Abstract
Baicalein and baicalin, the major bioactive compounds found in the Chinese herb Scutellaria baicalensis, have been shown to be effective against cancer, bacterial infections and oxidative stress diseases. However, little is known about their mechanisms of action. To probe whether iron homeostasis modulation may play a role in their bioactivity, we have investigated their iron binding characteristics under physiologically relevant conditions. A 2:1 baicalein-ferrous complex was readily formed in 20 mM phosphate buffer, pH 7.2, with a binding constant ~2-9 × 1011 M-2, whereas a 1:1 baicalein-ferric complex was formed, under the same conditions, with an apparent binding constant ~1-3 × 106 M-1. Baicalein appears to bind the ferrous ion more strongly than ferrozine, a well known iron(II) chelator. Using 1 H NMR and Zn2+ and Ga3+ as probes, the iron-binding site on baicalein was elucidated to be at the O6/O7 oxygen atoms of the A-ring. No binding was observed for baicalin under the same NMR conditions. Furthermore, baicalein strongly inhibits the Fe-promoted Fenton chemistry via a combination of chelation and radical scavenging mechanism while baicalin can provide only partial protection against radical damage. These results indicate that baicalein is a strong iron chelator under physiological conditions and hence may play a vital role in modulating the body's iron homeostasis. Modulation of metal homeostasis and the inhibition of Fenton chemistry may be one of the possible mechanisms for herbal medicine.
Keywords: Iron chelation, Baicalein, Baicalin, Fenton chemistry, Antioxidant
1. Introduction
Herbal medicine has been practiced in China and other countries for centuries, yet little is known about its mechanism of action at the molecular level [1]. Baicalein and baicalin are two of the major bioactive compounds found in the traditional Chinese medicinal herb Baikal skullcap (Scutellaria baicalensis Georgi), known as “Huang qin” in China and “Ogon” in Japan, which has been routinely administered in the treatment of disease-related symptoms such as fever, insomnia, and copious perspiration [2]. Used for centuries in traditional Asian medicine, S. baicalensis Georgi is increasingly being sold for health-promoting purposes in the United States and throughout the Europe [3]. Interestingly, several studies have reported that the active compounds in this herb can be effective against cancer, bacterial infections and oxidative stress diseases [4-6].
In recent years, baicalein and its glycoside baicalin (Fig. 1) have been the subjects of a number of studies that have produced promising results in diverse areas such as antioxidant [7], anti-inflammatory [8], and anticancer activities[9], as well as neuro-fibril disruption [10]. As early as the 1980s, baicalein was observed to inhibit iron-induced lipid peroxidation [11-13]. Similar effects were observed by Hamada et al., where baicalein was observed to be a potent radical scavenger, in rat and gerbil models, protecting neurons from FeCl3-induced epilepsy and ischemia-induced death [14]. More recently, it has been reported that baicalin can control iron overload in mouse models and reduce iron overload-induced liver damage in mouse models [15,16]. Despite these findings, the iron-binding properties of baicalein or baicalin have not been well characterized. And, as it has been widely reported, loosely bound iron in the cellular labile iron pool [17] can react with endogenous hydrogen peroxide to produce the short-lived and highly reactive hydroxyl radical (OH·) through the Fenton reaction (Eq. (1)). These hydroxyl radicals can in turn oxidize nucleic acids, proteins or cell membranes with the ensuing deleterious consequences for the organism.
Fig. 1.
Structures of baicalein (left) and baicalin (right).
Previous work in our laboratory has identified “iron-binding motifs” in plant polyphenolic compounds [18]. Strong iron binding by some phenolic compounds could potentially modulate iron homeostasis in the body and explain the reported bio-effects of plant phenolic compounds [18]. A look at the structures of baicalein and baicalin reveals that both flavonoids contain “iron-binding motifs” and thus are expected to bind iron. Nevertheless, the iron-binding properties and biochemical effects (e.g. promotion of Fenton chemistry) of both baicalein and baicalin under physiological conditions are unknown. The present work reports for the first time the binding of baicalein and baicalin to both Fe2+ and Fe3+ in biological pH and buffer (Note that Fe2+ and Fe3+ in Eq. (1) refer to Fe(II) and Fe(III), respectively.).
| (1) |
2. Materials and methods
2.1. Chemicals
Baicalein, baicalin and ferrozine (3-(2-pyridyl)-5,6-diphenyl-1,2,4-triazine-p-p′-disulfonic acid)) monosodium salt were purchased from Sigma-Aldrich Corp. (St. Louis, MO, USA). Baicalein and baicalin were freshly dissolved in ethanol while ferrozine monosodium salt was dissolved in deionized water. The sources of iron used were ferric chloride (Sigma-Aldrich) and ferrous ammonium sulfate (JT Baker, Philsburg, NJ, USA). Both were prepared fresh daily in 0.1 M HCl. Thiobarbituric acid (TBA) was prepared in a 50 mM NaOH solution and trichloroacetic acid (TCA) (Sigma-Aldrich) was dissolved in water. Double ionized water was used to dilute all other reagents. Potassium phosphate buffer (KPB, 20 mM) was prepared from a 1 M stock solution at pH 7.2. The solutions were kept in a refrigerator at 4 °C when not in use. All other chemicals used were reagent grade.
