Summary
Type III secretion-mediated cytotoxicity is one of the key virulence mechanisms of the opportunistic pathogen Pseudomonas aeruginosa. Prior data from several laboratories have established that metabolism is a key factor in the regulation of type III secretion gene expression in P. aeruginosa. Here we use a fluorescence-activated cell sorter (FACS)-based approach to investigate expression of type III secretion genes at a single-cell level. The data demonstrate that the metabolic state regulates the percentage of cells that are able to induce type III secretion gene expression under inducing conditions. We also present evidence that this regulation is the result of an effect of the growth conditions on the ability of P. aeruginosa to assemble a functional type III secretion apparatus. Preliminary data suggest that the metabolite that controls type III secretion gene expression is derived from acetyl-CoA and that this regulation may, in part, be mediated by changes in the intracellular concentration of cyclic-AMP.
Introduction
Pseudomonas aeruginosa is a common cause of ventilator-associated pneumonia and wound infections in patients that are immunocompromised due to an underlying condition, such as cancer patients (Velasco et al., 1997; Carratala et al., 1998), the elderly and burn patients (Holder et al., 2001a; Pirnay et al., 2003). One of the major virulence factors of P. aeruginosa is the type III secretion system, which is used to directly deliver toxins into the cytoplasm of infected host cells (Yahr et al., 1996; Olson et al., 1997). Epidemiological studies of P. aeruginosa isolates from patients have shown that the presence of a functional type III secretion system is associated with a higher incidence of systemic spread and poor clinical outcome (Roy-Burman et al., 2001; Hauser et al., 2002; Garau and Gomez, 2003).
To date, four type III secreted toxins (effectors) have been described in P. aeruginosa. Exoenzyme S (ExoS) and exoenzyme T (ExoT) are bi-functional proteins with an amino-terminal GAP domain and a carboxy-terminal ADP-ribosylating domain (Knight et al., 1995; Goehring et al., 1999; Pederson et al., 1999; Barbieri, 2000; Krall et al., 2000). The GAP domains of ExoS and ExoT are active towards small rho-like GTPases and effect disassembly of the actin cytoskeleton and cell rounding of infected host cells. The ADP-ribosylating activities of ExoS and ExoT have differing substrate specificity. ExoS targets small ras-like proteins, whereas ExoT appears to be specific for CrkI and CrkII, two signalling proteins involved in triggering phagocytosis (McGuffie et al., 1998; Sun and Barbieri, 2003). The two remaining effector proteins, exoenzyme Y (ExoY) and exoenzyme U (ExoU), have adenylate-cyclase and phospholipase activity respectively (Yahr et al., 1998; Satoet al., 2003). Curiously, most strains of P. aeruginosa only have the gene for either ExoS or ExoU, but not both (Feltman et al., 2001).
The expression of type III secreted toxins is induced on cell contact, and can be induced in vitro by the removal of calcium from the medium (Yahr et al., 1996; Frank, 1997; Vallis et al., 1999). This ability to induce effector expression under low-calcium conditions has also been linked to a more severe disease outcome.
Cell contact, or low-calcium conditions, appears to trigger induction of the system by way of a regulatory cascade that results in activation of ExsA, the main transcriptional regulator of the type III secretion regulon (Frank and Iglewski, 1991). This induction depends on the presence of a functional type III secretion system. Mutations that inactivate the type III secretion machinery result in an inability to induce effector expression under low-calcium conditions (Frank and Iglewski, 1991).
The activity of ExsA is controlled by interaction with another protein, ExsD. When ExsD is bound to ExsA, transcription of the type III secretion regulon is turned off (McCaw et al., 2002). Overexpression of ExsD therefore results in the inability to induce effector expression under low-calcium conditions, whereas deletion of exsD results in a constitutive expression phenotype. The ExsD–ExsA interaction, in turn is controlled by ExsC. ExsC is a small protein that can bind tightly to ExsD and thereby relieve the inactivation of ExsA by ExsD (Dasgupta et al., 2004). ExsC appears to be similar to a chaperone of a type III secreted protein and its activity is in turn controlled by a recently discovered, secreted protein, ExsE (Rietsch et al., 2005; Urbanowski et al., 2005). Upon triggering of the type III secretion machinery, ExsE is exported, freeing ExsC to interact with ExsD, which in turn liberates ExsA and results in induction of the regulon.
Expression of the type III secretion genes is subject to regulation by at least two global regulatory pathways. Cyclic AMP and the Pseudomonas catabolite repression homologue, Vfr, are required for expression of the type III secretion regulon (Wolfgang et al., 2003). Second, the GacA/GacS two-component regulatory system can repress expression of the type III secretion genes by modulating the activity of the regulator RsmA. The activity of the GacA/GacS pathway is, in turn, controlled by the hybrid two-component regulator RetS (Goodman et al.,2004; Laskowski et al., 2004; Zolfaghar et al., 2005). The molecular basis of neither regulation is, as yet, clear.
The third factor controlling exoenzyme expression is metabolic in nature, and much less well understood. Media composition, in particular salt concentration, contributes to the ability to induce ExoS expression (Hornef et al., 2000). Mutations in the genes for pyruvate dehydrogenase, aceA and aceB, were reported to result in the inability to induce exsA expression under low-calcium conditions (Dacheux et al., 2002). Similarly, plasmid insertions in gltR, the regulator of the glucose transporter, resulted in the inability to induce the type III secretion regulon (Wolfgang et al., 2003). Finally, overexpression of the histidine transporter and degradation genes abolished the ability to induce ExoS expression and rendered the bacteria non-cytotoxic (Rietsch et al., 2004). The latter phenotype depended on the presence of histidine in the medium and the ability to take up and metabolize histidine. Furthermore, the phenotype was suppressed by a transposon insertion in cbrA. The CbrAB two-component regulatory system is involved in sensing metabolic imbalance and adjusting gene-expression of catabolic operons accordingly (Nishijyo et al., 2001). Taken together these results suggest that metabolism can in fact exert a significant level of control over the ability to induce the type III secretion regulon.
We have used a fluorescence-activated cell sorter (FACS)-based assay to study the induction of ExoS expression to further define how metabolism controls the type III secretion regulon. Our results demonstrate that growth under metabolically unfavourable conditions in fact results in a type III secretion negative phenotype, suggesting that metabolism controls either assembly or activity of the type III secretion machinery. This finding allows us to integrate metabolic control into the model for regulation of ExsA activity and induction of the type III secretion regulon.
