Abstract
Introduction
While immunoassays have been used extensively for measurement of serum testosterone (T) and estradiol (E2) levels, there is concern about their specificity, particularly at low E2 levels as present in men.
Methods
We compared T and E2 measured by mass spectroscopy to levels measured by immunoassay in men (n = 313, age 22 to 91 years) and related these to volumetric bone mineral density (vBMD) at various skeletal sites.
Results
Serum T and non-SHBG bound (or bioavailable) T levels by immunoassay correlated well with the corresponding mass spectroscopy measurements (R = 0.90 and 0.95, respectively, P < 0.001); the correlations for serum E2 measured using the two techniques were less robust (R = 0.63 for total E2 and 0.84 for bioavailable E2, P < 0.001). Overall relationships between serum bioavailable T and E2 levels with vBMD at various skeletal sites were similar for the immunoassay and mass spectroscopic measures.
Conclusions
Although E2 levels with immunoassay correlate less well with the mass spectroscopic measurements than do the T measurements in men, our findings indicate that the fundamental relationships observed previously between vBMD and the sex steroids by immunoassay are also present with the mass spectroscopic measurements.
Key terms: Assays, Osteoporosis, Bone
Introduction
There is increasing evidence that sex steroids regulate bone mass and bone turnover in men and that declining sex steroids with age, particular declining non-sex hormone binding globulin (SHBG) bound (or bioavailable) sex steroids, may contribute to bone loss in aging men (1, 2). In previous studies, we (3-6) and others (7-12) have used immunoassays to measure testosterone (T) and estradiol (E2) levels in men. Estimation of the bioavailable fractions of T or E2 has been done by various methods, including ammonium sulfate precipitation (13, 14) or the use of mass action equations (15, 16). In general, these studies have found that serum bioavailable E2 (with E2 levels measured by immunoassay and the bioavailable fraction estimated using either method) correlated better with bone mineral density (BMD) and with rates of bone loss than did total E2 or total or bioavailable T levels (3-8).
Despite these extensive data using immunoassays, there has recently been concern about the use of immunoassays to measure sex steroids, particularly E2, in men due to the relatively low E2 levels present in men as compared to premenopausal women (17). This concern has been generated principally from studies using mass spectroscopy to measure the low E2 levels present in postmenopausal women (17), although it is clear that the mean E2 level in men is 2-3 times higher than the mean E2 level in postmenopausal women (3). To address this concern and to reexamine our previous work and conclusions regarding the relationships between sex steroids and bone mass in men, we remeasured T and E2 levels in our cohort of men in whom we had previously made these measurements using immunoassays (5, 6), examined the correlations between the mass spectroscopic and immunoassay derived values, and compared the relationship of each of these to volumetric BMD (vBMD) at various skeletal sites.
Methods
Subjects
We recruited subjects from an age-stratified, random sample of Rochester, MN residents who were selected using the medical records linkage system of the Rochester Epidemiology Project (18). This population is highly characteristic of the United States White population, but Blacks and Asians are underrepresented. The sample spanned ages from 22 to 93 yrs and included 325 men, with approximately 50 men per decade. Reflecting the ethnic composition of the community, 96% of the men were White. Twelve subjects were excluded from this analysis (6 subjects because of concomitant therapy with bisphosphonates [2], testosterone [2], raloxifene [1], or estrogen [1]; 4 subjects because of significant renal insufficiency [serum creatinine > 2 mg/dl]; 1 subject because of an inexplicably high bioavailable E2 level [> 60 pg/mL], and 1 subject who died soon after his scan and had multiple laboratory abnormalities prior to death). Thus, data were analyzed on a total of 313 men.
Sex steroid measurements
Fasting serum samples were obtained on all subjects. Total T was measured by a competitive immunoassay using direct, chemiluminescent technology (ACS 180, Bayer, Tarrytown, NY; interassay coefficients of variation [CV] < 11%, lower limit of detection 5 ng/dL). Cross-reactivity of this assay was 5% with dihydrotestosterone and < 1% for all other metabolites. Total E2 was measured using a double antibody radioimmunoassay (Diagnostic Products Corporation, Los Angeles, CA; interassay CV < 8%, lower limit of detection 5 pg/mL). Cross-reactivity of this assay was 12% with estrone and 6% or less for other estrogen metabolites. For the mass spectroscopy measurements, we used liquid chromatography-tandem mass spectrometry (LC-MS/MS, API 5000, Applied Biosystems-MDS Sciex, Foster City, CA). Testosterone was prepared by acetonitrile precipitation and high-throughput liquid chromatography (HTLC) extraction. This was followed by analysis on the LC-MS/MS equipped with a heated nebulizer ion source. For the 17β-E2 measurements, an organic extraction of serum was performed using methylene chloride to remove water-soluble conjugates and concentrate the specimen. After derivatization with dansyl chloride, the specimen was processed with high pressure liquid chromatography (HPLC) and then passed into the LC-MS/MS. Deuterated d5-17β-E2 and d3-testosterone served as internal standards. Values as low as 1 ng/dL for T and 1.25 pg/mL for E2 were detectable by this method. For T values of 8, 4, 2, and 1 ng/dL, the respective CVs were 7.5, 2.2, 6.3, and 28.8%. For E2 values of 10, 5, 2.5, and 1.25 pg/mL, the respective CVs were 2.3, 6.1, 14.5, and 13.4%.
