Abstract
Loss-of-function mutations of phosphatase/tensin homolog deleted on chromosome 10 (PTEN)-induced putative kinase 1 (Pink1) (also known as Park6) identified in familial forms of Parkinson's disease (PD) are associated with compromised mitochondrial function. Emerging data suggest that Pink1 is an essential pro-survival factor that is induced in response to oxidative stress. However, the mechanisms regulating Pink1 expression under stress conditions remain unknown. Forkhead box, subgroup O (FOXO) transcription factors carry out distinct biological functions in response to different extracellular signals. Notably, FOXO factors possess evolutionarily conserved roles in protecting cells from oxidative stress-induced death. Here we report that the FOXO family member FOXO3a controls Pink1 transcription in both mouse and human cells subjected to growth factor deprivation and that this regulation is exerted through evolutionarily conserved FOXO binding elements. Induction of Pink1 by FOXO3a is crucial for survival signals in lymphocytes, as depletion of Pink1 sensitizes these cells to death induced by deprivation of an essential growth factor. Our data reveal that the role of FOXO factors in protecting cells from growth factor deprivation-triggered apoptosis has been underestimated and that FOXOs mediate this protection by transactivating anti-apoptotic effectors like Pink1. Given the essential role of Pink1 in combating cell death, our findings may help to dissect the mechanisms by which FOXO proteins function as anti-oxidative stress factors.
Keywords: apoptosis, oxidative stress, transcription
Phosphatase/tensin homolog on chromosome 10 (PTEN)-induced putative kinase 1 (Pink1) was initially identified as a PTEN-inducible transcript and contains a serine/threonine kinase domain (1). Mutations in Pink1 (Park6) cause autosomal recessive Parkinson's disease (PD) but the precise role of Pink1 in the neuronal death process remains elusive. It is known that Pink1 expression can inhibit cell death in response to staurosporine (2). In addition, siRNA-induced Pink1 suppression sensitizes various mammalian cancer cell lines to multiple apoptotic stimuli (3, 4). Several lines of evidence suggest that Pink1 is an important mitochondrial protein and that loss-of-function of Pink1 is associated with dysregulated mitochondrial function. First, the Pink1 protein contains a putative mitochondrial localization sequence and is located to the mitochondria (5). Second, downregulation of Pink1 expression in the fly results in structural mitochondrial damage that impedes function, causes energy depletion, and shortens life span (6–8). Third, Pink1-dependent phosphorylation of HtrA2 modulates HtrA2's proteolytic activity, thereby increasing the resistance of cells to mitochondrial stress (9). Despite these findings, however, the molecular mechanism by which Pink1 influences cell survival is still not clear, and how Pink1 is induced and regulated in response to stress remains largely unknown.
Forkhead box, subgroup O (FOXO)3a belongs to the FOXO subfamily of transcription factors, and activation of FOXO3a generally leads to either cell cycle arrest or cell death. Recently, FOXO3a was shown to protect cells from oxidative stress-mediated apoptosis by upregulating magnesium superoxide dismutase (MnSOD) and peroxisomal catalase (10, 11). This anti-oxidative stress function of FOXO3a provides a possible explanation for the observation that the activation of the FOXO homologs Daf-16 and dFOXO extends life span in Caenorhabditis elegans and Drosophila, respectively (12, 13). In mammals, triple-mutant mice lacking FOXO1, FOXO3a, and FOXO4 possess hematopoietic stem cells (HSCs) that are defective in long-term repopulating activity and show increased cell cycling and apoptosis. These FOXO-deficient HSCs also exhibit a marked increase in reactive oxygen species (ROS) (14), implying an essential role for FOXOs in responses to physiologic oxidative stress. In previous work using microarray analysis, we demonstrated that FOXO3a is responsible for the transcriptional upregulation of the BH3-only family member Puma in response to growth factor deprivation (15). Genetic studies using Puma knockout mice confirmed the critical role of Puma in mediating pro-apoptotic functions downstream of FOXO activation. In this study, we report that FOXO3a activation induced by growth factor deprivation leads to upregulation of Pink1. In cytotoxic T cells, loss-of-function of Pink1 achieved by RNAi knockdown accelerates the death of these cells triggered by IL-2 withdrawal. Our findings reveal that Pink1 is a novel and important downstream mediator of FOXO3a and a regulator of the anti-oxidative stress response.
Results
Regulation of Pink1 Expression by PI3K/Akt/FOXO Signaling.
The FOXO factors are key downstream targets of insulin, growth factors, nutrients, and oxidative stress. The responses by the FOXO proteins to these stimuli coordinate a wide range of cellular outputs, including gluconeogenesis, neuropeptide secretion, atrophy, autophagy, apoptosis, cell cycle arrest, and stress resistance (16). The role of FOXO factors in mediating resistance to oxidative stress has been intensively studied in recent years. Two key molecules that are induced upon activation of FOXO signaling and can mediate oxidative stress resistance are MnSOD and catalase. Beta-catenin was reported as a positive modulator of FOXO-mediated transcription. Particularly, increased transcription of MnSOD occurs in a FOXO-dependent manner when beta-catenin forms a complex with FOXO (17–19). In an attempt to look for additional FOXO targets that can mediate an anti-oxidative stress response, we conducted microarray analyses and identified Pink1 as a potential FOXO-regulated gene. Therefore, we set out to examine the regulation and the biological function of Pink1, in association with the PI3K/Akt/FOXO signaling pathway.
