Abstract
Objective
To identify barriers to successful tuberculosis (TB) treatment in Tomsk, Siberia, by analysing individual and programmatic risk factors for non-adherence, default and the acquisition of multidrug resistance in a TB treatment cohort in the Russian Federation.
Methods
We conducted a retrospective cohort study of consecutively enrolled, newly detected, smear and/or culture-positive adult TB patients initiating therapy in a DOTS programme in Tomsk between 1 January and 31 December 2001.
Findings
Substance abuse was strongly associated with non-adherence [adjusted odds ratio (OR): 7.3; 95% confidence interval (CI): 2.89–18.46] and with default (adjusted OR: 11.2; 95% CI: 2.55–49.17). Although non-adherence was associated with poor treatment outcomes (OR: 2.4; 95% CI: 1.1–5.5), it was not associated with the acquisition of multi-drug resistance during the course of therapy. Patients who began treatment in the hospital setting or who were hospitalized later during their treatment course had a substantially higher risk of developing multidrug-resistant TB than those who were treated as outpatients (adjusted HRs: 6.34; 95% CI: 1.35–29.72 and 6.26; 95% CI: 1.02–38.35 respectively).
Conclusion
In this cohort of Russian TB patients, substance abuse was a strong predictor of non-adherence and default. DOTS programmes may benefit from incorporating measures to diagnose and treat alcohol misuse within the medical management of patients undergoing TB therapy. Multidrug-resistant TB occurred among adherent patients who had been hospitalized in the course of their therapy. This raises the possibility that treatment for drug-sensitive disease unmasked a pre-existing population of drug-resistant organisms, or that these patients were reinfected with a drug-resistant strain of TB.
Résumé
Objectif
Identifier les obstacles s’opposant au traitement avec succès de la tuberculose (TB) à Tomsk en Sibérie, par une analyse des facteurs de risque individuels et programmatiques de non-observance du traitement, d’abandon et d’acquisition d’une pharmacorésistance dans une cohorte traitée contre la TB en Fédération de Russie.
Méthodes
Nous avons mené une étude rétrospective sur une cohorte d’adultes récemment diagnostiqués comme tuberculeux par examen de frottis positif et/ou par culture et débutant un traitement dans le cadre d’un programme DOTS (autrefois appelé traitement de brève durée sous surveillance directe) à Tomsk, entre le 1er janvier et le 31 décembre 2001.
Résultats
Il existe de fortes associations entre la toxicomanie et la non observance du traitement (OR ajusté : 7,3 ; IC à 95% : 2,89-18,46) et son abandon (OR ajusté : 11,2 ; IC à 95% : 2,55-49,17). Si la non-observance du traitement est associée à un résultat thérapeutique insatisfaisant (OR : 2,4 ; IC à 95% : 1,1-5,5), elle ne l’est pas avec l’acquisition d’une pharmacorésistance. Les malades ayant débuté un traitement dans un cadre hospitalier ou ayant été hospitalisés ultérieurement au cours de leur traitement présentent un risque nettement plus élevé de développer une TB multirésistante que ceux traités en ambulatoire (OR ajustés : 6,34 ; IC à 95% 1,35-29,72 et 6,26 ; IC à 95% : 1,02-38,35, respectivement).
Conclusion
Dans cette cohorte de malades russes, la toxicomanie était un facteur prédictif fort de non-observance et d’abandon. Les programmes DOTS peuvent tirer profit de l’incorporation de mesures de diagnostic et de traitement des abus d’alcool dans le cadre d’une prise en charge médicale des malades traités contre la TB. Les TB multirésistantes apparaissent chez des malades observant leur traitement et hospitalisés dans le cadre de celui-ci. Cette observation laisse entrevoir la possibilité que le traitement des maladies pharmacosensibles démasque une population préexistante d’organismes pharmacorésistants ou que les malades concernés aient été réinfectés par une souche pharmacorésistante de TB.
Resumen
Objetivo
Identificar obstáculos al éxito del tratamiento de la tuberculosis (TB) en Tomsk (Siberia), analizando los factores de riesgo individuales y programáticos de incumplimiento y abandono del tratamiento y de adquisición de multirresistencia en una cohorte de pacientes tratados de TB en la Federación de Rusia.
Métodos
Hemos realizado un estudio de cohortes retrospectivo de pacientes adultos consecutivos con TB recién detectada (baciloscopia y/o cultivo positivo) en los que se inició un tratamiento con la estrategia DOTS (tratamiento breve bajo observación directa) en Tomsk entre el 1 de enero y el 31 de diciembre de 2001.
Resultados
El abuso de sustancias se asoció estrechamente al incumplimiento del tratamiento (OR ajustada: 7,3; IC95%: 2,89–18,46) y a su abandono (OR ajustada: 11,2; IC95%: 2,55–49,17). El incumplimiento se asoció a malos resultados terapéuticos (OR: 2,4; IC95%: 1,1–5,5), pero no a la adquisición de multirresistencia en el curso del tratamiento. En comparación con los pacientes que recibieron tratamiento ambulatorio, el riesgo de presentar multirresistencia fue significativamente mayor en aquellos que iniciaron el tratamiento en un hospital (HR ajustada: 6,34; IC95%: 1,35–29,72) o que fueron hospitalizados posteriormente en el curso de su tratamiento (HR ajustada: 6,26; IC95%: 1,02–38,35).
Conclusión
En esta cohorte de pacientes rusos con TB, el abuso de sustancias predijo bien el incumplimiento y el abandono del tratamiento. Los programas de DOTS pueden beneficiarse de la incorporación de medidas para diagnosticar y tratar el abuso de alcohol en pacientes sometidos a tratamiento antituberculoso. Se produjeron casos de TB multirresistente en pacientes que cumplieron su tratamiento pero fueron hospitalizados en el curso de éste. Esto plantea la posibilidad de que el tratamiento de la enfermedad sensible a los antituberculosos desenmascare una población preexistente de miroorganismos resistentes o de que estos pacientes se hayan reinfectado con cepas farmacorresistentes.