SHBG was measured using a chemiluminescent immunoassay (Diagnostic Products Corporation, Los Angeles, CA; interassay CV < 8%). In addition, the non-SHBG bound (bioavailable) fraction of total T and E2 was measured using a modification of the technique of O'Connor et al. (13) and Tremblay et al. (14), as previously described (3). The percent bioavailable fraction of T or E2 was then multiplied by the total T or E2 from either the immunoassay or mass spectroscopy methods to obtain the respective bioavailable T or E2 values based on the two methods.
Central QCT
As previously described (19, 20), single energy CT scans were made at the lumbar spine and proximal femur with a multi-detector Light Speed QX-I scanner (GE Medical Systems, Wakesha, WI), using a tube potential of 120 kVp, tube current of 80 mA, rotation time of 0.8 sec, table speed of 7.5 mm/rotation, detector collimation of 4 × 2.5 mm, and pitch of 0.75. Data sets were obtained in 20 sec for the spine and in 20 sec for the hip, eliminating significant motion artifacts. For the lumbar vertebrae, we analyzed the average of 5 slices obtained at the midportion of the L1, L2 and L3 vertebrae and a mean of these values was used. For the femoral neck, we assessed 5 reformatted oblique sections at the mid-portion of the femoral neck, between the superior aspect of the head of the femur and the inferior aspect of the inferior trochanter. For all scanning sites, slice width was 2.5 mm and the in-plane voxel size was 0.74 mm. Calibration standards scanned with the patient were used to convert CT numbers directly to equivalent vBMD in mg/cm3 (21). Image analysis and processing of the CT scans was done as previously described (19, 20).
Peripheral QCT
Single energy CT scans were made at two scanning sites in the distal radius and at two scanning sites in the distal tibia using the Densiscan 1000 (Scanco Medical AG, Bassersdorf, Switzerland), as previously described (19). All scans had a slice thickness of 1.5 mm and an in-plane voxel size of 0.35 mm for the radius and 0.45 mm for the tibia. From a digital image (scout view) of the lower forearm and lower leg, the joint space is visualized and a reference point is set electronically at the intersection of the joint space with radioulnar junction for the forearm and the tibiofibular junction for the distal leg. From this line, an automated program then selects a distal and a more proximal scanning site at both the distal radius and distal tibia. For the radius, the more distal of the two scanning sites (termed Rad-D) was located 7 to 20 mm and the more proximal scanning site (termed Rad-P) was located 48 to 55.5 mm from the reference line. For the tibia, the more distal scanning site (termed Tib-D) was located 20 to 33.5 mm and the more proximal scanning site (termed Tib-P) was located 63 to 70.5 mm from the reference line. Ten consecutive slices were made at the Rad-D and Tib-D sites, and six consecutive slices at the Rad-P and Tib-P sites. A surface detection algorithm delineates the bone that then is peeled pixel-by-pixel until core areas of 90%, 70%, and 50% remain. The outer 10% of bone is excluded to avoid partial volume effects from the bone edge. The 70% to 90% cross-sectional area contains cortical bone, and the inner 50% contains only trabecular bone. For the analyses presented here, the inner 50% at the Rad-D and Tib-D sites was used for trabecular vBMD, and the 70-90% value at the Rad-P and Tib-P sites was used for cortical vBMD. CVs were all < 0.5% using our own data from 2 repeat scans of 20 healthy young adults (19, 20).
Statistical analyses
Sex hormone values were summarized using means and standard deviations (SDs). Correlations between the immunoassay and mass spectroscopic values were made using Pearson correlations. Correlations of sex hormones with the bone structural and vBMD parameters were evaluated using Pearson's simple and partial correlation coefficients.