Results
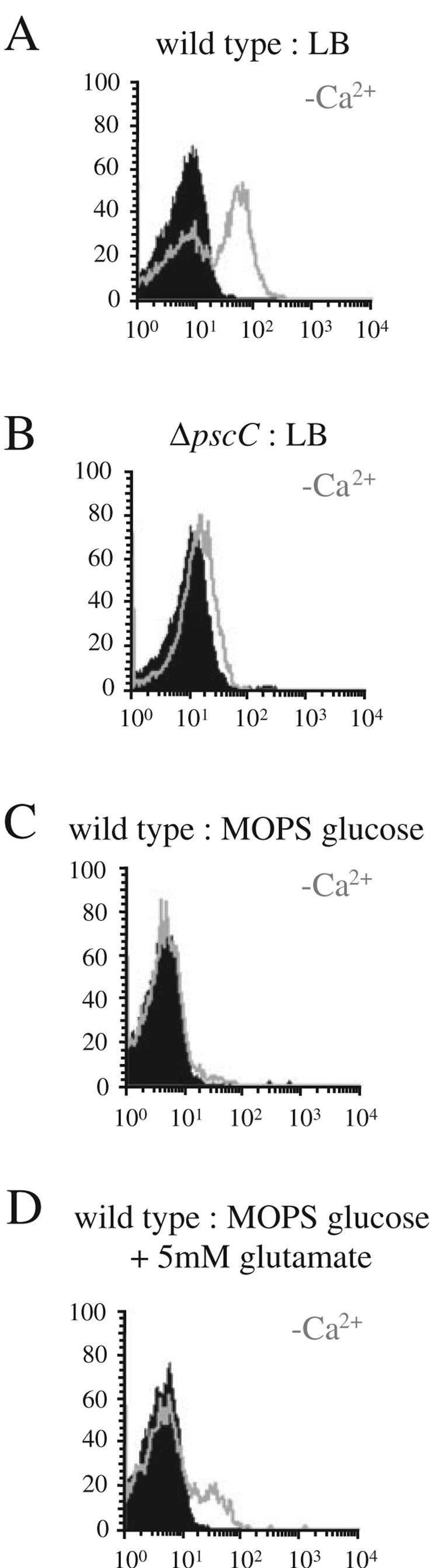
When measuring induction of gene expression using a conventional genetic reporter system, such as the lacZ gene, any change in expression is, by definition, a change in the expression of a population of cells. This type of method does not distinguish between changes in regulation that affect the entire population of cells uniformly, and regulation that only affects a subpopulation of cells. Using a FACS to monitor single cell gene expression of a green fluorescent protein (GFP) reporter inserted in the exoS locus of P. aeruginosa, we discovered that exoS is only induced in a subpopulation of cells under inducing conditions. In L-broth, roughly 45% of cells induce expression of the reporter upon addition of the chelator nitrilo-triacetic acid (NTA) (Fig. 1A). Furthermore, adding increasing amounts of chelator only serves to increase the percentage of cells that induce (Fig. S1A). Induction depends on the presence of a functional type III secretion system, because deletion of the structural genes pscC or pscJ results in an inability to induce GFP expression (Fig. 1B and data not shown). This mode of induction differs from induction of the lac or toluic acid promoters in P. aeruginosa (data not shown and Fig. S1B). In those cases, addition of increasing levels of inducer gradually raises expression of the entire population of cells rather than inducing expression in only a fraction of the population.
Fig. 1.
Induction of exoS expression monitored by FACS analysis using a GFP reporter. P. aeruginosa PAK (A, C and D) or PAK ΔpscC (B) harbouring the exoS∷GFP-lacZ reporter were grown in l-broth (A,B) or MOPS minimal media supplemented with 0.2% glucose (C) or 0.2% glucose and 5 mM glutamate (D). Relative fluorescence of individual bacteria is plotted on the x-axis. The y-axis denotes the number of bacteria of a given fluorescence intensity (10 000 bacteria were analysed). The grey overlay indicates the distribution for cultures in which calcium had been chelated by the addition of NTA to the media.
Induction of exoenzyme expression by addition of a chelator does not work in minimal glucose media (Fig. 1C). Addition of caseamino acids was able to restore induction of type III secretion genes in minimal glucose, suggesting that the inability to induce by addition of chelator was due to a change in growth conditions (data not shown). Interestingly, addition of just glutamate was able to partially restore the ability of P. aeruginosa to induce expression of exoS in minimal glucose media upon removal of calcium (Fig. 1D).
It had been previously reported that salt concentration strongly influences the intracellular glutamate concentration and also influences exoS induction, so we decided to determine if osmolarity can influence the percentage of cells that induce exoS expression. Indeed only about 4% of cells induce exoS expression in L-broth with 5 mM NaCl (Fig. 2A), whereas 90% induce in 200 mM NaCl (Fig. 2B). Increasing the concentration of salt in the medium correlated with an increase in the percentage of cells that were able to induce exoS expression upon removal of calcium from the medium (Fig. 2C). Salt could be replaced by sucrose in these experiments, suggesting that exoS expression is in fact controlled by osmolarity (Fig. 2D). We also performed shift experiments in which the exoS reporter strain was grown in Luria–Bertani (LB) with 5 mM or 200 mM NaCl and shifted to media of the opposite osmolarity. Bacteria that had been grown in low-osmolarity LB were able to quickly adapt to the high osmolarity phenotype of being permissive for induction, whereas bacteria that had been grown in high-osmolarity media and then shifted to low-osmolarity LB were still able to induce exoS expression to a significant extent, especially if the shift occurred at the time of induction (Fig. S2). The simplest interpretation of these data is that being permissive for induction of exoS expression is associated with a cellular component that is not rapidly turned over, suggesting that it is not simply the result of a shift in pool size of a given metabolite.
Fig. 2.
Osmolarity controls the ability to induce exoS expression. Strain PAK harbouring the exoS∷GFP-lacZ reporter was grown in LB containing the indicated concentration of NaCl (A, B) or 5 mM NaCl and 271 mM sucrose (D). The grey overlay indicates the distribution for cultures in which calcium had been chelated by the addition of NTA to the media. Panel C, a graph plotting the percentage of cells that induce exoS expression after removal of calcium from the media (y-axis) when grown in LB with the indicated NaCl concentration (x-axis). Panel E depicts levels of exoS expression as measured using the lacZ reporter in LB with 5 mM or 200 mM NaCl in the absence or presence of chelator (NTA). The actual activities are noted above each column of the graph.
While osmolarity controls the percentage of cells that induce exoS expression upon removal of calcium from the medium, osmolarity also affects the basal level of exoS expression in calcium-replete media. The basal level of exoS expression varies about fivefold between low- and high-osmolarity LB, as assayed using a lacZ reporter (Fig. 2E).
Induction of exoS in regulatory mutants under low- and high-salt conditions
To determine how this osmotic regulation integrates with the activity of the known regulators of the type III secretion regulon, we combined the reporter with deletions in exsD, exsE, pcrV and popN. ExsD is an anti-activator that binds ExsA and prevents induction of the type III secretion regulon. As mentioned in the Introduction, deletion of exsD results in constitutive expression of the type III secretion regulon. ExsE is a recently discovered negative regulator that is secreted via the type III secretion machinery upon triggering of secretion. ExsE binds ExsC and prevents activation of ExsA under conditions in which the secretion apparatus is closed. PcrV is also involved in the low-calcium response. PcrV is involved in inserting the translocase into the target cell membrane (Goure et al., 2004). It is likely to be surface exposed because antibodies directed against PcrV are commonly found in patient sera. Antibodies directed against PcrV are protective in an animal model and reduce cytotoxicity in an in vitro infection model (Sawa et al., 1999; Holder et al., 2001b; Moss et al., 2001; Frank et al., 2002; Faure et al., 2003). Like exsD mutants, deletion mutants lacking pcrV express the type III secretion regulon constitutively. popN mutants are deregulated for secretion and also express the type III secretion regulon constitutively (Sundin et al., 2004). PopN, like PcrV, is itself exported via the type III secretion apparatus and has been proposed to impose a block on secretion under repressing conditions. Unlike PcrV, PopN does not appear to be important for translocation of effectors into targeted host cells because a popN mutant is cytotoxic (Sundin et al., 2004).
