Abstract
Measles remains an important cause of pediatric morbidity and mortality in developing countries, especially among infants who are too young to receive the current licensed live attenuated measles vaccine. We developed two Sindbis virus DNA vaccines encoding the measles virus hemagglutinin (pMSIN-H) and fusion proteins (pMSINH-FdU) and examined their immunogenicities and protective efficacies when administered alone or followed by the live measles virus vaccine in cotton rats. Neutralizing antibodies, mucosal and systemic antibody-secreting cells, memory B cells, and gamma interferon-secreting T cells developed after priming and increased after boosting. pMSIN-H priming conferred 100% protection against pulmonary measles, whereas pMSINH-FdU protected only in conjunction with the live measles virus vaccine boost.
In recent years, periodic mass campaigns to immunize children with the currently licensed measles vaccines have succeeded in reducing the measles mortality burden in sub-Saharan Africa (2, 22). Nevertheless, measles still remains a major cause of morbidity and mortality among children in a number of developing countries (8, 10). Particularly at risk are young infants during the “window of vulnerability” (4 to 8 months of age), a period during which declining maternal antibodies are insufficient to protect against wild-type virus but can nevertheless interfere with successful immunization using the licensed live attenuated measles virus (MV) vaccine. Early attempts to use a high-dose vaccine in infants ∼6 months of age unexpectedly led to an increase in all-cause deaths among female children (1).
We developed two Sindbis virus-based DNA vaccines encoding the MV hemagglutinin (H) alone or together with the MV fusion (F) protein (pMSIN-H and pMSINH-FdU) (15, 21), intended to prime the immune systems of young infants 6 and 10 weeks of age (coadministered with DTP1 and DTP2) so that they could successfully respond to subsequent boosting with live measles virus vaccine given at 14 weeks of age (with DTP3).
Both vaccines were highly immunogenic for adult and newborn mice, even in the presence of maternal antibodies (5, 15, 21). They also elicited high levels of neutralizing antibodies in juvenile and infant rhesus macaques and protected them against respiratory challenge. In anticipation of the immunization regimen to be used in humans, these vaccines were given as a two-dose priming followed by a subsequent boosting with the live attenuated measles vaccine (16). A question that remained unanswered in the primate studies, however, was whether these vaccines could confer protection after priming with the DNA vaccines alone, prior to the boost with the attenuated measles vaccine. Additional unanswered questions concerned the characteristics of immune responses induced and the effector mechanisms associated with protection, mainly due to the limited availability of samples, which allowed only antibody measurements and basic cell-mediated-immunity assays.
In the present study, we examined the protective capacities of pMSIN-H and pMSINH-FdU administered alone or followed by a subsequent boost with live attenuated Edmonston Zagreb (EZ) measles virus vaccine in cotton rats. We also performed a detailed characterization of the B- and T-cell responses at the time of challenge.
To this end, cotton rats (6 to 12 weeks old) were immunized with two doses (100 μg/each) of pMSIN-H, pMSINH-FdU, or pSINCP (GMP pilot lots produced by Althea Technologies, Inc.) given on days 0 and 28 intramuscularly using a needle and syringe. Additional groups were boosted on day 56 with the attenuated EZ measles virus vaccine (Serum Institute of India) as recommended for humans: ∼5 × 104 50% tissue culture infective doses (TCID50) in 0.5 ml were delivered subcutaneously (s.c.). Animal procedures were conducted at Virion Systems Inc. (Rockville, MD) and approved by Virion Systems' animal care and use committee.
We examined the kinetics of appearance of MV-specific plaque reduction neutralizing (PRN) antibodies (15) in vaccinated and control animals. A single dose of pMSIN-H elicited a mean PRN antibody response that surpassed the minimum required for protection in humans (>120 mIU/ml) (6, 17). A further fourfold increase was observed in response to the second dose (geometric mean titers [GMTs], 200 and 900 mIU/ml on days 28 and 56, respectively) (Fig. 1A). Priming with pMSINH-FdU also elicited PRN antibodies, albeit at lower levels; a second immunization was necessary to achieve protective titers (GMTs, 25 and 135 mIU/ml on days 28 and 56, respectively).
