Abstract
Free-living amoebae represent a potential reservoir and predator of Salmonella enterica. Through the use of type III secretion system (T3SS) mutants and analysis of transcription of selected T3SS genes, we demonstrated that the Salmonella pathogenicity island 2 is highly induced during S. enterica serovar Typhimurium infection of Acanthamoeba polyphaga and is essential for survival within amoebae.
The importance of free-living protozoa, such as amoebae, as environmental reservoirs of food-borne pathogens is becoming increasingly recognized (1, 6, 9, 18). Such interactions may also have highly significant physiological implications, as amoebic passage of Legionella pneumophila enhanced bacterial virulence (3) and could resuscitate viable but nonculturable cells (20). Salmonella enterica serovars Typhimurium and Dublin have been shown to survive within Acanthamoeba polyphaga and Acanthamoeba rhysodes (5, 22), and induction of fis transcription, indicative of bacterial proliferation within contractile vacuoles, has been reported (3). The role of the Salmonella pathogenicity island 1 (SPI-1)-encoded type III secretion system (T3SS-1), which mediates forced bacterial uptake via subversion of actin dynamics, is unclear; however, an S. enterica serovar Dublin ΔhilA mutant lacking a key transcriptional activator of SPI-1 entered and survived within A. rhysodes at a level similar to that of the parent strain (22). A second type III secretion system encoded by SPI-2 (T3SS-2) and the PhoPQ two-component regulatory system are known to play key roles in the intracellular survival of Salmonella in mammalian cells, but their roles in interactions with protozoa have not been reported. In this study we investigated the roles of T3SS-1, T3SS-2, and PhoP in entry and survival of S. enterica serovar Typhimurium in A. polyphaga using defined mutant strains. Additionally, we quantified transcription of the S. enterica serovar Typhimurium SPI-1 gene sipC and the SPI-2 gene sseC (which encode components of the type III secretion translocon) within A. polyphaga.
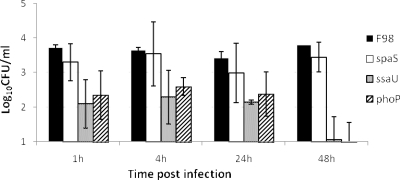
A gentamicin protection assay modified from that used to quantify Salmonella invasion of avian cells was used to assess uptake and intracellular survival in Acanthamoeba polyphaga Linc-1 grown in axenic culture in peptone-yeast extract-glucose medium (PYG) (1, 2). Amoebae were grown to 5 × 106 cells per ml initially in flasks and then in 24-well plates. S. enterica serovar Typhimurium strain F98 and previously described variants with mutations in the SPI-1 gene spaS and in the SPI-2 gene ssaU (genes well characterized for changes in their phenotype for virulence, cell invasion, and survival within macrophages [10, 11, 23]) and in phoP (13) were grown at 37°C in Luria-Bertani (LB) broth with shaking at 150 rpm to late logarithmic phase and added to amoebae at a multiplicity of infection (MOI) of 10. After 1 h of incubation at 37°C, the plates were centrifuged at 50 × g for 5 min. The medium was removed from the plates and replaced with PYG containing gentamicin sulfate at 100 μg ml−1. The plates were incubated at 37°C for 30 min and then centrifuged again before the medium was replaced with PYG containing 20 μg ml−1 gentamicin to inhibit growth of any Salmonella released from lysed amoebae. After 1, 4, 24, or 48 h of incubation, the medium was removed and amoebae were lysed by the addition of distilled water, and then serial dilutions of the lysates were plated onto nutrient agar to enumerate Salmonella bacteria after 24 h of incubation at 37°C. Following challenge, between 103 and 104 CFU of S. enterica serovar Typhimurium F98 per ml of cultured amoebae were recovered from amoebic cells (Fig. 1). Both the parent and ΔspaS mutant were recovered at similar levels from 1 h postinfection to 48 h postinfection. In contrast, recoveries of the ΔssaU mutant and ΔphoP mutant were significantly lower than recovery of the parent strain (P < 0.01) at each time point. There was also a decline in the number of viable bacteria recovered over time. The viability of amoebic cultures was slightly reduced following Salmonella infection, which may contribute to the gradual decline in bacterial numbers recovered over time. Counts of viable amoebae by microscopy and measurement of cytotoxicity by use of a commercial assay (Cytotox96; Promega, Southampton, Hampshire, United Kingdom) showed between 47 and 57% cell death in Salmonella-infected cells at 48 h postinfection, whereas uninfected controls showed 36% cell death. Differences in amoebic death between mutant strains were minimal and would not account for the differences in bacterial survival.
