Abstract
Objective
To reconstruct the historical changes in force of dengue infection in Singapore, and to better understand the relationship between control of Aedes mosquitoes and incidence of classic dengue fever.
Methods
Seroprevalence data were abstracted from surveys performed in Singapore from 1982 to 2002. These data were used to develop two mathematical models of age seroprevalence. In the first model, force of infection was allowed to vary independently each year, while in the second it was described by a polynomial function. Model-predicted temporal trends were analysed using linear regression. Time series techniques were employed to investigate periodicity in predicted forces of infection, dengue fever incidence and mosquito breeding.
Findings
Force of infection estimates showed a significant downward trend from 1966, when vector control was instigated. Force of infection estimates from both models reproduced significant increases in the percentage and average age of the population susceptible to dengue infections. Importantly, the year-on-year model independently predicted a five to six year periodicity that was also displayed by clinical incidence but absent from the Aedes household index.
Conclusion
We propose that the rise in disease incidence was due in part to a vector-control-driven reduction in herd immunity in older age groups that are more susceptible to developing clinical dengue.
Résumé
Objectif
Reconstituer les variations historiques à Singapour de la virulence de l’infection par la dengue et mieux comprendre la relation entre la lutte contre les moustiques Aedes et l’incidence de la dengue classique.
Méthodes
Des données de séroprévalence ont été extraites des enquêtes réalisées à Singapour entre 1982 et 2002. Ces données ont servi à développer deux modèles mathématiques de la séroprévalence en fonction de l’âge. Dans le premier modèle, on a laissé la virulence de l’infection varier chaque année de manière indépendante, tandis que, dans le second, cette virulence était décrite par une fonction polynomiale. On a analysé par régression linéaire les tendances temporelles prédites par les modèles. On a étudié la périodicité des previsions de la virulence de l’infection, de l’incidence de la dengue et de la reproduction des moustiques par des techniques utilisant des séries temporelles.
Résultats
La virulence estimée de l’infection présente une tendance à la baisse significative depuis 1966, année de lancement de la lutte antivectorielle. Les estimations de cette virulence fournies par les deux modèles reflètent des augmentations importantes du pourcentage et de l’âge moyen de la population susceptible de contracter la dengue. Point important : la modélisation d’année en année a prédit de manière indépendante une périodicité de cinq à six ans, que présente également l’incidence clinique, mais pas l’indice « maison » du moustique Aedes.
Conclusion
Notre interprétation est que l’augmentation de l’incidence de la maladie est en partie due à une baisse de l’immunité collective induite par la lutte antivectorielle dans les tranches d’âges supérieures, plus sensibles au développement de la dengue clinique.
Resumen
Objetivo
Determinar la evolución de la virulencia del dengue en Singapur, y profundizar en la relación entre el control del mosquito Aedes y la incidencia de la fiebre del dengue clásica.
Métodos
A partir de estudios realizados en Singapur entre 1982 y 2002, se reunieron datos de seroprevalencia que se utilizaron para desarrollar dos modelos matemáticos de seroprevalencia por edades. En el primer modelo se permitió que la virulencia de la infección variara de forma independiente cada año, mientras que en el segundo se describió mediante una función polinómica. Las tendencias temporales previstas por el modelo se analizaron mediante métodos de regresión lineal. Se emplearon técnicas de series temporales para investigar la periodicidad de la virulencia prevista de la infección, la incidencia de fiebre del dengue y la reproducción de los mosquitos.
Resultados
Las estimaciones de la virulencia de la infección mostraron una tendencia descendente considerable desde 1966, año en que se empezó a fomentar la lucha antivectorial. Las estimaciones de la virulencia de la infección obtenidas con los dos modelos reprodujeron aumentos importantes del porcentaje y la edad promedio de la población vulnerable al dengue. Un hallazgo importante es que el modelo de año en año predijo de forma independiente una periodicidad de cinco a seis años observable también en la incidencia clínica, pero no así en el índice de Aedes en los hogares.
Conclusión
El aumento de la incidencia de la enfermedad puede deberse en parte a una reducción, propiciada por la lucha antivectorial, de la inmunidad colectiva en los grupos de personas de edad avanzada que tienen mayor tendencia a desarrollar dengue clínico.
ملخص
الغرض
إعادة بناء التغيُّرات التاريخية في قوى العدوى بحمى الضنك في سنغافورة، والوصول إلى فەم أفضل للعلاقة بين مكافحة بعوض الزواعج ومعدل حدوث حمى الضنك المعەودة.
