Abstract
Double-stranded RNA (dsRNA) triggers RNA interference (RNAi) to silence genes of matching sequence. In some animals this experimentally induced silencing is transported between cells, and studies in the nematode Caenorhabditis elegans have shown that the dsRNA channel SID-1 is required for the import of such transported silencing signals. Gene silencing can also be triggered by endogenously expressed RNAi triggers, but it is unknown whether such silencing is transported between cells. Here, we show that, in C. elegans, SID-1 is required for efficient silencing of multicopy transgenes, indicating that mobile silencing signals contribute to transgene silencing. Further, most tissues can transport silencing initiated by the tissue-specific transgenic expression of RNAi triggers to other tissues, consistent with expressed RNAi triggers generating mobile silencing signals. Whereas the import of silencing signals requires SID-1, we found that mobile silencing signals generated by transgene-expressed RNAi triggers are exported to other tissues through a SID-1-independent mechanism. Furthermore, when RNAi triggers are expressed in ingested Escherichia coli, silencing signals can be transported to internal tissues from the gut lumen across gut cells that lack SID-1. Thus, C. elegans can transport endogenous and exogenous RNA silencing signals between many different tissues via at least 2 SID-1 independent export pathways.
Keywords: transcytosis, double-stranded RNA transport, intercellular signaling, non-cell autonomy, mobile silencing signals
Gene silencing mechanisms including RNA interference (RNAi), heterochromatin formation (1), and repeat-induced silencing (2) are associated with base-paired RNA structures. For example, double-stranded RNA (dsRNA) and hairpin RNA (hpRNA) can trigger RNAi to silence the expression of genes with matching sequence (3). These homology-driven silencing mechanisms are thought to protect the organism against viruses (4) and mobile repetitive elements such as transposons (5, 6). Consequently, these silencing mechanisms also reduce the expression of Caenorhabditis elegans transgenes that are present in multiple tandem copies (7). In plants, transgenic expression or introduction of an RNAi trigger locally within a tissue can silence the target gene or virus throughout the organism (8, 9). Similar systemic silencing can be triggered in some animals by ingestion, local introduction, or overexpression of RNAi triggers in some tissues (10, 11). In contrast to experimentally introduced RNAi triggers, whether RNAi triggers associated with endogenous gene silencing mechanisms (e.g., transcribed dsRNAs) are similarly transported between cells is unknown.
Several studies have provided insight into the distinct mechanisms used by plants and animals to transport experimentally introduced RNAi triggers between cells. Plants transport RNAi triggers between adjacent cells through dynamic intercellular bridges called plasmodesmata and between distant cells via the phloem vascular tissue (12, 8). In addition, genetic screens in plants have identified some of the proteins required for the generation or reception of transported silencing signals (13). Genetic screens in the nematode C. elegans have identified a dsRNA channel called SID-1 as a key component required for the import of silencing signals into all cells sensitive to systemic RNAi (14). It is not known whether SID-1 is required for export of silencing signals. Most animals, except some insects, have at least one SID-1 homolog. In mouse, the SID-1 homolog sidT1 is required for the import of lipid-modified siRNA into hepatocytes (15), suggesting that the import of RNAi triggers through SID-1 is a broadly conserved process among animals. In cultured Drosophila S2 cells, which lack SID-1 homologs, import of RNAi triggers occurs through a slow and energy-dependent process (16, 17). Expressing C. elegans SID-1 in these cells, however, enables rapid energy-independent dsRNA import (17), suggesting that SID-1 functions as a dsRNA channel.
Here, we show that C. elegans requires SID-1 to efficiently reduce the expression of multicopy transgenes, suggesting that transgene silencing in one cell produces mobile silencing signals that function to initiate and/or maintain transgene silencing in another cell. Tissue-specific expression of either hpRNA or dsRNA in many tissues results in SID-1-dependent systemic silencing. We confirm that SID-1 is required for the import of these silencing signals, but show that tissues that lack SID-1 can export these silencing signals. Finally, when RNAi triggers are expressed in ingested Escherichia coli, silencing signals can be transported from the gut lumen to internal tissues via gut cells that lack SID-1. Therefore, we conclude that silencing signals are exported from C. elegans tissues through multiple SID-1-independent pathways.
Results
Efficient Transgene Silencing Requires SID-1.
Transgene tandem arrays in C. elegans are subject to repeat-induced silencing that depends on genes required for RNAi and is more pronounced in mutants that show enhanced RNAi (e.g., eri-1, rrf-3) (18, 19). Repeat-induced silencing often occurs in a mosaic manner such that only a subset of cells containing the transgene tandem array shows silencing of transgene expression. It is unknown whether the silencing triggers, likely derived from aberrant expression of dsRNA and hpRNA from the tandem arrays, remain in the nucleus or are transported to the cytoplasm or between cells. If such RNAi triggers are transported between cells, then the extent of repeat-induced silencing should depended on SID-1, a dsRNA channel required for systemic RNAi (14).
