Abstract
Purpose
We have evaluated the effect of internal and external osteosynthesis devices on the efficacy of vancomycin treatment in a rabbit model of methicillin-resistant Staphylococcus aureus (MRSA)-induced post-traumatic osteomyelitis.
Methods
Double tibial osteotomies in female New Zealand rabbits were performed, inoculated with a MRSA strain, then fixed with an intramedullary rod. A debridement was performed 4 days later for each rabbit, and a bacterial count in pus was determined (B1). In the first group (G1), the osteosynthesis material was removed and replaced by a new sterile nail. In the second group (G2), the intramedullary rod was removed and then replaced by an external fixator. Immediately after surgery, G1 and G2 rabbits were treated with vancomycin (60 mg/kg twice a day). The animals were sacrificed at the end of a 5-day period, and a bacterial count in pus was performed again (B2).
Results
The difference of log10 colony forming units per milliliter (CFU/ml) (B2−B1) was −1.2 ± 0.5 and −2.9 ± 1.1, respectively, for G1 and G2.
Conclusion
The efficacy of vancomycin treatment increased after removal of the internal osteosynthesis device.
Keywords: Orthopedic device, Osteomyelitis, Trauma
Introduction
The pathogenesis of foreign body infection has been well known since Zimmerli’s work [1, 2]. He showed the presence of local granulocyte defect around the foreign body, explaining subsequent tendency to infection. Septic post-traumatic osteomyelitis is a difficult-to-treat complication associated with reconstructive surgery and joint replacement. Vital prognosis can be engaged and limb preservation is not always possible. Post-traumatic osteomyelitis requires lengthy treatment, and therapeutic failures are not rare. Removal of the osteosynthesis device is sometimes recommended [3]. However, some authors have reported clinical success in the treatment of post-traumatic osteomyelitis and prosthetic joint infection without removal of the orthopedic implant [4]. Others have reported clinical success in the treatment of early acute osteomyelitis after surgical debridement without the removal of the osteosynthesis device. Immediate antimicrobial chemotherapy and removal of the infected device are the preferred treatment methods for late-stage osteomyelitis.
Animal models were especially developed to study pathogenesis of bone infection or to evaluate antimicrobial agents [5, 1, 2], but not one was developed to evaluate a surgical procedure used to treat a post-traumatic osteomyelitis.
The aim of this experimental study was to compare two surgical procedures (external fixation and internal fixation after removal of internal osteosynthesis) on vancomycin efficacy in a rabbit model of methicillin-resistant Staphylococcus aureus-induced post-traumatic osteomyelitis.
Materials and methods
Microorganism
One clinical strain of methicillin-resistant Staphylococcus aureus (MRSA) isolated from blood culture was studied.
Medium
Mueller Hinton (MH) broth (Sanofi Diagnostics Pasteur, Marne la Coquette, France) supplemented with calcium (25 mg/l) and magnesium (12.5 mg/l) was used for susceptibility tests and killing curve experiments. Colony counts were determined on trypticase soy agar (TSA) (Difco) for the time-kill experiments.
In vitro studies
The minimal inhibitory concentration (MIC) of vancomycin for the MRSA strain was determined in cation-supplemented MH broth using the microdilution technique. Time-kill experiments were performed in glass flasks containing MH broth with an initial inoculum of 5 × 106 CFU/ml in the presence of vancomycin at 8× MIC. One flask without antibiotics served as a control. Surviving bacteria were counted after 0, 3, 6 and 24 h of incubation at 37°C, by subculturing 50-μl samples (in 0.9% NaCl) on TSA. A bactericidal effect was defined as a 3 log10 CFU/ml or greater decrease compared with the initial inoculum after 24 h of incubation.
Experimental post-traumatic osteomyelitis model
All the animal studies were carried out in accordance with the scientific procedures act (animals) 1986 and the code of practice for the housing and care of animals used in scientific procedures 1989.
Day 0: under general anesthesia, using an anteromedial approach, a bifocal tibial osteotomy was performed on a female New Zealand rabbit. The intermediate tibial fragment was infused in a 108 CFU/ml MRSA strain agar and then replaced in the rabbit’s leg. The double osteotomy was stabilized with an intramedullar nail (Kirshner’s wire 15/10) (Fig. 1a, b).
