Abstract
We prospectively determined the variability in radiologists' interpretation of screening mammograms and assessed the influence of type and number of readers on screening outcome. Twenty-one screening mammography radiographers and eight screening radiologists participated. A total of 106 093 screening mammograms were double-read by two radiographers and, in turn, by two radiologists. Initially, radiologists were blinded to the referral opinion of the radiographers. A woman was referred if she was considered positive at radiologist double-reading with consensus interpretation or referred after radiologist review of positive cases at radiographer double-reading. During 2-year follow-up, clinical data, breast imaging reports, biopsy results and breast surgery reports were collected of all women with a positive screening result from any reader. Single radiologist reading (I) resulted in a mean cancer detection rate of 4.64 per 1000 screens (95% confidence intervals (CI)=4.23–5.05) with individual variations from 3.44 (95% CI=2.30–4.58) to 5.04 (95% CI=3.81–6.27), and a sensitivity of 63.9% (95% CI=60.5–67.3), ranging from 51.5% (95% CI=39.6–63.3) to 75.0% (95% CI=65.3–84.7). Sensitivity at non-blinded, radiologist double-reading (II), radiologist double-reading followed by radiologist review of positive cases at radiographer double-reading (III), triple reading by one radiologist and two radiographers with referral of all positive readings (IV) and quadruple reading by two radiologists and two radiographers with referral of all positive readings (V) were as follows: 68.6% (95% CI=65.3–71.9) (II); 73.2% (95% CI=70.1–76.4) (III); 75.2% (95% CI=72.1–78.2) (IV), and 76.9% (95% CI=73.9–79.9) (V). We conclude that screener performance significantly varied at single-reading. Double-reading increased sensitivity by a relative 7.3%. When there is a shortage of screening radiologists, triple reading by one radiologist and two radiographers may replace radiologist double-reading.
Keywords: mammography, screening, radiologist, radiographer, inter-observer
Mammography screening significantly reduces breast cancer mortality (Otto et al, 2003; Tabar et al, 2003; Berry et al, 2005). Compared with single-reading, double interpretation of screening mammograms improves cancer detection rates by 6–15% (Brown et al, 1996; Harvey et al, 2003; Gur et al, 2004; Ciatto et al, 2005). Double interpretation can be performed in several ways. The second radiologist may be blinded to the first interpretation (i.e., blinded double-reading) or not (i.e., independent or non-blinded double-reading). Moreover, screening programmes apply variable methods for resolving reader disagreements. A woman may be recalled if only one reader considers the mammogram abnormal, without discussion of disagreement between readers; mammograms may be interpreted in consensus, in which recall occurs only with agreement of the radiologists involved; or a decision on reader disagreement may be obtained by panel arbitration. Alternatives for double interpretation by a second radiologist include second reading by a mammography radiographer or computer-aided detection devices (Pauli et al, 1996; Tonita et al, 1999; Gilbert et al, 2006, 2008).
In the Netherlands, a nation-wide programme provides biennial screening mammography for women aged 50–75 years. All mammograms are double-read in a non-blinded manner. In case of a discrepant reading, the two radiologists discuss the case together to reach consensus about referral.
One previous field study found a substantial variability in mammography interpretation among radiologists, which was responsible for potential delays in breast cancer diagnosis (Gur et al, 2004). In many studies, mammography test sets that may not adequately represent the actual screening setting are used, and data from real-world practice are therefore sparse (Elmore et al, 1994; Kerlikowske et al, 1998; Rutter and Taplin, 2000; Esserman et al, 2002). We therefore prospectively determined the variability in radiologists' interpretation of screening mammograms and assessed the influence of additional reading by radiologists and radiographers on mammography screening outcome at 2-year follow-up.
Materials and methods
Study population
We included all 106 093 mammograms of women aged 50–75 years who underwent biennial screening mammography at two specialized, analogue screening units in the southern Netherlands between 1 January 2003 and 1 July 2006. All women had given written informed consent to use their screening and follow-up data for evaluation purposes. According to the Dutch Central Committee on Research involving Human Subjects (CCMO), approval by our local Institutional Review Board was not required.
Screening procedure and mammogram readers
Details of the nation-wide screening programme and double-reading procedure by radiographers and radiologists have been described previously (Fracheboud et al, 2001; Duijm et al, 2004a, 2007). In brief, all 21 radiographers and 8 screening radiologists of the two units participated. Experience in screening mammography ranged from 1 to 124 months (mean, 69 months; median, 74 months) for radiographers, and from 39 to 95 months (mean, 79 months; median, 94 months) for radiologists, respectively. All radiologists read more than 6500 screening mammograms annually, and two radiologists are dedicated breast radiologists, who analyse the screening results and supervise quality assurance sessions with the other screening radiologists and radiographers.