FIG. 1.
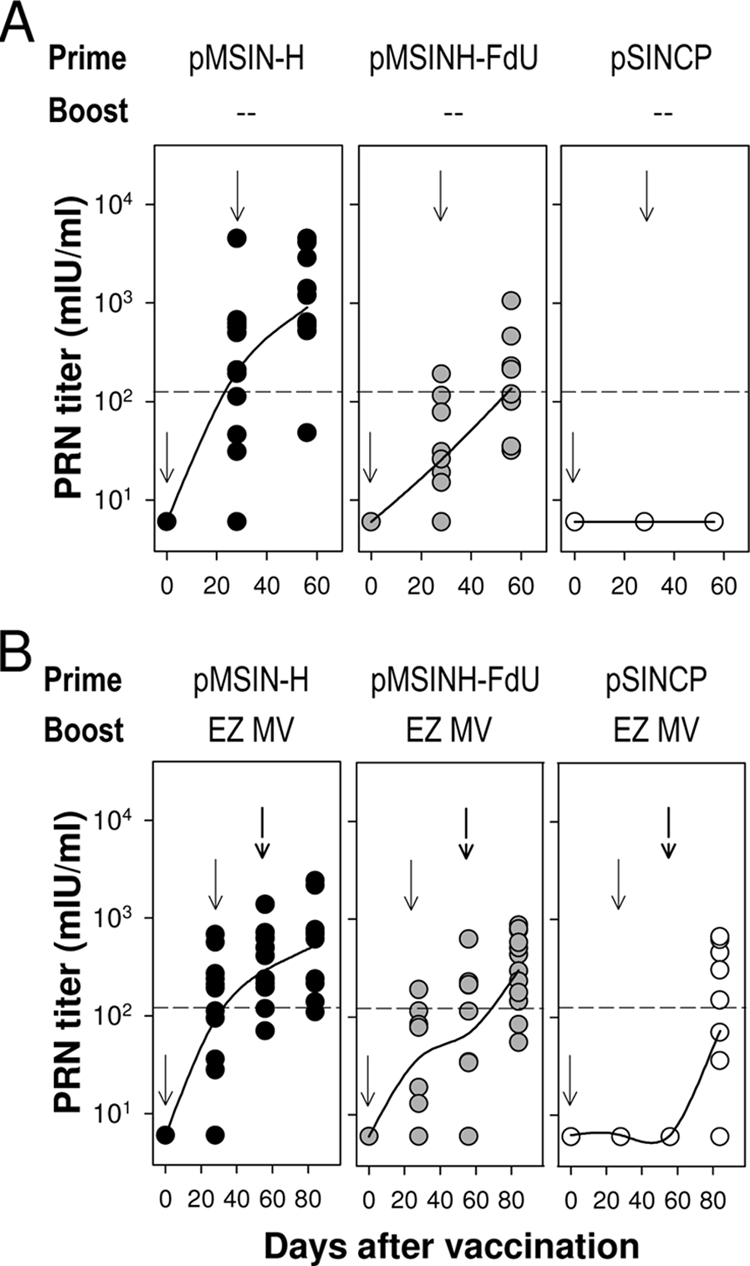
PRN titers elicited by Sindbis virus-based MV DNA vaccines administered alone as a “prime” or followed by a subsequent boost with the live attenuated EZ measles vaccine in a heterologous “prime-boost” regimen. (A) Cotton rats were primed with two doses (100 μg/each) of pMSIN-H, pMSINH-FdU, or pSINCP given intramuscularly on days 0 and 28. (B) DNA priming was followed by a single boost with the EZ measles vaccine (0.5 ml, ∼5 × 104 TCID50) given s.c. on day 56. Data represent individual titers from 11 to 12 animals per group. Lines are plotted upon the geometric mean titers. Arrows indicate each immunization. Dotted lines indicate the PRN titer considered to be protective in humans (120 mIU/ml).
