Abstract
The migration of neuronal growth cones, driving axon extension, is a fascinating process which has been subject of intense investigation over several decades. Many of the key underlying molecules, in particular adhesion proteins at the cell membrane which allow for target recognition and binding, and cytoskeleton filaments and motors which power locomotion have been identified. However, the precise mechanisms by which growth cones coordinate, in time and space, the transmission of forces generated by the cytoskeleton to the turnover of adhesion proteins are still partly unresolved. To get a better grasp at these processes, we put here in relation the turnover rate of ligand/receptor adhesions and the degree of mechanical coupling between cell adhesion receptors and the actin rearward flow. These parameters were obtained recently for N-cadherin and IgCAM based adhesions using ligand-coated microspheres in combination with optical tweezers and photo-bleaching experiments. We show that the speed of growth cone migration requires both a fairly rapid adhesion dynamics and a strong physical connection between adhesive sites and the cytoskeleton.
Key words: actin retrograde flow, molecular clutch, myosin, N-cadherin, IgCAM
Growth cones are motile structures at the distal extremity of axons responsible for pathfinding and neurite extension during nervous system development and repair (Fig. 1A). Growth cone advance relies on two coupled processes. First, an internal dynamics of the cytoskeletal network, with actin polymerization occurring at the leading edge, depolymerization in the central region, and myosin activity pulling on lamellipodial actin filaments.1 These integrated mechanisms altogether result in a continuous retrograde flow of actin (Fig. 1B). This flow provides the mechanical tension that drives axonal extension, through a connection to the dynamic array of microtubules that fills the axon and invades the growth cone central domain.2 Second, there is repeated formation and dissociation of transient contacts between growth cones and the extracellular matrix or adjacent cells. These contacts are mediated by trans-membrane cell adhesion molecules (CAMs), e.g. integrins,3 immunoglobulin CAMs (IgCAMs)4,5 and cadherins,6,7 which form specific ligand/receptor bonds with variable lifetimes. A still open question is how these two processes, i.e. actin flow and adhesion dynamic, are coordinated at the growth cone level and contribute to set migration speed. A thorough understanding of these mechanisms is important both from a fundamental perspective and for the design of new compounds to foster axon regeneration after injury.
Figure 1.
Growth cone advance and actin flow. (A) Growth cone from a 2 DIV rat hippocampal neuron plated on N-cadherin coated glass. This growth cone moved forward at a speed of about 1 µm/min (B) Raw fluorescence image of transfected actin-GFP. (C) Sequential actin-GFP images were subtracted, giving rise to intensity variations that display the movement of newly assembled actin (black). Note the rapid retrograde movement of actin spots (arrowheads), at a velocity of several µm/min.
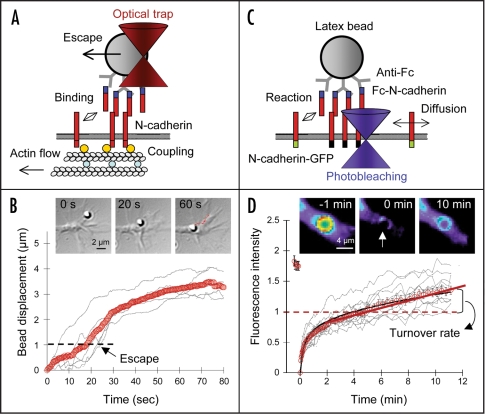
The coupling between actin-based motility and substrate adhesion has been shown for certain adhesion molecules such as NCAM and N-cadherin to involve a “molecular clutch” (Fig. 2). This mechanism implies a direct transmission of traction forces from the cytoskeleton to the substrate through a strong physical connection between the actin flow and ligand-bound adhesion receptors.8,9 The connection is likely provided by adaptor proteins that can make transient bridges between actin filaments and the cytoplasmic domain of adhesion molecules, i.e. α-and β-catenin in the case of N-cadherin,10 ankyrin and ezrin in the case of IgCAMs such as L1.11–14 These purely mechanical connections can also be accompanied by signalling events such as Rac-1 activation by N-cadherin liganding15 and phosphorylation of the L1 intracellular tail that regulates binding to ankyrin.11,12 When only few molecular bonds are formed, e.g. at low ligand density, coupling to the actin flow is not strong enough, resulting in “slippage.” In this process, transient bonds can be formed and broken repeatedly between ligand-occupied adhesion receptors and the actin network. This is how the speed of growth cone translocation usually reaches at most 1 µm/min, whereas the internal actin flow rate proceeds at a rate of several µm/min (Fig. 1A and B). Such slippage is best demonstrated by the use of optical tweezers to impose low forces on ligand-coated microspheres presented to the growth cone dorsal surface (Fig. 3A). Beads tend to move rearward as they couple to the actin flow, and then suddenly snap back into the trap center, when receptor-cytoskeleton bonds break16 (the force of optical tweezers is usually not enough to rupture ligand-receptor bonds, which remain intact at the cell surface). Thus, a step in which a nucleating cluster of adhesion receptors recruits a minimal number of intracellular partners allowing coupling to the actin flow, can be a rate-limiting factor in growth cone progression.