Two radiographers double-read each mammogram at the screening site immediately after the examination was completed. At subsequent screening examinations, the radiographers could view previous screening mammograms. The radiographers decided for each mammogram whether additional work-up was required (i.e., whether the mammogram was positive). For each positive mammogram, the radiographers recorded the woman's name and date of birth, the date of screening, and the mammographic findings on a form that was developed for this study. Mammographic findings were classified according to one of five categories of abnormal findings: suspicious high density (e.g., spiculated density or density with indistinct borders), suspicious microcalcifications (e.g., pleomorphic, branching, or amorphous/indistinct microcalcifications), high density in combination with microcalcifications, architectural distortion, or breast parenchyma asymmetry. The mammograms were then double-read by two radiologists, who were blinded to the referral opinion of the radiographers. The second reader was not blinded to the opinion of the first, but aimed not to see it before making his own decision. For each discrepant reading, the second reader recorded the woman's name and date of birth, the date of screening, and the assessment of both radiologists on a form that was developed for this study. The two screening radiologists then tried to reach consensus whether referral of the woman with a discrepant reading was indicated.
Referral
A woman was referred for additional workup (primary referral) if the mammogram was considered positive by both radiologists after initial double-reading or, in the case of a discrepant reading, if at least one radiologist considered referral necessary after consensus discussion. During monthly quality assurance sessions, mammograms that the radiographers had considered positive but that had not been referred by the radiologists were reviewed by two screening radiologists, who were now informed about the mammographic abnormalities detected by the radiographers. A woman was referred if, on review, at least one of the radiologists considered work-up to be essential (secondary referral).
Screening follow-up
The follow-up period included the time through the next screening round, with a screening interval of approximately 2 years. For all women with a screening mammogram that was considered positive by at least one of the radiologists or radiographers, we collected data on diagnostic procedures undertaken, breast cancer diagnosis, histopathology, and TNM (tumour–node–metastases) classification (UICC, 1987) to identify screen-detected cancers. Procedures for the detection of interval cancers (interval cancers are breast cancers that are diagnosed in women after a negative screening examination) have been described previously (Duijm et al, 2004b). To determine whether an interval cancer could potentially have been a screen-detected cancer if all positive radiographer readings had been referred, we investigated if the mammographic abnormalities on the diagnostic films corresponded to any abnormalities registered by the radiographers at screening.
Quality assurance
Throughout the study period, the radiologists reviewed breast cancer cases that were detected after secondary referral, as well as interval cancers. Every month, radiographers attended quality assurance sessions under the supervision of a breast radiologist. Together, they reviewed breast cancers that had been detected by radiologists only.
Statistical analysis
Main outcome measures were referral rate, cancer detection rate (CDR, defined as the number of cancers detected per 1000 women screened), sensitivity and specificity of mammography screening, positive predictive value (PPV) of referral and tumour stages of screen detected cancers at different screening strategies. 95% confidence intervals (CIs) were calculated. Distribution of outcome variables across radiologists and reading strategies was tested with the χ2 test; a P-value of less than 0.05 was considered to indicate statistical significance. Pearson's correlation coefficient was calculated for correlations between reader experience, referral rate, and cancer detection rate. All data were entered into an Excel spreadsheet (Microsoft, Redmond, WA, USA) and statistical analyses were performed using SAS V9.12 (SAS Institute Inc, Cary, NC, USA).
Results
Single-reading by radiologists
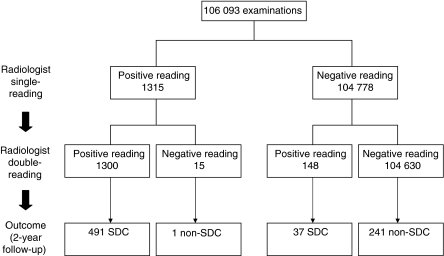
Of the 106 093 screens, 11 491 (10.8%) were initial (prevalent) screens and 94 602 (89.2%) were subsequent (incident) screens. Single-reading by radiologists would have resulted in 1315 referrals (referral rate 1.24%, 95% CI=1.17–1.31) (Figure 1). They would have detected a total of 492 cancers, corresponding to a breast cancer detection rate of 4.64 per 1000 screened women (95% CI=4.23–5.05). The sensitivity would have been 63.9% (95% CI=60.5–67.3), the specificity 99.2% (95% CI=99.2–99.3), and the PPV of referral 37.4% (95% CI=34.8–40.0) (Table 1).
Figure 1.
Radiologist single-reading vs radiologist double-reading: mammography-screening outcome at 2-year follow-up. At radiologist double-reading, a woman was referred for additional work-up if the mammogram was considered to be positive by both radiologists or, in the case of discrepant readings, if at least one radiologist considered referral necessary after consensus meeting. SDC=screen-detected cancer.