Priming with either DNA vaccine led to potent anamnestic responses after the EZ boost (Fig. 1B). Cotton rats primed with pMSIN-H achieved the highest PRN titers after the EZ boost. Increased (albeit still lower) PRN levels were also seen in cotton rats primed with pMSINH-FdU. Surprisingly, the EZ vaccine elicited a very modest response in cotton rats in the absence of DNA priming (GMT, 73 mIU/ml on day 84) (Fig. 1B); 50% of animals had PRN titers below the protective threshold. The virus-neutralizing capacity of the vaccine-induced antibodies was also measured by syncytium inhibition in B958 cells that express the simian homologue of the human MV receptor CD150/SLAM. A significant correlation was found between PRN and syncytium inhibition titers for all vaccine responders (r = 0.60; P < 0.01).
We further examined the responses elicited by the Sindbis virus DNA-measles virus vaccines by measuring the frequency of antibody-secreting plasma cells (ASC) in mucosal and systemic tissues 1 month after the last immunization and immediately prior to challenge. Lung tissue was dissected and homogenized and lymphocytes further purified through gradient centrifugation using Lympholyte (rat) (4). Cells from spleen and bone marrow (BM) were obtained as described previously (5). Peripheral blood lymphocytes (PBL) were separated from heparinized blood through gradient centrifugation. Cells were incubated overnight in nitrocellulose plates coated with MV lysate, and antibodies were detected with rabbit anti-cotton rat immunoglobulin G (IgG) (Virion Systems), followed by horseradish peroxidase-labeled goat anti-rabbit IgG.
MV-specific IgG ASC were detected in lungs and PBL following priming with either vaccine, which further increased after the EZ boost (Fig. 2A). ASC responses to the EZ live virus vaccine were comparable to those elicited by the DNA vaccines alone. Priming with pMSIN-H led to a significantly higher frequency of PBL IgG ASC (P < 0.001) than was the case with pMSINH-FdU; this increased response was also observed after the boost.
FIG. 2.
Vaccine-induced B- and T-cell responses and protection against respiratory measles virus challenge. (A) Frequency of MV-specific IgG-secreting cells in the lung and PBL. (B) MV-specific memory B cells measured in the BM. (C) IFN-γ-secreting T cells measured in PBL. (D) Protection against measles virus respiratory challenge. (E) Lung viral replication versus PRN antibody titers at the time of challenge. Cotton rats were immunized as described in the legend to Fig. 1. One month after the last immunization, lung, PBL, and BM were harvested from four animals in each group; the remaining animals were challenged with wild-type MV. Data represent mean frequencies of IgG ASC or IFN-γ SFC per 1 × 106 cells ± standard deviations for replicate cultures (A to C), mean log10 TCID50/g of lung tissue ± standard errors of the means (SEM) (D), or individual lung virus and PRN titers (E) from 7 to 16 animals per group from two separate experiments. Significant differences among groups are indicated: *, pMSIN-H and pMSINH-FdU versus pSINCP, P < 0.001; **, pMSIN-H versus pMSINH-FdU, P < 0.001. Group responses were compared using the t test, analysis of variance, or the Mann-Whitney rank sum test (if normality failed); P values of <0.05 were considered significant. The dotted line indicates the PRN titer considered to be protective in humans (120 mIU/ml).
