Abstract
Laser speckle imaging (LSI), a new technique that measures an index of plaque viscoelasticity, has been investigated recently to characterize atherosclerotic plaques. These prior studies demonstrated the diagnostic potential of LSI for detecting high-risk plaques and were conducted ex vivo. To conduct intracoronary LSI in vivo, the laser speckle pattern must be transmitted from the coronary wall to the image detector in the presence of cardiac motion. Small-diameter, flexible optical fiber bundles, similar to those used in coronary angioscopy, may be incorporated into an intravascular catheter for this purpose. A key challenge is that laser speckle is influenced by inter-fiber leakage of light, which may be exacerbated during bundle motion. In this study, we tested the capability of optical fiber bundles to transmit laser speckle patterns obtained from atherosclerotic plaques and evaluated the influence of motion on the diagnostic accuracy of fiber bundle-based LSI. Time-varying helium-neon laser speckle images of aortic plaques were obtained while cyclically moving the flexible length of the bundle to mimic coronary motion. Our results show that leached fiber bundles may reliably transmit laser speckle images in the presence of cardiac motion, providing a viable option to conduct intracoronary LSI.
Keywords: Biomedical optics, speckle, fiber optics, atherosclerosis, intracoronary, catheter
1 Introduction
Retrospective autopsy studies have indicated that the rupture of unstable coronary atherosclerotic plaque precedes the majority of ischemic cardiovascular events such as myocardial infarction.1 A certain type of plaque, termed the necrotic-core fibroatheroma (NCFA), is particularly vulnerable to rupture. Typical hallmarks of unstable NCFAs include the presence of a thin fibrous cap overlying a large necrotic lipid pool and macrophages near the fibrous cap.1,2 Laser speckle imaging (LSI) is a new technique that offers the unique capability of providing information about atherosclerotic plaque composition, morphology, and viscoelasticity, potentially yielding a powerful tool for detecting unstable NCFAs in patients prior to the occurrence of an acute coronary event.3,4
Laser speckle patterns, which appear as a granular pattern of high- and low-intensity spots, result from the interference of coherent light returning from a highly scattering medium. The dynamic behavior of laser speckle formation from scattering within tissue is highly dependent on the Brownian motion of suspended scatterers, which is in turn influenced by the viscoelasticity of the tissue. Consequently, in a NCFA, due to the relatively low viscosity of lipid, scatterers within the compliant necrotic core exhibit more rapid Brownian motion than the stiffer fibrous regions of the plaque. Since scatterer motion alters the optical path lengths of light within the tissue, the typical temporal behavior of the resulting speckle patterns is directly related to Brownian motion. As a result, the measurement of temporal intensity variations of laser speckle patterns provides information about the viscoelastic properties of the illuminated plaque. Using these principles, recent studies have demonstrated that measuring the intensity modulations of time-varying speckle patterns provides a highly sensitive method for distinguishing plaque type and evaluating collagen and lipid content ex vivo.4 By combining the analysis of spatial and temporal information from laser speckle patterns with a diffusion theory and Monte Carlo model to describe light propagation through tissue, a follow-up study demonstrated that it is possible to measure NCFA fibrous cap thickness from laser speckle patterns.5
Prior studies that have demonstrated the diagnostic potential of LSI for detecting high-risk plaques have been conducted ex vivo. To conduct intracoronary LSI in vivo, the laser speckle pattern must be transmitted from the coronary wall to the image detector. Small-diameter, flexible optical fiber bundles, similar to those used in coronary angioscopy,6 could be incorporated into an intravascular catheter for this purpose. However, the capability of fiber bundles to reliably transmit laser speckle data in the presence of motion such as that encountered in coronary arteries has not been evaluated. In this paper, we describe experiments that tested the capability of optical fiber bundles to transmit laser speckle patterns obtained from atherosclerotic plaques and evaluated the influence of motion on the diagnostic accuracy of optical fiber bundle-based LSI.
