Abstract
Several modifiable pre- and postnatal determinants of childhood overweight are known, but no one has examined how they influence risk of overweight in combination. We estimated the risk of overweight at age 3 years according to levels of maternal smoking during pregnancy, gestational weight gain, breastfeeding duration, and infant sleep duration. We studied 1,110 mother–child pairs in Project Viva, a prospective prebirth cohort study. The main outcome measure was child overweight (BMI for age and sex ≥95th percentile) at age 3. We ran logistic regression models with all four modifiable risk factors as well as the covariates maternal BMI and education, child race/ethnicity, and household income. From the model, we obtained the estimated probability of overweight for each of the 16 combinations of the four risk factors. During pregnancy, 9.8% of mothers smoked and 50% gained excessive weight. In infancy, 73% mother–child pairs breastfed for <12 m, and 31% of infants slept <12 h/day. Among the 3-year-old children in the cohort, 9.5% were overweight. In the prediction model, the estimated probability of overweight ranged from 0.06 among children exposed to favorable levels of all four risk factors, to 0.29 with adverse levels of all four. Healthful levels of four behaviors during early development predicted much lower probability of overweight at age 3 than adverse levels. Interventions to modify several factors during pregnancy and infancy could have substantial impact on prevention of childhood overweight.
INTRODUCTION
The obesity epidemic has spared no age group, even young infants (1). The implication is that prevention of childhood obesity should begin at the earliest stages of human development. In recent years, investigators have identified several potentially modifiable factors in the prenatal and early postnatal periods that increase the risk of overweight in childhood. These include maternal smoking during pregnancy (2-5), excessive gestational weight gain (6-10), reduced breastfeeding duration (11), rapid weight gain during the first few months of life (12-15), and short sleep duration (16,17).
Although studies have reported independent associations of each of these factors individually with later overweight, no previous study has assessed their combined impact. Such an assessment would provide estimates of the potential public health impact of developmentally appropriate interventions conducted among pregnant women and infants to prevent obesity later in childhood.
The purpose of this study was to estimate the probability of overweight at age 3 years for different combinations of four modifiable risk factors occurring prenatally and during the first year of life.
METHODS OF PROCEDURES
Participants
The subjects for this study were participants in Project Viva, a prospective, observational cohort study of gestational factors, pregnancy outcomes, and offspring health. We recruited women who were attending their initial prenatal visit at eight urban and suburban obstetrical offices of a multispecialty group practice in eastern Massachusetts. Eligibility criteria included fluency in English, gestational age <22 weeks at the initial prenatal clinical appointment, and singleton pregnancy. Details of recruitment and retention procedures are available elsewhere (18).
Of the 2,128 women who delivered a live infant, 1,579 were eligible for 3-year follow-up by virtue of having completed prenatal nutritional assessments and consenting for their children to be followed up. We collected follow-up information on 1,397 (88% of 1579), including in-person examinations on 1,283 (81%). For this analysis, we excluded 20 of the 1,283 participants whose gestational age at birth was <34 weeks, 28 participants missing height or weight at 3 years, and 125 participants missing values for any of the four risk factors, leaving a cohort for analysis of 1,110 mother–child pairs.
After obtaining informed consent, we performed in-person study visits with the mother at the end of the first and second trimesters of pregnancy, and with both mother and child immediately after delivery and at 6 months and 3 years after birth. Mothers completed mailed questionnaires at 1 and 2 years after birth. We also obtained information from medical records. Institutional review boards of participating institutions approved the study. All procedures were in accordance with the ethical standards for human experimentation established by the Declaration of Helsinki.
Comparison of the 1,110 participants in this analysis with the 1,579 who were eligible for 3-year follow-up showed a higher proportion of maternal white race (76% vs. 69%), college or graduate education (74% vs. 68%), and annual household income exceeding $70,000 (66 vs. 62%), and slightly lower smoking rates during pregnancy (10% vs. 12%) but did not differ on mean maternal prepregnancy BMI, gestational weight gain, or infant birth weight.