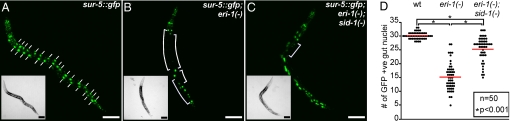
To determine whether repeat-induced silencing spreads beyond the cells in which silencing is initiated we compared the extent of silencing in sid-1(+) and sid-1(−) strains. To induce and monitor spontaneous transgene silencing we used a transgene, sur-5::gfp, which expresses nuclear-localized green fluorescent protein (GFP) in all somatic cells. sur-5::gfp expression is detected in all somatic cells in a wild-type genetic background (Fig. 1A), whereas sur-5::gfp expression is silenced in many cells in an eri-1(−) background ((19) and Fig. 1B). This silencing is readily observed in the large nuclei of the gut cells (Fig. 1A, dashes vs. Fig. 1B, brackets). In a wild-type background, 30.1 ± 0.2 gut nuclei show bright GFP fluorescence, whereas in an eri-1(−) background, only 15.3 ± 0.7 gut nuclei show bright GFP fluorescence (Fig. 1D). Although sur-5::gfp expression was not detectably altered in a sid-1(−) background (data not shown), we found that in a sid-1(−); eri-1(−) double mutant background, 25.6 ± 0.6 gut nuclei show bright GFP fluorescence (Figs. 1 C and D). This significant decrease in transgene silencing in the sid-1(−); eri-1(−) background compared with the eri-1(−) background (P < 0.001) shows that sid-1 is required for efficient repeat induced silencing and that, like RNAi, such silencing produces mobile silencing signals. Further, the significant transgene silencing seen in the sid-1(−); eri-1(−) double mutant background compared with that in the wild-type background (P < 0.001) likely reflects the extent of cell-autonomous transgene silencing. Thus, to efficiently silence transgene tandem arrays, C. elegans requires the transport of silencing signals derived from expressed transgenes between cells and/or tissues.
Fig. 1.
The efficiency of transgene silencing depends on SID-1. Representative wild-type (A), eri-1(−) (B), and eri-1(−);sid-1(−) (C) animals of the fourth larval stage (L4) that express nuclear-localized GFP in all somatic cells (sur-5::gfp) are shown. (Insets): bright-field images. Large gut nuclei (dashes in A) and regions showing silencing in the gut (brackets in B and C) are indicated. (Scale bars, 100 μm.) (D) Quantification of the extent of transgene silencing for each genotype shown in A--C. The number of GFP-positive gut nuclei in each of 50 L4 animals was counted. The average number (red line) for each genotype was significantly (P < 0.001; t test) different from that of the others.
Because the source for repeat-induced silencing triggers, the sur-5::gfp transgene, is expressed in all tissues, the observed sid-1-dependent silencing may reflect transport of silencing signals between cells within a tissue or may also involve transport between tissues. To determine whether silencing signals generated within a single tissue are transported between cells within that tissue, we examined silencing of the muscle-specific myo-3::gfp transgene (supporting information (SI) Fig. S1A). We found that this transgene is silenced in an eri-1(−) background (brackets in Fig. S1B), whereas sid-1(−); eri-1(−) animals appeared to have more body-wall muscle (bwm) cells that showed GFP fluorescence. To objectively compare the extent of silencing we measured the total GFP fluorescence per L4 animal for each genotype. This analysis showed that silencing is less efficient in the sid-1(−); eri-1(−) background than in the eri-1(−) background but more efficient than in the wild-type background (Fig. S1D). These results indicate that repeat-induced silencing signals are transported between cells within a single tissue.
Transgenic Expression of Either hpRNA or dsRNA Causes Transport of Silencing Between Multiple Tissues.
Our analysis of transgene silencing suggests that silencing signals produced in the nucleus may be transported between cells; however, other studies have produced conflicting data. These studies examined systemic silencing from tissue-specific expression of either inverted-repeat DNA constructs to produce hpRNA or a mix of sense- and antisense-oriented DNA constructs to produce dsRNA. For example, whereas the transport of RNAi from the pharynx to the bwm was detected when hpRNA was expressed in the pharynx (14), transport of RNAi from bwm cells to other tissues was detected only under certain environmental conditions when hpRNA was expressed in the bwm (20). Further, no transport of RNAi between cells was detected when hpRNA was expressed in the gut (21). Finally, RNAi induced by cell-specific expression of dsRNA was restricted to the cells expressing the dsRNA (22), suggesting that, in contrast to silencing triggered by transgenic expression of hpRNA, silencing triggered by transgenic expression of dsRNA may not be transported between cells. These observations suggest that the ability to export RNAi triggers may be restricted by cell type or RNA structure and may also be regulated by environmental conditions.