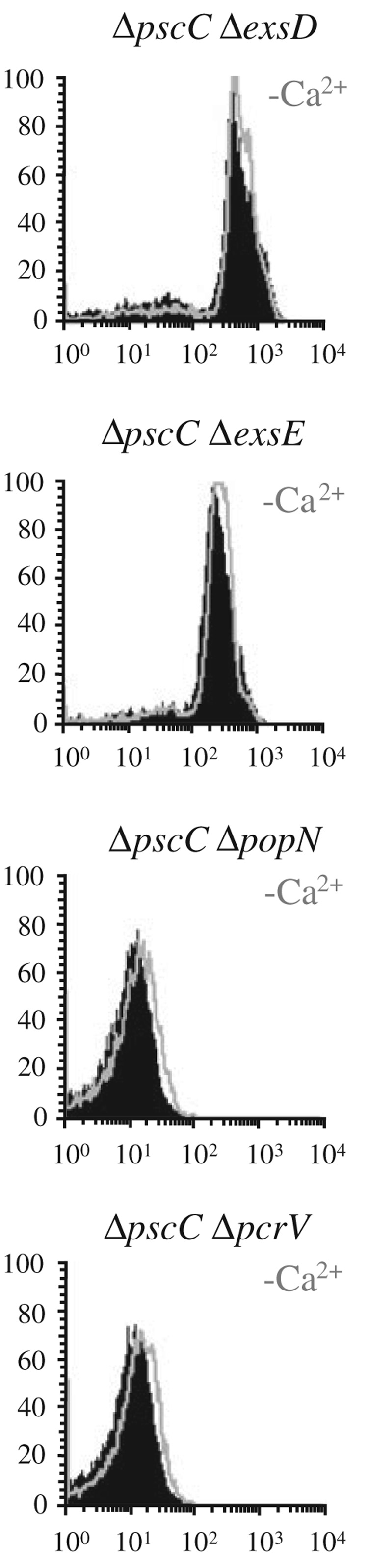
We grew exsD, exsE, pcrG and pcrV mutants of PAK harbouring the exoS-GFP reporter under low-salt and high-salt conditions and assayed exoS transcription under calcium-depleted and calcium-replete conditions. The exsD and exsE mutants displayed high levels of exoS transcription under all conditions tested (Fig. 3). This is in marked contrast to the popN and pcrV mutants, which only displayed high levels of transcription in the high-salt media (Fig. 3).
Fig. 3.
Effect of regulatory mutations on induction in high- and low-osmolarity media. Indicated mutants of strain PAK were grown in LB with low (5 mM) or high (200 mM) salt and assayed, by FACS, for exoS expression in the presence (black) or absence (grey overlay) of calcium using a chromosomal exoS-GFP reporter.
While exsD, exsE, popN and pcrV mutants have been described to constitutively express exoenzymes, they differ in one key respect. In exsD and exsE mutants, secretion is still low-calcium-dependent, whereas in popN and pcrV mutants, it is deregulated (McCaw et al., 2002; Sundin et al., 2002; 2004; Rietsch et al., 2005; Urbanowski et al., 2005). The constitutive induction phenotype of the popN and pcrV mutants could therefore be the result of constitutive secretion of ExsE and should therefore be type III secretion-dependent.
To determine if the deregulated expression of the type III secretion genes in the popN and pcrV mutants is type III-dependent, we combined the exsD, exsE, popN and pcrV mutations with a deletion in pscC, which encodes the outer membrane secretin of the type III secretion system. Indeed, expression of exoS continued unabated in the exsD pscC and exsE pscC mutants, but the popN pscC and the pcrV pscC double mutants, were unable to induce exoS expression, even under low-calcium conditions (Fig. 4). Taken together, these data suggest that the defect in exoS induction in low-osmolarity media behaves like a type III secretion defect and could therefore be the result of a defect in assembly of the type III secretion machinery. Interestingly, closer observation of the relative fluorescence of the exsD and exsE mutants harbouring the exoS-GFP reporter at low and high-osmolarity suggests that the basal level of transcription still varies with osmolarity and is therefore subject to a second, unrelated mechanism of regulation (Fig. 3).
Fig. 4.
exoS expression in various regulatory mutations in the context of a defective type III secretion apparatus. Derivatives of strain PAK bearing the indicated deletion mutations were assayed, by FACS, for exoS expression in LB with 200 mM NaCl in the presence (black) or absence (grey overlay) of calcium.
PcrV is not surface-displayed in P. aeruginosa grown in low-osmolarity media
To test directly if the type III secretion machinery is assembled properly in bacteria grown in low-osmolarity media, we tested if PcrV is displayed on the surface of these bacteria. As PcrV is itself secreted via the type III secretion machinery (Yahr et al., 1997), its surface localization should serve as a convenient indicator of the ability of the bacteria to assemble a functional type III secretion apparatus. We fixed bacteria grown in low- or high-osmolarity media before staining them with an anti-PcrV antiserum and subjecting them to immunofluorescence microscopy. PcrV is clearly displayed on the surface of bacteria grown in high-osmolarity media (Fig. 5A). This is not the case for bacteria grown under low-osmolarity conditions (Fig. 5A). The control strains, a pscC mutant lacking the secretin of the type III secretion machinery, and a pcrV mutant strain were both negative for the PcrV signal (Fig. 5A). Control staining of the outer membrane porin OprH demonstrated that growth under low-osmolarity conditions did not interfere with the immunofluorescence staining procedure. To obtain a more quantitative analysis of this result, the stained cells were analysed for their fluorescent signal by FACS. These data confirm the microscopy data (Fig. 5B). In high-osmolarity media about 40% of cells are positive for the PcrV signal, whereas in low-osmolarity media, less than 1% of the cells stained positive. The controls were also both negative. Western blot analysis demonstrated that PcrV is produced under both conditions, although the level of expression was about fivefold lower in low-osmolarity media than in high-osmolarity media (Fig. S3A), which is consistent with the drop in the basal level of exoS expression we observed. Furthermore, PcrV is not aberrantly released into the supernatant under low-calcium conditions but remains associated with the cells (Fig. S3B).
Fig. 5.
Assembly of the type III secretion machinery in low- and high-salt media.
A. Bacteria grown in LB with 5 mM or 200 mM NaCl were stained with the indicated primary antibody (anti-PcrV or anti-OprH antiserum) and detected using a AlexaFluor 488 coupled secondary antibody. Phase contrast and fluorescence images of representative fields were overlayed. The pcrV and pscC mutants were only grown in LB with 200 mM NaCl.
B. FACS analysis of antibody stained cells. All strains were stained with the PcrV and AlexaFluor-488 coupled secondary antibody as described above and analysed by FACS. The wild-type and exsE mutant strain were grown in LB with 5 mM NaCl (black) or 200 mM NaCl (grey). The pcrV and pscC mutant strains were only grown in LB with 200 mM NaCl.
One could argue that the inability to assemble the type III secretion machinery under low-osmolarity conditions is the result of the decrease in expression of the structural genes. Perhaps a critical threshold of expression is required to be able to assemble the machinery, and the decrease in transcription directly leads to the type III negative phenotype. To test this possibility we examined the ability of an exsE mutant, which expresses the type III secretion genes constitutively and to a high level, even in low-osmolarity media, to display PcrV on the surface of the bacterial cell. Even in the exsE mutant, however, the bacteria were not able to assemble a functional type III secretion machinery (Fig. 5B). This result suggests that the phenotype is the result of a defect in assembly or function of the machinery, or a post-transcriptional defect in expression, rather than a problem with the basal transcription level of the structural genes.
Tracing the nature of the metabolic cue
To begin to understand the nature of the metabolic cue that results in the type III secretion negative phenotype, we decided to use the FACS-based assay to monitor the effect of metabolic mutations on expression of exoS.
Pseudomonas aeruginosa is unable to induce exoS expression when grown in minimal medium with glucose as sole carbon source. Expression can be restored by adding back amino acids, for example, in the form of tryptone. We decided to use minimal medium with 0.1% tryptone and glutamate as a semi-defined condition to investigate this phenotype genetically. In this medium, about 35% of the cells induce exoS expression when calcium is removed from the medium, allowing for mutations that can shift the equilibrium towards either induction or repression. Based on the published metabolic conditions that affect expression, as well as preliminary data that suggested that adding back glutamate was able to partially restore induction (Fig. 1D), we decided to focus on mutations that impair the tricarboxylic acid cycle. Mutations in a large number of metabolic genes were analysed. The results of mutations that best define the metabolic pathway involved in the regulation are illustrated in Fig. 6. Deletion of aceA, one of the structural subunits of pyruvate dehydrogenase, was able to prevent induction in our media, whereas deletion of gltA (the main citrate synthetase) and prpC (citrates synthetase II), was able to enhance induction. Interestingly, the aceA mutation had no effect on induction in rich media in our hands (data not shown), somewhat contradicting previous data (Dacheux et al., 2002). However, subtle differences in media conditions or strains could account for this discrepancy. Taken together, these data suggest that acetyl-CoA, or a derivative thereof exerts metabolic control over exoS expression. Osmotic regulation and regulation seen when comparing minimal versus rich media are related and likely to be facets of the same regulatory phenomena, because the gltA and gltA prpC mutations also partially suppressed the inability of P. aeruginosa to induce exoS expression under low-osmolarity conditions (Table 1).
