Abstract
Chemical permeation enhancers (CPEs) have the potential to improve access of local anesthetics to the nerve, thereby improving nerve block performance. We assessed the effects of six CPEs on nerve blockade from tetrodotoxin (TTX) and from bupivacaine. Each of the six surfactants, representing three CPE sub-groups (anionic, cationic, and nonionic surfactants) was co-injected with TTX or bupivacaine at the sciatic nerve of Sprague-Dawley rats. Myotoxicity of CPEs, alone and with TTX, was assessed in vitro in C2C12 myotubes and in vivo via histological analysis. All enhancers produced marked concentration-dependent improvements in the frequency and duration of block with TTX but not bupivacaine. An in vitro toxicity assay showed a wide range of CPE myotoxicity, but in vivo histological assessment showed no signs of muscle or nerve damage at concentrations of CPEs that produced a half-maximal increase in the duration of block of TTX (except in the case of the cationic surfactant DDAB). This study demonstrates that CPEs can provide marked prolongation of nerve blockade from TTX but not bupivacaine, without apparent local tissue toxicity. These results may enhance the clinical applicability of TTX for prolonged-duration local anesthesia.
Keywords: Sciatic nerve blockade, Local anesthetic, Tetrodotoxin, Bupivacaine, Chemical permeation enhancer
Introduction
Local anesthetics must penetrate the epineurium, perineurium and endoneurium in order to reach their intended sight of action. Consequently, local anesthetics require much higher concentrations to be effective when used clinically than in isolated nerves [1–5]. Nerve block might therefore be enhanced by agents that increase small molecule flux across those biological barriers.
Chemical permeation enhancers (CPEs) have been used to increase the permeability of the lipid-protein barriers of the skin, and thereby increase drug flux, for over thirty years [6–9]. Surfactants, a heterogeneous group of amphiphilic organic molecules with hydrophilic heads and hydrophobic tails, are a well-known class of CPEs. Several sub-classes of surfactants (e.g., anionic, cationic, and nonionic) have been studied in the context of transdermal permeation, and are believed to reversibly modify lipids by adsorption at interfaces and removal of water-soluble agents that act as plasticizers [10, 11]. Cationic surfactants are known to produce greater increases in permeant flux than anionic surfactants, which, in turn, increase permeability more than nonionic surfactants [12–14]. Cationic surfactants, however, are generally more damaging to the skin [13]. A broad range of non-surfactant chemical enhancers has also been investigated (e.g., alcohols, sulfoxides, polyols, fatty acids, esters, terpenes, and cyclodextrins), with mechanisms of action that typically include denaturation of proteins within and between keratinocytes, and/or modification or disruption of lipids, resulting in increased lipid bilayer fluidity [10–15].
Here, we examine the hypothesis that CPEs will improve the quality of nerve blockade from local anesthetics. There are numerous compounds with known local anesthetic properties. Most of these have similar physicochemical characteristics and structures: an aromatic end, an ester or an amide linkage, and a tertiary amine [16]. We have selected one amino-amide from that group to test, bupivacaine, which is a partially neutral (pKa = 8.2), amphiphilic small molecule (288.43 g/mol) that binds the sodium channel on the cytosolic side of the axonal cell membrane. We also test a very different compound, tetrodotoxin (TTX), a very hydrophilic small molecule (318.28 g/mol) that blocks the sodium channel at site 1, on the outer surface of the neuron. It is a very potent local anesthetic, but does not cause the myo- and neurotoxicity associated with conventional local anesthetics. The potential enhancement of the nerve blockade with TTX is of particular interest to us because the principal limiting factor to its clinical use is its systemic toxicity [5, 17, 18]; it would be beneficial if CPEs reduced the TTX dose needed to provide a given duration of block. We also examine the effect of a range of CPEs differing in charge (cationic, anionic, and nonionic), and use cell culture and histology to assess the potential cytotoxicity and biocompatibility of the CPEs.
Materials & Methods
Animal Care
Young adult male Sprague-Dawley rats (350–420 g) were obtained from Charles River Laboratories (Wilmington, MA) and housed in groups of two per cage on a 6 a.m. to 6 p.m. light/dark cycle. All animals were cared for in accordance with protocols approved institutionally and nationally.
Chemical Enhancers & Solution Preparation
Representative enhancers from three different classes of surfactants were obtained from Sigma (St. Louis, MO): sodium lauryl sulfate (SLS) and sodium octyl sulfate (SOS), anionic surfactants; dodecyltrimethylammonium bromide (DDAB) and octyltrimethylammonium bromide (OTAB), cationic surfactants; and Polyoxyethylene (20) sorbitan monolaurate (T20) and Polyoxyethylene (20) sorbitan monooleate (T80), nonionic surfactants (Table 1). Tetrodotoxin (TTX) and bupivacaine (Sigma) solutions were prepared in saline individually and in combination with each enhancer the night before scheduled injections. TTX and bupivacaine concentrations were chosen to be near the bottom of their respective dose-response curves [19, 20]; enhancer concentrations were initially chosen to be approximately 50% of those used successfully in transdermal applications, followed by lower and higher concentrations as needed to obtain a dose-response curve.
Table 1.