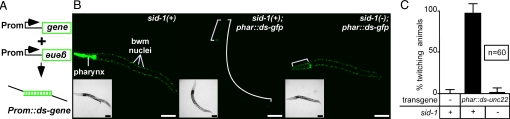
To directly compare systemic silencing triggered by dsRNA expression vs. by hpRNA expression, we expressed gfp-dsRNA in the pharynx, a tissue that shows readily detectable systemic silencing on gfp-hpRNA expression (14). We coinjected DNA constructs that express sense and anti-sense gfp RNA under the control of the pharynx-specific myo-2 promoter (Fig. 2A) into sid-1(+) and sid-1(−) strains that express GFP in both the pharynx and the bwm. In sid-1(+) animals GFP expression was silenced in both the pharynx and the bwm (Fig. 2B, brackets in Middle), but in sid-1(−) animals GFP expression was silenced only in the pharynx (Fig. 2B, bracket in Right). Thus, the nuclear expression of gfp-dsRNA results in the SID-1-dependent transport of RNAi from the pharynx to the bwm cells. To examine the silencing of endogenous genes, we introduced similar constructs that express sense and antisense fragments of the bwm-specific unc-22 gene in the pharynx. Animals that show unc-22 silencing are easily observed as they twitch in response to the paralyzing drug levamisole (3). Expression of the unc-22 sense and antisense constructs in the pharynx resulted in animals that show SID-1-dependent Unc-22 twitching phenotypes (Fig. 2C), demonstrating that expressed dsRNA produces a mobile silencing signal in the pharynx that can move to the bwm. These results and the results from Winston et al. (14) show that pharyngeal expression of either hpRNA or dsRNA can produce effective mobile silencing signals.
Fig. 2.
Double-stranded RNA expressed in the pharynx requires SID-1 to silence target gene in the body-wall muscles (bwm). (A) Schematic showing expression of dsRNA under the control of a specific promoter to silence a target gene (Prom::ds-gene). (B) gfp-dsRNA expression in the pharynx requires sid-1 to silence gfp in the bwm. Representative L4 animals that express gfp in the pharynx and the bwm in either the wild-type background (Left, Middle) or sid-1(−) background (Right), and that in addition express gfp-dsRNA under the control of a pharynx-specific promoter (Middle, Right) are shown. Silenced pharynx and bwm (brackets) are indicated. (Insets): bright-field images. (Scale bar, 100 μm.) (C) unc-22-dsRNA expressed in the pharynx requires sid-1 to silence unc-22 in the bwm. Wild-type animals and animals that express unc-22-dsRNA under the control of a pharynx promoter (phar::ds-unc-22) in either the wild-type or sid-1(−) backgrounds were scored for unc-22 silencing (% twitching). Significant unc-22 silencing was only detected in transgenic animals with a wild-type (sid-1(+)) genetic background (P < 0.0002). Error bars indicate 95% confidence intervals.
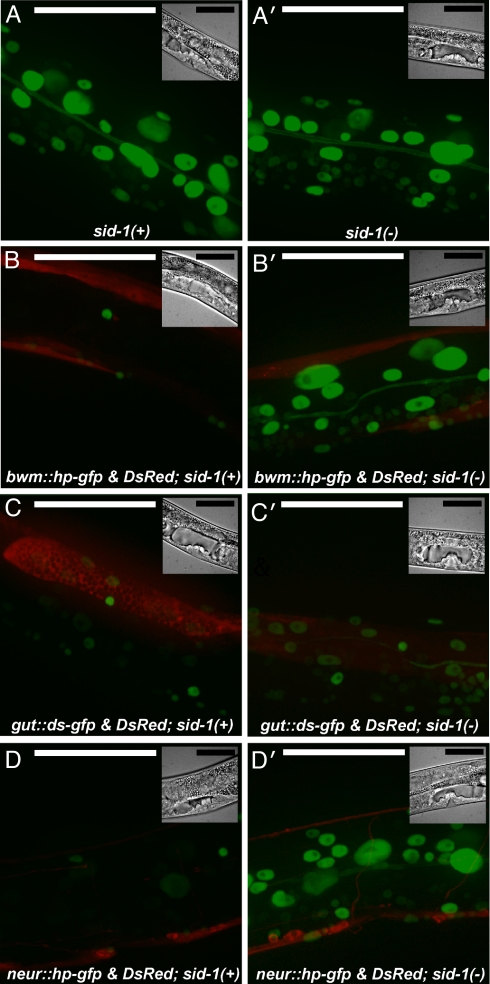
To determine whether nonpharyngeal cells can export silencing signals, we transformed animals that expressed nuclear-localized GFP in all cells (Fig. 3 A and A′) with constructs that coexpressed both gfp-hpRNA and a red fluorescent protein, DsRed, in bwm cells or that coexpressed gfp-dsRNA, and DsRed in the gut. The resulting tandem arrays were not integrated into the genome and were thus mitotically unstable (23) such that only a subset of cells expressed either DsRed and gfp-hpRNA (bwm) or DsRed and gfp-dsRNA (gut). Marking individual cells that express the RNAi trigger (red cells) within a tissue allows the detection of systemic silencing between cells within the same tissue and between cells in different tissues. We found that in addition to the silencing of GFP expression in the red gut and red bwm cells that express the RNAi trigger, silencing of GFP expression was also observed in nonred gut and nonred bwm cells, respectively (data not shown). Further, the muscle-expressed gfp-hpRNA and gut-expressed gfp-dsRNA constructs silenced GFP expression in cells of surrounding tissues (Fig. 3 B and C). Consistent with systemic silencing, when these constructs were introduced into a sid-1(−) background, silencing was restricted to the cells that expressed the RNAi trigger (Fig. 3 B′ and C′). Similar results were obtained when gfp-dsRNA was expressed in the bwm (data not shown). These results show that the silencing in cells that do not express the RNAi trigger is because of SID-1-dependent transport of RNAi from the bwm cells or from gut cells. Thus, the ability to export silencing is not restricted to pharyngeal cells.