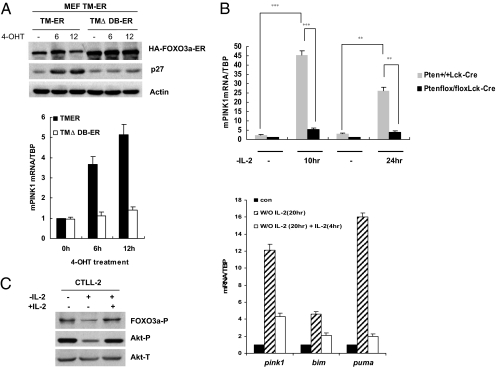
In previous work, we established murine embryonic fibroblast (MEF) lines that have a p53−/− genetic background and express either an HA-tagged FOXO3a-TM-ER (TM-ER) construct or a FOXO3a-TMΔDB-ER construct (15). We used these cell lines to examine Pink1 expression levels in response to 4-hydro-tamoxifen (4-OHT) treatment, using p27 induction as the readout to measure ectopically expressed FOXO3a activity. We first confirmed that the TM-ER and TMΔDB-ER transgenes were activated upon the addition of 4-OHT to the culture medium. As expected, p27 protein was upregulated only in TM-ER MEFs (Fig. 1A Upper). With respect to Pink1, a reliable commercial antibody that can detect endogenous Pink1 protein in MEFs is not yet available (data not shown), so that we measured Pink1 transcript levels instead for this study. Induction of FOXO3a-TM correlated with robust upregulation of Pink1 mRNA levels (Fig. 1A Lower).
Fig. 1.
Regulation of mPink1 by PI3k/Akt/FOXO signaling. (A) FOXO3a induces Pink1. (Upper) Validation of FOXO3a-expressing cell lines. p53−/− TM-ER or TMΔDB-ER FOXO3a-expressing MEFs were exposed to 4-OHT (1 μM) for 6 h or 12 h, and lysates were subjected to Western blotting using antibodies directed against the indicated proteins. Actin, loading control. (Lower) Pink1 mRNA levels were assessed in the MEFs in A Upper by qRT-PCR. TBP, normalization control. Unless otherwise stated, all results shown are one trial representative of at least 3 independent experiments. (B) Pten loss impairs Pink1 upregulation induced by IL-2 deprivation. RNA extracted from Pten+/+Lck-Cre or Ptenflox/floxLck-Cre activated T cells that were either left untreated or deprived of IL-2 for 10 or 24 h was subjected to qRT-PCR. The results presented are the mean and SEM of 2 independent experiments conducted in triplicate. **, P < 0.01; ***, P < 0.001. (C) Pink1 mRNA expression correlates inversely with PI3K/Akt activation. CTLL-2 cells were subjected to IL-2 withdrawal for 20 h and then treated with IL-2 for an additional 4 h. (Left) Lysates were prepared and analyzed by Western blotting with antibodies against the indicated proteins. P, phosphorylated; T, total. (Right) IL-2 restoration decreases Pink1 mRNA. qRT-PCR was performed to detect expression levels of Pink1 mRNA and Bim and Puma mRNAs (positive controls). Fold induction relative to TBP was determined as for A.
Because Pink1 was originally identified as a Pten-inducible gene, we theorized that deregulated Akt/FOXO signaling in the absence of Pten might alter Pink1 levels. To test this hypothesis, we took advantage of Ptenflox/flox Lck-Cre mice, which have a T cell-specific deletion of Pten (20). T cells of these mutant animals exhibit constitutive Akt activation and consequent inactivation of FOXO transcription factors (20). Strikingly, endogenous Pink1 mRNA levels were greatly decreased in activated Pten-deficient T cells compared to Pink1 levels in activated WT T cells (Fig. 1B), demonstrating that Pink1 expression is compromised in the absence of Pten. We next subjected cultured WT and Pten-deficient T cells to IL-2 deprivation, a condition that is known to inactivate PI3K/Akt and thus activate FOXOs. Strikingly, Pink1 transcript levels were significantly increased in Pten+/+ Lck-Cre cells in response to IL-2 withdrawal, whereas the loss of Pten dramatically impaired Pink1 induction in similarly treated Ptenflox/flox Lck-Cre T cells (Fig. 1B). These results indicate that unrestrained Akt signaling inversely correlates with Pink1 expression.
We further explored the PI3K/Akt/FOXO/Pink1 axis in CTLL-2 cells, an IL-2-dependent murine cytotoxic T lymphocyte cell line. Significant induction of Pink1 mRNA occurred in CTLL-2 cells subjected to IL-2 deprivation (Fig. 1C Right). Conversely, restoration of IL-2 to the culture medium largely reduced Pink1 transcript levels, coinciding with an increase in Akt activation and enhanced phosphorylation of FOXO3a at site Thr-32 (Fig. 1C Left). This regulation of Pink1 transcription mirrored that of Bim and Puma, two known FOXO targets that were analyzed as positive controls (Fig. 1C Right). Taken together, these results show that Pink1 mRNA expression correlates inversely with PI3K/Akt activation status. This observation raises the interesting possibility that FOXOs may be the transcription factors primarily responsible for directing Pink1 expression in response to growth factor deprivation.