Fig. 1.
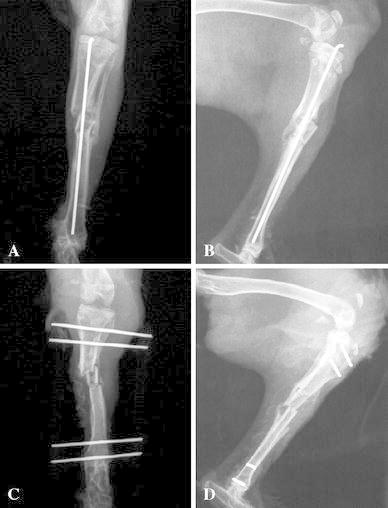
a Frontal radiogram of theright leg. The double tibial osteotomy was stabilized with an intramedullar rod.b Lateral radiogram of theright leg. The double tibial osteotomy was stabilized with an intramedullar rod.c Frontal radiogram of theright leg. The double tibial osteotomy was stabilized with a bilateral external fixator.d Lateral radiogram of theright leg. The double tibial osteotomy was stabilized with a bilateral external fixator
Day 4: an extensive surgical debridement (excluding resection of the intermediate tibial fragment) of the osteomyelitis was performed and the intramedullar nail was removed. Samples were collected, weighed, and suspended in 200 μl sterile saline. Serial dilutions (10−2, 10−4, 10−6) were made and plated on TSA, using a spiral plater (Spiral System; Interscience, Saint-Nom-La-Bretèche, France) to calculate the bacterial count (B1) in pus at the beginning of treatment. Animals were randomly assigned to one of two treatment groups. Group 1 (five rabbits): after debridement and surgical wash, a new internal osteosynthesis was performed with a sterile nail (Kirshner’s wire 15/10) and skin closure; group 2 (five rabbits): after debridement and surgical wash, skin closure was performed and a new osteosynthesis was performed using a homemade bilateral external fixator (Fig. 1c, d; 2). Vancomycin was administered intramuscularly twice daily (60 mg/kg b.i.d.) to all rabbits (groups 1 and 2) until day 8.
Fig. 2.
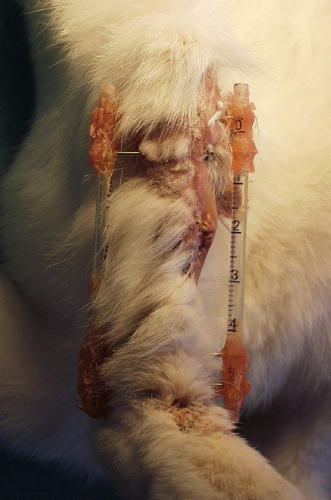
Frontal view of theright leg with homemade external fixator
Day 8: animals were sacrificed by means of a lethal thiopental injection. Samples were collected, weighed, and suspended in 200 μl sterile saline. Serial dilutions (10−2, 10−4, 10−6) were made and plated on TSA, using a spiral plater to calculate the bacterial count (B2) in pus at the end of treatment. A Δlog CFU/mg = B2−B1 expressing the decrease in bacterial count between day 4 and day 8 was calculated.
Pharmacokinetic studies
Vancomycin was administered intramuscularly to animals b.i.d. (60 mg/kg). Samples were collected at the end of the treatment. Assays of serum were performed to determine peak and trough levels of vancomycin.
Statistical analysis
Statistical analysis was performed using StatView software (Abacus Concepts, Berkeley, CA, USA). Analysis of variance (ANOVA) was used to compare the effects among the different groups, followed by Bonferroni’s test to compare the two groups. AP value ≤ 0.05 was considered significant.
Results
Susceptibility test
The minimum inhibitory concentration (MIC) of vancomycin for the S. aureus strain was 1 mg/l.
Time-kill curves
A decrease of 2 log10 CFU/ml was observed after 24 h with vancomycin at 8× MIC (Fig. 3).
Fig. 3.