Table 1. Inter-observer variability at single reading by eight radiologists (A–H): mammography screening outcome at 2-year follow-up.
| A | B | C | D | E | F | G | H | Total | |
|---|---|---|---|---|---|---|---|---|---|
| Total readings, no. (%) | 23 872 (11.3) | 28 244 (13.3) | 26 874 (12.7) | 26 754 (12.6) | 26 101 (12.3) | 28 317 (13.3) | 25 541 (12.0) | 26 483 (12.5) | 212 186 |
| 1st reading, no. (%) | 10 178 (42.6) | 11 614 (41.1) | 12 708 (47.3) | 11 403 (42.6) | 14 493 (55.5) | 16 467 (58.2) | 15 696 (61.5) | 13 534 (51.1) | 106 093 |
| 2nd reading, no. (%) | 13 694 (57.4) | 16 630 (58.9) | 14 166 (52.7) | 15 351 (57.4) | 11 608 (44.5) | 11 850 (41.8) | 9845 (38.5) | 12 949 (48.9) | 106 093 |
| No. of referrals (%) | 96 (0.9) | 147 (1.3) | 141 (1.1) | 158 (1.4) | 224 (1.5) | 188 (1.1) | 200 (1.3) | 161 (1.2) | 1315 (1.2) |
| Screen-detected cancers, no. (%) | 35 | 57 | 64 | 57 | 66 | 80 | 76 | 57 | 492 |
| CDR, per 1000 women (95% CI) | 3.44 (2.30–4.58) | 4.91 (3.64–6.18) | 5.04 (3.81–6.27) | 5.00 (3.70–6.29) | 4.55 (3.46–5.65) | 4.86 (3.80–5.92) | 4.84 (3.76–5.93) | 4.21 (3.12–5.30) | 4.64 (4.23–5.05) |
| Non-screen-detected cancers, no. | 33 | 32 | 36 | 19 | 29 | 41 | 41 | 47 | 278 |
| Cancer prevalence, per 1000 women (95% CI) | 6.68 (6.20–7.17) | 7.66 (7.18–8.15) | 7.87 (7.40–8.34) | 6.66 (6.12–7.12) | 6.55 (6.15–6.96) | 7.35 (6.15–6.96) | 7.45 (7.04–7.87) | 7.68 (7.24–8.13) | 7.26 (7.10–7.41) |
| Sensitivity, % (95% CI) | 51.5 (39.6–63.3) | 64.0 (54.1–74.0) | 64.0 (54.6–73.4) | 75.0 (65.3–84.7) | 69.5 (60.2–78.7) | 66.1 (57.7–74.5) | 65.0 (56.3–73.6) | 54.8 (45.2–64.4) | 63.9 (60.5–67.3) |
| Specificity, % (95% CI) | 99.4 (99.2–99.5) | 99.2 (99.1–99.4) | 99.4 (99.3–99.5) | 99.1 (98.9–99.3) | 98.9 (98.7–99.1) | 99.3 (99.2–99.5) | 99.2 (99.1–99.3) | 99.2 (99.1–99.4) | 99.2 (99.1–99.3) |
| PPV, % (95% CI) | 36.5 (26.8–46.1) | 3 8.8 (30.9–46.7) | 45.4 (37.2–53.6) | 36.1 (28.6–43.6) | 29.5 (23.5–35.4) | 42.6 (35.5–49.6) | 38.0 (31.3–44.7) | 35.4 (28.0–42.8) | 37.4 (34.8–40.0) |
CDR=cancer detection rate; CI=confidence interval; PPV=positive predictive value of referral.
Considerable individual variation existed between the radiologists regarding referral rates (range, 0.9–1.5%), breast cancer detection rates (range, 3.44–5.04 per 1000 screened women) and sensitivity (range, 51.5–75.0%; P=0.003). Sensitivity was significantly related to the referral rate (Pearson's correlation coefficient r=0.75, P=0.03). However, there was no correlation between radiologist experience (reading experience in number of months before the start of the study) and referral rate (Pearson's correlation coefficient r=0.11) or between radiologist experience and CDR (Pearson's correlation coefficient r=0.06).
Radiologist double-reading
The eight radiologists formed 28 different couples for double-reading. The total number of mammograms assessed per specific couple ranged from 757 to 7484 (median: 3691). Radiologist double-reading with consensus reading would have resulted in 1448 referrals. Compared with single-reading, the referral rate would increase from 1.24 to 1.36% (95% CI=1.30–1.43, P=0.011; Table 2 and Figure 1). The distribution of mammographic abnormalities of the 148 additional referrals with double-reading corresponded to the distribution observed at single-reading. With double-reading and consensus discussion, 37 additional cancers were detected. One interval cancer had initially been considered positive by the first reader, but had been assessed negative at consensus reading. Double-reading increased the mean CDR from 4.64 to 4.98 (P=0.26), with individual radiologist values ranging from 4.44 (106/23,872; 95% CI=3.60–5.28) to 5.62 (151/26 874; 95%CI=4.73–6.51). Mean sensitivity increased from 63.9 to 68.6% (P=0.02), with individual radiologist values varying between 62.0% (106/171; 95% CI=54.7–69.3) and 77.0% (127/165; 95% CI=70.5–83.4). At double-reading, the two specialized breast radiologists (screener E and F) detected 67.6% (25/37) of the cancers found only by the second reader and they acquired the highest sensitivity values of 77.0% (95% CI=70.5–83.4) and 70.7% (95% CI=64.7–76.7), respectively.
