Abstract
We report here the case of Whipple's disease in a 60-year-old man with severe arthralgia and systemic disorders but without gastrointestinal manifestations. The patient had different clinical diagnoses over a period of 14 years. We identified Tropheryma whipplei by real-time PCR. Molecular typing was also performed by sequencing the 16S-23S rRNA intergenic spacer region and domain III of the 23S rRNA gene.
CASE REPORT
A 60-year-old Italian man was admitted in March 2008 to the Istituto Fiorentino di Cura e Assistenza, Florence, Italy, after a 14-year history of multiple hospital admissions for recurring episodes of intermittent, migratory arthralgia and fever but without gastrointestinal (GI) signs such as abdominal pain, diarrhea, malabsorption, and weight loss. On admission, he was walking with assistance by crutches.
The patient's clinical history reported that the first episode of intermittent signs of arthralgia was in 1994 with a diagnosis of seronegative polyarthritis and suspected rheumatoid arthritis. He was treated for 4 years with nonsteroidal antiinflammatory drugs (NSAIDs) that produced relief from the symptoms until a severe relapse occurred in 2000. The patient presented with severe joint arthritis, particularly in the extremities of his legs and arms, which was mostly painful in the morning and required corticosteroid treatment. In 2001, due to the progressive worsening of symptoms coupled with the onset of skeletal muscle myalgia, the patient was admitted for the first time to the Department of Internal Medicine, Istituto Fiorentino di Cura e Assistenza (Florence, Italy). Extensive polyarthralgia of the extremities, as well as the shoulders, ankles, knees, fingers, and lower maxillary bones, was recorded. Electromyography, nerve conduction velocity, and somatosensory-evoked potential were recorded on bilateral stimulation of the median nerve and the posterior tibial nerve. Distal polyneuropathy was present with significant alterations. Laboratory tests indicated abnormal values for erythrocyte sedimentation rate (75 mm/h), C-reactive protein (45 mg/liter), hemoglobin (12.1 g/dl), sideremia (27 μg/dl), fibrinogen (600 mg/dl), and subclass immunoglobulin G levels (C3, 141 mg/dl; C4, 25 mg/dl), coupled with lymphocytopenia and neutrophilia. Rheumatoid factor and antinuclear antibodies were absent.
On suspicion of GI disorders, endoscopic examination was performed, which revealed yellow-white, friable mucosa in the descending duodenum. Duodenal biopsies showed foamy macrophages in the lamina propria. The macrophage cytoplasm contained large amounts of periodic acid-Schiff (PAS)-positive, diastase-resistant, Ziehl-Neelsen-negative particles (Fig. 1A). The diagnosis was clinically compatible with Whipple's disease without GI symptoms. During the hospitalization, the patient was treated with doxycycline (100 mg/twice daily), methylprednisolone (8 mg/daily), and methotrexate (5 mg/weekly). The antibiotic treatment was prolonged for 4 months at home. Clinical response was positive, with a rapid remission of symptoms. The patient remained free from symptoms for 3 years. In 2004 and 2006, he showed recurrent episodes of arthralgia and was treated with methylprednisolone and cyclophosphamide combination therapy.
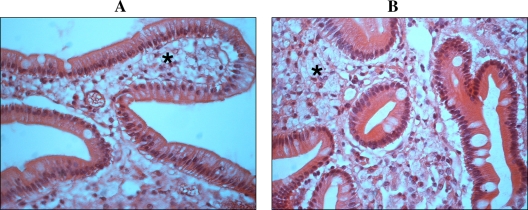
FIG. 1.
Histology of the intestinal biopsies stained with the PAS reagent (magnification, ×40). Shown are a sample from 2001 (A) and a sample from 2008 (B). Photographs show intestinal lamina propria infiltrated with macrophages with PAS-positive inclusions (asterisks). Deposits of lipid droplets are illustrated as empty spaces.
In February 2008, the patient had unexpected, rapid, and severe diffuse polyarthralgia with inflammatory signs, persistent fatigue, pulmonary chest pain, dyspnea, and heart problems but no GI involvement. He was then readmitted to the same hospital, where Whipple's disease was confirmed based on past history and clinical and laboratory findings. In particular, radiography revealed degenerative polyarthritis with bone demineralization and destructive joint changes at the extremities, knees, and fingers. Pulmonary involvement with pleural effusion was also detected. An echo-color Doppler of the heart showed a calcified bicuspid aortic valve without any abscess or cardiac vegetation.