We next examined the presence of MV-specific memory B cells in the BM. Cells were allowed to expand and differentiate in vitro into ASC through a 6-day incubation period in the presence of mitogens (0.002% lectin from Phytolacca americana, Escherichia coli O55:B5 lipopolysaccharide [50 μg/ml], and CpG oligodeoxynucleotide 1826 [5′-TCCATGACGTTCCTGACGTT-3′; 0.3 μg/ml]). The frequency of MV-specific and total IgG ASC was then quantified by enzyme-linked immunospot (ELISPOT) assay (7) (Fig. 2B). Both vaccines elicited a pool of memory B cells that further augmented after the EZ boost. In agreement with previous findings, the highest frequencies of B memory cells were seen for animals primed with pMSIN-H, whereas the weakest responses, comparable to those elicited by pMSINH-FdU alone, were observed for unprimed animals that received the EZ boost. A similar pattern of B memory responses, although of lower magnitude, was observed in spleen tissue (data not shown).
Although antibodies constitute the recognized correlate of protection, T cells are believed to help control virus replication, and any new measles vaccine is expected to elicit strong cell-mediated immunity. We examined our vaccines' capacities to generate MV-specific gamma interferon (IFN-γ)-secreting T cells in PBL at the moment of challenge (Fig. 2C). Lymphocytes were stimulated in vitro for 48 to 72 h with MV lysate, and the frequencies of IFN-γ spot-forming cells (SFC) were quantified by ELISPOT assay. Animals immunized with either DNA vaccine exhibited significant levels of IFN-γ SFC; a slight yet significantly higher T-cell frequency was found in animals primed with pMSINH-FdU (P < 0.001). Responses increased after the EZ boost. The frequencies of IFN-γ SFC in the prime-boost groups surpassed those of animals that received only the EZ vaccine (P < 0.001).
Protection against respiratory MV infection was assessed through intranasal challenge with 2 × 106 to 2 × 107 TCID50 of virulent MV (Edmonston strain). Titers of virus were measured in lungs 4 days later (15). All 16 unimmunized controls (pSINCP) exhibited significant virus replication in their lungs (Fig. 2D). In contrast, no animal immunized with pMSIN-H alone had detectable virus (100% protection). Among cotton rats immunized with pMSINH-FdU alone, 3 out of 16 had virus in the lungs (81% protection) (Fig. 2D). As expected, the highest virus load was found in cotton rats that received the empty plasmid pSINCP. Regardless of the vaccine used for priming, all animals were protected after the EZ boost. In the absence of priming, a single dose of EZ vaccine conferred 75% protection; two out of eight animals had detectable virus in their lungs, and the mean viral titer was similar to that of the pMSINH-FdU-primed group.
When we compared protection against infection with PRN titers at the time of challenge, we observed that all animals with a PRN titer of ≥120 mIU/ml were protected (i.e., had no detectable virus in the lungs) (Fig. 2E). In contrast, all cotton rats with titers of ≤14 mIU/ml exhibited virus replication in the lungs. Among those with titers of >14 but <120 mIU/ml, only 1 out of 9 (11%) had detectable virus in the lung. A significant reduction in virus replication in cotton rats even in the presence of low levels of neutralizing antibodies has also been reported by others (20).
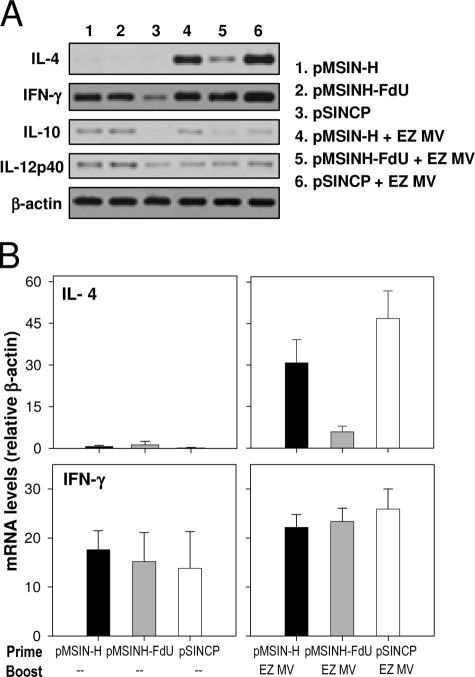
Finally, we examined the profile of cytokine gene expression in the lungs 4 days after challenge using reverse transcription-PCR (3) (Fig. 3). We hypothesized that the vaccines and immunization regimen might induce different cytokine patterns, which could influence protection. The most salient findings from this analysis were the following: (i) absence of interleukin 4 (IL-4) expression in animals that received a two-dose priming series of pMSIN-H or pMSINH-FdU (although IL-4 was produced after the EZ boost); (ii) high levels of IFN-γ produced in animals primed with either pMSIN-H or pMSINH-FdU; and (iii) low expression of IL-4 in all animals primed with pMSINH-FdU after the boost. No overt differences were seen in the expression of IL-12 and IL-10.