Measurements
Risk factors
We focused on factors during the prenatal period and infancy that have been associated with childhood overweight, are likely to be causal, and are potentially modifiable. Foregoing nuance for simplicity, we elected to use each of the four risk factors in dichotomous form. That decision limited the estimated probabilities to a tractable 16 combinations of the four risk factors.
Smoking during pregnancy
We asked mothers at both first and second trimester visits about their cigarette smoking habits before and during pregnancy. In Project Viva (2) and other cohort studies (3-5), smoking during pregnancy is associated with increased risk of overweight during childhood. A meta-analysis of 14 studies shows that the adjusted odds ratio for overweight among children of women who smoked during pregnancy was 1.50 (95% confidence interval (CI) 1.36−1.65) (19). In this analysis, we compared women who smoked during pregnancy with those who never smoked or quit before becoming pregnant.
Gestational weight gain
At the first trimester study visit, we asked mothers to report their weights just before they became pregnant. From medical records, we collected all weights measured during prenatal visits. We calculated total weight gain by subtracting pre-pregnancy weight from the last prenatal weight. In the Project Viva cohort, we have recently shown that excessive weight gain during pregnancy is associated with increased risk of overweight at age 3; compared with inadequate gain, the odds ratio for BMI exceeding the 95th percentile (vs. <50th percentile) was 4.35 (95% CI 1.69, 11.24) (7). Other preliminary data support this association (6,8-10). In this analysis, we expressed gestational weight gain in categories based on the 1990 recommendations of the Institute of Medicine (20), which advise less weight gain for mothers with higher pre-pregnancy BMI. For example, for a woman of normal weight BMI (19.8−26.0 kg/m2), the Institute of Medicine guidelines recommend a total weight gain during pregnancy of 11.5−16 kg, whereas for a woman with high BMI (26.1−29.0 kg/m2) the recommended gain is 7−11.5 kg. We used this same range, 7−11.5 kg, for obese women (>29.0 kg/m2). In this analysis, we compared excessive maternal weight gain with adequate or inadequate gain, and to be consistent across all variables we defined normal childhood weight as BMI 5th–85th percentile rather than <50th percentile.
Breastfeeding duration
At 6, 12, and 24 months, we asked mothers whether they were still breastfeeding at all. If they had stopped, we asked them the children's age at cessation. Although the magnitude of the association between breastfeeding and later overweight is still controversial (21-23), the only meta-analysis examining duration of breastfeeding demonstrated a substantial decrease in odds (4% (95% CI 2−6%)) of later overweight for each additional month of breastfeeding (11). In this analysis, we compared any breastfeeding duration of <12 months with at least 12 months, which is recommended by the American Academy of Pediatrics (24).
Daily sleep during infancy
At 6 and 12 months postpartum, we asked mothers to quantify the average amount of daily sleep their children obtained over the past month. The questions prompted them to include morning and afternoon naps in addition to nighttime sleep. Using these questions, we have recently shown in the Project Viva cohort that average daily sleep duration from 6 to 24 months is inversely associated with overweight at 3 years (16). In that analysis, comparing average daily sleep duration of <12 with at least 12 h/day, the odds ratio for overweight was 2.04 (95% CI 1.07, 3.91). Here, we limited the sleep variable to 6 and 12 months because we were interested in the first year after birth. We used the mean of 6-month and 12-month sleep among the 914 participants with data at both time-points; in the remainder of participants one time point was missing, so we used the nonmissing value to represent the mean. In this analysis, we compared daily sleep duration of <12 h with at least 12 h, as in our published work (16).
Outcome measures
At 3 years, we measured weight using a calibrated scale (Seca model 881; Seca, Hanover, MD) and length using a calibrated stadiometer (Shorr Productions, Olney, MD), and we calculated age- and sex-specific BMI z-scores using US national reference data (25). We defined overweight as BMI ≥95th percentile for age and sex, and used BMI 5th–85th percentile as the comparison.