FOXO3a Binds to the Murine Pink1 Promoter and Activates Pink1 Transcription upon Cytokine Deprivation.
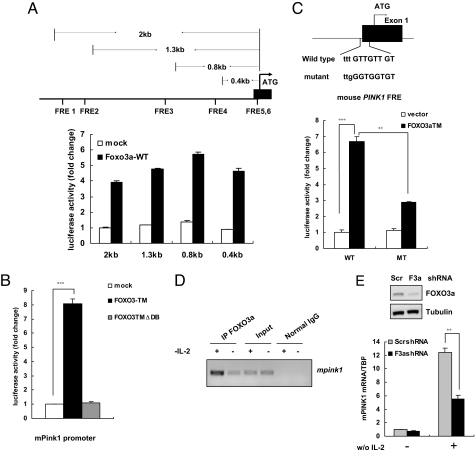
To test whether FOXO3a had any effect on the murine Pink1 promoter, we analyzed the sequence of this promoter and found 6 potential FOXO-binding-elements (FBEs), according to published consensus and suboptimal FBE sequences (21). To determine whether any of these putative FBEs were functional, we generated luciferase reporter constructs containing genomic DNA segments encompassing 2.0, 1.3, 0.8, or 0.4 kb of the DNA sequence immediately 5′ to the Pink1 ATG site (Fig. 2A Upper). 293T cells were cotransfected with WT FOXO3a or mutated FOXO3a (defective DNA binding function) plus one of the panel of Pink1 luciferase reporter constructs. WT FOXO3a transactivated all Pink1 luciferase reporters to comparable levels (Fig. 2A Lower). In contrast, no transactivation was detected upon cotransfection of 293T with the mutated FOXO3a (Fig. 2B). Two functional FBEs were identified within Pink1 exon1 through analysis of the 0.4-kb promoter construct (Fig. 2C Upper). To determine whether these putative FBEs could mediate transactivation of the Pink1 promoter by FOXO3a, 3 single-nucleotide substitution mutations were introduced into these 2 FBEs to eliminate its potential recognition by FOXO3a. Whereas the 0.4-kb Pink1 promoter region containing the unaltered FBEs showed strong transactivation, the same 0.4-kb region harboring the mutated sites was much less efficiently transactivated by WT FOXO3a (Fig. 2C Lower). These results demonstrate that Pink1 is a direct FOXO3a target and that this transactivation occurs through FBEs present within the Pink1 promoter region.
Fig. 2.
Murine Pink1 is a direct FOXO3a target. (A) Structure and validation of luciferase reporters driven by murine Pink1 promoter. (Upper) Schematic diagram of the WT murine Pink1 gene, showing the putative FBE sites and the segments of the candidate mPink1 promoter region that were cloned into the pGL3 basic luciferase reporter plasmid. (Lower) Transactivation of the Pink1 promoter by WT FOXO3a. 293T cells were cotransfected with a plasmid bearing a Pink1 promoter region fragment plus a plasmid expressing WT FOXO3a or were mock transfected. Luciferase activity was measured in triplicate after 48 h and normalized to an internal transfection efficiency control (β-galactosidase). (B) Functional FOXO is required for Pink1 induction. 293T cells were cotransfected with the 0.4-kb Pink1 promoter luciferase reporter plus vector alone, or FOXO3aTM, or FOXO3a TMΔDB. Luciferase activity was assessed 48 h after transfection as for A. The results presented correspond to the mean and SEM of 3 independent experiments conducted in triplicate. ***, P < 0.001. (C) FOXO3a transactivates the Pink1 promoter. (Upper) The sequence of the putative FBEs in the mouse Pink1 0.4-kb promoter region and the substitution mutations introduced into this FBE sequence are shown. (Lower) Mutations of the FBEs impair transactivation of the Pink1 promoter by FOXO3a. 293T cells were cotransfected with either control vector or WT FOXO3a plus 0.4-kb Pink1 promoter luciferase reporters containing either intact (WT) or mutated (MT) FBEs. Results were expressed as for A and represent the mean and error of 3 independent experiments conducted in triplicate. **, P < 0.01; ***, P < 0.001. (D) FOXO3a physically binds to the mPink1 promoter. CTLL-2 cells were cultured in IL-2−free medium for 12 h and ChIP assays to determine the binding of FOXO3a to the mPink1 FBE were conducted as described in Materials and Methods. (E) Knockdown of FOXO3a attenuates Pink1 induction. (Upper) CTLL-2 cells expressing either scrambled shRNA (Scr) or FOXO3a shRNA (F3a) were subjected to IL-2 withdrawal and protein lysates were assessed by Western blotting to detect FOXO3a protein. Tubulin, loading control. (Lower) RNA from the CTLL-2 cells in E Upper was analyzed by qRT-PCR to detect mPink1 transcripts. TBP, internal control. The data correspond to the mean and SEM of 2 independent experiments conducted in triplicate. **, P < 0.01.