2 Methods
2.1 Laser Speckle Imaging
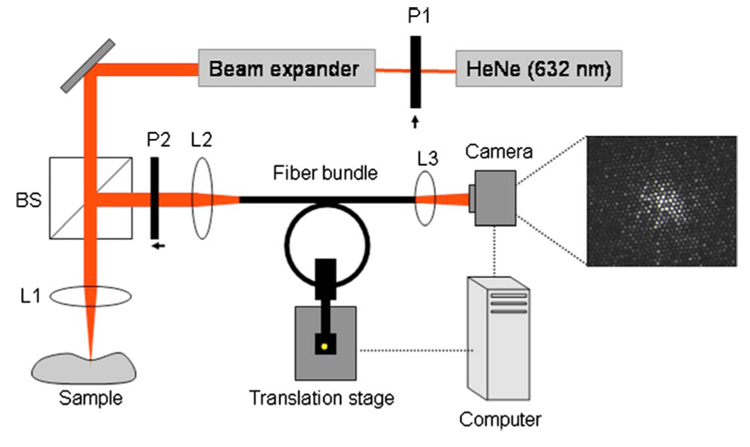
Thoracic and abdominal aortic specimens were obtained from nine human cadavers. Immediately following harvest, the aortas were stored in phosphate buffered saline (PBS). The time between autopsy and imaging did not exceed 48 hours. A bench-top system was constructed to acquire laser speckle images of aortic plaques using optical fiber bundles (Fig. 1). Two leached optical fiber bundles (OFB-A model #1119555 and OFB-B model #1119395, SCHOTT North America, MA, Elmsford, New York), and one commercially available angioscope (Vecmova, Fibertech, Japan) were tested in this study. The beam from a polarized helium-neon laser (632 nm) was expanded (5X), passed through a 50:50 beamsplitter, and focused to an approximately 50-µm-diameter spot on the luminal surface of the plaque samples. Cross-polarized laser speckle patterns were imaged (magnification=0.33) onto the distal end of the fiber bundle. For each bundle, the characteristic speckle size, determined by the resolution of the imaging system, was approximately matched to the individual fiber size. Each fiber bundle was inserted within a plastic tubing (diameter=3 mm) maintained at a 2-cm radius of curvature to mimic the curvature of the human left anterior descending (LAD) coronary artery. A computer-controlled motorized stage was used to control the motion of the optical fiber bundle (Fig. 1). The motorized stage was programmed (using a controller ESP 300, Newport) to mimic the coronary wall motion waveform over the cardiac cycle with a maximum peak-to-peak velocity of 12 mm/s perpendicular to the axis of the bundle.7 The proximal end of each bundle was imaged using an objective lens, and images were acquired using a CMOS camera (model #PL-A741, Pixelink, Ottawa, Canada). Time-varying laser speckle images of aortic plaques were obtained using each optical fiber bundle at a rate of 240 frames per second. To ensure accurate registration with histology, each imaging site was marked with two India ink spots to delineate the diameter of the imaged speckle pattern on the lesion. A set of 18 randomly selected aortic plaque samples was imaged by all three bundles under stationary conditions and during bundle motion.
Fig. 1.
Experimental setup for LSI. To obtain laser speckle images through the fiber bundles, light (632 nm) from the helium-neon source was focused on a sample, and the resulting speckle pattern was imaged on the distal end of the fiber bundle. The fiber bundle was inserted through plastic tubing wound over a 2-cm radius of curvature to mimic the human LAD curvature. The proximal end of the bundle was imaged using a CCD camera, and speckle images were obtained at a high frame rate. The fiber bundle was connected to a motorized stage that modulated the motion of the bundle (in the direction perpendicular to the page) to mimic human coronary motion over the cardiac cycle (L1, L2, L3=lenses; P1, P2=polarizers; BS =beamsplitter).