ملخص
الغرض
استەدفت ەذە الدراسة تحديد عوائق نجاح معالجة السل في مدينة تومسك، بسيبريا، عن طريق تحليل عوامل الاختطار الفردية والبرنامجية المؤدية إلى عدم الامتثال للمعالجة، والتخلُّف عنەا، واكتساب المقاومة للأدوية المتعدِّدة، وذلك في مجموعة أترابية تُعالج من السل في الاتحاد الروسي.
الطريقة
أجرينا دراسة أترابية استعادية لمجموعة من مرضى السل البالغين الإيجابيي اللطاخة و/أو الإيجابيي المزرعة، الذين اكتُشفوا حديثاً والتحقوا تباعاً ببرنامج المعالجة القصيرة الأمد للسل تحت الإشراف المباشر في مدينة تومسك، في المدة من 1 كانون الثاني/يناير إلى 31 كانون الثاني/يناير 2001.
الموجودات
لوحظ ارتباط قوي بين معاقرة مواد الإدمان وبين عدم الامتثال (نسبة الاحتمال المصحَّحة: 7.3؛ عند فاصلة ثقة 95 %: 2.89 – 18.46)، وكذلك بين التخلُّف عن المعالجة (نسبة الاحتمال المصحَّحة: 11.2؛ عند فاصلة ثقة 95 %: 2.55 – 49.17). وبرغم ارتباط عدم الامتثال بضعف حصائل المعالجة (نسبة الاحتمال المصحَّحة: 2.4؛ عند فاصلة ثقة 95 %: 1.1 – 5.5)، إلا أن عدم الامتثال لم يرتبط باكتساب المقاومة للأدوية المتعدِّدة أثناء المعالجة. ولوحظ أن المرضى الذين بدأوا المعالجة في المستشفى أو أُدخلوا المستشفى بعد ذلك أثناء المعالجة، كانوا أكثر تعرُّضاً لمخاطر اكتساب السل المقاوم لأدوية متعدِّدة (معدل المخاطرة المصحَّح: 6.34؛ عند فاصلة ثقة 95 %: 1.35 – 29.72)، بالمقارنة مع من عولجوا كمرضى خارجيـين (معدل المخاطرة المصحَّح: 6.26؛ عند فاصلة ثقة 95 %: 1.02 – 38.35).
الاستنتاج
في ەذە المجموعة من مرضى السل الروسيـين، كانت معاقرة مواد الإدمان عاملاً قوياً في التنبُّؤ بعدم الامتثال للمعالجة والتخلُّف عنەا. ويمكن لبرامج المعالجة القصيرة الأمد للسل تحت الإشراف المباشر أن تستفيد من عملية إدماج تدابير تشخيص ومعالجة إساءة استعمال الكحول في المعالجة الطبية للمرضى الذين يُعالجون من السل. وقد لوحظ وقوع السل المقاوِم لأدوية متعدِّدة بين المرضى الممتثلين للمعالجة الذين أُدخلوا المستشفى أثناء المعالجة. وەذا يطرح إمكانية أن تكون معالجة الأمراض الحساسة للأدوية قد كشفت عن وجود كائنات حيَّة مقاومة للأدوية، أو أن يكون ەؤلاء المرضى قد عاودتەم العدوى بذرية لجراثيم السل مقاومة للأدوية.
Introduction
Background
After a long period of decline, tuberculosis (TB) incidence and mortality in the Russian Federation rose dramatically in the 1990s and peaked in 2000.1 During the same period, the proportion of notified TB patients cured by therapy fell precipitously from 90% in 1985 to an estimated 72% in 2000. Despite the Russian Federation’s introduction and gradual uptake over the past decade of the DOTS strategy, treatment success rates have remained consistently low even though case notifications have declined.2 WHO attributes these high failure rates to drug resistance and high rates of default and death among Russian patients receiving DOTS.3
Before addressing these problems to improve DOTS outcomes, it is necessary to identify the proximal causes of death, default and the acquisition of drug resistance among TB therapy patients. In an earlier study, we reported the causes of death of patients undergoing DOTS treatment in Tomsk, Siberia, from January 2002 to December 2003.4 We observed a 9.6% death rate during TB treatment – due not only to TB but also to co-morbid conditions such as alcoholism and cardiovascular disease. We also found that both alcoholism and late presentation contributed substantially to mortality.
Here, we present data on programmatic and individual risk factors for non-adherence, default and the acquisition of multidrug resistance (MDR) in a DOTS treatment cohort in Tomsk. Based on our findings, we propose several specific interventions that may improve treatment outcomes and reduce the acquisition of drug resistance in patients undergoing TB therapy in this setting.
Methods
Setting and programme description
We conducted this study in the Tomsk oblast of western Siberia, where the incidence and mortality rates for TB in 2001 were 109.3 and 18.3 per 100 000, respectively. Rates of MDR in Tomsk were among the highest reported worldwide; MDR among newly diagnosed patients rose from 6.5% in 1999 to 12.1% by 2002. In 1995 Tomsk was one of the first Russian Federation oblasts to implement the DOTS strategy.
Tomsk City TB Services (TTBS) oversees diagnosis, treatment and reporting of adult patients with TB. Suspects undergo sputum smear microscopy and culture at the time of diagnosis. Those who are culture-positive also undergo drug sensitivity testing to isoniazid, rifampicin, ethambutol, streptomycin and kanamycin. Susceptibility is determined using the absolute concentration method on Lowenstein-Jensen medium, based on the following drug concentrations: isoniazid 1 μg/ml, rifampicin 40 μg/ml, ethambutol 5 μg/ml and streptomycin 10 μg/ml. Massachusetts State Laboratory Institute, a supranational reference laboratory, provides external quality control.
Patients diagnosed with active TB are treated according to WHO recommendations.5 Those with multidrug-resistant TB (MDR-TB) are switched to an individualized regimen based on the drug resistance profile. Treatment is offered three ways: under direct supervision in an inpatient setting, at one of three outpatient clinics or through home-based care. Patients receive drugs daily in each of the outpatient settings. Home-based care is provided for those who are unable to attend outpatient clinics, with nurses delivering drugs directly to the patients. Some patients self-administered drugs during weekends and holidays, and a small proportion self-administered over half of their medications. Government social services provide free passes for public transport to all patients treated in ambulatory settings. Travel expenses are not provided for patients who have no public transport services. Patients undergoing TB treatment are assessed with repeat sputum smear, culture and drug-sensitivity testing (DST) in months 2, 3 and 5 as well as at the end of treatment and at six-month intervals thereafter.
