Abstract
Niemann-Pick type C disease (NPC) is a sphingolipid storage disorder that results from inherited deficiencies of intracellular lipid trafficking proteins, and is characterized by an accumulation of cholesterol and glycosphingolipids in late endosomes and lysosomes. Patients with this disorder develop progressive neurological impairment that often begins in childhood, is ultimately fatal and is currently untreatable. How impaired lipid trafficking leads to neurodegeneration is largely unknown. Here we review NPC clinical features and biochemical defects, and discuss model systems used to study this disorder. Recent studies have established that NPC is associated with an induction of autophagy, a regulated and evolutionarily conserved process by which cytoplasmic proteins are sequestered within autophagosomes and targeted for degradation. This pathway enables recycling of limited or damaged macromolecules to promote cell survival. However, in other instances, robust activation of autophagy leads to cell stress and programmed cell death. We summarize evidence showing that autophagy induction and flux are increased in NPC by signaling through the class III-PI3K/Beclin-1 complex. We propose that an imbalance between induction and flux through the autophagic pathway contributes to cell stress and neuronal loss in NPC and related sphingolipid storage disorders, and discuss potential therapeutic strategies for modulating activity of this pathway.
Keywords: Autophagy, Beclin-1, central nervous system, lysosome, mouse model, mTOR, neurodegeneration, Niemann-Pick type C disease
I Niemann-Pick type C disease
The sphingolipid storage diseases are a collection of ~40 genetically distinct disorders caused by inherited deficiencies of lysosomal hydrolytic activities or lipid transport which result in intracellular accumulations of cholesterol and lipids in the endosomal/lysosomal network. These disorders have an aggregate prevalence of approximately 1 in 8000 births worldwide (1). Among this group is Niemann-Pick type C disease (NPC, MIM 257220), an autosomal recessive, progressive neurovisceral disorder characterized by abnormal cholesterol trafficking and by intracellular accumulations of unesterified cholesterol and glycosphingolipids in late endosomes and lysosomes (2–8). NPC is caused by loss of function mutations in either Npc1 (9) or Npc2 (10), genes whose protein products mediate proper intracellular lipid transport through pathways that are incompletely understood.
NPC patients develop symptoms over a wide range of ages (11), and there is no strict correlation between disease-causing mutations and the severity of the clinical phenotype (4, 12). While patients may initially exhibit systemic findings such as hepatosplenomegaly or obstructive jaundice, all eventually develop neurological and/or psychiatric symptoms, the severity of which is inversely associated with lifespan (13, 14). Patients presenting with neurological symptoms during the first two years of life, classified as the most extreme, infantile cases of NPC, demonstrate delayed motor development and hypotonia, and typically die within their first 5 years (15). The classical form of NPC, which encompasses ~60–70% of cases, presents between 3 and 15 years of life. Parents of these patients are often the first to notice troublesome signs of the disease, such as loss of speech or increasing clumsiness. The disorder progresses to cause a constellation of neurological symptoms that may include cerebellar ataxia, dysarthria, dysphagia, cataplexy, seizures, dystonia, vertical gaze palsy, progressive dementia and death by 8 – 25 years (11). Adult onset cases of NPC are well documented although much less common, and their unusual clinical presentation often initially leads to misdiagnosis. Most adults with NPC develop symptoms in their second or third decades of life, though onset can occur as late as the mid-sixth decade (16). These adult patients develop symptoms similar to the classical form of juvenile NPC and usually die when in their late 30’s or 40’s. At the current time, there are no effective treatments available to patients with this devastating disorder.
II NPC pathology and biochemical defects
While heterogeneity characterizes the clinical presentation of NPC, there is less pathological heterogeneity at the biochemical level. The vast majority of cases show prominent accumulations of unesterified cholesterol, sphingolipids and complex gangliosides in late endosomes and lysosomes of most cells types of the body (2–4, 11). Notably, a subset of patients with certain mutations demonstrate much less lipid storage (17), and central nervous system (CNS) pathology is accompanied by particularly prominent storage of sphingolipids and complex gangliosides. These lipid accumulations result from impairments of cholesterol and sphingolipid trafficking that cause a multifaceted toxic response in the liver, spleen and brain.
A. Histopathology
The progression of NPC in the periphery is characterized by an enlargement of the liver and spleen that results from the presence of lipid-laden macrophages, termed foam cells. Kupffer cells in the liver and splenic macrophages, predominantly in the red pulp, exhibit marked cytoplasmic vacuolization due to an accumulation of cholesterol, phospholipids and glycolipids.
Impairment of lipid trafficking also affects the CNS and results in neuron loss throughout the brain (18). The occurrence of swollen neuronal cell bodies in many brain regions, including cortex, basal ganglia, thalamus, cerebellum and brainstem is also characteristic of NPC and reflects lipid accumulation within late endosomes and lysosomes. The cell body of these neurons and their axonal processes also contain granular material, which by electron microscopy appears as membrane-bound polymorphous bodies containing loosely packed lamellae or as dense osmiophilic inclusions. Additional neuronal pathology includes the formation of ectopic dendrites, swelling of proximal axons to so-called meganeurites, and the presence of axonal spheroids, changes indicative of a neuroaxonal dystrophy (18). Like several other neurodegenerative diseases, NPC is classified as a tauopathy due to the presence of intracellular aggregates of the microtubule binding protein tau, which are biochemically identical to aggregates in Alzheimer disease (19). These changes in the NPC brain are accompanied by gliosis in grey and white matter, microglial activation and cerebral atrophy.
B. Biochemistry
Lipid trafficking defects within the NPC brain reflect defects in the pathway by which cholesterol and other lipids arrive in neurons and are sorted intracellularly. Studies in normal mice have demonstrated that cholesterol accumulation in the CNS markedly increases during the first three weeks a life, a period of late development and myelination (20, 21). Lipoprotein cholesterol from the periphery does not cross the blood brain barrier to enter the CNS, even during this early phase of brain development, and therefore de novo synthesis is required to meet the needs of both the developing and mature nervous system (22–24). Neurons and other CNS cell types obtain the cholesterol they require though endogenous synthesis or by uptake of lipoprotein cholesterol particles produced and released within the nervous system, possibly by astrocytes (25). Once target cells internalize these particles, unesterified cholesterol and companion lipids are trafficked from the endosomal/lysosomal system to compartments such as the Golgi and endoplasmic reticulum for processing and to serve as substrates for further reactions (26). Derivatives of cholesterol include oxysterols, which signal a decrease in the de novo synthesis of cholesterol and therefore provide negative feedback (27, 28).
In NPC fibroblasts, lipoprotein cholesterol particles are internalized without disruption but become entrapped within trafficking vesicles, particularly late endosomes and lysosomes (Fig. 1) (29–31). The insufficient efflux of cholesterol and lipids out of these structures causes a two-fold problem: unesterified cholesterol, sphingolipids and complex gangliosides accumulate within vesicles in the cell’s cytoplasm, while there is a simultaneous paucity of cholesterol and other essential lipids in organelles to which these compounds were originally destined. Decreased cholesterol in the endoplasmic reticulum, Golgi apparatus and other internal organelles causes deleterious effects on a multitude of processes dependent on proper membrane composition (32). Disrupted trafficking also results in a paucity of substrate for further synthetic reactions. For example, decreased availability of unesterified cholesterol impairs production of oxysterols, which may lead to increased cholesterol synthesis and an exacerbation of cholesterol accumulation in NPC cells (33).