Figure 2.
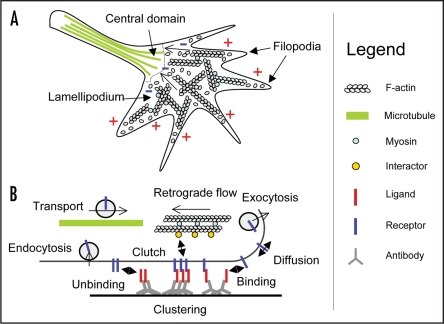
Molecular components involved in growth cone migration. (A) Top view diagram showing filopodia which sense the environment, a flat lamellipodium which is the site of actin dynamics and the thicker central domain and axon which contain dynamic microtubules. The plus signs are sites of actin polymerization and the minus signs indicate actin depolymerization. (B) Side view showing the life cycle of ligand/receptor adhesions.
Figure 3.
Optical tweezers and FRAP experiments to measure ligand/receptor and receptor/cytoskeleton dynamics. (A) Optical tweezers experiments performed on ligand-coated beads placed on the growth cone dorsal surface.16 (B) The distance traveled rearward with respect to the trap center is measured. A 2 min trajectory is indicated in red. A pooled parameter called coupling index taking into account the latency for bead escape, as well as the mean velocity and lateral diffusion of the bead, measures the strength of receptor/cytoskeleton interactions. (C) FRAP experiments on membrane GFP-tagged molecules accumulated at ligand-coated microspheres having sedimented on growth cones. The fluorescence intensity is normalized to represent the receptor enrichment level at the bead contact. (D) The recovered intensity is fit by a diffusion/reaction model, which yields a collective equilibrium turnover rate of ligand/receptor bonds. In red is the average of a series of individual curves (grey).