To characterize the physiological interaction between endogenous FOXO3a protein and FOXO binding sites within the murine Pink1 promoter, chromatin immunoprecipitation (ChIP) assays were performed using CTLL-2 cells deprived of IL-2. Primers flanking the putative Pink1 FBEs were used for the PCR. IL-2 starvation significantly enriched the association of FOXO3a with the Pink1 promoter (Fig. 2D), indicating that FOXO3a is recruited to the Pink1 promoter when the cells are starved of IL-2. Finally, we tested whether knockdown of FOXO3a in CTLL-2 cells could attenuate Pink1 induction in response to cytokine withdrawal. Indeed, depletion of FOXO3a resulted in markedly reduced Pink1 mRNA induction in the absence of IL-2 (Fig. 2E). Taken together, our results suggest that FOXO3a is a key transcriptional regulator that positively modulates Pink1 expression in response to cytokine deprivation.
FOXO-Dependent Transactivation of the Human Pink1 Promoter in Response to Attenuated PI3K/Akt Signaling.
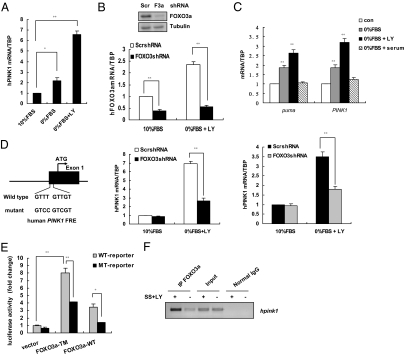
Because loss-of-function recessive mutations in the human Pink1 gene have been identified in autosomal recessive and some sporadic forms of PD, we set out to determine whether the Akt/FOXO/Pink1 axis we identified in mouse cells was conserved in human cells. To answer this question, we assessed Pink1 mRNA levels in MCF-7 cells subjected to growth factor starvation. Quantitative real-time (qRT)-PCR analyses revealed modest upregulation of Pink1 in serum-starved cells, whereas a more robust induction of Pink1 was observed when serum starvation was combined with inhibition of PI3K/Akt signaling by the PI3K inhibitor LY294002 (Fig. 3A). This synergy between serum starvation and LY294002 treatment is likely due to the greater upregulation of FOXO3a transcripts that occurs in the presence of LY294002 compared to FOXO3a levels induced by serum starvation alone (Fig. 3B Middle and data not shown). Knockdown of FOXO3a by shRNA largely impaired the induction of Pink1 transcripts compared to Pink1 mRNA levels in cells treated with control scrambled (Scr) shRNA (Fig. 3B Bottom). We also examined whether FOXO3 is responsible for Pink1 induction in a neuroblastoma cell line. Pink1 expression was significantly induced in SHSY5Y cells in serum-free medium or upon serum starvation plus LY294002 treatment, while adding back serum into the medium profoundly decreased Pink1 expression (Fig. 3C Upper). Furthermore, FOXO3 knockdown in SHSY5Y cells markedly attenuated Pink1 upregulation in the presence of LY294002 under serum starvation conditions (Fig. 3C Lower). These data demonstrate that FOXO3a regulates Pink1 expression levels in human cells subjected to growth factor deprivation.
Fig. 3.
FOXO regulates the human Pink1 promoter. (A) Inactivation of Akt signaling increases hPink1 transcription. MCF-7 cells were cultured for 24 h under standard conditions or in medium containing 0% serum or 0% serum plus 25 μM LY294002 (LY). RNA was extracted and levels of hPink1 expression were assessed by qRT-PCR. The data represent the mean and SEM of 4 independent experiments. *, P < 0.05; **, P < 0.01. (B) Depleting FOXO3a abrogates Pink1 induction in response to inactivated PI3K/Akt signaling. (Top) Protein lysates from MCF-7 cells expressing scrambled shRNA (Scr) or FOXO3a shRNA (F3a) were assessed by Western blotting to detect FOXO3a. (Middle and Bottom) RNA from the cells in A was analyzed by qRT-PCR to detect FOXO3a (Middle) or Pink1 (Bottom). Data represent the mean and SEM of 3 independent experiments conducted in triplicate. **, P < 0.01. (C) FOXO3a-dependent induction of Pink1 in SHSY5Y neuroblastoma cells. (Upper) SHSY5Y cells were cultured for 24 h under standard conditions or in medium containing 0% serum, or 0% serum plus 25 μM LY294002 (LY), or serum starved for 24 h and then cultured in 10% FBS for additional 3 h. qRT-PCR was performed to detect expression levels of Pink1 mRNA and Puma mRNAs (positive control). Fold induction relative to TBP was determined as for A. (Lower) SHSY5Y cells were treated as in B Bottom, and RNA was analyzed by qRT-PCR to detect Pink1. The results presented correspond to the mean and SEM of 3 independent quantitative RT-PCR experiments. **, P < 0.01. (D) The sequence of a highly conserved putative FBE in the human Pink1 promoter (WT) and substitution mutations introduced into this FBE (mutant) are shown. (E) FBE mutation impairs hPink1 promoter transactivation by FOXO3a. 293T cells were transfected with hPink1 promoter reporter plasmids containing an intact (WT) or mutated (MT) FBE, in the presence of vector alone, or FOXO3a-TM, or WT FOXO3a (FOXO3a-WT). Data represent the mean and error of 3 independent experiments conducted in triplicate. *, P < 0.05; **, P < 0.01. (F) FOXO3a physically binds to the hPink1 promoter. ChIP assays were performed as for Fig. 2D using MCF-7 cells that were left untreated or serum-starved (SS) for 24 h in the presence of 25 μM LY294002 (LY).