الطريقة
استخلصت المعطيـات حول الانتشار المصلي من المسوحات التي أجريت في سنغافورة في الفترة بين 1982 و2002، واستخدمت ەذە المعطيات لإعداد نموذجَيْن رياضيَـيْن للانتشار المصلي والعمري. وقد سمح لقوى العدوى في النموذج الأول بأن تخضع لاختلافات مستقلة كل عام، فيما وصفت في النموذج الثاني بأنەا وظيفة متعدِّدة الحدود. ثم حلِّلت الاتجاەات الزمنية وفق التوقعات الخاصة للنموذج باستخدام التحوُّف الخطي، كما استخدم أسلوب السلاسل الزمنية لتقصِّي الدورية في القوى المتوقعة للعدوى، وفي معدل حدوث حمى الضنك وفي تفقيس البعوض.
الموجودات
أظەرت تقديرات الاتجاە لقوى العدوى انحداراً يعتد بە إحصائياً بدءاً من عام 1966، عندما بدئت مكافحة نواقل المرض. وقد أعادت تقديرات قوى العدوى المستمدَّة من كلا النموذجين إحداث زيادات يعتد بەا إحصائياً في النسبة المئوية وفي الأعمار الوسطية للسكان الذين لديەم الاستعداد للعدوى بحمى الضنك. والأمر الەام والجديـر بالذكر أن النموذج الـمُرْتَكِز على الانتقال من سنة لأخرى توقّع بشكل مستقل عن غيرە دوريـة تستغرق 5 – 6 سنوات، وقد اتَّضح ذلك بالمعدلات السريرية للانتشار مع غياب مؤشِّر الزواعج لدى السكان.
الاستنتاج
نفترض أن الازدياد في معدلات حدوث المرض نجم جزئياً عن جەود مكافحة النواقل التي أدَّت إلى نقص في مناعة القطيع لدى مجموعات الأعمار الأعلى والتي تعد أكثر استعداداً للإصابة بحمى الضنك السريرية.
Introduction
Dengue fever is a viral infection transmitted by Aedes mosquitoes that has recently re-emerged globally as the most important arboviral disease.1 There are four antigenically distinct dengue virus serotypes (DEN 1–4) that induce permanent serotype-specific, IgG antibody-mediated protective immunity following first infection.2 Dengue fever presents as a spectrum of increasingly severe clinical manifestations ranging from classic dengue fever to dengue haemorrhagic fever to dengue shock syndrome,2 although the distinction between these conditions is often blurred.3 In an endemic situation, the majority of dengue infections are subclinical and the risk factors for severity of clinical outcome include age, viral strain, host genetics and time between heterotypic infection.4–6 Classic dengue fever is most commonly associated with primary viral infection, and in fully naive individuals the probability of developing clinical disease increases with age.5 Early studies in the Philippines demonstrated that the risk of classic disease was very high in young adults following primary infection,7,8 while more recent findings in Indonesia and Thailand indicate that most classic illness in children is the result of secondary infection.9,10 Due to the complex set of factors that contribute to risk of dengue haemorrhagic fever, as well as the extremely low incidence of dengue haemorrhagic fever in Singapore, this analysis has limited its scope to classic dengue illness.
Ae. aegypti, the primary vector for dengue fever, is well adapted to breeding in human-made breeding sites in urban and periurban environments.11 Dengue is now endemic in over 100 countries, with a dramatic increase in incidence and geographical range recorded in recent years. Reasons for this increase include growing levels of urbanization, international trade and travel disseminating both the vector and viruses, and the inadequacies of current methods to reduce dengue transmission.12
Singapore is one of the few settings that have recorded sustained suppression of the vector population. The dengue control programme combines all WHO-recommended control activities, including public health education and community participation, active breeding site detection, environmental management, reactive insecticide fogging, and geo-referenced entomologic and clinical surveillance systems.13 Since the first legislation to enforce vector control was introduced in 1966, Singapore has seen the Aedes household index (the percentage of all properties with breeding sites of Aedes mosquitoes) reduced from over 50% to less than 1% (Fig. 1).14 However, a contradictory phenomenon is occurring in Singapore, whereby the incidence of dengue fever has recently increased despite the success of the vector control programme. Notably, the overwhelming majority of cases in recent years have been as classic dengue fever, with dengue haemorrhagic fever representing less than 1% of the 21 000 officially reported cases between 2000 and 2004.15,16
Fig. 1.
