Abstract
The zinc finger-containing protein A20 is a negative regulator of TNF-induced JNK (c-Jun-N-terminal kinase) and NFκB (nuclear factor κB) signaling. A20 is an unusual enzyme that contains both ubiquitinating and deubiquitinating activities. Although A20 is mostly localized in the cytosol, our recent studies reveal that a fraction of A20 can associate with a lysosome-interacting compartment in a manner that requires its carboxy terminal zinc fingers, but independent of its ubiquitin modifying activities. Whether the lysosome-associated A20 has a function in cellular signaling is unclear. Here, we demonstrate that A20 is capable of targeting an associated signaling molecule such as TRAF2 to the lysosomes for degradation. This process is dependent on the membrane tethering zinc finger domains of A20, but does not require A20 ubiquitin modifying activity. Our findings suggest a novel mode of A20 action that involves lysosomal targeting of signal molecules bound to A20.
Keywords: A20, TRAF2, NFκB, Ubiquitin, Lysosomal degradation
1. Introduction
Receptors of the proinflammatory cytokine TNF (tumor necrosis factor) superfamily mediate cellular responses to a variety of environmental stimuli [1,2]. On binding to TNF, TNF receptors (TNFR) assemble a membrane-associated protein complex comprising the adaptor protein TRADD (TNF receptor associated death domain), a Ser/Thr protein kinase RIP1 (receptor interacting protein), and a TRAF (TNF receptor-associated factor) domain-containing protein TRAF2 [3–8]. Subsequently, a series of intracellular events are induced, leading to the activation of the transcription factor NFκB. TNF also activates JNK kinase through TRAF2, which in turn activates another transcription factor c-Jun [9–11]. The TNFR activation may promote either cell survival or death depending on the relative output of the life and death signals associated with these TNFR-activated processes.
The activation of NFκB by TNF induces the expression of many genes including the zinc finger protein A20, which turns out to be a potent inhibitor of TNF signals [12–20]. Genetic ablation of A20 in mice causes prolonged TNF activation, which is associated with sustained inflammatory responses and cachexia [19]. Thus, A20- dependent negative regulatory circuit plays a pivotal role in restricting the intensity and duration of TNF signaling in vivo.
The mechanism by which A20 regulates TNF signals was proposed to involve its dual ubiquitin-modifying activities, which can act on a substrate such as RIP1 to alter its stability. Specifically, it has been shown that A20 is capable of removing Lys63-linked ubiquitin chains from RIP1 using its amino-terminal OTU (ovarian tumor) domain-dependent deubiquitinating activity. Subsequently, it assembles Lys48-linked ubiquitin chains on RIP1 using its carboxy terminal zinc finger domains to promote the proteasomal degradation of RIP1 [21]. Although downregulation of RIP1 by A20 appears to contribute to its NFκB inhibitory function [22], many studies demonstrated that A20 mutants lacking ubiquitin-modifying activities can still function and inhibit TNFα activity under various conditions [15,23–26]. These observations indicate that additional mechanism(s) may be employed by A20 to regulate TNF signaling.
In the search for a new mechanism of A20 action, we recently discovered that a fraction of endogenous and ectopically expressed A20 is localized to a perinuclear membrane compartment that interacts dynamically with the lysosomes [26]. This localization requires the carboxy terminal zinc finger domains of A20. Coincidentally, the NFκB inhibitory activity of A20 in many cell lines also relies on these domains [15,23–26]. This correlation suggests that A20 may be able to target signaling molecules to the lysosomes for degradation, and therefore regulate the response of cells to TNF signals.
In this report, we demonstrate that A20 can recruit an associated signaling molecule such as TRAF2 to a lysosome-interacting compartment for degradation. As anticipated, the downregulation of TRAF2 requires the membrane tethering zinc finger domains of A20. Interestingly, our results show that A20 dependent downregulation of endogenous TRAF2 only occurs under certain conditions that promote TNF-induced apoptosis, suggesting a regulatory role for A20-mediated lysosomal degradation in a delicate balance between life and death during TNF signaling.
2. Materials and methods
2.1. Constructs, antibodies, and chemicals
The pRK-Flag-A20 wt, pRK-HA-A20, pRK-Flag-TRAF2 plasmids and the plasmid expressing TRAF2ΔRING were described previously [15,27,28]. The plasmids expressing various A20 mutants were described previously [26]. The plasmid expressing TRAF2 1-310 was generated by site directed mutagenesis. The pLNCX2-Flag-TRAF2 plasmid was constructed by ligating the TRAF2 coding sequence into the XhoI and NotI sites of the pLNCX2 vector (Clontech). The plasmid expressing CFP-tagged TRAF2 was constructed by ligating the PCR amplified TRAF2 coding sequence into the HindIII and KpnI sites of the pECFP-N1 vector (Clontech). The pEYFP-A20 plasmid was constructed by ligating the PCR amplified A20 coding sequence into the BglII and SalI sites of the pEYFP-C1 vector (Clontech). Flag, Myc, and A20 (59A426) monoclonal antibodies were purchased from Sigma, Roche, and Imgenex, respectively. Phospho-JNK and JNK antibodies were purchased from Cell signaling, Inc. The TRAF2 antibodywas purchased from Santa Cruz. Chloroquine and actinomycin D were purchased from Sigma. MG132 was purchased from Calbiochem. TWEAK and Lysotraker were purchased from Invitrogen.