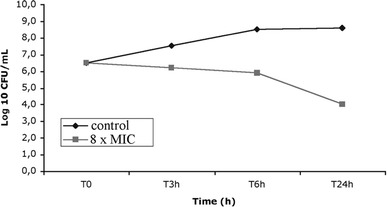
Time-kill curve of vancomycin
Pharmacokinetic study
Peak and trough vancomycin concentrations were 46.5 ± 7.4 and 14.8 ± 3.5 mg/l, respectively.
Experimental post-traumatic osteomyelitis
At day 4, the bacterial counts (B1) in pus were 6.3 ± 0.8 log10 CFU/mg and 6.8 ± 0.9 log10 CFU/mg in groups 1 and 2, respectively; there was no difference between the two groups (P = 0.465, B1 group 1 vs B1 group 2; Bonferroni’s test after ANOVA) (Table 1). None of the rabbits was cured after 4 days of antibiotic treatment with or without the removal of the internal osteosynthesis device. At day 8, the bacterial counts in pus were 5.2 ± 0.4 log10 CFU/mg and 3.9 ± 1.7 log10 CFU/mg in groups 1 and 2, respectively. On this occasion, there was a significant decrease in bacterial count between these groups (Δlog = B2−B1) (P = 0.0123, Δlog group 1 vs Δlog group 2, Bonferroni’s test after ANOVA). The efficacy of vancomycin treatment increased after removal of the internal osteosynthesis device.
Table 1.
Efficacy of vancomycin in a rabbit model of post-traumatic osteomyelitis
| Group (no. of rabbits) | B1 (log10 CFU/mg) mean ± SD | B2 (log10 CFU/mg) mean ± SD | Δlog (log10 CFU/mg) mean ± SD |
|---|---|---|---|
| G1 (5) | 6.3 ± 0.8 | 5.2 ± 0.4 | −1.1 ± 0.5* |
| G2 (5) | 6.8 ± 0.9 | 3.9 ± 1.7 | −2.9 ± 1.1* |
*P = 0.0123, Δlog group 1 vs Δlog group 2, Bonferroni’s test after ANOVA
Discussion
Infections related to osteosynthesis devices often result in dramatic consequences for the patient. Prolonged hospitalization with intravenous antimicrobial chemotherapy and repeated surgical procedures are common, and even amputation and death may occur. Treatment of post-traumatic osteomyelitis is still a challenge for the physician and the surgeon, and the agents of nosocomial bone infections are often MRSA strains [6]. Few investigators have suggested treating infections related to orthopedic devices without initial surgery [3, 4]. Instead, immediate antimicrobial chemotherapy and removal of the infected device is preferred [7].
Glycopeptides are effective against all staphylococcal strains and have been studied extensively in the treatment of bone infections [8]. Although sporadic strains of vancomycin-resistant S. aureus have been isolated in several countries [9, 10, 11], glycopeptides are still considered the gold standard in treatment of MRSA-induced bone infections [11]. Vancomycin is a time-dependent, antimicrobial drug. Pharmacokinetic studies showed a peak concentration equivalent to ten times the MIC. Time-kill curves (8× MIC) showed a reduction of 2 log at 24 h. Thus, the vancomycin regimen chosen for this study (60 mg/kg b.i.d.) seems to be an adequate treatment in this model. Jacqueline et al. [12] demonstrated that continuous infusion and intramuscular (twice daily) administration of vancomycin have a similar efficacy in a rabbit endocarditis model. Intramuscular administration allowed to avoid stabling, rabbits being free of movement in their cages.
A limitation of the present study could be the use of a single drug regimen, because it has been shown in clinical studies that dual drug therapy may be more adequate [13]. Zimmerli et al. showed the high efficacy of regimen including rifampin on implant-related bacterial infection [13]. However, vancomycin alone can still be considered a valuable regimen in other experimental animal models of infection with foreign bodies [12].
In our study, we evaluated the effect of removing the infected device on the efficacy of the antibiotic treatment. Samples collected at day 4 confirmed infection at the surgical site. Since the decrease in bacterial count between day 8 and day 4 can be calculated for each rabbit, the power of the statistical test is increased. In addition, the rabbit model is applicable to human pathology and clinical management of infection.