Fig. 6.
Metabolic mutations control exoS induction. Wild-type P. aeruginosa as well as the indicated mutants were grown in MOPS minimal media supplemented with 0.1% tryptone and 20 mM glutamate. exoS expression was induced by removal of calcium from the media and analysed by FACS in the presence (black) or absence (grey overlay) of calcium (aceE, pyruvate dehydrogenase subunit E1, gltA, citrate synthetase 1, prpC, citrate synthetase 2). The percentage of cells that induce exoS expression in the absence of calcium is indicated in the upper right-hand corner of each graph.
Table 1.
Intracellular cAMP concentrations in low- and high-osmolarity media.
| Strain | [NaCl] in media | Intracellular [cAMP] in µM | % of cells that induce exoS expression upon removal of calcium | Basal level of exoS expressiona |
|---|---|---|---|---|
| PAK | 5 mM | 1.2 ± 0.4 | 4% | 48 ± 6 |
| PAK | 200 mM | 5.1 ± 0.8 | 70% | 214 ± 5 |
| PAK ΔgltA ΔprpC | 5 mM | 2.7 ± 1.0 | 15% | 56 ± 3 |
| PAK/pP35-cyaB | 5 mM | 3.3 ± 0.8 | 23% | nd |
β-Galactosidase activity in Miller Units.
nd, not determined.
The nature of the metabolite or structural change in the cell (e.g. change in composition of the cell wall) that regulates the type III secretion machinery assembly is as yet unclear. Mutations in ackA and pta do not affect regulation, suggesting that acetylphosphate, which has been linked to regulation of SPI1 gene expression in Salmonella (Lawhon et al., 2002), is not a factor (data not shown). Similarly, no striking changes were observed in phospholipid head-group or fatty-acid side-chain composition (data not shown). While there are still other metabolites that derive from acetyl-CoA, future research will be aimed at understanding the nature of the assembly defect, which may shed light on the metabolite underlying the regulation.
While trying to express GFP under the control of the Escherichia coli lac promoter, in low- and high-osmolarity media we discovered that expression of GFP in P. aeruginosa was strongly regulated by the osmolarity of the media (data not shown). This defect could be partially suppressed by replacing the wild-type lac promoter with the cAMP-independent lacUV5 promoter, suggesting that changes in osmolarity result in changes in cAMP levels in P. aeruginosa. As cAMP regulates exoS expression, we measured cAMP levels in bacteria grown in low- and highosmolarity media. Growth in high-osmolarity media resulted in higher levels of cAMP than growth in low-osmolarity media (Table 1), suggesting that changes in intracellular cAMP concentration may be responsible, at least in part, for the inability to induce expression in low-osmolarity LB. Consistent with this, overexpression of the adenylate cyclase CyaB was able to partially restore the ability to induce exoS expression in low-osmolarity media (Table 1). Interestingly, the ΔgltA ΔprpC double mutant, which also partially restored the low-calcium induction of exoS expression in low-osmolarity media, resulted in elevated levels of cAMP. Compared with wild type, however, the ΔgltA ΔprpC double mutation did not significantly elevate the basal level of exoS transcription (Table 1), suggesting that the two modes of regulation are distinct.
Discussion
A series of papers have established that the regulation of the type III secretion genes in P. aeruginosa is intimately tied to the metabolic state of the cell. Pyruvate dehydrogenase mutants have been described as being noncytotoxic and unable to induce type III secretion gene expression. Osmolarity has been described as influencing exoS expression. Aberrant overexpression of a histidine transporter and subsequent uptake and metabolism of histidine also renders cells non-cytotoxic. Using a FACS-based assay to study induction of exoS at the single-cell level, we were able to determine that metabolically unfavourable conditions change the percentage of the population that can induce exoS expression, as well as the absolute level of expression per cell. We furthermore present data that the decrease in the percentage of cells that induce exoS expression is in fact due to a defect affecting either assembly or activity of the machinery. This defect causes a type III secretion negative phenotype, which in turn prevents induction by removal of calcium from the media.
Metabolism plays an important role in regulation of virulence factors in a wide variety of pathogens. In Salmonella, expression of the SPI1 type III secretion system depends on the alarmone ppGpp (Pizarro-Cerda and Tedin, 2004) and requires high salt and anaerobic growth (Lee and Falkow, 1990). Short chain fatty acids may also affect invasion gene expression in Salmonella typhimurium by altering levels of acetyl phosphate (Lawhon et al., 2002). In Listeria, pyruvate dehydrogenase is required for growth in macrophage (O’Riordan et al., 2003). Expression of cholera toxin in classical strains of Vibrio cholerae can be induced by growth at 30°C in media with a decreased pH (Waldor and Mekalanos, 1996), whereas induction in the El Tor biotype can be induce in vitro by growth under AKI conditions (Iwanaga et al., 1986), which involve a fixed regimen of anaerobic, followed by aerobic growth, suggesting that there is a physiological cue that governs expression of this key virulence factor. Even with many examples to draw upon, for the most part, the mechanism by which metabolism exerts its control is unclear.
By using a combination of β-galactosidase assay and FACS analysis, we were able to determine that, in P. aeruginosa, growth under metabolically unfavourable conditions affects expression of the type III secretion genes in two ways. It reduces the basal level of exoS expression and decreases the percentage of cells that induce exoS expression upon removal of calcium from the medium. The two phenomena can be separated genetically, because in the exsD or exsE mutants, exoS expression is induced in all cells, even under metabolically unfavourable conditions, but the absolute level of expression per cell is reduced.
The block imposed on exoS expression by growth in metabolically unfavourable conditions behaves, genetically, like a type III secretion mutant. Mutant strains lacking exsD or exsE do not require an intact type III secretion system for constitutive expression of exoS, whereas mutants that do require an intact type III secretion system, pcrV and popN, are metabolically regulated. The decrease in the percentage of cells that induce exoS expression upon calcium removal correlates with an inability to display PcrV on the surface demonstrating that assembly or function of the type III secretion machinery is impaired. Constitutive expression of the type III secretion regulon in an exsE mutant was not able to restore the ability of the bacteria to assemble a functional type III secretion apparatus in low-osmolarity LB. The immunofluorescence data presented in this paper clearly demonstrate that P. aeruginosa assembles a functional type III secretion apparatus even under calcium-replete conditions. Furthermore, the data suggest that P. aeruginosa, on average, only assembles one or two type III secretion complexes per cell and that these secretion machineries are not necessarily located directly at the pole of the cell. Experiments using GFP labelled components of the apparatus will be needed to confirm and extend these observations.