Figure 1. Lipid trafficking defects in Niemann-Pick type C disease.
Left panel. Under normal conditions, lipoprotein cholesterol particles enter the endosomal network after binding to cell surface receptors. Unesterified cholesterol and companion lipids are trafficked from the endosomal/lysosomal system to the Golgi, endoplasmic reticulum and other intracellular compartments.
Right panel. NPC1 or NPC2 loss of function inhibits the egress of unesterified cholesterol and sphingolipids from late endosomes and lysosomes, resulting in their accumulation at these sites and a paucity of these lipids at the intracellular compartments to which they were destined.
Abbreviation: ER, endoplasmic reticulum.
III Genetics of NPC
Almost a century has passed since the original reports by Albert Niemann and Ludwick Pick described patients with hepatosplenomegaly and rapidly progressive brain damage. Since then, biochemical and genetic studies have helped define the classification of the Niemann-Pick disease group. Niemann-Pick types A and B disease (MIM 257200 and 607616), distinct from NPC, are caused by loss of function mutations in the acid sphingomyelinase gene, encoding an enzyme that metabolizes sphingomyelin to ceramide and phosphocholine. While fibroblasts from NPC patients may show reduced sphingomyelinase activity in culture, this reduction is due to an imbalanced lipid and cholesterol environment, and removal of cholesterol from the media recovers enzymatic activity (34). In contrast, NPC fibroblasts exhibit an accumulation of unesterified cholesterol indicative of a defect in cholesterol trafficking. The major complementation group for NPC (11) was mapped to the proximal long arm of chromosome 18, and mutations in the Npc1 gene were found in ~95% of patients with this disease (9). The remaining 5% of NPC patients were subsequently found to have loss of function mutations in a separate gene, Npc2 (10).
The Npc1 gene encodes a multipass transmembrane protein, NPC1, which is localized to late endosomes and lysosomes (35, 36). The encoded protein is predicted to contain a sterol-sensing domain, similar to that found in regulators of cholesterol metabolism and in Patched, a receptor for the secreted signaling protein Hedgehog (9, 37). Experimental evidence indicates that NPC1 is involved in the efflux of LDL-derived unesterified cholesterol from late endosomes and lysosomes to other intracellular compartments (30, 31). Multiple disease-causing mutations within the Npc1 coding region have been identified which lead to its loss of function (38). The NPC1 protein is conserved within the animal kingdom, and loss of function mutations in orthologous genes in yeast (39), worms (40), flies (41), mice (42), cats (43) and dogs (44) disrupt lipid trafficking. Introduction of the yeast Npc1 ortholog, Ncrp1, into Chinese hamster ovary cells deficient in NPC1 is sufficient to reverse lipid trafficking defects associated with Npc1 deficiency, demonstrating that this protein is functionally conserved from yeast to mammals (39).
The Npc2 gene encodes a small, soluble protein that is localized to lysosomes, but is also secreted (10, 45). Loss of function mutations in this gene result in a disease phenotype that is clinically and biochemically indistinguishable from the disease caused by Npc1 mutations. The NPC2 protein binds cholesterol, and structural analysis has identified a hydrophobic pocket that serves as a putative cholesterol-binding domain (46). Disease causing mutations in Npc2 that affect amino acid residues surrounding this pocket impair cholesterol binding (47). Analysis of Npc1/Npc2 double mutant mice demonstrates that the disease phenotype is similar to that occurring in Npc1 or Npc2 single mutants, providing genetic evidence that the NPC1 and NPC2 proteins function in the same pathway to regulate intracellular lipid transport (48).
IV NPC1 deficient mice
A. Phenotype
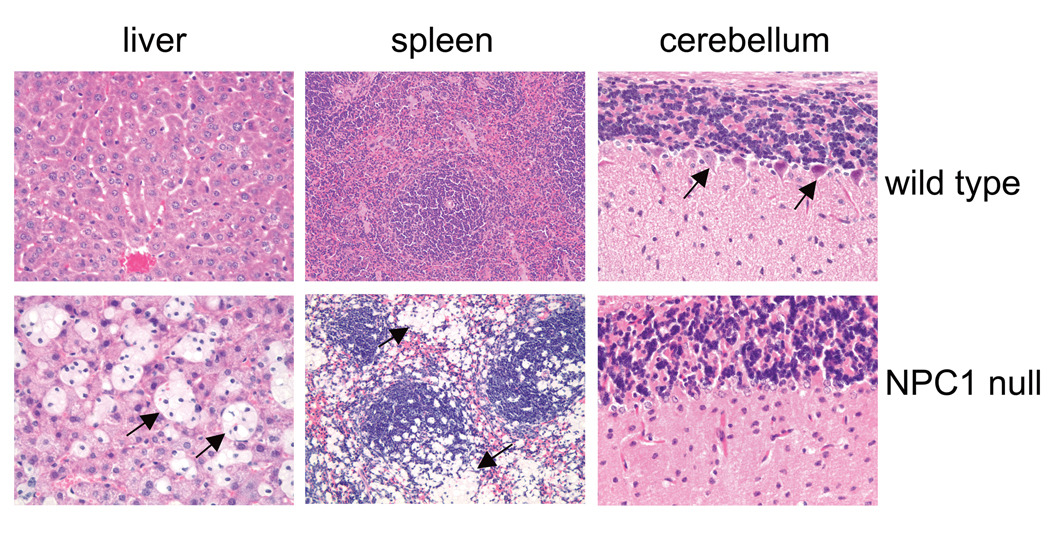
The most commonly used mammalian model of NPC, BALB/cNctr-Npc1m1N/J mice (Jackson Laboratory #003092), contains a transposon insertion into Npc1 exon 9 (49). Animals homozygous for this insertional mutation fail to express functional NPC1 protein and exhibit a degenerative phenotype that begins around 7 weeks (49, 50). The severity of the phenotype progresses with age and includes weight loss, tremors, ataxia, and death by 10–12 weeks. Liver and spleen contain an abundance of foam cells, reminiscent of the histopathological changes seen in humans with this disorder (Fig. 2). In vivo knockdown of Npc1 only in the liver of normal mice recapitulates the hepatic pathology associated with NPC, generating a novel model to examine liver dysfunction associated with disease independent of CNS involvement (51).
Figure 2. NPC histology.
Foamy macrophages are abundant in the liver and spleen of NPC1 deficient mice (examples at arrowheads). In the brain, loss of cerebellar Purkinje cells is a prominent pathologic feature. These large, pyramidal-shaped neurons normally reside at the interface of the molecular layer and granular cell layer of the cerebellar folia (examples at arrowheads). These cells die in NPC1 deficient mice and are replaced by reactive astrocytes.