Other measures
Using a combination of self-administered questionnaires and interviews, we collected information about maternal pre-pregnancy weight, height, age, education, and parity, father's weight and height, child's age, sex, and race/ethnicity, and household income. We calculated birth weight for gestational age z-score as a measure of fetal growth (26). As a measure of infant weight gain, we calculated the difference between weight-for-age z-scores at 6 months and at birth (25). Both fetal growth and rapid early infancy weight gain are directly associated with later BMI in childhood or adulthood (12-15,27). In this analysis, we considered fetal growth and infant weight gain to be “nonmodifiable” factors that might be intermediates in the causal pathways between the four risk factors and childhood obesity. At age 3, we estimated energy intake from a validated food frequency questionnaire (28), a weighted average of hours of television watched on weekdays and weekend days, and physical activity by asking mothers to report whether their children were less active, more active, or about the same as other children. As these three variables reflect energy intake and expenditure, we also considered them to be potential mediators.
Statistical analysis
To obtain estimates of the independent association of each risk factor with the outcome, we used logistic regression models that included all four modifiable risk factors as well as the four “nonmodifiable” determinants maternal prepregnancy BMI and education, child race/ ethnicity, and household income. Here, we consider these four factors nonmodifiable because usually they are fixed from the few months before pregnancy through 1 year after pregnancy. In preliminary modeling, we also included six additional potential confounding variables, namely maternal age and parity, paternal BMI, duration of gestation, and child sex and exact age. Because their inclusion did not materially change the effect estimates for the four risk factors, we left them out of the final model. Note that child age and sex are de facto in the final model because the BMI percentile calculation includes them.
Using parameter estimates from the multivariate logistic models, we estimated the predicted probability of overweight for each of the 16 combinations of the four risk factor variables. To estimate predicted probabilities, we needed to choose fixed values for the nonmodifiable determinants, and we selected a “typical” participant. By typical, we mean an individual with the modal values of race/ethnicity, education, and income, and the mean value of BMI, i.e, one who was white, and who had a mother with a bachelor's degree and a BMI of 24.5 kg/m2, and a household income between $40,000 and $70,000. In this article, we show estimates for the 16 combinations of the four risk factors. In secondary analyses, we additionally adjusted for the two potentially mediating variables fetal growth and infant weight gain from birth to 6 months. We conducted all analyses using SAS, version 9.1 (Cary, NC).
RESULTS
At age 3, mean (s.d.) for BMI z-score was 0.46 (1.0) and 9.5% were overweight. The proportion of mothers who smoked during pregnancy was 9.8%; 50% experienced excessive weight gain. Seventy-three percent of infants were breastfed for <12 months, and 31% slept an average of <12 h/day. Table 1 shows the extent to which adverse levels of each of the four risk factors were independently associated with overweight at 3 years. The four factors were associated with odds ratios in the 1.3−1.8 range, some with wider CIs than others.
Table 1.
Prevalence of four modifiable and several nonmodifiable factors during pregnancy and infancy, and adjusted odds of overweight at 3 years of age
|
Overweight at age 3 years |
|||
|---|---|---|---|
| No (BMI 5th—8th percentile) n = 791 | Yes (BMI > 95th percentile) n = 105 | Adjustedaodds of overweight | |
