm³. c including five patients who were previously at stage II with CD4 count > 350/mm³.
Discussion
When the WHO 2003–2006 guidelines for starting HAART in settings with available CD4 count were applied to our cohort of adult HIV-1 seroconverters in Abidjan,6,7 we noted a probability of reaching criteria for HAART initiation of 9%, 16%, 24%, 36% and 44% at 1, 2, 3, 4 and 5 years, respectively. Unsurprisingly, these probabilities were 19–33% higher when applying alternative early-initiation criteria, which would include all patients to treatment who were at WHO stage III irrespective of CD4 count, and all patients at stage II with CD4 < 350/mm³. By contrast, when applying the WHO 2003–2006 guidelines for starting HAART in settings where CD4 counts are not available, the probability of starting HAART was 14–34% lower, which illustrates the difficulty in finding a suitable surrogate for CD4 counts.
Rates of survival, WHO stage IV, and CD4 counts < 200/mm³ at 5 years’ in our Ivorian population did not differ greatly from those reported in industrialized countries.8,9 Our results do, however, contrast with findings from studies in the Central African Republic, Haiti, Kenya, Thailand, and Uganda that have suggested that median time from seroconversion to first HIV diseases was shorter in these settings than in industrialized countries.10–14
In Kenya, estimates of the median duration to CDC stage IV-A and IV-C disease were 3.5 years and 4.4 years in a cohort of female sex workers.14 In the Ugandan cohort, only 17% of patients remained symptom-free 6 years after seroconversion.12 In the Thai cohort, median time to clinical AIDS and to a CD4 cell count < 200/mm³ was 7.4 years and 6.9 years, respectively.11 In the study from the Central African Republic, 35% of HIV-1 seroconverters died 3–5 years after seroconversion, and all died at AIDS stage.13 In Haiti, median time to first HIV symptoms was 3 years and the median survival time to death was 7.4 years.10
It is well known that there are differences between sub-Saharan Africa and industrialized countries with respect to HIV-morbidity. In African HIV-positive adults, tuberculosis and severe bacterial diseases are the most frequent causes of HIV morbidity.4,15 They are also early diseases, which can occur in patients with CD4 counts greater than 200 cells/mm³. Malaria is also recognized as another HIV-related disease that can occur soon after seroconversion.16 The early occurrence of both malaria and severe bacterial diseases is one of the main reasons that cotrimoxazole prophylaxis is recommended at an earlier stage in Africa than it is in Europe or in the USA.17
Since 1994, all HIV-infected adults attending the blood donor centre of Abidjan have been prescribed cotrimoxazole prophylaxis, irrespective of their CD4 count and of their clinical stage. Thus, all patients included in the Primo-CI cohort received cotrimoxazole, including those at WHO stage I and with CD4 > 500/mm³, and those included before the 1999 Côte d’Ivoire national consensus on cotrimoxazole. This decision to provide prophylaxis was made by staff at the blood donor medical centre, and although it might have seemed an over-reaction 10 years ago, it has recently been recognized as a valid option.18 This prophylaxis may have contributed to a decrease in early morbidity in our study group and could explain the difference in morbidity between our cohort and those in other low-resource settings.
Limitations
First, for our data to be generalizable, our population should be representative of the overall population of recently HIV-infected adult in Abidjan. Unfortunately, there is no way to compare our self-selected population of urban voluntary blood donors with the overall population of recent seroconverters, as our cohort is the only source of data on recent HIV seroconverters in Abidjan.
Second, most of the data we present here shows probabilities of remaining event-free from the date of inclusion in the cohort, and not from seroconversion. However, our population included recent seroconverters with median time from seroconversion to enrolment estimated at about nine months. Analyses to estimate the probability of remaining event-free from the estimated date of seroconversion systematically gave probabilities that were slightly higher than those calculated using time since enrolment in the cohort. Because the real date of seroconversion was unknown and because the natural history of HIV-infection including the risk of very early mortality and severe morbidity within the first months of seroconversion has never been described in Côte d’Ivoire we chose to use time since inclusion rather than that since the estimated date of seroconversion in our analyses.