Global loss of NPC1 expression leads to CNS pathology including vacuolated neurons that accumulate glucosylceramide, lactosylceramide, complex gangliosides and cholesterol (52–54), abnormally swollen axons, and decreased myelination in the cerebral white matter (55). A prominent feature of CNS pathology is loss of Purkinje cells, the major neuronal cell type transmitting output from the cerebellum (56). These highly specialized neurons are responsible for timing and smoothness of movements, and dysfunction of or lesions within the cerebellum can lead to ataxia. Loss of Purkinje cells is initially apparent in the cerebellar vermis by 7 weeks. Progressive Purkinje cell loss proceeds from the anterior to posterior vermis, yet largely spares lobules IX and X, even in mice with end-stage disease (56). Interestingly, 2-year-old Npc1 +/− mice also exhibit mild Purkinje cell loss suggesting that haploinsufficiency for NPC1 may have adverse consequences in the aging brain (57).
B. Lipid trafficking defects
NPC1 deficient mice have been used to explore how lipid trafficking defects lead to progressive neurodegeneration. Attention has focused on the role of glycosphingolipids, including complex gangliosides such as GM2, which are not commonly found in the adult brain but accumulate in NPC neurons (54). These gangliosides are involved in normal brain development, and their presence in the NPC brain has been implicated in pathogenesis, including the formation of ectopic dendrites that branch from swollen axons (18). These abnormally long and inappropriately located processes may contribute to neuronal dysfunction. Based on this notion, on indications that some glycosphingolipids are toxic, and on findings that cholesterol accumulation in the NPC brain is ganglioside-dependent (58), glycosphingolipid synthesis has emerged as a therapeutic target in NPC and related sphingolipid storage disorders (59–61).
Trafficking defects also lead to diminished production of cholesterol derivatives including neurosteroids, which normally help mediate brain development, growth and differentiation (62). A single injection of the neurosteroid allopregnanolone, whose production is significantly decreased in NPC mice, is sufficient to promote Purkinje cell survival and delay disease phenotype (63). Although initial evidence suggested that this beneficial effect was mediated through GABAA receptor signaling, more recent studies indicate that it occurs by activation of the pregnane X receptor (PXR) or liver X receptor (LXR) (64). These steroid receptors are ligand activated transcription factors that bind the oxysterols 25-hydroxycholesterol and 27-hydroxycholesterol (27, 28). Pathways activated by the receptors promote efflux of cholesterol from late endosomes and lysosomes, and suppress endogenous cholesterol synthesis. Exogenous application of either 25-hydroxycholesterol or 27-hydroxycholesterol is sufficient to correct sterol defects in NPC1 deficient human fibroblasts and to mobilize cholesterol from the late endosomal/lysosomal compartment (65). LXR agonists also promote cholesterol efflux from the NPC mouse brain, slow neurodegeneration and prolong life (66), suggesting that activation of these pathways is a promising therapeutic strategy.
C. Cell autonomous versus non-cell autonomous cell death
Defects in lipid trafficking that occur in NPC lead to cell autonomous neuronal death. This was established by an analysis of chimeric mice that express functional NPC1 protein in only a subset of cells (67). In this model, NPC1 deficient Purkinje cells died, even when neighboring cells harbored normal levels of NPC1 protein. Conversely, Purkinje cells with functional NPC1 were spared, even in an NPC1-deficient environment. Examination of NPC1 deficient Purkinje cells in mutant mice revealed evidence of autophagy, suggesting that this pathway is activated in the NPC brain and may play a role in pathogenesis.
V Autophagy
Autophagy is a conserved cellular pathway that mediates the turnover of damaged or aged macromolecules and organelles. Three types of autophagy have been described, including microautophagy, chaperone mediated autophagy and macroautophagy. For the purposes of this review, we will discuss only macroautophagy and refer to it simply as “autophagy”.
At basal levels, autophagy is a house keeping mechanism for the constitutive degradation of long-lived proteins and other macromolecules inaccessible to the ubiquitin-proteasome pathway. Regulated degradation is governed by activation of autophagy related genes, or atg’s (68). Autophagy begins with the generation of an isolation membrane, a crescent shaped double membrane that originates from either smooth ER or another subcellular location that has yet to be defined (Fig. 3) (69). This isolation membrane surrounds cargo targeted for degradation. During the engulfment process, two ubiquitin-like conjugation reactions are required for progression through autophagy: the formation of an Atg12-Atg5-Atg16 complex and the conjugation of Atg8 (also known as microtubuole-associated protein 1 light chain 3 protein, LC3) to phosphatidylethanolamine (70). This latter step is commonly used as a biochemical marker for the induction of autophagy (71, 72). Following engulfment, mature autophagosomes fuse with lysosomes to enable degradation of contents and recycling of their components, a process indicative of flux through the autophagic pathway (73).
Figure 3. The autophagic pathway.
Autophagy occurs at low, basal levels and can be induced by multiple stimuli, including amino acid starvation or mTOR inhibition. It begins with the formation of an isolation membrane, a crescent-shaped sequestering membrane also termed a phagophore, and the induction of a ubiquitin-like conjugation reaction that couples soluble LC3-I with phosphatidylethanolamine. Lipidated LC3-II studs the phagophore membrane as it engulfs macromolecules, organelles and other cargo destined for degradation. Mature autophagosomes fuse with lysosomes to form autolysosomes, thereby degrading cargo and enabling components to be recycled. Not shown is the fusion of some autophagosomes with endosomes to form so-called amphisomes, another vesicle that fuses with lysosomes to facilitate cargo degradation.
Abbreviations: mTOR, mammalian target of rapamycin; LC3, microtubuole-associated protein 1 light chain 3 protein
Autophagy can be induced above basal levels by several stimuli, including amino acid starvation or cellular stress. This induction is mediated by signaling through the mammalian target of rapamycin (mTOR) or the Class III-PI3K/Beclin-1 complex (74). mTOR is a negative regulator of autophagy and its chemical inhibition by rapamycin relieves this blockade (74, 75). The Class III-PI3K/Beclin-1 complex is necessary for the induction of autophagy and can activate autophagy independent of mTOR inhibition (76).
Autophagy is necessary for normal CNS development and function, and has been implicated in the pathogenesis of a diverse array of neurodegenerative diseases, including protein aggregation disorders such as Alzheimer (77), Parkinson (78), and Huntington disease (79), and the sphingolipid storage disorders (80). For age-related diseases associated with protein misfolding, inducing autophagy in model systems enhances the clearance of misfolded or mutant proteins that are not efficiently degraded by the proteasome. This enhancement of protein clearance abrogates disease phenotypes (81), indicating that autophagy may have beneficial effects in some disease processes. Conversely, genetic deletion in mouse CNS of either of two autophagy related genes, atg5 (82) or atg7 (83), leads to neurodegeneration and the accumulation of ubiquitinated protein aggregates, demonstrating that basal autophagy contributes significantly to protein quality control and cell survival in the nervous system.