Fig. 3.
RNAi triggers expressed in multiple tissues cause systemic silencing of target genes. gfp RNAi triggers expressed in the bwm, in the gut or in neurons show SID-1-dependent systemic silencing of GFP expression in adjacent tissues. Green channel images of representative L4 animals that express nuclear-localized gfp in all somatic cells (sur-5::gfp) in a wild-type (A) or a sid-1(−) (A′) background; and merged green and red channel images of wild-type (B–D) or sid-1(−) (B′–D′) L4 animals that in addition coexpress gfp-hpRNA and DsRed under the control of a bwm-specific promoter (B and B′) or gfp-dsRNA and DsRed under the control of a gut-specific promoter (C and C′) or gfp-hpRNA and DsRed under the control of a neuronal promoter (D and D′) are shown. In addition to the silencing of gfp expression in red cells that express the RNAi trigger, silencing of gfp expression in nonred cells was seen in 100%, 80%, and 100% of wild-type animals that express RNAi triggers in the body-wall muscles, in the gut, and in neurons, respectively. Consistent with SID-1 dependent transport of silencing signals, in the sid-1(−) background, gfp silencing was restricted to red cells that express the RNAi trigger (100%). (Insets): bright-field images. (Scale bar, 50 μm.)
Neurons perform RNAi inefficiently (3) and some neurons (e.g., GABA-ergic motor neurons) do not show detectable RNAi-mediated silencing in wild-type animals. To determine whether neurons can export a silencing signal, we introduced constructs that coexpress gfp-hpRNA and DsRed in neurons into a strain that expresses nuclear-localized GFP in all cells. We found that GFP expression is silenced in most neurons that express gfp-hpRNA (Fig. 3 D and D′), with the notable exception of the GABA-ergic motor neurons, which were not detectably silenced (data not shown). Although the nonred neurons continued to show bright GFP expression, there was robust silencing of GFP expression in all other tissues (Fig. 3D). As expected for a transported silencing signal, silencing in nonred cells was not detectable in sid-1(−) animals (Fig. 3D′). Thus, these results suggest that neurons, which perform RNAi inefficiently, can export silencing signals.
SID-1 Is Not Required for Export of RNAi Triggers.
Because sid-1 is not detectably expressed in neurons (14), the ability of neurons to efficiently export silencing signals suggests that sid-1 may not be required for export. To determine whether SID-1 is required for the export of silencing signals, we generated mosaic sid-1(−) animals in which a tissue that lacks sid-1 expresses gfp-hpRNA (exporting tissue) and monitored the silencing of gfp in another tissue that expresses a rescuing sid-1(+) cDNA (importing tissue). If sid-1 is not required for export then silencing will be observed in the sid-1(+) importing tissue, whereas, if sid-1 is required for export, then silencing will not be observed in the importing tissue.
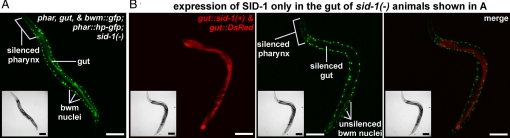
We first expressed gfp-hpRNA specifically in the pharynx of sid-1(−) animals that express gfp in the pharynx, gut, and body-wall muscle cells (Fig. 4A). In these animals, GFP expression is detectably silenced only in the pharynx. To determine whether silencing signals derived from the RNAi trigger expressed in the sid-1(−) pharynx were exported, we expressed sid-1(+) only in the gut of these animals. GFP expression was silenced specifically in the gut, whereas in the bwm cells, which were sid-1(−), GFP expression was not detectably silenced (Fig. 4B). Thus, silencing signals derived from the expression of gfp-hpRNA are exported from the pharynx independent of SID-1.
Fig. 4.
Expression of RNAi triggers in sid-1(−) tissues can silence target genes in sid-1(+) tissues. (A) Representative sid-1(−) young adult that expresses GFP in the pharynx, gut, and bwm, but that expresses gfp-hpRNA only in the pharynx (phar::hp-gfp). Only GFP expression in the pharynx is silenced as indicated. (Inset): bright-field image. (B) Representative young adult of the same genotype as in A, but in which wild-type sid-1 cDNA (sid-1(+)) and DsRed have been coexpressed in the gut. Red channel showing specific expression of sid-1(+) in the gut (Left); green channel showing specific silencing of gfp expression in the pharynx and in the gut (Middle); and merge of images from both channels (Right) are shown. (Insets): bright-field images. (Scale bar, 100 μm.)