To demonstrate the efficacy of optical fiber bundle-based LSI for identifying high-risk NCFAs, we used the fiber bundle that provided the highest tolerance to motion, whose selection is described in Sec. 2.2. Using this bundle, 74 aortic plaques were imaged during both stationary and moving conditions. Following imaging, all aortic plaques were fixed in 10% formalin, then embedded and sectioned for histological processing. Sections were cut across the India ink spots and stained with hematoxylin-eosin and trichrome stains. The histological sections were interpreted by a pathologist blinded to the LSI data. Plaques were classified into the following groups1: (1) intimal hyperplasia (IH), (2) calcific ©, (3) pathological intimal thickening (PIT), and (4) NCFA. Morphometric measurements of fibrous cap thickness were obtained from the digitized histopathology slides. NCFAs with a minimum fibrous cap thickness <100 µm were further classified as high-risk NCFAs.8
2.2 Laser Speckle Analysis
Time-varying laser speckle patterns obtained using the optical fiber bundles under stationary and moving conditions were analyzed using cross-correlation techniques to determine the speckle decorrelation time constant, τ, which is inversely proportional to the rate of change of the speckle pattern.3,4 The value of the normalized 2-D cross-correlation between the first acquired speckle image and each image in the time-varying series was computed and plotted as a function of time to obtain the speckle temporal decorrelation curve for each sample. The time constant τ for each plaque was computed by single exponential fitting of the region of the speckle decor-relation curve in which the cross-correlation value dropped to 75% of the maximum4 for all bundles under stationary and moving conditions. Plaque decorrelation time constants were compared using linear regression analysis and paired t-tests for all three bundles. The average difference (error) in time constant measurements under stationary and moving conditions was measured and expressed as a percentage of the time constant measured under stationary conditions. The fiber bundle with the highest tolerance to motion was selected as having the highest correlation, lowest error, and minimal statistically significant difference in measurement of plaque time constants under stationary and moving conditions. The efficacy of this bundle for identifying high-risk NCFAs was tested during stationary conditions and bundle motion. From histological diagnoses, the time constant value associated with each lesion was assigned to one of five plaque groups for all 74 aortic plaques, and the average time constant and standard error for each group were computed. The differences between average time constant measurements for all plaque groups were compared using two-way (for plaque type and patient within each plaque group) analysis of variance (ANOVA) tests; the pair-wise comparisons between the high-risk NCFA group and other plaque groups were evaluated using the Dunnett’s t-test. In all cases, a p-value <0.05 was considered statistically significant.
3 Results
3.1 Optical Fiber Bundles for LSI