Although the induction of autophagy may alleviate disease in some instances, excessive autophagy can have deleterious effects in others. In mouse embryonic fibroblasts deficient in apoptosis (bax/bak −/− cells), the classic programmed cell death pathway, application of death stimuli such as staurosporin or etoposide triggers an apoptosis-independent cell death pathway (84). Further genetic manipulation of these cells through RNAi knockdown of two autophagy regulators, atg5 and beclin-1, inhibited cell death, demonstrating the occurrence of a death program mediated by autophagy. Excessive activation or failure to complete autophagy has since been implicated in numerous disease processes, including some neurodegenerative diseases.
VI Autophagy in NPC and other lysosomal storage disorders
Induction of autophagy in NPC1 deficient mice was first reported by Ko et al, who observed frequent autophagic vacuoles in Purkinje cells of mutant mice accompanied by increased accumulation of lipidated LC3 in cerebellar lysates (67). Similar findings have since been reported by our lab and others (85, 86), confirming autophagy induction in the NPC brain, and extending this observation to additional pathologically affected organs, such as liver, and to primary human fibroblasts from NPC patients. Flux through the autophagic pathway is also increased in NPC1 deficient cells, as confirmed by monitoring the degradation of long-lived proteins and by establishing that inhibition of lysosomal proteases increases lipidated LC3 levels in mutant cells (86); in contrast, inhibition of lysosome function does not alter LC3 levels in pathological situations characterized by impaired autophagosome-lysosome fusion (73). The induction of autophagy in NPC1 deficiency is not mediated by signaling through the Akt-mTor-p70 S6K pathway, but rather by the class III-PI3K/Beclin-1 complex (86). Beclin-1 expression is mildly elevated in NPC1 deficient mouse tissue and human fibroblasts, and Beclin-1 knock-down by siRNA decreases long-lived protein degradation (86). Induction of autophagy and increased Beclin-1 expression are similarly observed in NPC2 deficient primary human fibroblasts and in control cells treated with U18666A (86–88), a compound known to induce an accumulation of unesterified cholesterol. In contrast, Gaucher disease fibroblasts, which traffic sphingolipids normally (26), show wild type levels of Beclin-1 and LC3-II, suggesting that autophagy induction is sensitive to alterations in these lipids (86). We envision that the class III-PI3K/Beclin-1 complex senses changes due to the dysregulation of lipid trafficking in NPC and other disorders and activates autophagy.
While NPC1 deficiency is characterized by elevated induction and flux through the autophagic pathway, ubiquitinated proteins accumulate in the mutant mouse brain. These ubiquitinated proteins can be visualized in cerebellar tissue of NPC1 deficient mice by immunohistochemistry (56). Fractionation studies indicate that much of this material is in the endosomal/lysosomal compartment (85). The paradoxical accumulation of ubiquitinated proteins in the setting of increased autophagic degradation suggests that flux though this pathway is not sufficient to handle the quantity of proteins targeted for degradation (Fig. 4). Situations where induction and flux are disproportionate to each other may lead to autophagic stress, a possible mediator of neuronal dysfunction and a precursor to cell death (89).
Figure 4. Autophagic induction and flux in NPC and other lysosomal storage disorders.
In healthy cells, macromolecules are efficiently targeted for degradation by autophagy in a process characterized by proportionate induction and flux through the pathway. This steady-state equilibrium is disrupted in NPC and other lysosomal storage diseases. In NPC, our working model holds that sphingolipid trafficking defects alter membrane lipid composition and induce autophagy through the activity of the class III-PI3K/Beclin-1 complex. This induction can be measured experimentally by an accumulation of LC3-II in NCP1- or NPC2-deficient protein lysates. The enhanced induction of autophagy occurs coordinately with a more modest increase in autophagic flux, resulting in the accumulation of ubiquitinated proteins in tissues from NPC patients and animal models. Disproportionate induction and flux through the autophagic pathway can lead to cell stress (so-called “autophagic stress”) and may trigger cell death, with dire consequences for NPC patients.
Abbreviation: LC3, microtubuole-associated protein 1 light chain 3 protein
Impaired flux through the autophagic pathway has also been observed in lysosomal storage disorders characterized by deficiencies of specific lysosomal hydrolases, including Pompe disease (90), mucolipidosis type IV (91), multiple sulfatase deficiency and mucopolysaccharidosis type IIIA (92). Mouse models of many of these disorders exhibit increased lipidated LC3, an accumulation of enlarged autophagosomes, and impaired autophagosome-lysosome fusion resulting in decreased substrate degradation. A similar block of autophagosome-lysosome fusion has been documented in a mouse model of juvenile neuronal ceroid lipofucinosis, a degenerative disorder caused by mutations in a novel, multipass transmembrane protein localized to late endosomes and lysosomes (93). These data suggest that impaired flux through the autophagic pathway is common to many lysosomal storage diseases, and that an imbalance between induction and flux may contribute to the pathology of this diverse array of disorders by causing autophagic stress or triggering autophagic cell death.
VII Clinical implications and outstanding research questions
Current therapeutic options for patients with the sphingolipid storage disorders are limited (see Table 1 for a summary of NPC therapeutic strategies). For diseases caused by deficiencies of lysosomal hydrolases, ideal therapies would replace the deficient enzymes, however administration presents a significant challenge. Intravenous infusion of active enzymes has yielded some success in Gaucher disease, Fabry disease, mucopolysaccharidosis I and glycogen storage disease type II (94). Yet, this approach treats only the systemic and not CNS disease manifestations because of limited blood brain barrier permeability. Similarly, bone marrow transplantation is effective at reducing systemic but not neurological symptoms in NPC (95), as is the case for most, but not all sphingolipid storage disorders (96). An alternative treatment strategy is substrate reduction therapy, which aims to reduce accumulating material in endosomes and lysosomes based on the notion that excess substrate contributes to pathogenesis. In the case of NPC, reduction of intracellular cholesterol by a combination of cholestyramine, lovastatin and nicotinic acid was able to lower liver and blood levels of cholesterol, but had little effect on disease progression (97, 98). For disorders characterized by neurodegeneration and the accumulation of glycosphingolipids in the CNS, small molecule inhibitors of glycosphingolipid synthesis have attracted considerable attention. Imino-sugars such as miglustat (N-butyl-deoxynojirimycin) are reported to act through this mechanism, delay symptom onset and prolong survival in mouse models of NPC (60) and Sandhoff disease (99). A recently completed clinical trial of miglustat in a small number of NPC patients similarly yielded a mild, encouraging effect after one year of treatment (61).
Table 1.