Results
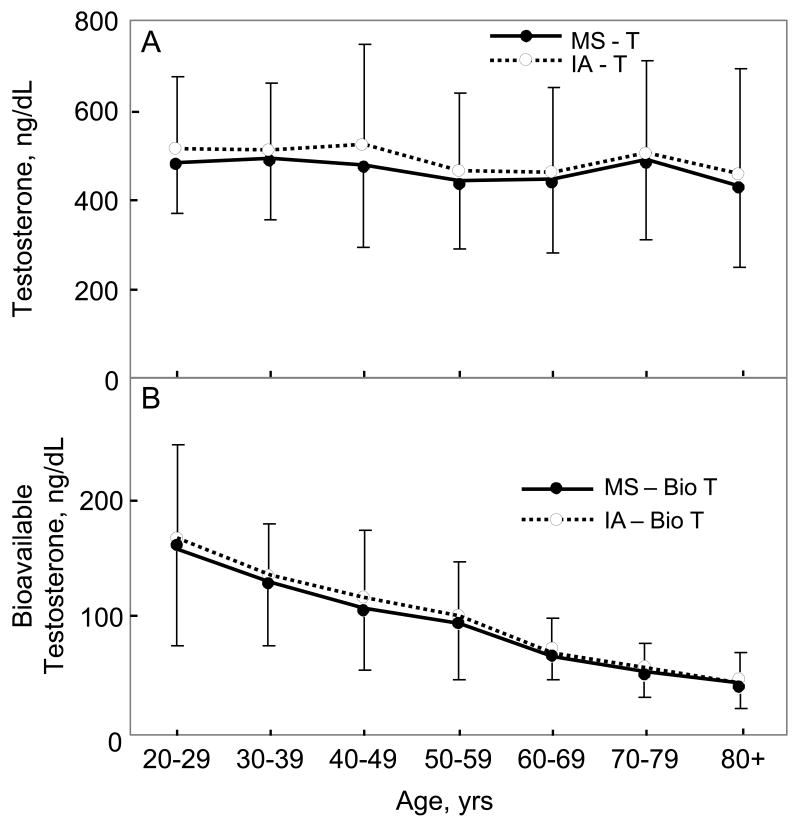
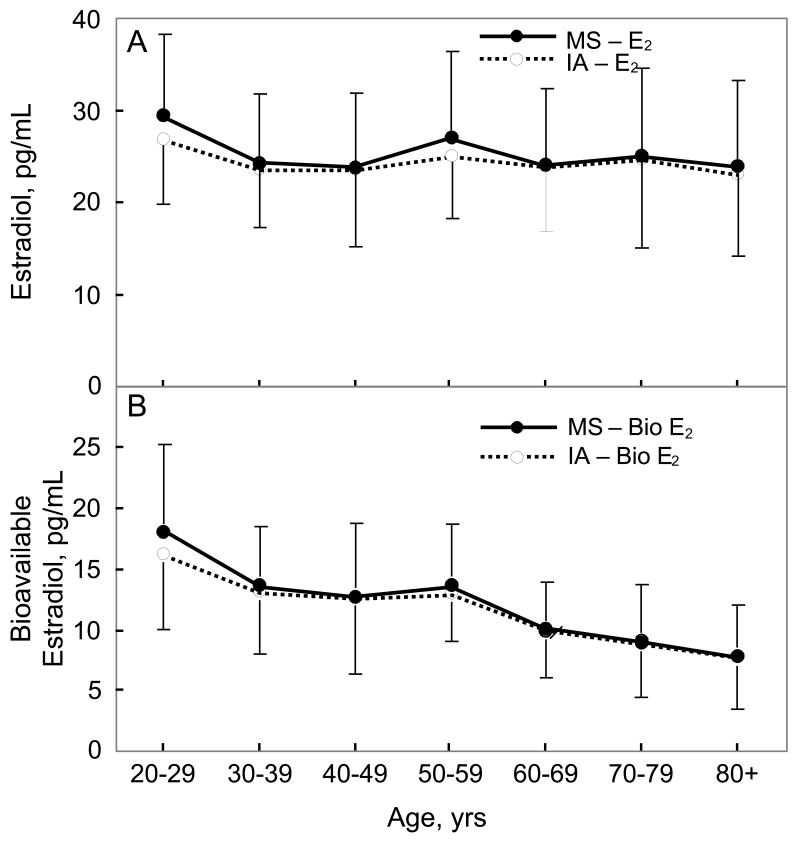
Table 1 provides a summary of the values for the sex steroid and BMD variables in the study group. Figure 1A shows the changes in serum T levels measured either by immunoassay or by mass spectroscopy as a function of age, and Figure 1B shows the corresponding plots for serum bioavailable T levels. As is evident, the pattern of changes with age were virtually identical with both assays. The correlation with age for total T levels was -0.09 (P = 0.122) and -0.09 (P = 0.117) using the immunoassay and mass spectroscopy measurements, respectively, and -0.65 (P < 0.0001) and -0.70 (P < 0.0001) for bioavailable T levels using the immunoassay and mass spectroscopy measurements, respectively. Figures 2A and 2B show the corresponding data for serum E2 and bioavailable E2 levels with the two assays, again demonstrating very similar age-related changes in these variables. The correlation with age for total E2 levels was -0.05 (P = 0.433) and -0.07 (P = 0.214) using the immunoassay and mass spectroscopy measurements, respectively, and -0.45 (P < 0.001) and -0.50 (P < 0.0001) for bioavailable E2 levels using the immunoassay and mass spectroscopy measurements, respectively.
