Abstract
Three phenotypically distinct cell types are present in human neuroblastomas (NB) and NB cell lines: I-type stem cells, N-type neuroblastic precursors, and S-type Schwannian/melanoblastic precursors. The stimulation of human N-type neuroblastoma cell proliferation by normal human bone marrow monocytic cell conditioned medium (BMCM) has been demonstrated in vitro, a finding consistent with the high frequency of bone marrow (BM) metastases in patients with advanced NB. Inorganic arsenic trioxide (As2O3), already clinically approved for the treatment of several hematological malignancies, is currently under investigation for NB. Recent studies show that As2O3 induces apoptosis in NB cells. We examined the impact of BMCM on growth and survival of As2O3-treated NB cell lines, to evaluate the response of cultured NB cell variants to regulatory agents. We studied the effect of BMCM on survival and clonogenic growth of eleven As2O3-treated NB cell lines grown in sparsely seeded, non-adherent, semi-solid cultures. As2O3 had a strong inhibitory effect on survival of all tested NB cell lines. BMCM augmented cell growth and survival and reversed the inhibitory action of As2O3 in all tested cell lines, but most strongly in N-type cells. While As2O3 effectively reduced survival of all tested NB cell lines, BMCM effectively impacted its inhibitory action. Better understanding of micro-environmental regulators affecting human NB tumor cell growth and survival may be seminal to the development of therapeutic strategies and clinically effective agents for this condition.
Keywords: Neuroblastoma, Bone metastasis, Arsenic trioxide, Human bone marrow cell-derived factor
Introduction
Neuroblastoma (NB), the most common extra-cranial solid tumor of childhood, arises in the developing neural crest [1]. Although intensive chemotherapy and radiotherapy supported by bone marrow transplantation and retinoic acid treatment have improved survival, most advanced NB patients eventually develop progressive disease, refractory to continued therapy [2]. Consistent with their origin from multi-potent neural crest cells, tumors often consist of multiple cell phenotypes [3]. Examination of the cellular heterogeneity and malignant potential of human NB cell lines and clones supports the observation that phenotypes characterized in cell lines are present in tumors [4]. Studies of NB cell growth, differentiation, and malignancy have resulted in the identification of three distinct cellular phenotypes [4]: (1) I-type stem cells, (2) S-type substrate-adherent, Schwannian/melanoblastic precursors, and (3) N-type neuroblastic/neuroendocrine precursors. I-type stem cells are exquisitely anchorage-independent and highly efficient in the formation of colonies in semi-solid medium. As assessed by tumor formation in nude mice, and anchorage-independent growth in soft agar, I-type stem cells are significantly more malignant than either N- or S-type cells [4]. N- and S-type cells are anchorage-dependent: N cells form colonies with low efficiency, while S cells grow poorly, if at all, in non-adherent, semi-solid cultures [4].
Bone marrow (BM) is the most common site of metastasis in NB, occurring in about 70% of metastatic NB patients [5]. Clinically, this stage is often associated with refractory disease and fatal outcome. The relatively high frequency of BM metastases in patients with advanced NB suggests the presence of BM micro-environmental elements that are favorable to NB tumor cell growth. We have previously presented evidence of pronounced augmentation of N-type NB cell colony formation in semi-solid non-adherent cultures, elicited by conditioned media from normal human BM monocytic cell cultures [6]. More recently, inorganic arsenic trioxide (As2O3) has been clinically approved for the treatment of several hematologic malignancies [7–9] and is currently under investigation for NB [10]. Recent reports have conclusively shown that As2O3 exerts its action through apoptotic processes while studies with cell lines show that As2O3 has a similar apoptotic effect on NB cells to that on malignant hematopoietic cells [10–14].
On the basis of clinical studies showing that As2O3 induces remission with minimal toxicity in patients with BM-related malignant diseases (acute promyelocytic leukemia and multiple myeloma) by apoptotic mechanisms [7–9], we evaluated the impact of BMCM on growth and survival of As2O3-treated NB cell lines, representing each of the NB cell types described above.
Methods
Cells Five N-type (IMR 32, IMR 5, KELLY, SY5Y, SK-N-SH), three I-type (NUB 6, MC, LAN 5) and three S-type (SHEP, SK-N-LO, LS) well established human NB cell lines were maintained in RPMI 1640 medium containing 10% fetal bovine serum.
Preparation of BMCM BM samples were obtained by informed consent from normal healthy adult donors. Low-density cells were suspended in serum-free RPMI 1640 culture medium (Biological Industries, Bet Haemek, Israel), at 5 × 106 cells/ml and incubated for 96 h. Cell-free conditioned medium (CM) was obtained by centrifugation and filtration of the 96-h incubated culture medium and tested for activity.