In contrast, when strong connection is formed and if the substrate is resistant enough, then the molecular clutch engages and the cell reacts. In growth cones from Aplysia bag cells, forces were imposed on microspheres coated with ApCAM (the homolog of vertebrate NCAM) using a microneedle to locally block the retrograde actin flow. This was systematically followed by a protrusion of the microtubule-rich central domain towards those stiff contacts and forward expansion of the actin-rich lamellipodium.9 These phenomena were later shown to be controlled by a src protein kinase.17 In the case of rat hippocampal neurons, a dramatic accumulation of actin at N-cadherin coated microspheres is observed when the latter are restrained from moving rearward by a microneedle.16 This phenomenon is mediated by a connection between N-cadherin and α-catenin, likely triggering local actin polymerization. By careful analysis of the bead trajectories at varying ligand densities and computation of the latency for bead escape when the optical trap is applied continuously, one can extract a quantitative index of receptor-cytoskeleton coupling (Fig. 3B). Overall, a strong correlation was observed between such coupling index and the velocity of growth cone migration on N-cadherin substrates, both by varying N-cadherin ligand density and by expressing mutated N-cadherin molecules, supporting the clutch concept.16 This mechanism is consistent with in vivo experiments showing that overexpression of the N-cadherin intracellular tail in retinal ganglion cells results in severely impaired axon outgrowth.18 As a negative example of the clutch model, beads coated with fibronectin (our unpublished data) or anti-α1 integrin antibodies3 couple weakly to the actin flow in growth cones while, in parallel, the migration of growth cones on fibronectin- or collagen-coated substrates is rather limited.6,19
Molecular mechanisms parallel to the “clutch” can also be involved in growth cone migration. For example, IgCAM adhesions can not only couple to the rearward actin flow but also to static components of the cytoskeleton. Indeed, a 30% fraction of TAG-1 or anti-L1 coated beads can stay immobile on the growth cone surface.12,20 These contrasting behaviors are likely mediated by interactions between the IgCAM intracellular domain and different binding partners (ankyrin vs. ERM),13 and may be responsible for the pauses which alternate with phases of growth cone advance. Also, homophilic adhesions between molecules of cadherin-11 couple very weakly to the actin flow, but promote substantial growth cone migration when cadherin-11 is presented as a substrate. This effect seems to be mediated by an independent interaction with the FGF receptor, which triggers actin dynamic through a signaling cascade.21,22
As growth cones migrate, adhesion sites must be recycled at a rate that somehow matches the speed of migration. Adhesion turnover can be schematically decomposed in several sequential phases (Fig. 2B). (1) Initiation of a first single ligand/receptor bond powered by membrane diffusion23 and followed by trapping through a key/lock interaction; (2) Formation of small adhesion clusters through the recruitment of more ligand/receptor pairs, and possibly stabilized by cis-oligomerization (cadherins through the same interface as the trans-dimer, IgCAMs through FnIII domains). These clusters might form very transiently and serve as sites of actin recruitment, as demonstrated for N-cadherin;16 (3) contact maturation and possible reinforcement by connection to the cytoskeleton (as demonstrated for integrins in fibroblasts24); (4) Adhesion rupture, which can proceed through ligand/receptor dissociation triggered by cytoskeleton tension. Indeed, the intrinsic lifetime of ligand/receptor bonds such as cadherins, is sensitive to the mechanical force applied on them.25 Furthermore, the loosening of receptor/cytoskeketon connections can cause inside-out rupture of ligand/receptor bonds. This was demonstrated for fibronectin/integrin interactions by the fact that when fibronectin coated-beads reach the base of a fibroblast lamellipodium, they spontaneously detach from the cell surface.26 In the case of very sticky ligand/receptor interactions such as SynCAM homophilic adhesions,27 this process can actually be a limiting step that slows down growth cone advance. Indeed, SynCAM couples very well to the actin flow, but is unable to support growth cone migration.16 Finally, adhesion rupture might also proceed through membrane rupture, the adhesion receptors being extracted from the cell membrane and left behind on the substrate (demonstrated for integrins at the tail of fibroblasts28).