Table 1.
Summary of sex steroid and volumetric bone mineral density (vBMD) variables in the study group. Data are mean ± SD.
| N | 313 |
| Age, years | 56.4 ± 18.4 |
| Sex steroids | |
| Immunoassay Testosterone, ng/dl | 492.9 ± 196.5 |
| Mass spectroscopy Testosterone, ng/dL | 467.8 ± 173.4 |
| Immunoassay bioavailable Testosterone, ng/dl | 95.5 ± 58.3 |
| Mass spectroscopy bioavailabe Testosterone, ng/dL | 89.7 ± 50.5 |
| Immunoassay Estradiol, pg/mL | 24.0 ± 8.0 |
| Mass spectroscopy Estradiol, pg/mL | 24.9 ± 8.6 |
| Immunoassay bioavailable Estradiol, pg/mL | 11.3 ± 5.4 |
| Mass spectroscopy bioavailable Estradiol, pg/mL | 11.6 ± 5.5 |
| Trabecular vBMD | |
| Vertebrae, mg/cm3 | 155.8 ± 38.6 |
| Femur neck, mg/cm3 | 204.8 ± 50.6 |
| Distal radius, mg/cm3 | 307.3 ± 69.7 |
| Distal tibia, mg/cm3 | 323.8 ± 67.7 |
| Cortical vBMD | |
| Femur neck, mg/cm3 | 553.1 ± 71.3 |
| Distal radius, mg/cm3 | 1639.9 ± 69.5 |
| Distal tibia, mg/cm3 | 1543.7 ± 64.0 |
Figure 1.
(A) Total and (B) bioavailable testosterone (T) levels as a function of age using either the mass spectroscopic (MS) or immunoassay (IA) measurement methods.
Figure 2.
(A) Total and (B) bioavailable estradiol (E2) levels as a function of age using either the mass spectroscopic (MS) or immunoassay (IA) measurement methods.
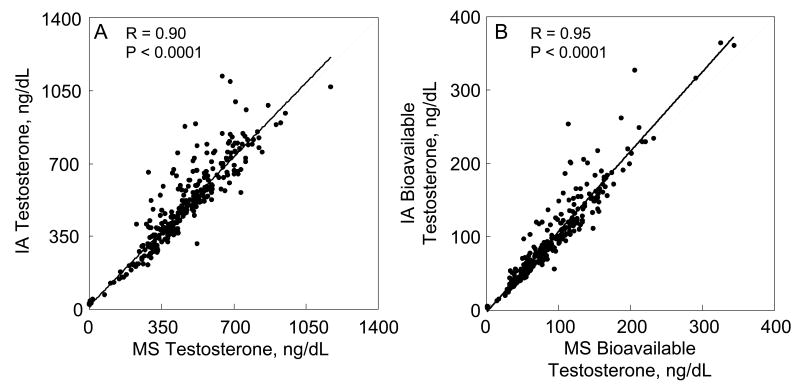
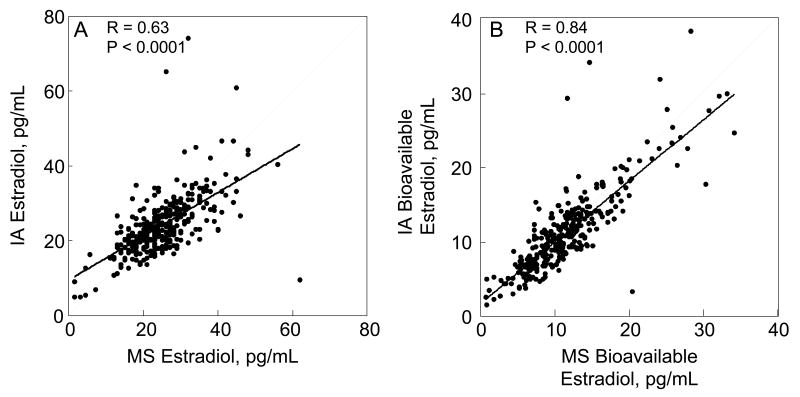
As shown in Figure 3A, serum T measured by immunoassay was highly correlated with that measured by mass spectroscopy. Similarly, when either the immunoassay or mass spectroscopic T measurement was used to calculate bioavailable T levels, the two measures correlated well (Figure 3B). By contrast, serum E2 measurements by immunoassay were only moderately correlated with the corresponding measurements by mass spectroscopy, with the immunoassay value being lower than the mass spectroscopic value at high E2 levels and higher than the mass spectroscopic value at low E2 levels (Figure 4A). However, when each of these measures were used to calculate bioavailable E2 levels, the correlations between bioavailable E2 levels measured by the two assays was considerably better than the correlation between the assays for total E2 levels (Figure 4B). Since our estimation of bioavailable E2 levels was based on ammonium sulfate precipitation of SHBG, this finding suggested that mass spectroscopically determined total E2 might correlate more strongly with SHBG levels. This was indeed the case, with a correlation between SHBG levels and the mass spectroscopic E2 of 0.16 (P = 0.006) and between SHBG levels and the immunoassay E2 of 0.004 (P = 0.940). The net effect of this was to improve the