Summary
Tissue differentiation and signal transduction involve dramatic changes in gene expression. These changes can be brought about by the expression or activation of sequence-specific transcription factors. In order to regulate their target genes, such factors must navigate the intricate chromatin environment and engage the complex basal transcriptional machinery. We discuss three mechanisms through which signaling pathways can interact with complexes that alter chromatin structure or recruit RNA polymerase II. Signals that promote differentiation may alter the properties of such transcriptional regulatory complexes by incorporating tissue-specific subunits. Alternatively, adaptor subunits specialized to interact with specific transcription factors may allow a single complex to respond to multiple signals. Finally, individual regulatory proteins may integrate a variety of signals, allowing crosstalk between pathways.
Introduction
Developmental signals induce programs of gene expression that lead to the differentiation of a diverse array of cell types and tissues. A precise interplay between cis-acting elements and trans-acting factors allows signaling pathways to activate or repress the expression of specific genes and to maintain these expression patterns in differentiated tissues. Transcription initiation represents a key regulatory step in this process. It requires the directed assembly of a pre-initiation complex, consisting of general transcription factors (GTFs) and RNA polymerase II (pol II), on a core promoter. Sequence-specific DNA-binding proteins bound to enhancer regions assist the assembly and function of this pre-initiation complex by facilitating the recruitment of multisubunit regulatory complexes. These complexes may remodel chromatin, modify histones, or directly recruit the basal transcriptional machinery [1,2]. Recent studies have described a variety of mechanisms by which signaling pathways interact with GTFs and regulatory complexes. These interactions may also allow crosstalk between different signaling pathways at the level of target gene regulation in the nucleus.
In addition to pol II, the basal transcriptional machinery consists of a group of GTFs (TFIIA, B, D, E, F and H) that assemble on the core promoter, position pol II on the start site and trigger mRNA synthesis [3]. Recognition of the core promoter DNA sequence by TFIID is mediated by TATA-binding protein (TBP) and its associated factors (TAFs), which can also interact with sequence-specific activators [3]. Metazoans have evolved multiple TFIID-related complexes specific for particular cell types or promoter sequences, adding another level of complexity to transcriptional regulation [4]. One type of regulatory complex, the multisubunit Mediator complex, directly binds pol II and other GTFs and can recruit them to target promoters, as well as contributing to transcriptional initiation downstream of pol II recruitment [5]. The Mediator complex is brought to the promoter region by interactions with sequence-specific transcription factors through a variety of adaptor subunits [6].
Eukaryotic DNA is packaged into nucleosomes and higher-order chromatin structures that have a repressive effect on transcription. Heterochromatin assembly can maintain genes in a stably repressed state, but even in euchromatic regions nucleosomes may interfere with DNA sequence recognition by transcription factors and with the binding or progression of pol II. Two classes of transcriptional regulatory complexes act by changing chromatin structure. ATP-dependent chromatin remodeling complexes alter the pattern of contacts between DNA and histones to expose DNA sequences that would otherwise be occluded by nucleosomes. This can be accomplished by nucleosome sliding, DNA bulging, nucleosome eviction, or changing the structure of the nucleosome by incorporation of histone variants [7]. The core ATPase subunit of each chromatin remodeling complex determines its mechanism of action [8], while a variety of accessory subunits have functions that are less well understood.
Histone-modifying complexes alter chromatin structure by covalently modifying specific amino acids in the histone tails. These modifications may directly influence chromatin compaction, or may create docking sites for specific proteins that include chromatin remodeling proteins, GTFs, heterochromatin proteins, and other factors [9]. Acetylation of lysines in the histone tails by histone acetyltransferases (HATs) contributes to transcriptional activation, while methylation of lysines or arginines or ubiquitylation of lysines can create marks for either transcriptional activation or repression, depending on the specific residue modified [9]. For instance, the Polycomb group proteins, which maintain a silenced chromatin state on critical developmental genes, include a histone methyltransferase for lysine 27 of histone H3 as well as a complex (Polycomb Repression Complex 1, PRC1) that recognizes this specific modification [10]. Interestingly, some enzymes initially characterized for their effects on histones can also modify sequence-specific transcription factors, altering their activities and providing a mechanism for feedback [11,12]. Recent studies have begun to elucidate how intercellular signals use this multitude of transcriptional regulatory complexes to establish transcriptional programs during development.
Differentiation-dependent switches in transcriptional regulatory complexes
One mechanism by which signals that lead to cell differentiation can regulate the transcription of multiple genes in a coordinated manner is by altering the cellular transcriptional machinery. A striking example of this strategy is the Drosophila testis, in which multiple tissue-specific TAF homologues as well as the TBP-related factor TRF2 are required for the transcription of genes involved in spermatid differentiation [13,14]. These TAFs appear to counteract the repressive effect of the Polycomb complex PRC1 on certain spermatid-specific genes. They do this both by sequestering PRC1 components in the nucleolus, and by recruiting Trithorax, which promotes transcriptional activation by methylating lysine 4 of histone H3 [15]. In mammals, TAF7L replaces TAF7 and TRF2 replaces TBP late in spermatogenesis [16–18], and TAF4b is required for normal germ cell development in both the testis and ovary [19,20]. The role of TAF4b in ovarian granulosa cells appears to be partially mediated by induction of the transcription factor c-Jun [21]. Interestingly, incorporation of TAF4b enhances the ability of the TFIID complex to recognize the core promoter of c-jun and other ovary-specific genes, and alters its conformation so as to increase its accessibility to activators [22].