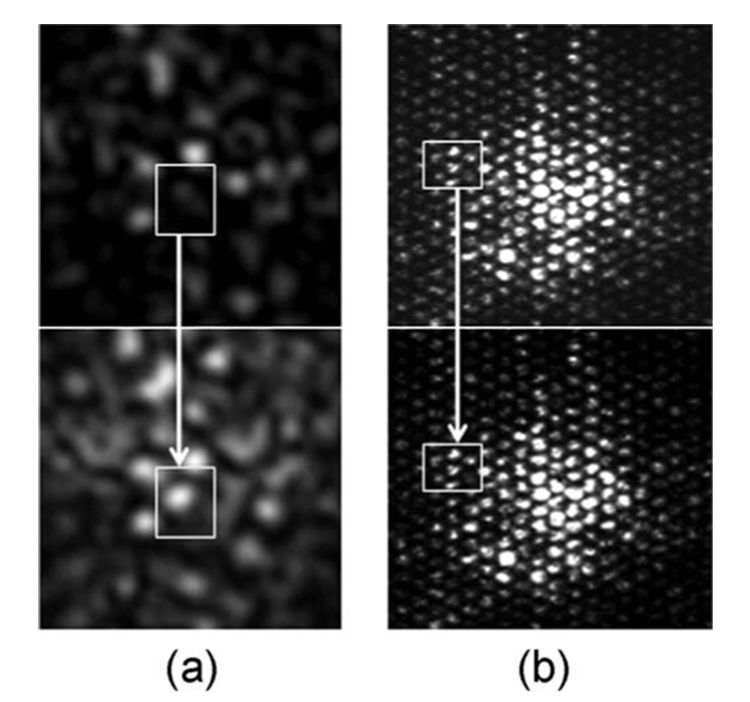
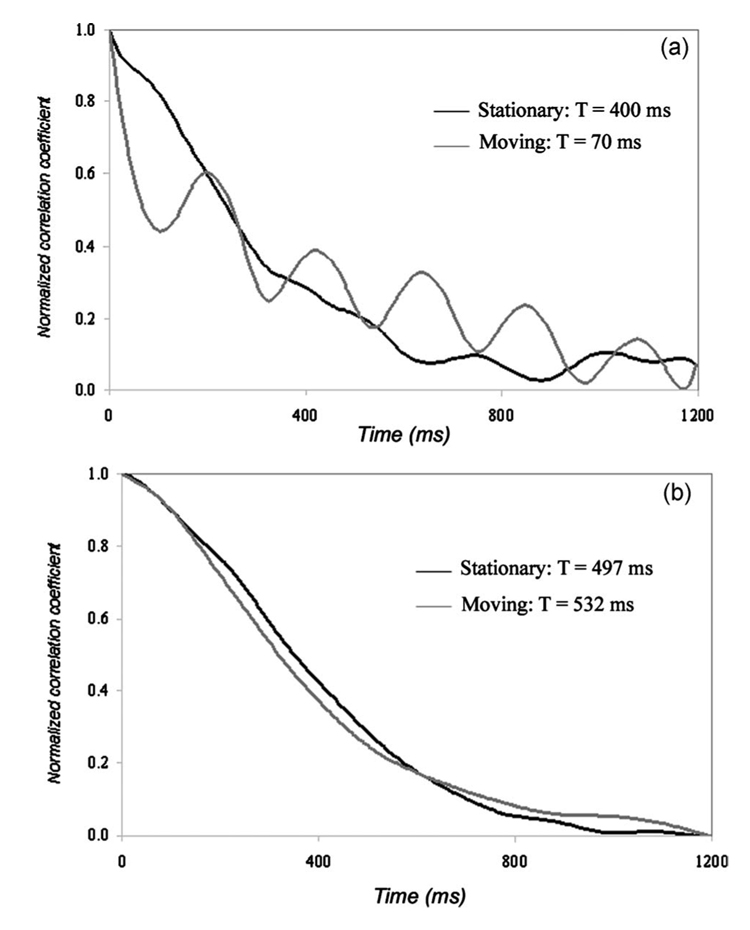
Visual inspections of the speckle pattern confirmed our expectation that bundle motion interferes with the characteristic temporal evolution of laser speckle. Figure 2 shows the laser speckle patterns of a card paper obtained using an angioscope [Fig. 2(a)] and a leached fiber bundle [Fig. 2(b)] while the flexible shafts of the bundles were moved. Although the card paper exhibited a frozen speckle pattern when the system was stationary, rapid speckle motion was observed even with the slightest motion in the angioscope, as shown in Fig. 2(a), which was obtained 100 ms after the onset of motion. On the other hand, the leached fiber bundle showed a high tolerance to bundle motion with a negligible inter-fiber effect, as shown in Fig. 2(b). This was also observed in laser speckle patterns of atherosclerotic plaques obtained during fiber bundle motion. Figure 3 shows normalized speckle decorrelation curves obtained from a fibrous plaque using the angioscope [Fig. 3(a)] and leached fiber bundle, OFB-A [Fig. 3(b)]. Normalized speckle decorrelation curves obtained using the angioscope [Fig. 3(a)] during stationary and moving conditions showed larger differences that resulted from the significant temporal modulation of the speckle pattern during motion, with an error of ~80% in measuring the plaque time constant. However, the normalized speckle decorrelation curves obtained from a leached bundle [Fig. 3(b)] showed a high correspondence during stationary and moving conditions of the fiber bundle for identical motion conditions. In this case, a negligible inter-fiber effect during motion resulted in a 7% error in measuring the plaque time constant.
Fig. 2.
Laser speckle images of card paper obtained through (a) an angioscope, and (b) a leached optical fiber bundle. Images were obtained 100 ms apart during motion of the fiber bundles. One would expect the speckle pattern from the card paper to appear frozen in time with negligible speckle decorrelation. However, as shown in (a), even the slightest motion of the angioscope caused a significant modulation of the speckle pattern resulting from inter-fiber crosstalk due to light leakage between individual fiber cores. In contrast, in the leached fiber bundle in (b), bundle motion had a negligible effect in the temporal evolution of the speckle pattern due to low crosstalk between fibers.
Fig. 3.
Normalized speckle decorrelation curves of a fibrous plaque during stationary and moving conditions using (a) an angioscope, and (b) a leached fiber bundle. In (a), the angioscope showed larger differences in speckle decorrelation curves during motion, resulting in an error of over 80% in measuring time constants. The speckle decorrelation curve shown in grey was significantly modulated by cardiac motion, as shown in the plot. In (b), the speckle decorrelation curves obtained from the leached fiber bundle measured under moving conditions corresponded well with measurements under stationary conditions. The error in measuring time constant of the fibrous plaque was 7% using the leached fiber bundle.