2.2. Cell lines
293T and COS7 cells were purchased from ATCC, and maintained according to standard protocols. Wild type and A20 deficient mouse embryonic cells were kindly provided by Averil Ma (University of California San Francisco). To make a 293T cell line expressing Flag-tagged TRAF2, a retroviral packaging cell line (GP2-293) was co-transfected with pLNCX2-Flag-TRAF2 and a plasmid expressing the VSV-G protein. The retrovirus-containing medium was collected 72 h after transfection, and used to infect 293Tcells. TRAF2-expressing cells were maintained in a medium containing neomycin (500 μg/ml). Individual clones were selected and analyzed for TRAF2 expression.
2.3. RNA preparation and RT–PCR
Total RNA was extracted using TRIzol reagent (Invitrogen), and subsequently purified using an RNeasy MinElute Cleanup kit (QIA-GEN). For RT–PCR, cDNA was synthesized using the SuperScript™ First-Strand kit (Invitrogen). The following primers were used in PCR to detect the mRNA expression of mouse A20: forward, 5′-TTTGAGCAATATGCGGAAAGC; Reverse, 5′-ATTTTGTCTGAAATGACAATG. Actin was used as a control. Forward, 5′-GCTCCGGCATGTGCAA; Reverse, 5′-AGGATCTTCATGAGGTAGT.
2.4. Transfection, immunoblotting, and pulse chase experiments
Transfectionwas done either with Polyfect (Qiagen) for COS7 cells, or with the TransIT293 reagent (Mirus, Madison, WI) for 293T cells. Immunoblotting was performed according to standard protocol. Pulse chase experiments were performed as described previously [29,30]. Briefly, cells were starved in a Met/Cys free medium for 40 min, and then incubated in a medium containing 35S-Met/Cyc for 20 min. Cells were then incubated in a chase medium containing excessive amount of unlabeled Met/Cys. Cell extracts were prepared in a RIPA buffer (50 mM Tris–HCl pH 7.5, 1% NP40, 0.1% SDS, 0.5% sodium deoxycholate, 150 mM sodium chloride, 2 mM EDTA and a protease inhibitor cocktail). TRAF2 was immunoprecipitated from detergent extracts and analyzed by SDS–PAGE and autoradiography. To inhibit the lysosome function, unless otherwise specified in the figure legends, 50 mM NH4Cl or 100 μMchloroquinewas included in the starvation and chase medium.
2.5. Immunostaining and cell imaging
Immunofluorescence experiments were performed as described previously [31]. Briefly, cells fixed in a phosphor saline solution (PBS) containing 4% paraformaldehyde were permeabilized in a solution containing 0.1% NP40 and 4% normal donkey serum. Cells were then stained with the primary and secondary antibodies in the same buffer. Cells were imaged using either a Zeiss Axiovert fluorescence microscope or a Zeiss LSM510 laser scanning confocal microscope equipped with a 63X oil immersion Plan-Apochromat objective (N/A 1.4).
3. Results
3.1. A20 induces the degradation of ectopically expressed TRAF2
To test whether A20 was capable of targeting its associated signaling molecules to the lysosomes for degradation, we investigated one candidate—the TNF receptor-associated factor 2 (TRAF2) for the following reasons. First, A20 interacts directly with TRAF2 [15]. Second, A20 can block both JNK (Fig. 1A, B) and NFκB activation [15,17] induced by overexpression of TRAF2. Since TRAF2 overexpression activates these signaling events via distinct downstream components [32], a simple explanation of these observations is that A20 may directly inhibit TRAF2 to terminate these signals. Otherwise, A20 would have to act on multiple molecules downstream of TRAF2.
Fig. 1.
A20 induces the degradation of ectopically expressed TRAF2. (A) Downregulation of TRAF2 expression by A20. Detergent extracts of 293T cells transfected with a GFP-expressing plasmid together with the indicated plasmids were analyzed by immunoblotting with antibodies against the indicated proteins. WCE, whole cell extract. (B) As in (A), except that primary MEF cells were used. (C) As in (A), except that YFP-tagged A20 was also tested and that transfected COS7 cells were directly lysed in the Laemmli buffer. (D) Immunofluorescence experiment confirms the downregulation of TRAF2 by A20. A clone derived from a single 293T cell stably expressing Flag-TRAF2 was transfected with a plasmid expressing HA-tagged A20 (HA-A20), and stained with antibodies to visualize the indicated proteins. The left panel shows an untransfected control. Images were obtained using a 20X Plan-Neofluar objective (N/A 0.4). Note that the A20 and TRAF2 vesicles were not visible at this resolution. (E) Pulse chase experiment demonstrates that TRAF2 is unstable in A20 expressing cells. The graph shows the quantification of the experiment.
We first analyzed the effect of A20 overexpression on the steady state level of TRAF2. 293T cells transfected with a plasmid expressing Flag-tagged TRAF2 together with either a control or an A20 expressing plasmid were subjected to extraction by detergent. Immunoblotting showed that expression of A20 dramatically reduced the TRAF2 protein level (Fig. 1A, lane 4 versus lane 3). This observation was further confirmed in primary MEF cells (Fig. 1B). In addition, expression of YFP-tagged A20 in COS7 cells similarly downregulated TRAF2 (Fig. 1C, lane 3 versus lane 1). Because this experiment was performed by lysing cells directly in the Laemmli buffer, the loss of TRAF2 in A20-expressing cells could not be due to partition of TRAF2 into a detergent insoluble fraction. Immunofluorescence experiments further confirmed that TRAF2 was indeed downregulated in A20- expressing cells (Fig. 1D).