overall relationship between bioavailable E2 derived using the two assays as compared to the relationship for total E2: At high E2 levels by mass spectroscopy, the correspondingly higher SHBG level tended to lower the bioavailable E2 levels derived from mass spectroscopy in this estimation, and vice versa at low E2 levels, bringing the slope of the correlation in Figure 4B closer to unity as compared to the slope in Figure 4A.
Figure 3.
Correlation between (A) total and (B) bioavailable testosterone levels using either the mass spectroscopic (MS) or immunoassay (IA) measurement methods. Solid line indicates the regression relationship and the dashed line represents a slope of unity.
Figure 4.
Correlation between (A) total and (B) bioavailable estradiol levels using either the mass spectroscopic (MS) or immunoassay (IA) measurement methods. Solid line indicates the regression relationship and the dashed line represents a slope of unity.
Finally, we compared correlations of total and bioavailable T as well as total and bioavailable E2 with trabecular and cortical vBMD at various skeletal sites (Table 2). As is evident, unadjusted, age adjusted, and age and body mass index (BMI)-adjusted correlations between the sex steroid measures and vBMD were virtually identical, regardless of whether immunoassays or mass spectroscopy was used to measure the sex steroid levels. Specifically, total T, whether measured by immunoassay or mass spectroscopy, did not correlate with trabecular or cortical vBMD, wheras total E2 correlated with trabecular vBMD, but not cortical vBMD (except for a correlation noted between cortical vBMD at the tibia and the immunoassay total E2). Both bioavailable T and E2 assessed using either the immunoassay or mass spectroscopy, showed significant (and similar) correlations with trabecular and vBMD at the various sites. In the overall group of subjects, age or age and BMI adjustment eliminated (or markedly reduced) the significance of the correlations between bioavailable T and vBMD at cortical and trabecular sites. By contrast, while age or age and BMI adjustment eliminated the significance of the correlations between bioavailable E2 and vBMD at cortical sites, most of these correlations remained significant at trabecular sites even adjusting for these covariates, and these findings were similar whether the immunoassay or mass spectroscopy was used for the E2 measurements.
Table 2.
Unadjusted/age adjusted/age and body mass index adjusted Pearson correlation coefficients between sex steroids measured either by immunoassay or mass spectroscopy and volumetric bone mineral density (vBMD) at various skeletal sites in men.