These basic processes can be accompanied by more complex and active phenomena, e.g. involving forward surface transport as shown for NCAM29 or internal trafficking in the case of L1.30 By interacting with the clathrin adaptor AP-2 through a specific RSLE motif in its intracellular tail, L1 can undergo endocytosis in the central domain and exocytosis at the periphery of the growth cone.30 This mechanism generates a density gradient of L1 molecules which accelerates the formation of bonds with a variety of ligands, including L1 itself. The use of an L1-GFP construct in which the N-terminal GFP could be rapidly cleaved off by thrombin, together with L1-Fc microspheres manipulated by optical tweezers showed that local exocytosis of L1-rich vesicles at the growth cone periphery indeed participates in enhancing the formation of L1 homophilic contacts.31 We did not observe such internal traffic for N-cadherin within the growth cone, partly because of a difficulty to introduce a fluorescent protein tag in the ectodomain, which otherwise perturbs the adhesive function. However, the use of an N-cadherin molecule with triple mutation in the juxta-membrane domain that abolishes binding to p120 catenin, involved in the export of N-cadherin to the cell surface, suggested that recycling events might also play a role.32
By measuring the fluorescence recovery after photobleaching (FRAP) of GFP-tagged receptors transiently trapped at ligand-coated microspheres and analyzing the curves using a diffusion/reaction model, we were able to compute the equilibrium turnover rates of ligand/receptor pairs in controlled adhesive contacts involving many simultaneous bonds (Fig. 3C and D). We found that mature L1 homophilic adhesions recycle fast compared to other IgCAMs such as TAG-1/NrCAM adhesions,20 likely owing to the specific internalization motif present in L1. Indeed, the recycling rate was reduced by a factor of 3 after truncation of the L1 intracellular tail, which prevented endocytosis.31 N-cadherin homophilic adhesions have an intermediate turnover rate, which is sensitive to the binding to catenin partners.32 Using these measurements as well as data from the literature, we plotted the impact of both receptor-cytoskeleton coupling and adhesion turnover rate on neurite outgrowth (Fig. 4A and B), which is strongly proportional to growth cone velocity.16 The graphs show that a strong coupling between ligand-occupied receptors and the actin flow is necessary, but not sufficient for neurite extension (Fig. 4C). Another requirement is that the turnover of ligand/receptor adhesions lies in an optimal range: not too high, otherwise bonds detach before coupling can occur, and not too slow either, since sticky bonds which do not rupture paralyze growth cone progression (Fig. 4D). A similar bell-shape curve between the strength of cell-substrate adhesion and cell migration speed was demonstrated for fibroblasts33 and keratocytes,34 indicating that these coupled mechanisms are fundamental to cell migration. To fully understand the quantitative relationship between adhesion turnover and the clutch process, it would be helpful to add data to this preliminary graph. For example, the extracellular matrix molecule laminin is known to support axon growth very efficiently but, to our knowledge, neither the coupling to the actin flow in growth cones or the adhesive turnover rate of integrins has been evaluated yet. Conversely, NCAM was shown to couple well to the actin flow35 and induce neurite outgrowth,4,36 but measurements of the lifetime of NCAM homophilic adhesions within growth cones are still lacking.
Figure 4.
Impact of ligand/receptor turnover rate and receptor/cytoskeletal coupling on neurite outgrowth. (A and B) Example of 2 DIV rat hippocampal neurons plated on N-cadherin-Fc coated substrate and transfected at 1 DIV with N-cadherin-GFP. (A) DIC image. (B) Fluorescence image. The longest neurite, most likely the axon, is outlined by arrowheads. (C and D) In both graphs, the y-axis represents the longest neurite length after two days plating on ligand-coated glass. (Red) Rat hippocampal neurons transfected with either wild type or mutated N-cadherin molecules, interacting with purified N-cadherin ligands.16,32 The scale in red intensity represents from dark to light: wild type N-cadherin, N-cadherin deleted of the whole ectodomain, N-cadherin truncated in the C-terminal region binding to β-catenin, N-cadherin with triple mutation in the juxta-membrane domain interacting with p120, and wild type N-cadherin in the presence of cytochalasin D. (Blue) Neurons transfected with either wild type L1 (dark) or L1 truncated in the intracellular tail (light), interacting with purified L1.31 Neurite growth on L1 was estimated from references.7,14,30 (Grey) Interaction between endogenous SynCAM1 molecules expressed on growth cones and SynCAM-Fc ligands coated on microspheres or flat glass.16 The turnover of SynCAM homophilic interactions was estimated from SynCAM-coated Quantum dots detaching from neurons transfected with SynCAM1.42 (Green) Neuroblastoma cells expressing NrCAM-GFP in contact with TAG-1 coated microspheres.20 DRG neurite growth on TAG-1 was taken from reference.43 (Orange) The coupling index was taken from optical tweezers experiments using anti-β1 integrin coated beads interacting with DRG neurons,3 the turnover rate was inferred from FRAP experiments on fibroblast focal contacts44 and neurite growth on fibronectin was estimated from reference.19 We omitted statistics for clarity. The SEM are usually in the order of 5–15% of the mean, for sample sizes of typically 20–30 beads (coupling index and turnover rate) and 40–100 transfected cells (neurite length).