| Modifiable risk factors | N (% of subjects) | ||
| Prenatal smoking | |||
| No | 725 (92) | 88 (84) | 1.0 (Ref) |
| Yes | 66 (8) | 17 (16) | 1.71 (0.90, 3.25) |
| Gestational weight gainb | |||
| Inadequate or adequate | 407 (51) | 45 (43) | 1.0 (Ref) |
| Excessive | 384 (49) | 60 (57) | 1.32 (0.85, 2.03) |
| Breastfeeding duration (m) | |||
| >12 | 222 (28) | 18 (17) | 1.0 (Ref) |
| <12 | 569 (72) | 87 (83) | 1.50 (0.86, 2.64) |
| Infant sleep (h/day) | |||
| >12 | 563 (71) | 60 (57) | 1.0 (Ref) |
| <12 | 228 (29) | 45 (43) | 1.83 (1.17, 2.85) |
| Nonmodifiable factors included in multivariate model | |||
| Maternal education | |||
| Some college or less | 198 (25) | 37 (35) | 1.27 (0.66, 2.43) |
| BA or BS | 301 (38) | 43 (41) | 1.41 (0.82, 2.42) |
| Graduate degree | 292 (37) | 25 (24) | 1.0 (Ref) |
| Household income | |||
| Don't know or missing | 57 (7) | 10 (10) | 1.32 (0.61, 2.87) |
| <$40,000 | 86 (11) | 15 (14) | 0.73 (0.32, 1.66) |
| $40,000−$70,000 | 144 (18) | 25 (24) | 1.07 (0.61, 1.89) |
| >$70,000 | 504 (64) | 55 (52) | 1.0 (Ref) |
| Child race/ethnicity | |||
| White | 571 (72) | 68 (65) | 1.0 (Ref) |
| Black | 81 (10) | 15 (14) | 1.02 (0.49, 2.13) |
| Hispanic | 27 (3) | 8 (8) | 1.86 (0.72, 4.78) |
| Other | 112 (14) | 14 (13) | 0.91 (0.48, 1.74) |
| Mean (s.d.) | |||
| Maternal pre-pregnancy BMI (kg/m2) | 24.0 (4.7) | 27.5 (6.4) | 1.11 (1.07, 1.15) |
| Other non-modifiable factors | |||
| N (% of subjects) | |||
| Nulliparous | 386 (49) | 39 (37) | |
| Child sex (F) | 407 (51) | 42 (40) | |
| Weight gain birth to 6 m | |||
| Quartile 1−3 | 602 (81) | 51 (52) | |
| Quartile 4 | 144 (19) | 48 (48) | |
| Mean (s.d.) | |||
| Maternal age at enrolment (y) | 32.6 (5.0) | 33.0 (4.7) | |
| Paternal BMI (kg/m2) | 26.1 (3.6) | 28.6 (4.8) | |
| Gestation length (weeks) | 39.6 (1.4) | 39.5 (1.4) | |
| Fetal growth (birth weight/gestational age z score) | 0.13 (0.93) | 0.53 (0.84) | |
| Child age at outcome measurement (y) | 3.3 (0.3) | 3.3 (0.3) | |
Data from 1,110 mother—infant pairs participating in Project Viva.
Model covariates include all four modifiable risk factors and the four “nonmodifiable” variables maternal education and BMI, household income, and child race/ethnicity.
Gestational weight gain categories according to 1990 recommendations of the Institute of Medicine (19).
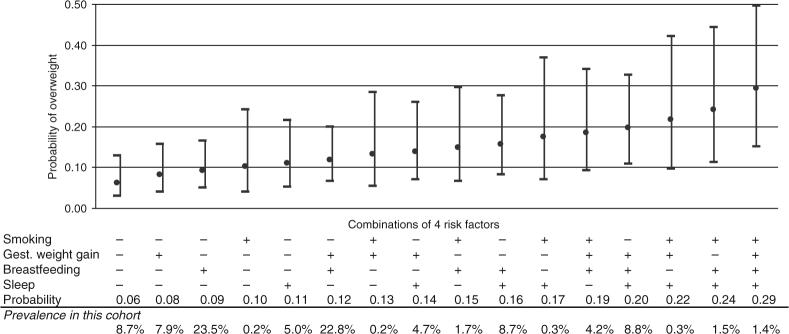
We next modeled the covariate-adjusted predicted probability of 3-year overweight for varying combinations of the four modifiable risk factors. In Figure 1, we show predicted probabilities for the 16 combinations of the four factors. Participants with favorable levels of all four risk factors had a predicted probability of 3-year overweight of 6%, whereas those with adverse levels of all four had a probability of 29%. Mixtures of favorable and adverse risk factor levels resulted in probabilities between these extremes.
Figure 1.
| Definitions of risk factors | + | − |
|---|---|---|
| Smoking in pregnancy | Yes | No |
| Gestational weight gaina | “Excessive” | “Adequate or inadequate” |
| Breastfeeding duration (months) | <12 | ≥12 |
| Daily sleep in infancy (h/day) | <12 | ≥12 |
In additional models that included the potentially mediating variables fetal growth and infant weight gain, the range of probabilities for the 16 combinations of the four risk factors was from 0.04 to 0.20, which is slightly lower and narrower than the main result of 0.06−0.29. With these additional variables in the model, odds ratios for the four risk factors were not much changed: 1.6 instead of 1.7 for maternal smoking, 1.2 vs. 1.3 for gestational weight gain, 1.8 vs. 1.5 for breastfeeding, and 1.9 vs. 1.8 for infant sleep duration. A similar mediation analysis with 3-year energy intake, television watching, and physical activity showed similar results, i.e., a range of probabilities of 0.04−0.20.