To Measure Cell Viability Cell lines (2 × 104 cells/ml) were incubated for 24 h in non-adherent cultures with or without 0.6, 1.2, 2.5 and 5 μM As2O3 (Sigma) and/or 1.2, 2.5, 5, and 10% BMCM. Using XTT reagent (Biological Industries, Bet Haemek, Israel), survival was quantified by colorimetric measurement of mitochondria-mediated tetrazolium salt reduction, resulting in colored formazan compounds [15]. Disruption of the mitochondrial transmembrane potential is one of the early intracellular events to occur following induction of apoptosis. A decrease in cellular ability to reduce XTT, a yellow water-soluble tetrazolium salt, 4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate, is widely used for the colorimetric non-radioactive quantification of cellular viability. This tetrazolium salt is cleaved to formazan by the “succinate-tetrazolium reductase” system (EC1.3.99.1) which belongs to the respiratory chain of the mitochondria and is active only in metabolically active cells. The rate of tetrazolium reduction is proportional to the extent of cell viability [15].The results (mean ± SD) of triplicate cultures represent absorbance at 450 nm after 5 h incubation with XTT.
To Determine Colony Formation As previously described [6], cells were seeded at 500 cells/ml in semi-solid non-adherent cultures with or without As2O3 and/or BMCM at the above concentrations. Colonies were counted after 8–10 days of incubation. All determinations were done in triplicate and are presented as mean ± SD.
Results
BMCM Inhibits As2O3-induced Cell Death
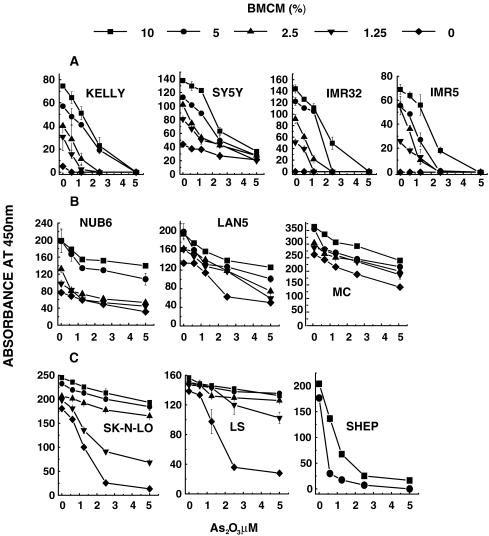
In agreement with recent reports, we observed that in the micro-molar range As2O3 had a concentration-dependent cytotoxic effect on the 11 cell lines representing all three NB cell types. To examine the impact of BMCM on As2O3-induced cell death, non-adherent cultures of three I-type (NUB 6, MC, LAN 5), three S-type (SHEP, SK-N-LO, LS) and five N-type (IMR 32, IMR 5, KELLY, SY5Y, SK-N-SH) NB cell lines were incubated for 24 h with increasing concentrations of both BMCM and As2O3. Viability was strongly reduced in all five of the anchorage-dependent N-type cell lines in non-adherent cultures in the absence of both As2O3 and BMCM. Fig. 1a shows the results of four out of five cell lines (SK-N-SH not shown). Addition of increasing concentrations of BMCM, without As2O3, to the culture medium resulted in significantly increased cell viability. As2O3 at 2.5 μM considerably reduced viability in all five cell lines treated with a low 2.5% concentration of BMCM, while higher BMCM concentrations (5% and 10%) considerably reduced or abrogated this cytotoxic action of As2O3 at 2.5 μM (Fig. 1a). As2O3 at 5 μM, however, was cytotoxic to the same extent regardless of BMCM.
Fig. 1.
Dose-dependent inhibition of As2O3-induced cell death by BMCM (at concentrations indicated in the box) in non-adherent cultures of: a Anchorage-dependent N-type cells. b Anchorage-independent I-type cells. c Anchorage-dependent S-type cells. Viability was quantified, using the XTT reagent by colorimetric measurement of mitochondria-mediated tetrazolium salt reduction, resulting in colored formazan compounds. The results (mean ± SD) of triplicate cultures represent absorbance at 450 nm after 5 h incubation with XTT
Consistent with their anchorage-independence, the I-type cell lines survived well in non-adherent cultures in the absence of BMCM (Fig. 1b). In contrast to N-type cells, BMCM increased cell survival only very slightly, and As2O3-induced cytotoxicity was also weaker than in the other cell types. None the less, similarly to the other cell types, BMCM reduced the cytotoxic activity of As2O3 in I-type cells (Fig. 1b).