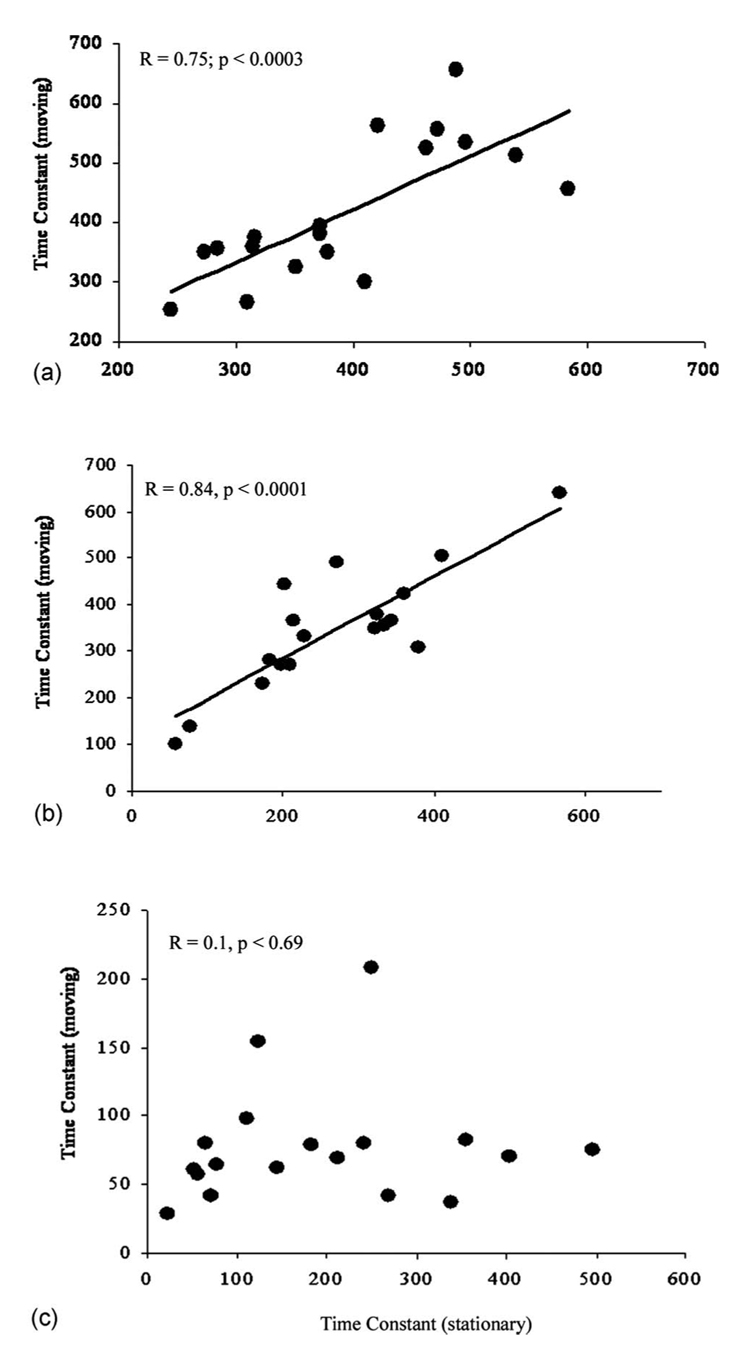
In Fig. 4, plaque time constants that were measured while the bundles were stationary are plotted against those measured during bundle motion for all three bundles, with the results of the linear regression analysis and paired t-tests presented in Table 1. For the two leached fiber bundles OFB-A and OFB-B, a good correlation was found between plaque time constants measured during stationary and moving conditions [Figs. 4(a) and 4(b)]. The leached fiber bundle OFB-A provided a high correlation between time constants (R=0.75, p <0.0003) and the lowest error (16%) in measuring plaque time constants during bundle motion compared to stationary conditions. The results of paired t-tests for this bundle showed that plaque time constant measurements during motion were not significantly different (p=0.21) from those measured by the stationary bundle (Table 1). The leached fiber bundle OFB-B also provided a high correlation between time constants (R=0.84, p<0.0001); however, the error in measuring plaque time constants during bundle motion compared to stationary conditions was 41%. For this bundle, paired t-tests showed a statistically significant difference (p<0.0003) between stationary and moving bundle conditions. Using the angioscope, decorrelation time constants measured during bundle motion showed no correlation with those measured under stationary conditions (R=0.1, p=0.69), as shown in Fig. 4(c). The angioscope provided the highest error in measuring time constants during bundle motion compared to stationary conditions (47%), and paired t-tests showed a statistically significant difference (p<0.003). Among the tested bundles, the leached fiber bundle OFB-A provided the best combination of highest correlation (R=0.75), lowest error (16%), and least significant difference (p=0.21) in measuring plaque time constants during our motion test, and was selected as having the highest tolerance to bundle motion.
Fig. 4.