Table 2. Breast cancers and tumour characteristics at different reading strategies.
| Reading strategy | Single radiologist reading | Radiologist double-reading | Single radiologist reading and radiographer double-reading with referral of all positive readings | Radiologist double-reading followed by radiologist review of positive cases at radiographer double-reading | Double-reading by radiologists and radiographers with referral of all positive readings |
|---|---|---|---|---|---|
| Referral rate, % (95% CI) | 1.24 (1.17–1.31) | 1.36 (1.30–1.43) | 1.96 (1.87–2.04) | 1.48 (1.41–1.55) | 2.04 (1.95–2.12) |
| Mammographic abnormality, no. (%) | 1315 | 1448 | 2076 | 1570 | 2161 |
| Density | 877 (66.7) | 967 (66.8) | 1386 (66.8) | 1041 (66.3) | 1448 (67.0) |
| Microcalcifications | 257 (19.5) | 289 (20.0) | 447 (21.5) | 331 (21.1) | 464 (21.5) |
| Density with microcalcifications | 106 (8.1) | 114 (7.9) | 140 (6.7) | 116 (7.4) | 145 (6.7) |
| Architectural distortion | 44 (3.3) | 47 (3.2) | 62 (3.0) | 51 (3.2) | 64 (3.0) |
| Breast parenchyma asymmetry | 31 (2.4) | 31 (2.1) | 41 (2.0) | 31 (2.0) | 40 (1.9) |
| Breast cancers, no. | 492 | 528 | 579 | 564 | 592 |
| CDR, per 1,000 women (95% CI) | 4.64 (4.23–5.05) | 4.98 (4.55–5.40) | 5.46 (5.01–5.90) | 5.32 (4.88–5.75) | 5.58 (5.13–6.03) |
| Sensitivity, % (95% CI) | 63.9 (60.5–67.3) | 68.6 (65.3–71.9) | 75.2 (72.1–78.2) | 73.2 (70.1–76.4) | 76.9 (73.9–79.9) |
| Specificity, % (95% CI) | 99.2 (99.2–99.3) | 99.1 (99.1–99.2) | 98.6 (98.5–98.7) | 99.0 (99.0–99.1) | 98.5 (98.4–98.6) |
| PPV of referral, % (95% CI) | 37.4 (34.8–40.0) | 36.5 (34.0–38.9) | 27.9 (26.0–29.8) | 35.9 (33.6–38.3) | 27.4 (25.5–29.3) |
| Type of breast cancer, no. (%) | |||||
| DCIS | 80 (16.3) | 90 (17.0) | NA | 98 (17.4) | NA |
| Invasive | 412 (83.7) | 438 (83.0) | NA | 466 (82.6) | NA |
| T1a–c | 321 (77.9) | 342 (78.1) | NA | 366 (78.5) | NA |
| T2 | 88 (21.4) | 93 (21.2) | NA | 96 (20.6) | NA |
| Unknown | 3 (0.7) | 3 (0.7) | NA | 4 (0.9) | NA |
CI=confidence interval; CDR=cancer detection rate; DCIS=ductal carcinoma in situ; PPV=positive predictive value.
NA: several cancers were diagnosed as interval cancers or detected at subsequent screening. Consequently, exact tumour stages of these cancers at the time of the index screening examination are not available.
Radiologist double-reading with referral of all abnormal mammograms, instead of referral following consensus in case of discrepant interpretations, would have resulted in 1473 referrals (referral rate, 1.39%, 95% CI=1.32–1.46), 530 screen detected cancers (CDR, 5.00, 95% CI=4.57–5.42), and a sensitivity of 68.8% (95% CI=65.6–72.1). This reading strategy added 25 referrals, which were considered positive only by the first reader (15 referrals) or second reader (10 referrals), to the 1448 referrals at radiologist double-reading with consensus reading.