Despite their anchorage dependence, untreated S-type cells survived in non-adherent cultures while a concentration of 2.5 μM As2O3 was sufficient to reduce viability in all three S-type cell lines. Increasing concentrations of BMCM, however, effectively reversed this As2O3-induced cytotoxic action (Fig. 1c).
BMCM Reverses As2O3-induced Inhibition of Colony Formation
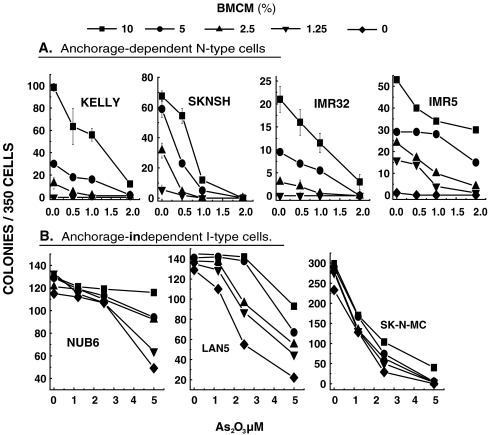
As we have previously reported [6], anchorage-dependent N-type cell lines failed to form colonies in sparsely seeded semi-solid cultures without BMCM (Fig. 2a). Consistent with its cytotoxic activity, As2O3 caused considerable dose-dependent inhibition of BMCM-induced NB cell colony formation by all five of the anchorage-dependent N-type cell lines. Figure 2a shows the results of four out of five N-type cell lines (results of SY5Y not shown). At increasing concentrations of BMCM, higher concentrations of As2O3 were required to successfully inhibit cell growth of all five cell lines tested.
Fig. 2.
BMCM reverses As2O3-induced inhibition of colony formation. Cells were seeded at 500 cells/ml in semi-solid non-adherent cultures ± As2O3 and/or BMCM at the above concentrations. Colonies were counted after 8–10 days of incubation. All determinations were done in triplicate and are presented as mean ± SD
Figure 2b demonstrates the reversal of As2O3 inhibitory action on anchorage-independent I-type cell colony formation. In contrast to N-type cells, these cells form colonies very efficiently in semi-solid medium in the absence of BMCM. In the presence of increasing concentrations of BMCM, however, As2O3-induced inhibitory action is considerably reversed.
Discussion
Malignant cells from solid tumors often metastasize to the bone marrow (BM). In patients with neuroblastoma, metastatic disease in the BM is observed more frequently than at any other site; indeed, more than 70% of patients with NB have BM metastases at diagnosis of stage IV disease, with a significantly poorer prognosis [5]. Our previous report provides evidence that non-stimulated low-density BM cells release activity which strongly augments proliferation of N-type cell lines derived from human NB tumors [6]. This finding is consistent with the relatively high frequency of BM metastases in patients with advanced NB. Cells from various types of tumors, similarly to N-type NB cells, are anchorage-dependent. By detachment from the primary tumor they are deprived of vital adhesion signals. Denial of their extra-cellular matrix causes these cells to undergo programmed cell death [16]. This detachment-induced apoptosis may provide protection against the metastatic spread of tumor cells. To mimic such detachment from the matrix in vitro, we used culture conditions under which cells could not attach themselves to a surface or to each other. In the case of N-type NB cells, this resulted in cell death within 24 h. Our study provides evidence that the presence of BMCM in non-adherent sparsely seeded cultures increases viability of anchorage-dependent N-type cells and thereby enables them to form colonies. In contrast, anchorage-independent I-type NB cells, which were unaffected by the absence of adhesion, survived and formed similar numbers of colonies regardless of whether BMCM was present or absent. Treatment with As2O3 dose-dependently reduced viability and inhibited growth of all the I-type cell lines, and of all of the BMCM-treated N-type cell lines. Increasing the concentration of BMCM in these cultures, however, resulted in the reversal of the inhibitory action of As2O3.
N and I-type cells, similarly to neuronal cells, produce neurotransmitters and grow as aggregates of small round cells with neuritic processes, In contrast, S-type cells are devoid of neurotransmitter synthesis and are larger, flattened and substrate-adherent, resembling epithelial or fibroblast cells. Previous studies have shown that growth arrest induced by suspension of certain fibroblasts does not result in apoptosis and indeed may be reversible [17]. We have previously shown that, in contrast to anchorage-dependent N-type cells and similarly to fibroblasts, colony formation by S-type cells was not induced by BMCM [18]. Despite these discrepancies, As2O3 effectively reduced the survival of all three S-type cell lines while increasing concentrations of BMCM resulted in the reversal of As2O3 activity (Fig. 1c).