Intriguingly, we found a conserved FBE in the human Pink1 promoter region (Fig. 3D). A luciferase reporter construct containing either the putative WT FBE or a mutated version of this FBE was introduced into 293T cells. In the case of the WT FBE, induction of luciferase reporter activity was detected when either FOXO3-TM or WT FOXO3 was cotransfected (Fig. 3E). In contrast, mutation of the putative FBE coincided with significantly reduced induction of luciferase reporter activity (Fig. 3E). To determine whether FOXO3 proteins associate with the human Pink1 promoter in vivo, we performed ChIP assays. A markedly enhanced interaction between the putative hPink1 FBE and endogenous FOXO3a protein was detected in MCF-7 cells subjected to a combination of serum starvation and LY294002 treatment (Fig. 3F). These data indicate that FOXO3 directs human Pink1 transcription by acting on the hPink1 promoter.
Pink1 Promotes the Survival of Cytokine-Deprived T Cells by Modulating Glutathione Levels.
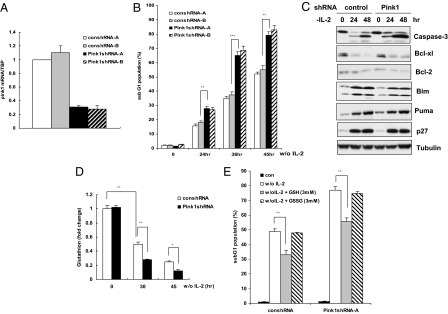
We next investigated the biological consequences of Pink1 induction in cytokine-deprived murine T cells. It is known that the proliferation of CTLL-2 cells depends on IL-2 and that depletion of IL-2 leads to the apoptosis of these cells. After confirming that endogenous Pink1 could be depleted in CTLL-2 cells by specific shRNA treatment (Fig. 4A), we subjected Pink1 knockdown CTLL-2 cells to IL-2 withdrawal and examined apoptosis. Strikingly, and consistent with its protective role in various organs and cell types (22–24), Pink1 depletion in CTLL-2 cells profoundly increased the proportion of apoptotic cells in the treated population (Fig. 4B). This increase in apoptotic cells coincided with a marked enhancement of caspase-3 cleavage (activation) (Fig. 4C, row 1), suggesting that Pink1 plays a critical role in the survival of CTLL-2 cells in the absence of an essential cytokine.
Fig. 4.
Pink1 depletion sensitizes T cells to cytokine withdrawal-induced death. (A) Validation of mPink1 shRNAs. CTLL-2 cells were transfected with 2 independent sets of control (con) shRNA or mPink1 shRNA constructs and levels of mPink1 mRNA were determined by qRT-PCR. (B) Reduction of Pink1 increases apoptosis. CTLL-2 cells expressing the indicated shRNAs were cultured in the presence or absence of IL-2 for the indicated times and apoptosis was determined by PI staining and flow cytometry. Data shown are the mean percentage of apoptotic cells ± SD from 3 independent experiments. **, P < 0.01; ***, P < 0.001. (C) Reduction of mPink1 increases caspase-3 cleavage. CTLL-2 cells expressing either control shRNA or Pink1 shRNA were deprived of IL-2 for the indicated time point and cell lysates were subjected to Western blotting to detect the indicated proteins. (D) Reduction of mPink1 correlates with decreased GSH levels. CTLL-2 cells expressing either control shRNA or Pink1 shRNA were cultured in the presence or absence of IL-2 for 30 or 45 h and intracellular GSH levels were measured in triplicate as described in Materials and Methods. Results represent the mean and error of 4 independent experiments conducted in triplicate each time. *, P < 0.05; **, P < 0.01. (E) GSH is a cause, not a consequence, of cell death associated with Pink1 depletion. CTLL-2 cells expressing control or Pink1 shRNA were cultured for 45 h in medium lacking IL-2 but containing 3 mM reduced GSH or oxidized GSSG. Cell death was measured in triplicate by PI staining and flow cytometry. Results shown are the mean percentage of apoptotic cells ± SD from 4 independent experiments. **, P < 0.01.