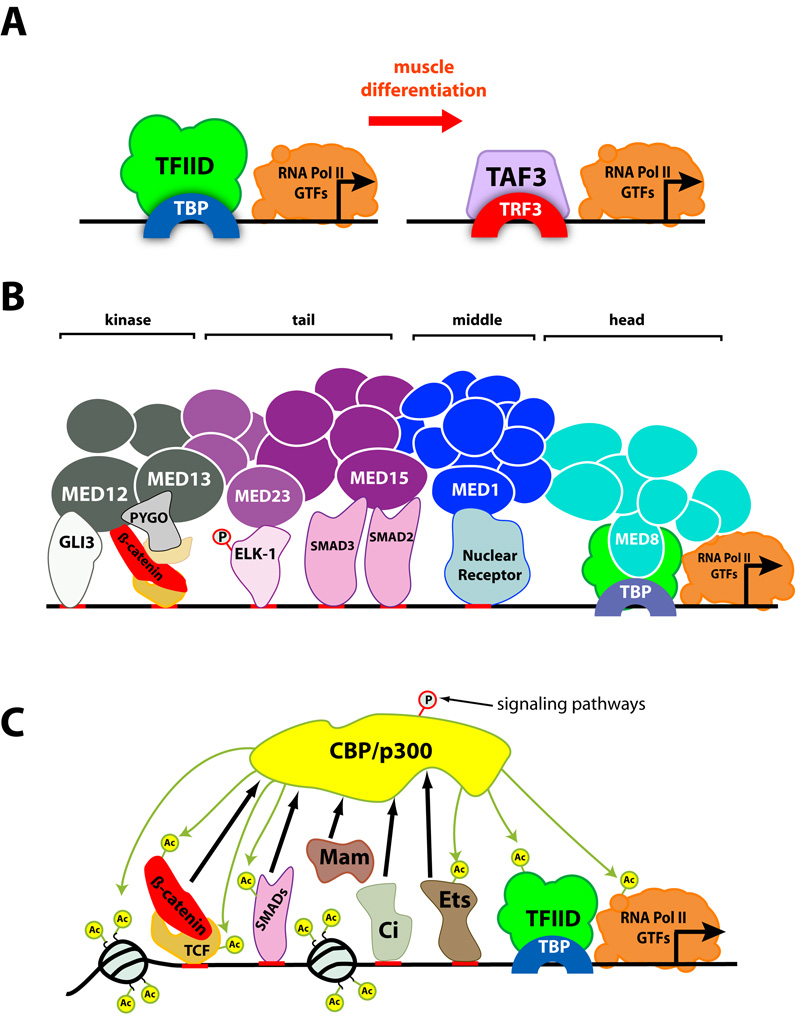
An even more dramatic transformation of the core promoter complex occurs when mouse myoblasts differentiate into myotubes: TFIID disappears and is replaced by a smaller complex containing only TAF3 and the TBP homologue TRF3 [23] (Fig. 1A). This substitution may prevent the transcription of genes expressed in undifferentiated cells, as well as allowing the transcription of muscle-specific genes such as myogenin. It is not yet clear how transcription of housekeeping genes is maintained following the loss of TFIID, nor how the noncoding RNAs transcribed by pol I and pol III are affected. The replacement seems to occur at the transcriptional level and is presumably controlled by myogenic transcription factors, although the precise mechanism has not been determined. TRF3 is also essential for haematopoiesis in zebrafish, suggesting that it may substitute for TBP in additional cell lineages [24].
Figure 1. Mechanisms for interaction between signaling pathways and the general transcriptional machinery.
(A) Developmental signals that trigger differentiation can result in the replacement of a core transcriptional complex. When myoblasts differentiate into myotubes, TFIID is replaced by a smaller complex containing TAF3 and the TBP homologue TRF3. This complex recognizes the promoters of muscle-specific genes. (B) Transcriptional complexes can contain subunits specialized to act as adaptors for signal-regulated transcription factors. The Mediator complex interacts with pol II and GTFs through its head module (turquoise), and with multiple DNA-binding proteins and coactivators through individual adaptor subunits in its middle (blue), tail (purple) and kinase (gray) modules. Only transcription factors regulated by signaling pathways are shown; they are represented on a single enhancer region for convenience, but would in fact recruit the complex to different target genes. (C) Some transcriptional regulators can integrate multiple inputs, allowing crosstalk between signaling pathways. The histone acetyltransferase CBP is recruited (black arrows) by a large number of signal-regulated and other transcription factors, again represented on a single hypothetical enhancer. Some of these factors are themselves acetylated by CBP (green arrows). The activity and binding preferences of CBP itself can also be regulated by post-translational modifications controlled by signaling pathways.
An analogous switch in a chromatin remodeling complex occurs during neuronal differentiation in the mouse spinal cord. Two subunits of the SWI/SNF-related chromatin remodeling complex present in neural progenitor cells, BAF45a and BAF53a, are replaced in differentiating neurons by the BAF45b, BAF45c, and BAF53b isoforms [25]. This switch is functionally important, since BAF53b has an essential role in activity-dependent dendritic outgrowth that cannot be substituted by BAF53a [26]. BAF53b appears to target the transcription factor Calcium-responsive transactivator (CREST) to the promoters of genes involved in dendritic development [26]. In contrast, overexpression of BAF45a increases the number of proliferating neural progenitor cells and interferes with neuronal differentiation [25]. Additional mechanisms for wholesale transformation of transcriptomes through changes in the transcription machinery may come to light with further analysis of the transcriptional regulatory complexes that are present in differentiated cell types in vivo, rather than in a small number of tissue culture cell lines.
