Abstract
Electromyography (EMG) studies are useful in the anatomical localization of nerve injuries and, in most cases, isolating lesions to a single nerve root level. Their utility is important in identifying specific nerve-root-level injuries where surgical or interventional procedures may be warranted. In this case report, an individual presented with right upper extremity radicular symptoms consistent with a clinical diagnosis of cervical radiculopathy. EMG studies revealed that the lesion could be more specifically isolated to the T1 nerve root and, furthermore, provided evidence that the abductor pollicis brevis receives predominantly T1 innervation.
Keywords: electromyography, EMG, electrodiagnostics, T1 radiculopathy, abductor pollicis brevis (APB), T1-T2 disc herniation
Introduction
Neurological deficits involving the cervical nerve roots (C5–T1) supplying the upper extremities are a common indication for obtaining electromyographic studies. However, radiculopathies involving the first thoracic nerve root (T1) are reported to be the least common with <1% of cervical root lesions identified at T1 [1, 2]. One confounding reason may simply be that most comprehensive studies investigating cervical radiculopathy exclude T1 nerve root pathology from their analysis [3]. This may be due in part to the fact that common medical teaching of upper extremity myotomal innervations merges C8 and T1 nerve root levels and does not specifically assign any individual muscle to be predominantly T1. In the following case, further proof is provided to support a growing body of evidence that the abductor pollicis brevis is predominantly innervated by the T1 nerve root level [3–8].
Case report
A 63-year-old male presented with 6-week history of right-sided neck–arm pain and numbness after hauling heavy loads. At the time of injury, the patient reports progressive achiness at the right periscapular region followed by radiating pain and numbness down the medial aspect of the right upper extremity. Paresthesias were noted predominantly in the right fourth and fifth digits with subjective complaints of loss of dexterity in the right hand manifesting as occasionally dropping objects and deterioration of penmanship.
The patient reported no weakness, sudden trauma, prodromal illness, or previous injury. His symptoms are relatively unchanged from the onset of injury. Activities of daily living have been modified to decrease or avoid painful activities or movements. Certain cervical positions, especially neck flexion, exacerbate both pain and numbness sensations. For the past 6 weeks, he has tried relative rest and chiropractic treatments with little change in symptomology.
On general examination, the patient presented as a well-nourished age-appropriate individual with normal gait. Inspection revealed no gross evidence of deformity or atrophy of the posterior spine and shoulders. There was otherwise normal muscle bulk and tone in bilateral upper extremities with no fasciculations. There was also no evidence of Horner’s syndrome.
Range of motion examination of the cervical spine revealed full lateral bending and rotation with minimally decreased range in flexion and extension with end range axial lower neck pain. Spurling’s maneuver to the right reproduced concordant pain with exacerbation of radicular symptoms down the right upper extremity. There were no positive signs of impingement on examination of the right shoulder. Strength examination revealed 4/5 strength on right thumb abduction and 5/5 strength in all other muscles of both upper extremities. Sensation examination was diminished over the medial aspect of the right arm extending into the medial hand. Reflexes were present and symmetric at bilateral biceps, brachioradialis, and triceps. Phalen’s maneuver and Tinel’s sign were negative. There were no myelopathic signs on examination.
Electromyographic study was performed 6 weeks after onset of symptoms. Nerve conduction studies of sensory nerves were normal and symmetric (Table 1). The importance of this finding reflects relative continuity of sensory neurons originating in the dorsal root ganglion distally to the hand at the point of the recording electrodes. This would indicate that a postganglionic lesion or injury is not present and, if a lesion does exist, it would be preganglionic. Furthermore, this normal finding demonstrates no evidence of median or ulnar sensory nerve compression at the hand or wrist, essentially ruling out a median mononeuropathy at the wrist which would indicate carpal tunnel syndrome.
Table 1.