NPC therapeutic targets and strategies
| Mechanism of Action | Therapeutic | Refs |
|---|---|---|
| I. Current and former strategies | ||
| A. Introduction of functional Npc1 or Npc2 | Bone Marrow transplantaion | #95 |
| B. Substrate reduction therapy | ||
| i. Reduce intracellular cholesterol | Cholestyramine, lovastatin and nicotinic acid | #97, 98 |
| ii. Reduce glycosphingolipid accumulation | Miglustat (N-butyl-deoxynojirimycin) | #61 |
| II. Possible future strategies | ||
| A. Activate endogenous pathways that promote cholesterol efflux | LXR or PXR agonist | #63–66 |
| B. Chemical modulation of autophagy | Rapamycin derivatives or novel autophagy inhibitors | #67, 85, 86 |
Despite these various treatment strategies, only modest clinical improvements have been achieved and new therapeutic approaches are needed. The recently described relationship between autophagy and NPC may lead to novel therapeutic targets. A critical, unanswered question is whether activation of this pathway in NPC patients is detrimental and promotes cell death, or is compensatory and promotes cell survival. To resolve this issue, autophagy will need to be modulated in NPC model systems and effects on disease phenotype established. If it is determined that increasing autophagy is beneficial, then administration of compounds like rapamycin would be an interesting therapeutic strategy. Rapamycin, in the form of Rapamune© (also termed sirolimus), has been tested in humans as an immune suppressant for organ transplant patients (100). Alternatively, if autophagy is found to enhance the NPC phenotype, small molecule inhibitors of this pathway may provide therapeutic benefit. A search for compounds that specifically modulate the activity of the autophagic pathway in the CNS will likely yield important reagents for investigational studies of disease mechanisms, and may provide lead compounds for the development of new treatments.
Acknowledgements and funding
We thank the reviewers for their helpful comments on our manuscript. Work in our laboratory on Niemann-Pick C disease is supported by grants from the National Institutes of Health (F31 NS51143 to C.D.P., R03 NS057150 to A.P.L.).
Footnotes
Further reading, resources and contacts
Publications
There are several excellent, recent reviews on autophagy and disease, including:
Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell, 2008; 132:27-42.
Mizushima N, Levine B, Cuervo AM, and Klionsky DJ. Autophagy fights disease through cellular self-digestion. Nature, 2008, 451:1069-1075.
Websites
The Ara Parseghian Medical Research Foundation supports scientific research on Niemann-Pick type C disease and related disorders. Their website includes information about the Foundation, the diseases they target with research support, and links to other sources of information. www.parseghian.org
The National Niemann-Pick Disease Foundation provides support for patients and their families, and promotes scientific research on the Niemann-Pick disease group. www.nnpdf.org
References
- 1.Meikle PJ, et al. Prevalence of lysosomal storage disorders. Jama. 1999;281(3):249–254. doi: 10.1001/jama.281.3.249. [DOI] [PubMed] [Google Scholar]
- 2.German DC, et al. Neurodegeneration in the Niemann-Pick C mouse: glial involvement. Neuroscience. 2002;109(3):437–450. doi: 10.1016/s0306-4522(01)00517-6. [DOI] [PubMed] [Google Scholar]
- 3.Patel SC, et al. Localization of Niemann-Pick C1 protein in astrocytes: implications for neuronal degeneration in Niemann-Pick type C disease. Proc Natl Acad Sci U S A. 1999;96(4):1657–1662. doi: 10.1073/pnas.96.4.1657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vanier MT, Millat G. Niemann-Pick disease type C. Clin Genet. 2003;64(4):269–281. doi: 10.1034/j.1399-0004.2003.00147.x. [DOI] [PubMed] [Google Scholar]
- 5.Higgins JJ, et al. A clinical staging classification for type C Niemann-Pick disease. Neurology. 1992;42(12):2286–2290. doi: 10.1212/wnl.42.12.2286. [DOI] [PubMed] [Google Scholar]
- 6.Karten B, et al. Cholesterol accumulates in cell bodies, but is decreased in distal axons, of Niemann-Pick C1-deficient neurons. J Neurochem. 2002;83(5):1154–1163. doi: 10.1046/j.1471-4159.2002.01220.x. [DOI] [PubMed] [Google Scholar]
- 7.Karten B, et al. Trafficking of cholesterol from cell bodies to distal axons in Niemann Pick C1-deficient neurons. J Biol Chem. 2003;278(6):4168–4175. doi: 10.1074/jbc.M205406200. [DOI] [PubMed] [Google Scholar]
- 8.Reid PC, Sugii S, Chang TY. Trafficking defects in endogenously synthesized cholesterol in fibroblasts, macrophages, hepatocytes, and glial cells from Niemann-Pick type C1 mice. J Lipid Res. 2003;44(5):1010–1019. doi: 10.1194/jlr.M300009-JLR200. [DOI] [PubMed] [Google Scholar]
- 9.Carstea ED, et al. Niemann-Pick C1 disease gene: homology to mediators of cholesterol homeostasis. Science. 1997;277(5323):228–231. doi: 10.1126/science.277.5323.228. [DOI] [PubMed] [Google Scholar]
- 10.Naureckiene S, et al. Identification of HE1 as the second gene of Niemann-Pick C disease. Science. 2000;290(5500):2298–2301. doi: 10.1126/science.290.5500.2298. [DOI] [PubMed] [Google Scholar]
- 11.Patterson MC, et al. Niemann-Pick disease type C: a lipid trafficking disorder, in The Metabolic and Moleular Bases of Inherited Disease. 8th edition. New York: McGraw-Hill; 2001. pp. 3611–3634. [Google Scholar]
- 12.Yamamoto T, et al. Genotype-phenotype relationship of Niemann-Pick disease type C: a possible correlation between clinical onset and levels of NPC1 protein in isolated skin fibroblasts. J Med Genet. 2000;37(9):707–712. doi: 10.1136/jmg.37.9.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Imrie J, et al. Niemann-Pick disease type C in adults. J Inherit Metab Dis. 2002;25(6):491–500. doi: 10.1023/a:1021259403196. [DOI] [PubMed] [Google Scholar]
- 14.Turpin JC, Masson M, Baumann N. Clinical aspects of Niemann-Pick type C disease in the adult. Dev Neurosci. 1991;13(4–5):304–306. doi: 10.1159/000112177. [DOI] [PubMed] [Google Scholar]
