Abstract
Vascular access failure is a major clinical problem for patients on hemodialysis but therapeutic approaches aimed at improving arteriovenous fistula (AVF) dysfunction remain elusive. Using a murine AVF model, Juncos et al. demonstrate for the first time that the stress protein heme oxygenase-1 (HO-1) is induced in AVFs, and that HO-1 is a critical determinant of AVF survival.
Hemodialysis vascular access failure is the dominant cause of morbidity in patients with end stage renal disease and contributes substantially to the economic burden of this patient population. Vascular access complications account for about 20% of the hospitalization of hemodialysis patients in the United States with an annual cost of $1 billion dollars (1). The native arteriovenous fistula (AVF) is the preferred form of dialysis access but suffers from frequent failure. According to an analysis of the pooled data by the Dialysis Outcomes Quality Initiative committee, the primary patency of AVFs was 85% at 1 year and 75% at 2 years after excluding patients with primary failure, the rate of which may be up to 50% in some centers. The major cause of AVF dysfunction is neointimal hyperplasia leading to the development of venous stenosis and subsequent thrombosis. Significantly, neointimal hyperplasia has also been implicated in the failure of AVFs to mature and accommodate the increased rate of blood flow required for effective dialysis. Histologically, these neointimal lesions consist of proliferating and migrating vascular smooth muscle cells (VSMCs), infiltrating inflammatory cells, substantial amounts of extracellular matrix, and angiogenesis in both the adventitia and intima of the lesion (see 2). Multiple factors are believed to contribute to the pathogenesis of neointimal hyperplasia following the creation of AVFs, including surgical injury, hemodynamic stress, platelet activation, endothelial dysfunction, oxidative stress, inflammation, and uremia. While ever more sophisticated surveillance techniques can reliable detect subcritical and critical stenoses, therapeutic interventions that target this lesion and substantially prolong patency remain elusive.
Heme oxygenase-1 (HO-1) as a highly inducible stress protein that mediates important vasoprotective actions in the circulation (see 3). Pharmacological induction of HO-1 or adenovirus-mediated HO-1 gene delivery attenuates neointimal hyperplasia in injured arteries and retards the development of atherosclerotic plaques in murine models of atherosclerosis, whereas inhibition of HO-1 activity enhances lesion formation. In addition, HO-1-deficient mice display enhanced neointimal thickening following wire-induced arterial injury, robust VSMC proliferation in a murine vein graft model, and accelerated lesion formation in an atherogenic rodent model. Moreover, HO-1 may also regulate the vascular response to injury in humans since a long guanine thymidine dinucleotide repeat [(GT)n] microsatellite polymorphism in the human HO-1 promoter that is linked with impaired inducibility is associated with susceptibility to restenosis in patients undergoing percutaneous transluminal angioplasty or coronary artery stenting. Interestingly, we recently demonstrated that the same length polymorphism in the HO-1 promoter is associated with stenosis-related AVF failure in Chinese hemodialysis patients (4). In particular, we found that long GT repeats [(GT)n ≥ 30)] in the HO-1 promoter are associated with a higher frequency of access failure and poorer patency of AVFs. Based on these findings, we speculated that longer GT repeats in the HO-1 promoter might limit gene transcription and consequently offset the protective effect of HO-1 against vascular injury.
In this issue of Kidney International, Juncos and colleagues (5) directly demonstrate for the first time that HO-1 plays a critical role in promoting AVF survival. The authors took advantage of a recently developed murine AVF model (6) that recapitulates the salient features of dysfunctional AVFs in humans, including neointima formation and thrombosis, to show that HO-1 gene deletion exacerbates neointimal hyperplasia and AVF failure. In this model, an AVF is constructed by an end-to-end anastomosis between the mouse right common carotid artery and jugular vein. Robust induction of both HO-1 mRNA and protein is observed within one week in the venous wall of the fistula, predominantly by cells with phenotypic characteristics of VSMCs. Notably, AVFs remain fully patent for up to 3 weeks in all wild-type animals. In contrast, induction of HO-1 is not observed in the fistula of homozygous HO-1-deficient mice and the AVF patency rate declines by one-third in these animals. In addition, the impaired patency rate in HO-1-null animals is associated with more exuberant venous neointimal hyperplasia, as reflected by increases in venous wall thickening and decreases in luminal area/venous wall cross-section in HO-1-deficient mice compared to wild-type animals. Finally, the authors also examined the effect of HO-1 gene deletion on the expression of genes that may contribute to AVF failure, and found that the expression of plasminogen activator inhibitor -1 (PAI-1), monocyte chemoattractant protein-1 (MCP-1), and matrix metalloproteinase-2 and -9 are markedly elevated in AVFs of HO-1-deficient mice compared to wild-type animals.