FIG. 3.
Pulmonary cytokine gene expression in cotton rats after vaccination and MV challenge. (A) Cytokine mRNA expression in lung homogenates measured by reverse transcription-PCR. (B) Relative expression of IL-4 and IFN-γ. Amplified products were quantified by Southern blotting and normalized to the level of β-actin (housekeeping gene). Data represent mean mRNA cytokine expression from four animals ± standard errors of the means.
A novel contribution of this study is the demonstration that both Sindbis virus DNA vaccines protect against respiratory MV infection when administered alone, which could not be assessed with monkeys due to the difficulty of working with this model and the need to prioritize the vaccine/regimen testing. The detailed analysis of B-cell responses and cytokine mRNA expression at the time of challenge is also new; these outcomes could not be studied in monkeys due to the limited tissue available.
Cotton rats are permissive for MV replication, thereby allowing assessment of the protective capacity of new vaccine candidates (12, 23). For many years, a drawback of this model had been the lack of reagents and established methods to assess immune responses. Herein, we measured MV-specific B-cell responses and demonstrated the capacity of our Sindbis virus-MV DNA vaccines to stimulate mucosal and circulating IgG ASC as well as memory B cells residing in the BM. To the best of our knowledge, this is the first description of such responses to a measles (or any other) vaccine in cotton rats. The IgG ASC detected in circulation likely represent a pool of B cells available to migrate to mucosal sites. Memory B cells in the BM and spleen are presumably involved in maintaining a sustained level of circulating PRN antibodies, which has been directly correlated with protection (6). Our data also show that PRN titers associated with protection in humans (>120 to 200 mIU/ml) (6, 17) are also indicative of protection in cotton rats. Among the effector mechanisms measured in our study, the level of neutralizing antibodies, the PBL IgG ASC (probably homing to the mucosa), and the B memory cells appeared to be those most closely correlated with protection in this model. On the other hand, the T-cell responses detected after vaccination did not appear to be a determinant of protection. Priming with pMSINH-FdU, for example, elicited a high frequency of IFN-γ SFC, which surpassed that produced by pMSIN-H, and yet this vaccine conferred only partial protection whereas pMSIN-H was fully protective. This apparent lack of association between T-cell responses and protection against pulmonary MV infection in cotton rats has been reported by others (19). It is possible that whereas T cells may not be able to prevent infection, they may nevertheless help to prevent further virus spread, thereby preventing more severe disease and complications.
The cytokine profile elicited by the vaccines and evoked by the virus challenge suggests that both DNA plasmids produce a predominant Th1-type response with IFN-γ secretion in the absence of IL-4 whereas the EZ vaccine favors a Th2/Th1-type response with production of IL-4 and IFN-γ. Both cytokines were induced by the prime-boost regimen. We and others have shown the production of IFN-γ in PBL from juvenile and infant monkeys immunized with MV DNA vaccines (14, 16, 18). Howe et al. showed CD4-mediated Th1/Th2-type cytokine secretion in MV-immune individuals (9). Infants prone to Th2-type biased responses can greatly benefit from a vaccine-driven Th1-type cytokine milieu that would facilitate an IgG class switch and avidity maturation of antibodies, found to be deficient in early infancy (11). The extremely low IL-4 responses in animals primed with pMSINH-FdU and boosted with the EZ vaccine were noteworthy. The absence of CD4+ T cells that can secrete IL-4 could help explain, in part, the lower titers of antibody produced by this construct.