One important question is how these observations obtained from simplified in vitro systems using stiff substrates of well-defined geometry coated with specific purified proteins at controlled density, translate to the in vivo situation. There, the 3D substrate is comprised of extracellular matrix and multiple cell types, co-expressing many different CAMs that can bind simultaneously in various stoechiometries and also generating local gradients of chemo-attractant and chemo-repulsive signals.37 Substrate flexibility is probably an important factor, since axons grow more slowly when neurons are plated on a layer of fibroblasts expressing CAMs21 than when molecules are immobilized on a substrate.16,30 This preference for cells to move on stiff substrates, called durotaxis, has been well described for fibroblasts.38 Another specific feature of the in vivo situation is the existence of decision points, often correlated with the presence of guidepost cells where growth cones make a pause and often change shape and reorient before turning to another direction.39 This type of behavior has been successfully mimicked in vitro using artificial guideposts made of fibronectin or laminin coated microspheres.40 Whereas growth cones display a fairly continuous displacement on a homogeneous substrate,16 the presence of these guideposts make growth cones either slow down, pause or even collapse, or conversely accelerate, depending on the CAM grafted on the bead.40,41 Finally, the shape itself of the growth cone can be an indicator of its motile state:39 this is also true in vitro where small growth cones are often the most rapid whereas large and flat growth cones stay rather immobile. Thus, although the in vivo situation seems at first sight awfully complex, some general trends can be explained given a small number of interacting molecular species and rather simple bio-chemical and mechanical models.
In conclusion, the dynamic regulation of growth cone advance can take place at several levels: (1) the actin-associated proteins controlling actin dynamics (nucleation, polymerization, sequestering, branching); (2) the activity of motors pulling on the actin network, generating the retrograde flow; (3) the intracellular adaptor proteins that link actin to the CAMs; (4) the membrane delivery and retrieval of CAMs; (5) the ligand/receptor interaction properties themselves; and (6) the processes regulating microtubule assembly and microtubule/actin interactions at the base of the growth cone. The orchestration in time and space of all these processes generates the movement and reactivity of growth cones necessary to lead axons to their target cells.
Acknowledgements
I thank D. Choquet for continuous support, C. Breillat, D. Bouchet, A. Frouin and B. Tessier for neuronal cultures and molecular biology, P. Gonzales for coverslip preparation, R.M. Mège and M. Lambert for a long standing collaboration, J. Falk, C. Sarrailh, T. Galli, P. Alberts and L. Danglot, for the gift of reagents and helpful discussions, and L. Bard, C. Dequidt, E. Saint-Michel and European Synapse Summer School students for obtaining some of the data presented here. I acknowledge financial support from the French Ministry of Research and CNRS, Conseil Régional Aquitaine, AFM, ARC and INSERM.