Third, we censored follow-up data at HAART initiation, with the potential of informative censoring in survival analyses and morbidity-free probability analyses.19 On the other hand, this potential bias did not affect estimates of remaining free of criteria for HAART initiation, because all patients who started HAART in the cohort met the HAART starting criteria before their follow-up was censored.
In conclusion, our data provide valuable estimates of the probability of reaching the criteria for HAART initiation by time from first contact in a population of west African recent seroconverters. They support the hypothesis that median time to AIDS and survival in Africa do not differ from those in industrialized countries.20 However, our data also emphasize the significant incidence of the early morbidity in the first years following seroconversion in a west African urban population. This latter finding provides strong support in favour of more readily available HIV testing in this population so that HIV infection may be diagnosed and patients receiving care and follow-up as early as possible within the course of HIV disease. ■
Acknowledgments
This study was supported by the French National Agency for Research on AIDS (ANRS, Paris, France) and the Ministry of Public Health of Côte d’Ivoire, within the collaborative Programme PAC-CI.
Footnotes
Competing interests: none declared.
References
- 1.UNAIDS. WHO. AIDS epidemic update 2005. http://www.unaids.org/Epi2005/doc/report.html
- 2.Salamon R, Marimoutou C, Ekra D, Minga A, Nerrienet E, Huet C, et al. Clinical and biological evolution of HIV-1 seroconverters in Abidjan, Cote d’Ivoire, 1997-2000. J Acquir Immune Defic Syndr. 2002;29:149–57. doi: 10.1097/00042560-200202010-00007. [DOI] [PubMed] [Google Scholar]
- 3.Anglaret X, Dakoury-Dogbo N, Bonard D, Toure S, Combe P, Ouassa T, et al. Causes and empirical treatment of fever in HIV-infected adult outpatients, Abidjan, Cote d’Ivoire. AIDS. 2002;16:909–18. doi: 10.1097/00002030-200204120-00011. [DOI] [PubMed] [Google Scholar]
- 4.Anglaret X, Messou E, Ouassa T, Toure S, Dakoury-Dogbo N, Combe P, et al. Pattern of bacterial diseases in a cohort of HIV-1 infected adults receiving cotrimoxazole prophylaxis in Abidjan, Côte d’Ivoire. AIDS. 2003;17:575–84. doi: 10.1097/00002030-200303070-00013. [DOI] [PubMed] [Google Scholar]
- 5.Djomand G, Roels T, Ellerbrock T, Hanson D, Diomande F, Monga B, et al. Virologic and immunologic outcomes and programmatic challenges of an antiretroviral treatment pilot project in Abidjan, Côte d’Ivoire. AIDS. 2003;17:S5–15. doi: 10.1097/00002030-200317003-00002. [DOI] [PubMed] [Google Scholar]
- 6.WHO. Scaling up antiretroviral therapy in resource-limited settings: treatment guidelines for a public health approach. Vol. 2004: http://www.who.int/3by5/publications/en/arv_eng.pdf [PubMed]
- 7.WHO. Antiretroviral therapy for HIV infection in adults and adolescents in resource-limited settings: towards universal access: recommendations for a public health approach (2006 revision). Available from: http://www.who.int/hiv/pub/guidelines/WHO%20Adult%20ART%20Guidelines.pdf