To control for the possibility that export from the pharynx may have occurred through SID-1 because of ectopic SID-1 expression in the pharynx, we examined the SID-1 dependence of silencing in multiple tissues. When we introduced constructs that express gfp-hpRNA in all neurons of sid-1(−) animals that express GFP in a subset of neurons and in bwm cells, no silencing of GFP expression was observed. However, subsequent introduction of constructs that express sid-1(+) specifically in bwm cells resulted in robust silencing of GFP expression in bwm cells (Fig. S2), consistent with SID-1-independent export of silencing signals from neurons to bwm. Similarly, silencing signals derived from RNAi triggers expressed in sid-1(−) pharynx silenced GFP expression in sid-1(+) bwm cells (Fig. S2), consistent with SID-1-independent export of silencing signals from pharynx to bwm cells. Taken together, the data suggest that silencing signals derived from expressed RNAi triggers can be exported from tissues independent of SID-1.
Ingested RNAi Triggers Are Transported Across Gut Cells Independent of SID-1.
A common method to deliver RNAi triggers to C. elegans is to feed them E. coli that expresses dsRNA or hpRNA. Ingestion of the RNAi trigger results in systemic silencing of the target gene in most tissues (24). Silencing in gut cells by ingested RNAi triggers requires SID-1 and SID-2, a transmembrane protein found almost exclusively on the gut luminal membrane (25). How the ingested RNAi triggers or a silencing signal derived from such triggers are delivered into the gut or transported from the gut to other internal tissues is unknown. To determine whether SID-1 is required in the gut for the transport of silencing signals from the lumen to internal tissues during feeding RNAi, we generated sid-1(−) mosaic animals where wild-type SID-1 was expressed only in bwm cells and examined silencing of bwm-expressed genes in response to feeding RNAi. If SID-1 is not required in the gut for ingested silencing signals to pass through the gut cells to silence genes in the bwm, then the RNAi trigger is likely transported from the gut lumen to the bwm cells without entry into the cytoplasm of gut cells. If SID-1 is required in the gut for ingested silencing signals to silence genes in the bwm, then the RNAi triggers likely enter the cytoplasm of gut cells before subsequent transport to the bwm cells.
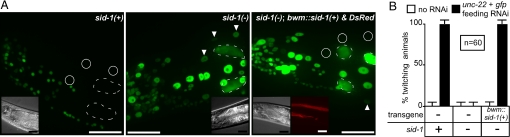
To induce feeding RNAi of genes in the bwm, we used E. coli that express gfp-hpRNA with an unc-22 loop, which triggers the silencing of both gfp and unc-22 likely because of the production of unc-22 dsRNA in the E. coli (14). When wild-type animals that express nuclear-localized GFP in all somatic cells (sur-5::gfp) were fed E. coli that express gfp-hpRNA with an unc-22 loop, GFP expression was efficiently silenced in all nonneuronal cells (Fig. 5A, Left). This silencing was abolished in sid-1(−) animals. However, when a construct that expresses sid-1(+) in the bwm and a construct that expresses DsRed in the bwm were coinjected into these sid-1(−) animals, the resultant red sid-1(+) bwm cells were silenced in response to feeding RNAi (Fig. 5A, compare Middle and Right images). Consistent with a lack of ectopic SID-1 expression in the gut, GFP expression was not detectably silenced in the gut cells of sid-1(−) animals that express sid-1(+) in bwm cells. Thus, the ingested gfp-hpRNA is transported from the lumen of a sid-1(−) gut to sid-1(+) bwm. We then examined unc-22 silencing in the above strains. Silencing unc-22 results in animals that respond to the drug levamisole by twitching, and in a wild-type background, 100% of the animals twitched when fed E. coli that express gfp-hpRNA with an unc-22 loop (Fig. 5B). This twitching was abolished in sid-1(−) animals (Fig. 5B, 0% twitching) but was rescued when sid-1(+) was expressed only in the bwm of sid-1(−) animals (Fig. 5B, 100% twitching). Thus, the ingested gfp-hpRNA with unc-22 loop and unc-22 dsRNA were exported from a sid-1(−) gut to silence unc-22 in the sid-1(+) bwm. These results suggest that ingested RNAi triggers can be transported from the gut lumen into the animal independent of SID-1. To control for undetected misexpression of SID-1 in gut cells from the bwm-specific promoter, we isolated sid-1 genetic mosaics by mitotic segregation of a transgene array that expresses GFP in all cell types and that expresses sid-1 from its native promoter, which is active in all nonneuronal cells. Rare mosaic animals that had lost the array in all gut cells, as monitored by lack of gut GFP expression, but still expressed GFP in bwm and/or hypodermal cells were identified and placed as L4 larvae on E. coli that express gfp-hpRNA. Consistent with the analysis using tissue-specific promoters, compared with control animals, we observed modest silencing of GFP expression (8/8 animals). Therefore, during feeding RNAi, ingested RNAi triggers can be transported from the gut lumen to cells in other tissues without entry into the cytoplasm of gut cells.
Fig. 5.