Observed annual average Aedes household index and annual clinical incidence of dengue fever
a 2005 household index estimate is not yet available.
Intuitively, it would be expected that a decrease in the mosquito population would lower the force of infection (the per capita rate at which susceptible individuals acquire infection) and consequently decrease disease incidence. While the Aedes household index and the observed increases in average age of clinical dengue17 (Fig. 2) are consistent with a decrease in the force of infection, the disease incidence continues to climb. The number of confirmed dengue cases climbed from a record level of 9292 in 2004 to a new high of over 13 800 in 2005 (Fig. 1).18 Determining the temporal pattern of force of infection is essential to understanding the extent to which vector control in Singapore has reduced the intensity of dengue transmission, thus helping to clarify the unprecedented rise in disease incidence.
Fig. 2.
Observed average age of clinical cases of dengue fever reported in Singaporea
a National dengue control activities were instigated in 1966.
Force of infection has been used widely to understand the intensity of disease transmission within a community.19–23 In an endemic situation, the force of infection can be approximated by the reciprocal of the average age of infection.19 However, the force of infection has likely been in a dynamic state in Singapore due to the long-term programme to reduce Aedes mosquitoes. While estimates of force of dengue infection have never been published for Singapore, seroprevalence surveys have been performed on an ad hoc basis for several decades. These data provide a historical record of the percentage of the population that have ever been infected by dengue virus. Here we develop a mathematical model that allows the changes in force of dengue infection in Singapore to be reconstructed from these published, age-stratified seroprevalence data. The modelling procedures and resulting insights have major implications for routine surveillance activities, the long-term monitoring of control activities and the choice of strategies aimed at controlling dengue fever, not only in Singapore but across all endemic settings.
Materials and methods
Age-stratified seroprevalence data
Five age-stratified seroprevalence surveys of dengue IgG antibodies conducted in Singapore were identified in the literature. These surveys, conducted in 1982, 1991, 1993, 1999 and 2002, included serosamples from a total of 3954 individuals (Table 1).17,24,25 The studies reported the number sampled and number seropositive in a variety of different age categories. For model fitting purposes, the midpoint age (to the nearest half year) for each category was used.
Table 1. Summary of deviance and percentage deviance explained by models.
| Survey | Number sampleda | Total deviance | Year-on-year modelb | Polynomial modelb |
|---|---|---|---|---|
| 1982 | 425 (8) | 181.3 | 13.3 (92.66%) | 18.4 (89.84%) |
| 1991 | 1409 (11) | 531.4 | 25.8 (95.14%) | 29.6 (94.43%) |
| 1993 | 912 (7) | 344.9 | 17.5 (94.92%) | 18.1 (94.74%) |
| 1999 | 901 (16) | 297.2 | 24.5 (91.74%) | 26.4 (91.10%) |
| 2002 | 298 (3) | 48.2 | 7.2 (85.15%) | 7.5 (84.36%) |
| Overall | 3945 (45) | 1403.1 | 88.3 (93.70%) | 100.1 (92.86%) |
a Number of age classes presented in parentheses. b Percentage explained presented in parentheses.
The surveys performed in 1982, 1991 and 1993 used the hemagglutination-inhibition procedure.26 All three surveys used dengue serotype 2 virus as the antigen; however, the hemagglutination-inhibition test is broadly cross-reactive and would detect antibodies to all four serotypes of dengue virus.27 For the 1982 survey, a titre of < 8 was considered negative, while in 1991 and 1993 a titre of < 10 was considered negative. The surveys performed in 1999 and 2002 both used the PanBio (PanBio, Brisbane, Australia) dengue IgG-ELISA test kit. Similar to the hemagglutination-inhibition test, the IgG-ELISA procedure does not distinguish viral serotype, and seroprevalence from these surveys is based on infection with any of the four dengue serotypes. For the purposes of this paper, the relative difference in seroprevalence between age classes, not absolute seroprevalence, is important. Therefore, while the hemagglutination-inhibition and IgG-ELISA tests may have relatively low specificity and have likely underestimated seroprevalence in Singapore, there is no evidence that specificity for either test is age-dependent so this should not significantly affect the proportional increase in seroprevalence between age classes.