To dissect the molecular pathways acting downstream of Pink1 following cytokine withdrawal, we first examined the protein levels of several key Bcl-2 family members in IL-2-deprived CTLL-2 cells. Bcl-2 family members are essential for modulating mitochondria-mediated cell death, particularly in T cells deprived of cytokines. In the case of IL-2 deprivation-induced death, the main executioners are Puma and Bim, since a compound knockout of both of these BH3-only proteins exhibits marked resistance to cytokine withdrawal-induced death (15). In contrast, the levels of the anti-apoptotic family members Bcl-2 and Bcl-xL are downregulated in the absence of IL-2. In our study, we found reduced levels of Bcl-2 and Bcl-xL, whereas levels of Puma and Bim were greatly increased (Fig. 4C, rows 2–5). However, we failed to detect any difference between cells expressing control shRNA and those expressing Pink1 shRNA. We also assessed the protein levels of the cell cycle checkpoint regulator p27 and found no effect when Pink1 was depleted.
Another factor that can promote the survival of IL-2-deprived T cells regardless of Bcl-2 level is reduced glutathione (GSH) (25). When we examined endogenous GSH levels in CTLL-2 cells subjected to IL-2 withdrawal, we observed a time-dependent decrease in the level of this molecule (Fig. 4D). Strikingly, Pink1 knockdown profoundly accelerated this decrease in GSH (Fig. 4D). Downregulation of GSH has been correlated with the increase in apoptosis induced by IL-2 withdrawal (25). To exclude the possibility that our observed decrease in endogenous GSH levels was merely a consequence of the increased death exhibited by Pink1 knockdown cells, we added exogenous GSH to the culture medium of IL-2-deprived CTLL-2 cells. Exogenous GSH is known to influence intracellular GSH levels, which in turn can rescue cells from apoptosis (25). Consistent with these previous findings, we found that the addition of reduced GSH, but not oxidized GSH (GSSG), to cultures of IL-2-deprived CTLL-2 cells prevented cell death in both the control shRNA group and the Pink1 knockdown group (Fig. 4E). Thus, the reduction in endogenous GSH observed in IL-2-deprived CTLL-2 cells is the cause, rather than the consequence, of cell death. Taken together, our data demonstrate an essential role for Pink1 in promoting cell survival under conditions of cytokine withdrawal via regulation of intracellular GSH levels.
Discussion
Oxidative stress that causes mitochondrial dysfunction has been implicated in the neurodegenerative process prominent in diseases such as PD. It has been suggested that the molecular mechanism by which loss-of-function recessive mutations in the Pink1 gene lead to PD pathogenesis may be impairment of mitochondrial function. Indeed, defects in mitochondrial respiration have been observed in the cerebral cortex of Pink1−/− mice exposed to either hydrogen peroxide or mild heat shock (23). Accumulating data in several different species imply that Pink1 may provide critical protection against both intrinsic and environmental stresses through its effects on mitochondria. Thus, loss of Pink1 may constitute a pathogenic mechanism that leads to abnormal cell death.
Although the role of Pink1 in protecting against oxidative stress-induced cell death has been intensively studied in neuronal systems, the biological functions of Pink1 in other tissues remain largely unknown. Before our work, the only information available on Pink1 regulation was based on one study involving overexpression of Pten (1). In our study, we have identified FOXO3a as a key transcription factor directing the expression of Pink1 in cells deprived of growth factors, a culture condition known to activate FOXOs. In triple-mutant mice deficient for FOXO1, FOXO3a, and FOXO4, HSCs exhibit an aberrant increase in ROS, which in turn leads to decreased HSC numbers and impaired long-term repopulating activity (14). Taking these 2 observations together, we believe that Pink1 may be an essential downstream FOXO3a target that mediates the anti-oxidative stress functions of this family of transcription factors.
In evaluating the degree of oxidative stress in a cell or an organism, one of the most important parameters to consider is levels of reduced GSH. It has been proposed that the downregulation of GSH observed in PD is associated with mitochondrial damage caused by oxidative stress and that this decrease in GSH accelerates the death of dopaminergic neurons (26). Consistent with this finding, we found that depletion of Pink1 in lymphocytes via shRNA treatment led to a dramatic decrease in GSH that sensitized T cells to death caused by IL-2 withdrawal. Our results thus support a pro-survival role for Pink1 that takes the form of guarding mitochondrial function in the face of pathological oxidative stress. The molecular mechanisms by which reduced GSH levels are regulated in a Pink1-dependent manner require further investigation. Notably, DJ-1/PARK7, another PD-associated protein, physically associates with Pink1 to form a complex that exerts a protective effect against oxidative stress (27). DJ-1 has been shown to stabilize and thus enhance the transcriptional activity of Nrf-2, a master regulator of anti-oxidant responses (28). Does Pink1 act on the same pathway? And what is the role of DJ-1 in cytokine deprived T cells? A mouse model bearing T cells deficient for both DJ-1 and Pink1 may reveal the answer. Investigation of these questions should help to elucidate the functions of Pink1 under physiological oxidative stress conditions.