The results described in this study corroborate those reported for other animal species (5, 16, 21). The superior immunogenicity of pMSIN-H over pMSINHF-dU, for example, has now been confirmed in several animal models. Although the reasons are unclear, it seems as if the simultaneous expression of H and F were (in some way) interfering with the proper functioning of B and T cells, resulting in suboptimal humoral responses. MV-infected or transfected cells coexpressing H and F have been shown to impair T-cell responses when transferred to cotton rats (13). Further studies will be needed to rule out potential immunosuppression by pMSINH-FdU in humans.
Most importantly, the protective efficacy observed herein for cotton rats fully agrees with results obtained with rhesus macaques (16). A compilation of immune responses and protection outcomes for pMSIN-H and pMSINH-FdU in different models is shown in Table 1. The data further support the reliability of the cotton rat as a practical and helpful model for the evaluation of respiratory virus vaccine candidates. Our cotton rat model results provide encouragement for further studies of the Sindbis virus-based DNA vaccines in humans; a phase 1 trial of the two measles virus DNA vaccines was recently successfully completed.
TABLE 1.
Immune responses and protection induced by pMSIN-H and pMSINH-FdU in different animal models
| Vaccine(s) | Response induced ina:
|
|||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adult mice
|
Neonatal mice
|
Rhesus macaques
|
Cotton rats
|
|||||||||||
| PRN | IFN-γ | IgG ASC | PRN | IFN-γ | IgG ASC | PRN | IFN-γ | IgG ASC | Protective efficacye (%) | PRN | IFN-γ | IgG ASC | Protective efficacy (%) | |
| pMSIN-H | +++ | +++ | NDb | +++ | ++ | ++ | +++ | ++ | ++ | ND | ++ | + | ++ | 100 |
| pMSINH-FdU | ++ | +++ | ND | ++ | ++ | ++ | + | ++ | ND | ND | + | ++ | ++ | 81 |
| pSINCPd | −c | − | ND | − | − | − | − | − | ND | 0 | − | − | − | 0 |
| pMSIN-H + EZ | ND | ND | ND | +++ | ++ | ++ | ++++ | ++ | ND | 100 | ++ | ++ | ++ | 100 |
| pMSINH-FdU + EZ | ND | ND | ND | ++ | ++ | ++ | ++++ | +++ | ND | 67-81 | ++ | ++ | ++ | 100 |
| pSINCP + EZ | ND | ND | ND | + | + | ++ | + | ND | 0-50 | + | + | ++ | 75 | |
Responses were as follows. For PRN: +, <200 mIU/ml; ++, 200 to 1,000 mIU/ml; +++, 1,000 to 10,000 mIU/ml; ++++, >10,000 mIU/ml. For IFN-γ and IgG ASC: +, <50 SFC; ++, 50 to 200 SFC; +++, >200 SFC per 1 × 106 PBL (rhesus macaques and cotton rats) or spleen cells (mouse). Protective efficacy, percentage of animals protected against respiratory measles challenge. For adult mice, see references 15 and 21; for neonatal mice, see reference 5; for rhesus macaques, see reference 16.
ND, not done.
−, no response.
pSINCP, pSINCP and naïve controls.
EZ was given by aerosol or s.c.; all macaques primed with pMSIN-H were protected regardless of the route of EZ administration.