Study design
We conducted a retrospective cohort study of newly detected smear- and/or culture-positive TB patients aged over 17 who were notified under DOTS and began TB treatment during the period from 1 January to 31 December 2001. We excluded patients who were admitted to psychiatric hospitals, were in prison, died within one month of beginning therapy or did not live within Tomsk city limits. Individual and programmatic risk factors as well as outcomes were assessed by reviewing patients’ charts and TB treatment records, and through a TB database set up by the TTBS. We then assessed risk factors for non-adherence, default and the development of MDR during therapy.
Exposure assessment
For each patient, we recorded the following information collected routinely for all patients undergoing TB therapy under the TTBS: age, gender, address, history of previous TB treatment, clinical signs at presentation, date of diagnosis, all sputum-smear results, all culture results, all drug-sensitivity profiles, number of missed doses, dates of missed doses, date of end of treatment, date of default, date of death, co-morbidities including HIV, employment status at beginning of treatment, history of previous incarceration and diagnosis of chronic alcoholism and/or drug addiction by a narcologist. Alcoholism is often underdiagnosed in the Russian Federation, therefore we also recorded any note of alcohol abuse that occurred during the treatment period. We classified patients’ proximity to their assigned clinic on the basis of their home address and the accessibility of public transport. Patients were classified as having co-morbidities potentially associated with side effects if they reported renal insufficiency, liver disease, diabetes mellitus, gastric ulcers, malignancies, cholecystitis or neurosyphilis.
Outcome assessment
We classified patients as non-adherent if they missed more than 20% of the prescribed doses during the treatment period recommended by WHO. In a sensitivity analysis, we identified patients who missed more than 50% of their prescribed doses. Treatment outcomes, including default, were classified according to WHO guidelines.6 Patients were classified as having acquired MDR during or subsequent to therapy if they were sensitive to either isoniazid or rifampicin on their first DST but were noted to be resistant to both agents on any later DST.
Statistical analysis
For univariate analyses of non-adherence and default, we used logistic regression to estimate odds ratios (ORs) and 95% confidence intervals (CIs). The Mantel-Haenszel χ² method or Fisher’s exact test were used to calculate p-values. Statistical tests were two-sided. We used separate logistic regression models to perform multivariate analyses of the outcomes, adherence and default. The multivariate model included relevant variables with p-values less than 0.2 in univariate analysis, and those for which we had strong expectation of an association. As a sensitivity analysis we repeated the multivariate analysis of risk factors for non-adherence, excluding those people who had self-administered more than 40% of their doses. We also assessed the univariate association between non-adherence and a binary variable, summarizing treatment outcomes as either poor (death, default or failure) or good (cure or treatment completion).
Kaplan–Meier survival analysis was used to estimate the time from initiation of therapy to acquisition of MDR-TB. For patients who did not reach the end-point, the data were censored at the time of their last DST. The MDR acquisition time was taken as the mid-point between the last DST without MDR and the first DST with MDR. The log rank test was used to compare time to MDR between strata. The Cox proportional hazards model was used for multivariate analysis. In a sub-analysis, we also assessed risk factors for early (within four months of treatment initiation) and late (6 months after treatment initiation) acquisition of MDR. Patients who acquired early MDR were excluded from the analysis of risk factors for late MDR. Analyses were performed using Stata (version 9.0) and SAS (version 9.1) software.
Ethics approval
This study was approved by the respective institutional review boards at Tomsk State Medical University on 21 June 2004 and at Brigham and Women’s Hospital on 17 September 2004.
Results
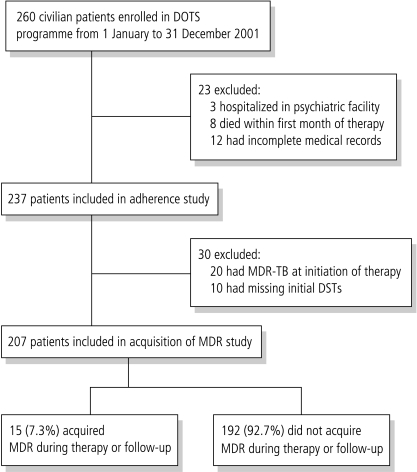
Of the 260 civilian adult patients enrolled in the DOTS treatment programme during the study period, 3 were residents of psychiatric facilities, 8 died during the first month of therapy and 12 had missing treatment records (Fig. 1). The remaining 237 patients were included in the analysis of non-adherence and default; there were 148 men and 89 women and the mean age was 40. Primary MDR was found in 20 (8.4%) of the patients, and 82 (34.5%) were found to be resistant to at least one drug at the time of diagnosis. Excluded patients were more likely to be male, unemployed, homeless and substance abusers. Among the 237 patients included in the study, 20 had MDR on initiation of therapy and 10 had missing initial DSTs; the remaining 207 participants were included in the analysis of MDR acquisition. The 30 patients with initial MDR or missing DSTs were more likely to be male and illicit drug users. All patients were HIV tested, but since only two were found to be HIV-positive we did not include HIV in our subsequent analyses.
Fig. 1.
Flow chart of study participants
DSTs, drug-sensitivity testing; MDR-TB, multidrug resistant TB.
Treatment outcomes are presented in Table 1 (available at http://www.who.int/bulletin/volumes/85/9/06-038331/en/index.html). More than half of those who died (8/14) did so within the first month of treatment. The overall mortality of patients undergoing DOTS therapy for TB is underestimated, as the data presented in Table 1 exclude results on these patients. Twenty-one (8.8%) of the patients in our cohort defaulted on therapy and 37 (15.6%) took fewer than 80% of their observed prescribed doses. Fifteen patients (6.3%) acquired MDR during the study period, seven during the course of treatment and eight during post-treatment follow-up.