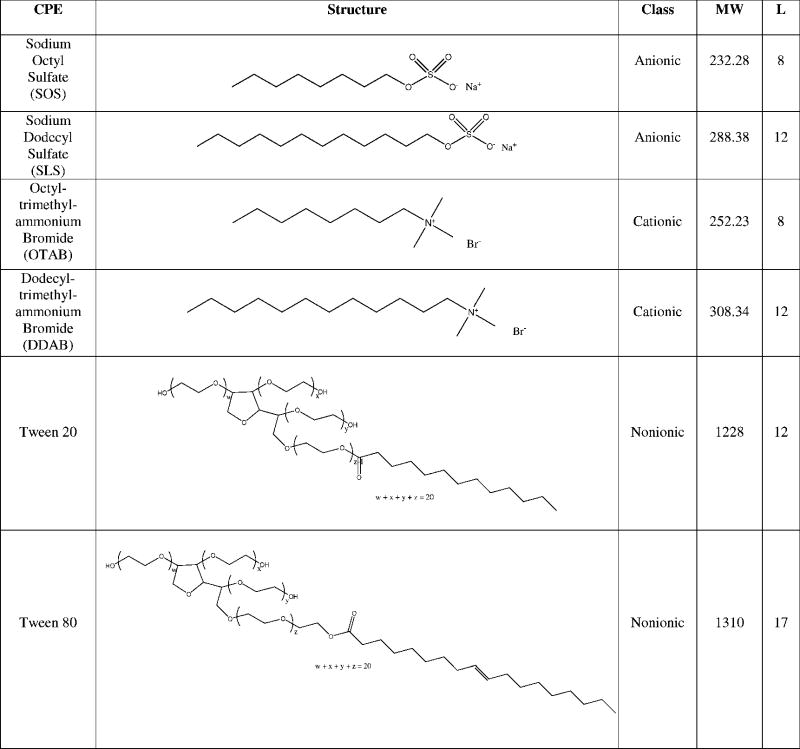
|
Sciatic Nerve Blockade Technique
Animals were cared for in compliance with protocols approved by the Massachusetts Institute of Technology (MIT) Committee on Animal Care, in conformity with the NIH guidelines for the care and use of laboratory animals (NIH publication #85-23, revised 1985). Rats were anesthetized using isoflurane in oxygen. A 23-gauge needle was introduced posteromedial to the greater trochanter, and 0.3 mL of the drug +/− enhancer solution was injected upon contacting bone, depositing the injectate over the sciatic nerve. To generate the dose-response curves for the CPEs, four animals were injected with each CPE at 5 different concentrations (each alone and with 30 μM TTX; see Figure 1). Larger sample sizes were obtained at the following concentrations that were important for further experiments: 11 mM SOS, 2 mM SLS, 9 mM SLS, 17 mM SLS, 35 mM SLS, 46 mM T80 (n = 8); 4 mM SLS (n = 16); and 23 mM T80 (n = 22). For CPEs injected with 1.39 mM bupivacaine, n = 4 for each CPE-bupivacaine combination. A total of 12 animals were injected with 1.39 mM bupivacaine alone (no CPE).
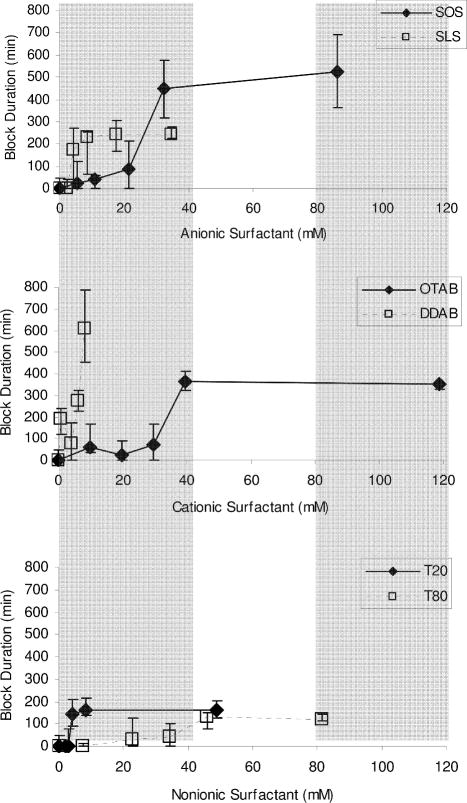
Figure 1.
Effect of CPE concentration on the duration of sensory block from 30 μm TTX. Durations are expressed as medians with 25th and 75th percentiles (n ≥ 4; see Methods). SOS = sodium octyl sulfate; SLS = sodium lauryl sulfate; OTAB = octyltrimethylammonium bromide; DDAB = dodecyltrimethylammonium bromide; T20 = polyoxyethylene (20) sorbitan monolaurate; T80 = polyoxyethylene (20) sorbitan monooleate.
The concentration of TTX used in this study (30 μM) was selected from a previously published [21] dose-response curve (concentration of TTX vs. duration of nerve blockade) so as to be very close to the lower inflection point of that curve, allowing for the most clear-cut observation of any improvement. The median duration of block was zero minutes. The concentration of bupivacaine was similarly determined from a dose-response curve performed here; the concentration used in this study (1.39 mM) was selected to be equieffective to the concentration of TTX, i.e. a median duration of block of zero, and close to the lower inflection point of its dose-response curve. We note that the concentrations of the local anesthetics used, though small, were not completely ineffective: the mean durations of block ± standard deviations from 30 μm TTX and 1.39 mM bupivacaine used were 27.92 ± 50.39 and 22.38 ± 34.90 minutes, respectively.