Referral of all positive readings at radiologist single-reading combined with radiographer double-reading
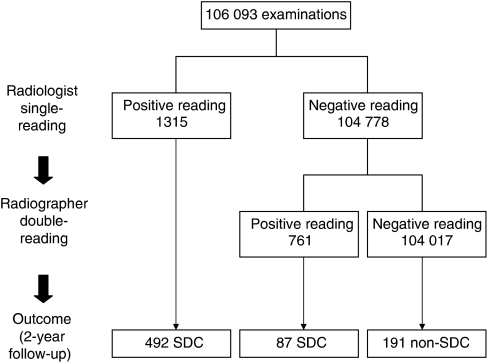
Addition of radiographer double-reading to radiologist single-reading, in combination with referral of all mammograms that were considered abnormal by the radiologist and/or radiographers, would have resulted in a referral rate of 1.96%. (95% CI=1.87–2.04) and a CDR of 5.46 (95% CI=5.01–5.90; Table 2 and Figure 2). Compared with radiologist single-reading, additional reading by the pair of radiographers would have resulted in a significant increase of the referral rate (from 1.24 to 1.96%, P<0.001), the CDR (from 4.64 to 5.46, P=0.006), and the sensitivity (from 63.9 to 75.2%, P<0.001), but in a decreased PPV of referral (from 37.4 to 27.9%, P<0.001). The exact tumour stages of all 87 cancers, detected only by radiographers, could not be determined, as 27 of these cancers evolved as interval cancers (n=19) or were detected at subsequent screening (n=8). Given the hypothetical situation of referring all women with abnormal mammograms at triple reading, double-reading by radiologists would have led to a lower CDR of 4.98 than triple reading by one radiologist and a pair of radiographers (CDR, 5.46; P=0.12).
Figure 2.
Radiologist single-reading combined with radiographer double-reading: mammography-screening outcome at 2-year follow-up. A woman was referred for additional work-up if the mammogram was considered to be positive at radiologist single-reading and/or at radiographer double-reading. SDC=screen-detected cancer.
Radiologist double-reading using secondary referral
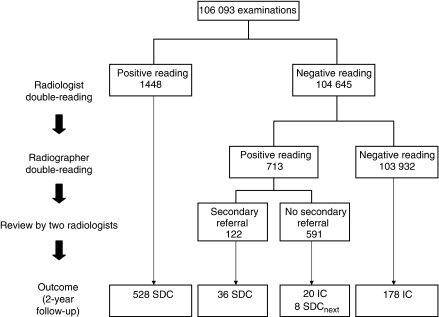
Radiologist double-reading, followed by review of mammograms that were considered abnormal at radiographer double-reading only, reflected the actual screening situation. Of the 713 examinations that were considered abnormal by radiographers only, 122 were referred upon review by two screening radiologists (Figure 3). Compared with cancers identified at radiologist double-reading, the 36 cancers detected after secondary referral included a higher percentage of ductal carcinomas in situ (22.2% (8/36) vs 17.0% (90/528), P=0.4) and a larger proportion of invasive tumours were <20 mm (T1a-c, 85.7% (24/28) vs 78.1% (342/438), P=0.3, Table 2). Replacement of screening strategy IV by strategy III (i.e., replacement of the second radiologist by a pair of radiographers, in combination with referral of all radiographer positive readings rather than review of radiographer positive readings) would have resulted in a similar cancer detection rate (5.46 vs 5.32, P=0.66) and sensitivity (75.2 vs 73.2%, P=0.22), but a lower PPV of referral (27.9 vs 35.9%, P<0.001).
Figure 3.
Radiologist double-reading followed by radiologist review of positive cases at radiographer double-reading: mammography-screening outcome at 2-year follow-up. SDC=screen-detected cancer; SDCnext=cancer detected at subsequent screening; IC=interval cancer.
Double-reading by radiologists and radiographers with referral of all radiographer-positive readings
This screening strategy would have resulted not only in the highest referral rate (2.04%, 95% CI=1.95–2.12), but also in the highest CDR (5.58, 95% CI=5.13–6.03) and sensitivity (76.9%, 95% CI=73.9–79.9; Table 2). Compared with secondary referral of radiographer-positive readings upon review, referral of all radiographer-positive readings would have led to the detection of 28 additional cancers, including 20 interval cancers and 8 mammographic abnormalities that proved to be malignant at subsequent screening.
Discussion
At single-reading, we observed large variations in screening outcome among the eight radiologists. The referral rates ranged from 0.9 to 1.5%, sensitivity of breast screening for cancer detection from 51.5 to 75.0% and PPV of referral from 29.5 to 45.4%. Previous studies have shown that the agreement among radiologists interpreting a test set of mammograms is relatively low (Elmore et al, 1994; Kerlikowske et al, 1998). However, data from other population-based series are very rare. The variation in CDR we observed, from 3.4 to 5.0, is comparable with the 2.6–5.4 range that was found in a US study (Gur et al, 2004). In line with the latter study, our results also showed that higher recall rates were significantly correlated with increased detection rates. Owing to differences between the screening programmes and study designs, further comparison between both studies is limited. The US study showed higher referral rates (range, 7.7–17.2%), PPV values were not given and sensitivity could not be calculated, as interval cancers were not included in the analysis. In a retrospective study of a random sample of screening mammograms, Elmore et al (2002) found a large variability range among community radiologists regarding false-positive rates, which was not eliminated after adjustment for patient, radiologist, and testing characteristics. The screening radiologists in our study had ample experience in breast screening before the start of the study, which probably explains that we found no correlation between number of months of reading experience of the radiologist and the referral rate or the CDR. Each radiologist reads a high volume of screening mammograms annually and participates in quality assurance programmes. These reading conditions might imply that their individual contributions to the skill mix has leveled off over time, but we nevertheless found large inter-observer variability in screening outcome. At double-reading, the two breast radiologists tended to have the best screening results with the highest sensitivities and the highest number of cancers detected by the second reader only, but otherwise we do not have a plausible explanation for this observation.