2.2. Instrumentation
UV/Visible (UV/Vis) spectroscopic studies were conducted in a Perkin Elmer Lambda 25 spectrometer at room temperature (25 °C). Standard 1 cm path length quartz cuvettes were used to hold sample and reference solutions. Electrospray ionization mass spectrometry experiments were conducted in a Sciex API 150 EX (Ontario, Canada) instrument with a Turbo Spray ion source and a nitrogen gas for mobilizations. The vacuum pressure was set at 1.4 Torr and the ion spray voltage was set at 5000 Volts. The sample was introduced at a flow rate of 70 μL min-1 with a Harvard syringe pump (Holliston, MA, USA). Due to the possibility of the formation of salt bridges in the ion optics, potassium buffer was not used as a solvent. Instead, both the baicalein/baicalin and iron solutions were dissolved in a 1:1 methanol: water mixture with 1% acetic acid. All mass spectrometry data were analyzed with the program The Analyst.
1H NMR studies were carried out on a Bruker AC 300 spectrometer. Stock solutions of baicalein (300 mM) and baicalin (300 mM) were freshly prepared in DMSO-d6. Zinc acetate (300 mM) and gallium chloride (600 mM) were prepared in D2O and D2O/HCl, respectively. The titrations were done in a 50/50 (v/v) DMSO-d6/D2O solvent buffered with 50 mM Tris-HCl at pH 7.20. The pH of the solutions was determined with a Corning pH meter adapted with a Sigma-Aldrich micro combination electrode. The pH meter readings for D2O were recorded as pH values, i.e. uncorrected for the effect of deuterium.
2.3. 2-Deoxyribose degradation assays
The 2-deoxyribose degradation assays were performed following the procedure described by Lopes et al. [19], with some modifications in the following manner: in a 2 mL eppendorf tube baicalein/baicalin (or EDTA) and Fe2+ solutions were mixed and let incubating for 40 min. When EDTA was added first, baicalein/baicalin was incubated in the EDTA-Fe2+ system for an additional 30 min. Then phosphate buffer, 2-deoxyribose (1 mM), ascorbic acid (200 μM) and hydrogen peroxide (200 μM) were added in the mentioned order (final concentrations shown in parenthesis). The reaction was stopped after 10 min with 500 μL of 10% thrichloroacetic acid (TCA) and then, to facilitate the observation of the oxidation products, 500 μL of 1% 2-thiobarbaturic (TBA) were added. Lastly, the eppendorf tubes were heated in a dry bath for 15 min at 80 °C. The absorbance of the complex was monitored at 532 nm.
2.4. Studies on Fe2+ competition between ferrozine and baicalein
Ferrozine was mixed with Fe2+ at a 3:1 stoichiometric ratio (60 μM:20 μM) in a 1-cm path length quartz cuvette containing 1 mL of 20 mM phosphate buffer at pH 7.2. Then, 40 μL of baicalein (40 μM) were added to the Ferrozine3-Fe2+ complex after it reached equilibrium (ca. 5 min). The absorbance of this complex was spectrophotometrically monitored each hour for over 6 h.
2.5. Measurements of binding constants
The conditional binding constants (apparent binding constants) for the formation of the baicalein-ferrous and the baicalein-ferric complexes, in 20 mM phosphate buffer at pH 7.2, were measured by UV/Vis spectroscopic methods. This was done first by finding the molar stoichiometry of the complexes formed by the titration of 10 μM baicalein with increasing amounts of Fe2+ or Fe3+ and monitoring the change of absorbance at 440 nm where the complexes absorb while the ligand does not. Two methods were employed to find the binding constants. The first method, based on molar absorptivity, is similar to the one previously described by Gibbs in the determination of the binding constant of the ferrozine3-Fe2+ complex [20]. The second method [21] used was previously described in the determination of the apparent binding constant of the quercetin-Fe2+ complexes [18]. The spectroscopic changes at 364 nm were used for the calculations. The equations are given in Supplementary material.