DISCUSSION
In this study, the predicted probability of overweight in childhood varied from 6 to 29%, depending on the levels of four potentially modifiable risk factors in pregnancy and infancy— maternal smoking during pregnancy, gestational weight gain, breastfeeding duration, and daily sleep duration during infancy. This wide range of risk of childhood overweight implies that developmentally appropriate interventions to modify these factors may have substantial impact on preventing childhood obesity and its consequences.
Research into developmental origins of health and disease suggests that factors operating before and soon after birth can alter occurrence of chronic disease throughout the lifecourse (29). Some critics, however, have questioned the magnitude of public health impact of modifying the pre- and postnatal environment. A previous attempt at quantifying long-term effects invoked birth weight as the single perinatal factor (30). But birth weight, though easily measured, is unfortunately a poor proxy for true etiologic factors operating before and after birth (31). Likewise, although several studies implicate rapid infant weight gain in the genesis of later obesity (12-15), its modifiability is debatable beyond the potential effect of infant feeding, which we included as a risk factor in our analysis. In this study, we focused on four factors that are likely to be causal and potentially modifiable through clinical or public health interventions.
In most populations, the number of children with extreme risk factor profiles, e.g., those resulting in a 29% risk in this study, is likely to be small. Nevertheless, many mother–infant pairs in the intermediate categories would have room for substantial risk reduction. Consider, for example, a 3-year-old whose mother had smoked and gained an excessive amount of weight during pregnancy, and who had been breastfed for <12 months, but slept for at least 12 h/day during infancy. The estimated probability of overweight for such a child would be 19% (Figure 1), which is likely to approximate the prevalence of overweight in US children by 2010 (32). If the mother had not smoked or gained excessive weight during pregnancy and had breastfed for at least 12 months, the probability of overweight at 3 would be only 6%, assuming sleep duration did not change.
These are potentially achievable changes. In the developed world, smoking rates among pregnant women have risen and then declined in the past several decades (33), and smoking cessation before or even during pregnancy is attainable (34). Although no randomized controlled trials exist to show that moderating gestational weight gain is feasible, one trial with historical controls showed reduction in risk of excessive weight gain among low-income women (35). In addition, wide fluctuations have occurred over the past 50 years in the amount of weight women gain during pregnancy, likely due to health professionals changing their recommendations over time (36). After falling in the early to mid 20th century, breastfeeding rates have now risen from their nadir >30 years ago (37), and a large randomized trial of breastfeeding promotion resulted in substantial increases in duration and exclusivity of breastfeeding (38). Sleep duration during infancy appears modifiable as well (39).
This study has several strengths. They include prospective data collection and the ability to adjust for several important confounding factors. The study also has several potential limitations. Gestational diabetes is associated with offspring overweight in some studies (40), but was not prevalent enough in our cohort (n = 42, 3.8%) to include as a risk factor. Including gestational diabetes status as a covariate hardly changed the estimated range of probabilities (from 0.06−0.29 to 0.06−0.30). Our main outcome was overweight at age 3. Overweight at this age does not predict adult consequences as well as overweight later in childhood (41) but can nevertheless presage serious adverse health consequences in childhood itself (42).
Some of our exposure–outcome associations were not “statistically significant” according to the common but arbitrary standard of a P value <0.05. Nonetheless, as detailed in the Methods and Procedures section, other studies and meta-analyses have shown similar magnitudes of association with more precision than was possible in our single study, and the imprecision of our estimates is manifest in the width of the CIs (Figure 1). As in other studies (43), maternal BMI was a strong predictor of childhood obesity. Prevention of maternal obesity entering pregnancy is a public health priority, but was not the focus of this study, in which we evaluated modifiable factors once pregnancy begins.