Assessment of Sciatic Nerve Blockade
In all experiments, the experimenter was blinded as to what treatment any given rat had received. Presence and extent of nerve blockade was investigated as previously described [5, 19, 20, 22, 23]. As described above, the test agents were injected at a relatively proximal location on the sciatic nerve near the greater trochanter. Testing of nerve blockade was performed at a distal site in dermatomes innervated by the sciatic nerve, the sole of the foot. Therefore, deficits in sensation there could be attributed to interruption of conduction along the sciatic nerve (nerve blockade), rather than local infiltration with drug. Thermal nociception of each leg was assessed in each hindpaw, with the right (uninjected) leg serving as an untreated control.
Thermal nociception was assessed by a modified hotplate test. Hind paws were exposed in sequence (left then right) to a 56°C hot plate (Model 39D Hot Plate Analgesia Meter, IITC Inc., Woodland Hills, CA). The time (latency) until paw withdrawal was measured with a stopwatch. If the animal did not remove its paw from the hot plate within 12 seconds, it was removed by the experimenter to avoid injury to the animal or the development of hyperalgesia. Testing was conducted at the following intervals after injection: 30 min, 60 min, then hourly four times, then every two hours. Injection occurred early in the morning and testing continued until nerve blockade resolved.
The duration of thermal nociceptive block was calculated as the time required for thermal latency to return to a value of seven seconds from a higher value. Seven seconds is the midpoint between a baseline thermal latency of approximately two seconds in adult rats, and a maximal latency of twelve seconds. Latencies longer than seven seconds were considered to be effective blocks.
As previously reported [5, 23], the experimenter demonstrated >99% successful blocks with 0.1–0.3 mL of 0.25%–0.5% (8.7–17 mM) bupivacaine, therefore suggesting that differences in block duration reflected actual pharmacological differences rather than operator error.
Tissue Harvesting and Histology
Animals were euthanized with carbon dioxide, and the sciatic nerves and adjacent tissues were harvested for histology. Tissues were fixed in Accustain (formalin-free fixative, Sigma), embedded in paraffin, sectioned, and stained with hematoxylin and eosin by the Department of Comparative Medicine at MIT (fee for service), using standard techniques. Tissues were all harvested at 4 days, as this is a time point at which local anesthetic myotoxicity and biomaterial-related inflammation can be seen.
Cell Culture
C2C12, a mouse myoblast cell line (American Type Culture Collection, ATCC, CRL-1772, Manassas, VA) was cultured to proliferate in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 20% Fetal Bovine Serum and 1% Penicillin Streptomycin (Pen Strep). All cell culture supplies were purchased from Invitrogen (Carlsbad, CA) unless otherwise noted. Cells were plated in 24-well tissue culture plates with 50,000 cells/mL/well in DMEM supplemented with 2% Horse Serum and 1% Pen Strep, and left to differentiate into myotubules for 10–14 days. During differentiation, media was exchanged every 2 to 3 days. After one week of myotube differentiation, 100 μL of the 10x enhancer +/μ TTX containing media was added to 900 μL of fresh media; 100 μL of PBS was added in control wells. The enhancer solution was prepared by dissolving the enhancer in PBS and stirring overnight. The solution was filtered aseptically using a 0.2 μm syringe filter. The TTX, or PBS for groups not containing drug, was added aseptically to the enhancer solution. At 2, 8, 24, or 96 hours the plates were assayed as described below, as these time points bracketed the time frame of the in vivo experiments. Cells were maintained at 37°C in 5% CO2 balance air. N = 4 in all cases.
Viability Assessment
Cell viability was assessed using a colorimetric assay (MTT kit, Promega G4100 Madison, WI) in which a yellow tetrazolium salt (MTT) is metabolized in live cells to form insoluble purple formazan crystals. The purple crystals are solubilized by the addition of a detergent, and the color is quantified spectrophotometrically. At each time point 150 μL of MTT reagent was added to the cells. Following a four hour incubation at 37 °C, 1 mL of solubilization solution (detergent) was added. The absorbance was read at 570 nm using the SpectraMax 384 Plus fluorometer (Molecular Devices, Sunnyvale, CA) after overnight incubation. Cells were also monitored visually to confirm the results of the MTT assay. Each plate contained media-only wells whose absorbance was subtracted from the rest of the plate as noise. All groups were then normalized to blank media.
Statistical Analysis
In vivo neurobehavioral data were not normally distributed, and are therefore presented as medians with 25th and 75th percentiles and compared by Mann-Whitney U-test. Cell survival assay results are described parametrically with means and standard deviations, and compared by t-tests and analysis of variance (ANOVA). Statistical significance, for both parametric and nonparametric tests, was defined as P < 0.05, and α = 0.05 in all cases. Statistical analyses were performed on MATLAB (MathWorks, Natick, MA).
Results
Effect of enhancers on nerve blockade with TTX
Injection of 0.3 mL of 30 μM TTX caused sensory blockade in 29% of animals tested (n = 24). The median duration of block was 0 minutes, with 25th and 75th percentiles of 0 minutes and 47 minutes, respectively. The selected concentration was based on previous observations [5] and chosen for these studies such that even small improvement of nerve blockade could easily be detected. Dose-response curves were obtained for the duration of block from 30 μM TTX with varying concentrations of SLS and SOS (anionic surfactants), DDAB and OTAB (cationic surfactants), and T20 and T80 (nonionic surfactants) individually (Table 1; Figure 1). The group that received DDAB was also injected with 30 μM TTX in 32 mM of the enhancer, but those data are not included in Figure 1 because none of those rats’ nerve blocks resolved (returned to normal function) within a 7-day observation period.