Epidemiological and entomologic data
Official records of annual classic dengue fever incidence from 1960 (when official records began) to 2002 (the latest serosurvey year), and the Aedes household index collated between 1966 (when records began) and 2002, were obtained from the appropriate Singapore government departments (Fig. 1). It is worth noting most dengue cases in Singapore acquire the infection locally, rather than through imported cases.28
The average age of dengue fever cases was calculated from the number of cases recorded in seven age categories (0–4, 5–14, 15–24, 25–34, 35–44, 45–54 and ≥ 55 years) for each year from 1975, when age-stratified records were first available, to 2002. The best estimate of the average age of clinical disease was calculated using the mid-age of each category, assuming 65 years for the oldest age class.
The basic model
The model population age structure was divided into 0.5 yearly increments. The age-specific seroprevalence, ia, in year t was described by
 |
for individuals aged 0.5 years, assuming there are no remaining maternal antibodies by six months of age, and
for all other ages ≥ 1 year, in which a is the age years and λt is the age-independent force of infection in year t. To illustrate this model, consider a serologic survey conducted in the year 2000. The observed difference in mean seroprevalence from this survey between five-year-olds (i.e. born in 1995) and six-year-olds (i.e. born in 1994) is a measure of the force of infection between 1994 and 1995. Our model assumes the seroresponse following first infection is lifelong.9 The model further assumes an endemic situation in 1960 with the age-specific seroprevalence profile in that year given by
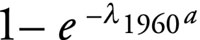 . .
|
Fitting the year-on-year model
The force of infection in each year from 1960 to 2002 was varied to maximize the likelihood between the observed and model-predicted age seroprevalence.29 The time period over which the force of infection was allowed to fluctuate was chosen as clinical incidence measures are only available from 1960 and the latest seroprevalence survey was conducted in 2002. The significance of this full year-on-year model (with 43 independent estimates of λt from t = 1960 to t = 2002) was assessed by comparing the model deviance with total deviance, using the likelihood ratio test.30 Model deviance was calculated using model-estimated seroprevalence for each age group, whereas total deviance was calculated by fitting the overall mean seroprevalence across all ages in all surveys.
Fitting the polynomial model
A simplified model was also fitted in which the temporal profile of the force of infection from 1960 onwards was described by a polynomial of the form
 |
in which j is a positive integer from 0 to k. The likelihood ratio test30 was used to determine the minimal adequate model with the lowest polynomial order (i.e. the minimum value of k, whereby moving to a higher order k+1 polynomial did not result in a statistically significant decrease in the amount of deviance).31 All model fitting was performed using the Solver add-in function in Excel 2000 (Microsoft, Reading, United Kingdom).
Estimating the proportion and average age of susceptibles
The proportion of each age fully susceptible to any dengue virus infection in year t was simply taken as 1-ia,t. The total susceptible proportion of the population was then calculated as
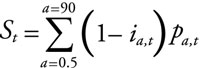 |
where p is the proportion of the population aged a years in year t, which was estimated from official Singapore government figures, and assuming a maximum age of 90 years in the population. The average age of the susceptible proportion of the population in a given year, At, was calculated as
 |
St and At were calculated for both the year-on-year and polynomial models.
Trends in model outputs
For both models, temporal trends in the predicted outcomes following the instigation of control activities in 1966 were investigated using linear regression conducted in Stata version 8 (StataCorp, College Station, TX, United States of America).
Periodicity
Autocorrelation analysis was used to compare the periodicity of clinical incidence, Aedes household index and predicted local year-on-year forces of infection.32 Each of the three time series was first made stationary by taking the natural log, regressing the logged value against time to determine the log-linear trend, and finally subtracting this trend from the logged value. All transformations and autocorrelations were conducted in Stata version 8.
Results
The reconstructed year-on-year changes in force of dengue infection are shown in Fig. 3. The observed and predicted age-stratified seroprevalence recorded in each cross-sectional survey are shown in Fig. 4, Fig. 5, Fig. 6, Fig. 7 and Fig. 8. The model produced a highly significant fit to the data (χ² = 2630, degrees of freedom, d.f. = 42, P ~ 0) explaining almost 94% of the variation in seroprevalence (Table 1).
Fig. 3.
Annual forces of infection predicted by models
a Per capita rate at which susceptible individuals acquire infection.
Fig. 4.
Seroprevalence observed in the 1982 survey and fitted age estimates using models
Fig. 5.
Seroprevalence observed in the 1991 survey and fitted age estimates using models
Fig. 6.