Plaque time constants measured under stationary conditions plotted against those measured during bundle motion for (a) the leached fiber bundle OFB-A, (b) the leached fiber bundle OFB-B, and (c) the angioscope. The leached fiber bundles both show high correlation in time constants while the angioscope showed no correlation in measurements of time constants under stationary and moving conditions.
Table 1.
Results for comparison of the measurement of plaque time constants during stationary and moving conditions for each bundle.
| Fiber bundle | R-value (linear regression analysis) |
Error | p-value (paired t-test) |
|---|---|---|---|
| OFB-A | R=0.75; p<0.0003 | 16% | p=0.21 |
| OFB-B | R=0.84; p<0.0001 | 41% | p<0.0003 |
| Angioscope | R=0.1; p=0.69 | 47% | p<0.003 |
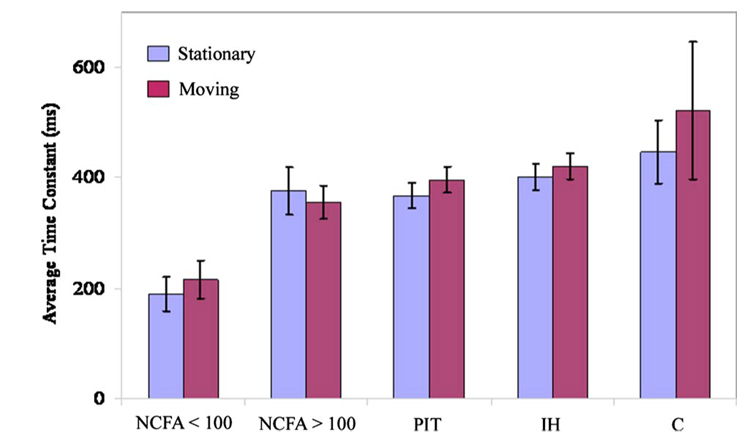
The efficacy of the leached fiber bundle OFB-A for identifying high-risk NCFAs with LSI was then evaluated using all 74 aortic specimens. The aortic specimens were histologically classified as IH (n=17), C (n=9), PIT (n=29), NCFA with cap thickness >100 µm (n=11), and NCFA with cap thickness <100 µm (n=8). The average time constants computed for the different plaque groups under both stationary and moving conditions are plotted in Fig. 5. Paired t-tests showed that for all plaque groups, differences in decorrelation time constants measured during stationary and moving conditions were not statistically significant (p=0.07). The high-risk NCFA group with fibrous cap thickness <100 µm had the lowest average time constant, and the calcific group had the highest time constant for both the stationary and moving bundle conditions. The results of the Dunnett’s t-test to compare pair-wise differences in plaque time constants between the high-risk NCFA group and other plaque groups are tabulated in Table 2 for both stationary and moving conditions of the bundle. In all cases, plaque time constants measured for the high-risk NCFA group were significantly different from each of the other groups, even during bundle motion (p <0.05), as shown in Table 2. Additionally, paired t-tests showed no statistically significant differences in plaque time constants measured for the high-risk NCFA group under stationary and moving conditions of the bundle (p=43), suggesting that high-risk NCFAs could be identified in the presence of fiber bundle motion using the leached fiber bundle OFB-A.
Fig. 5.
Average plaque time constants measured under stationary and moving conditions obtained using the leached fiber bundle (OFB-A) with a partial core size of 0.36. No statistically significant differences occurred between time constants obtained during stationary and moving conditions (paired t-tests: p=0.21). The high-risk plaque group defined as NCFAs with fibrous cap thickness <100 µm could be distinguished with highly statistically significant differences from each of the other groups in the presence of cardiac motion (p<0.05 in all cases).
Table 2.
Results of the Dunnett’s t-test comparing time constants measured for high-risk NCFA group with fibrous cap thickness <100 µm with other plaque groups under stationary and moving conditions of the leached fiber bundle. OFB-A.