In most cases, the enhancers demonstrated a concentration-dependent increase in block duration before reaching a plateau. This maximal duration of block was used as a measure of efficacy for all CPEs except for DDAB, where we used the longest duration of block from which the animals recovered. There was a range of maximum durations of block due to the CPEs, the greatest being seen with DDAB (median maximum duration of block = 606 minutes; Figure 1, Table 2a). All the CPEs increased the percentage of animals that developed effective nerve block from 29.2% with TTX alone to 88–100% (Table 2b). At the concentrations needed to cause this maximum percentage of block, all CPEs significantly improved TTX block duration (for SOS, SLS, OTAB, DDAB, T20, and T80, p < 0.001 compared to the duration of block from TTX alone). In general, the maximum prolongation of TTX block by the cationic surfactants (OTAB, DDAB) was greater than prolongation by all other CPEs (p ≪ 0.05) except SOS (p > 0.10). The maximum prolongation of TTX block by the nonionic surfactants (T20, T80) was generally less than that by the others (p < 0.05 by Mann-Whitney U-test).
As a measure of the potency of the block-prolonging effect of the CPEs, we determined by interpolation the concentration of each enhancer required to achieve block lasting 100 minutes (the EC100min, Table 2a). T20, SLS, and DDAB were markedly more potent (lower EC100min) than T80, SOS, and OTAB (Figure 1; Table 2a). Within the classes of enhancers where the members differed only in the length of the carbon chain (SOS-SLS; OTAB-DDAB), those with the eight-carbon chain were generally less potent than those with the twelve-carbon chain. (In the case of the polysorbate compounds (T20, T80), that relationship was reversed, but there are other significant differences in their structures.) Each enhancer was also injected alone, without TTX, to confirm that the increased duration of block was not due to analgesic or toxic effects of the enhancers themselves; with one exception (32 mM DDAB), the CPE alone did not cause nerve blockade.
Effect of enhancers on nerve blockade with bupivacaine
We determined that 1.39 mM (0.04%) bupivacaine had performance characteristics comparable to 30 μm TTX with respect to percentage of animals blocked and median duration of block (Table 3) by producing a dose-response curve of sciatic nerve blocks with six bupivacaine concentrations spanning the range 0.69 mM – 6.9 mM, (n = 4–12 rats per concentration). We tested 1.39 mM bupivacaine with each of the CPEs at a concentration that created a half-maximal increase in the duration of block of TTX, the EC50eff (Table 2a). None of the 6 enhancers tested in these experiments resulted in a statistically significant change in the duration of bupivacaine nerve block (Table 3). This lack of enhancement of the duration of block by CPEs was also found at higher and more effective concentrations of bupivacaine: 2.1 mM (0.06%; n = 4) and 3.08 mM (0.1%; n = 8) bupivacaine gave median durations of block of 75 (45 – 90) min and 45 (0 – 60) minutes, respectively. Addition of the EC50eff of SLS to those concentrations of bupivacaine resulted in durations of block of 60 (45 – 60) (n = 4) and 30 (0 – 75) min (n = 4), which were not statistically significantly different from the results obtained without SLS.
In vitro toxicity
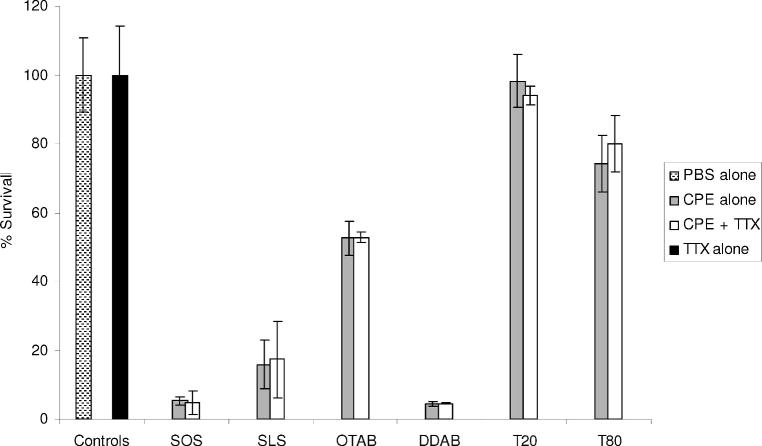
C2C12 myotube cultures were exposed to each enhancer at its EC50eff with and without TTX and assayed for viability after 2 hours (Figure 2). The most toxic enhancers were DDAB and SOS, followed by SLS, OTAB, T80, and T20, in order of decreasing toxicity. C2C12 viability decreased with increased duration of exposure to all CPEs except T20, which remained at untreated-control levels after an 8-hour exposure (data not shown). Addition of TTX to the cell culture medium did not impact cell survival when given alone or in the presence of enhancers.
Figure 2.
Survival of C2C12 myotubes after a 2-hour exposure to the EC50eff of each CPE alone and with 30 μM TTX, in vitro. Controls include cells exposed to PBS or TTX alone. Data are shown as means ± standard deviations (n = 4). SOS = sodium octyl sulfate; SLS = sodium lauryl sulfate; OTAB = octyltrimethylammonium bromide; DDAB = dodecyltrimethylammonium bromide; T20 = polyoxyethylene (20) sorbitan monolaurate; T80 = polyoxyethylene (20) sorbitan monooleate.