- 15.Vanier MT, et al. Niemann-Pick disease group C: clinical variability and diagnosis based on defective cholesterol esterification. A collaborative study on 70 patients. Clin Genet. 1988;33(5):331–348. doi: 10.1111/j.1399-0004.1988.tb03460.x. [DOI] [PubMed] [Google Scholar]
- 16.Sevin M, et al. The adult form of Niemann-Pick disease type C. Brain. 2007;130(Pt 1):120–133. doi: 10.1093/brain/awl260. [DOI] [PubMed] [Google Scholar]
- 17.Millat G, et al. Niemann-Pick disease type C: spectrum of HE1 mutations and genotype/phenotype correlations in the NPC2 group. Am J Hum Genet. 2001;69(5):1013–1021. doi: 10.1086/324068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Walkley SU, Suzuki K. Consequences of NPC1 and NPC2 loss of function in mammalian neurons. Biochim Biophys Acta. 2004;1685(1–3):48–62. doi: 10.1016/j.bbalip.2004.08.011. [DOI] [PubMed] [Google Scholar]
- 19.Auer IA, et al. Paired helical filament tau (PHFtau) in Niemann-Pick type C disease is similar to PHFtau in Alzheimer's disease. Acta Neuropathol (Berl) 1995;90(6):547–551. doi: 10.1007/BF00318566. [DOI] [PubMed] [Google Scholar]
- 20.Dietschy JM, Turley SD. Control of cholesterol turnover in the mouse. J Biol Chem. 2002;277(6):3801–3804. doi: 10.1074/jbc.R100057200. [DOI] [PubMed] [Google Scholar]
- 21.Quan G, et al. Ontogenesis and regulation of cholesterol metabolism in the central nervous system of the mouse. Brain Res Dev Brain Res. 2003;146(1–2):87–98. doi: 10.1016/j.devbrainres.2003.09.015. [DOI] [PubMed] [Google Scholar]
- 22.Herz J, Bock HH. Lipoprotein receptors in the nervous system. Annu Rev Biochem. 2002;71:405–434. doi: 10.1146/annurev.biochem.71.110601.135342. [DOI] [PubMed] [Google Scholar]
- 23.Turley SD, et al. Brain does not utilize low density lipoprotein-cholesterol during fetal and neonatal development in the sheep. J Lipid Res. 1996;37(9):1953–1961. [PubMed] [Google Scholar]
- 24.Dietschy JM, Turley SD. Cholesterol metabolism in the brain. Curr Opin Lipidol. 2001;12(2):105–112. doi: 10.1097/00041433-200104000-00003. [DOI] [PubMed] [Google Scholar]
- 25.Mauch DH, et al. CNS synaptogenesis promoted by glia-derived cholesterol. Science. 2001;294(5545):1354–1357. doi: 10.1126/science.294.5545.1354. [DOI] [PubMed] [Google Scholar]
- 26.Pagano RE. Endocytic trafficking of glycosphingolipids in sphingolipid storage diseases. Philos Trans R Soc Lond B Biol Sci. 2003;358(1433):885–891. doi: 10.1098/rstb.2003.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fu X, et al. 27-hydroxycholesterol is an endogenous ligand for liver X receptor in cholesterol-loaded cells. J Biol Chem. 2001;276(42):38378–38387. doi: 10.1074/jbc.M105805200. [DOI] [PubMed] [Google Scholar]
- 28.Janowski BA, et al. An oxysterol signalling pathway mediated by the nuclear receptor LXR alpha. Nature. 1996;383(6602):728–731. doi: 10.1038/383728a0. [DOI] [PubMed] [Google Scholar]
- 29.Ory DS. Niemann-Pick type C: a disorder of cellular cholesterol trafficking. Biochim Biophys Acta. 2000;1529(1–3):331–339. doi: 10.1016/s1388-1981(00)00158-x. [DOI] [PubMed] [Google Scholar]
- 30.Liscum L, Ruggiero RM, Faust JR. The intracellular transport of low density lipoprotein-derived cholesterol is defective in Niemann-Pick type C fibroblasts. J Cell Biol. 1989;108(5):1625–1636. doi: 10.1083/jcb.108.5.1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sokol J, et al. Type C Niemann-Pick disease. Lysosomal accumulation and defective intracellular mobilization of low density lipoprotein cholesterol. J Biol Chem. 1988;263(7):3411–3417. [PubMed] [Google Scholar]
- 32.Wojtanik KM, Liscum L. The transport of low density lipoprotein-derived cholesterol to the plasma membrane is defective in NPC1 cells. J Biol Chem. 2003;278(17):14850–14856. doi: 10.1074/jbc.M300488200. [DOI] [PubMed] [Google Scholar]
- 33.Vance JE. Lipid imbalance in the neurological disorder, Niemann-Pick C disease. FEBS Lett. 2006;580(23):5518–5524. doi: 10.1016/j.febslet.2006.06.008. [DOI] [PubMed] [Google Scholar]
- 34.Thomas GH, et al. Correction of sphingomyelinase deficiency in Niemann-Pick type C fibroblasts by removal of lipoprotein fraction from culture media. J Inherit Metab Dis. 1989;12(2):139–151. doi: 10.1007/BF01800716. [DOI] [PubMed] [Google Scholar]
- 35.Higgins ME, et al. Niemann-Pick C1 is a late endosome-resident protein that transiently associates with lysosomes and the trans-Golgi network. Mol Genet Metab. 1999;68(1):1–13. doi: 10.1006/mgme.1999.2882. [DOI] [PubMed] [Google Scholar]
- 36.Neufeld EB, et al. The Niemann-Pick C1 protein resides in a vesicular compartment linked to retrograde transport of multiple lysosomal cargo. J Biol Chem. 1999;274(14):9627–9635. doi: 10.1074/jbc.274.14.9627. [DOI] [PubMed] [Google Scholar]
- 37.Watari H, et al. Mutations in the leucine zipper motif and sterol-sensing domain inactivate the Niemann-Pick C1 glycoprotein. J Biol Chem. 1999;274(31):21861–21866. doi: 10.1074/jbc.274.31.21861. [DOI] [PubMed] [Google Scholar]
- 38.Runz H, et al. NPC-db, a Niemann-Pick type C disease gene variation database. Hum Mutat. 2008;29(3):345–350. doi: 10.1002/humu.20636. [DOI] [PubMed] [Google Scholar]
- 39.Malathi K, et al. Mutagenesis of the putative sterol-sensing domain of yeast Niemann Pick C-related protein reveals a primordial role in subcellular sphingolipid distribution. J Cell Biol. 2004;164(4):547–556. doi: 10.1083/jcb.200310046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sym M, Basson M, Johnson C. A model for niemann-pick type C disease in the nematode Caenorhabditis elegans. Curr Biol. 2000;10(9):527–530. doi: 10.1016/s0960-9822(00)00468-1. [DOI] [PubMed] [Google Scholar]
- 41.Huang X, et al. A Drosophila model of the Niemann-Pick type C lysosome storage disease: dnpc1a is required for molting and sterol homeostasis. Development. 2005;132(22):5115–5124. doi: 10.1242/dev.02079. [DOI] [PubMed] [Google Scholar]
- 42.Pentchev PG, et al. The cholesterol storage disorder of the mutant BALB/c mouse. A primary genetic lesion closely linked to defective esterification of exogenously derived cholesterol and its relationship to human type C Niemann-Pick disease. J Biol Chem. 1986;261(6):2772–2777. [PubMed] [Google Scholar]
- 43.Brown DE, et al. Feline Niemann-Pick disease type C. Am J Pathol. 1994;144(6):1412–1415. [PMC free article] [PubMed] [Google Scholar]