While it is interesting to speculate about the role of osmolarity in regulation of this important virulence factor, it should be noted that changes in osmolarity alone are not sufficient to signal whether or not an environment is metabolically favourable. Simply increasing the salt concentration in minimal glucose media was not able to restore the ability of P. aeruginosa to induce exoS expression, while addition of glutamate did partially restore the ability to induce, suggesting that osmolarity, which was our primary means of controlling the metabolic ‘on’ and ‘off’ condition in this paper, was in fact merely a convenient way of skewing metabolism in a way that allows or prevents exoS induction upon removal of calcium from the media. The original thought in using osmolarity to manipulate the type III regulon stemmed from the observation that addition of glutamate to minimal glucose media was able to partially restore exoS induction. Increases in osmolarity result in significant increases in intracellular glutamate, and presumably alter flux through the citric acid cycle. Using the FACS assay, we therefore probed the citric acid cycle using metabolic mutation in order to triangulate the metabolite that controls exoS expression. Our efforts were met with partial success, because we were able to demonstrate that mutations in the citrate synthetases were able to increase the percentage of cells that induce exoS expression in a semi-defined media. Mutations in pyruvate dehydrogenase, on the other hand, reduced the percentage of cells that were able to induce exoS upon removal of calcium from the media. These experiments suggest that the critical metabolite regulating the type III regulon is acetyl-CoA or derived from acetyl-CoA. Deletion of both the gene for acetate kinase and that encoding phospho-transacetylase, ackA and pta respectively, did not affect the ability of the bacteria to induce exoS expression, suggesting that acetyl-phosphate is not, in fact, the controlling metabolite. Similarly, there were no overwhelming changes in the composition of the headgroups or side-chains of the phospholipids in the inner and outer membrane of P. aeruginosa grown under our test conditions. There are other metabolites that branch from acetyl-CoA. The gltA prpC double mutant was able to partially restore the ability of bacteria to induce exoS expression, but not elevate the basal level of expression, suggesting that these mutations restore the ability of the bacteria to assemble a functional type III secretion apparatus. As the type III secretion apparatus is a complex machinery that spans both the inner and the outer membrane, as well as the peptidoglycan layer of the cell, it seems reasonable to propose that the inability to induce exoS expression under metabolically unfavourable conditions is the result of a change in the envelope that prevents either assembly or proper function of the apparatus. Experiments are currently underway to better understand how the machinery is assembled. With regard to metabolic regulation, a better understanding of how this complex apparatus is assembled should lead to insights that will help characterize the assembly defect under metabolically unfavourable conditions and further focus our efforts to understand this remarkable phenomenon.
There are several global regulatory pathways that impact the expression of the type III secretion genes. The P. aeruginosa CRP homologue, Vfr, and the adenylate cyclases CyaA and CyaB are critically important for the expression of the type III secretion regulon. Interestingly, we were able to demonstrate that changes in osmolarity also alter the intracellular concentration of cAMP, and that artificially increasing the level of cAMP in the cell by over-expression of an adenylate cyclase was able to partially restore the ability to induce exoS expression, even in low-osmolarity media. As the gltA prpC double null mutation also resulted in elevated levels of cAMP, compared with the parental strain, it suggests that the cAMP pathway regulates, among other things, a gene or genes that are important for the proper assembly of the type III secretion machinery. Another way of interpreting the latter result is that the metabolic signal that derives form acetyl-CoA controls the activity of one or both adenylate cyclases. The second global regulatory pathway, the GacA/GacS system, however, does not appear to be relevant in this context of metabolic regulation, because expression of exoS in a gacS mutant is still media-dependent.
The fact that metabolism can severely impact the expression and assembly of the type III secretion machinery of P. aeruginosa is of considerable interest. It suggests that, if the controlling metabolic pathway is fully elucidated, it will provide a new target for antimicrobial development, with several potential benefits. As the controlling factor is a metabolite, it may be possible to use it as a lead compound for inhibitor development. Second, if the metabolite is an essential component of the cell, developing inhibitors of its synthesis may result in a bactericidal compound which, even if present at subinhibitory concentrations, can still impact the assembly of a key virulence factor.
We have presented evidence that metabolism controls expression of the type III secretion regulon in P. aeruginosa at both the level of basal transcription, as well as the assembly of the apparatus. These observations suggest that P. aeruginosa undergoes physiological changes when growing in the host, which directly influence the ability of the bacterium to express key virulence factors and open exciting new possibilities for therapeutic intervention.
Experimental procedures
Bacterial strains and media
All P. aeruginosa strains described in this study are listed in Table 2. E. coli DH5α λpir and SM10 λpir were used, respectively, for cloning and conjugative transfer of plasmids into P. aeruginosa and were derived from laboratory stocks. Bacteria were routinely grown in L-broth (10 g tryptone, 5 g yeast extract, 5 g NaCl per litre). For experiments in which the effect of osmolarity was taken into account, bacteria were grown in LB (containing 10 g tryptone and 5 g yeast extract per litre) supplemented with 10 mM MgCl2 and either 5 mM or 200 mM NaCl. MOPS minimal media were prepared as described (Neidhardt et al., 1974).
Table 2.
Strains and plasmids.
| Strain | Relevant genotype | Reference/source |
|---|---|---|
| RP631 | P. aeruginosa strain PAK, wild-type | Takeya and Amako (1966) |
| RP1407 | PAK ΔpcrV | Rietsch et al. (2005) |
| RP1370 | PAK ΔpscC | Rietsch et al. (2005) |
| RP955 | PAK ΔexoS: GFP-lacZ | Rietsch et al. (2004) |
| RP1224 | PAK ΔexoS∷ GFP-lacZ ΔexsD | Rietsch et al. (2005) |
| RP1389 | PAK ΔexoS∷ GFP-lacZ ΔexsE | Rietsch et al. (2005) |
| RP1221 | PAK ΔexoS∷ GFP-lacZ ΔpscC | Rietsch et al. (2005) |
| RP1192 | PAK ΔexoS∷ GFP-lacZ ΔpcrV | This study |
| RP1509 | PAK ΔexoS∷ GFP-lacZ ΔpopN | This study |
| RP1302 | PAK ΔexoS∷ GFP-lacZ ΔexsD ΔpscC | This study |
| RP1438 | PAK ΔexoS∷ GFP-lacZ ΔexsE ΔpscC | Rietsch et al. (2005) |
| RP1305 | PAK ΔexoS∷ GFP-lacZ ΔpcrV ΔpscC | This study |
| RP1303 | PAK ΔexoS∷ GFP-lacZ ΔpopN ΔpscC | This study |
| RP1150 | PAK ΔexoS∷ GFP-lacZ ΔaceA | This study |
| RP987 | PAK ΔexoS∷ GFP-lacZ ΔgltA | This study |
| RP1152 | PAK ΔexoS∷ GFP-lacZ ΔgltA ΔprpC | This study |
| Plasmid | Description | Reference/source |
| pPSV35 | Shuttle vector with gentamicin-resistance gene (aacC1), PA origin, lacIq, and the lacUV5 promoter and MCS of pUC18 | Rietsch et al. (2005) |
| pP35-cyaB | cyaB gene cloned into pPSV35 | This study |
Plasmids and strain construction
Deletion mutations were constructed using plasmid pEXG2 (Rietsch et al., 2005). In-frame deletions were constructed by first generating two PCR products that carried the flanking regions for the deletion site. The two products were then spliced together in a second PCR reaction. The internal primers for the two PCR products defined the site of the deletion, as well as complementary 24 bp sequences (5′-TTCAGCATGCTTGCGGCTCGAGTT-3′ and 5′-AACTCGAGCCGCAAGCATGCTGAA-3′ respectively), that allowed the two initial PCR products to be spliced together to generate the in-frame deletion. All primer sequences are available upon request.
β-Galactosidase assays
Cells were permeabilized with chloroform/SDS, and β-galactosidase activity was assayed as described previously (Miller, 1992).
FACS analysis
Overnight cultures of strains were diluted 1:200 into the appropriate fresh media and grown for 2 h at 37°C. At this point, the cultures were split into two and diluted into an equal volume of prewarmed media with or without NTA (final concentration 10 mM). Bacteria were then incubated with aeration for another 3 h at 37°C and subsequently chilled on ice for at least 10 min. The chilled bacteria were diluted into PBS and assayed for fluorescence using a FACScalibur flow cytometer (Becton Dickinson).