Table 1. Outcomes for Tomsk TB treatment cohort (n = 237).
| Treatment resolution | Adherent | Not-adherent | Total | Proportion | |
|---|---|---|---|---|---|
| Successful outcome | Cured | 137 | 19 | 156 | 0.66 |
| Treatment completed | 2 | 0 | 2 | 0.01 | |
| Poor outcomea | Failed | 30 | 6 | 36 | 0.15 |
| Default | 13 | 8 | 21 | 0.09 | |
| Died | 5 | 1 | 6 | 0.03 | |
| Transferred | Transferred out | 8 | 3 | 11 | 0.05 |
| Transferred to DOTS Plus | 5 | 0 | 5 | 0.02 |
a Crude odds ratio, OR, for poor outcome given non-adherence = 2.43 (95% confidence interval, CI: 1.05–5.53).
Baseline characteristics of those who defaulted or were non-adherent are given in Table 2. In a multivariate model, substance abuse was identified as the only factor that was strongly associated with non-adherence with odds ratios for baseline alcohol dependence – 4.38 (95% CI: 1.58–12.60); reported alcohol use in a patient during therapy – 6.35 (95% CI: 2.27–17.75); and intravenous drug use – 16.64 (95% CI: 3.24–85.56) (Table 3). The adjusted odds ratio of non-adherence for those with any kind of substance abuse was 7.30 (95% CI: 2.89–18.46). Substance abuse was also strongly associated with default, with an odds ratio of 15.57 (95% CI: 3.46–70.07) among those with baseline alcoholism and 5.14 (95% CI: 0.87–30.25) for those with reported alcohol use. Patients with any form of substance abuse had an adjusted odds ratio for default of 11.20 (95% CI: 2.55–49.17). When this analysis was repeated, excluding patients for whom more than 40% of doses were self-administered, the odds ratios changed by less than 20%.Table 1 (available at http://www.who.int/bulletin/volumes/85/9/06-038331/en/index.html) shows that non-adherence was associated with poor treatment outcomes (OR: 2.43, 95% CI: 1.05–5.53).
Table 2. Characteristics of DOTS treatment cohort in Tomsk (n = 237).
| Characteristics | Non-adherent n = 38 | Adherent n = 199 | P-value | Defaulter n = 21 | Non-defaulter n = 216 | P-value |
|---|---|---|---|---|---|---|
| Gender | 0.92 | 0.68 | ||||
| male | 24 | 124 | 14 | 134 | ||
| female | 14 | 75 | 7 | 82 | ||
| Age group | 0.36 | 0.21 | ||||
| ≤ 40 | 16 | 100 | 13 | 103 | ||
| > 40 | 22 | 99 | 8 | 113 | ||
| Unemployed | 0.02 | < 0.01 | ||||
| yes | 25 | 91 | 16 | 100 | ||
| no | 13 | 108 | 5 | 116 | ||
| Previously incarcerated | 0.11 | 0.41 | ||||
| yes | 10 | 31 | 5 | 36 | ||
| no | 28 | 168 | 16 | 180 | ||
| Alcoholism noted on treatment initiation | 0.11 | < 0.01 | ||||
| yes | 13 | 44 | 14 | 43 | ||
| no | 25 | 155 | 7 | 173 | ||
| Alcohol abuse noted after treatment initiation | < 0.01 | 0.90 | ||||
| yes | 12 | 24 | 3 | 33 | ||
| no | 26 | 175 | 18 | 183 | ||
| Intravenous drug user at treatment initiation | < 0.01 | 0.90 | ||||
| yes | 6 | 4 | 1 | 9 | ||
| no | 32 | 195 | 20 | 207 | ||
| Any substance abuse | < 0.01 | < 0.01 | ||||
| yes | 30 | 71 | 18 | 33 | ||
| no | 8 | 128 | 3 | 183 | ||
| Impaired mobility | 0.35 | 0.97 | ||||
| yes | 2 | 20 | 2 | 20 | ||
| no | 36 | 179 | 19 | 196 | ||
| Co-morbid conditions associated with side-effects | 0.66 | 0.02 | ||||
| yes | 7 | 31 | 7 | 31 | ||
| no | 31 | 168 | 14 | 185 | ||
| MDR at treatment initiation | 0.65 | 0.88 | ||||
| yes | 4 | 16 | 2 | 18 | ||
| no | 34 | 177 | 19 | 192 | ||
| Sputum smear positivity at treatment initiation | 0.46 | 0.26 | ||||
| yes | 17 | 102 | 13 | 106 | ||
| no | 21 | 97 | 8 | 110 | ||
| Cavitary disease | 0.76 | 0.92 | ||||
| yes | 26 | 141 | 15 | 152 | ||
| no | 12 | 58 | 6 | 64 | ||
| Transport time to clinic | 0.85 | 0.63 | ||||
| < 20 minutes | 11 | 65 | 7 | 69 | ||
| 20–40 minutes | 21 | 100 | 12 | 109 | ||
| > 40 minutes | 6 | 34 | 2 | 38 |
MDR, multidrug resistant (TB).
Table 3. Multivariable analysis of risk factors associated with non-adherence and default in a Tomsk TB treatment cohort.
| Outcome |
||||
|---|---|---|---|---|
| Non-adherence multivariate OR (95% CI) | Default multivariate OR (95% CI) | |||
| Male | 0.66 | 0.28–1.55 | 0.85 | 0.27–2.61 |
| Age > 40 | 0.84 | 0.37–1.90 | 1.98 | 0.65–6.08 |
| Unemployed | 1.15 | 0.49–2.69 | 2.62 | 0.76–9.06 |
| Previously incarcerated | 1.06 | 0.39–2.86 | 0.69 | 0.20–2.41 |
| Baseline alcoholism noted on initiation of treatment | 4.48 | 1.58–12.68 | 15.57 | 3.46–70.02 |
| Alcohol abuse first noted after initiation of treatmenta | 6.35 | 2.27–17.75 | 5.14 | 0.87–30.25 |
| Intravenous drug user at initiation of treatment | 16.64 | 3.24–85.56 | 2.58 | 0.21–30.96 |
| Any substance abuseb | 7.30 | 2.89–18.46 | 11.20 | 2.55–49.17 |
| Co-morbid conditions associated with side-effectsa | NIc | 7.20 | 1.94–26.75 | |
CI, confidence interval; OR, odds ratio. a Included in default model only. Other variables included in both models. b Included in model excluding alcoholism and drug use variables. c Not included.