Preparations of BMCM are presently being subjected to procedures for the isolation of the molecular mediator of the activity described above. Isolation and identification of the agent(s) responsible for this potent regulatory activity in BM CM may be useful for devising strategies for the detection of responsive NB primary tumor cells and may be crucial for the development of more effective therapeutic strategies for this often fatal disease.
Acknowledgement
We would like to thank Mrs. Lilly Schaechter and Ms. Einat Budowski for editorial help in the preparation of this manuscript.
Footnotes
This study has been presented in part at the 4th International Conference on Tumor Microenvironment (March 6–10, 2007), Florence Italy.
References
- 1.Castleberry RP (1997) Biology and treatment of neuroblastoma. Pediatr Clin North Am 44:919–937 [DOI] [PubMed]
- 2.Matthay KK, Villablanca JG, Seeger RC et al (1999) Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. Children’s Cancer Group. N Engl J Med 341:1165–1173 [DOI] [PubMed]
- 3.Lastowska M, Cullinane C, Variend S et al (2001) Comprehensive genetic and histopathological study reveals three types of human neuroblastoma tumors. J Clin Oncol 19:3080–3090 [DOI] [PubMed]
- 4.Walton JD, Kattan DR, Thomas SK et al (2004) Characteristics of stem cells from human neuroblastoma cell lines and in tumors. Neoplasia 6:838–845 [DOI] [PMC free article] [PubMed]
- 5.DuBois SG, Kalika Y, Lukens JN et al (1999) Metastatic sites in stage IV and IVS neuroblastoma correlate with age, tumor biology, and survival. J Pediatr Hematol Oncol 21:181–189 [DOI] [PubMed]
- 6.Hahn T, Or R, Segall H et al (1998) Human bone marrow-derived mitogenic stimulation selective for breast carcinoma and neuroblastoma cells. Int J Cancer 78:624–628 [DOI] [PubMed]
- 7.Gazitt Y, Akay C (2005) Arsenic trioxide: an anti cancer missile with multiple warheads. Hematology 10(3):205–213 [DOI] [PubMed]
- 8.Zhu XH, Shen YL, Jing Y et al (1999) Apoptosis and growth inhibition in malignant lymphocytes after treatment with arsenic trioxide at clinically achievable concentrations. J Natl Cancer Inst 91:772–778 [DOI] [PubMed]
- 9.Gesundheit B, Shapira MY, Ackerstein A et al (2006) A patient with progressive multiple myeloma treated successfully with arsenic trioxide after allogeneic bone marrow transplantation. Acta Haematol 117:119–121 [DOI] [PubMed]
- 10.Pettersson HM, Karlsson J, Pietras A et al (2007) Arsenic trioxide and neuroblastoma cytotoxicity. J Bioenerg Biomembr 39:35–41 [DOI] [PubMed]
- 11.Karlsson J, Edsjö A, Påhlman S et al (2005) Multi-drug-resistant neuroblastoma cells are responsive to arsenic trioxide at both normoxia and hypoxia. Mol Cancer Ther 4:1128–1135 [DOI] [PubMed]
- 12.Karlsson J, ØRa I, Pörn-Ares I et al (2004) Arsenic trioxide-induced death of neuroblastoma cells involves activation of bax and does not require p53. Clin Cancer Res 10:3179–3188 [DOI] [PubMed]
- 13.Akao Y, Nakagawa Y, Akiyama K (1999) Arsenic trioxide induces apoptosis in neuroblastoma cell lines through the activation of caspase-3 in vitro. FEBS Lett 455:59–62 [DOI] [PubMed]
- 14.Øra I, Bondesson L, Jonsson C et al (2000) Arsenic trioxide inhibits neuroblastoma growth in vivo and promotes apoptotic cell death in vitro. Biochem Biophys Res Commun 277:179–185 [DOI] [PubMed]
- 15.Roehm NW, Rodgers GH, Hatfield SM, Glasebrook AL (1991) An improved colorimetric assay for cell proliferation and viability utilizing the tetrazolium salt XTT. J Immunol Methods 142:257–265 [DOI] [PubMed]
- 16.Frisch SM, Francis H (1994) Disruption of epithelial cell–matrix interactions induces apoptosis. J Cell Biol 124:619–626 [DOI] [PMC free article] [PubMed]
- 17.Ben-Ze’ev A, Farmer SR, Penman S (1980) Protein synthesis requires cell-surface contact while nuclear events respond to cell shape in anchorage-dependent fibroblasts. Cell 21:365–372 [DOI] [PubMed]
- 18.Kuci Z, Bruchelt G, Seitz G, Karov Y, Or R, Handgretinger R, Niethammer D, Hahn T (2002) Bone marrow derived factors support growth of N-type but not of melanocytic neuroblastoma cells. Anticancer Res 22:4325–4329 [PubMed]