| NCFA> 100 µm | PIT | IH | C | |
|---|---|---|---|---|
| Stationary | p<0.003 | p<0.0003 | p<0.0001 | p<0.003 |
| Moving | p<0.01 | p<0.001 | p<0.0003 | p<0.05 |
4 Discussion
The intricate interplay between biomechanical, compositional, and morphological factors influences plaque stability. Novel techniques that measure these factors may be invaluable for identifying rupture-prone plaques, for guiding treatment, and for investigating mechanisms associated with plaque stabilization therapies. Prior studies, conducted ex vivo, have shown that LSI measures an index of plaque viscoelasticity, which is related to plaque composition and morphology.4 While LSI shows great promise for assessing high-risk plaques, in order for it to be useful clinically, laser speckle measurements must be obtained from the coronary vasculature in vivo. Intracoronary LSI requires the development of a catheter to illuminate and image diagnostic-quality laser speckle patterns from the coronary wall. The catheter could be similar in design to an angioscope, which utilizes a small-diameter optical fiber bundle to illuminate and obtain images of the arterial wall. In this study, three leached optical fiber bundles, including a commercially available angioscope that consisted of a multicore image fiber, were tested for their efficacy in conducting LSI during stationary conditions and under conditions that simulated cardiac motion. We have found that conventional angioscopes may be incapable of conducting LSI. Commercially available angioscopes, similar to the angioscope tested in this study, are often designed to conduct white-light endoscopy and provide high-quality color images to evaluate coronary plaques in vivo. For this purpose, multicore image fiber bundles may be used in which multiple optical fiber cores are fused together in close proximity to achieve high fiber density to improve resolution. Due to the close proximity between fibers over the entire length of the image fiber, light leaks between the individual fiber cores. Unlike conventional angioscopy, laser speckle patterns formed by interference of coherent light are highly sensitive to motion. The high fiber density fused in close proximity over the length of the angioscope may contribute to light leakage (crosstalk) between individual fibers, which may be exacerbated during cardiac motion and potentially corrupt laser speckle patterns of the arterial wall. Thus, the ability to conduct LSI via an intracoronary device is contingent on the reliability of an optical guide to transmit arterial laser speckle images, especially in the presence of cardiac motion.
The leached fiber bundles evaluated in this study both showed a high correlation in plaque time constants measured under stationary conditions and under conditions that simulated cardiac motion, suggesting that these bundles may be suitable for intracoronary LSI. Leached fiber bundles are composed of multiple optical fibers, each consisting of a primary acid-resistant cladding and a secondary acid-soluble cladding that bonds the individual fiber elements. The distal and proximal ends of the bundle are protected and the bundle is immersed in an acid bath to leach away the secondary cladding, which produces a highly flexible fiber bundle. This process results in a fiber bundle that is highly flexible with larger separation between individual optical fibers along the length of the bundle. This larger inter-fiber separation over the flexible part of the bundle may result in reduced inter-fiber crosstalk, potentially allowing these bundles to be more conducive to the transmission of laser speckle images under motion conditions as compared to multicore image fibers used in angioscopes. Of the two leached fiber bundles evaluated in this study, we found that the error in measuring plaque time constants under moving conditions was significantly lower (~16%) for the fiber bundle (OFB-A) with the lowest partial-core size (core area÷individual fiber area) of 0.36 compared to the bundle OFB-B that had a partial-core size of 0.45 (error ~41%). In addition, results of the paired t-tests showed no significant difference in plaque time constants measured during stationary and moving conditions using this bundle, suggesting that the leached fiber bundle OFB-A could be suitable for use in the intracoronary LSI device. Using this fiber bundle, high-risk NCFAs could be identified from a randomly selected group of 74 aortic plaques, even in the presence of motion (p<0.05 in all cases). This study shows that the ability to conduct LSI using optical fiber bundles depends on the inter-fiber light leakage, which is related to the separation between fiber cores. A leached optical fiber bundle with a low partial-core size may elicit reduced crosstalk between fiber cores, thus allowing the reliable transmission of laser speckle patterns during motion.
While commercially available angioscopes with low pixilation artifacts are highly suitable for conducting white-light angioscopy, they may need to be optimized for LSI by incorporating low crosstalk fiber bundles. In the current study, the distal and proximal ends of the fiber bundles were kept fixed while the fiber bundle shaft was moved using a computer-controlled motorized stage to mimic coronary motion over the cardiac cycle. Under in vivo conditions, motion of the distal end of the fiber bundle relative to the coronary wall may also add an undesired speckle decorrelation that may reduce the diagnostic value of intracoronary LSI. One way to minimize this effect would be to design an intracoronary LSI device that maintains constant contact with the coronary wall during the imaging period, which is typically less than 100 ms. Similar well-established catheter designs used for intracoronary temperature measurement have been developed.9,10 Thermography studies have demonstrated the utility and safety of contact-based intra-arterial catheters in which flexible thermosensors maintain contact with the vessel wall to measure local temperature changes associated with increased inflammation, and without requiring an interruption of the coronary blood flow. Alternately, cardiac gating methods may be employed that would allow for a sufficient temporal window during the resting phase of the cardiac cycle to obtain diagnostic-quality speckle data. To obviate the influence of blood in the field of view, intracoronary saline flushing, which has been successfully implemented for optical coherence tomography and angioscopy procedures to temporarily displace blood by injecting a bolus of saline,11 can be utilized in conjunction with LSI to enable unobstructed imaging of the coronary wall.