Transcriptional complexes contain subunits dedicated to receiving specific signals
Such dramatic alterations of the transcriptional machinery would be unsuitable for cells that must remain poised to respond to a variety of possible signals by transcribing the appropriate target genes. An alternative strategy requires multiple forms of a transcriptional regulatory complex to coexist in the same cells, where they can respond to different factors. A single complex may also contain multiple subunits specialized to act as adaptors or transducers for specific transcription factors or developmental signals.
The SWI/SNF chromatin remodeling complex is required for the transcription of many genes through interactions with a variety of transcription factors. In addition to the subunit isoforms specific to neuronal differentiation described above, two distinct forms of the complex are defined by mutually exclusive but unrelated accessory subunits. In addition to a common set of core subunits, the BAF complex contains the BAF250 subunit, while PBAF instead contains BAF180 and BAF200 [27]. BAF and PBAF coexist in the same cells, but have distinct developmental functions. Two mouse BAF250 isoforms are required for embryonic stem cell maintenance and mesoderm formation, while BAF180 affects the development of the heart and placenta [28–30]. In Drosophila, the BAF250 orthologue Osa has important roles early in development, including repressing target genes of Wingless (Wg), a Wnt family signaling protein [31]. In contrast, the fly homologues of BAF180 and BAF200 are required only at late stages of development [32](Carrera et al., submitted). These phenotypic differences may reflect recruitment by different sequence-specific transcription factors. BAF250 homologues have been shown to interact directly with the glucocorticoid receptor, Zeste, the GATA transcription factor Pannier, its coactivator Chip, and acidic activators [33–37], while human BAF200 can bind to Serum response factor [38] and BAF180 regulates genes that are targets of the retinoic acid receptor [30]. A third subunit, Supporter of Activation of Yellow Protein (SAYP), specific to the Drosophila PBAF complex may interact with additional transcription factors [32]. However, if BAF250, BAF200 and BAF180 were only required to recruit the SWI/SNF complex to a variety of promoters, it is unclear why their presence should be mutually exclusive. Perhaps these accessory subunits also confer distinct chromatin remodeling properties on the BAF and PBAF complexes.
Several other examples of interactions between subunits of transcriptional regulatory complexes and specific transcription factors downstream of signaling pathways have been described. The Nucleosome Remodeling Factor (NURF) chromatin remodeling complex interacts with the ecdysone receptor and with Ken & Barbie, an inhibitor of JAK/STAT target genes, and requires the NURF301 subunit to regulate targets of these transcription factors [39,40]. Target genes of the Notch signaling pathway are repressed in the absence of Notch signaling by the histone chaperone Anti-silencing function 1 (Asf1), which is recruited through interactions with the Notch-specific corepressor Hairless [41]. Wnt target genes are repressed by a remodeling complex that is recruited at least in part through interactions of the chromodomain protein CHD8/Kismet with β-catenin; this mechanism may limit the maximal activation of these genes by Wnt signaling [42].
The Mediator complex, which is broadly required for both basal and activated transcription by pol II, provides a striking example of a general complex containing multiple specialized subunits. This complex can be divided into head, middle and tail modules based on electron microscopic visualization and biochemical dissociation studies, and large and small forms of the complex are distinguished by the presence or absence of an additional kinase module consisting of four subunits [43]. The head module is required for interactions with pol II and the general transcription factor TFIIF [44], while the tail module interacts with sequence-specific activators [45]. The middle module contributes to both functions, in part by controlling the conformation of the entire complex [46]. A number of Mediator complex subunits act as adaptors for specific transcription factors regulated by signaling pathways (Fig. 1B). MED1 interacts with ligand-bound nuclear receptors [47]; MED15 interacts with Smad2 and Smad3, which transduce TGF-β family signals [48]; and MED23 interacts with Elk-1 that has been phosphorylated by MAP kinase [49]. MED12 and MED13, subunits of the accessory kinase module, interact with the coactivator Pygopus (Pygo) and with β-catenin to turn on Wingless target genes [50,51], as well as with Gli3, a transcription factor controlled by Sonic hedgehog signaling [52]. Numerous other transcription factors have been shown to interact with the subunits mentioned above and with additional subunits [6]. The Mediator complex thus seems to consist of a core functional unit and a collection of adaptor subunits each able to interact with multiple transcription factors. This organization may be necessary to enable its recruitment to essentially all pol II target genes.
There are intriguing indications that the Mediator complex may itself be functionally regulated by certain signaling pathways. The MED1 subunit is phosphorylated by activated ERK, enhancing its association with the Mediator complex [53,54]. This phosphorylation can be stimulated by the thyroid and steroid hormones that use MED1 as an adaptor subunit [54]. In yeast, phosphorylation of SRB9/MED13 by PKA enhances its ability to repress the expression of genes induced by nutrient deprivation [55]. Two different transcriptional activators, VP16 and Sterol regulatory element-binding protein (SREBP), have been shown to induce different conformational changes when bound to the Mediator complex [56], suggesting that binding of one activator could facilitate or inhibit interactions of the complex with another. The Mediator complex can also feed back on transcription factors; for instance, the subunit Cdk8 phosphorylates the intracellular domain of Notch, promoting its degradation [57]. These findings suggest that the Mediator complex is not simply a passive array of adaptors, but may play an active role in signal-regulated transcription.