Most measures were from self-report, including prepregnancy weight, smoking, breastfeeding, and infant sleep, and loss to follow-up was not random. These factors could have introduced bias. We obtained results from one cohort of mothers and children from Massachusetts. The quantitative results may not be directly applicable to other populations, chiefly because the frequencies of risk factors or overweight may differ. That is not a true limitation of the study, rather just a reminder that absolute risks differ across populations as covariate frequencies do. For example, in our calcuations of predicted probabilities if we had chosen as a “typical” participant, a woman with BMI of 30 instead of the sample population mean of 24.5 kg/ m2, the range of predicted probabilities would have been 0.11− 0.43 instead of 0.06−0.29. Regardless, more important than the exact probabilities is the message that varying levels of potentially modifiable early developmental factors predict a wide range of probabilities of childhood overweight.
In summary, we found that combinations of four modifiable factors in pregnancy and infancy can predict quite low or quite high risk of childhood overweight, in this cohort from well under 10 to almost 30%. Given the short- and long-term adverse consequences of childhood obesity, these findings suggest how crucial early developmental factors may be in determining obesity-related consequences across the life course. They also provide a strong rationale for testing developmentally appropriate pre- and postnatal interventions to prevent childhood overweight.
ACKNOWLEDGEMENTS
We thank the staff and participants of Project Viva. This work was funded by grants from the US National Institutes of Health (HD 34568, HL 64925, HL 68041, HL 075504, HD 044807), Harvard Medical School and the Harvard Pilgrim Health Care Foundation.
Footnotes
DISCLOSURE
In the past 12 months, M.W.G. has received grant funding from sanofiaventis, but that grant was not related to this analysis. All other authors declare no conflicts of interest.
REFERENCES
- 1.Kim J, Peterson KE, Scanlon KS, et al. Trends in overweight from 1980 through 2001 among preschool-aged children enrolled in a health maintenance organization. Obesity (Silver Spring) 2006;14:1107–1112. doi: 10.1038/oby.2006.126. [DOI] [PubMed] [Google Scholar]
- 2.Oken E, Huh SY, Taveras EM, Rich-Edwards JW, Gillman MW. Associations of maternal prenatal smoking with child adiposity and blood pressure. Obes Res. 2005;13:2021–2028. doi: 10.1038/oby.2005.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen A, Pennell ML, Klebanoff MA, Rogan WJ, Longnecker MP. Maternal smoking during pregnancy in relation to child overweight: follow-up to age 8 years. Int J Epidemiol. 2006;35:121–130. doi: 10.1093/ije/dyi218. [DOI] [PubMed] [Google Scholar]
- 4.Power C, Jefferis BJ. Fetal environment and subsequent obesity: a study of maternal smoking. Int J Epidemiol. 2002;31:413–419. [PubMed] [Google Scholar]
- 5.von Kries R, Toschke AM, Koletzko B, Slikker W., Jr Maternal smoking during pregnancy and childhood obesity. Am J Epidemiol. 2002;156:954–961. doi: 10.1093/aje/kwf128. [DOI] [PubMed] [Google Scholar]
- 6.Fisch RO, Bilek MK, Ulstrom R. Obesity and leanness at birth and their relationship to body habitus in later childhood. Pediatrics. 1975;56:521–528. [PubMed] [Google Scholar]
- 7.Oken E, Taveras EM, Kleinman KP, Rich-Edwards JW, Gillman MW. Gestational weight gain and child adiposity at age 3 years. Am J Obstet Gynecol. 2007;196:322.e1–322.e8. doi: 10.1016/j.ajog.2006.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sharma AJ, Cogswell ME, Grummer-Strawn LM. The association between pregnancy weight gain and childhood overweight is modified by mother's pre-pregnancy BMI. Pediatr Res. 2005;58:1038. [Google Scholar]