Acknowledgments
The authors thank SCHOTT North America Inc., for providing us with the samples of leached fiber bundles evaluated in this study.
Contributor Information
Seemantini K. Nadkarni, Harvard Medical School and Wellman Center for Photomedicine, Massachusetts General Hospital, Boston, Massachusetts, 02114
Brett E. Bouma, Harvard Medical School and Wellman Center for Photomedicine, Massachusetts General Hospital, Boston, Massachusetts, 02114
Dvir Yelin, Harvard Medical School and Wellman Center for Photomedicine, Massachusetts General Hospital, Boston, Massachusetts, 02114.
Amneet Gulati, Wellman Center for Photomedicine, Massachusetts General Hospital, Boston, Massachusetts, 02114.
Guillermo J. Tearney, Harvard Medical School and Wellman Center for Photomedicine, Massachusetts General Hospital, Boston, Massachusetts, 02114
References
- 1.Virmani R, Kolodgie FD, Burke AP, et al. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler., Thromb., Vasc. Biol. 2000;20(5):1262–1275. doi: 10.1161/01.atv.20.5.1262. [DOI] [PubMed] [Google Scholar]
- 2.Schroeder AP, Falk E. Vulnerable and dangerous coronary plaques. Atherosclerosis. 1995;118 Suppl. S:141–149. [PubMed] [Google Scholar]
- 3.Tearney GJ, Bouma BE. Atherosclerotic plaque characterization by spatial and temporal speckle pattern analysis. Opt. Lett. 2002;27(7):533–535. doi: 10.1364/ol.27.000533. [DOI] [PubMed] [Google Scholar]
- 4.Nadkarni SK, Bouma BE, Helg T, et al. Characterization of atherosclerotic plaques by laser speckle imaging. Circulation. 2005;112(6):885–892. doi: 10.1161/CIRCULATIONAHA.104.520098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nadkarni SK, Bilenca A, Bouma BE, et al. Measurement of fibrous cap thickness in atherosclerotic plaques by spatiotemporal analysis of laser speckle images. J. Biomed. Opt. 2006;11(2):21006. doi: 10.1117/1.2186046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Uchida Y, Fujimori Y, Hirose J, et al. Percutaneous coronary angioscopy. Jpn. Heart J. 1992;33(3):271–294. doi: 10.1536/ihj.33.271. [DOI] [PubMed] [Google Scholar]
- 7.Johnson KR, Patel SJ, Whigham A, et al. Three-dimensional, time-resolved motion of the coronary arteries. J. Cardiovasc. Magn. Reson. 2004;6(3):663–673. doi: 10.1081/jcmr-120038086. [DOI] [PubMed] [Google Scholar]
- 8.Davies MJ. Anatomic features in victims of sudden coronary death. Coronary artery pathology. Circulation. 1992;85(1 Suppl):I19–I24. [PubMed] [Google Scholar]
- 9.Madjid M, Willerson JT, Casscells SW. Intracoronary thermography for detection of high-risk vulnerable plaques. J. Am. Coll. Cardiol. 2006;47(8 Suppl):C80–C85. doi: 10.1016/j.jacc.2005.11.050. [DOI] [PubMed] [Google Scholar]
- 10.Naghavi M, Madjid M, Gul K, et al. Thermography basket catheter: in vivo measurement of the temperature of atherosclerotic plaques for detection of vulnerable plaques. Cathet. Cardiovasc. Interv. 2003;59(1):52–59. doi: 10.1002/ccd.10486. [DOI] [PubMed] [Google Scholar]
- 11.MacNeill BD, Jang IK, Bouma BE, et al. Focal and multi-focal plaque macrophage distributions in patients with acute and stable presentations of coronary artery disease. J. Am. Coll. Cardiol. 2004;44(5):972–979. doi: 10.1016/j.jacc.2004.05.066. [DOI] [PubMed] [Google Scholar]