Ingested RNAi triggers can silence target genes in animals that lack SID-1 in the gut. (A) Expression of sid-1(+) in bwm is sufficient to silence gfp expression in bwm of sid-1(−) animals by feeding RNAi. Representative adult animals that express nuclear-localized gfp in all somatic cells (sur-5::gfp) and that are wild-type (Left), sid-1(−) (Middle), or mosaic sid-1(−) animals that express sid-1(+) only in the bwm (Right) are shown. Animals were fed E. coli that express gfp-hpRNA with an unc-22 loop sequence. bwm nuclei that express GFP (arrowheads) or that show silencing of GFP expression (circle) and gut nuclei (dashed outline), which do not show detectable silencing in sid-1(−) animals, are indicated. Most neuronal nuclei are not detectably silenced in all cases. (Insets): DIC and Red channel images. (Scale bar, 25 μm.) (B) Expression of sid-1(+) in bwm is sufficient to silence unc-22 expression in sid-1(−) animals by feeding RNAi. Wild-type, sid-1(−), and mosaic sid-1(−) animals that express sid-1(+) under the control of a bwm promoter (bwm::sid-1(+)) were fed either E. coli (white bar) or E. coli that express gfp-hpRNA with an unc-22 loop sequence (black bar) and were scored for unc-22 silencing (% twitching). All animals also express sur-5::gfp (as in A). n = 60 animals, error bars indicate 95% confidence intervals.
Discussion
We found that SID-1-dependent mobile silencing signals contribute to transgene silencing in C. elegans. In addition, cells of all tested tissue types can export silencing signals derived from expressed RNAi triggers. Finally, we found that C. elegans cells can use SID-1-independent pathway(s) to export silencing signals upon transgenic expression of or ingestion of RNAi triggers.
Export of RNA Silencing from Cells That Express RNAi Triggers.
Import of RNAi triggers into cells is readily observed in many organisms upon injection of dsRNA into the body cavity (11). For example, in adult Drosophila, intraabdominal injection of dsRNA can silence target genes in the central nervous system (26). In C. elegans, injection of dsRNA into the pseudocoelomic body cavity can cause silencing of target genes in most tissues (3). The ability of cells to import RNAi triggers, suggests that other cells within the organism may act as sources of RNAi triggers. Studies that used tissue-specific expression of RNAi triggers to examine the ability of cells to export silencing signals suggested that only a subset of cells within the organism could export silencing signals. For example, in the C. elegans gut and in Drosophila, silencing by transgenic expression of hpRNAs was apparently restricted to cells that express the RNAi trigger (21, 27). However, we found that expression of RNAi triggers in the cells of most C. elegans tissues, including gut cells and neurons, results in systemic silencing. Therefore, we propose that a mechanism to export silencing signals derived from expressed RNAi triggers exists in most tissues but may be active to different extents in different tissues.
SID-1-Independent Export.
Although C. elegans tissues require SID-1 to import silencing signals, we found that they do not require SID-1 to export silencing signals (Fig. 4 and Fig. S2). Exported silencing signals may be produced in the nucleus or in the cytoplasm. Recent reports show that RNAi that occurs in the nucleus uses effector proteins distinct from those used in the cytoplasm (28), and similar specialized protein machinery may generate the transported silencing signal in either the nucleus or cytoplasm. Intriguingly, many of the mRNAs and microRNAs that are transported in vesicles between cultured mammalian mast cells are not found in the cytoplasm of the exporting cell, consistent with a nuclear origin for these transported RNAs (29).
Why Do Animal Cells Export RNA Silencing?
Numerous dsRNAs or hpRNAs are expressed as precursors to endogenous small RNAs that regulate gene expression (30). These small RNAs include small-interfering RNAs (siRNAs) from natural antisense transcripts, endogenous siRNAs, and microRNAs. Because our results show that RNAi triggers expressed from transgene tandem arrays are transported between animal cells, we propose that silencing signals derived from endogenously expressed dsRNAs or hpRNAs may be transported between cells. A possible function of such transport may be to directly coordinate the expression of target genes between cells in a multicellular organism. Additionally, transport of silencing signals may play a role in the strong silencing of transposons in the germ line (5, 6). However, because sid-1(−) mutants do not have an obvious developmental or behavioral phenotype (14), the transport of endogenous silencing signals may be regulated or may occur only under certain physiological or environmental conditions. For example, exposure to viruses may lead to the systemic transport of silencing signals to curtail the spread of the infection. In support of this possibility, virulence factors of some plant viruses specifically inhibit the transport of silencing signals between cells (4). However, evaluation of this hypothesis in C. elegans awaits the identification of a virus that infects C. elegans in nature.
Transcytosis of Ingested RNAi Triggers?
In addition to the transport of silencing signals between cells within an organism, robust uptake of RNAi triggers from the environment into the organism is readily observed in some animals (10). For example, ingestion of E. coli that expresses RNAi triggers results in systemic silencing of the target gene in most tissues in C. elegans and in planaria. However, how silencing signals are exported to internal tissues from the gut lumen is unknown.