FIG. 1.
Survival of S. enterica serovar Typhimurium F98 parent and SPI-1 (spaS), SPI-2 (ssaU), and phoP mutant strains within A. polyphaga in axenic culture. The values are the mean numbers of Salmonella recovered ± standard errors of the means (error bars) from six replicate experiments. Salmonella was added at an MOI of 10 in these experiments.
To determine intra-amoebic transcription of the T3SS genes sseC and sipC and the housekeeping gene yejA as an internal standard, invasion assays were performed in 20-ml flasks as described above. The sseC and sipC genes were selected as representatives of the SPI-2 and SPI-1 T3SS, respectively, as the gene products are expressed within the host cell as part of the translocon stabilizing the needle-like apparatus and we have previously demonstrated their expression within porcine and murine macrophages (14). After 1 or 4 h of incubation, flasks were centrifuged to obtain a cell pellet from which total RNA was extracted with TRI reagent and treated in solution with Turbo DNase (Ambion, Inc., Austin, TX), followed by on-column DNase treatment with RNase-free DNase. Transcription at time zero was determined in Salmonella grown in LB broth as described above. Real-time reverse transcriptase PCR (RT-PCR) was performed with the MJ Research/Bio-Rad Opticon 2 system with quantitative RT-PCR Mastermix (Eurogentec, Seraing, Belgium) using previously described conditions (14). To quantify transcription, the 2ΔΔCt method was used for data analysis (12), and transcription was reported as n-fold induction normalized to the internal standard and relative to the control at time zero (14). RT-PCR data were analyzed by Student's t test with a P value of >0.05 considered statistically significant.
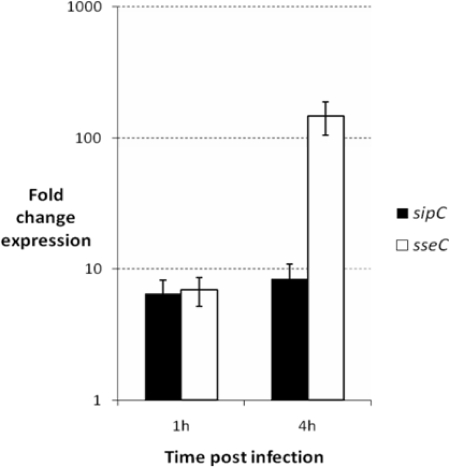
Significant induction of both the SPI-1 gene sipC and the SPI-2 gene sseC was found to occur relative to yejA within amoebic cells at 1 h postinfection (Fig. 2), with an approximately sixfold increase over Salmonella grown in LB broth (P > 0.02). At 4 h postinfection, there was no significant change in transcription of sipC. In contrast, a dramatic increase in transcription of the SPI-2 gene sseC was found at 4 h. Transcription within amoebic cells was increased over Salmonella grown in LB broth by a mean value of 147-fold (P = 0.001). Previously we have shown sseC to be expressed by S. enterica serovar Typhimurium within porcine and murine macrophages at 4 h postinfection (14). A range of SPI-2 genes are also strongly induced following S. enterica serovar Typhimurium infection of J774A.1 murine macrophage-like cells (4) and epithelial cells (8).
FIG. 2.
Relative transcription of SPI-1 (sipC) and SPI-2 (sseC) genes of S. enterica serovar Typhimurium F98 within amoebic cells 1 and 4 h postinfection. Salmonella was added to the amoebae at an MOI of 10. Changes in expression were determined by real-time quantitative RT-PCR compared to transcription of bacteria grown in LB broth and using the housekeeping gene yejA as an internal standard. Data shown are the mean values for the change in expression ± standard errors of the means (error bars) from eight replicate experiments.
Taken together, these data indicate that the SPI-2-encoded T3SS influences survival within amoebic cells, as is the case with macrophages. Previously we have shown that mutation of ssaU reduces the ability of a range of Salmonella serovars to survive within phagocytes and cause systemic infection (10, 11, 23). The failure of the ΔphoP mutant to survive in A. polyphaga cells further supports the theory that survival within amoebae and survival within macrophages are largely analogous, as the PhoPQ two-component regulatory system, although associated with regulation of many genes, is key to activation of the SPI-2-encoded T3SS within phagocytes (7, 15). In contrast, mutation of spaS had a minor effect on Salmonella survival within amoebae, consistent with findings using a ΔhilA mutant (17). Nevertheless, transcription of the SPI-1 gene sipC was induced within amoebae, in contrast to findings with S. enterica serovar Typhimurium in J774A.1 murine macrophage-like cells, where sipC transcription was downregulated 50-fold compared to growth in culture medium (4). Recently, however, SPI-1 genes were found to be induced after infection of epithelial cells, indicating that the intracellular program of bacterial gene expression is sensitive to cell type (8).