Infections related to orthopedic devices are frequent. Tattevin and colleagues [14] have demonstrated that the success rate for the treatment of joint prosthesis infection without removal of the orthopedic device was better if surgical revision was performed before the fourth day after clinical signs appeared, suggesting that the best chance of clinical success is correlated with early and efficient treatment. Microbial adherence is recognized as the first step of foreign body infection [7, 8, 15, 16, 17]. It is a complex phenomenon involving bacterial factors (bacterial species, production of adhesins forming glycocalyx or slime), foreign body design and hydrophobicity, and host factors [18, 19, 20]. In clinical practice, it is quite easy to modify the bacterial factors. Surgical debridement and excision of infected tissues decrease the local bacterial count, and removal of the orthopedic device alters microbial adherence.
Moreover, orthopedic device removal (and that of fibrous tissues surrounding it) allows improvement of antibiotic diffusion in the infected site and eventually prevents a cellular penetration ofStaphylococcus aureus that may be considered as a cause of clinical failure.
The choice of a double tibial osteotomy was made in order to infuse the intermediate tibial fragment in a MRSA strain agar and eventually emphasize the local bone and soft tissue infections. During the surgical debridement, the intermediate tibial fragment wasn't removed because of early bone healing; then this fragment wasn’t considered as a sequestrum.
It’s likely that if the treatment had been prolonged more than 4 days, both groups might have been cured. However, the duration of treatment was deliberately restricted to 4 days as we aimed to show a difference of efficacy of antimicrobial treatment between the two groups. Furthermore, treatment efficacy should be obtained as soon as possible; thus the first days of treatment are critical. Consequently, extensive surgical debridement and double antimicrobial regimen are recommended. In this experimental study, removal of the osteosynthesis device allowed an increased efficacy of vancomycin at day 4.
This study only compared two different surgical techniques in the management of an infection related to the implantation of an osteosynthesis device, although clinical management of patients with post-traumatic osteomyelitis may be more complex, depending on the type of microorganism, status of the skin and soft tissues, patient status, and stability of the osteosynthesis device. However, the efficacy of vancomycin treatment increased significantly after removal of the internal osteosynthesis device, confirming the necessity of removing such a device whenever it is clinically possible.
References
- 1.Zimmerli W, Lew PD, Waldvogel FA. Pathogenesis of foreign body infection. Evidence for a local granulocyte defect. J Clin Invest. 1984;73:1191–1200. doi: 10.1172/JCI111305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zimmerli W, Waldvogel FA, Vaudaux P, Nydegger UE. Pathogenesis of foreign body infection: description and characteristics of an animal model. J Infect Dis. 1982;146:487–497. doi: 10.1093/infdis/146.4.487. [DOI] [PubMed] [Google Scholar]
- 3.Stein A, Bataille JF, Drancourt M, Curvale G, Argenson JN, Groulier P, Raoult D. Ambulatory treatment of multidrug-resistantStaphylococcus-infected orthopedic implants with high-dose oral co-trimoxazole (trimethoprim-sulfamethoxazole) Antimicrob Agents Chemother. 1998;42:3086–3091. doi: 10.1128/aac.42.12.3086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brouqui P, Rousseau MC, Stein A, Drancourt M, Raoult D. Treatment of pseudomonas æruginosa infected orthopedic prostheses with ceftazidime–ciprofloxacin antibiotic combination. Antimicrob Agents Chemother. 1995;39:2423–2425. doi: 10.1128/AAC.39.11.2423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yin LY, Lazzarini L, Li F, Stevens CM, Calhoun JH. Comparative evaluation of tigeciline and vancomycin, with and without rifampicin, in the treatment of methicillin-resistantStaphylococcus aureus experimental osteomyelitis in a rabbit model. J Antimicrob Chemother. 2005;55:995–1002. doi: 10.1093/jac/dki109. [DOI] [PubMed] [Google Scholar]