Nerve conduction values of sensory nerves
| Nerve | Site | Onset lat (ms) | Norm onset | Amplitude | Norm amp | Duration (ms) | Seg name | Distance (cm) | Velocity (m/s) | Norm vel |
|---|---|---|---|---|---|---|---|---|---|---|
| L med/antbr | Forearm | |||||||||
| Abv elbow | 1.7 | 10.9 | Abv elbow–forearm | 9.0 | 52.9 | |||||
| R med/antbr | Forearm | |||||||||
| Abv elbow | 1.3 | 10.5 | Abv elbow–forearm | 8.5 | 65.4 | |||||
| L median | Second digit | |||||||||
| Wrist | 3.2 | <3.2 | 20.2 | >10 | ||||||
| R median | Second digit | |||||||||
| Mid palm | 1.4 | 15.2 | 1.4 | Mid palm–second digit | 8.0 | 57.1 | >45.0 | |||
| Wrist | 3.2 | <3.2 | 16.6 | 1.8 | Wrist–mid palm | 8.5 | 47.2 | |||
| L ulnar | Fifth digit | |||||||||
| Wrist | 2.7 | <3.2 | 30.1 | >10 | 2.7 | Wrist–fifth digit | 0.0 | |||
| R ulnar | Fifth digit | |||||||||
| Palm | 1.6 | 22.1 | 1.6 | Palm–fifth digit | 8.0 | 50.0 | ||||
| Wrist | 2.7 | <3.2 | 22.4 | >10 | 1.1 | Wrist–palm | 7.0 | 63.6 | >45 |
Motor nerve studies revealed mildly prolonged distal onset latency and severely reduced amplitude of the right median motor nerve (Table 2). This finding points to an underlying axonal loss which may have occurred anywhere between the anterior horn cells within the spinal cord to the distal recording motor site. The degree of axonal loss could explain the mildly prolonged median motor distal latency due to unreliable determination of accurate onset or, alternatively, it may have been secondary to a mild, clinically insignificant, median mononeuropathy at the wrist consistent with carpal tunnel syndrome. The muscle tested in this median motor examination was the abductor pollicis brevis with reported C8–T1 nerve root innervation. However, the normal sensory conduction study suggests that the site of injury was between the anterior horn cell and the dorsal root ganglion.
Table 2.
Nerve conduction values of motor nerves
| Nerve | Site | Onset lat (ms) | Norm onset | Amplitude | Norm amp | Duration (ms) | Seg name | Distance (cm) | Velocity (m/s) | Norm vel |
|---|---|---|---|---|---|---|---|---|---|---|
| R median | Wrist | 4.2 | <3.6 | 0.7 | >4 | Pron ter–wrist | 25.0 | 51.0 | >50.0 | |
| Pron ter | 9.1 | 0.5 | ||||||||
| R ulnar | Wrist | 2.8 | <3.6 | 3.8 | >3 | 24.0 | 53.3 | >50.0 | ||
| Abv FCU | 7.3 | 3.1 | Abv uln grv–abv FCU | 12.0 | 50.0 | >50.0 | ||||
| Abv uln grv | 9.7 | 3.1 |
Electromyographic needle examination revealed abnormal spontaneous activity in the form of positive sharp waves and fibrillations in C8–T1 innervated muscles (Table 3). The right abductor pollicis brevis revealed the most noted abnormal changes in the form of severe spontaneous activity, discrete recruitment pattern, and decreased recruitment interval. The right first dorsal interosseus, flexor carpi ulnaris, and abductor digiti minimi muscles revealed mild abnormal spontaneous activity and normal recruitment pattern. There was also mild abnormal spontaneous activity at the right lower cervical paraspinal muscles. All remaining muscles innervated by cervical nerve roots cranial to C8 showed no abnormal spontaneous activity, normal motor unit configuration, and normal motor unit recruitment. These electrodiagnostic studies are consistent with a severe right C8 but, more likely, T1 radiculopathy predominantly involving the anterior ramus division.
Table 3.