Our study found a 7.3% relative increase in CDR with the use of radiologist double-reading, which is comparable with those reported previously (Taplin et al, 2000; Harvey et al, 2003). The variability range in screening performance among the radiologists was reduced, but not eliminated, after double-reading. Two-thirds of the cancers, detected by the second reader only, were found by two radiologists. This observation indicates that there is room for a further increase in cancer detection by the other second readers. Beam et al (1996) found that specific pairs of radiologists achieve better detection rates than other pairs. We had 28 possible combinations of radiologist couples for double-reading and, taking into account the sequence of the first and second reader, even 56 couples. The numbers per specific couple varied widely and were too small to analyse Beam's belief and would be of no practical value in our screening setting; our schedules are too complex to allow specific pairs of radiologists to perform double interpretation.
Owing to a shortage of radiologists in screening programmes, such as in the United Kingdom and the United States (D'Orsi et al, 2005), it would be of practical value if dedicated radiographers can replace a screening radiologist as the second reader. Compared with radiologist double-reading, triple reading by one radiologist and a pair of radiographers was characterized by a 40% relative increase in a number of referrals, but an absolute referral rate of 1.96% is still low compared with other screening programmes (Smith-Bindman et al, 2003). The data in our study were not suited to assess the performance of individual radiologist–radiographer double-reading, as the radiographers had reported the outcome after consensus reading.
As reported previously, the highest CDR and sensitivity would have been obtained by quadruple reading by two radiologists and two radiographers, followed by referral of all radiologist and/or radiographer-positive readings (Duijm et al, 2007). The 2.04% referral rate at this reading strategy would still be cost effective in the Dutch screening programme (Otten et al, 2005; Groenewoud et al, 2007). There is a delicate balance between referral rate and cancer detection rate (Yankaskas et al, 2001). The Dutch breast screening programme is characterized by low referral rates and relatively high-interval cancer rates. In this study, we found that adding readers resulted in an increased referral rate and cancer detection rate. The current conversion of the Dutch breast screening programme from analogue to digital screening and the simultaneous introduction of screening BI-RADS categories may lower the threshold for referral (American College of Radiology, 2003; Pisano et al, 2005). The impact of these alterations on referral rate, detection rate and interval cancer rate is an important issue of future research.
Our study has certain limitations. First, the second readers were not fully blinded to the first reader's report, as this would be too complex in the current screening practice. The second readers aim not to see the opinion of the first reader before making their own decision about referral, but knowing the report of the first reader might have influenced their interpretation and, as a consequence, their detection rates and sensitivity concerning both single and double-reading. At present, digital mammography is introduced in the Dutch nation-wide breast cancer screening programme. The conversion from analogue to digital screening will be completed within 2 years. Non-blinded double-reading will be replaced by blinded double-reading, and the individual scores of the first and second reader will automatically be documented. The performance of each radiologist will be monitored and used for quality assurance. A second limitation of our study is the inability to assess the screening accuracy of all different pair of radiologists because the number of readings per couple sometimes was too small for interpretation. Differences between pairs may exist, however (Beam et al, 1996). Moreover, it is most likely that radiographer outcome parameters would have been better if only experienced radiographers were used. Although one may prefer that only experienced radiographers read mammograms, we had all radiographers participating. This approach least affected our daily screening practice, as it would be impossible to schedule specific pairs of radiographers to perform double-reading. Again, variations in the number of mammograms per specific radiographer couple precluded a proper statistical analysis of differences in screening outcome parameters among couples.
Finally, the sensitivity of breast cancer screening with 2-year follow-up will be sensitive to the number of interval cancers that developed in the second year after screening. van Dijck et al (1993) showed that a considerable percentage of interval cancers in a biennial screening programme appear de novo between two screening rounds. We related screening outcome parameters to the total breast cancer incidence after 2 years of follow-up rather than after 1 year of follow-up, as this will provide full information about the interval cancer rate and the total costs of follow-up in a biennial screening programme (Duijm et al, 2004b, 2008). Review of late interval cancers is part of the quality assurance and evaluation of the Dutch breast cancer screening programme.