3. Results and discussion
3.1. UV/Vis studies of the interactions between baicalein and baicalin with Fe2+/Fe3+
Baicalein (10 μM) were titrated firstly with increasing amounts of Fe2+ in 20 mM KPB at pH 7.2. The titration was monitored by UV/Vis (Fig. 2A). Two absorbance bands were observed at 260 nm and 354 nm for the baicalein and are generally ascribed to π → π* electronic transitions from the conjugated 3-ring system. As Fe2+ was added, the baicalein absorbance peaks centered at 260 nm and 354 nm decreased in intensity while the absorbance in the regions between 290 nm and 316 nm and after 402 nm increased in intensity. Three isosbestic points were observed at ca. 287 nm, 315 nm and 402 nm, suggesting clean formation of the baicalein-Fe2+ complex. The spectroscopic changes upon iron-binding may be attributed to the further delocalization of π electrons of the conjugated ring system of baicalein over the metal, as discussed similarly for the quercetin-Fe complexes [18]. The titration curve (Fig. 2A inset) suggests a 2:1 molar ratio of baicalein to Fe2+ in the complex. The same stoichiometry was obtained by analyzing the spectroscopic changes at the 260 nm and 354 nm peaks. The complexation reaction was completed within a minute under the conditions applied.
Fig. 2.

Spectrophotometric titration of 10 μM baicalein with (A) Fe2+ (1-10 μM in 1 μM increments, and finally 2 μM to a final concentration of 12 μM) and 11.7 μM baicalein with (B) Fe3+ (2-20 μM in 2 μM increments, and finally 5 μM to a final concentration of 25 μM) in 20 mM KPB, pH 7.2. Insets: absorbance of the baicalein-iron complex at 440 nm as a function of the concentration of (A) Fe2+ and (B) Fe3+.
A similar titration with Fe3+ was carried out. The spectra for this titration are shown in Fig. 2B. The binding with Fe3+ appeared kinetically slower than that with Fe2+, as the titration took ca. 2 min to reach equilibrium. This may be ascribed to a greater interaction of Fe3+ with the phosphate ions of the buffer, which compete for binding [22]. The overall spectroscopic changes upon Fe3+-binding were similar to those of Fe2+-binding but the isosbestic points were at slightly different positions (285 nm, 320 nm and 406 nm) and the intensity increase at wavelengths >406 nm was much smaller (ε440 = 4100 M-1 cm-1) than that of Fe2+ (ε440 = 8600 M-1 cm-1). The ε values are smaller than those of the quercetin-Fe complexes, but they are in the same order of magnitude [18,22]. The major difference observed in the titration curve (Fig. 2B inset) with Fe3+ is the saturation point, which suggests the formation of a 1:1 complex between baicalein and Fe3+. The same stoichiometry was obtained by analyzing spectroscopic changes at the other peaks in the UV region as well as by a Job analysis (data not shown).
Similar titrations with baicalin were attempted but the data interpretation was hampered by the intrinsic instability and degradation of baicalin in the system. It has been reported [23] recently that baicalin is very unstable in aqueous solution. It degrades rapidly at neutral or basic pH, via possibly a phenoxyl radical and hydrolysis pathway, to finally produce a quinone form of baicalein, accompanied by the release of the sugar group [23]. The mixture of baicalin, its degraded species, and, possibly, their iron-adducts, coupled with the complex dynamic processes of the species prevent us from a clear interpretation of the UV/Vis spectra.
3.2. Electrospray ionization mass spectrometry (ESI-MS) studies
Further studies on the complexation of iron by baicalein were carried out in a mass spectrometer equipped with an ion-spray ionization source. A 1:1 (v/v) mixture of water and methanol, with 1% acetic acid to facilitate ionization, was used as the solvent. Fig. 3 shows the mass spectra for a mixture of 10 μM of baicalein with 10 μM of freshly prepared Fe2+ or Fe3+ solution. In both cases, the protonated ligand, [baicalein + H]+, was observed at m/z = 271.7 (or 271.8), and species corresponding to the formation of a 2:1 complex between baicalein and iron were observed in each case: a baicalein2-Fe2+ complex (m/z = 595.2, [FeII(B2-H)]+, Fig. 3A) when Fe2+ was used and a baicalein2-Fe3+ complex (m/z = 594.3, [FeIII(B-H)2]+, Fig. 3B) when Fe3+ was used. A close look at the isotopic pattern of the baicalein-Fe complex suggests it fits well the isotopic distribution of iron. The formation of a baicalein2-Fe3+ complex is probably favored by the acidic conditions used in the ESI-MS study. This observation was corroborated by a spectrophotometric titration of 10 μM baicalein with Fe3+ (2-20 μM in 2 μM increments) in 30 mM NaAc buffer, pH 4.5 (Fig. S1 in Supplementary material).
Fig. 3.

Electrospray ionization mass spectra of 10 μM baicalein with (A) 10 μM Fe2+ and (B) 10 μM Fe3+ in 1:1 methanol:water (v/v, with 1% acetic acid).