Survival within protozoa may represent an important environmental reservoir of Salmonella and confer resistance to predation in the gastrointestinal tracts of ruminants. In addition, such interactions may have exerted an evolutionary pressure leading to bacterial divergence, including variation in the lipopolysaccharide O-side chain of Salmonella and the development of virulence factors (16, 24, 25). Recent studies have indicated that Shiga toxin (Stx) of the food-borne pathogen enterohemorrhagic Escherichia coli (EHEC) aids resistance to grazing protozoa and may account for the very high stx carriage rates in EHEC isolated from ruminants (19). Furthermore, the type II and IV protein secretion systems of L. pneumophila aid survival in both waterborne amoebae and alveolar macrophages during human infection (17, 21). Taken together with our finding that S. enterica serovar Typhimurium SPI-2 is induced and required for survival in A. polyphaga, such data suggest that traits we primarily consider virulence factors for bacterial pathogenesis in animals and humans may have originally evolved to play other roles in microbial ecology.
Acknowledgments
B.B. was funded by a Society for Applied Microbiology “Students into Work” grant. P.J.L. was supported by the “Veterinary Research Training Initiative” funded by the Department of Environment Food and Rural Affairs and the Higher Education Funding Council for England.
Footnotes
Published ahead of print on 23 January 2009.
REFERENCES
- 1.Axelsson-Olsson, D., J. Waldenstrom, T. Broman, B. Olsen, and M. Holmberg. 2005. Protozoan Acanthamoeba polyphaga as a potential reservoir for Campylobacter jejuni. Appl. Environ. Microbiol. 71:987-992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Birtles, R. J., T. J. Rowbotham, R. Michel, D. G. Pitcher, B. Lascola, S. Alexiou-Daniel, and D. Raoult. 2000. ‘Candidatus Odyssella thessalonicensis’ gen. nov., sp. nov., an obligate intracellular parasite of Acanthamoeba species. Int. J. Syst. Evol. Microbiol. 50:63-72. [DOI] [PubMed] [Google Scholar]
- 3.Cirillo, J. D., S. L. Cirillo, L. Yan, L. E. Bermudez, S. Falkow, and L. S. Tompkins. 1999. Intracellular growth in Acanthamoeba castellanii affects monocyte entry mechanisms and enhances virulence of Legionella pneumophila. Infect. Immun. 67:4427-4434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eriksson, S., S. Lucchini, A. Thompson, M. Rhen, and J. C. Hinton. 2003. Unravelling the biology of macrophage infection by gene expression profiling of intracellular Salmonella enterica. Mol. Microbiol. 47:103-118. [DOI] [PubMed] [Google Scholar]
- 5.Gaze, W. H., N. Burroughs, M. P. Gallagher, and E. M. Wellington. 2003. Interactions between Salmonella typhimurium and Acanthamoeba polyphaga, and observation of a new mode of intracellular growth within contractile vacuoles. Microb. Ecol. 46:358-369. [DOI] [PubMed] [Google Scholar]
- 6.Gourabathini, P., M. T. Brandl, K. S. Redding, J. H. Gunderson, and S. G. Berk. 2008. Interactions between food-borne pathogens and protozoa isolated from lettuce and spinach. Appl. Environ. Microbiol. 74:2518-2525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Groisman, E. A. 2001. The pleiotropic two-component regulatory system PhoP-PhoQ. J. Bacteriol. 183:1835-1842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hautefort, I., A. Thompson, S. Eriksson-Ygberg, M. L. Parker, S. Lucchini, V. Danino, R. J. Bongaerts, N. Ahmad, M. Rhen, and J. C. Hinton. 2008. During infection of epithelial cells Salmonella enterica serovar Typhimurium undergoes a time-dependent transcriptional adaptation that results in simultaneous expression of three type 3 secretion systems. Cell. Microbiol. 10:958-984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huws, S. A., R. J. Morley, M. V. Jones, M. R. Brown, and A. W. Smith. 2008. Interactions of some common pathogenic bacteria with Acanthamoeba polyphaga. FEMS Microbiol. Lett. 282:258-265. [DOI] [PubMed] [Google Scholar]