Our prospective field study also has strengths. It is the largest study of double interpretation reported to date and is unique in assessing screening outcome from a single-reading setting to a quadruple reading setting. We were able to assess screening performance in clinical practice at different screening strategies, without having to rely on test sets. Complete follow-up data were obtained in essentially all women, allowing us to identify the false-negative examinations accurately and to calculate sensitivity of breast screening in addition to cancer detection rates.
In summary, we found large variations in individual radiologist's screening performance. Compared with single-reading, radiologists' double-reading significantly increased sensitivity. Triple reading by one radiologist and two radiographers may be an alternative to radiologist double-reading in programmes with a shortage of radiologists. Highest sensitivity was obtained by quadruple interpretation with referral of all radiologist- and radiographer-positive readings.
Conflict of interest
The authors state no conflict of interest.
References
- American College of Radiology (2003) Breast Imaging Reporting and Data System (BI-RADS), 4th edn. American College of Radiology: Reston, VA [Google Scholar]
- Beam CA, Sullivan DC, Layde PM (1996) Effect of human variability on independent double reading in screening mammography. Acad Radiol 3: 891–897 [DOI] [PubMed] [Google Scholar]
- Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, Mandelblatt JS, Yakovlev AY, Habbema JDF, Feuer EJ, the Cancer Intervention and Surveillance Modeling Network (CISNET) Collaborators (2005) Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med 353: 1784–1792 [DOI] [PubMed] [Google Scholar]
- Brown J, Bryan S, Warren R (1996) Mammography screening: an incremental cost effectiveness analysis of double vs single reading of mammograms. Br Med J 312: 809–812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciatto S, Ambrogetti D, Bonardi R, Catarzi S, Risso G, Rosselli Del Turco M, Mantellini P (2005) Second reading of screening mammograms increases cancer detection and recall rates. Results in the Florence screening programme. J Med Screen 12: 103–106 [DOI] [PubMed] [Google Scholar]
- D'Orsi C, Tu SP, Nakano C, Carney PA, Abraham LA, Taplin SH, Hendrick RE, Cutter GR, Berns E, Barlow WE, Elmore JG (2005) Current realities of delivering mammography services in the community: do challenges with staffing and scheduling exist? Radiology 235: 391–395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duijm LEM, Groenewoud JH, Hendriks JHCL, de Koning HJ (2004a) Independent double reading of screening mammograms in the Netherlands: effect of arbitration following reader disagreements. Radiology 231: 564–570 [DOI] [PubMed] [Google Scholar]
- Duijm LEM, Groenewoud JH, Jansen FH, Fracheboud J, van Beek M, de Koning HJ (2004b) Mammography screening in the Netherlands: delay in the diagnosis of breast cancer after breast cancer screening. Br J Cancer 91: 1795–1799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duijm LEM, Groenewoud JH, Fracheboud J, de Koning HJ (2007) Additional double reading of screening mammograms by radiologic technologists: impact on screening performance parameters. J Natl Cancer Inst 99: 1162–1170 [DOI] [PubMed] [Google Scholar]
- Duijm LEM, Groenewoud JH, Fracheboud J, Plaisier ML, Martin van Ineveld B, Roumen RMH, van Beek M, de Koning HJ (2008) Utilization and cost of diagnostic imaging and biopsies following positive screening mammography, 2000–2005. Eur Radiol 18: 2390–2397 [DOI] [PubMed] [Google Scholar]
- Elmore JG, Wells CK, Lee CH, Howard DH, Feinstein AR (1994) Variability in radiologists' interpretations of mammograms. New Engl J Med 331: 1493–1499 [DOI] [PubMed] [Google Scholar]
- Elmore JG, Miglioretti DL, Reisch LM, Barton MB, Kreuter W, Christiansen CL, Fletcher SW (2002) Screening mammograms by community radiologists: variability in false-positive rates. J Natl Cancer Inst 94: 1373–1380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esserman L, Cowley H, Eberle C, Kirkpatrick A, Chang S, Berbaum K, Gale A (2002) Improving the accuracy of mammography: volume and outcome relationships. J Natl Cancer Inst 94: 369–375 [DOI] [PubMed] [Google Scholar]
- Fracheboud J, de Koning HJ, Boer R, Groenewoud JH, Verbeek ALM, Broeders MJM, van Ineveld M, Hendriks JHCL, de Bruyn AE, Holland R, van der Maas PJ (2001) Nationwide breast cancer screening programme fully implemented in the Netherlands. Breast 10: 6–11 [DOI] [PubMed] [Google Scholar]