To test if A20-mediated downregulation of TRAF2 was due to increased TRAF2 turnover, we measured the stability of TRAF2 protein in a pulse chase experiment. To this end, cells expressing TRAF2 were radiolabeled with 35S-Met/Cys and subsequently incubated in a chase medium containing excess unlabeled Met/Cys. The immuno-precipitation experiment showed that expression of A20 significantly shortened the half life of TRAF2 (Fig. 1E). Together, these results suggest that A20 can reduce the level of TRAF2 by accelerating its degradation.
To determine the amino acid sequence in TRAF2 that is important for A20-dependent degradation, we generated TRAF2 truncation mutants that lacked either the N-terminal RING domain or the C-terminal TRAF domain (Fig. 2A). The RING domain of TRAF2 contains ubiquitin ligase activity that is required for its function in TNF dependent JNK signaling [32–35]. Interestingly, expression of A20 only had a moderate effect on the half-life of the TRAF2ΔRING mutant (Fig. 2B, lanes 5–8 versus lanes 1–4). In contrast, a TRAF2 mutant lacking the TRAF domain (TRAF2 1-310) was destabilized by A20 similarly to wild type TRAF2 (Fig. 2B). Since the TRAF2ΔRING and TRAF2 1-310 mutants both interact with A20 [15], it appears that the association of TRAF2 with A20 alone is not sufficient to trigger efficient degradation of TRAF2.
Fig. 2.
The RING finger domain of TRAF2 is required for A20-induced turnover. (A) Schematic representation of the TRAF2 constructs (Znf, Zinc finger; CC, coiled-coil; RING, really interesting gene; TRAF, TNF receptor associated factor). (B) Degradation of the TRAF2 variants in A20 expressing cells. Numbers underneath the graphs indicate the relative amount of TRAF2.
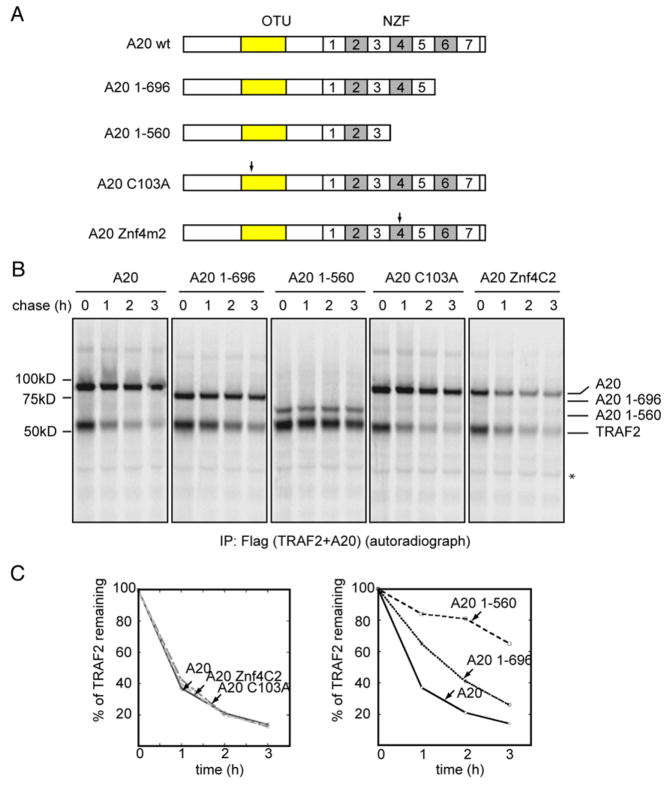
3.2. The degradation of TRAF2 requires the membrane tethering activity of A20
A previous study showed that A20 contains both ubiquitin ligase and deubiquitinating activities, which are required for targeting RIP1 to the proteasome for degradation [21]. We therefore tested whether the ubiquitin proteasome system plays a role in A20-mediated degradation of TRAF2. Pulse chase experiments showed that the degradation of TRAF2 in A20-expressing cells was not blocked by treating these cells with the proteasome inhibitor MG132 (Fig. 3 lanes 5–8). These data argue against the involvement of the proteasome in this process. To further study this issue, we analyzed TRAF2 degradation in cells expressing A20 mutants defective in either ubiquitination or deubiquitination. Indeed, both the ubiquitinating (A20 Znf4C2) and deubiquitinating defective A20 mutants (A20 C103A) were as effective as wild type A20 in downregulating TRAF2 (Fig. 4). Together, these data strongly indicate that A20 is able to induce TRAF2 turnover by an ubiquitin proteasome independent pathway.
Fig. 3.
The degradation of TRAF2 in A20-expressing cells is not mediated by the proteasome. The degradation of TRAF2 in 293T cells expressing wild type A20 was analyzed by pulse-chase experiment. Where indicated, cells were treated with a proteasome inhibitor MG132 (20 μM). The graph shows the quantification of the experiment.
Fig. 4.