Acknowledgments
We thank Paul Rota (CDC, Atlanta, GA) for providing the EZ live attenuated measles virus vaccine, Kevin Yim, Jorge Blanco, and Marina Boukhvalova from Virion Systems, Inc. (Rockville, MD), for assistance in animal studies, cytokine testing, and critical readings of the manuscript, and Yu Lim from the CVD Applied Immunology Section for outstanding assistance with PRN assays.
This work was supported by a grant from the Bill and Melinda Gates Foundation to M. M. Levine.
We declare no conflict of interest.
Footnotes
Published ahead of print on 7 January 2009.
REFERENCES
- 1.Aaby, P., K. Knudsen, H. Whittle, I. M. Lisse, J. Thaarup, A. Poulsen, M. Sodemann, M. Jakobsen, L. Brink, U. Gansted, et al. 1993. Long-term survival after Edmonston-Zagreb measles vaccination in Guinea-Bissau: increased female mortality rate. J. Pediatr. 122904-908. [DOI] [PubMed] [Google Scholar]
- 2.Arevshatian, L., C. Clements, S. Lwanga, A. Misore, P. Ndumbe, J. Seward, and P. Taylor. 2007. An evaluation of infant immunization in Africa: is a transformation in progress? Bull. W. H. O. 85449-457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blanco, J. C., J. Y. Richardson, M. E. Darnell, A. Rowzee, L. Pletneva, D. D. Porter, and G. A. Prince. 2002. Cytokine and chemokine gene expression after primary and secondary respiratory syncytial virus infection in cotton rats. J. Infect. Dis. 1851780-1785. [DOI] [PubMed] [Google Scholar]
- 4.Capozzo, A. V., L. Cuberos, M. M. Levine, and M. F. Pasetti. 2004. Mucosally delivered Salmonella live vector vaccines elicit potent immune responses against a foreign antigen in neonatal mice born to naive and immune mothers. Infect. Immun. 724637-4646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Capozzo, A. V., K. Ramirez, J. M. Polo, J. Ulmer, E. M. Barry, M. M. Levine, and M. F. Pasetti. 2006. Neonatal immunization with a Sindbis virus-DNA measles vaccine induces adult-like neutralizing antibodies and cell-mediated immunity in the presence of maternal antibodies. J. Immunol. 1765671-5681. [DOI] [PubMed] [Google Scholar]
- 6.Chen, R. T., L. E. Markowitz, P. Albrecht, J. A. Stewart, L. M. Mofenson, S. R. Preblud, and W. A. Orenstein. 1990. Measles antibody: reevaluation of protective titers. J. Infect. Dis. 1621036-1042. [DOI] [PubMed] [Google Scholar]
- 7.Crotty, S., R. D. Aubert, J. Glidewell, and R. Ahmed. 2004. Tracking human antigen-specific memory B cells: a sensitive and generalized ELISPOT system. J. Immunol. Methods 286111-122. [DOI] [PubMed] [Google Scholar]
- 8.Grais, R. F., C. Dubray, S. Gerstl, J. P. Guthmann, A. Djibo, K. D. Nargaye, J. Coker, K. P. Alberti, A. Cochet, C. Ihekweazu, N. Nathan, L. Payne, K. Porten, D. Sauvageot, B. Schimmer, F. Fermon, M. E. Burny, B. S. Hersh, and P. J. Guerin. 2007. Unacceptably high mortality related to measles epidemics in Niger, Nigeria, and Chad. PLoS Med. 4e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Howe, R. C., N. Dhiman, I. G. Ovsyannikova, and G. A. Poland. 2005. Induction of CD4 T cell proliferation and in vitro Th1-like cytokine responses to measles virus. Clin. Exp. Immunol. 140333-342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moss, W. J. 2007. Measles still has a devastating impact in unvaccinated populations. PLoS Med. 4e24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nair, N., H. Gans, L. Lew-Yasukawa, A. C. Long-Wagar, A. Arvin, and D. E. Griffin. 2007. Age-dependent differences in IgG isotype and avidity induced by measles vaccine received during the first year of life. J. Infect. Dis. 1961339-1345. [DOI] [PubMed] [Google Scholar]