Seroprevalence observed in the 1993 survey and fitted age estimates using models
Fig. 7.
Seroprevalence observed in the 1999 survey and fitted age estimates using models
Fig. 8.
Seroprevalence observed in the 2002 survey and fitted age estimates using models
A third-order polynomial was the lowest-order model that was not significantly different from the year-on-year model (χ² = 23.6, d.f. = 39, P = 0.976) and could not be significantly improved by moving to a higher, forth-order polynomial (χ² = 1.30, d.f. = 1, P = 0.254). Estimates and 95% confidence intervals for the parameters describing the polynomial are shown in Table 2 and the predicted temporal changes in force of infection in Fig. 3. The polynomial model fit to the age seroprevalence surveys, which explained almost 93% of the deviance (Table 1), are shown in Fig. 4, Fig. 5, Fig. 6, Fig. 7 and Fig. 8.
Table 2. Parameter value estimates for third order polynomial model describing the forces of dengue virus infection in Singapore, 1960–2002.
| Parameter | Fitted value | Lower 95% CIa | Upper 95% CIa |
|---|---|---|---|
| α0 | –2.954 | –3.039 | –2.869 |
| α1 | 0.217 | 0.210 | 0.223 |
| α2 | –2.115 × 10–2 | –2.141 × 10–2 | –2.092 × 10–2 |
| α3 | 3.771 × 10–4 | 3.687 × 10–4 | 3.842 × 10–4 |
CI, confidence interval. a Limits determined using likelihood ratio test.
Fig. 3 shows that over the period of effective vector control from 1966 onwards, there were highly significant negative trends in the forces of infection predicted by both the year-on-year model (correlation coefficient = –0.545, P = 4.837 × 10–4) and the simplified polynomial model (correlation coefficient = –0.766, P = 3.342 × 10–8).
There were also significant positive trends in the percentage (Fig. 9) and average age (Fig. 10) of the population susceptible to infection by any dengue virus serotype from 1966 onwards. These trends resulted from the force of infection estimates from both the year-on-year and polynomial models (Table 3). The observed average age of clinical dengue cases was consistently higher than the model-estimated average age of the susceptible population (Fig. 10). This is consistent with the probability of infection resulting in clinical disease being greater in older susceptible individuals.5
Fig. 9.
Estimated changes in the percentage of the population susceptible to infection by all dengue virus serotypes
Fig. 10.
Estimated changes in the average age of the population susceptible to infection by all dengue virus serotypes
Table 3. Correlation coefficients describing trends since instigation of control in 1966 to 2002.
| Model outcome | Correlation coefficient |
|
|---|---|---|
| year-on-year model | polynomial model | |
| Force of infectiona | –0.545 (P = 4.837 × 10–4) | –0.766 (P = 3.342 × 10–8) |
| Proportion of population susceptible | 0.825 (P = 3.332 × 10–10) | 0.879 (P = 8.031 × 10–18) |
| Average age of susceptible population | 0.991 (P = 3.692 × 10–32) | 0.994 (P = 1.096 × 10–35) |
a Per capita rate at which susceptible individuals acquire infection.
The forces of infection predicted by the year-on-year model show high levels of inter-annual variation, with the major peaks in transmission intensity corresponding well with the recorded peaks in clinical incidence (Fig. 11 and Fig. 12). The autocorrelograms for clinical incidence, Aedes household index and predicted year-on-year force of infection from 1966 to 2002 are shown in Fig. 13. There was a strong similarity between the correlogram for the model-predicted forces of infection, with a distinct 5–6 year periodicity, and the profile and periodicity observed in clinical incidence. By contrast, the Aedes household index displayed no such periodicity.
Fig. 11.
Observed annual clinical incidence and predicted annual forces of infection, 1960–1987
a Per capita rate at which susceptible individuals acquire infection.
Fig. 12.
Observed annual clinical incidence and predicted annual forces of infection, 1988–2002
a Per capita rate at which susceptible individuals acquire infection.
Fig. 13.
Correlogram of the observed annual clinical incidence, Aedes household index and predicted annual forces of infection, 1966–2002
Discussion
Adhering to all WHO recommendations, Singapore has dramatically reduced the percentage of households with Aedes mosquitoes since the inception of its vector control programme. The fact that incidence of clinical dengue fever has recently increased despite this sustained reduction in the Aedes mosquito population has been difficult to explain.