These findings by Juncos et al. (5) are highly significant and set the stage for further studies exploring the role of HO-1 in AVF survival. The ability of HO-1 to suppress MCP-1 expression suggests that recruitment of inflammatory cells into the neointima may be enhanced in HO-1-deficient animals. Furthermore, the striking increase in PAI-1 expression in the fistula of HO-1-null mice may contribute to a procoagulant state in these animals. A comprehensive examination of the extent of leukocyte infiltration, fibrin formation, and platelet thrombosis in AVFs from both wild-type and HO-1-null mice would confirm whether HO-1 is exerting anti-inflammatory and antithrombotic effects in this setting. In addition, studies exploring how HO-1 is regulating cellular hyperplasia within the lesion would provide important mechanistic information. Is HO-1 influencing the rate of VSMC growth or apoptosis, or both? Alternatively, the capacity of HO-1 to inhibit gelatinase activity may also serve to reduce the number of VSMCs in the lesion by restricting their migration from the media to the intima. The synthesis of extracellular matrix components is another critical determinant of lesion size and may provide an additional pathway by which HO-1 regulates neointimal thickening. While loss of HO-1 clearly promotes AVF failure, it will be important to extend this study beyond three weeks and determine if overexpression of HO-1 will prolong the lifespan of AVFs in wild-type animals. Furthermore, elucidation of the kinetics as well as the stimulus for HO-1 induction in AVFs would be of interest. Determination of the reaction product(s) that mediate the protective action of HO-1 would also represent a logical extension of the present study. Finally, it will be essential to validate the beneficial actions of HO-1 on AVF stenosis and dysfunction in larger animals that utilize more relevant peripheral vessels.
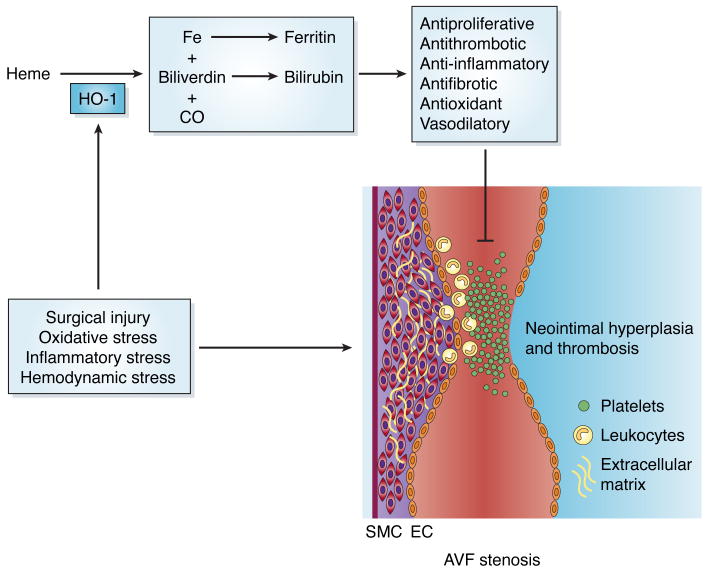
A model demonstrating the protective role of HO-1 in AFVs is shown in Figure 1. While the mechanism responsible for the induction of HO-1 in AVFs is not known, surgical injury and increases in oxidative and inflammatory stress that occur following the placement of a fistula are likely to contribute. In addition, changes in hemodynamics following the creation of AVFs may be particularly relevant since increases in venous flow will elevate fluid shear stress and circumferential wall strain, which are established inducers of HO-1 (7). The induction of HO-1 serves an important adaptive protective response in AVFs by generating a cascade of reaction products. HO-1 catalyzes the oxidative degradation of heme into equimolar amounts of carbon monoxide (CO), free iron, and biliverdin. Biliverdin is subsequently metabolized to bilirubin by biliverdin reductase and iron leads to the induction of ferritin. These HO-1 products are known to exert potent antiproliferative, antithrombotic, anti-inflammatory, antifibrotic, antioxidant, and vasodilatory actions that will mitigate AVF stenosis (3). Significantly, the local administration of biliverdin/bilirubin or CO protects against neointimal hyperplasia in other models of vascular injury (3,8).
Figure 1.
Model for the protective role of heme oxygenase-1 (HO-1) in arteriovenous fistula (AVF) failure. Several factors (surgical injury, oxidative stress, inflammatory stress, and hemodynamic stress) associated with the creation of an AVF leads to the induction of HO-1 in the fistula. HO-1 mediates the oxidative metabolism of heme to carbon monoxide (CO), iron (Fe), and biliverdin. Subsequently, biliverdin is converted to bilirubin, while iron stimulates ferritin expression. These HO-1 products possess potent antiproliferative, antithrombotic, anti-inflammatory, antifibrotic, antioxidant, and vasodilatory effects that limit AVF stenosis.