GI endoscopic examination showed that the second portion of the duodenum was granulomatous, and histological examination of the duodenal mucosa showed that the lamina propria was infiltrated by PAS-positive histiocytes (Fig. 1B). Blood samples, duodenal biopsies, and paraffin-embedded slices of the duodenal specimens (which had been taken in 2001) that were suspected to be infected with Tropheryma whipplei were sent to the Istituto Superiore di Sanità, Department of Infectious, Parasitic, and Immune-Mediated Diseases, where the samples were examined by PCR.
Results for blood cultures and a search for parasites were both negative. The PCR result for eubacterial 16S rRNAs (27f, 5′-GAGAGTTTGATCCTGGCTCAG-3′; 1495r, 5′-CTACGGCTACCTTGTTACGA-3′) from the DNA sample from blood was also negative. DNAs were extracted from the duodenal biopsy samples by the Nucleospin tissue kit (Macherey-Nagel GmbH, Düren, Germany), and protocols for human tissue and paraffin-embedded slices were carried out according to the manufacturer's instructions. Five microliters (100 ng) of DNA was used for real-time PCR SYBR green amplification in the LightCycler instrument (Roche Diagnostics, GmbH, Germany) with the T. whipplei-specific primer pairs whipp-frw2/whipp-rev (357 bp) for the hsp65 gene target (9). Samples were also checked for the presence of PCR inhibitors using the LightCycler control kit DNA for the human β-globin gene (Roche Diagnostics, GmbH, Germany) according to the manufacturer's instructions. Duodenal biopsies taken in 2001 and 2008 were both positive for the hsp65 gene fragment. To further confirm the bacterial identification, all of the 16S rRNAs were amplified using the universal primers for eubacteria, and the PCR products from all specimens were analyzed by automated DNA sequencing. The nucleotide sequences obtained showed 100% identity within the hsp65 (GenBank accession number AF184091) and the 16S rRNA (GenBank accession number AF202891) genes of T. whipplei (25).
In order to characterize the strain of T. whipplei detected in tissue samples, the 16S-23S rRNA gene intergenic spacer region and domain III of the 23S rRNA gene were amplified and sequenced as previously described (14, 15). Both samples presented the same pattern and were classified as type 2A with 100% nucleotide identity (Fig. 2).
FIG. 2.
Sequence analysis alignment (partial sequences) of the two biopsy samples (from 2001 and 2008) with the 16S-23S rRNA intergenic spacer region (type 2) and the 23S rRNA domain III (type A). Dots and underlined letters symbolize identity and target nucleotides, respectively. *, GenBank accession no. AF100950; **, GenBank accession no. AF148136.
Based on the characteristics from pathological and molecular findings, the definitive diagnosis of Whipple's disease was made. The patient is now under continuous administration of 4 g trimethoprim-sulfamethoxazole per day, orally.
Whipple's disease is a rare, chronic, multisystemic disease caused by the gram-positive bacterium T. whipplei. The natural source of this environmentally ubiquitous bacterium has not been defined, and patient-to-patient transmission has never been reported (28). The pathogenesis of Whipple's disease is not yet well understood. Predisposition to infection due to undefined immunological disorders (8) and/or genetic risk factors (12) has been postulated. However, the disease occurs most frequently in middle-aged men, as repeatedly reported (12, 19, 27).
Usually the small intestine is involved, but the disease may affect virtually any organ. Clinical manifestations are quite diverse and include all major organs and tissues (11, 19).
In some patients, the migratory arthralgia with nondeforming polyarthritis may precede the onset of typical GI symptoms (16). In exceptional cases, Whipple's disease can also occur without any classical intestinal signs for the whole duration of infection (13). In this circumstance, especially when NSAID therapy is ineffective, a biopsy of the intestines should be performed (22, 23).