- 44.Kuwamura M, et al. Type C Niemann-Pick disease in a boxer dog. Acta Neuropathol (Berl) 1993;85(3):345–348. doi: 10.1007/BF00227733. [DOI] [PubMed] [Google Scholar]
- 45.Mutka AL, et al. Secretion of sterols and the NPC2 protein from primary astrocytes. J Biol Chem. 2004;279(47):48654–48662. doi: 10.1074/jbc.M405345200. [DOI] [PubMed] [Google Scholar]
- 46.Friedland N, et al. Structure of a cholesterol-binding protein deficient in Niemann-Pick type C2 disease. Proc Natl Acad Sci U S A. 2003;100(5):2512–2517. doi: 10.1073/pnas.0437840100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ko DC, et al. The integrity of a cholesterol-binding pocket in Niemann-Pick C2 protein is necessary to control lysosome cholesterol levels. Proc Natl Acad Sci U S A. 2003;100(5):2518–2525. doi: 10.1073/pnas.0530027100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sleat DE, et al. Genetic evidence for nonredundant functional cooperativity between NPC1 and NPC2 in lipid transport. Proc Natl Acad Sci U S A. 2004;101(16):5886–5891. doi: 10.1073/pnas.0308456101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Loftus SK, et al. Murine model of Niemann-Pick C disease: mutation in a cholesterol homeostasis gene. Science. 1997;277(5323):232–235. doi: 10.1126/science.277.5323.232. [DOI] [PubMed] [Google Scholar]
- 50.Morris MD, et al. Lysosome lipid storage disorder in NCTR-BALB/c mice. I. Description of the disease and genetics. Am J Pathol. 1982;108(2):140–149. [PMC free article] [PubMed] [Google Scholar]
- 51.Rimkunas VM, et al. In vivo antisense oligonucleotide reduction of NPC1 expression as a novel mouse model for Niemann Pick type C- associated liver disease. Hepatology. 2008;47(5):1504–1512. doi: 10.1002/hep.22327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Walkley SU. Pyramidal neurons with ectopic dendrites in storage diseases exhibit increased GM2 ganglioside immunoreactivity. Neuroscience. 1995;68(4):1027–1035. doi: 10.1016/0306-4522(95)00208-z. [DOI] [PubMed] [Google Scholar]
- 53.Xie C, et al. Cholesterol is sequestered in the brains of mice with Niemann-Pick type C disease but turnover is increased. J Neuropathol Exp Neurol. 2000;59(12):1106–1117. doi: 10.1093/jnen/59.12.1106. [DOI] [PubMed] [Google Scholar]
- 54.Zervas M, Dobrenis K, Walkley SU. Neurons in Niemann-Pick disease type C accumulate gangliosides as well as unesterified cholesterol and undergo dendritic and axonal alterations. J Neuropathol Exp Neurol. 2001;60(1):49–64. doi: 10.1093/jnen/60.1.49. [DOI] [PubMed] [Google Scholar]
- 55.Takikita S, et al. Perturbed myelination process of premyelinating oligodendrocyte in Niemann-Pick type C mouse. J Neuropathol Exp Neurol. 2004;63(6):660–673. doi: 10.1093/jnen/63.6.660. [DOI] [PubMed] [Google Scholar]
- 56.Higashi Y, et al. Cerebellar degeneration in the Niemann-Pick type C mouse. Acta Neuropathol (Berl) 1993;85(2):175–184. doi: 10.1007/BF00227765. [DOI] [PubMed] [Google Scholar]
- 57.Yu W, et al. Neurodegeneration in heterozygous Niemann-Pick type C1 (NPC1) mouse: implication of heterozygous NPC1 mutations being a risk for tauopathy. J Biol Chem. 2005;280(29):27296–27302. doi: 10.1074/jbc.M503922200. [DOI] [PubMed] [Google Scholar]
- 58.Gondre-Lewis MC, McGlynn R, Walkley SU. Cholesterol Accumulation in NPC1-Deficient Neurons Is Ganglioside Dependent. Curr Biol. 2003;13(15):1324–1329. doi: 10.1016/s0960-9822(03)00531-1. [DOI] [PubMed] [Google Scholar]
- 59.Cox T, et al. Novel oral treatment of Gaucher's disease with N-butyldeoxynojirimycin (OGT 918) to decrease substrate biosynthesis. Lancet. 2000;355(9214):1481–1485. doi: 10.1016/S0140-6736(00)02161-9. [DOI] [PubMed] [Google Scholar]
- 60.Zervas M, et al. Critical role for glycosphingolipids in Niemann-Pick disease type C. Curr Biol. 2001;11(16):1283–1287. doi: 10.1016/s0960-9822(01)00396-7. [DOI] [PubMed] [Google Scholar]
- 61.Patterson MC, et al. Miglustat for treatment of Niemann-Pick C disease: a randomised controlled study. Lancet Neurol. 2007;6(9):765–772. doi: 10.1016/S1474-4422(07)70194-1. [DOI] [PubMed] [Google Scholar]
- 62.Mellon SH. Neurosteroid regulation of central nervous system development. Pharmacol Ther. 2007;116(1):107–124. doi: 10.1016/j.pharmthera.2007.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Griffin LD, et al. Niemann-Pick type C disease involves disrupted neurosteroidogenesis and responds to allopregnanolone. Nat Med. 2004;10(7):704–711. doi: 10.1038/nm1073. [DOI] [PubMed] [Google Scholar]
- 64.Langmade SJ, et al. Pregnane X receptor (PXR) activation: a mechanism for neuroprotection in a mouse model of Niemann-Pick C disease. Proc Natl Acad Sci U S A. 2006;103(37):13807–13812. doi: 10.1073/pnas.0606218103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Frolov A, et al. Cholesterol overload promotes morphogenesis of a Niemann-Pick C (NPC)-like compartment independent of inhibition of NPC1 or HE1/NPC2 function. J Biol Chem. 2001;276(49):46414–46421. doi: 10.1074/jbc.M108099200. [DOI] [PubMed] [Google Scholar]
- 66.Repa JJ, et al. Liver X receptor activation enhances cholesterol loss from the brain, decreases neuroinflammation, and increases survival of the NPC1 mouse. J Neurosci. 2007;27(52):14470–14480. doi: 10.1523/JNEUROSCI.4823-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ko DC, et al. Cell-autonomous death of cerebellar purkinje neurons with autophagy in Niemann-Pick type C disease. PLoS Genet. 2005;1(1):81–95. doi: 10.1371/journal.pgen.0010007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Klionsky DJ, et al. A unified nomenclature for yeast autophagy-related genes. Dev Cell. 2003;5(4):539–545. doi: 10.1016/s1534-5807(03)00296-x. [DOI] [PubMed] [Google Scholar]
- 69.Xie Z, Klionsky DJ. Autophagosome formation: core machinery and adaptations. Nat Cell Biol. 2007;9(10):1102–1109. doi: 10.1038/ncb1007-1102. [DOI] [PubMed] [Google Scholar]