The A20-dependent degradation of TRAF2 requires the membrane tethering activity of A20, but not its ubiquitin-modifying function. (A) Schematic representation of the A20 constructs (OTU, ovarian tumor; NZF, Npl4 zinc finger). Arrows indicate the location of the mutations. (B) The degradation of TRAF2 in 293Tcells expressing the indicated A20 variants was analyzed by pulse chase experiment. Asterisk indicates a non-specific protein. (C) Quantification of the experiment in (B).
Since we previously found that a fraction of A20 is localized to a lysosome-associated compartment via its carboxy terminal zinc finger domains, we analyzed the degradation of TRAF2 in cells expressing A20 mutants defective in membrane association. Immunoblotting experiments have shown that these A20 variants were expressed at a slightly higher level than wild type A20 [26]. Nonetheless, compared with wild type A20, the A20 mutant lacking the last two zinc fingers (A20 1-696) was less active, and the A20 mutant lacking the last four zinc fingers (A20 1-560) was more severely impaired in down-regulating TRAF2 (Fig. 4). Intriguingly, we previously showed that the A20 1-696 mutant retained partial interaction with the lysosomes, whereas the A20 1-560 mutantwas exclusively localized to the cytosol [26]. Thus, the ability of A20 to induce TRAF2 degradation correlates with its association with the lysosomes. Since A20 mutants defective in membrane association are also less active in inhibiting NFκB signals [26], these results suggest that A20 mediated lysosomal degradation may contribute to its regulation of the TNF signal.
3.3. A20 recruits TRAF2 to a lysosome-associated compartment for degradation
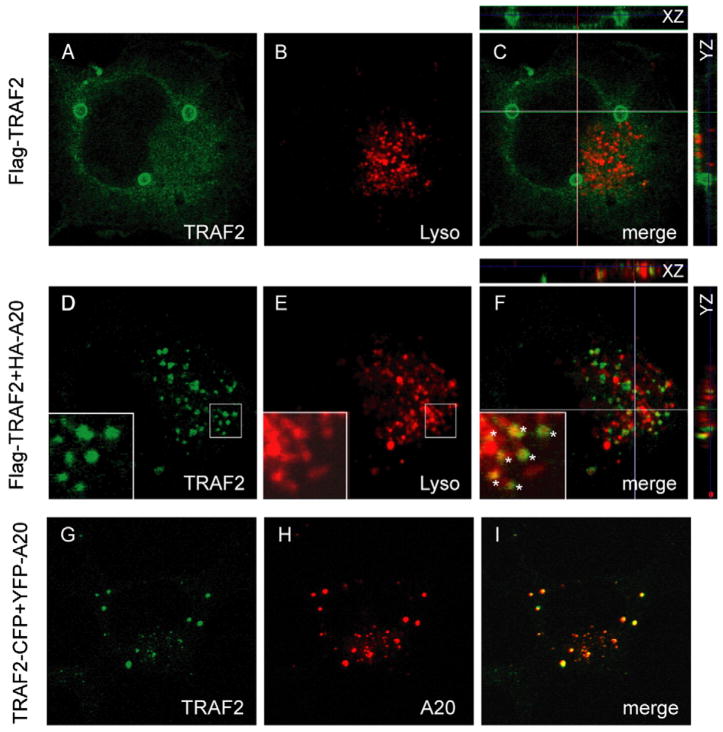
We next examined the localization of TRAF2 in COS7 cells in the presence or absence of ectopically expressed A20. When TRAF2 was expressed alone, it was mostly localized in the cytosol. A fraction of TRAF2 was associated with membrane vesicles that constantly fused to form a few large round-shaped vesicles (Figs. 5A–C). The biological relevance of these vesicles was unclear, but they did not display any overlapping localization with the lysosomes. In contrast, when A20 was co-expressed with TRAF2, the TRAF2 protein level was reduced, and the remaining TRAF2 was mostly concentrated in a juxtanuclear region as speckles reminiscent of the A20 staining pattern (Figs. 5D–F) [26]. Indeed, these TRAF2-containing dots did colocalize precisely with A20 (Figs. 5G–I), confirming the reported interaction between A20 and TRAF2 [15]. Consistent with our previous observation that A20-containing vesicles display dynamic interactions with the lysosomes [26], the TRAF2-containing vesicles in A20-expressing cells also overlapped significantly with the lysosomes (see insets in Fig. 5). Thus, A20 is able to recruit TRAF2 to vesicles that are in association with the lysosomes, which perhaps leads to the degradation of TRAF2 by a lysosome dependent mechanism.
Fig. 5.
A20 recruits TRAF2 to a lysosome-associated compartment. (A–C) COS7 cells expressing Flag-TRAF2 were stained with Lysotracker (red) and an anti-Flag antibody (green). (D–F) COS7 cells co-expressing Flag-TRAF2 and HA-A20 were stained with Lysotracker (red) and an anti-Flag antibody (green). The insets showa magnified view of the indicated region (box). Asterisks indicate some examples of TRAF2-containing vesicles that are either colocalized or tethered with the lysosomes. The smaller panels in (C) and (F) show vertical sections along the XZ and YZ axes indicated by the lines. (G–I) COS7 cells co-expressing TRAF2-CFP and YFP-A20 were fixed and imaged. All images were obtained with a Zeiss LSM510 laser scanning confocal microscope.