Four main suggestions have been proposed to reconcile this contradictory phenomenon: (1) A shift in dengue virus transmission from the household to other sites, such as schools and workplaces.17 (2) Increased dengue virus transmission by other Aedes mosquito species, most notably Ae. albopictus, which is not targeted by current control activities. However, evidence suggests that the pattern of dengue incidence in Singapore is strongly spatially associated with that of Ae. aegypti populations and not Ae. albopictus.33 (3) Increased reporting probability of clinical cases. Though this is likely correct for the more distant past, it does not reconcile the year-on-year increases in incidence seen in Singapore recently. (4) The decrease in Aedes mosquitoes in Singapore caused a reduction in herd immunity and this process, coupled with the increased importation of dengue viruses into Singapore, led to an increase in transmission and a subsequent increase in disease incidence.34
While these theories may help to explain the increased clinical incidence, none addresses the central issue of whether vector control has reduced force of infection. In fact, they all invoke a shift or increase in dengue transmission not captured by the Aedes household index. However, the patterns from the age-seroprevalence surveys, as elucidated by the simple mathematical models, are robustly consistent with there being a general trend of decreasing force of dengue infection since the introduction of vector control measures in 1966. Confidence in the temporal trends in force of infection is strengthened by the correspondence in peaks of predicted transmission with observed disease incidence (Fig. 11 and Fig. 12), and the predicted periodicity of force of infection with observed periodicity of incidence of clinical disease over the same time period (Fig. 13). It should be stressed that these predictions were made completely independent of clinical incidence data and reveal latent properties of the age seroprevalence data uncovered by the model. It is also noteworthy that the Aedes household index failed to display any periodicity in line with disease incidence, supporting the argument that the main driver of the interepidemic interval is herd immunity rather than vector population dynamics.35
Therefore, if there has been a decrease in force of infection, how can the increase in disease incidence be explained? Endemic stability is an epidemiological state in which host, disease agent, vector and environment coexist with little or no clinical disease.36 The concept of endemic stability has recently been proposed as a general hypothesis for the contradictory phenomenon of decreasing force of infection and increasing clinical incidence for diseases that meet two criteria: (1) the probability of infection resulting in clinical disease is more likely in older than in younger individuals, and (2) initial infection decreases the probability of subsequent infection.37 Classic dengue fever fits both of the necessary criteria for endemic stability to occur. Infection with dengue virus confers life-long immunity to the infecting viral serotype. Furthermore, there is substantial evidence that older individuals are more likely to contract classic dengue fever than younger individuals.5
We propose that the marked reduction in force of dengue infection in Singapore, due to mosquito control, resulted not only in an overall reduction in herd immunity but an increase in the average age of first infection, which led to an increase in the incidence of clinical disease by exposing a higher proportion of older age groups to infection. From a surveillance perspective, the performance of the model highlights the potential importance of regular cross-sectional, age-stratified serologic surveys as a surveillance tool for monitoring the impact of dengue prevention activities and helping to plan for potential epidemics. Most endemic communities throughout the world have poor surveillance capabilities for both Aedes mosquitoes and dengue.38 This lack of surveillance means that an emergency response to epidemic dengue has become common practice. But by the time a response is carried out, transmission is usually at or near its peak, at which time vector control has little impact. Due to their relative ease and low cost, age-stratified seroprevalence surveys have proven to be a useful tool in surveillance of several directly transmitted diseases, most notably chickenpox and measles.31 More recently, researchers have investigated the applicability of age seroprevalence survey data to help monitor the transmission of vector-borne diseases such as malaria, trypanosomiasis and cutaneous leishmaniasis.20,21,39
Our findings have significant implications for dengue prevention and control. Due to the absence of any prophylaxis for dengue, prevention of the disease has focused almost exclusively on controlling the primary vector, Aedes aegypti. Singapore has been a world leader in Aedes control for decades, yet if Singapore is unable to interrupt dengue transmission, it will likely be difficult for other endemic communities to do so. These findings suggest that a step-change in the effectiveness of current vector control measures is essential to fight dengue and avoid the unwanted effects of piecemeal control activities. ■
Footnotes
Competing interests: PG Coleman and DW Kelly are shareholders in Oxitec Limited, a private company developing novel genetic technologies for the control of insect populations.
References
- 1.CDC dengue fever home page. Washington: US Centers for Disease Control and Prevention; 2005. Available from: http://www.cdc.gov/ncidod/dvbid/dengue/index.htm
- 2.Dengue and dengue haemorrhagic fever WHO Fact Sheet No. 117. Geneva: WHO; 2002.