Footnotes
Previously published online as a Cell Adhesion & Migration E-publication: http://www.landesbioscience.com/journals/celladhesion/article/7274
References
- 1.Diefenbach TJ, Latham VM, Yimlamai D, Liu CA, Herman IM, Jay DG. Myosin 1c and myosin IIB serve opposing roles in lamellipodial dynamics of the neuronal growth cone. J Cell Biol. 2002;158:1207–1217. doi: 10.1083/jcb.200202028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schaefer AW, Schoonderwoert VT, Ji L, Mederios N, Danuser G, Forscher P. Coordination of actin filament and microtubule dynamics during neurite outgrowth. Dev Cell. 2008;15:146–162. doi: 10.1016/j.devcel.2008.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schmidt CE, Dai J, Lauffenburger DA, Sheetz MP, Horwitz AF. Integrin-cytoskeletal interactions in neuronal growth cones. J Neurosci. 1995;15:3400–3407. doi: 10.1523/JNEUROSCI.15-05-03400.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Friedlander DR, Milev P, Karthikeyan L, Margolis RK, Margolis RU, Grumet M. The neuronal chondroitin sulfate proteoglycan neurocan binds to the neural cell adhesion molecules Ng-CAM/L1/NILE and N-CAM, and inhibits neuronal adhesion and neurite outgrowth. J Cell Biol. 1994;125:669–680. doi: 10.1083/jcb.125.3.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haspel J, Friedlander DR, Ivgy-May N, Chickramane S, Roonprapunt C, Chen S, et al. Critical and optimal Ig domains for promotion of neurite outgrowth by L1/Ng-CAM. J Neurobiol. 2000;42:287–302. [PubMed] [Google Scholar]
- 6.Bixby JL, Zhang R. Purified N-cadherin is a potent substrate for the rapid induction of neurite outgrowth. J Cell Biol. 1990;110:1253–1260. doi: 10.1083/jcb.110.4.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lemmon V, Burden SM, Payne HR, Elmslie GJ, Hlavin ML. Neurite growth on different substrates: permissive versus instructive influences and the role of adhesive strength. J Neurosci. 1992;12:818–826. doi: 10.1523/JNEUROSCI.12-03-00818.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mitchison T, Kirschner M. Cytoskeletal dynamics and nerve growth. Neuron. 1988;1:761–772. doi: 10.1016/0896-6273(88)90124-9. [DOI] [PubMed] [Google Scholar]
- 9.Suter DM, Errante LD, Belotserkovsky V, Forscher P. The Ig superfamily cell adhesion molecule, apCAM, mediates growth cone steering by substrate-cytoskeletal coupling. J Cell Biol. 1998;141:227–240. doi: 10.1083/jcb.141.1.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yamada S, Pokutta S, Drees F, Weis WI, Nelson WJ. Deconstructing the cadherin-cateninactin complex. Cell. 2005;123:889–901. doi: 10.1016/j.cell.2005.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Garver TD, Ren Q, Tuvia S, Bennett V. Tyrosine phosphorylation at a site highly conserved in the L1 family of cell adhesion molecules abolishes ankyrin binding and increases lateral mobility of neurofascin. J Cell Biol. 1997;137:703–714. doi: 10.1083/jcb.137.3.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gil OD, Sakurai T, Bradley AE, Fink MY, Cassella MR, Kuo JA, et al. Ankyrin binding mediates L1CAM interactions with static components of the cytoskeleton and inhibits retrograde movement of L1CAM on the cell surface. J Cell Biol. 2003;162:719–730. doi: 10.1083/jcb.200211011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nishimura K, Yoshihara F, Tojima T, Ooashi N, Yoon W, Mikoshiba K, et al. L1-dependent neuritogenesis involves ankyrinB that mediates L1-CAM coupling with retrograde actin flow. J Cell Biol. 2003;163:1077–1088. doi: 10.1083/jcb.200303060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cheng L, Itoh K, Lemmon V. L1-mediated branching is regulated by two ezrin-radixin-moesin (ERM)-binding sites, the RSLE region and a novel juxtamembrane ERM-binding region. J Neurosci. 