In vivo toxicity
The sciatic nerves and surrounding muscle of rats injected with the EC50eff of each enhancer (i.e., the same concentration used in vitro), with and without TTX, were examined for evidence of inflammation and tissue injury four days after injection (Figures 3A–F). Four animals were injected in each group.
Figure 3.
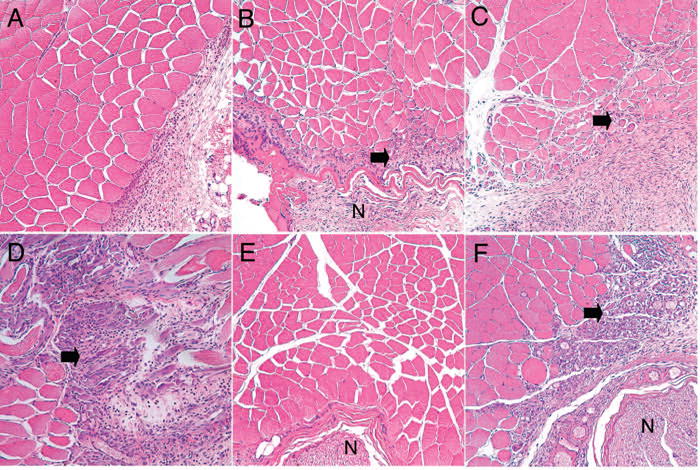
Histology of the site of injection CPEs at EC50eff with or without TTX 4 days after injection. (A) Skeletal muscle and nerve from regions injected with T20 (shown), SOS, SLS, OTAB or T80 at their EC50eff showed no evidence of injury to muscle or nerve. T20 at higher concentrations, of 21.4 mM (B) and 81.4 mM (C), led to increased inflammation with muscle fiber atrophy (arrows). DDAB at its EC50eff (D) showed surface inflammation similar to that seen with the higher T20 concentrations. Injections of T20 (shown, E), T80, SOS, SLS, and OTAB with TTX showed no evidence of injury to muscle or nerve. Injection of DDAB with TTX (F) showed findings similar to those seen with DDAB in the absence of TTX. N = nerve. Magnification = 200X.
Animals injected with the EC50eff of SOS, SLS, OTAB, T20 and T80 showed no significant muscle or nerve injury, although some samples in all groups showed mild inflammation with macrophages and lymphocytes around the muscle and nerve, without evidence of infiltration, fibrosis, or atrophy within the muscle or nerve (Figure 3A). Because T20 at its EC50eff showed no evidence of toxicity in vitro or in vivo, additional concentrations were tested to determine the highest sub-toxic concentration. T20 at 24.4 and 81.4 mM showed progressively worsening (mild to moderate) muscle atrophy and inflammation (Figures 3B–C), similar in type but not severity to that seen with the DDAB EC50eff (Figure 3D). (Note that 81.4 mM is more than twenty times the EC50eff of T20.) Samples from animals injected with DDAB consistently showed moderate to severe infiltration of macrophages and lymphocytes, atrophy and degeneration of muscle fibers, and fibrosis of the tissue (Figure 3D). An additional two animals were injected with 3% (97.3 mM) DDAB (the concentration at which animals developed irreversible nerve block). These showed deep and severe tissue damage, including ischemic necrosis, accompanied by severe and extensive inflammation.
Animals injected with the EC50eff of the enhancers together with 30 μm TTX showed the same histological results as those without TTX (Figures 3E–F). Again, some of the samples exposed to DDAB showed severe lymphocytic inflammatory infiltration of muscle with degenerative changes, regenerative changes, and fibrosis (Figure 3F). These samples also showed a mild lymphocytic infiltrate of nerve and focal fat necrosis.
Indicators of nerve fiber injury, including fibrosis and myelin ovoids, were not seen in any samples, but subtle degrees of damage to myelinated nerve fibers cannot be accurately assessed using paraffin-embedded, hematoxylin-eosin-stained sections.
Discussion
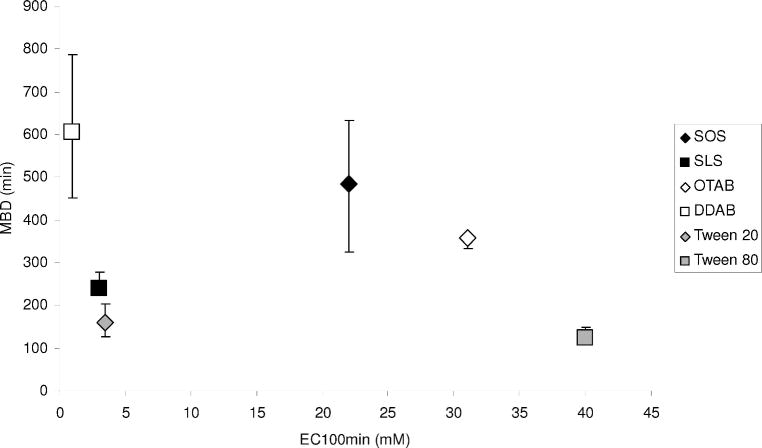
All the surfactant CPEs examined caused a marked concentration-dependent prolongation of TTX block, presumably by increasing penetration to the nerve surface (TTX acts on the cell surface). Though there was a considerable range in the magnitude of enhancement, no individual CPE or class of CPE (anionic, cationic, or nonionic surfactant) clearly performed better than all the others. In general, the magnitude of the maximal improvement in duration of block (the maximum block duration, MBD) did not correlate well with the potency (EC100min, Figure 4). The nonionic agents’ block prolongations, though significant, were shorter than those of the other CPEs. This is consistent with the effects of surfactants on permeant flux across the stratum corneum and epidermis of the skin [13]. However, it is important to be careful in making comparative statements regarding potency, since the shapes of the dose-response curves for each CPE are not always similar. In contrast to their marked enhancement of TTX-induced nerve blockade, CPEs had no effect on the duration of block of a concentration of bupivacaine that was equieffective to the TTX concentration enhanced by CPEs. One CPE also had no effect on two higher and more effective concentrations of bupivacaine.