3.3. Measurements of the binding affinity of baicalein with iron
The conditional binding constants of baicalein with Fe2+ and Fe3+ were studied in 20 mM KPB, pH 7.2 at 298 K, described in the experimental section. It was estimated from method 1 that the apparent binding constant for the (baicalein)2-Fe2+ complex is ~9 × 1011 M-2 and the apparent binding constant for the baicalein-Fe3+ complex is ~3 × 106 M-1, while the corresponding values from method 2 were ~2 × 1011 M-2 and ~1 × 106 M-1. The values obtained by both methods are in good agreement and the differences are within a factor of ca. 4. Under similar conditions, it appears that baicalein binds Fe2+ one order of magnitude higher than quercetin does (5 × 1010 M-2, 2:1 complex) [18], suggesting that baicalein may be more effective than quercetin in modulating iron homeostasis under physiological conditions.
3.4. Competition for Fe2+ between baicalein and ferrozine
The above binding constant data suggest a strong binding between Fe2+ and baicalein under physiologically relevant conditions. In order to verify this strong binding, competition experiments between baicalein and a well known Fe2+-chelator, ferrozine (K (β3) = 3.65 × 1015 [20]), were further carried out in 20 mM KBP buffer (pH 7.2) at 298 K. Fig. 4 shows the absorbance changes in time of the ferrozine3-Fe2+ complex (20 μM) after the addition of a stoichiometric amount of baicalein. During the first hour, little change was observed for the Fe-ferrozine peak at 562 nm, but an increase in intensity at ca. 400-450 nm was observed, possibly due to the formation of a hybrid ferrozine-Fe2+-baicalein complex. Similar hybrid iron complexes have been reported with amino acids and ferrozine [24]. After the first hour, a steady decline in the absorbance of ferrozine3-Fe2+ peak (562 nm) coupled with a simultaneous increase in the 400-450 nm absorbance region was observed. This spectral change suggests that the formation of the baicalein-Fe2+ complex is concurrent with the dissociation of the ferrozine3-Fe2+ complex. This experiment clearly demonstrates that baicalein strongly binds Fe2+ under physiological relevant conditions and that the binding is stronger than the well known Fe2+-chelator ferrozine. This observation is consistent with the calculated binding constants.
Fig. 4.

Competitive Fe2+ binding study between ferrozine and baicalein mixed at stoichiometric ratios, 3 mol equiv. of ferrozine, 1 mol equiv. of Fe2+, and 2 mol equiv. of baicalein. (a) UV/Vis spectra of 60 μM ferrozine in 1 mL of 20 mM phosphate buffer at pH 7.2. (b) red line, addition of Fe2+ (20 μM), (c) green line: addition of baicalein (40 μM). Lines (c) to (i): hourly scans for 6 h. (For interpretation of the references in colour in this figure legend, the reader is referred to the web version of this article.)
3.5. 1H NMR studies of the metal binding site
1H NMR titration studies were performed to identify the likely site of iron binding on baicalein. Diamagnetic metal ions Zn2+ and Ga3+ were used as probes for Fe2+ and Fe3+, respectively, as the iron complexes were not detectable by NMR. All studies were done in a 50/50 (v/v) solvent of DMSO-d6/D2O-Tris-HCl (50 mM, pH 7.2). The 1H NMR peak assignments for baicalein were based on those reported by Lim and coworkers [25].
The 1H NMR spectra of the titration of 5 mM baicalein with Zn2+ was shown in Fig. 5. As Zn2+ was added, little change was observed for most of the baicalein peaks; however the H8 proton experienced a large change by shifting downfield (ca. 0.17 ppm). The H3 proton peak also slightly shifted downfield (ca. 0.02 ppm). At 1 mol equiv. Zn2+ added (Fig. 5d), a precipitate formed thus lower intensity of the signals was observed. This NMR change suggests that the metal-binding site is close to the H8 proton, i.e. the O7 and O6 site on the A-ring. This binding site was further confirmed by similar experiments performed by using baicalin, which has the O7 site blocked by a glucose unit (Fig. 1). No NMR change was observed after the addition of Zn2+ into baicalin (Supplementary material, Figs. S2-S4), confirming that the O7 site is critical for Zn2+ binding.
Fig. 5.
1H NMR titration of 5 mM baicalein (a) with (b)¼, (c) ½, and (d) 1 mol equiv. of Zn2+ in 50 mM Tris-HCl, pH 7.2 (50/50, v/v, DMSO-d6/D2O-Tris-HCl).
The titration of baicalein with Ga3+ showed similar 1H NMR shifts compared to those seen with Zn2+ (Fig. 6), i.e., H3 had a slight downfield shift (ca. 0.06 ppm) while H8 had a large downfield shift (ca. 0.16 ppm), suggesting that Ga3+ binds the same site as Zn2+. However, as more Ga3+ was added (beyond 1 mol equiv.), new NMR peaks appeared, indicating the formation of multiple species. No effort was made to identify these species.
Fig. 6.
1H NMR of 5 mM baicalein (a) and after the addition of ½ mol equiv. Ga3+ (b) in 50 mM Tris-HCl, pH 7.2 (50/50, v/v, DMSO-d6/D2O-Tris-HCl).