- 3.Murgue B, Deparis X, Chungue E, Cassar O, Roche C. Dengue: an evaluation of dengue severity in French Polynesia based on an analysis of 403 laboratory-confirmed cases. Trop Med Int Health. 1999;4:765–73. doi: 10.1046/j.1365-3156.1999.00478.x. [DOI] [PubMed] [Google Scholar]
- 4.Kuno G. Review of the factors modulating dengue transmission. Epidemiol Rev. 1995;17:321–35. doi: 10.1093/oxfordjournals.epirev.a036196. [DOI] [PubMed] [Google Scholar]
- 5.Halstead SB. Epidemiology of dengue and dengue hemorrhagic fever In: Gubler D, Kuno G, eds. Dengue and dengue hemorrhagic fever Wallingford/New York: CAB International; 1997. [Google Scholar]
- 6.Guzman MG, Kouri G, Valdes L, Bravo J, Alvarez M, Vazques S, et al. Epidemiologic studies on dengue in Santiago de Cuba, 1997. Am J Epidemiol. 2000;152:793–9. doi: 10.1093/aje/152.9.793. [DOI] [PubMed] [Google Scholar]
- 7.Siler JF, Hall MW, Hitchens AP. Dengue, its history, epidemiology, mechanism of transmission, etiology, clinical manifestations, immunity and prevention. Philippine Journal of Science. 1926;29:1–304. [Google Scholar]
- 8.Simmons JS, St John JH, Reynolds FHK. Experimental studies of dengue. Philippine Journal of Science. 1931;44:1–251. [Google Scholar]
- 9.Graham RR, Juffrie M, Tan R, Hayes CG, Laksono I, Ma’roef C, et al. A prospective seroepidemiologic study on dengue in children four to nine years of age in Yogyakarta, Indonesia. Am J Trop Med Hyg. 1999;61:412–9. doi: 10.4269/ajtmh.1999.61.412. [DOI] [PubMed] [Google Scholar]
- 10.Vaughn DW, Green S, Kalayanarooj S, Innis BL, Nimmannitya S, Suntayakorn S, et al. Dengue viremia titer, antibody response pattern, and virus serotype correlate with disease severity. J Infect Dis. 2000;181:2–9. doi: 10.1086/315215. [DOI] [PubMed] [Google Scholar]
- 11.Uribe LJ. The problems of Aedes aegypti control in the Americas. Bull Pan Am Health Organ. 1983;17:133–41. [PubMed] [Google Scholar]
- 12.Gubler DJ. Dengue and dengue hemorrhagic fever: its history and resurgence as a global public health problem. In: Gubler DJ, Kuno G, eds. Dengue and dengue hemorrhagic fever Wallingford/New York: CAB International; 1997. [Google Scholar]
- 13.Guidelines for dengue surveillance and mosquito control. Geneva: WHO; 2003.
- 14.Dengue surveillance in Singapore, 2000. Epidemiological News Bulletin. 2001;27(2). Singapore: Ministry of Health; 2001.
- 15.Communicable diseases surveillance in Singapore Singapore: Ministry of Health; 2003.
- 16.Dengue surveillance in Singapore, 2004. Epidemiological News Bulletin. 2005;31(1). Singapore: Ministry of Health; 2004.
- 17.Ooi EE, Hart TJ, Tan HC, Chan SH. Dengue seroepidemiology in Singapore. Lancet. 2001;357:685–6. doi: 10.1016/S0140-6736(00)04137-4. [DOI] [PubMed] [Google Scholar]
- 18.Weekly Infectious Disease Bulletin, epidemiological week 52, vol. 1, no. 52. Singapore: Ministry of Health; December 31, 2005.