Figure 4.
Maximum block duration (MBD) plotted against interpolated EC100min values for each CPE indicate no correlation (R2 = 0.08). Anionic, cationic, and nonionic surfactants are grouped by color (black, grey, and white, respectively). Block durations are expressed as medians with 25th and 75th percentiles (n ≥ 4; see Methods).
CPEs varied widely in the cyotoxicity of their EC50eff. In general, the agents that produced the longest maximal block durations were more toxic in cell culture (Figure 5, R2 = 0.66). There was no correlation between the EC100min and toxicity (R2 = 0.11). With the cationic surfactants, toxicity increased with molecular weight and carbon-chain length, while it decreased with the same parameters in anionic surfactants.
Figure 5.
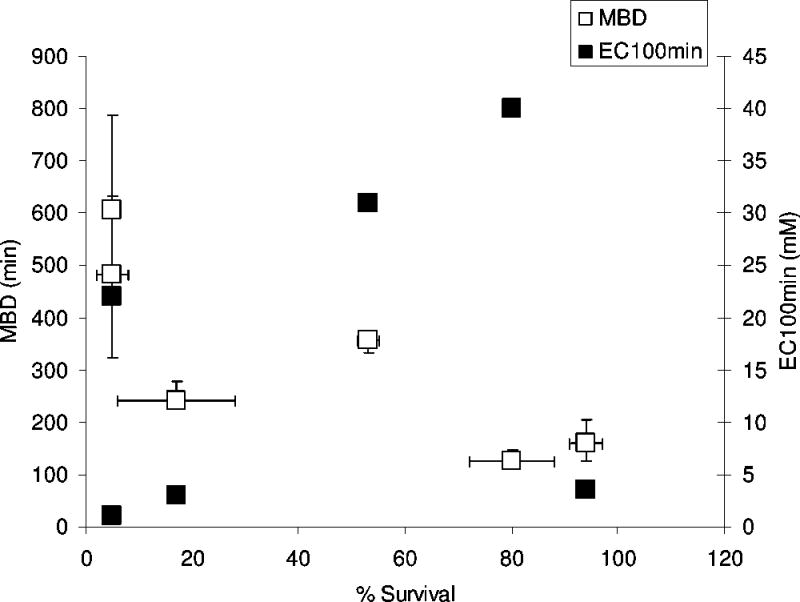
Maximum block duration (MBD) and EC100min plotted against cell survival. The EC50eff is the CPE concentration that caused a half-maximal increase in block duration. Cell survival data are means with standard deviations from C2C12 cells exposed to each CPE at its EC50eff for 2 hours from Figure 2). Block durations are expressed as medians with 25th and 75th percentiles.
In assessing the trade-off between maximum block duration and cytotoxicity (Table 4), T20 would appear to have the most favorable relevant ratios. However, this does not mean that T20 would be the optimal enhancer for clinical use. The in vivo data showed that all compounds, with the notable exception of DDAB, caused little or no tissue injury when delivered at the same concentrations as used in vitro (the EC50eff, which had caused approximately half-maximal increase in duration of block from TTX). This discrepancy between in vitro and in vivo results is not surprising. It may be explained by differences between cultured cell lines and in vivo tissue, but it is also possible that the local concentration of the CPEs dissipates rapidly after injection in vivo. We have seen a similar discrepancy with conventional local anesthetics [19]. Tetrodotoxin itself caused little or no toxicity, with or without enhancers, a finding consistent with prior experience [19]. The in vivo results suggest that enhancer toxicity can be minimal or non-existent within a concentration range that results in significant block duration, and that the most efficacious compounds could be used rather than those with the best toxicity profile from in vitro studies. There was little or no evidence of direct nerve injury in all the CPEs investigated, including concentrations of DDAB that resulted in long-term loss of nerve function. We note, however, that the use of paraffin-embedded tissue is relatively insensitive for the evaluation of damage to myelinated fibers. This study did not determine the ideal dose of TTX and enhancer to provide the longest safe nerve block.