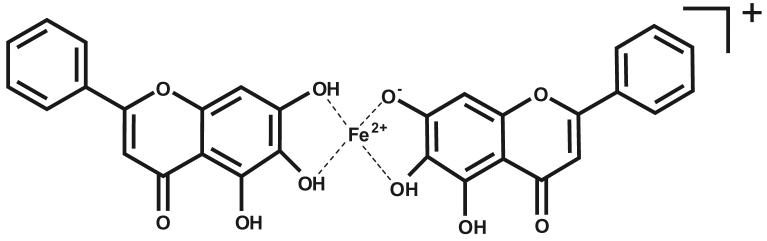
Based on the evidence from our NMR studies, it can be concluded that the metal is located between the hydroxyl groups at carbons 6 and 7 in the Zn- or Ga-baicalein complexes. Due to the similar coordination properties of Fe2+ and Zn2+, as well as Fe3+ and Ga3+, and considering the results from the UV/Vis and ESI-MS studies, it can be suggested that both Fe2+ and Fe3+ bind baicalein at the same site as Zn2+or Ga3+ does, i.e., the O6 and O7 site on the A-ring. Fig. 7 shows the proposed structure for the baicalein-Fe2+ species.
Fig. 7.
Proposed structure of the baicalein2-Fe2+ complex (m/z = 595.3) observed under the described ESI-MS conditions.
3.6. Inhibition of the Fenton chemistry by baicalein and baicalin
2-Deoxyribose degradation assays were performed to assess the capability of baicalein and baicalin to inhibit the formation of hydroxyl radicals promoted by Fenton reaction. Fig. 8A shows the absorbance (average of triplicate) at 532 nm of the malonaldehyde-TBA complex as a function of the concentration of Fe2+ in the absence of the flavones (a), in the presence of 10 μM baicalin (b), and 10 μM baicalein (c). It is clearly seen that baicalein can completely inhibit Fenton-induced radical damage even at Fe2+ concentrations up to 20 μM. At higher concentrations of Fe2+ up to 50 μM, the curve rose a bit but the inhibition is still significant (Fig. S5 in Supplementary material). Under the same conditions, baicalin can only partially protect the 2-deoxyribose molecule from Fenton radical damage. As demonstrated by the NMR study, baicalin does not bind iron strongly; the partial radical damage protection of baicalin is likely due to its radical scavenging activity [26].
Fig. 8.

Absorbance at 532 nm of 2-deoxyribose degradation assays in the presence and absence of baicalein, baicalin, and EDTA with error bars shown (n = 3). (A): (a) Fe2+ (0 to 20 μM) only; (b) with 10 μM baicalin; and (c) with 10 μM baicalein. (B): (a) Fe2+ with equimolar EDTA (0-20 μM) in the absence of baicalein; (b) after the addition of 10 μM baicalein; and (c) 10 μM of baicalein in the absence of EDTA.
To clarify the mechanism by which baicalein can inhibit Fenton-induced radical damage (acts as an antioxidant), i.e., whether it is by iron chelation or radical scavenging, Fe2+-EDTA system was used to promote Fenton chemistry. Since EDTA has an iron binding constant several orders of magnitude higher than that of baicalein, baicalein can only act as a radical scavenger in the Fe-EDTA system. Fig. 8B shows that in the presence of EDTA, Fe2+ can still generate radicals (a), although not at the same rate as when Fe2+ is unchelated. In the presence of baicalein, a lesser radical-mediated damage was observed (b), which is expected for a radical scavenger. Line (c) in Fig. 8B shows that when baicalein acted as a chelator (without EDTA in the system), no radical-mediated damage was observed. This indicates that chelating iron is the key to baicalein's inhibition of Fenton chemistry-induced radical damage. Thus, baicalein can be a more potent antioxidant by chelating Fe2+ than by scavenging radicals.
Our data (Fig. 8 and Fig. S5) demonstrate that baicalein can inhibit iron-promoted Fenton chemistry beyond the stoichiometries of the formed complexes. A similar phenomenon has also been observed for quercetin [18]. The anti-Fenton activity may be due to a combined effect of chelation and radical scavenging activities of baicalein (or quercetin) and its Fe-complexes. In addition, the baicalein-Fe(II/III) complexes could catalytically scavenge radicals in the sysytem via a mechanism similar to the one proposed by Zhao et al. for the Fe-tannin system [27].