Immunofluorescence microscopy
Bacterial cells were stained based on a previously described protocol (Watarai et al., 2001). Overnight cultures of each strain were diluted 1:200 and grown in the appropriate media until mid-log. At this point 0.5 ml of culture were centrifuged, the supernatant was discarded and the bacteria were resuspended in a 2% solution of paraformaldehyde in KPO4 buffer (150 mM, pH 7.4) and incubated on ice for 30 min. The bacteria were subsequently pelleted, washed twice with PBS containing 2% goat serum (PBS-G) and resuspended in 40 µl of PBS-G. After 30 min of incubation at 37°C, the primary antibody was added at a final dilution of 1:250 (by addition of 10 µl of a 1:50 dilution in PBS-G) and the cells were incubated another 45 min at 37°C. At this point, 1 ml of PBSG was added to each sample and the cells were again pelleted, washed once with 1 ml of PBS-G and resuspended in 50 µl of PBS-G containing a 1:500 dilution of a Alexa-fluor 488 conjugated goat-anti-rabbit secondary antibody (Molecular Probes). After 45 min of incubation at 37°C, in the dark, 1 ml of PBS-G was added to each sample and the cells were pelleted, washed twice with PBS-G, and resuspended in 30 µl PBS. Ten microlitres of each sample was spread evenly on a polylysine-coated microscopy slide, dried, covered with a drop of VectaShield mounting medium (Vector Laboratories) and a coverslip.
cAMP measurements
Bacteria were diluted 1:200 from overnight cultures and grown to mid-log in the appropriate medium at 37°C. The cultures were then chilled on ice for 5–10 min and 1 ml was removed and placed into a microcentrifuge tube. The bacteria were pelleted and washed twice with sterile, ice-cold saline (0.9% NaCl), then resuspended in 200 µl of ice-cold ddH2O. One hundred microlitres of the suspension were used to determine the OD600, the other half was extracted with HCl (0.1 N final concentration) for 15 min on ice. The extract was cleared by pelleting the cells. The supernatant was then transferred into a fresh microcentrifuge tube and dried in a SpeedVac. The amount of cAMP in the extract was determined using a commercial kit (Cayman) and the intracellular concentration was extrapolated, based on published values (D’Souza-Ault et al., 1993), by using the OD600 to determine the volume of the free intracellular water of the extracted bacteria. Changes in free intracellular water due to changes in osmolarity were taken into account.
Supplementary Material
The following supplementary material is available for this article online:
FACS analysis of a plasmid-based exoS promoter (A) or toluic acid promoter (B) driving GFP expression in P. aeruginosa strain PAK. The concentration of inducer NTA (A) or toluic acid (B) is given in mM in the right hand corner of each plot.
Shift experiments: PAK ΔexoS∷GFP-lacZ (RP955) was grown overnight in LB with either 5 mM NaCl (low) or 200 mM NaCl (high), diluted 1:200 into fresh media and grown at 37°C with aeration. After 2 h, the culture was split and diluted 1:1 into LB with or without 10 mM NTA. Cultures were shifted between media of different osmolarity by pelleting cells in a microcentrifuge, washing them once with the medium the cells were being shifted to and resuspending them in the new medium at the indicated times (T0, at the time of diluting the overnight, or T2 after 2 h of incubation, at the time of the shift to media with or without NTA). After 3 h of incubation the cultures were chilled on ice and GFP fluorescence was assayed by FACS analysis [black, calcium replete; grey, in the presence of chelator (NTA)].
PcrV production and localization in low- and highosmolarity LB. (A) Western blot of cell lysates of PAK ΔexoS∷GFP-lacZ(RP955) grown in LB with 5 mM NaCl (low) or 200 mM NaCl (high). Cells were grown to mid-log, pelleted and resuspended in SDS-sample buffer at an OD600 of 1. 10 µl of each lysate, and of a 1:4 and 1:16 dilution thereof, were separated by 10% SDS-PAGE and transferred to nitrocellulose. The blot was probed with a rabbit anti-PcrV antiserum and developed using a secondary antibody conjugated to HRP. After the first hybridization, the blot was stripped using a commercial stripping reagent (Pierce) and reprobed using a commercial mouse monoclonal-antibody directed against the α-subunit of RNA polymerase (Neoclone). (B) RP955 was grown in low- or high-osmolarity LB for 2 h at which point the culture was split and diluted 1:1 into LB of the same osmolarity with or without the chelator NTA. After 2 h of growth the cultures were chilled on ice and fractionated into cell pellet and supernatant fractions (supernatant proteins were precipitated using trichloroacetic acid at a final concentration of 10%). All fractions were subjected to SDS-PAGE, transferred to nitrocellulose and probed with an anti-PcrV antiserum.
This material is available as part of the online article from http://www.blackwell-synergy.com
Acknowledgements
We would like to thank Dr Robert Hancock for the anti-OprH antiserum, as well as Dr Ina Attree for the anti-PcrV antiserum. This research was supported by National Institutes of Health Grant AI26289 to J. J. M.
References
- Barbieri JT. Pseudomonas aeruginosa exoenzyme S, a bifunctional type-Ill secreted cytotoxin. Int J Med Microbiol. 2000;290:381–387. doi: 10.1016/S1438-4221(00)80047-8. [DOI] [PubMed] [Google Scholar]
- Carratala J, Roson B, Fernandez-Sevilla A, Alcaide F, Gudiol F. Bacteremic pneumonia in neutro-penic patients with cancer: causes, empirical antibiotic therapy, and outcome. Arch Intern Med. 1998;158:868–872. doi: 10.1001/archinte.158.8.868. [DOI] [PubMed] [Google Scholar]
- D’Souza-Ault MR, Smith LT, Smith GM. Roles of N-acetylglutaminylglutamine amide and glycine betaine in adaptation of Pseudomonas aeruginosa to osmotic stress. Appl Environ Microbiol. 1993;59:473–478. doi: 10.1128/aem.59.2.473-478.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dacheux D, Epaulard O, de Groot A, Guery B, Leberre R, Attree I, et al. Activation of the Pseudomonas aeruginosa type III secretion system requires an intact pyruvate dehydrogenase aceAB operon. Infect Immun. 2002;70:3973–3977. doi: 10.1128/IAI.70.7.3973-3977.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasgupta N, Lykken GL, Wolfgang MC, Yahr TL. A novel anti-anti-activator mechanism regulates expression of the Pseudomonas aeruginosa type III secretion system. Mol Microbiol. 2004;53:297–308. doi: 10.1111/j.1365-2958.2004.04128.x. [DOI] [PubMed] [Google Scholar]
- Faure K, Fujimoto J, Shimabukuro DW, Ajayi T, Shime N, Moriyama K, et al. Effects of monoclonal anti-PcrV antibody on Pseudomonas aeruginosa-induced acute lung injury in a rat model. J Immune Based Ther Vaccines. 2003;1:2. doi: 10.1186/1476-8518-1-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feltman H, Schulert G, Khan S, Jain M, Peterson L, Hauser AR. Prevalence of type III secretion genes in clinical and environmental isolates of Pseudomonas aeruginosa. Microbiology. 2001;147:2659–2669. doi: 10.1099/00221287-147-10-2659. [DOI] [PubMed] [Google Scholar]