- 70.Ohsumi Y, Mizushima N. Two ubiquitin-like conjugation systems essential for autophagy. Semin Cell Dev Biol. 2004;15(2):231–236. doi: 10.1016/j.semcdb.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 71.Kabeya Y, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. Embo J. 2000;19(21):5720–5728. doi: 10.1093/emboj/19.21.5720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tanida I, Ueno T, Kominami E. Human light chain 3/MAP1LC3B is cleaved at its carboxyl-terminal Met121 to expose Gly120 for lipidation and targeting to autophagosomal membranes. J Biol Chem. 2004;279(46):47704–47710. doi: 10.1074/jbc.M407016200. [DOI] [PubMed] [Google Scholar]
- 73.Tanida I, et al. Lysosomal turnover, but not a cellular level, of endogenous LC3 is a marker for autophagy. Autophagy. 2005;1(2):84–91. doi: 10.4161/auto.1.2.1697. [DOI] [PubMed] [Google Scholar]
- 74.Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290(5497):1717–1721. doi: 10.1126/science.290.5497.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Noda T, Ohsumi Y. Tor, a phosphatidylinositol kinase homologue, controls autophagy in yeast. J Biol Chem. 1998;273(7):3963–3966. doi: 10.1074/jbc.273.7.3963. [DOI] [PubMed] [Google Scholar]
- 76.Yamamoto A, Cremona ML, Rothman JE. Autophagy-mediated clearance of huntingtin aggregates triggered by the insulin-signaling pathway. J Cell Biol. 2006;172(5):719–731. doi: 10.1083/jcb.200510065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Nixon RA. Autophagy, amyloidogenesis and Alzheimer disease. J Cell Sci. 2007;120(Pt 23):4081–4091. doi: 10.1242/jcs.019265. [DOI] [PubMed] [Google Scholar]
- 78.Pan T, et al. The role of autophagy-lysosome pathway in neurodegeneration associated with Parkinson's disease. Brain. 2008 doi: 10.1093/brain/awm318. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 79.Ravikumar B, et al. Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet. 2004;36(6):585–595. doi: 10.1038/ng1362. [DOI] [PubMed] [Google Scholar]
- 80.Kiselyov K, et al. Autophagy, mitochondria and cell death in lysosomal storage diseases. Autophagy. 2007;3(3):259–262. doi: 10.4161/auto.3906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Williams A, et al. Aggregate-prone proteins are cleared from the cytosol by autophagy: therapeutic implications. Curr Top Dev Biol. 2006;76:89–101. doi: 10.1016/S0070-2153(06)76003-3. [DOI] [PubMed] [Google Scholar]
- 82.Kuma A, et al. The role of autophagy during the early neonatal starvation period. Nature. 2004;432(7020):1032–1036. doi: 10.1038/nature03029. [DOI] [PubMed] [Google Scholar]
- 83.Komatsu M, et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature. 2006;441(7095):880–884. doi: 10.1038/nature04723. [DOI] [PubMed] [Google Scholar]
- 84.Shimizu S, et al. Role of Bcl-2 family proteins in a non-apoptotic programmed cell death dependent on autophagy genes. Nat Cell Biol. 2004;6(12):1221–1228. doi: 10.1038/ncb1192. [DOI] [PubMed] [Google Scholar]
- 85.Liao G, et al. Cholesterol accumulation is associated with lysosomal dysfunction and autophagic stress in Npc1 −/− mouse brain. Am J Pathol. 2007;171(3):962–975. doi: 10.2353/ajpath.2007.070052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pacheco CD, Kunkel R, Lieberman AP. Autophagy in Niemann-Pick C disease is dependent upon Beclin-1 and responsive to lipid trafficking defects. Hum Mol Genet. 2007;16(12):1495–1503. doi: 10.1093/hmg/ddm100. [DOI] [PubMed] [Google Scholar]
- 87.Cheng J, et al. Cholesterol depletion induces autophagy. Biochem Biophys Res Commun. 2006;351(1):246–252. doi: 10.1016/j.bbrc.2006.10.042. [DOI] [PubMed] [Google Scholar]
- 88.Lajoie P, et al. The lipid composition of autophagic vacuoles regulates expression of multilamellar bodies. J Cell Sci. 2005;118(Pt 9):1991–2003. doi: 10.1242/jcs.02324. [DOI] [PubMed] [Google Scholar]
- 89.Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132(1):27–42. doi: 10.1016/j.cell.2007.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Raben N, Roberts A, Plotz PH. Role of autophagy in the pathogenesis of Pompe disease. Acta Myol. 2007;26(1):45–48. [PMC free article] [PubMed] [Google Scholar]
- 91.Vergarajauregui S, et al. Autophagic Dysfunction in Mucolipidosis Type Iv Patients. Hum Mol Genet. 2008 doi: 10.1093/hmg/ddn174. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Settembre C, et al. A block of autophagy in lysosomal storage disorders. Hum Mol Genet. 2008;17(1):119–129. doi: 10.1093/hmg/ddm289. [DOI] [PubMed] [Google Scholar]
- 93.Cao Y, et al. Autophagy is disrupted in a knock-in mouse model of juvenile neuronal ceroid lipofuscinosis. J Biol Chem. 2006;281(29):20483–20493. doi: 10.1074/jbc.M602180200. [DOI] [PubMed] [Google Scholar]
- 94.Beck M. New therapeutic options for lysosomal storage disorders: enzyme replacement, small molecules and gene therapy. Hum Genet. 2007;121(1):1–22. doi: 10.1007/s00439-006-0280-4. [DOI] [PubMed] [Google Scholar]
- 95.Hsu YS, et al. Niemann-Pick disease type C (a cellular cholesterol lipidosis) treated by bone marrow transplantation. Bone Marrow Transplant. 1999;24(1):103–107. doi: 10.1038/sj.bmt.1701826. [DOI] [PubMed] [Google Scholar]
- 96.Escolar ML, et al. Transplantation of umbilical-cord blood in babies with infantile Krabbe's disease. N Engl J Med. 2005;352(20):2069–2081. doi: 10.1056/NEJMoa042604. [DOI] [PubMed] [Google Scholar]
- 97.Patterson MC, et al. The effect of cholesterol-lowering agents on hepatic and plasma cholesterol in Niemann-Pick disease type C. Neurology. 1993;43(1):61–64. doi: 10.1212/wnl.43.1_part_1.61. [DOI] [PubMed] [Google Scholar]
- 98.Sylvain M, et al. Magnetic resonance spectroscopy in Niemann-Pick disease type C: correlation with diagnosis and clinical response to cholestyramine and lovastatin. Pediatr Neurol. 1994;10(3):228–232. doi: 10.1016/0887-8994(94)90028-0. [DOI] [PubMed] [Google Scholar]
- 99.Jeyakumar M, et al. Enhanced survival in Sandhoff disease mice receiving a combination of substrate deprivation therapy and bone marrow transplantation. Blood. 2001;97(1):327–329. doi: 10.1182/blood.v97.1.327. [DOI] [PubMed] [Google Scholar]
- 100.Gaumann A, Schlitt HJ, Geissler EK. Immunosuppression and tumor development in organ transplant recipients: the emerging dualistic role of rapamycin. Transpl Int. 2008;21(3):207–217. doi: 10.1111/j.1432-2277.2007.00610.x. [DOI] [PubMed] [Google Scholar]