A single protein can integrate a variety of signaling information
The complexes discussed above contain subunits specialized to receive information from particular signaling pathways. In contrast, other transcriptional regulators appear to be capable of integrating a broad array of signaling information through a single protein. The histone acetyltransferases (HATs) CREB Binding Protein (CBP) and p300 provide one such example. These molecules are recruited by β-catenin to activate Wnt-mediated transcription, by Smads to activate TGF-β-mediated transcription, by Mastermind (Mam) to activate Notch-mediated transcription, by Cubitus interruptus (Ci) to activate Hedgehog-mediated transcription, by Stats to activate cytokine-mediated transcription, and by Ets proteins to activate MAPK-mediated transcription, as well as by many other transcription factors [58–63] (Fig. 1C). At least some of these interactions seem to be direct, suggesting that CBP has integrated multiple adaptor domains into a single molecule. Several other HAT complexes, including STAGA, TFTC and Tip60, are recruited to transcriptional activators through the TRRAP subunit, a very large protein containing multiple sites for protein-protein interactions [64]. Such a mechanism may promote cooperative recruitment of HAT activity by different transcription factors bound to the same enhancer region.
Interestingly, CBP acetylates not only histone substrates, but many transcription factors as well. Such acetylation can either enhance or inhibit their transcriptional activity by altering nuclear localization, protein stability, DNA binding, or interaction with coactivators [65]. For instance, CBP/p300 is recruited to the promoters of Wnt target genes by interactions with β-catenin and the DNA-binding protein TCF. In addition to acetylating histones, it acetylates β-catenin, reducing its ability to activate myc expression and its affinity for TCF, and TCF, reducing its affinity for β-catenin [66–69]. Conversely, intercellular signaling can lead to posttranslational modifications of CBP itself, regulating its activity [11] and thus indirectly influencing other signaling pathways. An example is phosphorylation of CBP by IKKα, which promotes CBP binding to the transcription factor NF-κB rather than to p53, leading to tumor cell proliferation rather than apoptosis [70]. These bidirectional interactions between CBP and signaling pathways allow it to integrate multiple inputs and translate them into changes in gene expression.
Conclusions
A diverse set of mechanisms allow signal-regulated transcription factors to recruit the general transcriptional machinery and alter target gene expression. Complexes that alter chromatin structure or recruit RNA polymerase may interact with transcription factors through large multifunctional subunits or individual specialized subunits. In many cells, different forms of the same complex can be recruited to different promoters, and may also have distinct functional properties. Subunits of regulatory complexes can themselves be the targets of regulation by signaling pathways. A special case of this is the replacement of entire transcriptional regulatory complexes in specific developing tissues, but complexes can also be altered by post-translational modifications or conformational changes induced by transcription factor binding. Such effects provide a mechanism through which signaling pathways can influence each other in the nucleus by indirectly altering the potential to regulate target gene expression.
Acknowledgements
The manuscript was improved by the critical comments of Sergio Astigarraga, Kerstin Hofmeyer, Kevin Legent, Jean-Yves Roignant, Josie Steinhauer, and Naoko Tanese. Work in this area in the authors’ laboratory is supported by the National Institutes of Health (grant GM56131) and the Irma T. Hirschl/Monique Weill-Caulier Trust.
Glossary
- BAF
BRG1- or hBRM Associated Factors. A chromatin remodeling complex of the SWI/SNF family, containing the BAF250 subunit.
- CBP
(cAMP-Response Element-Binding Protein, CREB) Binding Protein. A commonly used histone acetyltransferase.
- CHD8
CHromoDomain protein 8. An ATP-dependent chromatin remodeling protein.
- GTFs
General Transcription Factors. A term used to describe TFIIA, TFIIB, TFIIC, TFIID, TFIIE, TFIIF, and TFIIH.
- HAT
Histone Acetyl Transferase. A complex that acetylates lysines in histone tails, promoting transcriptional activation.
- NURF
Nucleosome Remodeling Factor. A chromatin remodeling complex that uses the ISWI ATPase subunit.
- PBAF
Polybromo-Associated BAF. A complex identical to BAF except that it contains BAF180 and BAF200 instead of BAF250.
- pol II
RNA polymerase II. The primary RNA polymerase for protein-coding mRNAs. Pol I transcribes primarily ribosomal RNA, and pol III transfer RNA and other non-coding RNAs.
- STAGA
Spt3-TAF9-GCN5L Acetylase. A histone acetyltransferase complex.
- SWI/SNF
SWItching defective and Sucrose-Non Fermenting. A chromatin remodeling complex first identified in yeast through these two phenotypes.
- TAFs
TBP-Associated Factors. A set of proteins that associate with TBP to constitute TFIID.
- TBP
TATA-binding Protein. The protein within TFIID that recognizes the TATA box within the core promoter.
- TFTC
TBP-free TAFII-containing complex. A histone acetyltransferase complex.
- Tip60
Tat Interactive Protein (60kD). A histone acetyltransferase complex.
- TRF3
TBP-related Factor 3. A protein that can substitute for TBP to recognize a different set of core promoters.