Electromyographic needle examination
| Side | Muscle | Nerve | Root | INS | FIBS | PSW | FAS | AMP | DUR | Configuration | PAT | REC INT |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R | AbdPollBrev | Median | C8–T1 | Nml | 2+ | 3+ | 0 | Nml | Nml | Di/Tri phasic | Discrete | Dec |
| R | 1stDorInt | Ulnar | C8–T1 | Nml | 1+ | 1+ | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | FlexCarRad | Median | C6–7 | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | LatHdTriceps | Radial | C6–7 | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | Biceps | Musc | C5–6 | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | Deltoid Mid | Axillary | C5–6 | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | ExtIndicis | Radial | C7–8 | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri Phasic | Full | Nml |
| R | FlexCarUln | Ulnar | C8–T1 | Nml | 1+ | 1+ | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | ABDDigMin | Ulnar | C8–T1 | Nml | 1+ | 1+ | 0 | Nml | Nml | Di/Tri phasic | Full | Nml |
| R | CervPara Up | Rami | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | |||
| R | CervPara Mid | Rami | Nml | 0 | 0 | 0 | Nml | Nml | Di/Tri phasic | |||
| R | CervPara Low | Rami | Nml | 0 | 1+ | 0 | Nml | Nml | Di/Tri phasic |
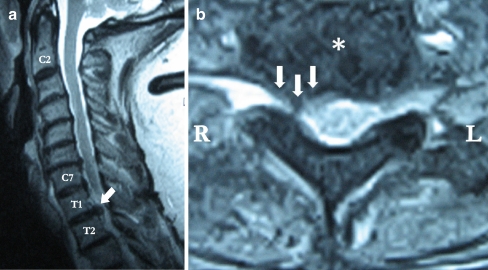
A cervical spine MRI was performed to examine the cervical nerve roots. This study demonstrated a right posterolateral and foraminal disc extrusion 4 mm in AP length at the T1–T2 intervertebral level (Fig. 1). Furthermore, there was no significant pathology related to the C8 nerve root at the C7–T1 disc level. The findings at that level were minimal with only a mild central disc bulge.
Fig. 1.
Cervical MRI shows right posterolateral T1–T2 intervertebral disc protrusion. a Sagittal image of cervical MRI demonstrating T1–T2 disc protrusion (arrow). b Axial image of cervical MRI demonstrating T1–T2 intervertebral disc (asterisk) with right posterolateral protrusion (arrows)
Based on the history, physical exam, electrodiagnostic studies, and the MRI, the final diagnosis arrived at was right posterolateral T1–T2 intervertebral disc protrusion causing clinically significant right T1 radiculopathy.
After discussion of potential treatment options, the patient chose to undergo conservative management. The patient was subsequently referred to physical therapy for postural exercises and retraining, manual chin retractions, anterior chest wall stretching, shoulder retractions, and soft tissue massage to improve segmental motions.
Discussion
Thoracic disc herniation is a rare condition that occurs in only about 1% of all disc herniations [9–11]. When they do occur, they are typically found in older patients and are predominantly found in the lower thoracic spine [11–13]. The most common disc level for thoracic disc herniation was found to be the T11–12 level with 26–50% of the total number [14–16]. A classic study reviewing the occurrence of 258 thoracic disc herniations found the majority (66%) occurred in the lower third of the thoracic spine (between T9–10 and T12–L1), then middle third (28% between T5–6 and T8–9), followed least by the upper thoracic spine (6% between T1–2 and T4–5) [17]. Of the small number of thoracic disc herniations that occur at the upper third thoracic level (between T1–2 and T4–5), T1–2 is the most common [17].
Thoracic disc disease and diabetes mellitus represent two of the most frequent etiologies for the development of thoracic radiculopathy [18]. Degeneration is favored as the prevailing cause for thoracic disc herniation, and the lower thoracic segments are most at risk because of the increased motion present at these levels [19]. When thoracic disc herniations do occur, they occur either in a posterior-central or a posterior-lateral direction [20]. At the upper thoracic levels, one reason for more lateral disc herniation may be due to the absence of Luschka joints beginning at the C7–T1 level [21].
There are many theoretical reasons for the decreased incidence of clinically significant thoracic disc herniations including the fact that disc heights tend to be smaller and discs have less volume [19]. It may also be due to decreased weight bearing loads in flexion and extension, as the primary movements of the thoracic spine are rotation and lateral bending [22]. Regarding certain cases of T1–2 intervertebral disc herniation, reports indicate a causal relationship secondary to increased mechanical stresses following postsurgical treatment at the cervical levels [23, 24].