- Gilbert FJ, Astley SM, McGee MA, Gillan MGC, Boggis CRM, Griffiths PM, Duffy SW (2006) Single reading with computer-aided detection and double reading of screening mammograms in the United Kingdom National Breast Screening Program. Radiology 241: 47–53 [DOI] [PubMed] [Google Scholar]
- Gilbert FJ, Astley SM, Gillan MG, Agbaje OF, Wallis MG, James J, Boggis CR, Duffy SW, II CADET Group (2008) Single reading with computer-aided detection for screening mammography. N Engl J Med 359: 1675–1684 [DOI] [PubMed] [Google Scholar]
- Groenewoud JH, Otten JDM, Fracheboud J, Draisma G, van Ineveld BM, Holland R, Verbeek ALM, de Koning HJ (2007) Cost-effectiveness of different reading and referral strategies in mammography screening in the Netherlands. Breast Cancer Res Treat 102: 211–218 [DOI] [PubMed] [Google Scholar]
- Gur D, Sumkin JH, Hardesty LA, Clearfield RJ, Cohen CS, Ganott MA, Hakim CM, Harris KM, Poller WR, Shah R, Wallace LP, Rockette HE (2004) Recall and detection rates in screening mammography. A review of clinical experience – implications for practice guidelines. Cancer 100: 1590–1594 [DOI] [PubMed] [Google Scholar]
- Harvey SC, Geller B, Oppenheimer RG, Pinet M, Riddell L, Garra B (2003) Increase in cancer detection and recall rates with independent double interpretation of screening mammography. Am J Roentgenol 180: 1461–1467 [DOI] [PubMed] [Google Scholar]
- Kerlikowske K, Grady D, Barclay J, Frankel SD, Ominsky SH, Sickles EA, Ernster V (1998) Variability and accuracy in mammographic interpretation using the American College of Radiology Breast Imaging Reporting and Data System. J Natl Cancer Inst 90: 1801–1809 [DOI] [PubMed] [Google Scholar]
- Otten JD, Karssemeijer N, Hendriks JHCL, Groenewoud JH, Fracheboud J, Verbeek ALM, de Koning HJ, Holland R (2005) Effect of recall rate on earlier screen detection of breast cancers based on the Dutch peformance indicators. J Natl Cancer Inst 97: 748–754 [DOI] [PubMed] [Google Scholar]
- Otto SJ, Fracheboud J, Looman CWN, Broeders MJM, Boer R, Hendriks JHCL, Verbeek ALM, de Koning HJ (2003) Initiation of population-based mammography screening in Dutch municipalities and effect on breast-cancer mortality: a systematic review. Lancet 361: 1411–1417 [DOI] [PubMed] [Google Scholar]
- Pauli R, Hammond S, Cooke J, Ansell J (1996) Comparison of radiographer/radiologist double film reading with single reading in breast cancer screening. J Med Screen 3: 18–22 [DOI] [PubMed] [Google Scholar]
- Pisano EA, Gatsonis C, Hendrick E, Yaffe M, Baum JK, Acharyya S, Conant EF, Fajardo LL, Bassett L, D'Orsi C, Jong R, Rebner M, for the Digital Mammographic Imaging Screening Trial (DMIST) Investigators Group (2005) Diagnostic performance of digital vs film mammography for breast-cancer screening. New Engl J Med 353: 1773–1783 [DOI] [PubMed] [Google Scholar]
- Rutter CM, Taplin SH (2000) Assessing mammographers' accuracy. A comparison of clinical and test performance. J Clin Epidemiol 53: 443–450 [DOI] [PubMed] [Google Scholar]
- Smith-Bindman R, Chu PW, Miglioretti DL, Sickles EA, Blanks R, Ballard-Barbash R, Bobo JK, Lee NC, Wallis MG, Patnick J, Kerlikowske K (2003) Comparison of screening mammography in the United States and the United Kingdom. JAMA 290: 2129–2137 [DOI] [PubMed] [Google Scholar]
- Tabar L, Yen MF, Vitak B, Chen HHT, Smith RA, Duffy SW (2003) Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet 361: 1405–1410 [DOI] [PubMed] [Google Scholar]
- Taplin SH, Rutter CM, Elmore JG, Seger D, White D, Brenner RJ (2000) Accuracy of screening mammography using single vs independent double interpretation. Am J Roentgenol 174: 1257–1262 [DOI] [PubMed] [Google Scholar]
- Tonita JM, Hillis JP, Lim C (1999) Medical radiologic technologist review: effects on a population-based breast cancer screening program. Radiology 211: 529–533 [DOI] [PubMed] [Google Scholar]
- UICC (International Union against Cancer) (1987) TNM Classification of Malignant Tumours, 4th rev edn Springer Verlag: Berlin [Google Scholar]
- van Dijck JA, Verbeek ALM, Hendriks JHCL, Holland R (1993) The current detectability of breast cancer in a mammographic screening program. A review of the previous mammograms of interval and screen-detected cancers. Cancer 72: 1933–1938 [DOI] [PubMed] [Google Scholar]
- Yankaskas BC, Cleveland RJ, Schell MJ, Kozar R (2001) Association of recall rates with sensitivity and positive predictive values of screening mammography. Am J Roentgenol 177: 543–549 [DOI] [PubMed] [Google Scholar]