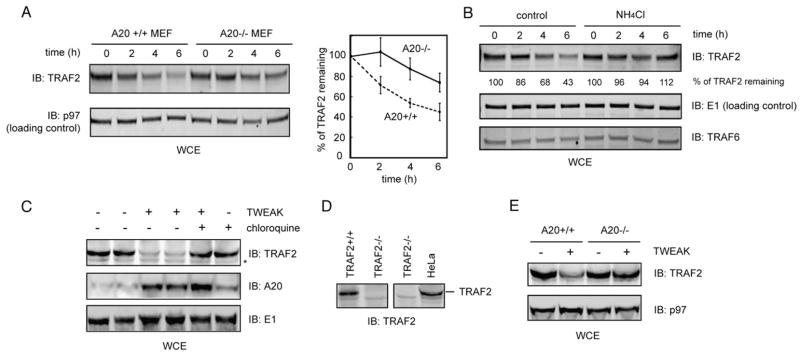
To test this possibility, 293T cells co-expressing TRAF2 and A20 were treated with two lysosomal inhibitors, ammonium chloride and chloroquine. The stability of TRAF2 in untreated control cells and the inhibitor-treated cells were analyzed by pulse chase experiments. Our data showed that the A20-induced degradation of TRAF2 could be inhibited by these inhibitors (Figs. 6A, B), confirming the involvement of the lysosomes in this process.
Fig. 6.
Lysosome dependent degradation of TRAF2 in A20-expressing cells. (A, B) Pulse chase experiments show the inhibition of TRAF2 degradation in A20 expressing cells by lysosome inhibitors. Graphs show the quantification of the experiments. Note that data from two independent experiments for chloroquine treatment (0 and 50 μM) were labeled in the graph in (B) as dots. Chloroquine at 100 μM appears to reduce the labeling efficiency. The lines indicate the average of the two experiments. (C) Characterization of A20 deficient (A20−/−) MEF cells. A20 mRNA level in the indicated cells was determined by RT–PCR. (D) Accumulation of TRAF2 in A20 deficient primary MEF cells. Wild type and A20 deficient MEF cells were transfected with the indicated plasmids together with Flag-TRAF2 and GFP (transfection efficiency control). Whole cell extracts (WCE) were analyzed by immunoblotting. Shown is a gel representing two independent experiments.
To see if A20 is required for TRAF2 turnover, we used A20 deficient primary MEF cells (Fig. 6C). Indeed, ectopically expressed TRAF2 was stabilized in cells lacking A20. Moreover, expression of A20 in A20 deficient cells completely restored the degradation of TRAF2, resulting in a lower level of TRAF2 (Fig. 6D). These results further demonstrate that the stability of ectopically expressed TRAF2 is regulated by A20.
3.4. A20-dependent degradation of endogenous TRAF2
We next examined whether the stability of endogenous TRAF2 is influenced by A20. Intriguingly, expression of A20 did not result in significant loss of endogenous TRAF2 protein (data not shown), which is consistent with a previous report [36]. Moreover, we and others have found that in cells treated with TNFα alone, endogenous TRAF2 is not degraded [37,38]. On the other hand, endogenous TRAF2 was found to be rapidly downregulated in MEF cells treated with TNFα together with actinomycin D, a condition that promotes TNFα-induced apoptotic signals [39]. Although the degradation of TRAF2 under this condition may be to some extent influenced by a proteasome inhibitor and the level of the ubiquitin ligase Siah2, neither the proteasome inhibitor nor genetic ablation of Siah2 completely stabilizes TRAF2, suggesting that a proteasome independent mechanismmay also account for the degradation of TRAF2 under this condition [39]. We therefore tested whether the lysosomes may play a role in TRAF2 turnover under this condition and whether the loss of TRAF2 is dependent on A20. We analyzed the level of endogenous TRAF2 by immunoblotting and found that it was indeed decreased in cells treated with TNFα and actinomycin D (Fig. 7A, lanes 1–4). The downregulation of TRAF2 was at least partially dependent on A20 because the stability of TRAF2 was increased in A20 deficient MEF cells (Fig. 7A, lanes 5–8 versus lanes 1–4, also see the graph in Fig. 7A). Additionally, pretreating cells with a lysosomal inhibitor attenuated TRAF2 degradation (Fig. 7B), suggesting that the degradation of TRAF2 may take place in the lysosome.
Fig. 7.
A20-dependent degradation of endogenous TRAF2. (A) Impaired degradation of endogenous TRAF2 in A20 deficient MEF cells. Wild type (A20+/+) or A20 deficient (A20−/−) MEF cells were treated with TNFα (40 ng/ml) and actinomysin D (1μg/ml) for the indicated time points. Cell extracts were analyzed by immunoblotting (IB). Graph shows the quantification of four independent experiments. Error bars represent S.D. (n=4). (B) Inhibition of TRAF2 degradation by a lysosomal inhibitor. HeLa cells were treated with TNFα and actinomysin D for the indicated time periods and analyzed as in (A). Where indicated, cells were pretreated with NH4Cl for 1 h. Numbers indicate the relative level of TRAF2. Shown is a gel representing 3 independent experiments. (C) TWEAK induces the loss of endogenous TRAF2 by a lysosome dependent mechanism. HeLa cells were treated with the indicated protein/chemical (TWEAK, 100 ng/ml; chloroquine, 80 μM) for 6 h. Cell extracts were analyzed by immunoblotting. Asterisk indicates a non-specific band. (D) The specificity of the TRAF2 antibody as demonstrated by immunoblotting analyses using whole cell extract from the indicated cell lines. (E) TWEAK-mediated TRAF2 turn over requires A20. Immortalized wild type (+/+) and A20 knock-out cells were treated for 6 h as indicated. Whole cell extract was subjected to immunoblotting analysis.