The strong chelation of iron by baicalein, shown by our results, appears to be in general agreement with the observations that flavonoids with an “iron-binding motif” [18] can chelate iron under a number different of conditions such as buffer, pH and solvent [28-30]. Among the three possible metal-binding sites on baicalein, the site comprised by the two hydroxyl groups at the 6 and 7 positions on the A-ring appears to be the strongest site for both Fe2+ and Fe3+ (also Zn2+ and Ga3+). A recent NMR and DFT study has demonstrated that a strong intermolecular H-bond (C5-OH⋯O=C4) is formed between the 4-carbonyl and the 5-OH group in both baicalein and baicalin [31]. This H-bond may prevent metal-binding at the 4-carbonyl and the 5-OH positions. Furthermore, in baicalin, the 7-OH position was blocked by a glucose group. This may explain the apparent non interaction between baicalin and Zn2+ in our NMR study. However, this does not exclude a possible binding under alternative conditions. For example, Dong et al. [5] reported that a solid 2:1 baicalin-Fe2+ complex was isolated (yield 53%) after refluxing baicalin and FeCl2 for 6 h, at 65 °C, in a basic solution (pH 9) of ascorbic acid and ethanol. However, as has been discussed in this paper, the intrinsic instability and rapid degradation of baicalin in aqueous media [23] prevents us from performing a detailed solution study on the iron-baicalin interactions in KBP buffer. The sugar moiety of baicalin could also possibly bind the metal via the hydroxyl or carbonyl groups, however, this kind of binding was not observed in our NMR study (Figs. S3 and S4 in Supplementary material). This observation is in general agreement with what Gyurcsik and Nagy concluded [32]; metal-sugar binding strength is so weak in neutral or acidic aqueous solution that the sugar molecules do not readily replace the water molecules in the first coordination sphere of the metal ions.
Recent in vivo and in vitro studies have found that the supplementation baicalein and its glycoside baicalin have multiple bio-effects. Moreover, the protecting effects on organs like heart, liver and brain have been linked to their antioxidant activities [6,15,33]. Correspondingly, quite a few diseases including arteriosclerosis, diabetes, cancer, Parkinson's disease and Alzheimer's disease have been linked to an increased radical production inside the body [34,35]. Therefore, mitigating the production of endogenous radical species could be essential to prolonging the well-being of human beings. The evidence presented here argues that baicalein can inhibit the production of endogenous hydroxyl radicals produced through the Fenton reaction by forming stable and inert complexes with iron. The 2-deoxyribose degradation assays clearly show that the bulk of baicalein's antioxidant activity is derived from its iron-chelation property rather than its radical scavenging activity. The apparent binding constants of baicalein with respect to Fe2+ and Fe3+ indicate that baicalein could be a strong iron chelator under physiological conditions and thus may modulate iron homeostasis in the body. The NMR studies conducted here identified the metal binding site as the dihydroxyl moiety at carbons 6 and 7 on the A-ring. On the other hand, although baicalin appears not to bind iron strongly under the conditions described here, it can be converted to baicalein by betaglucoronidase, an enterobacterial enzyme, inside the body [36].
The biological role of an iron chelator in living organisms affected by oxidative stress was recently investigated by Imlay et al. [37]. In this study, the addition of a cell-permeable iron chelator to bacteria lacking three H2O2 scavenging enzymes caused rapid DNA damage and eventual death under aerobic conditions [30]. Baicalein could inhibit the Fenton reaction in vivo by the same mechanism. Additionally, electrovoltametry experiments performed by Cheng et al. showed that baicalein can partially inhibit the Fenton reaction in the presence of iron chelator ATP [38], offering further evidence that baicalein can deactivate iron, stopping the generation of hydroxyl radicals.
Plant phenolic compounds have been shown to be readily absorbed by the body and distributed to key organs including the liver and the brain [39]. Underscoring the possible importance of baicalein's iron affinity, a bioavailability study [40] of orally administered “Shosaiko-to” (a seven-herb formulation that contains “Huang qin” (S. baicalensis)) extracts did not show baicalin in the plasma, however, the plasma showed baicalein at concentrations as high as 0.62 μg mL-1 (ca. 2.3 μM) up to 12 h after administrating the extracts. Moreover, 0.3 μg mL-1 (ca. 1.1 μM) of baicalein were still detectable in the plasma 24 h after administration [33]. Lee Chao and coworkers reported that baicalein metabolites have an apparent 8 h elimination half life, a long time for a metabolite, and that baicalein is more rapidly absorbed than baicalin [41]. Consequently, once inside the body, baicalein can stay long enough to affect the body's iron homeostasis and thus could reduce oxidative stress damage within the cell. The findings described here may have a real impact on the understanding of the mechanisms of action of baicalein and S. baicalensis Georgi. Abnormal accumulation of iron and other metals in the body, as well as redox metal-promoted oxidative stress, have been linked to a large number of diseases [42-44]. Thus, compounds containing iron-binding motifs, found in many medicinal herbs [45], could potentially modulate metal homeostasis and inhibit Fenton chemistry, serving as possible mechanisms of action for the medicinal effects of these herbs.
Supplementary Material
Acknowledgements
Financial support from the National Institutes of Health and University of Massachusetts Dartmouth is greatly acknowledged. This publication/project was made possible by Grant 1 R21 AT002743-02 from the National Center for Complementary and Alternative Medicine (NCCAM). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NCCAM, or the National Institutes of Health. We thank Drs. C. Neto and S. Cai (UMass Dartmouth, MA, USA) for helpful discussions. We thank the anonymous reviewers for their insightful comments.