About three-quarters of patients with joint manifestations also complain of intestinal problems over a temporal interval of 6 years. In most patients, duodenal and jejunal biopsy specimens contain macrophages with PAS-positive granules corresponding to bacteria. Nevertheless, some patients have no intestinal symptoms, and a few have normal intestinal histological findings (12).
Cardiovascular complications caused by T. whipplei have been described. Clinical manifestations of heart disease are less evident and are characterized by the presence of systolic murmurs, congestive heart failure, and nonspecific electrocardiogram changes with aortic or mitral valve insufficiency (3, 6, 18, 26). Interestingly, postmortem reports of patients with Whipple's disease describe that more than 50% have cardiovascular involvement based on histopathology and molecular technique (11, 20).
Less-common symptoms of Whipple's disease include central nervous system manifestations, cutaneous nonspecific dermatitis/panniculitis, muscle manifestations, peripheral neuropathy, and lung manifestations, which have been characterized by pleuritic chest pain, chronic nonproductive cough, dyspnea, and pleural effusion (8, 19, 24, 27).
We have described here an unusual and atypical case of Whipple's disease. The patient in this report showed multiorgan involvement, including erosive and destructive polyarthritis, muscle myalgia, polyneuropathy, and pulmonary and cardiological implications with uncertain etiology, but not weight loss or GI symptoms typically seen with Whipple's disease. The first clinical diagnosis was made 7 years after the initial signs and was based on histological and laboratory suspicions. The definitive diagnosis was made by molecular identification of T. whipplei in GI biopsies.
As suggested in recent papers, our case clearly demonstrates and underlines the importance of performing biopsies/histopathological analyses and molecular assays to confirm the bacterial identification and place the diagnosis of Whipple's disease on solid ground. This aspect is a focal point in suspected Whipple's disease, particularly in the absence of GI symptoms and/or when PCR analyses show negative results for blood/body fluids (5, 17, 21).
The patient's biopsies were histologically characterized by foamy macrophages containing granules that stained positive with PAS reagent. It must be stressed here the difficulties in the isolation of T. whipplei in pure culture, which remains limited to specialized research laboratories. PCR assays and electron microscopy are alternative diagnostic methods. However, PCR techniques should be limited to accredited laboratories, and the amplified DNA should be verified by sequencing, as electron microscopy is too complex and time consuming (29).
We also characterized the T. whipplei strain type by considering the analyses of the 16S-23S rRNA spacer region and the 23S rRNA domain III to possibly detect strain differences between the 2001 and 2008 biopsy samples. We found the same strain type, 2A, in both tissue sections. All this not only confirms identification but also makes it quite plausible that the bacterium was not eradicated. In fact, although antibiotic therapy consistently leads to remission, relapses can occur, and when the disease is untreated the outcome is fatal.
In vitro antibiotic susceptibility studies of T. whipplei have shown that doxycycline and sulfamethoxazole are effective against the organism (4). However, doxycycline alone seems to be more bacteriostatic than bactericidal (30), and this is one explanation for recurrence of disease in our case. Moreover, the protracted NSAID therapy probably favored bacterial persistence, because the immunosuppressive treatment could increase tissue colonization (16).
Current recommendations for first-line treatment are tetracycline administration followed by a course of at least 1 year of trimethoprim-sulfamethoxazole treatment for the blood-brain barrier, because relapse is frequent (30%) (17).
To our knowledge, this is a rare case of destructive polyarthritis due to T. whipplei without GI signs and with multiorgan repercussions in a patient not previously diagnosed with Whipple's disease. In fact, in the literature we have found very few reports of bone deformity or severe damage associated with Whipple's disease (1, 2, 7, 10). Furthermore, our report highlighted the helpfulness and benefit of molecular methods for diagnosis of Whipple's disease, remarkably in the absence of GI findings and with concomitant secondary diseases where definitive diagnosis can be difficult. Additionally, the molecular strain typing is an important laboratory tool for follow-up investigations and control of therapeutic success or failure.
Acknowledgments
We thank Francesco Pallone (Cattedra di Gastroenterologia, Dipartimento di Medicina Interna, Università Tor Vergata, Rome, Italy) for critically reading the manuscript and Maria Losardo for technical help.