- TRRAP
TRansformation-tRansactivation domain-associated protein. A large subunit of several histone acetyltransferase complexes that interacts with activators.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
* of special interest
** of outstanding interest
- 1.Li B, Carey M, Workman JL. The role of chromatin during transcription. Cell. 2007;128:707–719. doi: 10.1016/j.cell.2007.01.015. [DOI] [PubMed] [Google Scholar]
- 2.Naar AM, Lemon BD, Tjian R. Transcriptional coactivator complexes. Annu Rev Biochem. 2001;70:475–501. doi: 10.1146/annurev.biochem.70.1.475. [DOI] [PubMed] [Google Scholar]
- 3.Thomas MC, Chiang CM. The general transcription machinery and general cofactors. Crit Rev Biochem Mol Biol. 2006;41:105–178. doi: 10.1080/10409230600648736. [DOI] [PubMed] [Google Scholar]
- 4.Reina JH, Hernandez N. On a roll for new TRF targets. Genes Dev. 2007;21:2855–2860. doi: 10.1101/gad.1623207. [DOI] [PubMed] [Google Scholar]
- 5.Malik S, Roeder RG. Dynamic regulation of pol II transcription by the mammalian Mediator complex. Trends Biochem Sci. 2005;30:256–263. doi: 10.1016/j.tibs.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 6.Kim YJ, Lis JT. Interactions between subunits of Drosophila Mediator and activator proteins. Trends Biochem Sci. 2005;30:245–249. doi: 10.1016/j.tibs.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 7.Saha A, Wittmeyer J, Cairns BR. Chromatin remodelling: the industrial revolution of DNA around histones. Nat Rev Mol Cell Biol. 2006;7:437–447. doi: 10.1038/nrm1945. [DOI] [PubMed] [Google Scholar]
- 8.Fan HY, Trotter KW, Archer TK, Kingston RE. Swapping function of two chromatin remodeling complexes. Mol Cell. 2005;17:805–815. doi: 10.1016/j.molcel.2005.02.024. [DOI] [PubMed] [Google Scholar]
- 9.Kouzarides T. Chromatin modifications and their function. Cell. 2007;128:693–705. doi: 10.1016/j.cell.2007.02.005. [DOI] [PubMed] [Google Scholar]
- 10.Muller J, Kassis JA. Polycomb response elements and targeting of Polycomb group proteins in Drosophila. Curr Opin Genet Dev. 2006;16:476–484. doi: 10.1016/j.gde.2006.08.005. [DOI] [PubMed] [Google Scholar]
- 11.Kalkhoven E. CBP and p300: HATs for different occasions. Biochem Pharmacol. 2004;68:1145–1155. doi: 10.1016/j.bcp.2004.03.045. [DOI] [PubMed] [Google Scholar]
- 12.Huang J, Berger SL. The emerging field of dynamic lysine methylation of non-histone proteins. Curr Opin Genet Dev. 2008 doi: 10.1016/j.gde.2008.01.012. [DOI] [PubMed] [Google Scholar]
- 13.Hiller M, Chen X, Pringle MJ, Suchorolski M, Sancak Y, Viswanathan S, Bolival B, Lin TY, Marino S, Fuller MT. Testis-specific TAF homologs collaborate to control a tissue-specific transcription program. Development. 2004;131:5297–5308. doi: 10.1242/dev.01314. [DOI] [PubMed] [Google Scholar]
- 14.Kopytova DV, Krasnov AN, Kopantceva MR, Nabirochkina EN, Nikolenko JV, Maksimenko O, Kurshakova MM, Lebedeva LA, Yerokhin MM, Simonova OB, et al. Two isoforms of Drosophila TRF2 are involved in embryonic development, premeiotic chromatin condensation, and proper differentiation of germ cells of both sexes. Mol Cell Biol. 2006;26:7492–7505. doi: 10.1128/MCB.00349-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Chen X, Hiller M, Sancak Y, Fuller MT. Tissue-specific TAFs counteract Polycomb to turn on terminal differentiation. Science. 2005;310:869–872. doi: 10.1126/science.1118101.. This paper shows that TAFs specific to the Drosophila testis promote activation of genes important for spermatid differentiation by relocalizing PRC1 components to the nucleolus and by promoting lysine methylation of H3K4.
- 16.Pointud JC, Mengus G, Brancorsini S, Monaco L, Parvinen M, Sassone-Corsi P, Davidson I. The intracellular localisation of TAF7L, a paralogue of transcription factor TFIID subunit TAF7, is developmentally regulated during male germ-cell differentiation. J Cell Sci. 2003;116:1847–1858. doi: 10.1242/jcs.00391. [DOI] [PubMed] [Google Scholar]
- 17.Zhang D, Penttila TL, Morris PL, Roeder RG. Cell- and stage-specific high-level expression of TBP-related factor 2 (TRF2) during mouse spermatogenesis. Mech Dev. 2001;106:203–205. doi: 10.1016/s0925-4773(01)00439-7. [DOI] [PubMed] [Google Scholar]
- 18.Martianov I, Brancorsini S, Gansmuller A, Parvinen M, Davidson I, Sassone-Corsi P. Distinct functions of TBP and TLF/TRF2 during spermatogenesis: requirement of TLF for heterochromatic chromocenter formation in haploid round spermatids. Development. 2002;129:945–955. doi: 10.1242/dev.129.4.945. [DOI] [PubMed] [Google Scholar]
- 19.Freiman RN, Albright SR, Zheng S, Sha WC, Hammer RE, Tjian R. Requirement of tissue-selective TBP-associated factor TAFII105 in ovarian development. Science. 2001;293:2084–2087. doi: 10.1126/science.1061935. [DOI] [PubMed] [Google Scholar]
- 20.Falender AE, Freiman RN, Geles KG, Lo KC, Hwang K, Lamb DJ, Morris PL, Tjian R, Richards JS. Maintenance of spermatogenesis requires TAF4b, a gonad-specific subunit of TFIID. Genes Dev. 2005;19:794–803. doi: 10.1101/gad.1290105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Geles KG, Freiman RN, Liu WL, Zheng S, Voronina E, Tjian R. Cell-type-selective induction of c-jun by TAF4b directs ovarian-specific transcription networks. Proc Natl Acad Sci U S A. 2006;103:2594–2599. doi: 10.1073/pnas.0510764103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Liu WL, Coleman RA, Grob P, King DS, Florens L, Washburn MP, Geles KG, Yang JL, Ramey V, Nogales E, et al. Structural changes in TAF4b-TFIID correlate with promoter selectivity. Mol Cell. 2008;29:81–91. doi: 10.1016/j.molcel.2007.11.003.. This paper shows that incorporation of TAF4b into the TFIID complex alters both its structure and its core promoter specificity, increasing activator binding to the target c-jun promoter.