TTX-based local anesthetics could potentially offer substantial advantages over conventional local anesthetics. TTX does not cause local neurotoxicity [21], unlike the well-documented neurotoxicity of conventional local anesthetics [24]; neither does it cause myotoxicity, which is a common feature of conventional local anesthetics [25]. It is also less likely to produce the seizures and/or cardiac arrest resistant to resuscitative measures that are seen with conventional local anesthetics [26–29]. The principal obstacle to the clinical use of TTX is its potential for profound systemic toxicity [17] due to systemic distribution, particularly to the phrenic nerves, leading to respiratory paralysis. In these experiments, CPEs dramatically increased the median duration of block from a very low concentration of TTX (e.g. from 0 to 353 minutes by use of OTAB). These durations of block far exceed those that could be achieved even by toxic, near-lethal concentrations of TTX in the absence of vasoconstrictors. For example, 50 μm TTX applied in the same manner without CPEs resulted in an average duration of block of approximately 150 minutes, but with a 20% mortality rate [22]. Although we have not demonstrated this formally, it would seem to follow from these facts that the use of CPEs would result in a marked improvement in the therapeutic index of TTX (the ratio of the effective to the lethal dose).
This study was not designed to address the mechanism by which the CPEs enhanced the duration. However, it is possible that the difference in the physicochemical properties between TTX and bupivacaine can explain the difference in the effect of CPEs on nerve blockade from the two compounds. TTX is extremely hydrophilic, having an obligate charge, while bupivacaine – like all amino-ester and amino-amide local anesthetics – can be conditionally hydrophobic due to its aromatic moiety and tertiary amine. There is a pH-dependent equilibrium between the cationic protonated form of bupivacaine that is water soluble and the neutral form that is soluble in organic solvents (i.e., is hydrophobic), and therefore partitions relatively easily into cell membranes and other biological barriers. We speculate that the relatively pronounced improvement in block from TTX with CPEs relates to its lack of hydrophobicity, whereas bupivacaine does not benefit because its structure already permits easy crossing of biological barriers. We have previously described that high (millimolar) concentrations of adrenergic antagonists, far in excess of the range in which they are active on adrenergic receptors, greatly prolonged the duration of block by TTX [20]. The results presented here support the view that the prolongation of nerve block by those polycyclic compounds was due to flux enhancement (i.e. those compounds were acting as CPEs). This study did not reveal a consistent pattern in the effect of hydrophobic chain length on duration of block.
Acknowledgments
Financial support: GM073626 (to DSK) from NIGMS (National Institute of General Medical Sciences); T32/DC000038 (to EJS) from NIH/NIDCD (National Institute of Deafness and Other Communication Disorders).
Table of Abbreviations
- DDAB
dodecyltrimethylammonium bromide
- EC100min
concentration of enhancer that prolongs the block from 30 μM TTX to 100 minutes
- EC50eff
concentration of enhancer that produces 50% of the maximum prolongation of block achieved with that enhancer with 30 μm TTX
- OTAB
octyltrimethylammonium bromide
- SLS
sodium lauryl sulfate (AKA sodium dodecyl sulfate)
- SOS
sodium octyl sulfate
- T20
polyoxyethylene (20) sorbitan monolaurate (polysorbate 20)
- T80
polyoxyethylene (20) sorbitan monooleate (polysorbate 80)
- TTX
tetrodotoxin
References
- 1.Schwartz JR, Ulbricht W, Wanger HH. The rate of action of tetrodotoxin on myelinated nerve fibers of Xenopus laevis and Rana esculenta. J Physiol (Lond) 1973;233:167–194. doi: 10.1113/jphysiol.1973.sp010304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hahin R, Strichartz G. Effects of deuterium oxide on the rate and dissociation constants for saxitoxin and tetrodotoxin action. Voltage-clamp studies on frog myelinated nerve. J Gen Physiol. 1981;78(2):113–139. doi: 10.1085/jgp.78.2.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chernoff DM, Strichartz GR. Kinetics of local anesthetic inhibition of neuronal sodium currents. pH and hydrophobicity dependence. Biophys J. 1990;58(1):69–81. doi: 10.1016/S0006-3495(90)82354-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lee-Son S, Wang GK, Concus A, Crill E, Strichartz G. Stereoselective inhibition of neuronal sodium channels by local anesthetics. Evidence for two sites of action? Anesthesiology. 1992;77(2):324–335. doi: 10.1097/00000542-199208000-00016. [DOI] [PubMed] [Google Scholar]
- 5.Kohane DS, Yieh J, Lu NT, Langer R, Strichartz GR, Berde CB. A re-examination of tetrodotoxin for prolonged duration local anesthesia. Anesthesiology. 1998;89(1):119–131. doi: 10.1097/00000542-199807000-00019. [DOI] [PubMed] [Google Scholar]