- 8.Collaborative Group on AIDS Incubation and HIV Survival including the CASCADE EU Concerted Action. Concerted Action on SeroConversion to AIDS and Death in Europe. Time from HIV-1 seroconversion to AIDS and death before widespread use of highly-active antiretroviral therapy: a collaborative re-analysis. Lancet. 2000;355:1131–7. doi: 10.1016/S0140-6736(00)02061-4. [DOI] [PubMed] [Google Scholar]
- 9.Goujard C, Bonarek M, Meyer L, Bonnet F, Chaix ML, Deveau C, et al. CD4 cell count and HIV DNA level are independent predictors of disease progression after primary HIV type 1 infection in untreated patients. Clin Infect Dis. 2006;42:709–15. doi: 10.1086/500213. [DOI] [PubMed] [Google Scholar]
- 10.Deschamps MM, Fitzgerald DW, Pape JW, Johnson WD. HIV infection in Haiti: natural history and disease progression. AIDS. 2000;14:2515–21. doi: 10.1097/00002030-200011100-00014. [DOI] [PubMed] [Google Scholar]
- 11.Rangsin R, Chiu J, Khamboonruang C, Sirisopana N, Eiumtrakul S, Brown AE, et al. The Natural History of HIV-1 Infection in Young Thai Men After Seroconversion. J Acquir Immune Defic Syndr. 2004;36:622–9. doi: 10.1097/00126334-200405010-00011. [DOI] [PubMed] [Google Scholar]
- 12.Morgan D, Mahe C, Mayanja B, Whitworth J. Progression to symptomatic disease in people infected with HIV-1 in rural Uganda: prospective cohort study. BMJ. 2002;324:193–6. doi: 10.1136/bmj.324.7331.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Begaud E, Feindirongai G, Versmisse P, Ipero J, Leal J, Germani Y, et al. Broad spectrum of coreceptor usage and rapid disease progression in HIV-1-infected individuals from Central African Republic. AIDS Res Hum Retroviruses. 2003;19:551–60. doi: 10.1089/088922203322230914. [DOI] [PubMed] [Google Scholar]
- 14.Anzala OA, Nagelkerke NJ, Bwayo JJ, Holton D, Moses S, Ngugi EN, et al. Rapid progression to disease in African sex workers with human immunodeficiency virus type 1 infection. J Infect Dis. 1995;171:686–9. doi: 10.1093/infdis/171.3.686. [DOI] [PubMed] [Google Scholar]
- 15.Lucas SB, Hounnou A, Peacock C, Beaumel A, Djomand G, N’Gbichi JM, et al. The mortality and pathology of HIV infection in a West African city. AIDS. 1993;7:1569–79. doi: 10.1097/00002030-199312000-00005. [DOI] [PubMed] [Google Scholar]
- 16.Whitworth J, Morgan D, Quigley M, Smith A, Mayanja B, Eotu H, et al. Effect of HIV-1 and increasing immunosuppression on malaria parasitaemia and clinical episodes in adults in rural Uganda: a cohort study. Lancet. 2000;356:1051–6. doi: 10.1016/S0140-6736(00)02727-6. [DOI] [PubMed] [Google Scholar]
- 17.WHO. WHO Expert Consultation on Cotrimoxazole Prophylaxis in HIV Infection. WHO technical report series. Reference number: WHO/HIV/2006.01. Available from: http://www.who.int/hiv/pub/meetingreports/ctx/en/
- 18.Mermin J, Lule JR, Ekwaru JP, Pitter C. Should cotrimoxazole prophylaxis be taken by all adults with HIV in Africa? AIDS. 2005;19:845–6. doi: 10.1097/01.aids.0000168986.12832.a4. [DOI] [PubMed] [Google Scholar]
- 19.Laird NM. Missing data in longitudinal studies. Stat Med. 1988;7:305–15. doi: 10.1002/sim.4780070131. [DOI] [PubMed] [Google Scholar]
- 20.Morgan D, Mahe C, Mayanja B, Okongo J, Lubega R, Whitworth J. HIV-1 infection in rural Africa: is there a difference in median time to AIDS and survival compared with that in industrialized countries? AIDS. 2002;16:597–603. doi: 10.1097/00002030-200203080-00011. [DOI] [PubMed] [Google Scholar]