This work was supported by the Italian Ministry of Research and University, Rome, Italy, FIRB grant Costruzione di un Laboratorio Nazionale per le Resistenze agli Antimicrobici.
Footnotes
Published ahead of print on 17 December 2008.
REFERENCES
- 1.Akar, Z., N. Tanriover, S. Tüzgen, B. Canbaz, H. Erman, B. Oz, and C. Kuday. 2002. Intracerebral Whipple disease: unusual location and bone destruction. Case report. J. Neurosurg. 97988-991. [DOI] [PubMed] [Google Scholar]
- 2.Ayoub, W. T., D. E. David, D. Torretti, and F. J. Viozzi. 1982. Bone destruction and ankylosis in Whipple's disease. J. Rheumatol. 9930-931. [PubMed] [Google Scholar]
- 3.Bostwick, D. G., K. G. Bensch, J. S. Burke, M. E. Billingham, D. C. Miller, J. C. Smith, and D. F. Keren. 1981. Whipple's disease presenting as aortic insufficiency. N. Engl. J. Med. 305995-998. [DOI] [PubMed] [Google Scholar]
- 4.Boulos, A., J. M. Rolain, M. N. Mallet, and D. Raoult. 2005. Molecular evaluation of antibiotic susceptibility of Tropheryma whipplei in axenic medium. J. Antimicrob. Chemother. 55178-181. [DOI] [PubMed] [Google Scholar]
- 5.Buckle, M. J., R. W. Ellis, M. Bone, and H. Lockman. 2008. Neurologically presenting Whipple disease: case report and review of the literature. J. Clin. Pathol. 611140-1141. [DOI] [PubMed] [Google Scholar]
- 6.Celard, M., G. de Gevigney, S. Mosnier, P. Buttard, Y. Benito, J. Etienne, and F. Vandenesch. 1999. Polymerase chain reaction analysis for diagnosis of Tropheryma whippelii infective endocarditis in two patients with no previous evidence of Whipple's disease. Clin. Infect. Dis. 291348-1349. [DOI] [PubMed] [Google Scholar]
- 7.Dearment, M. C., T. A. Woodward, D. M. Menke, P. W. Brazis, L. W. Bancroft, and S. T. Persellin. 2003. Whipple's disease with destructive arthritis, abdominal lymphadenopathy, and central nervous system involvement. J. Rheumatol. 301347-1350. [PubMed] [Google Scholar]
- 8.Desnues, B., M. Ihrig, D. Raoult, and J. L. Mege. 2006. Whipple's disease: a macrophage disease. Clin. Vaccine Immunol. 13170-178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dreier, J., F. Szabados, A. von Herbay, T. Kroger, and K. Kleesiek. 2004. Tropheryma whipplei infection of an acellular porcine heart valve bioprosthesis in a patient who did not have intestinal Whipple's disease. J. Clin. Microbiol. 424487-4493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Durand, D. V., C. Lecomte, P. Cathébras, H. Rousset, P. Godeau, et al. 1997. Whipple disease. Clinical review of 52 cases. Medicine (Baltimore) 76170-184. [DOI] [PubMed] [Google Scholar]
- 11.Dutly, F., and M. Altwegg. 2001. Whipple's disease and “Tropheryma whippelii.” Clin. Microbiol. Rev. 14561-583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fenollar, F., X. Puechal, and D. Raoult. 2007. Whipple's disease. N. Engl. J. Med. 35655-66. [DOI] [PubMed] [Google Scholar]
- 13.Gagné, M., J. Brown, A. Lussier, M. Rola-Pleszczynski, and M. Camerlain. 1983. Maladie de Whipple sans manifestations digestives: arthropathie de diagnostic tardif. Union Med. Can. 112628-632. [PubMed] [Google Scholar]
- 14.Hinrikson, H. P., F. Dutly, and M. Altwegg. 2000. Evaluation of a specific nested PCR targeting domain III of the 23S rRNA gene of “Tropheryma whippelii” and proposal of a classification system for its molecular variants. J. Clin. Microbiol. 38595-599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hinrikson, H. P., F. Dutly, S. Nair, and M. Altwegg. 1999. Detection of three different types of “Tropheryma whippelii” directly from clinical specimens by sequencing, single-strand conformation polymorphism (SSCP) analysis and type-specific PCR of their 16S-23S ribosomal intergenic spacer region. Int. J. Syst. Bacteriol. 491701-1706. [DOI] [PubMed] [Google Scholar]
- 16.Mahnel, R., A. Kalt, S. Ring, A. Stallmach, W. Strober, and T. Marth. 2005. Immunosuppressive therapy in Whipple's disease patients is associated with the appearance of gastrointestinal manifestations. Am. J. Gastroenterol. 1001167-1173. [DOI] [PubMed] [Google Scholar]
- 17.Maiwald, M., A. von Herbay, and D. A. Relman. 2006. Whipple's disease, p. 1496-1500. In M. Feldman, L. S. Friedman, and L. Brandt (ed.), Sleisenger & Fordtran's gastrointestinal and liver disease, 8th ed. W. B. Saunders, Philadelphia, PA.