| T | Bioavailable T | E2 | Bioavailable E2 | |||||
|---|---|---|---|---|---|---|---|---|
| Immunoassay | Mass spec | Immunoassay | Mass spec | Immunoassay | Mass spec | Immunoassay | Mass spec | |
| Trabecular vBMD | ||||||||
| Vertebrae | 0.09/0.05/0.05 | 0.04/-0.01/-0.01 | 0.49***/0.14*/0.14* | 0.50***/0.11/0.11 | 0.18**/0.19***/0.19*** | 0.18**/0.16**/0.17** | 0.41***/0.19***/0.19*** | 0.44***/0.19***/0.19*** |
| Femur neck | -0.04/-0.11/-0.06 | -0.05/-0.12*/-0.08 | 0.40***/0.02/0.06 | 0.42***/0.01/0.05 | 0.15*/0.16**/0.17** | 0.14*/0.13*/0.12* | 0.39***/0.17**/0.17** | 0.42***/0.19**/0.17** |
| Distal radius | 0.08/0.06/0.06 | 0.05/0.02/0.03 | 0.31***/0.12*/0.13* | 0.31***/0.11*/0.12* | 0.15**/0.15**/0.15** | 0.19***/0.17**/0.17** | 0.27***/0.14*/0.14* | 0.31***/0.18**/0.18** |
| Distal tibia | -0.01/-0.04/-0.03 | -0.01/-0.04/-0.04 | 0.21***/0.01/0.02 | 0.23***/0.02/0.03 | 0.08/0.07/0.07 | 0.08/0.05/0.05 | 0.20***/0.07/0.06 | 0.22***/0.07/0.07 |
| Cortical vBMD | ||||||||
| Femur neck | 0.10/0.09/0.06 | 0.10/0.08/0.05 | 0.19**/0.04/0.01 | 0.20***/0.04/0.02 | 0.09/0.09/0.09 | 0.10/0.08/0.10 | 0.16**/0.06/0.06 | 0.18**/0.07/0.09 |
| Distal radius | 0.10/0.06/0.06 | 0.06/0.02/0.01 | 0.39***/0.05/0.05 | 0.40***/0.03/0.03 | 0.10/0.09/0.09 | 0.08/0.05/0.05 | 0.29***/0.07/0.07 | 0.29***/0.04/0.04 |
| Distal tibia | 0.08/0.05/0.01 | 0.06/0.02/-0.03 | 0.34***/0.08/0.06 | 0.35***/0.06/0.04 | 0.16**/0.16**/0.16** | 0.08/0.05/0.06 | 0.30***/0.13*/0.14* | 0.27***/0.07/0.08 |
P < 0.05
P < 0.01
P < 0.001
Discussion
We demonstrate in the present study that, in men, serum T levels measured by immunoassay were highly correlated with T measurements using mass spectroscopy. Further, since the non-SHBG (or bioavailable) T level was estimated by multiplying the total T value with the fraction that was not precipitated by ammonium sulfate, bioavailable T levels based on either method for measuring total T levels were also highly correlated. By contrast, total E2 levels measured by immunoassay correlated less well with the corresponding E2 levels measured by mass spectroscopy. Interestingly, correlations between bioavailable E2 levels based on the two assay methods were better than for total E2 levels, since total E2 levels measured by mass spectroscopy correlated better with SHBG levels than did E2 levels by immunoassay. This had the net effect of bringing the slope of the relationship between bioavailable E2 levels estimated using the two methods closer to unity. Since E is known to stimulate hepatic SHBG production (22), the observed better correlation between E2 levels and serum SHBG levels with the mass spectroscopy assay, as compared to the immunoassay, would suggest that the mass spectroscopy measurement is providing a more accurate measure of serum E2 levels in men.
Despite this, when we compared correlations of vBMD at various skeletal sites with total and bioavailable T or E2 levels derived from either assay, we obtained virtually identical values for the unadjusted and age-adjusted correlation coefficients. These findings suggest that conclusions regarding relationships of sex steroid levels to cross-sectional or longitudinal changes in BMD by us (3-6) and others (7-12) using immunoassays are generally valid. Nonetheless, the greater accuracy of the mass spectroscopy measurements likely make this the preferred method for assessing sex steroid levels, particularly E2 levels, in men. Thus, when serum total estradiol levels need to be obtained in men to search, for example, for possible aromatase defects or other abnormalities of estrogen metabolism, our data would suggest that mass spectroscopy would be the preferred method for assessing total estradiol levels clinically in men. However, since mass spectroscopy is not currently widely available and has considerably higher cost than immunoassays, our findings would suggest that use of a validated immunoassay is likely to provide overall results for correlations with bone density similar to those observed using mass spectroscopy, at least in men.
A caveat to our findings is that we used carefully validated, highly sensitive and specific immunoassays [as previously described (3)] for T and E2 to compare with the mass spectroscopy values. Since not all immunoassays have similar performance characteristics, it may be necessary for investigators to perform at least an initial validation of the immunoassay being used against mass spectroscopy; preferably, this could be done by the vendors marketing the immunoassays. For example, a recent study from the Dubbo Osteoporosis Epidemiology Study (23) found a weaker association between the immunoassay for total T used by that group and mass spectroscopy measurements than the concordance noted between the two methods in our study. Moreover, the even lower values for T and E2 in postmenopausal women, as compared to men (3), may well require mass spectroscopy as the principal approach, as has been recently suggested (17).