Withdrawal of a growth factor or cytokine usually leads to cell cycle arrest or apoptosis. The essential roles of FOXO family members in mediating these events are well documented (15). Thus, it seems a paradox that Pink1 is both a pro-survival molecule and a transcriptional target of the pro-apoptotic factor FOXO3a. However, our results clearly show that loss-of-function of Pink1 is associated with abnormal and massive cell death. We propose that the induction of pro-survival factors like Pink1 by FOXO3a upon growth factor starvation allows short-term protection of these cells during the early phase (within 12 h, as neither GSH decrease nor apoptosis was detected, data not shown) until growth factors are once more available, at which time the cells can resume cell cycle progression. In contrast, prolonged starvation (>24 h) synergizes with the robust upregulation of pro-apoptotic factors like Puma and Bim. These potent BH3-only family members may overcome the survival effect initiated in the early stages of growth factor deprivation and drive the starved cells into apoptosis. The biological significance of the induction of Pink1 by FOXO family members in response to growth factor/PI3k/Akt signaling may be examined in organs sensitive to growth hormone levels such as insulin responsive tissues and in stem cells that are sensitive to redox contents. Investigation of such responses may shed light on the biological functions of Pink1 in the context of PI3K/Akt signaling.
Materials and Methods
Cell Culture and Manipulation.
To generate FOXO3a-related stable cell lines, MEFs were infected with pBabe FOXO3a-TMER or FOXO3a-TMΔDB-ER constructs and selected with 2 μg/mL puromycin for 72 h (29).
T cell purification, activation, and cell viability analyses were performed as previously reported (15).
Constructs and Antibodies.
The pECE-HA-FOXO3a-TM and TMΔDB expression plasmids provided by M. Greenberg (Harvard Medical School, Boston), and shRNA sequences against murine FOXO3a and human FOXO3a, were as described previously (15). Pink1 shRNA and control shRNA sequences were based on siRNA oligo sequences from Dharmacon. pSIRIPP retroviral vector or pLL3.7 lentiviral vector was used for cloning and delivering shRNA. Stable cells are pools of clones selected by puromycin.
ChIP Assays.
ChIP assays were performed as described previously (30). For additional information see SI Materials and Methods.
Supplementary Material
Acknowledgments.
We thank Drs. Michael Greenberg (Children's Hospital, Harvard Medical School, Boston), Zhuohua Zhang (Burnham Institute for Medical Research, La Jolla, CA), and Bingwei Lu (Department of Pathology, Stanford University School of Medicine, Palo Alto, CA) for reagents and discussions and Dr. Mary Saunders for scientific editing. We are grateful for excellent technical support provided by Joan Dacosta and Lily Zhou. This work is supported by the National Natural Science Foundation of China (Grant 30700130 to H.Y.) and the 973 Project of the Ministry of Science and Technology of China (Grant 2009CB522200 to H.Y.).
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/cgi/content/full/0901104106/DCSupplemental.
References
- 1.Unoki M, Nakamura Y. Growth-suppressive effects of BPOZ and EGR2, two genes involved in the PTEN signaling pathway. Oncogene. 2001;20(33):4457–4465. doi: 10.1038/sj.onc.1204608. [DOI] [PubMed] [Google Scholar]
- 2.Petit A, et al. Wild-type PINK1 prevents basal and induced neuronal apoptosis, a protective effect abrogated by Parkinson disease-related mutations. J Biol Chem. 2005;280(40):34025–34032. doi: 10.1074/jbc.M505143200. [DOI] [PubMed] [Google Scholar]
- 3.MacKeigan JP, Murphy LO, Blenis J. Sensitized RNAi screen of human kinases and phosphatases identifies new regulators of apoptosis and chemoresistance. Nat Cell Biol. 2005;7(6):591–600. doi: 10.1038/ncb1258. [DOI] [PubMed] [Google Scholar]
- 4.Deng H, Jankovic J, Guo Y, Xie W, Le W. Small interfering RNA targeting the PINK1 induces apoptosis in dopaminergic cells SH-SY5Y. Biochem Biophys Res Commun. 2005;337(4):1133–1138. doi: 10.1016/j.bbrc.2005.09.178. [DOI] [PubMed] [Google Scholar]
- 5.Valente EM, et al. Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science. 2004;304(5674):1158–1160. doi: 10.1126/science.1096284. [DOI] [PubMed] [Google Scholar]
- 6.Clark IE, et al. Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature. 2006;441(7097):1162–1166. doi: 10.1038/nature04779. [DOI] [PubMed] [Google Scholar]
- 7.Yang Y, et al. Mitochondrial pathology and muscle and dopaminergic neuron degeneration caused by inactivation of Drosophila Pink1 is rescued by Parkin. Proc Natl Acad Sci USA. 2006;103(28):10793–10798. doi: 10.1073/pnas.0602493103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang D, et al. Antioxidants protect PINK1-dependent dopaminergic neurons in Drosophila. Proc Natl Acad Sci USA. 2006;103(36):13520–13525. doi: 10.1073/pnas.0604661103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Plun-Favreau H, et al. The mitochondrial protease HtrA2 is regulated by Parkinson's disease-associated kinase PINK1. Nat Cell Biol. 2007;9(11):1243–1252. doi: 10.1038/ncb1644. [DOI] [PubMed] [Google Scholar]