The literature regarding symptomology of T1 herniated disc radiculopathy supports the findings of this case, as well as rarer type presentations. Studies have reported cases of paraparesis and myelopathy [25, 26]. However, typical reports include symptoms of hand weakness and pain along the medial arm [4, 27–30]. Also, considering possible T1 contribution to the sympathetic trunk, there are reported cases of oculosympathetic paralysis or Horner’s syndrome [27, 28, 31]. An isolated case of a patient with chronic chest pain underwent an exhaustive medical workup before the etiology was attributed to a T1–2 disc herniation compressing the T1 nerve root [24].
In this case, electromyography assisted in the diagnosis of a T1 radiculopathy with findings that support a predominant T1 innervation to the abductor pollicis brevis. The finding of significantly decreased motor amplitude of the median nerve points to an underlying axonal lesion affecting the C8–T1 myotome. Furthermore, the spontaneous activity found in C8–T1 innervated muscles (first dorsal interosseus, flexor carpi ulnaris, and abductor digiti minimi) point to a C8 and T1 distribution as well as the spontaneous activity found in the lower cervical paraspinals. The abnormal spontaneous activity in the lower cervical paraspinal muscles can be debated as to their nerve root etiology. There is considerable nerve root overlap in paraspinal innervation and the abnormal spontaneous activity in the lower cervical paraspinals could originate from either C8 and/or T1 nerve root levels. However, the robust findings of severe abnormal spontaneous activity and decreased recruitment pattern–interval of the abductor pollicis brevis supports a T1 radiculopathy. This, in light of a disc herniation at the T1–2 intervertebral level, supports the growing body of evidence that the APB is primarily T1 innervated [3–8].
In summary, the traditional nomenclature of myotomal distribution of the upper extremity links C8 and T1 nerve roots when describing the muscular innervations of the forearm and hand. There are no classically taught innervations that assign any muscle of the upper extremity with mainly T1 innervation. The electrophysiologic findings in this case help purport the more accepted claim that the APB is the only muscle in the upper extremity with chiefly T1 innervation.
References
- 1.Murphey F, Simmons JC, Brunson B (1973) Surgical treatment of laterally ruptured cervical disc. Review of 648 cases, 1939 to 1972. J Neurosurg 38:679–683 [DOI] [PubMed]
- 2.Levin KH (2002) Electrodiagnostic approach to the patient with suspected radiculopathy. Neurol. Clin N Am 20:397–421 [DOI] [PubMed]
- 3.Levin KH, Maggiano HJ, Wilbourn AJ (1996) Cervical radiculopathies: comparison of surgical and EMG localization of single-root lesions. Neurology 46:1022–1025 [DOI] [PubMed]
- 4.Levin KH (1999) Neurologic manifestations of compressive radiculopathy of the first thoracic root. Neurology. 53:1149–1151 [DOI] [PubMed]
- 5.Levin KH, Wilbourn AJ, Maggiano HJ (1998) Cervical rib and median sternotomy-related brachial plexopathies: a reassessment. Neurology 50:1407–1413 [DOI] [PubMed]
- 6.Wilbourn AJ, Ferrante MA (2000) Clinical electromyography. In: Joynt RJ, Griggs RC (eds) Baker’s clinical neurology on CDROM. Lippincott, Williams & Wilkins, Philadelphia, pp 7592–8248. record
- 7.Wilbourn AJ (1985) Electrodiagnosis of plexopathies. Neuro Clin 3:511–529 [PubMed]
- 8.Ferrante MA, Wilbourn AJ (2002) Electrodiagnostic approach to the patient with suspected brachial plexopathy. Neurol Clin N Am 20:423–450 [DOI] [PubMed]