- 12.Niewiesk, S. 1999. Cotton rats (Sigmodon hispidus): an animal model to study the pathogenesis of measles virus infection. Immunol. Lett. 6547-50. [DOI] [PubMed] [Google Scholar]
- 13.Niewiesk, S., I. Eisenhuth, A. Fooks, J. C. Clegg, J. J. Schnorr, S. Schneider-Schaulies, and V. ter Meulen. 1997. Measles virus-induced immune suppression in the cotton rat (Sigmodon hispidus) model depends on viral glycoproteins. J. Virol. 717214-7219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pan, C. H., A. Valsamakis, T. Colella, N. Nair, R. J. Adams, F. P. Polack, C. E. Greer, S. Perri, J. M. Polo, and D. E. Griffin. 2005. Inaugural article: modulation of disease, T cell responses, and measles virus clearance in monkeys vaccinated with H-encoding alphavirus replicon particles. Proc. Natl. Acad. Sci. USA 10211581-11588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pasetti, M. F., E. M. Barry, G. Losonsky, M. Singh, S. M. Medina-Moreno, J. M. Polo, J. Ulmer, H. Robinson, M. B. Sztein, and M. M. Levine. 2003. Attenuated Salmonella enterica serovar Typhi and Shigella flexneri 2a strains mucosally deliver DNA vaccines encoding measles virus hemagglutinin, inducing specific immune responses and protection in cotton rats. J. Virol. 775209-5217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pasetti, M. F., A. Resendiz-Albor, K. Ramirez, R. Stout, M. Papania, R. J. Adams, F. P. Polack, B. J. Ward, D. Burt, S. Chabot, J. Ulmer, E. M. Barry, and M. M. Levine. 2007. Heterologous prime-boost strategy to immunize very young infants against measles: pre-clinical studies in rhesus macaques. Clin. Pharmacol. Ther. 82672-685. [DOI] [PubMed] [Google Scholar]
- 17.Plotkin, S. A. 2008. Vaccines: correlates of vaccine-induced immunity. Clin. Infect. Dis. 47401-409. [DOI] [PubMed] [Google Scholar]
- 18.Premenko-Lanier, M., P. A. Rota, G. H. Rhodes, W. J. Bellini, and M. B. McChesney. 2004. Protection against challenge with measles virus (MV) in infant macaques by an MV DNA vaccine administered in the presence of neutralizing antibody. J. Infect. Dis. 1892064-2071. [DOI] [PubMed] [Google Scholar]
- 19.Pueschel, K., A. Tietz, M. Carsillo, M. Steward, and S. Niewiesk. 2007. Measles virus-specific CD4 T-cell activity does not correlate with protection against lung infection or viral clearance. J. Virol. 818571-8578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schlereth, B., J. K. Rose, L. Buonocore, V. ter Meulen, and S. Niewiesk. 2000. Successful vaccine-induced seroconversion by single-dose immunization in the presence of measles virus-specific maternal antibodies. J. Virol. 744652-4657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Song, M. K., C. J. Vindurampulle, A. V. Capozzo, J. Ulmer, J. M. Polo, M. F. Pasetti, E. M. Barry, and M. M. Levine. 2005. Characterization of immune responses induced by intramuscular vaccination with DNA vaccines encoding measles virus hemagglutinin and/or fusion proteins. J. Virol. 799854-9861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.World Health Organization. 2007. Progress in global measles control and mortality reduction, 2000-2006. Wkly. Epidemiol. Rec. 48418-424. [PubMed] [Google Scholar]
- 23.Wyde, P. R., K. J. Stittelaar, A. D. Osterhaus, E. Guzman, and B. E. Gilbert. 2000. Use of cotton rats for preclinical evaluation of measles vaccines. Vaccine 1942-53. [DOI] [PubMed] [Google Scholar]