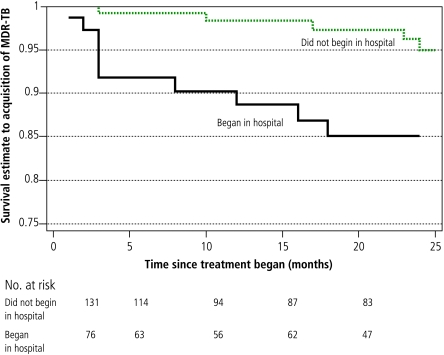
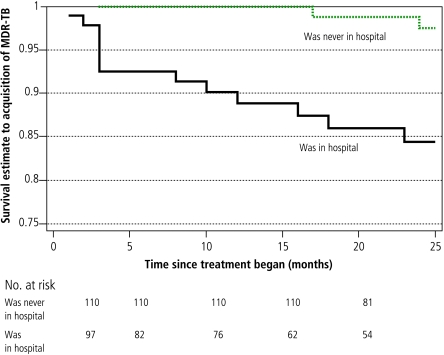
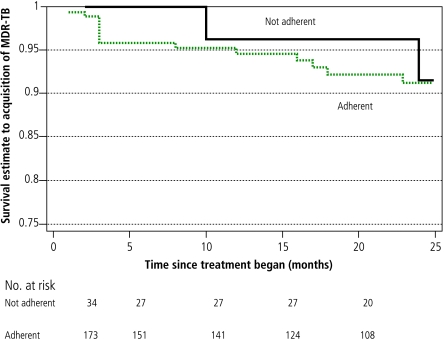
Sputum-smear positivity was the only factor associated significantly with baseline MDR in both a univariate analysis (OR=2.4, 95% CI: 1.04–5.57) and in a multivariate logistic regression model that included age and substance abuse (OR = 3.28, 95% CI: 1.24–8.68). Factors associated significantly with MDR acquisition in a univariate analysis included substance abuse, hospitalization (both at initiation of treatment and later in the course of therapy) and failure to self-administer therapy (Figs. 2, 3, 4; and Table 4, available at http://www.who.int/bulletin/volumes/85/9/06-038331/en/index.html). In the multivariate Cox proportional hazards model, treatment received in the hospital setting (either at initiation of therapy or later) was the only remaining independent risk factor for MDR acquisition. Patients who received treatment in the hospital setting had a substantially higher risk of developing MDR-TB than those whose treatment was confined to the outpatient sector. This was true for those who began DOTS treatment in the hospital setting (adjusted hazard ratio, HR: 6.34; P = 0.02) and those who were hospitalized only later in their treatment course (adjusted HR: 6.26; P = 0.04).
Fig. 2.
Kaplan-Meier survival curves for substance abuse as a factor associated with time to acquisition of multidrug resistance
Fig. 3.
Kaplan-Meier survival curves for hospitalization as a factor associated with time to acquisition of multidrug resistance
Fig. 4.
Kaplan-Meier survival curves for failure to self-administer therapy as a factor associated with time to acquisition of multidrug resistance
Table 4. Factors associated with acquisition of multidrug resistance in univariate and multivariate analyses.
| Cohort characteristics | Number of events | Person time in months | Univariate hazard ratio | P-value | Multivariate hazard ratio | P-value |
|---|---|---|---|---|---|---|
| Age | ||||||
| ≤ 40 | 7 | 2442 | 1.06 | 0.90 | 0.70 | 0.52 |
| > 40 | 8 | 2586 | ||||
| Gender | ||||||
| male | 11 | 2920 | 1.93 | 0.24 | 1.67 | 0.39 |
| female | 4 | 2108 | ||||
| Not-adherent | ||||||
| yes | 2 | 736 | 0.81 | 0.77 | 1.61 | 0.53 |
| no | 13 | 4267 | ||||
| Substance abuse | ||||||
| yes | 10 | 1944 | 2.88 | 0.04 | 1.96 | 0.26 |
| no | 5 | 3084 | ||||
| Side-effects | NI | |||||
| yes | 4 | 855 | 1.69 | 0.39 | ||
| no | 11 | 4173 | ||||
| Baseline cavity present | NI | |||||
| yes | 11 | 3432 | 1.25 | 0.69 | ||
| no | 4 | 1596 | ||||
| Previously incarcerated | NI | |||||
| yes | 3 | 656 | 1.56 | 0.51 | ||
| no | 12 | 4372 | ||||
| Smear ++ or +++ | NI | |||||
| yes | 4 | 1584 | 0.79 | 0.68 | ||
| no | 11 | 3444 | ||||
| Began treatment in hospitala | ||||||
| yes | 10 | 1703 | 3.8 | 0.01 | 6.34 | 0.02 |
| no | 5 | 3325 | ||||
| Hospitalized later during therapy only | ||||||
| yes | 13 | 2195 | 8.18 | < 0.001 | 6.26 | 0.047 |
| no | 2 | 2833 | ||||
| Self-administered treatment | NI | |||||
| yes | 2 | 1847 | 0.25 | 0.03 | ||
| no | 13 | 3181 |
a Individuals who were hospitalized at initiation of therapy as well as later were included only in the hospitalized at initiation category.
Table 5 (available at http://www.who.int/bulletin/volumes/85/9/06-038331/en/index.html) demonstrates the differing risk factors for early and late acquisition of MDR – of the seven patients who developed MDR within four months of initiating treatment, all had cavitary disease at baseline and six began treatment in the hospital. In a multivariate analysis, those who initiated treatment in the hospital were more likely to develop early MDR, but this finding failed to achieve statistical significance (adjusted HR: 7.18, P = 0.07). In contrast, univariate risk factors for MDR after 6 months of treatment included male gender (HR: 5.12, P = 0.06), substance abuse (HR: 11.22, P = 0.004) and absence of smear positivity (HR: 0, P = 0.01). In a multivariate Cox proportional hazards model substance abuse was the only statistically significant factor (adjusted HR: 9.09, P = 0.04), although patients who had been hospitalized at some point during their illness were also more likely to develop late MDR (HR: 4.52, P = 0.07).