We found that ingested RNAi triggers apparently do not require SID-1 to pass through the C. elegans intestine to silence target genes in sid1(+) tissues that are not exposed to the environment (Fig. 5). SID-1 independent transport of RNAi triggers across the gut was more obvious in sid-1(−) animals where SID-1 was overexpressed in the body-wall muscles (bwm) by using a bwm-specific promoter than in sid-1(−) mosaic animals where SID-1 was expressed under its own promoter in nongut tissues. The difference in the level of silencing observed in the 2 experiments may reflect undetected SID-1 misexpressed in the gut or differences in SID-1 expression levels in target tissues. Knockdown of some vesicle transport proteins in C. elegans results in resistance to feeding RNAi (16), suggesting that ingested RNAi triggers are taken up into vesicles. A parsimonious model that explains the SID-1-independent import of ingested RNAi triggers is transcytosis (31) of the ingested RNAi triggers across the gut. Specifically, we propose that ingested RNAi triggers are endocytosed into vesicles that bud off from the gut lumen, and that these vesicles then release their cargo into the pseudocoelomic cavity by fusing to the basal membrane of the gut. Because most tissues in C. elegans are exposed to the pseudocoelomic cavity, the ingested RNAi triggers can then be taken up through SID-1 into most tissues resulting in systemic silencing throughout the animal.
Materials and Methods
All C. elegans strains were generated and maintained using standard methods (32). Transgenic animals were generated by injecting DNA in 10 mM Tris-Cl pH 8.5 into the germ line by using standard methods (23), and at least 3 independent transgenic lines were analyzed for each experiment. Representative L4 or adult animals were imaged by using exposure times that avoid saturation of pixels by the brightest cells on an Axiovert 200 microscope (Zeiss) and all images within a figure were adjusted in the same way by using Photoshop (Adobe) for display and to enable comparison. Feeding RNAi and unc-22 silencing measurements were adapted from standard procedures (33). By using Wilson's estimates and the proportion of pooled values, 95% confidence intervals for a single proportion and P values for comparison of two proportions were calculated, respectively (34). Detailed procedures are provided in the SI Materials and Methods, and a list of primers used to generate PCR fragments is available in Table S1.
Supplementary Material
Acknowledgments.
We thank Jessica Tanis, Daniel Chase, and members of the Hunter lab; particularly Jennifer Whangbo, Deborah de Jong, Jacqueline M Brooks, Andrea Hinas, Daniel Schott, and Joseph Shih for critical comments on the manuscript; the Caenorhabditis Genetics Center for strains; Yunsoo A Kim for an independent measurement of data in Fig. 1D; and Carlo Garcia for making PCR products for myo-2::unc22sense, myo-2::unc-22antisense, and PF25B3.3::DsRed expression. This work was supported by National Institutes of Health Grant GM069891 (to C.P.H.) and an American Heart Association postdoctoral fellowship (A.M.J.).
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/cgi/content/full/0809760106/DCSupplemental.
References
- 1.White SA, Allshire RC. RNAi-mediated chromatin silencing in fission yeast. Curr Top Microbiol Immunol. 2008;320:157–183. doi: 10.1007/978-3-540-75157-1_8. [DOI] [PubMed] [Google Scholar]
- 2.Hsieh J, Fire A. Recognition and silencing of repeated DNA. Annu Rev Genet. 2000;34:187–204. doi: 10.1146/annurev.genet.34.1.187. [DOI] [PubMed] [Google Scholar]
- 3.Fire A, et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391:806–811. doi: 10.1038/35888. [DOI] [PubMed] [Google Scholar]
- 4.Li F, Ding SW. Virus counterdefense: Diverse strategies for evading the RNA silencing immunity. Annu Rev Microbiol. 2006;60:503–531. doi: 10.1146/annurev.micro.60.080805.142205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ketting RF, Haverkamp TH, van Luenen HG, Plasterk RH. Mut-7 of C. elegans, required for transposon silencing and RNA interference, is a homolog of Werner syndrome helicase and RNaseD. Cell. 1999;99:133–141. doi: 10.1016/s0092-8674(00)81645-1. [DOI] [PubMed] [Google Scholar]
- 6.Tabara H, et al. The rde-1 gene, RNA interference, and transposon silencing in C elegans. Cell. 1999;99:123–132. doi: 10.1016/s0092-8674(00)81644-x. [DOI] [PubMed] [Google Scholar]
- 7.Kelly WG, Xu S, Montgomery MK, Fire A. Distinct requirements for somatic and germline expression of a generally expressed Caenorhabditis elegans gene. Genetics. 1997;146:227–238. doi: 10.1093/genetics/146.1.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Palauqui JC, Elmayan T, Pollien JM, Vaucheret H. Systemic acquired silencing: Transgene-specific post-transcriptional silencing is transmitted by grafting from silenced stocks to nonsilenced scions. EMBO J. 1997;16:4738–4745. doi: 10.1093/emboj/16.15.4738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Voinnet O, Baulcombe DC. Systemic signaling in gene silencing. Nature. 1997;389:553. doi: 10.1038/39215. [DOI] [PubMed] [Google Scholar]