- 23. Deato MD, Tjian R. Switching of the core transcription machinery during myogenesis. Genes Dev. 2007;21:2137–2149. doi: 10.1101/gad.1583407.. The authors demonstrate a dramatic transformation of the basal transcriptional machinery when myoblasts differentiate into myotubes. TFIID disappears and is replaced by TRF3 and TAF3, which are required for the transcription of muscle-specific genes such as myogenin.
- 24.Hart DO, Raha T, Lawson ND, Green MR. Initiation of zebrafish haematopoiesis by the TATA-box-binding protein-related factor Trf3. Nature. 2007;450:1082–1085. doi: 10.1038/nature06349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lessard J, Wu JI, Ranish JA, Wan M, Winslow MM, Staahl BT, Wu H, Aebersold R, Graef IA, Crabtree GR. An essential switch in subunit composition of a chromatin remodeling complex during neural development. Neuron. 2007;55:201–215. doi: 10.1016/j.neuron.2007.06.019.. The authors identify subunits of the SWI/SNF chromatin remodeling complex that undergo isoform switching during neuronal differentiation in the mouse spinal cord. One progenitor-specific subunit isoform, BAF45a, is necessary and sufficient for continued cell proliferation.
- 26.Wu JI, Lessard J, Olave IA, Qiu Z, Ghosh A, Graef IA, Crabtree GR. Regulation of dendritic development by neuron-specific chromatin remodeling complexes. Neuron. 2007;56:94–108. doi: 10.1016/j.neuron.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 27.Mohrmann L, Verrijzer CP. Composition and functional specificity of SWI2/SNF2 class chromatin remodeling complexes. Biochim Biophys Acta. 2005;1681:59–73. doi: 10.1016/j.bbaexp.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 28.Gao X, Tate P, Hu P, Tjian R, Skarnes WC, Wang Z. ES cell pluripotency and germ-layer formation require the SWI/SNF chromatin remodeling component BAF250a. Proc Natl Acad Sci U S A. 2008;105:6656–6661. doi: 10.1073/pnas.0801802105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yan Z, Wang Z, Sharova L, Sharov AA, Ling C, Piao Y, Aiba K, Matoba R, Wang W, Ko MS. BAF250B-associated SWI/SNF Chromatin-remodeling Complex is Required to Maintain Undifferentiated Mouse Embryonic Stem Cells. Stem Cells. 2008 doi: 10.1634/stemcells.2007-0846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Z, Zhai W, Richardson JA, Olson EN, Meneses JJ, Firpo MT, Kang C, Skarnes WC, Tjian R. Polybromo protein BAF180 functions in mammalian cardiac chamber maturation. Genes Dev. 2004;18:3106–3116. doi: 10.1101/gad.1238104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Collins RT, Treisman JE. Osa-containing Brahma chromatin remodeling complexes are required for the repression of Wingless target genes. Genes Dev. 2000;14:3140–3152. doi: 10.1101/gad.854300.. This paper shows that Osa, a subunit specific to one form of the Brahma chromatin remodeling complex, has a specific developmental function, repressing target genes regulated by the Wnt protein Wingless.