- 9.Seidman DS, Laor A, Shemer J, Gale R, Stevenson DK. Excessive maternal weight gain during pregnancy and being overweight at 17 years of age. Pediatr Res. 1996;39:112A. [Google Scholar]
- 10.Schack-Nielsen L, Mortensen EL, Michaelsen KF, Sorensen TIA. High maternal pregnancy weight gain is associated with an increased risk of obesity in childhood and adulthood independent of maternal BMI. Pediatr Res. 2005;58:1020. [Google Scholar]
- 11.Harder T, Bergmann R, Kallischnigg G, Plagemann A. Duration of breastfeeding and risk of overweight: a meta-analysis. Am J Epidemiol. 2005;162:397–403. doi: 10.1093/aje/kwi222. [DOI] [PubMed] [Google Scholar]
- 12.Monteiro PO, Victora CG. Rapid growth in infancy and childhood and obesity in later life—a systematic review. Obes Rev. 2005;6:143–154. doi: 10.1111/j.1467-789X.2005.00183.x. [DOI] [PubMed] [Google Scholar]
- 13.Baird J, Fisher D, Lucas P, et al. Being big or growing fast: systematic review of size and growth in infancy and later obesity. BMJ. 2005;331:929–934. doi: 10.1136/bmj.38586.411273.E0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stettler N, Stallings VA, Troxel AB, et al. Weight gain in the first week of life and overweight in adulthood: a cohort study of European American subjects fed infant formula. Circulation. 2005;111:1897–1903. doi: 10.1161/01.CIR.0000161797.67671.A7. [DOI] [PubMed] [Google Scholar]
- 15.Ong KK, Loos RJ. Rapid infancy weight gain and subsequent obesity: systematic reviews and hopeful suggestions. Acta Paediatr. 2006;95:904–908. doi: 10.1080/08035250600719754. [DOI] [PubMed] [Google Scholar]
- 16.Taveras EM, Rifas-Shiman SL, Oken E, Gunderson EP, Gillman MW. Short sleep duration in infancy and risk of childhood overweight. Arch Pediatr Adolesc Med. 2008;162:305–311. doi: 10.1001/archpedi.162.4.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Reilly JJ, Armstrong J, Dorosty AR, et al. Early life risk factors for obesity in childhood: cohort study. BMJ. 2005;330:1357–1363. doi: 10.1136/bmj.38470.670903.E0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gillman MW, Rich-Edwards JW, Rifas-Shiman SL, et al. Maternal age and other predictors of newborn blood pressure. J Pediatr. 2004;144:240–245. doi: 10.1016/j.jpeds.2003.10.064. [DOI] [PubMed] [Google Scholar]
- 19.Oken E, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes (Lond) 2008;32:201–210. doi: 10.1038/sj.ijo.0803760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Institute of Medicine . Nutrition During Pregnancy. National Academy Press; Washington, DC: 1990. Part I, Weight Gain. [Google Scholar]
- 21.Owen CG, Martin RM, Whincup PH, Smith GD, Cook DG. Effect of infant feeding on the risk of obesity across the life course: a quantitative review of published evidence. Pediatrics. 2005;115:1367–1377. doi: 10.1542/peds.2004-1176. [DOI] [PubMed] [Google Scholar]
- 22.Owen CG, Martin RM, Whincup PH, et al. The effect of breastfeeding on mean body mass index throughout life: a quantitative review of published and unpublished observational evidence. Am J Clin Nutr. 2005;82:1298–1307. doi: 10.1093/ajcn/82.6.1298. [DOI] [PubMed] [Google Scholar]
- 23.Arenz S, Ruckerl R, Koletzko B, von Kries R. Breast-feeding and childhood obesity—a systematic review. Int J Obes Relat Metab Disord. 2004;28:1247–1256. doi: 10.1038/sj.ijo.0802758. [DOI] [PubMed] [Google Scholar]
- 24.Committee on Nutrition of the American Academy of Pediatrics . Pediatric Nutrition Handbook. 5th edn. American Academy of Pediatrics; Elk Grove Village, IL: 2004. [Google Scholar]
- 25.National Center for Health Statistics CDC Growth Charts, United States. 2000 < http://www.cdc.gov/growthcharts/>.