Table 5. Factors associated with early and late acquisition of MDR in univariate and multivariate analyses.
| Cohort characteristics | Early MDR (n = 7) |
Late MDR (n = 8) |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Univariate HR | P-value | Multivariate HR | P-value | Univariate HR | P-value | Multivariate HR | P-value | ||
| Age | NI | NI | |||||||
| ≤ 40 | 1.28 | 0.74 | 0.90 | 0.89 | |||||
| > 40 | |||||||||
| Gender | NI | ||||||||
| male | 0.89 | 0.88 | 5.12 | 0.06 | 2.58 | 0.389 | |||
| female | |||||||||
| Not-adherent | NI | ||||||||
| yes | 0 | 0.12 | 0 | NA | 1.86 | 0.47 | |||
| noa | |||||||||
| Substance abuse | NI | ||||||||
| yes | 1.04 | 0.96 | 11.22 | 0.004 | 9.09 | 0.046 | |||
| no | |||||||||
| Side effects | |||||||||
| yes | 3.53 | 0.12 | 2.92 | 0.16 | 0.65 | 0.67 | NI | ||
| no | |||||||||
| Baseline cavity present | NI | NI | |||||||
| yes | Infb | 0.025 | Infb | NA | 0.47 | 0.30 | |||
| no | 0 | ||||||||
| Previously incarcerated | NI | NI | |||||||
| yes | 0.97 | 0.98 | 2.20 | 0.37 | |||||
| no | |||||||||
| Smear ++ or +++ | |||||||||
| yes | 2.86 | 0.17 | 1.16 | 0.84 | 0c | 0.01 | 0c | ||
| no | |||||||||
| Began treatment in hospital | NI | ||||||||
| yes | 10.87 | 0.006 | 7.18 | 0.07 | 1.98 | 0.34 | |||
| no | |||||||||
| Hospitalized later during therapy only | NI | ||||||||
| yes | 1.50 | 0.72 | 3.53 | 0.17 | 4.52 | 0.07 | |||
| no | |||||||||
MDR, multidrug resistant (TB). a No early cases of acquired MDR were non-adherent. b All early cases of acquired MDR had cavitary disease. c No late cases were smear-positive.
Notably, non-adherence was not a risk factor for either early or late acquisition of MDR. This finding remained true when we conducted a sensitivity analysis in which patients were classified non-adherent if they missed 40% or more of their prescribed doses.
Discussion
In this study of non-adherence, default and acquisition of MDR among TB patients in Tomsk, substance abuse and in-hospital care were identified as two potential obstacles to effective treatment. These results suggest that DOTS programmes might be more likely to achieve TB control targets if they include interventions aimed at improving adherence by diagnosing and treating substance abuse concurrently with standard TB therapy. They also raise the possibility that some patients with apparent drug-sensitive disease also may be infected with drug-resistant strains that are unmasked upon initiation of therapy. Some patients also might be reinfected with drug-resistant strains in the hospital setting, a possibility which emphasizes the need for effective infection-control measures within facilities that care for patients with active disease.
Despite the implementation of a DOTS programme and the provision of extensive social services to patients undergoing TB therapy, non-adherence and default continued in a substantial proportion of those who initiated treatment in Tomsk. Like TB patients throughout the world, these patients were burdened with a wide array of social and medical problems: many were unemployed, had been in prison or had significant co-morbid conditions. Despite this, alcohol and injection drug use were the only independent risk factors for non-adherence and default that we identified. These findings echo those of numerous previous studies that found substance abuse to be the single major factor associated most strongly with non-compliance with TB regimens.7–15 Our results also agree with these previous studies’ findings that non-adherence has important adverse effects on the outcomes of TB treatment16,17 – 66% of all poor outcomes experienced in our cohort occurred among the 16% of patients who did not adhere to therapy.
Despite the clear need for new approaches to this problem, to date there has been relatively little research on treatment options for patients with chronic infectious diseases and concomitant substance misuse or psychiatric problems. The few TB programmes that have explicitly offered patients treatment for substance abuse generally have demonstrated better outcomes than “unexpanded” DOTS programmes.18 Some even achieve very high cure rates among patient populations in which alcoholism or injection drug use are common.19 Disappointingly, these successes have not yet led to widespread integration of substance-abuse care for these patients.
This failure has at least three possible explanations. The first is the general reluctance to tinker with the specialized “vertical” DOTS approach, given its success in improving case completion and cure rates in developing and less-developed countries over the past two decades.20 Closely related to this are the numerous obstacles faced by multidisciplinary approaches to research and patient care, including the lack of a shared language and space among care providers from different specialties and mutual lack of knowledge of other treatment approaches.21 Often the care of TB patients and those with substance-use disorders is relegated to highly specialized practitioners; this offers little opportunity for meaningful interaction or exchange between disciplines. Finally, until recently many physicians without specific expertise in managing alcohol disorders and injection drug misuse have assumed that these conditions’ treatments are too complex and intensive to be carried out simultaneously with the treatment of another complex disease. However, recent evidence suggests that brief interventions, social skills training, behaviour contracting and pharmacotherapy are among the most effective approaches for treatment of substance-use disorders.22–24 These data raise the possibility that integrated management of these most vulnerable TB patients may be within the reach of a unified TB care facility.
Our study also suggests that non-adherence did not contribute to either the early or late occurrence of MDR among patients receiving DOTS in this setting. We considered several other possible explanations for the observation that a group of adherent patients developed MDR-TB within 24 months of initiating therapy. First, we speculated that MDR acquisition might be associated with disease severity, which might in turn be linked to hospitalization. Since the number of new mutations that code for drug resistance will be a function of the bacterial load, it follows that those with a greater disease burden would be at higher risk of developing these mutations.25 Having adjusted for disease severity by controlling for the presence or absence of cavitary disease and sputum-smear status, we found that these markers of disease severity were strongly correlated with early acquisition of MDR but not associated with late acquisition. These data suggest that these patients may harbour multiple different strains of Mycobacterium TB, some of which may be drug-resistant. In these mixed infections, standard short-course therapy may have unmasked the drug-resistant strain population by suppressing the previously dominant drug-sensitive strain. Indeed, van Rie et al. have described this mechanism in a high-burden population in South Africa.26 In that study, adherence to a first-line drug therapy was shown to select for a resistant population, while non-adherence led to re-emergence of the drug-susceptible strains.