- 32.Chalkley GE, Moshkin YM, Langenberg K, Bezstarosti K, Blastyak A, Gyurkovics H, Demmers JA, Verrijzer CP. The transcriptional coactivator SAYP is a trithorax group signature subunit of the PBAP chromatin remodeling complex. Mol Cell Biol. 2008;28:2920–2929. doi: 10.1128/MCB.02217-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Inoue H, Furukawa T, Giannakopoulos S, Zhou S, King DS, Tanese N. Largest subunits of the human SWI/SNF chromatin-remodeling complex promote transcriptional activation by steroid hormone receptors. J Biol Chem. 2002;277:41674–41685. doi: 10.1074/jbc.M205961200. [DOI] [PubMed] [Google Scholar]
- 34.Nie Z, Xue Y, Yang D, Zhou S, Deroo BJ, Archer TK, Wang W. A specificity and targeting subunit of a human SWI/SNF family-related chromatin-remodeling complex. Molecular & Cellular Biology. 2000;20:8879–8888. doi: 10.1128/mcb.20.23.8879-8888.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kal AJ, Mahmoudi T, Zak NB, Verrijzer CP. The Drosophila brahma complex is an essential coactivator for the trithorax group protein zeste. Genes Dev. 2000;14:1058–1071. [PMC free article] [PubMed] [Google Scholar]
- 36.Heitzler P, Vanolst L, Biryukova I, Ramain P. Enhancer-promoter communication mediated by Chip during Pannier-driven proneural patterning is regulated by Osa. Genes Dev. 2003;17:591–596. doi: 10.1101/gad.255703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Neely KE, Hassan AH, Brown CE, Howe L, Workman JL. Transcription activator interactions with multiple SWI/SNF subunits. Mol Cell Biol. 2002;22:1615–1625. doi: 10.1128/MCB.22.6.1615-1625.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhang X, Azhar G, Zhong Y, Wei JY. Zipzap/p200 is a novel zinc finger protein contributing to cardiac gene regulation. Biochem Biophys Res Commun. 2006;346:794–801. doi: 10.1016/j.bbrc.2006.05.211. [DOI] [PubMed] [Google Scholar]
- 39. Badenhorst P, Xiao H, Cherbas L, Kwon SY, Voas M, Rebay I, Cherbas P, Wu C. The Drosophila nucleosome remodeling factor NURF is required for Ecdysteroid signaling and metamorphosis. Genes Dev. 2005;19:2540–2545. doi: 10.1101/gad.1342605.. This paper shows that a mutation in the NURF subunit NURF301 prevents pupariation and specifically disrupts the expression of target genes of the ecdysone receptor. The NURF complex also physically interacts with the receptor in a ligand-dependent manner.
- 40.Kwon SY, Xiao H, Glover BP, Tjian R, Wu C, Badenhorst P. The nucleosome remodeling factor (NURF) regulates genes involved in Drosophila innate immunity. Dev Biol. 2008;316:538–547. doi: 10.1016/j.ydbio.2008.01.033. [DOI] [PubMed] [Google Scholar]
- 41.Goodfellow H, Krejci A, Moshkin Y, Verrijzer CP, Karch F, Bray SJ. Gene-specific targeting of the histone chaperone asf1 to mediate silencing. Dev Cell. 2007;13:593–600. doi: 10.1016/j.devcel.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 42.Thompson BA, Tremblay V, Lin G, Bochar DA. CHD8 is an ATP-dependent Chromatin Remodeling Factor That Regulates {beta}-catenin Target Genes. Mol Cell Biol. 2008 doi: 10.1128/MCB.00322-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Boube M, Joulia L, Cribbs DL, Bourbon HM. Evidence for a mediator of RNA polymerase II transcriptional regulation conserved from yeast to man. Cell. 2002;110:143–151. doi: 10.1016/s0092-8674(02)00830-9. [DOI] [PubMed] [Google Scholar]
- 44.Takagi Y, Calero G, Komori H, Brown JA, Ehrensberger AH, Hudmon A, Asturias F, Kornberg RD. Head module control of mediator interactions. Mol Cell. 2006;23:355–364. doi: 10.1016/j.molcel.2006.06.007. [DOI] [PubMed] [Google Scholar]
- 45.Kang JS, Kim SH, Hwang MS, Han SJ, Lee YC, Kim YJ. The structural and functional organization of the yeast mediator complex. J Biol Chem. 2001;276:42003–42010. doi: 10.1074/jbc.M105961200. [DOI] [PubMed] [Google Scholar]
- 46.Baidoobonso SM, Guidi BW, Myers LC. Med19(Rox3) regulates Intermodule interactions in the Saccharomyces cerevisiae mediator complex. J Biol Chem. 2007;282:5551–5559. doi: 10.1074/jbc.M609484200. [DOI] [PubMed] [Google Scholar]
- 47.Belakavadi M, Fondell JD. Role of the mediator complex in nuclear hormone receptor signaling. Rev Physiol Biochem Pharmacol. 2006;156:23–43. doi: 10.1007/s10254-005-0002-0. [DOI] [PubMed] [Google Scholar]
- 48.Kato Y, Habas R, Katsuyama Y, Naar AM, He X. A component of the ARC/Mediator complex required for TGF beta/Nodal signalling. Nature. 2002;418:641–646. doi: 10.1038/nature00969. [DOI] [PubMed] [Google Scholar]
- 49. Boyer TG, Martin ME, Lees E, Ricciardi RP, Berk AJ. Mammalian Srb/Mediator complex is targeted by adenovirus E1A protein. Nature. 1999;399:276–279. doi: 10.1038/20466.. This was one of the first papers to show that a specific Mediator subunit is involved in signal transduction by binding to the Elk-1 transcription factor once it has been phosphorylated by the ERK kinase.
- 50.Carrera I, Janody F, Leeds N, Duveau F, Treisman JE. Pygopus activates Wingless target gene transcription through the mediator complex subunits Med12 and Med13. Proc Natl Acad Sci U S A. 2008;105:6644–6649. doi: 10.1073/pnas.0709749105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kim S, Xu X, Hecht A, Boyer TG. Mediator is a transducer of Wnt/beta-catenin signaling. J Biol Chem. 2006;281:14066–14075. doi: 10.1074/jbc.M602696200. [DOI] [PubMed] [Google Scholar]
- 52.Zhou H, Kim S, Ishii S, Boyer TG. Mediator modulates Gli3-dependent Sonic hedgehog signaling. Mol Cell Biol. 2006;26:8667–8682. doi: 10.1128/MCB.00443-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Pandey PK, Udayakumar TS, Lin X, Sharma D, Shapiro PS, Fondell JD. Activation of TRAP/mediator subunit TRAP220/Med1 is regulated by mitogen-activated protein kinase-dependent phosphorylation. Mol Cell Biol. 2005;25:10695–10710. doi: 10.1128/MCB.25.24.10695-10710.2005.. This paper shows that phosphorylation of the Mediator complex subunit MED1 by MAP kinase regulates its levels, subnuclear localization and coactivation strength.