- Frank DW. The exoenzyme S regulon of Pseudomonas aeruginosa. Mol Microbiol. 1997;26:621–629. doi: 10.1046/j.1365-2958.1997.6251991.x. [DOI] [PubMed] [Google Scholar]
- Frank DW, Iglewski BH. Cloning and sequence analysis of a trans-regulatory locus required for exoenzyme S synthesis in Pseudomonas aeruginosa. J Bacteriol. 1991;173:6460–6468. doi: 10.1128/jb.173.20.6460-6468.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank DW, Vallis A, Wiener-Kronish JP, Roy-Burman A, Spack EG, Mullaney BP, et al. Generation and characterization of a protective monoclonal antibody to Pseudomonas aeruginosa PcrV. J Infect Dis. 2002;186:64–73. doi: 10.1086/341069. [DOI] [PubMed] [Google Scholar]
- Garau J, Gomez L. Pseudomonas aeruginosa pneumonia. Curr Opin Infect Dis. 2003;16:135–143. doi: 10.1097/00001432-200304000-00010. [DOI] [PubMed] [Google Scholar]
- Goehring UM, Schmidt G, Pederson KJ, Aktories K, Barbieri JT. The N-terminal domain of Pseudomonas aeruginosa exoenzyme S is a GTPase-activating protein for Rho GTPases. J Biol Chem. 1999;274:36369–36372. doi: 10.1074/jbc.274.51.36369. [DOI] [PubMed] [Google Scholar]
- Goodman AL, Kulasekara B, Rietsch A, Boyd D, Smith RS, Lory S. A signaling network reciprocally regulates genes associated with acute infection and chronic persistence in Pseudomonas aeruginosa. Dev Cell. 2004;7:745–754. doi: 10.1016/j.devcel.2004.08.020. [DOI] [PubMed] [Google Scholar]
- Goure J, Pastor A, Faudry E, Chabert J, Dessen A, Attree I. The V antigen of Pseudomonas aeruginosa is required for assembly of the functional PopB/PopD translocation pore in host cell membranes. Infect Immun. 2004;72:4741–4750. doi: 10.1128/IAI.72.8.4741-4750.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauser AR, Cobb E, Bodi M, Mariscal D, Valles J, Engel JN, Rello J. Type III protein secretion is associated with poor clinical outcomes in patients with ventilator-associated pneumonia caused by Pseudomonas aeruginosa. Crit Care Med. 2002;30:521–528. doi: 10.1097/00003246-200203000-00005. [DOI] [PubMed] [Google Scholar]
- Holder IA, Neely AN, Frank DW. Type III secretion/intoxication system important in virulence of Pseudomonas aeruginosa infections in burns. Burns. 2001a;27:129–130. doi: 10.1016/s0305-4179(00)00142-x. [DOI] [PubMed] [Google Scholar]
- Holder IA, Neely AN, Frank DW. PcrV immunization enhances survival of burned Pseudomonas aeruginosa-infected mice. Infect Immun. 2001b;69:5908–5910. doi: 10.1128/IAI.69.9.5908-5910.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornef MW, Roggenkamp A, Geiger AM, Hogardt M, Jacobi CA, Heesemann J. Triggering the ExoS regulon of Pseudomonas aeruginosa: a GFP-reporter analysis of exoenzyme (Exo) S, ExoT and ExoU synthesis. Microb Pathog. 2000;29:329–343. doi: 10.1006/mpat.2000.0398. [DOI] [PubMed] [Google Scholar]
- Iwanaga M, Yamamoto K, Higa N, Ichinose Y, Nakasone N, Tanabe M. Culture conditions for stimulating cholera toxin production by Vibrio cholerae O1 El Tor. Microbiol Immunol. 1986;30:1075–1083. doi: 10.1111/j.1348-0421.1986.tb03037.x. [DOI] [PubMed] [Google Scholar]
- Knight DA, Finck-Barbancon V, Kulich SM, Barbieri JT. Functional domains of Pseudomonas aeruginosa exoenzyme S. Infect Immun. 1995;63:3182–3186. doi: 10.1128/iai.63.8.3182-3186.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krall R, Schmidt G, Aktories K, Barbieri JT. Pseudomonas aeruginosa ExoT is a Rho GTPase-activating protein. Infect Immun. 2000;68:6066–6068. doi: 10.1128/iai.68.10.6066-6068.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laskowski MA, Osborn E, Kazmierczak BI. A novel sensor kinase-response regulator hybrid regulates type III secretion and is required for virulence in Pseudomonas aeruginosa. Mol Microbiol. 2004;54:1090–1103. doi: 10.1111/j.1365-2958.2004.04331.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawhon SD, Maurer R, Suyemoto M, Altier C. Intestinal short-chain fatty acids alter Salmonella typhimurium invasion gene expression and virulence through BarA/SirA. Mol Microbiol. 2002;46:1451–1464. doi: 10.1046/j.1365-2958.2002.03268.x. [DOI] [PubMed] [Google Scholar]
- Lee CA, Falkow S. The ability of Salmonella to enter mammalian cells is affected by bacterial growth state. Proc Natl Acad Sci USA. 1990;87:4304–4308. doi: 10.1073/pnas.87.11.4304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCaw ML, Lykken GL, Singh PK, Yahr TL. ExsD is a negative regulator of the Pseudomonas aeruginosa type III secretion regulon. Mol Microbiol. 2002;46:1123–1133. doi: 10.1046/j.1365-2958.2002.03228.x. [DOI] [PubMed] [Google Scholar]
- McGuffie EM, Frank DW, Vincent TS, Olson JC. Modification of Ras in eukaryotic cells by Pseudomonas aeruginosa exoenzyme S. Infect Immun. 1998;66:2607–2613. doi: 10.1128/iai.66.6.2607-2613.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller JH. A Short Course in Bacterial Genetics: a Laboratory Manual and Handbook for Escherichia coli and Related Bacteria. Plainview, New York: Cold Spring Harbor Laboratory Press; 1992. [Google Scholar]
- Moss J, Ehrmantraut ME, Banwart BD, Frank DW, Barbieri JT. Sera from adult patients with cystic fibrosis contain antibodies to Pseudomonas aeruginosa type III apparatus. Infect Immun. 2001;69:1185–1188. doi: 10.1128/IAI.69.2.1185-1188.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidhardt FC, Bloch PL, Smith DF. Culture medium for enterobacteria. J Bacteriol. 1974;119:736–747. doi: 10.1128/jb.119.3.736-747.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishijyo T, Haas D, Itoh Y. The CbrA-CbrB two-component regulatory system controls the utilization of multiple carbon and nitrogen sources in Pseudomonas aeruginosa. Mol Microbiol. 2001;40:917–931. doi: 10.1046/j.1365-2958.2001.02435.x. [DOI] [PubMed] [Google Scholar]
- O’Riordan M, Moors MA, Portnoy DA. Listeria intracellular growth and virulence require host-derived lipoic acid. Science. 2003;302:462–464. doi: 10.1126/science.1088170. [DOI] [PubMed] [Google Scholar]
- Olson JC, McGuffie EM, Frank DW. Effects of differential expression of the 49-kilodalton exoenzyme S by Pseudomonas aeruginosa on cultured eukaryotic cells. Infect Immun. 1997;65:248–256. doi: 10.1128/iai.65.1.248-256.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pederson KJ, Vallis AJ, Aktories K, Frank DW, Barbieri JT. The amino-terminal domain of Pseudomonas aeruginosa ExoS disrupts actin filaments via small-molecular-weight GTP-binding proteins. Mol Microbiol. 1999;32:393–401. doi: 10.1046/j.1365-2958.1999.01359.x. [DOI] [PubMed] [Google Scholar]
- Pirnay JP, De Vos D, Cochez C, Bilocq F, Pirson J, Struelens M, et al. Molecular epidemiology of Pseudomonas aeruginosa colonization in a burn unit: persistence of a multidrug-resistant clone and a silver sulfadiazine-resistant clone. J Clin Microbiol. 2003;41:1192–1202. doi: 10.1128/JCM.41.3.1192-1202.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizarro-Cerda J, Tedin K. The bacterial signal molecule, ppGpp, regulates Salmonella virulence gene expression. Mol Microbiol. 2004;52:1827–1844. doi: 10.1111/j.1365-2958.2004.04122.x. [DOI] [PubMed] [Google Scholar]