The study of Juncos and colleagues (5) provides a strong rationale for the development of therapeutic interventions that target HO-1 in AVF failure. Given their superficial position, the local application of pharmacological inducers of HO-1 to AVFs offers one promising approach. A large number of chemically diverse inducers of HO-1 have been identified and many of them are capable of inhibiting neointima formation in animal models of balloon angioplasty. Interestingly, some of the therapeutic approaches being developed to treat AVF failure may mediate their beneficial effect through the induction of HO-1. We recently demonstrated that local application of far infrared radiation improves blood flow and patency of AVFs as well as systemic inflammation in hemodialysis patients, and subsequently found that HO-1 mediates the anti-inflammatory effect of far infrared therapy (9,10). Similarly, rapamycin and paclitaxel, which have been shown to suppress luminal stenosis in different animal models of AVF failure, exert their antiproliferative effect on VSMCs via the induction of HO-1 (see 3,4). While these studies attest to the therapeutic potential of using HO-1 inducers, the GT length polymorphism in the HO-1 promoter may limit the utility of this approach in some patients. Increasing HO-1 expression via viral-mediated delivery of HO-1 circumvents this problem and may provide a more specific approach in raising HO-1 protein levels. Alternatively, the local delivery of the HO-1 products affords another potential therapeutic avenue for the treatment of AVF failure.
In conclusion, the study by Juncos and colleagues (5) is HOming in on AVF survival, and has identified HO-1 as a critical determinant of AVF function. Treatment modalities that locally target HO-1 or its reaction products to AVFs represent a promising new approach in overcoming the recalcitrant clinical problem of AVF stenosis.
Acknowledgments
The authors are supported, in part, by grants from NIH (HL59976 and HL749976), the Taipei Veterans General Hospital (V95-ER-003, V96-ER-B2-009), and the National Science Council of Taiwan (NSC95-2314-B-075-070, NSC96-2314-B-010-045).
References
- 1.Feldman HI, Kobrin S, Wasserstein A. Hemodialysis vascular access morbidity. J Am Soc Nephrol. 1996;7:523–535. doi: 10.1681/ASN.V74523. [DOI] [PubMed] [Google Scholar]
- 2.Roy-Chaudhury P, Lee TC. Vascular stenosis: biology and interventions. Curr Opin Nephrol Hypertens. 2007;16:516–522. doi: 10.1097/MNH.0b013e3282efa57f. [DOI] [PubMed] [Google Scholar]
- 3.Durante W, Johnson RK, Johnson RA. Role of carbon monoxide in cardiovascular function. J Cell Mol Med. 2006;10:672–686. doi: 10.1111/j.1582-4934.2006.tb00427.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lin C-C, Yang W-C, Lin SJ, et al. Length polymorphism in heme oxygenase-1 is associated with arteriovenous fistula patency in hemodialysis patients. Kidney Int. 2006;69:165–172. doi: 10.1038/sj.ki.5000019. [DOI] [PubMed] [Google Scholar]
- 5.Juncos JP, Tracz MJ, Croatt AJ, et al. Genetic deficiency of HO-1 markedly impairs the functionality and form of an arteriovenous fistula in the mouse. Kidney Int. 2007 doi: 10.1038/ki.2008.110. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Castier Y, Lehoux S, Hu Y, et al. Characterization of neointima lesions associated with arteriovenous fistulas in a mouse model. Kidney Int. 2006;70:315–320. doi: 10.1038/sj.ki.5001569. [DOI] [PubMed] [Google Scholar]
- 7.Wagner C, Durante W, Christodoulides N, Hellums JD, Schafer AI, et al. Hemodynamic forces induce the expression of heme oxygenase in cultured vascular smooth muscle cells. J Clin Invest. 1997;100:589–596. doi: 10.1172/JCI119569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ollinger R, Bilban M, Erat A, et al. Bilirubin: a natural inhibitor of vascular smooth muscle cell proliferation. Circulation. 2005;112:1030–1039. doi: 10.1161/CIRCULATIONAHA.104.528802. [DOI] [PubMed] [Google Scholar]
- 9.Lin C-C, Chang CF, Lai MY, et al. Far infrared therapy: a novel treatment improving access blood flow and unassisted patency of arteriovenous fistula in hemodialysis patients. J Am Soc Nephrol. 2007;18:985–992. doi: 10.1681/ASN.2006050534. [DOI] [PubMed] [Google Scholar]
- 10.Lin C-C, Liu X-M, Peyton KJ, et al. Far infrared therapy inhibits vascular endothelial inflammation via the induction of heme oxygenase-1. Arterioscler Thromb Vasc Biol. 2008 doi: 10.1161/ATVBAHA.107.160085. [DOI] [PMC free article] [PubMed] [Google Scholar]