- 9.Alberico AM, Sahni KS, Hall JA Jr et al (1986) High thoracic disc herniation. Neurosurgery 19:449–451 [DOI] [PubMed]
- 10.Arce CA, Dohrmann GJ (1985) Herniated thoracic disks. Neurol Clin 3:383–392 [PubMed]
- 11.Singounas EG, Kypriades EM, Kellerman AJ et al (1992) Thoracic disc herniation. Analysis of 14 cases and review of the literature. Acta Neurochir (Wien) 116:49–52 [DOI] [PubMed]
- 12.Awwad EE, Martin DS, Smith KR et al (1991) Asymptomatic versus symptomatic herniated thoracic discs: their frequency and characteristics as detected by computed tomography after myelography. Neurosurgery 28:180–186 [DOI] [PubMed]
- 13.Brown CW, Deffer PA Jr, Akmakjian J et al (1992) The natural history of thoracic disc herniation. Spine 17:s97–s102 [DOI] [PubMed]
- 14.Blumenkopf B (1988) Thoracic intervertebral disc herniations: diagnostic value of magnetic resonance imaging. Neurosurgery 23:36–40 [DOI] [PubMed]
- 15.Tahmouresie A (1980) Herniated thoracic intervertebral disc—an unusual presentation: case report. Neurosurgery 7:623–625 [DOI] [PubMed]
- 16.Videman T, Battié MC, Gill K et al (1995) Magnetic resonance imaging findings and their relationships in the thoracic and lumbar spine. Insights into the etiopathogenesis of spinal degeneration. Spine 20:928–935 [DOI] [PubMed]
- 17.Arce CA, Dohrmann GJ (1985) Thoracic disc herniation. Improved diagnosis with computed tomographic scanning and a review of the literature. Surg Neurol 23:356–361 [DOI] [PubMed]
- 18.O’Connor RC, Andary MT, Russo RB et al (2002) Thoracic radiculopathy. Phys Med Rehabil Clin N Am 13:623–644 [DOI] [PubMed]
- 19.McInerney J, Ball PA (2000) The pathophysiology of thoracic disc disease. Neurosurg Focus 9:1–8 [DOI] [PubMed]
- 20.Stillerman CB, Chen TC, Couldwell WT et al (1988) Experience in the surgical management of 82 symptomatic herniated thoracic discs and review of the literature. J Neurosurg 88:623–633 [DOI] [PubMed]
- 21.Post NH, Cooper PR, Frempong-Boadu AK et al (2006) Unique features of herniated discs at the cervicothoracic junction: clinical presentation, imaging, operative management, and outcome after anterior decompressive operation in 10 patients. Neurosurgery 58:497–501 [DOI] [PubMed]
- 22.Parke WW (1982) Biomechanics of the spine. In: Rothman RH, Simeone FA (eds) The Spine, 2nd edn, vol 1. Saunders, Philadelphia, pp 47–49
- 23.Takagi H, Kawaguchi Y, Kanamori M et al (2002) T1–2 disc herniation following an en bloc cervical laminoplasty. J Ortho Sci 7:495–497 [DOI] [PubMed]
- 24.Frecerro D, Donovan DJ (2005) Adjacent segment degeneration at T1–T2 presenting as chest pain: case report. Spine 30:E655–E657 [DOI] [PubMed]
- 25.Negoveti L, Cerina V, Sajko T et al (2001) Intradural disc herniation at the T1–T2 level. Croat Med J 42:193–195 [PubMed]
- 26.Nakahara S, Sato T (1995) First thoracic disc herniation with myelopathy. Eur Spine J 4:366–367 [DOI] [PubMed]
- 27.Caner H, Kilinçoglu BF, Benli S et al (2003) Magnetic resonance image findings and surgical considerations in T1–2 disc herniation. Can J Neuro 30:152–154 [DOI] [PubMed]
- 28.Gelch MM (1978) Herniated thoracic disc at T1–2 level associated with Horner’s syndrome. Case report. J Neurosurg 48:128–129 [DOI] [PubMed]
- 29.Morgan H, Abood C (1998) Disc herniation at T1–2. Report of four cases and literature review. J Neurosurg 88:148–150 [DOI] [PubMed]
- 30.Lyu RK, Chang HS, Tang LM et al (1999) Thoracic disc herniation mimicking acute lumbar disc disease. Spine 24:416–418 [DOI] [PubMed]
- 31.Lloyd TV, Johnson JC, Paul DJ et al (1980) Horner’s syndrome secondary to herniated disc at T1–T2. Am J Roentgenol 134:184–185 [DOI] [PubMed]