Although TNFα alone is insufficient to activate apoptosis in many cell lines, a recent study showed that another TNF superfamily member TWEAK on its own is capable of inducing apoptosis via TNFα signaling in many types of cancer cells and transformed MEF. TWEAK can also sensitize these cells to TNFα-induced cell death, and it appears to act by triggering the degradation of a protein complex consisting of endogenous TRAF2 via a lysosome dependent mechanism [40]. We therefore tested whether TWEAK-induced TRAF2 loss requires A20. We first confirmed that endogenous TRAF2 could be downregulated in response to TWEAK treatment and it could be blocked by the lysosome inhibitor chloroquine, as previously demonstrated (Fig. 7C, D) [40]. TWEAK-induced TRAF2 losswas also observed in immortalized wild type MEF cells, but was significantly attenuated in A20 deficient MEF cells (Fig. 7E). Thus, A20 appears to play a role in TWEAK-induced TRAF2 downregulation.
4. Discussion
Previous work showed that A20 downregulates RIP1 via the ubiquitin proteasome system. It is thought that both the ubiquitin ligase and the deubiquitinating activities of A20 are required for RIP1 turnover and for the NFκB inhibitory function of A20 [21]. In this context, it is surprising that we and others found that the ubiquitin modifying activities of A20 can be dispensable for its function under many experimental conditions [15,23–26]. Perhaps, there exists an ubiquitin independent mechanism of A20 action.
In support of this idea, we recently demonstrated that A20 is also localized to a novel membrane compartment that dynamically communicates with the lysosomes. Intriguingly, A20 mutants defective in membrane association also contain reduced NFκB inhibitory activity, suggesting that A20 may function through this compartment to participate in cellular signaling [26]. One attractive model is that A20 may target signaling molecules to this lysosome-associated compartment for degradation, which may help to regulate the TNF signaling. However, direct evidence in support of this model has been lacking.
In this study, we demonstrate that A20 indeed has the capacity to target an associated signaling molecule such as TRAF2 to the lysosome-associated compartment for clearance. As expected, the downregulation of TRAF2 by A20 does not require its ubiquitin modifying function. Instead, the carboxy terminal zinc fingers of A20 that mediate its membrane association are involved. These results, together with previous observations that the same zinc finger domains are required for A20 function [15, 23–26], indicate that the membrane associated pool of A20 may be functionally critical for TNF signaling under certain conditions. Indeed, we showed that endogenous TRAF2 can be downregulated by A20 under a condition that promotes TNF-induced cell death, and the degradation of TRAF2 can be inhibited by a lysosome inhibitor. Along the same line, endogenous TRAF2 is also rapidly degraded in many types of cancer cells in response to TWEAK, another TNF superfamily member, which on its own is sufficient to induce TNFα dependent cell death [40]. Interestingly, the degradation of TRAF2 under this condition is not mediated by the ubiquitin proteasome system. Instead, TRAF2 is targeted to the lysosomes for degradation. We now demonstrate that TWEAK-mediated loss of TRAF2 is also dependent on A20.
It is likely that TRAF2 may not be the only substrate of A20. An attractive scenario is that A20-mediated lysosomal degradation of a collection of signaling molecules required for sustained NFκB and JNK activation may offer a powerful means to regulate these signaling activities. In this regard, identification of additional substrates that undergo A20-dependent lysosomal degradation during TNF signaling would help to further clarify this novel regulatory mechanism in TNF signaling.
Acknowledgments
We thank Michael Krause, Zheng-Gang Liu, Jonathan Ashwell, and Harris Bernstein for critical reading of the manuscript, Zheng-Gang Liu for TRAF2−/− MEF cells, Averil Ma for A20−/− MEF cells, Jennifer Macdonald for constructing the plasmids expressing the TRAF2 variants. This Research is supported by the intramural research program of the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health.
Abbreviations
- TNF
tumor necrosis factor
- JNK
c-Jun-N-terminal kinase
- NFκB
nuclear factor κB
- TRAF
TNF receptor-associated factor
- RIP
receptor interacting protein
- TNFR
TNF receptor
- OTU
ovarian tumor
- MEF
mouse embryonic fibroblast
Footnotes
Publisher's Disclaimer: This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution and sharing with colleagues.
Other uses, including reproduction and distribution, or selling or licensing copies, or posting to personal, institutional or third party websites are prohibited.