- 6.Cunha BA. Osteomyelitis in elderly patients. Clin Infect Dis. 2002;35:287–293. doi: 10.1086/341417. [DOI] [PubMed] [Google Scholar]
- 7.Jansen B, Peters G. Foreign body associated infection. J Antimicrob Chemother. 1993;32(Suppl A):69–75. doi: 10.1093/jac/32.suppl_A.69. [DOI] [PubMed] [Google Scholar]
- 8.Gracia E, Lacleriga A, Monzon M, Leiva J, Oteiza C, Amorena B. Application of a rat osteomyelitis model to compare in vivo and in vitro the antibiotic efficacy against bacteria with high capacity to form biofilms. J Surg Res. 1998;79:146–153. doi: 10.1006/jsre.1998.5416. [DOI] [PubMed] [Google Scholar]
- 9.Hamilton-Miller JM. Vancomycin-resistantStaphylococcus aureus: a real and present danger? Infection. 2002;30:118–124. doi: 10.1007/s15010-002-2160-8. [DOI] [PubMed] [Google Scholar]
- 10.Hiramatsu K. The emergence ofStaphylococcus aureus with reduced susceptibility to vancomycin in Japan. Am J Med. 1998;104:7S–10S. doi: 10.1016/S0002-9343(98)00149-1. [DOI] [PubMed] [Google Scholar]
- 11.Smith TL, Pearson ML, Wilcox KR, Cruz C, Lancaster MV, Robinson-Dunn B, Tenover FC, Zervos MJ, Band JD, White E, Jarvis WR. Emergence of vancomycin resistance inStaphylococcus aureus glycopeptide-intermediateStaphylococcus aureus working group. N Engl J Med. 1999;340:493–501. doi: 10.1056/NEJM199902183400701. [DOI] [PubMed] [Google Scholar]
- 12.Jacqueline C, Batard E, Perrez L, Boutoille D, Hamel A, Caillon J, Kergueris MF, Potel G, Bugnon D. In vivo efficacy of continuous infusion versus intermittent dosing of linezolid compared to vancomycin in a methicillin-resistantStaphylococcus aureus rabbit endocarditis model. Antimicrob Agents Chemother. 2002;46:3706–3711. doi: 10.1128/AAC.46.12.3706-3711.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zimmerli W, Widmer AF, Blatter M, Frei R, Ochsner PE. Role of rifampin for treatment of orthopedic implant-related staphylococcal infections: a randomized controlled trial. Foreign-body infection (FBI) study group. JAMA. 1998;279:1537–1541. doi: 10.1001/jama.279.19.1537. [DOI] [PubMed] [Google Scholar]
- 14.Tattevin P, Crémieux AC, P. Pottier P, D. Huten D, Carbon C (1998) Prosthetic joint infection: when can prosthesis salvage be considered? Clin Infect Dis 29:292–295 [DOI] [PubMed]
- 15.Besnier JM, Leport C. Effect of antibiotics on bacterial adherence to foreign material. Pathol Biol. 1990;38:243–248. [PubMed] [Google Scholar]
- 16.Gristina AG, Costerston JW. Bacterial adherence to biomaterials and tissues. The significance of its role in clinical sepsis. J Bone Joint Surg Am. 1985;67:264–273. [PubMed] [Google Scholar]
- 17.Monzon M, Garcia-Alvarez F, Lacleriga A, Amorena B. Evaluation of four experimental osteomyelitis infection models by using precolonized implants and bacterial suspensions. Acta Orthop Scand. 2002;73:11–19. doi: 10.1080/000164702317281341. [DOI] [PubMed] [Google Scholar]
- 18.Arens S, Eijer H, Schegel U, Printzen G, Perren SM, Hansis M. Influence of the design for fixation implants on local infection. An experimental study of DC-plates vs. Point-contact-fixators in rabbits. J Orthop Trauma. 1999;13:470–473. doi: 10.1097/00005131-199909000-00002. [DOI] [PubMed] [Google Scholar]
- 19.Arens S, Schlegel U, Printzen G, Ziegler WJ, Perren SM, Hansis M. Influence of the materials for fracture fixation implants on the development of local infection. An experimental study of steel vs. Titanium DC-plates in rabbits. J Bone Joint Surg Br. 1996;78:647–651. [PubMed] [Google Scholar]
- 20.Melcher GA, Claudi B, Schlegel U, Perren SM, Printzen G, Munzinger J. Influence of type of medullary nail on the development of local infection. An experimental study of solid and slotted nails in rabbits. J Bone Joint Surg Br. 1994;76:955–959. [PubMed] [Google Scholar]