- 26.Oken E, Kleinman KP, Rich-Edwards JW, Gillman MW. A nearly continuous measure of birth weight for gestational age using a United States national reference. BMC Pediatr. 2003;3:6. doi: 10.1186/1471-2431-3-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Oken E, Gillman MW. Fetal origins of obesity. Obes Res. 2003;11:496–506. doi: 10.1038/oby.2003.69. [DOI] [PubMed] [Google Scholar]
- 28.Blum RE, Wei EK, Rockett HR, et al. Validation of a food frequency questionnaire in Native American and Caucasian children 1 to 5 years of age. Matern Child Health J. 1999;3:167–172. doi: 10.1023/a:1022350023163. [DOI] [PubMed] [Google Scholar]
- 29.Gillman MW. Developmental origins of health and disease. N Engl J Med. 2005;353:1848–1850. doi: 10.1056/NEJMe058187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Barker DJ, Eriksson JG, Forsen T, Osmond C. Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol. 2002;31:1235–1239. doi: 10.1093/ije/31.6.1235. [DOI] [PubMed] [Google Scholar]
- 31.Gillman MW. Epidemiological challenges in studying the fetal origins of adult chronic disease. Int J Epidemiol. 2002;31:294–299. [PubMed] [Google Scholar]
- 32.Food and Nutrition Board–Institute of Medicine . Progress in preventing childhood obesity: how do we measure up? National Academies Press; Washington, DC: 2006. [Google Scholar]
- 33.Cnattingius S, Lambe M. Trends in smoking and overweight during pregnancy: prevalence, risks of pregnancy complications, and adverse pregnancy outcomes. Semin Perinatol. 2002;26:286–295. doi: 10.1053/sper.2002.34771. [DOI] [PubMed] [Google Scholar]
- 34.Lumley J, Oliver SS, Chamberlain C, Oakley L. Interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst Rev. 2004:CD001055. doi: 10.1002/14651858.CD001055.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Olson CM, Strawderman MS, Reed RG. Efficacy of an intervention to prevent excessive gestational weight gain. Am J Obstet Gynecol. 2004;191:530–536. doi: 10.1016/j.ajog.2004.01.027. [DOI] [PubMed] [Google Scholar]
- 36.National Research Council, Institute of Medicine . Workshop report. The National Academies Press; Washington, DC: 2007. Influence of pregnancy weight on maternal and child health. [Google Scholar]
- 37.Ryan AS, Wenjun Z, Acosta A. Breastfeeding continues to increase into the new millennium. Pediatrics. 2002;110:1103–1109. doi: 10.1542/peds.110.6.1103. [DOI] [PubMed] [Google Scholar]
- 38.Kramer MS, Chalmers B, Hodnett ED, et al. Promotion of Breastfeeding Intervention Trial (PROBIT): a randomized trial in the Republic of Belarus. JAMA. 2001;285:413–420. doi: 10.1001/jama.285.4.413. [DOI] [PubMed] [Google Scholar]
- 39.Mindell JA, Kuhn B, Lewin DS, et al. Behavioral treatment of bedtime problems and night wakings in infants and young children. Sleep. 2006;29:1263–1276. [PubMed] [Google Scholar]
- 40.Dabelea D, Hanson RL, Lindsay RS, et al. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes. 2000;49:2208–2211. doi: 10.2337/diabetes.49.12.2208. [DOI] [PubMed] [Google Scholar]
- 41.Whitaker RC, Wright JA, Pepe MS, Seidel KD, Dietz WH., Jr Predicting obesity in young adulthood from childhood and parental obesity. N Engl J Med. 1997;337:869–873. doi: 10.1056/NEJM199709253371301. [DOI] [PubMed] [Google Scholar]
- 42.Ebbeling CB, Pawlak DB, Ludwig DS. Childhood obesity: public-health crisis, common sense cure. Lancet. 2002;360:473–482. doi: 10.1016/S0140-6736(02)09678-2. [DOI] [PubMed] [Google Scholar]
- 43.Whitaker RC. Predicting preschooler obesity at birth: the role of maternal obesity in early pregnancy. Pediatrics. 2004;114:e29–e36. doi: 10.1542/peds.114.1.e29. [DOI] [PubMed] [Google Scholar]