We also assessed the possibility that patients who developed MDR did so through “amplification” of existing drug resistance. While this mechanism may have accounted for MDR acquisition in some cases, eight of the thirteen hospitalized patients with this outcome had fully susceptible disease on initiation of therapy.
Finally, we considered the possibility that some of these patients developed MDR-TB as a result of reinfection with a drug-resistant strain of TB. Reinfection of patients on therapy for drug-sensitive disease has been described in several different high-incidence settings and has been associated with nosocomial transmission.27–31 Usually, MDR-TB patients in the Russian Federation are not placed on respiratory precautions in the hospitals or clinics where they receive care, so there is opportunity for further spread of drug-resistant strains among patients receiving therapy for drug-sensitive disease. The finding that substance abuse was a risk factor for late occurrence of MDR also raises the possibility that these patients are at higher risk of exposure to drug-resistant disease or are more susceptible to reinfection than other patients. Future studies on the association between adherence and development of MDR would benefit from molecular typing of sequential isolates in patients undergoing therapy.
This study was limited by its retrospective study design, as sociodemographic and behavioural variables were abstracted from routine medical assessments conducted upon initiation of therapy. In particular, the diagnoses of alcohol and drug disorders were based on clinicians’ reports and were not made using a standardized instrument. Hence, it is likely that alcohol disorders were underreported and that only more severe cases came to clinical attention. This could have resulted in an underestimation of the effect of alcoholism if less severe cases were also associated with non-adherence. Systematic studies using standardized and validated alcohol assessment instruments will be needed to ascertain the full impact of alcohol disorders on patients’ ability to comply with TB treatment. ■
Acknowledgements
The authors wish to thank Natasha Arlyapova, Donna Barry, Doreen Balbuena, Lauren Doctoroff, Paul Farmer, Jennifer Furin, Timothy Holtz, Gwyneth Jones, Jim Yong Kim, Tatyana Lyagoshina, Sergey Pavlovich Mishustin, Joia Mukherjee, Ed Nardell, Michael Nikiforov, Alexander Pasechnikov, Genady Giorgevich Peremitin, Oksana Ponomarenko, Michael Rich, Sonya Shin, Olga Sirotkina, Tamara Tonkel and Askar Yedilbayev for their contributions to this study.
Footnotes
Funding and competing interests: Two authors (Keshavjee and Gelmanova) received partial salary support and/or travel support from the Bill & Melinda Gates Foundation and from the Eli Lilly International Foundation. Keshavjee received salary support from the Frank Hatch Scholars Program at Brigham & Women’s Hospital. No funder played any role in study design, data collection, analysis or interpretation; or in preparing, reviewing or approving the manuscript.
References
- 1.Toungoussova OS, Bjune G, Caugant DA. Epidemic of tuberculosis in the former Soviet Union: social and biological reasons. Tuberculosis (Edinb) 2006;86:1–10. doi: 10.1016/j.tube.2005.04.001. [Edinb] [DOI] [PubMed] [Google Scholar]
- 2.Shilova MV, Dye C. The resurgence of tuberculosis in Russia. Philos Trans R Soc Lond B Biol Sci. 2001;356:1069–75. doi: 10.1098/rstb.2001.0895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Global tuberculosis control – surveillance, planning and financing: Russian Federation. Geneva: WHO; 2005. Available at: http://www.who.int/tb/publications/global_report/2005/annex1/en/index15.html
- 4.Mathew TA, Ovsyanikova TN, Shin SS, Gelmanova I, Balbuena DA, Atwood S, et al. Causes of death during tuberculosis treatment in Tomsk Oblast, Russia. Int J Tuberc Lung Dis. 2006;10:857–63. [PubMed] [Google Scholar]
- 5.Treatment of tuberculosis: guidelines for national programmes. 3rd ed. Geneva: WHO; 2003. [Google Scholar]
- 6.Veen J, Raviglione MC, Rieder HL, Migliori GB, Graf P, Grzemska M, et al. Standardised outcome monitoring in Europe. Recommendations of a working group of WHO and IUATLD (Europe Region). Eur Respir J. 1998;12:505–10. doi: 10.1183/09031936.98.12020505. [DOI] [PubMed] [Google Scholar]
- 7.Kok-Jensen A. The prognosis of pulmonary tuberculosis in patients with abuse of alcohol. Scand J Respir Dis. 1970;51:42–8. [PubMed] [Google Scholar]
- 8.Corcoran R. Compliance with chemotherapy for tuberculosis. Ir Med J. 1986;79:87–90. [PubMed] [Google Scholar]
- 9.Cayla JA, Caminero JA, Rey R, Lara N, Valles X, Galdos-Tanguis H. Working group on completion of tuberculosis treatment in Spain. Current status of treatment completion and fatality among tuberculosis patients in Spain. Int J Tuberc Lung Dis. 2004;8:458–64. [PubMed] [Google Scholar]
- 10.Diel R, Niemann S. Outcome of tuberculosis treatment in Hamburg: a survey, 1997-2001. Int J Tuberc Lung Dis. 2003;7:124–31. [PubMed] [Google Scholar]
- 11.Ferrer X, Kirschbaum A, Toro J, Jadue J, Munoz M, Espinoza A. Compliance with tuberculosis treatment in adults in Santiago, Chile. Bol Oficina Sanit Panam. 1991;111:423–31. [PubMed] [Google Scholar]
- 12.Salami AK, Oluboyo PO. Management outcome of pulmonary tuberculosis: a nine-year review in Ilorin. West Afr J Med. 2003;22:114–9. doi: 10.4314/wajm.v22i2.27928. [DOI] [PubMed] [Google Scholar]