Footnotes
Appendix A. Supplementary material Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.jinorgbio.2008.11.003.
References
- [1].Stone R. Science. 2008;309:709–710. doi: 10.1126/science.319.5864.709. [DOI] [PubMed] [Google Scholar]
- [2].van Loon IM. Alt. Med. Rev. 1997;2:472–480. [Google Scholar]
- [3].Chen S, Ruan Q, Bednert E, Deptala A, Wang X. Cell Prolif. 2001;34:2933–2934. doi: 10.1046/j.0960-7722.2001.00213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Scheck AC, Perry K, Hank NC, Clark WD. BMC Complement. Altern. Med. 2006;27(6) doi: 10.1186/1472-6882-6-27. doi:10.1186/1472-6882-6-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Dong H, Bi P, Li S. Chem. Nat. Comp. 2005;41:158–161. [Google Scholar]
- [6].Gao Z, Huang K, Xu H. Pharmacol. Res. 2001;43:173–178. doi: 10.1006/phrs.2000.0761. [DOI] [PubMed] [Google Scholar]
- [7].Shao Hoek Z, Vanden Hoek TL, Quin Y, Becker LB, Schumacker PT, Li C, Dey L, Barth E, Halperin H, Rosen GM, Yuan C. Am. J. Physiol. Heart Circ. Physiol. 2002;282:H999–H1006. doi: 10.1152/ajpheart.00163.2001. [DOI] [PubMed] [Google Scholar]
- [8].Chou TC, Chang LP, Li CY, Wong CS, Yang SP. Anesth. Analg. 2003;97(6):1724–1729. doi: 10.1213/01.ANE.0000087066.71572.3F. [DOI] [PubMed] [Google Scholar]
- [9].Miocinovic R, McCabe NP, Keck RW, Jankun J, Hampton JA, Selman SH. Int. J. Oncol. 2005;26:241–246. [PubMed] [Google Scholar]
- [10].Zhu M, Rajamani S, Kaylor J, Hans S, Zhou F, Fink AL. J. Biol. Chem. 2004;279:26846–26857. doi: 10.1074/jbc.M403129200. [DOI] [PubMed] [Google Scholar]
- [11].Kimuya Y, Kubo M, Tani T, Arichi S, Okuda H. Chem. Pharmacol. Bull. 1981;29:2610–2617. doi: 10.1248/cpb.29.2610. [DOI] [PubMed] [Google Scholar]
- [12].Gao DY, Sakurai K, Katoh M, Chen JM, Ogiso T. Biochem. Mol. Biol. Int. 1996;39:215–225. doi: 10.1080/15216549600201221. [DOI] [PubMed] [Google Scholar]
- [13].Yoshino M, Murakami K. Anal. Biochem. 1998;257:40–44. doi: 10.1006/abio.1997.2522. [DOI] [PubMed] [Google Scholar]
- [14].Hamada H, Hiramatsu M, Edamatsu R, Mori A. Arch. Biochem. Biophys. 1993;306:261–266. doi: 10.1006/abbi.1993.1509. [DOI] [PubMed] [Google Scholar]
- [15].Zhao Y, Li H, Gao Z, Xu H. Eur. J. Pharmacol. 2005;509:195–200. doi: 10.1016/j.ejphar.2004.11.060. [DOI] [PubMed] [Google Scholar]
- [16].Zhang Y, Li H, Gao Z. Eur. J. Pharmacol. 2006;535:263–269. doi: 10.1016/j.ejphar.2006.01.067. [DOI] [PubMed] [Google Scholar]
- [17].Sergent O, Tomasi A, Ceccarelli D, Masini A, Nohl H, Cillard P, Cillard J, Vladirimov YA, Kozlov AV. Biometals. 2005;18:567–575. doi: 10.1007/s10534-005-8488-7. [DOI] [PubMed] [Google Scholar]
- [18].Guo M, Perez CA, Wei Y, Rapoza E, Su G, Bou-Abdallah F, Chasteen ND. Dalton Trans. 2007:4951–4961. doi: 10.1039/b705136k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Lopes GKB, Schulman HM, Hermes-Lima M. Biochim. Biophys. Acta. 1999;1472:142–152. doi: 10.1016/s0304-4165(99)00117-8. [DOI] [PubMed] [Google Scholar]
- [20].Gibbs CR. Anal. Chem. 1976;48:1197–1201. [Google Scholar]
- [21].Ojadi E. Ph.D. Thesis. Brandeis University; Boston, MA, USA: 1986. [Google Scholar]
- [22].Hajji H, Nkhili E, Tomao V, Dangles O. Free Rad. Res. 2006;40:303–320. doi: 10.1080/10715760500484351. [DOI] [PubMed] [Google Scholar]