- 18.Marín, M., P. Munoz, M. Sanchez, M. Del Rosal, M. Rodriguez-Creixems, and E. Bouza. 2007. Tropheryma whipplei infective endocarditis as the only manifestation of Whipple's disease. J. Clin. Microbiol. 452078-2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Marth, T., and D. Raoult. 2003. Whipple's disease. Lancet 361239-246. [DOI] [PubMed] [Google Scholar]
- 20.McAllister, H. A., Jr., and J. J. Fenoglio, Jr. 1975. Cardiac involvement in Whipple's disease. Circulation 52152-156. [DOI] [PubMed] [Google Scholar]
- 21.Prüss, H., J. Katchanov, R. Zschenderlein, C. Loddenkemper, T. Schneider, and V. Moos. 2007. A patient with cerebral Whipple disease with gastric involvement but no gastrointestinal symptoms: a consequence of local protective immunity? J. Neurol. Neurosurg. Psychiatr. 78896-898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Puéchal, X. 2001. Whipple disease and arthritis. Curr. Opin. Rheumatol. 1374-79. [DOI] [PubMed] [Google Scholar]
- 23.Puéchal, X., T. Schaeverbeke, J. Sibilia, A. Saraux, and J. D. Poveda. 2002. Polymerase chain reaction testing for Tropheryma whippelii in unexplained isolated cases of arthritis. Arthritis Rheum. 461130-1132. [DOI] [PubMed] [Google Scholar]
- 24.Puget, M., J. Iwaz, A. Tristan, and N. Streichenberger. 2006. Whipple's disease with muscle impairment. Muscle Nerve 34794-798. [DOI] [PubMed] [Google Scholar]
- 25.Raoult, D., H. Ogata, S. Audic, C. Robert, K. Suhre, M. Drancourt, and J. M. Claverie. 2003. Tropheryma whipplei Twist: a human pathogenic Actinobacteria with a reduced genome. Genome Res. 131800-1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Richardson, D. C., L. L. Burrows, B. Korithoski, I. E. Salit, J. Butany, T. E. David, and J. M. Conly. 2003. Tropheryma whippelii as a cause of afebrile culture-negative endocarditis: the evolving spectrum of Whipple's disease. J. Infect. 47170-173. [DOI] [PubMed] [Google Scholar]
- 27.Schneider, T., V. Moos, C. Loddenkemper, T. Marth, F. Fenollar, and D. Raoult. 2008. Whipple's disease: new aspects of pathogenesis and treatment. Lancet Infect. Dis. 8179-190. [DOI] [PubMed] [Google Scholar]
- 28.Schöniger-Hekele, M., D. Petermann, B. Weber, and C. Muller. 2007. Tropheryma whipplei in the environment: survey of sewage plant influxes and sewage plant workers. Appl. Environ. Microbiol. 732033-2035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Swartz, M. N. 2000. Whipple's disease—past, present, and future. N. Engl. J. Med. 342648-650. [DOI] [PubMed] [Google Scholar]
- 30.Van La, M., P. Barbry, D. Raoult, and P. Renesto. 2007. Molecular basis of Tropheryma whipplei doxycycline susceptibility examined by transcriptional profiling. J. Antimicrob. Chemother. 59370-377. [DOI] [PubMed] [Google Scholar]