- 10.Whangbo JS, Hunter CP. Environmental RNA interference. Trends Genet. 2008;24:297–305. doi: 10.1016/j.tig.2008.03.007. [DOI] [PubMed] [Google Scholar]
- 11.Jose AM, Hunter CP. Transport of sequence-specific RNA interference information between cells. Annu Rev Genet. 2007;41:305–330. doi: 10.1146/annurev.genet.41.110306.130216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Voinnet O, Vain P, Angell S, Baulcombe DC. Systemic spread of sequence-specific transgene RNA degradation in plants is initiated by localized introduction of ectopic promoterless DNA. Cell. 1998;95:177–187. doi: 10.1016/s0092-8674(00)81749-3. [DOI] [PubMed] [Google Scholar]
- 13.Dunoyer P, Himber C, Ruiz-Ferrer V, Alioua A, Voinnet O. Intra- and intercellular RNA interference in Arabidopsis thaliana requires components of the microRNA and heterochromatic silencing pathways. Nat Genet. 2007;39:848–856. doi: 10.1038/ng2081. [DOI] [PubMed] [Google Scholar]
- 14.Winston WM, Molodowitch C, Hunter CP. Systemic RNAi in C. elegans requires the putative transmembrane protein SID-1. Science. 2002;295:2456–2459. doi: 10.1126/science.1068836. [DOI] [PubMed] [Google Scholar]
- 15.Wolfrum C, et al. Mechanisms and optimization of in vivo delivery of lipophilic siRNAs. Nat Biotechnol. 2007;25:1149–1157. doi: 10.1038/nbt1339. [DOI] [PubMed] [Google Scholar]
- 16.Saleh MC, et al. The endocytic pathway mediates cell entry of dsRNA to induce RNAi silencing. Nat Cell Biol. 2006;8:793–802. doi: 10.1038/ncbl439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Feinberg EH, Hunter CP. Transport of dsRNA into cells by the transmembrane protein SID-1. Science. 2003;301:1545–1547. doi: 10.1126/science.1087117. [DOI] [PubMed] [Google Scholar]
- 18.Grishok A, Sinskey JL, Sharp PA. Transcriptional silencing of a transgene by RNAi in the soma of C. elegans. Genes Dev. 2005;19:683–696. doi: 10.1101/gad.1247705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim JK, et al. Functional genomic analysis of RNA interference in C. elegans. Science. 2005;208:1164–1167. doi: 10.1126/science.1109267. [DOI] [PubMed] [Google Scholar]
- 20.Timmons L, Tabara H, Mello CC, Fire AZ. Inducible systemic RNA silencing in Caenorhabditis elegans. Mol Biol Cell. 2003;14:2972–2983. doi: 10.1091/mbc.E03-01-0858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tijsterman M, May RC, Simmer F, Okihara KL, Plasterk RH. Genes required for systemic RNA interference in Caenorhabditis elegans. Curr Biol. 2004;14:111–116. doi: 10.1016/j.cub.2003.12.029. [DOI] [PubMed] [Google Scholar]
- 22.Esposito G, Di Schiavi E, Bergamasco C, Bazzicalupo P. Efficient and cell specific knock-down of gene function in targeted C. elegans neurons. Gene. 2007;395:170–176. doi: 10.1016/j.gene.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 23.Mello CC, Kramer JM, Stinchcomb D, Ambros V. Efficient gene transfer in C. elegans: Extrachromosomal maintenance and integration of transforming sequences. EMBO J. 1991;10:3959–3970. doi: 10.1002/j.1460-2075.1991.tb04966.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Timmons L, Fire A Specific interference by ingested dsRNA. Nature. 1998;395:854. doi: 10.1038/27579. [DOI] [PubMed] [Google Scholar]
- 25.Winston WM, Sutherlin M, Wright AJ, Feinberg EH, Hunter CP. Caenorhabditis elegans SID-2 is required for environmental RNA interference. Proc Natl Acad Sci USA. 2007;104:10565–10570. doi: 10.1073/pnas.0611282104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dzitoyeva S, Dimitrijevic N, Manev H. Gamma-aminobutyric acid B receptor 1 mediates behavior-impairing actions of alcohol in Drosophila: Adult RNA interference and pharmacological evidence. Proc Natl Acad Sci USA. 2003;100:5485–5490. doi: 10.1073/pnas.0830111100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roignant JY, et al. Absence of transitive and systemic pathways allows cell-specific and isoform-specific RNAi in Drosophila. RNA. 2003;9:299–308. doi: 10.1261/rna.2154103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guang S, et al. An Argonaute transports siRNAs from the cytoplasm to the nucleus. Science. 2008;321:537–541. doi: 10.1126/science.1157647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Valadi H, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654–659. doi: 10.1038/ncb1596. [DOI] [PubMed] [Google Scholar]
- 30.Okamura K, Lai EC. Endogenous small interfering RNAs in animals. Nat Rev Mol Cell Biol. 2008;9:673–678. doi: 10.1038/nrm2479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tuma PL, Hubbard AL. Transcytosis: Crossing barriers. Physiol Rev. 2003;83:871–932. doi: 10.1152/physrev.00001.2003. [DOI] [PubMed] [Google Scholar]
- 32.Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77:71–94. doi: 10.1093/genetics/77.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Parrish S, Fleenor J, Xu S, Mello C, Fire A. Functional anatomy of a dsRNA trigger: Differential requirement for the two trigger strands in RNA interference. Mol Cell. 2000;6:1077–1087. doi: 10.1016/s1097-2765(00)00106-4. [DOI] [PubMed] [Google Scholar]
- 34.Moore DS, McCabe GP. Introduction to the Practice of Statistics. 4th Ed. New York: W.H. Freeman; 2003. pp. 571–601. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.