Our findings using mass spectroscopy are also consistent with previous findings by us (3) and others (2) that, whereas total T or E2 change only modestly (or not at all) over life in men, bioavailable T and E2 levels decline substantially. While there is increasing evidence for possible independent effects of SHBG on target tissues (24), the stronger correlations in the present and previous (3-8) work demonstrating more robust associations between BMD and bioavailable sex steroids, as compared to total sex steroid levels, do support the concept that (at least for bone) the biologically relevant fraction of T and E2 may be the non-SHBG bound sex steroids. However, direct studies, which are difficult to do even in animals, are needed to rigorously test this assumption. Finally, cross-calibration of mass spectroscopy instrumentation should be easier in principle than standardization of immunoassays so that increased use of mass spectroscopy for measuring sex steroid levels should help in standardizing measurements across institutions and perhaps provide a common basis for studies examining criteria for sex steroid deficiency in men.
Acknowledgments
We would like to thank Lisa McDaniel, R.N. for recruitment of study subjects, Kelley Hoey for processing of the samples, Sara Achenbach for assistance with the statistical analyses, and Jim Peterson for making the figures.
Supported by NIH Grants AR027065 and 1UL1RR024150.
Footnotes
In men, measurement of serum testosterone and estradiol levels with immunoassays correlated with mass spectroscopic measurements, and correlations of sex steroids with volumetric bone mineral density were similar.
References
- 1.Khosla S, Melton LJ, Riggs BL. Estrogen and the male skeleton. J Clin Endocrinol Metab. 2002;87:1443–1450. doi: 10.1210/jcem.87.4.8417. [DOI] [PubMed] [Google Scholar]
- 2.Kaufman JM, Vermeulen A. The decline of androgen levels in elderly men and its clinical and therapeutic implications. Endocr Rev. 2005;26:833–876. doi: 10.1210/er.2004-0013. [DOI] [PubMed] [Google Scholar]
- 3.Khosla S, Melton LJ, III, Atkinson EJ, O'Fallon WM, Klee GG, Riggs BL. Relationship of serum sex steroid levels and bone turnover markers with bone mineral density in men and women: A key role for bioavailable estrogen. J Clin Endocrinol Metab. 1998;83:2266–2274. doi: 10.1210/jcem.83.7.4924. [DOI] [PubMed] [Google Scholar]
- 4.Khosla S, Melton LJ, Atkinson EJ, O'Fallon WM. Relationship of serum sex steroid levels to longitudinal changes in bone density in young versus elderly men. J Clin Endocrinol Metab. 2001;86:3555–3561. doi: 10.1210/jcem.86.8.7736. [DOI] [PubMed] [Google Scholar]
- 5.Khosla S, Melton LJ, III, Robb RA, Camp JJ, Atkinson EJ, Oberg AL, Rouleau PA, Riggs BL. Relationship of volumetric BMD and structural parameters at different skeletal sites to sex steroid levels in men. J Bone Miner Res. 2005;20:730–740. doi: 10.1359/JBMR.041228. [DOI] [PubMed] [Google Scholar]
- 6.Khosla S, Melton LJ, III, Achenbach SJ, Oberg AL, Riggs BL. Hormonal and biochemical determinants of trabecular microstructure at the ultradistal radius in women and men. J Clin Endocrinol Metab. 2006;91:885–891. doi: 10.1210/jc.2005-2065. [DOI] [PubMed] [Google Scholar]
- 7.Van Pottelbergh I, Goemaere S, Kaufman JM. Bioavailable estradiol and an aromatase gene polymorphism are determinants of bone mineral density changes in men over 70 years of age. J Clin Endocrinol Metab. 2003;88:3075–3081. doi: 10.1210/jc.2002-021691. [DOI] [PubMed] [Google Scholar]
- 8.Gennari L, Merlotti D, Martini G, Gonnelli S, Franci B, Campagna S, Lucani B, Canto ND, Valenti R, Gennari C, Nuti R. Longitudinal association between sex hormone levels, bone loss, and bone turnover in elderly men. J Clin Endocrinol Metab. 2003;88:5327–5333. doi: 10.1210/jc.2003-030736. [DOI] [PubMed] [Google Scholar]