- 54.Belakavadi M, Pandey PK, Vijayvargia R, Fondell JD. MED1 phosphorylation promotes its association with Mediator: implications for nuclear receptor signaling. Mol Cell Biol. 2008 doi: 10.1128/MCB.02191-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chang YW, Howard SC, Herman PK. The Ras/PKA signaling pathway directly targets the Srb9 protein, a component of the general RNA polymerase II transcription apparatus. Mol Cell. 2004;15:107–116. doi: 10.1016/j.molcel.2004.05.021. [DOI] [PubMed] [Google Scholar]
- 56. Taatjes DJ, Naar AM, Andel F, 3rd, Nogales E, Tjian R. Structure, function, and activator-induced conformations of the CRSP coactivator. Science. 2002;295:1058–1062. doi: 10.1126/science.1065249.. Electron microscopic structural studies show that binding of two different activators to the Mediator complex induces distinct conformational changes.
- 57.Fryer CJ, White JB, Jones KA. Mastermind recruits CycC:CDK8 to phosphorylate the Notch ICD and coordinate activation with turnover. Mol Cell. 2004;16:509–520. doi: 10.1016/j.molcel.2004.10.014. [DOI] [PubMed] [Google Scholar]
- 58.Hecht A, Vleminckx K, Stemmler MP, van Roy F, Kemler R. The p300/CBP acetyltransferases function as transcriptional coactivators of beta-catenin in vertebrates. EMBO J. 2000;19:1839–1850. doi: 10.1093/emboj/19.8.1839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Janknecht R, Wells NJ, Hunter T. TGF-beta-stimulated cooperation of smad proteins with the coactivators CBP/p300. Genes Dev. 1998;12:2114–2119. doi: 10.1101/gad.12.14.2114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fryer CJ, Lamar E, Turbachova I, Kintner C, Jones KA. Mastermind mediates chromatin-specific transcription and turnover of the Notch enhancer complex. Genes Dev. 2002;16:1397–1411. doi: 10.1101/gad.991602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chen Y, Goodman RH, Smolik SM. Cubitus interruptus requires Drosophila CREB-binding protein to activate wingless expression in the Drosophila embryo. Mol Cell Biol. 2000;20:1616–1625. doi: 10.1128/mcb.20.5.1616-1625.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Paulson M, Pisharody S, Pan L, Guadagno S, Mui AL, Levy DE. Stat protein transactivation domains recruit p300/CBP through widely divergent sequences. J Biol Chem. 1999;274:25343–25349. doi: 10.1074/jbc.274.36.25343. [DOI] [PubMed] [Google Scholar]
- 63.Li QJ, Yang SH, Maeda Y, Sladek FM, Sharrocks AD, Martins-Green M. MAP kinase phosphorylation-dependent activation of Elk-1 leads to activation of the co-activator p300. Embo J. 2003;22:281–291. doi: 10.1093/emboj/cdg028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Murr R, Vaissiere T, Sawan C, Shukla V, Herceg Z. Orchestration of chromatin-based processes: mind the TRRAP. Oncogene. 2007;26:5358–5372. doi: 10.1038/sj.onc.1210605. [DOI] [PubMed] [Google Scholar]
- 65.Yang XJ. Lysine acetylation and the bromodomain: a new partnership for signaling. Bioessays. 2004;26:1076–1087. doi: 10.1002/bies.20104. [DOI] [PubMed] [Google Scholar]
- 66.Li J, Sutter C, Parker DS, Blauwkamp T, Fang M, Cadigan KM. CBP/p300 are bimodal regulators of Wnt signaling. Embo J. 2007;26:2284–2294. doi: 10.1038/sj.emboj.7601667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Waltzer L, Bienz M. Drosophila CBP represses the transcription factor TCF to antagonize Wingless signalling. Nature. 1998;395:521–525. doi: 10.1038/26785.. This paper was an early demonstration that CBP has non-histone targets; in this case acetylation of TCF by CBP reduces its ability to bind to β-catenin and activate transcription.
- 68.Wolf D, Rodova M, Miska EA, Calvet JP, Kouzarides T. Acetylation of beta-catenin by CREB-binding protein (CBP) J Biol Chem. 2002;277:25562–25567. doi: 10.1074/jbc.M201196200. [DOI] [PubMed] [Google Scholar]
- 69.Levy L, Wei Y, Labalette C, Wu Y, Renard CA, Buendia MA, Neuveut C. Acetylation of beta-catenin by p300 regulates beta-catenin-Tcf4 interaction. Mol Cell Biol. 2004;24:3404–3414. doi: 10.1128/MCB.24.8.3404-3414.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Huang WC, Ju TK, Hung MC, Chen CC. Phosphorylation of CBP by IKKalpha promotes cell growth by switching the binding preference of CBP from p53 to NF-kappaB. Mol Cell. 2007;26:75–87. doi: 10.1016/j.molcel.2007.02.019.. This paper demonstrates that post-translational modification of CBP is a route for crosstalk between pathways. Phosphorylation of CBP by IKKα increases its binding to NF-κB and reduces its binding to p53.