- 6.Bauerova K, Matusova D, Kassai Z. Chemical enhancers for transdermal drug transport. Eur J Drug Metab Pharmacokinet. 2001;26(1–2):85–94. doi: 10.1007/BF03190381. [DOI] [PubMed] [Google Scholar]
- 7.Asbill CS, El-Kattan AF, Michniak B. Enhancement of transdermal drug delivery: chemical and physical approaches. Crit Rev Ther Drug Carrier Syst. 2000;17(6):621–658. [PubMed] [Google Scholar]
- 8.Kanikkannan N, Kandimalla K, Lamba SS, Singh M. Structure-activity relationship of chemical penetration enhancers in transdermal drug delivery. Curr Med Chem. 2000;7(6):593–608. doi: 10.2174/0929867003374840. [DOI] [PubMed] [Google Scholar]
- 9.Karande P, Jain A, Mitragotri S. Relationships between skin’s electrical impedance and permeability in the presence of chemical enhancers. J Control Release. 2006;110(2):307–313. doi: 10.1016/j.jconrel.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 10.Middleton JD. Mechanism of action of surfactants on water binding properties of isolated stratum corneum. J Soc Cosmet Chem. 1969;20:399–403. [Google Scholar]
- 11.Ribaud C, Garson JC, Doucet J, Leveque JL. Organization of stratum corneum lipids in relation to permeability: influence of sodium lauryl sulfate and preheating. Pharm Res. 1994;11(10):1414–1418. doi: 10.1023/a:1018987721531. [DOI] [PubMed] [Google Scholar]
- 12.Farber EM. Psoriasis. Grune and Stratton; Orlando: 1982. [Google Scholar]
- 13.Kushla GP, Zatz JL, Mills OH, Jr, Berger RS. Noninvasive assessment of anesthetic activity of topical lidocaine formulations. J Pharm Sci. 1993;82(11):1118–1122. doi: 10.1002/jps.2600821110. [DOI] [PubMed] [Google Scholar]
- 14.Shen WW, Danti AG, Bruscato FN. Effect of nonionic surfactants on percutaneous absorption of salicylic acid and sodium salicylate in the presence of dimethyl sulfoxide. J Pharm Sci. 1976;65(12):1780–1783. doi: 10.1002/jps.2600651222. [DOI] [PubMed] [Google Scholar]
- 15.Walker RB, Smith EW. The role of percutaneous penetration enhancers. Adv Drug Delivery Rev. 1996;18:295–301. [Google Scholar]
- 16.Stoelting RK, Miller RD. Local anesthetics: Basics of anesthesia. Churchill Livingstone, Inc; New York: 1994. [Google Scholar]
- 17.Kao CY. Tetrodotoxin, saxitoxin, and their significance in the study of excitation phenomena. Pharmacological Reviews. 1966;18:997–1049. [PubMed] [Google Scholar]
- 18.Kohane DS, Lu NT, Gokgol-Kline AC, Shubina M, Kuang Y, Hall S, Strichartz GR, Berde CB. The local anesthetic properties and toxicity of saxitonin homologues for rat sciatic nerve block in vivo. Reg Anesth Pain Med. 2000;25(1):52–59. doi: 10.1016/s1098-7339(00)80011-5. [DOI] [PubMed] [Google Scholar]
- 19.Kohane DS, Sankar WN, Shubina M, Hu D, Rifai N, Berde CB. Sciatic nerve blockade in infant, adolescent, and adult rats: a comparison of ropivacaine with bupivacaine. Anesthesiology. 1998;89(5):1199–1208. doi: 10.1097/00000542-199811000-00021. discussion 1110A. [DOI] [PubMed] [Google Scholar]
- 20.Kohane DS, Lu NT, Cairns BE, Berde CB. Effects of adrenergic agonists and antagonists on tetrodotoxin-induced nerve block. Reg Anesth Pain Med. 2001;26(3):239–245. doi: 10.1053/rapm.2001.23215. [DOI] [PubMed] [Google Scholar]
- 21.Sakura S, Bollen AW, Ciriales R, Drasner K. Local anesthetic neurotoxicity does not result from blockade of voltage-gated sodium channels. Anesth Analg. 1995;81(2):338–346. doi: 10.1097/00000539-199508000-00023. [DOI] [PubMed] [Google Scholar]
- 22.Padera RF, Tse JY, Bellas E, Kohane DS. Tetrodotoxin for prolonged local anesthesia with minimal myotoxicity. Muscle Nerve. 2006;34(6):747–753. doi: 10.1002/mus.20618. [DOI] [PubMed] [Google Scholar]
- 23.Masters DB, Berde CB, Dutta SK, Griggs CT, Hu D, Kupsky W, Langer R. Prolonged regional nerve blockade by controlled release of local anesthetic from a biodegradable polymer matrix. Anesthesiology. 1993;79(2):340–346. doi: 10.1097/00000542-199308000-00020. [DOI] [PubMed] [Google Scholar]
- 24.Bainton CR, Strichartz GR. Concentration dependence of lidocaine-induced irreversible conduction loss in frog nerve. Anesthesiology. 1994;91:657–667. doi: 10.1097/00000542-199409000-00020. [DOI] [PubMed] [Google Scholar]
- 25.Benoit PW, Yagiela A, Fort NF. Pharmacologic correlation between local anesthetic-induced myotoxicity and disturbances of intracellular calcium distribution. Toxicol Appl Pharmacol. 1980;52(2):187–198. doi: 10.1016/0041-008x(80)90105-2. [DOI] [PubMed] [Google Scholar]
- 26.Strichartz GR, Berde CB. Local Anesthetics, Anesthesia. Churchill Livingstone, Inc; New York: 1994. [Google Scholar]
- 27.Albright GA. Cardiac arrest following regional anesthesia with etidocaine or bupivacaine. Anesthesiology. 1979;51:285–287. doi: 10.1097/00000542-197910000-00001. [DOI] [PubMed] [Google Scholar]
- 28.Feldman HS, Arthur GR, Covino BG. Comparative systemic toxicity of convulsant and supraconvulsant doses of intravenous ropivacaine, bupivacaine, and lidocaine in the conscious dog. Anesth Analg. 1989;69:794–801. [PubMed] [Google Scholar]
- 29.Feldman HS, Arthur GR, Pitkanen M, Hurley R, Doucette AM, Covino BG. Treatment of acute systemic toxicity after the rapid intravenous injection of ropivacaine and bupivacaine in the conscious dog. Anesth Analg. 1991;73:373–384. [PubMed] [Google Scholar]