- Rietsch A, Wolfgang MC, Mekalanos JJ. Effect of metabolic imbalance on expression of type III secretion genes in Pseudomonas aeruginosa. Infect Immun. 2004;72:1383–1390. doi: 10.1128/IAI.72.3.1383-1390.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rietsch A, Vallet-Gely I, Dove SL, Mekalanos JJ. ExsE, a secreted regulator of type III secretion genes in Pseudomonas aeruginosa. Proc Natl Acad Sci USA. 2005;102:8006–8011. doi: 10.1073/pnas.0503005102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy-Burman A, Savel RH, Racine S, Swanson BL, Revadigar NS, Fujimoto J, et al. Type III protein secretion is associated with death in lower respiratory and systemic Pseudomonas aeruginosa infections. J Infect Dis. 2001;183:1767–1774. doi: 10.1086/320737. [DOI] [PubMed] [Google Scholar]
- Sato H, Frank DW, Hillard CJ, Feix JB, Pankhaniya RR, Moriyama K, et al. The mechanism of action of the Pseudomonas aeruginosa-encoded type III cyto-toxin, ExoU. EMBOJ. 2003;22:2959–2969. doi: 10.1093/emboj/cdg290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawa T, Yahr TL, Ohara M, Kurahashi K, Gropper MA, Wiener-Kronish JP, Frank DW. Active and passive immunization with the Pseudomonas V antigen protects against type III intoxication and lung injury. Nat Med. 1999;5:392–398. doi: 10.1038/7391. [DOI] [PubMed] [Google Scholar]
- Sun J, Barbieri JT. Pseudomonas aeruginosa ExoT ADP-ribosylates CT10 regulator of kinase (Crk) proteins. J Biol Chem. 2003;278:32794–32800. doi: 10.1074/jbc.M304290200. [DOI] [PubMed] [Google Scholar]
- Sundin C, Wolfgang MC, Lory S, Forsberg A, Frithz-Lindsten E. Type IV pili are not specifically required for contact dependent translocation of exoen-zymes by Pseudomonas aeruginosa. Microb Pathog. 2002;33:265–277. doi: 10.1006/mpat.2002.0534. [DOI] [PubMed] [Google Scholar]
- Sundin C, Thelaus J, Broms JE, Forsberg K. Polarisation of type III translocation by Pseudomonas aeruginosa requires PcrG, PcrV and PopN. Microb Pathog. 2004;37:313–322. doi: 10.1016/j.micpath.2004.10.005. [DOI] [PubMed] [Google Scholar]
- Takeya K, Amako K. A rod-shaped Pseudomonas phage. Virology. 1966;28:163–165. doi: 10.1016/0042-6822(66)90317-5. [DOI] [PubMed] [Google Scholar]
- Urbanowski ML, Lykken GL, Yahr TL. A secreted regulatory protein couples transcription to the secretory activity of the Pseudomonas aeruginosa type III secretion system. Proc Natl Acad Sci USA. 2005;102:9930–9935. doi: 10.1073/pnas.0504405102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallis AJ, Yahr TL, Barbieri JT, Frank DW. Regulation of ExoS production and secretion by Pseudomonas aeruginosa in response to tissue culture conditions. Infect Immun. 1999;67:914–920. doi: 10.1128/iai.67.2.914-920.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velasco E, Thuler LC, Martins CA, Dias LM, Goncalves VM. Nosocomial infections in an oncology intensive care unit. Am J Infect Control. 1997;25:458–462. doi: 10.1016/s0196-6553(97)90067-5. [DOI] [PubMed] [Google Scholar]
- Waldor MK, Mekalanos JJ. Vibrio cholerae: molecular pathogenesis, immune response and vaccine development. In: Paradise LJ, editor. Enteric Infections and Immunity. New York: Plenum Press; 1996. pp. 37–55. [Google Scholar]
- Watarai M, Andrews HL, Isberg RR. Formation of a fibrous structure on the surface of Legionella pneumophila associated with exposure of DotH and DotO proteins after intracellular growth. Mol Microbiol. 2001;39:313–329. doi: 10.1046/j.1365-2958.2001.02193.x. [DOI] [PubMed] [Google Scholar]
- Wolfgang MC, Lee VT, Gilmore ME, Lory S. Coordinate regulation of bacterial virulence genes by a novel adenylate cyclase-dependent signaling pathway. Dev Cell. 2003;4:253–263. doi: 10.1016/s1534-5807(03)00019-4. [DOI] [PubMed] [Google Scholar]
- Yahr TL, Goranson J, Frank DW. Exoenzyme S of Pseudomonas aeruginosa is secreted by a type III pathway. Mol Microbiol. 1996;22:991–1003. doi: 10.1046/j.1365-2958.1996.01554.x. [DOI] [PubMed] [Google Scholar]
- Yahr TL, Mende-Mueller LM, Friese MB, Frank DW. Identification of type III secreted products of the Pseudomonas aeruginosa exoenzyme S regulon. J Bacteriol. 1997;179:7165–7168. doi: 10.1128/jb.179.22.7165-7168.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yahr TL, Vallis AJ, Hancock MK, Barbieri JT, Frank DW. ExoY, an adenylate cyclase secreted by the Pseudomonas aeruginosa type III system. Proc Natl Acad Sci USA. 1998;95:13899–13904. doi: 10.1073/pnas.95.23.13899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zolfaghar I, Angus AA, Kang PJ, To A, Evans DJ, Fleiszig SM. Mutation of retS, encoding a putative hybrid two-component regulatory protein in Pseudomonas aeruginosa, attenuates multiple virulence mechanisms. Microbes Infect. 2005;7:1305–1316. doi: 10.1016/j.micinf.2005.04.017. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The following supplementary material is available for this article online:
FACS analysis of a plasmid-based exoS promoter (A) or toluic acid promoter (B) driving GFP expression in P. aeruginosa strain PAK. The concentration of inducer NTA (A) or toluic acid (B) is given in mM in the right hand corner of each plot.
Shift experiments: PAK ΔexoS∷GFP-lacZ (RP955) was grown overnight in LB with either 5 mM NaCl (low) or 200 mM NaCl (high), diluted 1:200 into fresh media and grown at 37°C with aeration. After 2 h, the culture was split and diluted 1:1 into LB with or without 10 mM NTA. Cultures were shifted between media of different osmolarity by pelleting cells in a microcentrifuge, washing them once with the medium the cells were being shifted to and resuspending them in the new medium at the indicated times (T0, at the time of diluting the overnight, or T2 after 2 h of incubation, at the time of the shift to media with or without NTA). After 3 h of incubation the cultures were chilled on ice and GFP fluorescence was assayed by FACS analysis [black, calcium replete; grey, in the presence of chelator (NTA)].
PcrV production and localization in low- and highosmolarity LB. (A) Western blot of cell lysates of PAK ΔexoS∷GFP-lacZ(RP955) grown in LB with 5 mM NaCl (low) or 200 mM NaCl (high). Cells were grown to mid-log, pelleted and resuspended in SDS-sample buffer at an OD600 of 1. 10 µl of each lysate, and of a 1:4 and 1:16 dilution thereof, were separated by 10% SDS-PAGE and transferred to nitrocellulose. The blot was probed with a rabbit anti-PcrV antiserum and developed using a secondary antibody conjugated to HRP. After the first hybridization, the blot was stripped using a commercial stripping reagent (Pierce) and reprobed using a commercial mouse monoclonal-antibody directed against the α-subunit of RNA polymerase (Neoclone). (B) RP955 was grown in low- or high-osmolarity LB for 2 h at which point the culture was split and diluted 1:1 into LB of the same osmolarity with or without the chelator NTA. After 2 h of growth the cultures were chilled on ice and fractionated into cell pellet and supernatant fractions (supernatant proteins were precipitated using trichloroacetic acid at a final concentration of 10%). All fractions were subjected to SDS-PAGE, transferred to nitrocellulose and probed with an anti-PcrV antiserum.
This material is available as part of the online article from http://www.blackwell-synergy.com