In most cases authors are permitted to post their version of the article (e.g. in Word or Tex form) to their personal website or institutional repository. Authors requiring further information regarding Elsevier’s archiving and manuscript policies are encouraged to visit: http://www.elsevier.com/copyright
References
- 1.Chen G, Goeddel DV. TNF-R1 signaling: a beautiful pathway. Science. 2002;296:1634–1635. doi: 10.1126/science.1071924. [DOI] [PubMed] [Google Scholar]
- 2.Karin M, Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol. 2005;5:749–759. doi: 10.1038/nri1703. [DOI] [PubMed] [Google Scholar]
- 3.Rothe M, Wong SC, Henzel WJ, Goeddel DV. A novel family of putative signal transducers associated with the cytoplasmic domain of the 75 kDa tumor necrosis factor receptor. Cell. 1994;78:681–692. doi: 10.1016/0092-8674(94)90532-0. [DOI] [PubMed] [Google Scholar]
- 4.Stanger BZ, Leder P, Lee TH, Kim E, Seed B. RIP: a novel protein containing a death domain that interacts with Fas/APO-1 (CD95) in yeast and causes cell death. Cell. 1995;81:513–523. doi: 10.1016/0092-8674(95)90072-1. [DOI] [PubMed] [Google Scholar]
- 5.Hsu H, Xiong J, Goeddel DV. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. Cell. 1995;81:495–504. doi: 10.1016/0092-8674(95)90070-5. [DOI] [PubMed] [Google Scholar]
- 6.Hsu H, Shu HB, Pan MG, Goeddel DV. TRADD–TRAF2 and TRADD–FADD interactions define two distinct TNF receptor 1 signal transduction pathways. Cell. 1996;84:299–308. doi: 10.1016/s0092-8674(00)80984-8. [DOI] [PubMed] [Google Scholar]
- 7.Hsu H, Huang J, Shu HB, Baichwal V, Goeddel DV. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity. 1996;4:387–396. doi: 10.1016/s1074-7613(00)80252-6. [DOI] [PubMed] [Google Scholar]
- 8.Yeh WC, et al. Early lethality, functional NF-kappaB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity. 1997;7:715–725. doi: 10.1016/s1074-7613(00)80391-x. [DOI] [PubMed] [Google Scholar]
- 9.Lee SY, et al. TRAF2 is essential for JNK but not NF-kappaB activation and regulates lymphocyte proliferation and survival. Immunity. 1997;7:703–713. doi: 10.1016/s1074-7613(00)80390-8. [DOI] [PubMed] [Google Scholar]
- 10.Natoli G, et al. Activation of SAPK/JNK by TNF receptor 1 through a noncytotoxic TRAF2-dependent pathway. Science. 1997;275:200–203. doi: 10.1126/science.275.5297.200. [DOI] [PubMed] [Google Scholar]
- 11.Papa S, Zazzeroni F, Pham CG, Bubici C, Franzoso G. Linking JNK signaling to NF-kappaB: a key to survival. J Cell Sci. 2004;117:5197–5208. doi: 10.1242/jcs.01483. [DOI] [PubMed] [Google Scholar]
- 12.Opipari AW, Jr, Boguski MS, Dixit VM. The A20 cDNA induced by tumor necrosis factor alpha encodes a novel type of zinc finger protein. J Biol Chem. 1990;265:14705–14708. [PubMed] [Google Scholar]
- 13.Opipari AW, Jr, Hu HM, Yabkowitz R, Dixit VM. The A20 zinc finger protein protects cells from tumor necrosis factor cytotoxicity. J Biol Chem. 1992;267:12424–12427. [PubMed] [Google Scholar]
- 14.Jaattela M, Mouritzen H, Elling F, Bastholm L. A20 zinc finger protein inhibits TNF and IL-1 signaling. J Immunol. 1996;156:1166–1173. [PubMed] [Google Scholar]
- 15.Song HY, Rothe M, Goeddel DV. The tumor necrosis factor-inducible zinc finger protein A20 interacts with TRAF1/TRAF2 and inhibits NF-kappaB activation. Proc Natl Acad Sci U S A. 1996;93:6721–6725. doi: 10.1073/pnas.93.13.6721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cooper JT, et al. A20 blocks endothelial cell activation through a NF-kappaB-dependent mechanism. J Biol Chem. 1996;271:18068–18073. doi: 10.1074/jbc.271.30.18068. [DOI] [PubMed] [Google Scholar]
- 17.Heyninck K, Beyaert R. The cytokine-inducible zinc finger protein A20 inhibits IL- 1-induced NF-kappaB activation at the level of TRAF6. FEBS Lett. 1999;442:147–150. doi: 10.1016/s0014-5793(98)01645-7. [DOI] [PubMed] [Google Scholar]
- 18.Eliopoulos AG, Blake SM, Floettmann JE, Rowe M, Young LS. Epstein–Barr virus-encoded latent membrane protein 1 activates the JNK pathway through its extreme C terminus via a mechanism involving TRADD and TRAF2. J Virol. 1999;73:1023–1035. doi: 10.1128/jvi.73.2.1023-1035.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee EG, et al. Failure to regulate TNF-induced NF-kappaB and cell death responses in A20-deficient mice. Science. 2000;289:2350–2354. doi: 10.1126/science.289.5488.2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Beyaert R, Heyninck K, Van Huffel S. A20 and A20-binding proteins as cellular inhibitors of nuclear factor-kappa B-dependent gene expression and apoptosis. Biochem Pharmacol. 2000;60:1143–1151. doi: 10.1016/s0006-2952(00)00404-4. [DOI] [PubMed] [Google Scholar]