2005;25:395–403. doi: 10.1523/JNEUROSCI.4097-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lambert M, Choquet D, Mege RM. Dynamics of ligand-induced, Rac1-dependent anchoring of cadherins to the actin cytoskeleton. J Cell Biol. 2002;157:469–479. doi: 10.1083/jcb.200107104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bard L, Boscher C, Lambert M, Mege RM, Choquet D, Thoumine O. A molecular clutch between the actin flow and N-cadherin adhesions drives growth cone migration. J Neurosci. 2008;28:5879–5890. doi: 10.1523/JNEUROSCI.5331-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Suter DM, Forscher P. Transmission of growth cone traction force through apCAM-cytoskeletal linkages is regulated by Src family tyrosine kinase activity. J Cell Biol. 2001;155:427–438. doi: 10.1083/jcb.200107063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Riehl R, Johnson K, Bradley R, Grunwald GB, Cornel E, Lilienbaum A, et al. Cadherin function is required for axon outgrowth in retinal ganglion cells in vivo. Neuron. 1996;17:837–848. doi: 10.1016/s0896-6273(00)80216-0. [DOI] [PubMed] [Google Scholar]
- 19.Gardiner NJ, Moffatt S, Fernyhough P, Humphries MJ, Streuli CH, Tomlinson DR. Preconditioning injury-induced neurite outgrowth of adult rat sensory neurons on fibronectin is mediated by mobilisation of axonal alpha5 integrin. Mol Cell Neurosci. 2007;35:249–260. doi: 10.1016/j.mcn.2007.02.020. [DOI] [PubMed] [Google Scholar]
- 20.Falk J, Thoumine O, Dequidt C, Choquet D, Faivre-Sarrailh C. NrCAM coupling to the cytoskeleton depends on multiple protein domains and partitioning into lipid rafts. Mol Biol Cell. 2004;15:4695–4709. doi: 10.1091/mbc.E04-03-0171. Epub 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saffell JL, Williams EJ, Mason IJ, Walsh FS, Doherty P. Expression of a dominant negative FGF receptor inhibits axonal growth and FGF receptor phosphorylation stimulated by CAMs. Neuron. 1997;18:231–242. doi: 10.1016/s0896-6273(00)80264-0. [DOI] [PubMed] [Google Scholar]
- 22.Boscher C, Mege RM. Cadherin-11 interacts with the FGF receptor and induces neurite outgrowth through associated downstream signalling. Cell Signal. 2008;20:1061–1072. doi: 10.1016/j.cellsig.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 23.Thoumine O, Saint-Michel E, Dequidt C, Falk J, Rudge R, Galli T, et al. Weak effect of membrane diffusion on the rate of receptor accumulation at adhesive contacts. Biophys J. 2005;89:40–42. doi: 10.1529/biophysj.105.071688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Choquet D, Felsenfeld DP, Sheetz MP. Extracellular matrix rigidity causes strengthening of integrin-cytoskeleton linkages. Cell. 1997;88:39–48. doi: 10.1016/s0092-8674(00)81856-5. [DOI] [PubMed] [Google Scholar]
- 25.Perret E, Benoliel AM, Nassoy P, Pierres A, Delmas V, Thiery JP, et al. Fast dissociation kinetics between individual E-cadherin fragments revealed by flow chamber analysis. Embo J. 2002;21:2537–2546. doi: 10.1093/emboj/21.11.2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nishizaka T, Shi Q, Sheetz MP. Position-dependent linkages of fibronectin-integrin-cytoskeleton. Proc Natl Acad Sci USA. 2000;97:692–697. doi: 10.1073/pnas.97.2.692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Breillat C, Thoumine O, Choquet D. Characterization of SynCAM surface trafficking using a SynCAM derived ligand with high homophilic binding affinity. Biochem Biophys Res Commun. 2007 doi: 10.1016/j.bbrc.2007.05.152. [DOI] [PubMed] [Google Scholar]