- 9.Eriksson AL, Lorentzon M, Mellstrom D, Vandenput L, Swanson C, Andersson N, Hammond GL, Jakobsson J, Rane A, Orwoll ES, Ljunggren O, Johnell O, Labrie F, Windahl SH, Ohlsson C. SHBG gene promoter polymorphisms in men are associated with serum sex hormone-binding globulin, androgen and androgen metabolite levels, and hip bone mineral density. J Clin Endocrinol Metab. 2006;91:5029–5037. doi: 10.1210/jc.2006-0679. [DOI] [PubMed] [Google Scholar]
- 10.Ensrud KE, Lewis CE, Lambert LC, Taylor BC, Fink HA, Barrett-Connor E, Cauley JA, Stefanick ML, Orwoll E, Group OFiMSR Endogenous sex steroids, weight change and rates of hip bone loss in older men: the MrOS study. 2006;17:1329–1336. doi: 10.1007/s00198-006-0088-z. [DOI] [PubMed] [Google Scholar]
- 11.Amin S, Zhang Y, Sawin CT, Evans SR, Hannan MT, Kiel DP, Wilson PWF, Felson DT. Association of hypogonadism and estradiol levels with bone mineral density in elderly men from the Framingham study. Ann Intern Med. 2000;133:951–963. doi: 10.7326/0003-4819-133-12-200012190-00010. [DOI] [PubMed] [Google Scholar]
- 12.Amin S, Zhang Y, Felson DT, Sawin CT, Hannan MT, Wilson PWF, Kiel DP. Estradiol, testosterone, and the risk for hip fractures in elderly men from the Framingham Study. Am J Med. 2006;119:426–433. doi: 10.1016/j.amjmed.2005.10.048. [DOI] [PubMed] [Google Scholar]
- 13.O'Connor S, Baker HWG, Dulmanis A, Hudson B. The measurement of sex steroid binding globulin by differential ammonium sulphate precipitation. J Steroid Biochem. 1973;4:331–339. doi: 10.1016/0022-4731(73)90002-2. [DOI] [PubMed] [Google Scholar]
- 14.Tremblay RR, Dube JY. Plasma concentrations of free and non-TeBG bound testosterone in women on oral contraceptives. Contraception. 1974;10:599–605. doi: 10.1016/0010-7824(74)90099-7. [DOI] [PubMed] [Google Scholar]
- 15.Vermeulen A, Verdonck L, Kaufman JM. A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab. 1999;84:3666–3672. doi: 10.1210/jcem.84.10.6079. [DOI] [PubMed] [Google Scholar]
- 16.Sodergard R, Backstrom T, Shanbhag V, Carstensen H. Calculation of free and bound fractions of testosterone and estradiol-17 beta to human plasma proteins at body temperature. J Steroid Biochem. 1982;16:801–810. doi: 10.1016/0022-4731(82)90038-3. [DOI] [PubMed] [Google Scholar]
- 17.Lee JS, Ettinger B, Stanczyk FZ, Vittinghoff E, Hanes V, Cauley JA, Chandler W, Settlage J, Beattie MS, Folkerd E, Dowsett M, Grady D, Cummings SR. Comparison of methods to measure low serum estradiol levels in postmenopausal women. J Clin Endocrinol Metab. 2006;91:3791–3797. doi: 10.1210/jc.2005-2378. [DOI] [PubMed] [Google Scholar]
- 18.Melton LJ., III History of the Rochester Epidemiology Project. Mayo Clin Proc. 1996;71:266–274. doi: 10.4065/71.3.266. [DOI] [PubMed] [Google Scholar]
- 19.Riggs BL, Melton LJ, III, Robb RA, Camp JJ, Atkinson EJ, Peterson JM, Rouleau PA, McCollough CH, Bouxsein ML, Khosla S. Population-based study of age and sex differences in bone volumetric density, size, geometry, and structure at different skeletal sites. J Bone Miner Res. 2004;19:1945–1954. doi: 10.1359/JBMR.040916. [DOI] [PubMed] [Google Scholar]
- 20.Camp JJ, Karwoski RA, Stacy MC, Atkinson EJ, Khosla S, Melton LJ, Riggs BL, Robb RA. A system for the analysis of whole-bone strength from helical CT images. Proceedings of SPIE. 2004;5369:74–88. [Google Scholar]
- 21.Cann CE. Quantitative CT for determination of bone mineral density: a review. Radiology. 1988;166:509–522. doi: 10.1148/radiology.166.2.3275985. [DOI] [PubMed] [Google Scholar]
- 22.Pardridge WM. Serum bioavailability of sex steroid hormones. Clin Endocrinol Metab. 1986;15:259–278. doi: 10.1016/s0300-595x(86)80024-x. [DOI] [PubMed] [Google Scholar]
- 23.Nguyen TV, Meier C, Handelsman DJ, Kraenzlin M, Kushnir MM, Rockwood AL, Meikle AW, Center JR, Eisman JA, Seibel MJ. Serum testosterone in elderly men measured by liquid chromatography-tandem mass spectrometry and radioimmunoassay: concordance and effect on epidemiologic association. J Bone Miner Res. 2007;22(Suppl 1):S277. Abstract. [Google Scholar]
- 24.Khosla S. Editorial: Sex hormone binding globulin: inhibitor or facilitator (or both) of sex steroid action? J Clin Endocrinol Metab. 2006;91:4764–4766. doi: 10.1210/jc.2006-1990. [DOI] [PubMed] [Google Scholar]