- 21.Wertz IE, et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-kappaB signalling. Nature. 2004;430:694–699. doi: 10.1038/nature02794. [DOI] [PubMed] [Google Scholar]
- 22.Heyninck K, Beyaert R. A20 inhibits NF-kappaB activation by dual ubiquitin-editing functions. Trends Biochem Sci. 2005;30:1–4. doi: 10.1016/j.tibs.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 23.Natoli G, et al. Nuclear factor kB-independent cytoprotective pathways originating at tumor necrosis factor receptor-associated factor 2. J Biol Chem. 1998;273:31262–31272. doi: 10.1074/jbc.273.47.31262. [DOI] [PubMed] [Google Scholar]
- 24.Klinkenberg M, Van Huffel S, Heyninck K, Beyaert R. Functional redundancy of the zinc fingers of A20 for inhibition of NF-kappaB activation and protein–protein interactions. FEBS Lett. 2001;498:93–97. doi: 10.1016/s0014-5793(01)02504-2. [DOI] [PubMed] [Google Scholar]
- 25.Evans PC, et al. Zinc-finger protein A20, a regulator of inflammation and cell survival, has de-ubiquitinating activity. Biochem J. 2004;378:727–734. doi: 10.1042/BJ20031377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li L, et al. Localization of A20 to a lysosome-associated compartment and its role in NFkappaB signaling. Biochim Biophys Acta. 2008;1783:1140–1149. doi: 10.1016/j.bbamcr.2008.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Huang J, et al. ZNF216 Is an A20-like and IkappaB kinase gamma-interacting inhibitor of NFkappaB activation. J Biol Chem. 2004;279:16847–16853. doi: 10.1074/jbc.M309491200. [DOI] [PubMed] [Google Scholar]
- 28.Wang YY, Li L, Han KJ, Zhai Z, Shu HB. A20 is a potent inhibitor of TLR3- and Sendai virus-induced activation of NF-kappaB and ISRE and IFN-beta promoter. FEBS Lett. 2004;576:86–90. doi: 10.1016/j.febslet.2004.08.071. [DOI] [PubMed] [Google Scholar]
- 29.Ye Y, Meyer HH, Rapoport TA. The AAA ATPase Cdc48/p97 and its partners transport proteins from the ER into the cytosol. Nature. 2001;414:652–656. doi: 10.1038/414652a. [DOI] [PubMed] [Google Scholar]
- 30.Wang Q, Li L, Ye Y. Regulation of retrotranslocation by p97-associated deubiquitinating enzyme ataxin-3. J Cell Biol. 2006;174:963–971. doi: 10.1083/jcb.200605100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ye Y, Shibata Y, Yun C, Ron D, Rapoport TA. A membrane protein complex mediates retro-translocation from the ER lumen into the cytosol. Nature. 2004;429:841–847. doi: 10.1038/nature02656. [DOI] [PubMed] [Google Scholar]
- 32.Xia ZP, Chen ZJ. TRAF2: a double-edged sword? Sci STKE. 2005;2005:pe7. doi: 10.1126/stke.2722005pe7. [DOI] [PubMed] [Google Scholar]
- 33.Deng L, et al. Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell. 2000;103:351–361. doi: 10.1016/s0092-8674(00)00126-4. [DOI] [PubMed] [Google Scholar]
- 34.Shi CS, Kehrl JH. Tumor necrosis factor (TNF)-induced germinal center kinase-related (GCKR) and stress-activated protein kinase (SAPK) activation depends upon the E2/E3 complex Ubc13-Uev1A/TNF receptor-associated factor 2 (TRAF2) J Biol Chem. 2003;278:15429–15434. doi: 10.1074/jbc.M211796200. [DOI] [PubMed] [Google Scholar]
- 35.Habelhah H, et al. Ubiquitination and translocation of TRAF2 is required for activation of JNK but not of p38 or NF-kappaB. EMBO J. 2004;23:322–332. doi: 10.1038/sj.emboj.7600044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lademann U, Kallunki T, Jaattela M. A20 zinc finger protein inhibits TNF-induced apoptosis and stress response early in the signaling cascades and independently of binding to TRAF2 or 14-3-3 proteins. Cell Death Differ. 2001;8:265–272. doi: 10.1038/sj.cdd.4400805. [DOI] [PubMed] [Google Scholar]
- 37.Li X, Yang Y, Ashwell JD. TNF-RII and c-IAP1 mediate ubiquitination and degradation of TRAF2. Nature. 2002;416:345–347. doi: 10.1038/416345a. [DOI] [PubMed] [Google Scholar]
- 38.Baud V, et al. Signaling by proinflammatory cytokines: oligomerization of TRAF2 and TRAF6 is sufficient for JNK and IKK activation and target gene induction via an amino-terminal effector domain. Genes Dev. 1999;13:1297–1308. doi: 10.1101/gad.13.10.1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Habelhah H, et al. Stress-induced decrease in TRAF2 stability is mediated by Siah2. EMBO J. 2002;21:5756–5765. doi: 10.1093/emboj/cdf576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vince JE, et al. TWEAK-FN14 signaling induces lysosomal degradation of a cIAP1- TRAF2 complex to sensitize tumor cells to TNFalpha. J Cell Biol. 2008;182:171–184. doi: 10.1083/jcb.200801010. [DOI] [PMC free article] [PubMed] [Google Scholar]