- 10.Kops GJ, et al. Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature. 2002;419(6904):316–321. doi: 10.1038/nature01036. [DOI] [PubMed] [Google Scholar]
- 11.Nemoto S, Finkel T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science. 2002;295(5564):2450–2452. doi: 10.1126/science.1069004. [DOI] [PubMed] [Google Scholar]
- 12.Lin K, Dorman JB, Rodan A, Kenyon C. daf-16: An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science. 1997;278(5341):1319–1322. doi: 10.1126/science.278.5341.1319. [DOI] [PubMed] [Google Scholar]
- 13.Giannakou ME, et al. Long-lived Drosophila with overexpressed dFOXO in adult fat body. Science. 2004;305(5682):361. doi: 10.1126/science.1098219. [DOI] [PubMed] [Google Scholar]
- 14.Tothova Z, et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell. 2007;128(2):325–339. doi: 10.1016/j.cell.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 15.You H, et al. FOXO3a-dependent regulation of Puma in response to cytokine/growth factor withdrawal. J Exp Med. 2006;203(7):1657–1663. doi: 10.1084/jem.20060353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Salih DA, Brunet A. FoxO transcription factors in the maintenance of cellular homeostasis during aging. Curr Opin Cell Biol. 2008;20(2):126–136. doi: 10.1016/j.ceb.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Essers MA, et al. Functional interaction between beta-catenin and FOXO in oxidative stress signaling. Science. 2005;308(5725):1181–1184. doi: 10.1126/science.1109083. [DOI] [PubMed] [Google Scholar]
- 18.Almeida M, Han L, Martin-Millan M, O'Brien CA, Manolagas SC. Oxidative stress antagonizes Wnt signaling in osteoblast precursors by diverting beta-catenin from T cell factor- to forkhead box O-mediated transcription. J Biol Chem. 2007;282(37):27298–27305. doi: 10.1074/jbc.M702811200. [DOI] [PubMed] [Google Scholar]
- 19.Hoogeboom D, et al. Interaction of FOXO with beta-catenin inhibits beta-catenin/T cell factor activity. J Biol Chem. 2008;283(14):9224–9230. doi: 10.1074/jbc.M706638200. [DOI] [PubMed] [Google Scholar]
- 20.Suzuki A, et al. T cell-specific loss of Pten leads to defects in central and peripheral tolerance. Immunity. 2001;14(5):523–534. doi: 10.1016/s1074-7613(01)00134-0. [DOI] [PubMed] [Google Scholar]
- 21.Biggs WH, III, Cavenee WK, Arden KC. Identification and characterization of members of the FKHR (FOX O) subclass of winged-helix transcription factors in the mouse. Mamm Genome. 2001;12(6):416–425. doi: 10.1007/s003350020002. [DOI] [PubMed] [Google Scholar]
- 22.Wood-Kaczmar A, et al. PINK1 is necessary for long term survival and mitochondrial function in human dopaminergic neurons. PLoS ONE. 2008;3(6):e2455. doi: 10.1371/journal.pone.0002455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gautier CA, Kitada T, Shen J. Loss of PINK1 causes mitochondrial functional defects and increased sensitivity to oxidative stress. Proc Natl Acad Sci USA. 2008;105(32):11364–11369. doi: 10.1073/pnas.0802076105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pridgeon JW, Olzmann JA, Chin LS, Li L. PINK1 protects against oxidative stress by phosphorylating mitochondrial chaperone TRAP1. PLoS Biol. 2007;5(7):e172. doi: 10.1371/journal.pbio.0050172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hyde H, Borthwick NJ, Janossy G, Salmon M, Akbar AN. Upregulation of intracellular glutathione by fibroblast-derived factor(s): Enhanced survival of activated T cells in the presence of low Bcl-2. Blood. 1997;89(7):2453–2460. [PubMed] [Google Scholar]
- 26.Di Monte DA, Chan P, Sandy MS. Glutathione in Parkinson's disease: A link between oxidative stress and mitochondrial damage? Ann Neurol. 1992;32(Suppl):S111–S115. doi: 10.1002/ana.410320719. [DOI] [PubMed] [Google Scholar]
- 27.Tang B, et al. Association of PINK1 and DJ-1 confers digenic inheritance of early-onset Parkinson's disease. Hum Mol Genet. 2006;15(11):1816–1825. doi: 10.1093/hmg/ddl104. [DOI] [PubMed] [Google Scholar]
- 28.Clements CM, McNally RS, Conti BJ, Mak TW, Ting JP. DJ-1, a cancer- and Parkinson's disease-associated protein, stabilizes the antioxidant transcriptional master regulator Nrf2. Proc Natl Acad Sci USA. 2006;103(41):15091–15096. doi: 10.1073/pnas.0607260103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.You H, Yamamoto K, Mak TW. Regulation of transactivation-independent proapoptotic activity of p53 by FOXO3a. Proc Natl Acad Sci USA. 2006;103(24):9051–9056. doi: 10.1073/pnas.0600889103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.You H, et al. p53-dependent inhibition of FKHRL1 in response to DNA damage through protein kinase SGK1. Proc Natl Acad Sci USA. 2004;101(39):14057–14062. doi: 10.1073/pnas.0406286101. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.