- 10.Jones, M. A., S. D. Hulme, P. A. Barrow, and P. Wigley. 2007. The Salmonella pathogenicity island 1 and Salmonella pathogenicity island 2 type III secretion systems play a major role in pathogenesis of systemic disease and gastrointestinal tract colonization of Salmonella enterica serovar Typhimurium in the chicken. Avian Pathol. 36:199-203. [DOI] [PubMed] [Google Scholar]
- 11.Jones, M. A., P. Wigley, K. L. Page, S. D. Hulme, and P. A. Barrow. 2001. Salmonella enterica serovar Gallinarum requires the Salmonella pathogenicity island 2 type III secretion system but not the Salmonella pathogenicity island 1 type III secretion system for virulence in chickens. Infect. Immun. 69:5471-5476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Livak, K. J., and T. D. Schmittgen. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402-408. [DOI] [PubMed] [Google Scholar]
- 13.Methner, U., P. A. Barrow, D. Gregorova, and I. Rychlik. 2004. Intestinal colonisation-inhibition and virulence of Salmonella phoP, rpoS and ompC deletion mutants in chickens. Vet. Microbiol. 98:37-43. [DOI] [PubMed] [Google Scholar]
- 14.Paulin, S. M., A. Jagannathan, J. Campbell, T. S. Wallis, and M. P. Stevens. 2007. Net replication of Salmonella enterica serovars Typhimurium and Choleraesuis in porcine intestinal mucosa and nodes is associated with their differential virulence. Infect. Immun. 75:3950-3960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Prost, L. R., M. E. Daley, M. W. Bader, R. E. Klevit, and S. I. Miller. 2008. The PhoQ histidine kinases of Salmonella and Pseudomonas spp. are structurally and functionally different: evidence that pH and antimicrobial peptide sensing contribute to mammalian pathogenesis. Mol. Microbiol. 69:503-519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rasmussen, M. A., S. A. Carlson, S. K. Franklin, Z. P. McCuddin, M. T. Wu, and V. K. Sharma. 2005. Exposure to rumen protozoa leads to enhancement of pathogenicity of and invasion by multiple-antibiotic-resistant Salmonella enterica bearing SGI1. Infect. Immun. 73:4668-4675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shin, S., and C. R. Roy. 2008. Host cell processes that influence the intracellular survival of Legionella pneumophila. Cell. Microbiol. 10:1209-1220. [DOI] [PubMed] [Google Scholar]
- 18.Snelling, W. J., J. E. Moore, J. P. McKenna, D. M. Lecky, and J. S. Dooley. 2006. Bacterial-protozoa interactions: an update on the role these phenomena play towards human illness. Microbes Infect. 8:578-587. [DOI] [PubMed] [Google Scholar]
- 19.Steinberg, K. M., and B. R. Levin. 2007. Grazing protozoa and the evolution of the Escherichia coli O157:H7 Shiga toxin-encoding prophage. Proc. Biol. Sci. 274:1921-1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Steinert, M., L. Emody, R. Amann, and J. Hacker. 1997. Resuscitation of viable but nonculturable Legionella pneumophila Philadelphia JR32 by Acanthamoeba castellanii. Appl. Environ. Microbiol. 63:2047-2053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Swanson, M. S., and B. K. Hammer. 2000. Legionella pneumophila pathogenesis: a fateful journey from amoebae to macrophages. Annu. Rev. Microbiol. 54:567-613. [DOI] [PubMed] [Google Scholar]
- 22.Tezcan-Merdol, D., M. Ljungstrom, J. Winiecka-Krusnell, E. Linder, L. Engstrand, and M. Rhen. 2004. Uptake and replication of Salmonella enterica in Acanthamoeba rhysodes. Appl. Environ. Microbiol. 70:3706-3714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wigley, P., M. A. Jones, and P. A. Barrow. 2002. Salmonella enterica serovar Pullorum requires the Salmonella pathogenicity island 2 type III secretion system for virulence and carriage in the chicken. Avian Pathol. 31:501-506. [DOI] [PubMed] [Google Scholar]
- 24.Wildschutte, H., and J. G. Lawrence. 2007. Differential Salmonella survival against communities of intestinal amoebae. Microbiology 153:1781-1789. [DOI] [PubMed] [Google Scholar]
- 25.Wildschutte, H., D. M. Wolfe, A. Tamewitz, and J. G. Lawrence. 2004. Protozoan predation, diversifying selection, and the evolution of antigenic diversity in Salmonella. Proc. Natl. Acad. Sci. USA 101:10644-10649. [DOI] [PMC free article] [PubMed] [Google Scholar]