- 28.Regen CM, Horwitz AF. Dynamics of beta1 integrin-mediated adhesive contacts in motile fibroblasts. J Cell Biol. 1992;119:1347–1359. doi: 10.1083/jcb.119.5.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sheetz MP, Baumrind NL, Wayne DB, Pearlman AL. Concentration of membrane antigens by forward transport and trapping in neuronal growth cones. Cell. 1990;61:231–241. doi: 10.1016/0092-8674(90)90804-n. [DOI] [PubMed] [Google Scholar]
- 30.Kamiguchi H, Yoshihara F. The role of endocytic l1 trafficking in polarized adhesion and migration of nerve growth cones. J Neurosci. 2001;21:9194–9203. doi: 10.1523/JNEUROSCI.21-23-09194.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dequidt C, Danglot L, Alberts P, Galli T, Choquet D, Thoumine O. Fast turnover of L1 adhesions in neuronal growth cones involving both surface diffusion and exo/endocytosis of L1 molecules. Mol Biol Cell. 2007;18:3131–3143. doi: 10.1091/mbc.E06-12-1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thoumine O, Lambert M, Mege RM, Choquet D. Regulation of N-cadherin dynamics at neuronal contacts by ligand binding and cytoskeletal coupling. Mol Biol Cell. 2006;17:862–875. doi: 10.1091/mbc.E05-04-0335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Palecek SP, Loftus JC, Ginsberg MH, Lauffenburger DA, Horwitz AF. Integrin-ligand binding properties govern cell migration speed through cell-substratum adhesiveness. Nature. 1997;385:537–540. doi: 10.1038/385537a0. [DOI] [PubMed] [Google Scholar]
- 34.Jurado C, Haserick JR, Lee J. Slipping or gripping? Fluorescent speckle microscopy in fish keratocytes reveals two different mechanisms for generating a retrograde flow of actin. Mol Biol Cell. 2005;16:507–518. doi: 10.1091/mbc.E04-10-0860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thompson C, Lin CH, Forscher P. An Aplysia cell adhesion molecule associated with site-directed actin filament assembly in neuronal growth cones. J Cell Sci. 1996;109:2843–2854. doi: 10.1242/jcs.109.12.2843. [DOI] [PubMed] [Google Scholar]
- 36.Doherty P, Skaper SD, Moore SE, Leon A, Walsh FS. A developmentally regulated switch in neuronal responsiveness to NCAM and N-cadherin in the rat hippocampus. Development. 1992;115:885–892. doi: 10.1242/dev.115.3.885. [DOI] [PubMed] [Google Scholar]
- 37.Tessier-Lavigne M. Wiring the brain: the logic and molecular mechanisms of axon guidance and regeneration. Harvey Lect. 2002;98:103–143. [PubMed] [Google Scholar]
- 38.Lo CM, Wang HB, Dembo M, Wang YL. Cell movement is guided by the rigidity of the substrate. Biophys J. 2000;79:144–152. doi: 10.1016/S0006-3495(00)76279-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Stirling RV, Dunlop SA. The dance of the growth cones—where to next? Trends Neurosci. 1995;18:111–115. [PubMed] [Google Scholar]
- 40.Kuhn TB, Schmidt MF, Kater SB. Laminin and fibronectin guideposts signal sustained but opposite effects to passing growth cones. Neuron. 1995;14:275–285. doi: 10.1016/0896-6273(95)90285-6. [DOI] [PubMed] [Google Scholar]
- 41.Fan J, Raper JA. Localized collapsing cues can steer growth cones without inducing their full collapse. Neuron. 1995;14:263–274. doi: 10.1016/0896-6273(95)90284-8. [DOI] [PubMed] [Google Scholar]
- 42.Thoumine O, Ewers H, Heine M, Groc L, Frischknecht R, Giannone G, et al. Probing the dynamics of protein-protein interactions at neuronal contacts by optical imaging. Chem Rev. 2008;108:1565–1587. doi: 10.1021/cr078204m. [DOI] [PubMed] [Google Scholar]
- 43.Felsenfeld DP, Hynes MA, Skoler KM, Furley AJ, Jessell TM. TAG-1 can mediate homophilic binding, but neurite outgrowth on TAG-1 requires an L1-like molecule and beta 1 integrins. Neuron. 1994;12:675–690. doi: 10.1016/0896-6273(94)90222-4. [DOI] [PubMed] [Google Scholar]
- 44.Wehrle-Haller B. Analysis of integrin dynamics by fluorescence recovery after photobleaching. Methods Mol Biol. 2007;370:173–202. doi: 10.1007/978-1-59745-353-0_13. [DOI] [PubMed] [Google Scholar]