- 19.Anderson RM, May RM. Infectious diseases of humans: dynamics and control Oxford/New York: Oxford University Press; 1991. [Google Scholar]
- 20.Drakeley CJ, Corran PH, Coleman PG, Tongren JE, McDonald SL, Carneiro I, et al. Estimating medium- and long-term trends in malaria transmission by using serological markers of malaria exposure. Proc Natl Acad Sci USA. 2005;102:5108–13. doi: 10.1073/pnas.0408725102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Feliciangeli MD, Campbell-Lendrum D, Martinez C, Gonzalez D, Coleman P, Davies C. Chagas disease control in Venezuela: lessons for the Andean region and beyond. Trends Parasitol. 2003;19:44–9. doi: 10.1016/S1471-4922(02)00013-2. [DOI] [PubMed] [Google Scholar]
- 22.Ferguson NM, Donnelly CA, Anderson RM. Transmission dynamics and epidemiology of dengue: insights from age-stratified sero-prevalence surveys. Philos Trans R Soc Lond B Biol Sci. 1999;354:757–68. doi: 10.1098/rstb.1999.0428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mossong J, Putz L, Schneider F. Sero-prevalence and force of infection of varicella-zoster virus in Luxembourg. Epidemiol Infect. 2004;132:1121–7. doi: 10.1017/S0950268804002754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goh KT. Dengue in Singapore Technical Monograph Series, no. 2. Singapore: Institute of Environmental Epidemiology, Ministry of the Environment; 1998. [Google Scholar]
- 25.Wilder-Smith A, Foo W, Earnest A, Sremulanathan S, Paton NI. Seroepidemiology of dengue in the adult population of Singapore. Trop Med Int Health. 2004;9:305–8. doi: 10.1046/j.1365-3156.2003.01177.x. [DOI] [PubMed] [Google Scholar]
- 26.Clarke DH, Casals J. Techniques for hemagglutination and hemagglutination-inhibition with arthropod-borne viruses. Am J Trop Med Hyg. 1958;7:561–73. doi: 10.4269/ajtmh.1958.7.561. [DOI] [PubMed] [Google Scholar]
- 27.Rigau-Perez JG, Gubler DJ, Vorndam AV, Clark GG. Dengue: A literature review and case study of travelers from the United States, 1986-1994. J Travel Med. 1997;4:65–71. doi: 10.1111/j.1708-8305.1997.tb00782.x. [DOI] [PubMed] [Google Scholar]
- 28.Ooi EE, Goh KT, Gubler DJ. Dengue prevention and 35 years of vector control in Singapore. Emerg Infect Dis. 2006;12:887–93. doi: 10.3201/10.3201/eid1206.051210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Crassidis JL, Junkins JL. Optimal estimation of dynamic systems Boca Raton: Chapman & Hall/CRC; 2004. [Google Scholar]
- 30.Kirkwood BR, Sterne JAC, Kirkwood BR. Essential medical statistics 2nd ed. Malden: Blackwell Science; 2003. [Google Scholar]
- 31.Grenfell BT, Anderson RM. The estimation of age-related rates of infection from case notifications and serological data. J Hyg (Lond) 1985;95:419–36. doi: 10.1017/s0022172400062859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shumway RH, Stoffer DS. Time series analysis and its applications New York: Springer; 2000. [Google Scholar]
- 33.Chung YK, Pang FY. Dengue virus infection rate in field populations of female Aedes aegypti and Aedes albopictus in Singapore. Trop Med Int Health. 2002;7:322–30. doi: 10.1046/j.1365-3156.2002.00873.x. [DOI] [PubMed] [Google Scholar]
- 34.Reiter P. Dengue Control in Singapore In: Goh KT, ed., Dengue in Singapore. Singapore: 1993; 213-242. [Google Scholar]
- 35.Hay SI, Myers MF, Burke DS, Vaughn DW, Endy T, Ananda N, et al. Etiology of interepidemic periods of mosquito-borne disease. Proc Natl Acad Sci USA. 2000;97:9335–9. doi: 10.1073/pnas.97.16.9335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Norval RAI, Perry BD. The Epidemiology of Theleriosis in Africa London: Academic Press; 1992. [Google Scholar]
- 37.Coleman PG, Perry BD, Woolhouse ME. Endemic stability — a veterinary idea applied to human public health. Lancet. 2001;357:1284–6. doi: 10.1016/S0140-6736(00)04410-X. [DOI] [PubMed] [Google Scholar]
- 38.Gubler DJ. How effectively is epidemiological surveillance used for dengue programme planning and epidemic response? Dengue Bulletin WHO 2002;26. [Google Scholar]
- 39.Davies CR, Llanos-Cuentas A, Canales J, Leon E, Alvarez E, Monge J, et al. The fall and rise of Andean cutaneous leishmaniasis: transient impact of the DDT campaign in Peru. Trans R Soc Trop Med Hyg. 1994;88:389–93. doi: 10.1016/0035-9203(94)90395-6. [DOI] [PubMed] [Google Scholar]