- 13.Santha T, Garg R, Frieden TR, Chandrasekaran V, Subramani R, Gopi PG, et al. Risk factors associated with default, failure and death among tuberculosis patients treated in a DOTS programme in Tiruvallur District, South India, 2000. Int J Tuberc Lung Dis. 2002;6:780–8. [PubMed] [Google Scholar]
- 14.Krupitsky EM, Zvartau EE, Lioznov DA, Tsoy MV, Egorova VY, Belyaeva TV, et al. Co-morbidity of infectious and addictive diseases in St. Petersburg and the Leningrad region, Russia. Eur Addict Res. 2006;12:12–9. doi: 10.1159/000088578. [DOI] [PubMed] [Google Scholar]
- 15.Fry RS, Khoshnood K, Vdovichenko E, Granskaya J, Sazhin V, Shpakovskaya L, et al. Barriers to completion of tuberculosis treatment among prisoners and former prisoners in St. Petersburg, Russia. Int J Tuberc Lung Dis. 2005;9:1027–33. [PubMed] [Google Scholar]
- 16.Burman WJ, Cohn DL, Rietmeijer CA, Judson FN, Sbarbaro JA, Reves RR. Noncompliance with directly observed therapy for tuberculosis. Epidemiology and effect on the outcome of treatment. Chest. 1997;111:1168–73. doi: 10.1378/chest.111.5.1168. [DOI] [PubMed] [Google Scholar]
- 17.Pablos-Méndez A, Knirsch CA, Barr RG, Lerner BH, Frieden TR. Nonadherence in tuberculosis treatment: predictors and consequences in New York City. Am J Med. 1997;102:164–70. doi: 10.1016/S0002-9343(96)00402-0. [DOI] [PubMed] [Google Scholar]
- 18.Chaulk CP, Kazandjian VA. Directly observed therapy for treatment completion of pulmonary tuberculosis: consensus statement of the public health tuberculosis guidelines panel. JAMA. 1998;279:943–8. doi: 10.1001/jama.279.12.943. [DOI] [PubMed] [Google Scholar]
- 19.Schluger N, Ciotoli C, Cohen D, Johnson H, Rom WN. Comprehensive tuberculosis control for patients at high risk for noncompliance. Am J Respir Crit Care Med. 1995;151:1486–90. doi: 10.1164/ajrccm.151.5.7735604. [DOI] [PubMed] [Google Scholar]
- 20.Raviglione MC, Pio A. Evolution of WHO policies for tuberculosis control, 1948-2001. Lancet. 2002;359:775–80. doi: 10.1016/S0140-6736(02)07880-7. [DOI] [PubMed] [Google Scholar]
- 21.Willenbring ML. Integrating care for patients with infectious, psychiatric, and substance use disorders: concepts and approaches. AIDS. 2005;19:S227–37. doi: 10.1097/01.aids.0000192094.84624.c2. [DOI] [PubMed] [Google Scholar]
- 22.Bertholet N, Daeppen JB, Wietlisbach V, Fleming M, Burnand B. Reduction of alcohol consumption by brief alcohol intervention in primary care: systematic review and meta-analysis. Arch Intern Med. 2005;165:986–95. doi: 10.1001/archinte.165.9.986. [DOI] [PubMed] [Google Scholar]
- 23.Goldstein MG, Whitlock EP, DePue J. Planning committee of the addressing multiple behavioral risk factors in primary care project. Multiple behavioral risk factor interventions in primary care. Am J Prev Med. 2004;27:61–79. doi: 10.1016/j.amepre.2004.04.023. [DOI] [PubMed] [Google Scholar]
- 24.Whitlock EP, Green CA, Polen MR. Behavioral counseling interventions in primary care to reduce risky/harmful alcohol use. Systematic evidence review. Rockville: Agency for Healthcare Research and Quality; 2004. Available at: www.ahrq.gov/clinic/serfiles.htm [PubMed]
- 25.Seung KJ, Gelmanova IE, Peremitin GG, Golubchikova VT, Pavlova VE, Sirotkina OB, et al. The effect of initial drug resistance on treatment response and acquired drug resistance during standardized short-course chemotherapy for tuberculosis. Clin Infect Dis. 2004;39:1321–8. doi: 10.1086/425005. [DOI] [PubMed] [Google Scholar]
- 26.van Rie A, Victor TC, Richardson M, Johnson R, van der Spuy GD, Murray EJ, et al. Reinfection and mixed infection cause changing Mycobacterium tuberculosis drug-resistance patterns. Am J Respir Crit Care Med. 2005;172:636–42. doi: 10.1164/rccm.200503-449OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Narvskaya O, Otten T, Limeschenko E, Sapozhnikova N, Graschenkova O, Steklova L, et al. Nosocomial outbreak of multidrug-resistant tuberculosis caused by a strain of Mycobacterium tuberculosis W-Beijing family in St. Petersburg, Russia. Eur J Clin Microbiol Infect Dis. 2002;21:596–602. doi: 10.1007/s10096-002-0775-4. [DOI] [PubMed] [Google Scholar]
- 28.Rivero A, Marquez M, Santos J, Pinedo A, Sanchez MA, Esteve A, et al. High rate of tuberculosis reinfection during a nosocomial outbreak of multidrug-resistant tuberculosis caused by Mycobacterium bovis strain B. Clin Infect Dis. 2001;32:159–61. doi: 10.1086/317547. [DOI] [PubMed] [Google Scholar]
- 29.Kruuner A, Hoffner SE, Sillastu H, Danilovits M, Levina K, Svenson SB, et al. Spread of drug-resistant pulmonary tuberculosis in Estonia. J Clin Microbiol. 2001;39:3339–45. doi: 10.1128/JCM.39.9.3339-3345.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Verver S, Warren RM, Beyers N, Richardson M, van der Spuy GD, Borgdorff MW, et al. Rate of reinfection tuberculosis after successful treatment is higher than rate of new tuberculosis. Am J Respir Crit Care Med. 2005;171:1430–5. doi: 10.1164/rccm.200409-1200OC. [DOI] [PubMed] [Google Scholar]
- 31.van Rie A, Warren R, Richardson M, Victor TC, Gie RP, Enarson DA, et al. Exogenous reinfection as a cause of recurrent tuberculosis after curative treatment. N Engl J Med. 1999;341:1174–9. doi: 10.1056/NEJM199910143411602. [DOI] [PubMed] [Google Scholar]