- [23].Xing J, Chen X, Zhong D. J. Pharmacol. Biomed. Anal. 2005;39:593–600. doi: 10.1016/j.jpba.2005.03.034. [DOI] [PubMed] [Google Scholar]
- [24].Berlett BS, Levine RL, Chock PB, Chevion M, Stadtman ER. Proc. Natl. Acad. Sci. USA. 2001;98:451–456. doi: 10.1073/pnas.021552298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Moon B, Lee Y, Ahn J, Lim Y. Y. Mag. Res. Chem. 2005;43:858–860. doi: 10.1002/mrc.1622. [DOI] [PubMed] [Google Scholar]
- [26].Gao Z, Yang X, Huang H, Xu H. Appl. Magnet. Res. 2000;19:35–44. [Google Scholar]
- [27].Zhao ZS, Khan S, O'Brien PJ. Biolchem. Pharmacol. 1998;56:825–830. doi: 10.1016/s0006-2952(98)00222-6. [DOI] [PubMed] [Google Scholar]
- [28].Engleman MD, Hutcheson R, Cheng IF. J. Agric. Food. Chem. 2005;53:2953–2960. doi: 10.1021/jf048298q. [DOI] [PubMed] [Google Scholar]
- [29].Botelho FV, Alvarez-Leite JI, Lemos VS, Pimenta AMC, Calado HDR, Matencio T, Miranda CT, Pereira-Maia EC. J. Inorg. Biochem. 2007;101:935–943. doi: 10.1016/j.jinorgbio.2007.03.007. [DOI] [PubMed] [Google Scholar]
- [30].Bodini ME, Copia G, Tapia R, Leighton F, Herrera L. Polyhedron. 1999;18:2233–2239. [Google Scholar]
- [31].Wolniak M, Oszmianskib J, Wawera I. Magn. Reson. Chem. 2008;46:215–225. doi: 10.1002/mrc.2165. [DOI] [PubMed] [Google Scholar]
- [32].Gyurcsik B, Nagy L. Coord. Chem. Rev. 2000;203:81–149. [Google Scholar]
- [33].Lee HJ, Noh YH, Lee DY, Kim YS, Kim KY, Chung YH, Lee WB, Kim SS. Eur. J. Cell Biol. 2005;84:897–905. doi: 10.1016/j.ejcb.2005.07.003. [DOI] [PubMed] [Google Scholar]
- [34].Harborne JB, Williams CA. Phytochemistry. 2000;50:481–504. doi: 10.1016/s0031-9422(00)00235-1. [DOI] [PubMed] [Google Scholar]
- [35].Schmitt-Schilling S, Schaffer S, Webber CC, Ecker GP, Müler WE. J. Physiol. Pharmacol. 2005;56:23–36. [PubMed] [Google Scholar]
- [36].Muto R, Motozuka T, Nakano M, Tatsumi Y, Sakamoto F, Kosaka N. J. Pharma. Soc. Jpn. 1998;118:79. doi: 10.1248/yakushi1947.118.3_79. [DOI] [PubMed] [Google Scholar]
- [37].Park S, You X, Imlay JA. Proc. Natl. Acad. Sci. USA. 2005;102:9317–9322. doi: 10.1073/pnas.0502051102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Cheng IF, Breen K. Biometals. 2000;13:77–83. doi: 10.1023/a:1009229429250. [DOI] [PubMed] [Google Scholar]
- [39].Spencer JPE, Mohsen MMA, Rice-Evans C. Arch. Biochem. Biophys. 2004;423:148–161. doi: 10.1016/j.abb.2003.11.010. [DOI] [PubMed] [Google Scholar]
- [40].Nishioka Y, Kyotani S, Miyamura M, Kusunose M. Chem. Pharmacol. Bull. 1992;40:1335–1337. doi: 10.1248/cpb.40.1335. [DOI] [PubMed] [Google Scholar]
- [41].Lai M, Hsiu S, Chen C, Hou Y, Lee Chao P. Biol. Pharmacol. Bull. 2003;26:79–83. doi: 10.1248/bpb.26.79. [DOI] [PubMed] [Google Scholar]
- [42].Richardson DR. NY Ann. Acad. Sci. 2004;1012:326–341. doi: 10.1196/annals.1306.026. [DOI] [PubMed] [Google Scholar]
- [43].Wei Y, Guo M. Angew. Chem. Int. Ed. 2007;46:4722–4725. doi: 10.1002/anie.200604859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Ghosh S, Mukherjee A, Sadler PJ, Verma S. Angew. Chem. Int. Ed. 2008;47:2217–2221. doi: 10.1002/anie.200705723. [DOI] [PubMed] [Google Scholar]
- [45].Perez CA, Tong Y, Guo M. Curr. Bioactive Comp. 2008;4:150–158. doi: 10.2174/157340708786305952. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.