Abstract
Neuronal nicotinic receptor α4 subunits associated with nicotinic α4β2 receptors are phosphorylated by cAMP-dependent protein kinase (PKA) and protein kinase C (PKC), but the stages of receptor formation during which phosphorylation occurs and the functional consequences of kinase activation are unknown. SH-EP1 cells transfected with DNAs coding for human α4 and/or β2 subunits were incubated with 32Pi, and PKA or PKC were activated by forskolin or phorbol 12,13-dibutyrate, respectively. Immunoprecipitation and immunoblotting of proteins from cells expressing α4β2 receptors or only α4 subunits were used to identify free α4 subunits, and α4 subunits present in immature α4β2 complexes and mature α4β2 pentamers containing complex carbohydrates. In the absence of kinase activation, phosphorylation of α4 subunits associated with mature pentamers was 3 times higher than subunits associated with immature complexes. PKA and PKC activation increased phosphorylation of free α4 subunits on different serine residues; only PKC activation phosphorylated subunits associated with mature α4β2 receptors. Activation of both PKA and PKC increased the density of membrane-associated receptors, but only PKC activation increased peak membrane currents. PKA and PKC activation also phosphorylated β2 subunits associated with mature α4β2 receptors. Results indicate that activation of PKA and PKC leads to the phosphorylation α4β2 receptors at different stages of receptor formation and maturation and has differential effects on the expression and function of α4β2 receptors.
Keywords: protein kinase A, protein kinase C, neuronal nicotinic receptor, phosphorylation, receptor formation, receptor function
Neuronal nicotinic acetylcholine (ACh) receptors composed of α4 and β2 subunits are widely distributed throughout the brain and represent the most abundant nicotinic receptor type in the brain (Zoli et al., 1995). These α4β2 receptors participate in numerous biochemical and physiological processes, have been implicated in several neurological and behavioral disorders including nocturnal frontal lobe epilepsy and Alzheimer’s disease, and may be responsible for the rewarding and addictive effects of nicotine (Picciotto et al., 2001; Tapper et al., 2004).
Studies have suggested that the expression and function of α4β2 receptors are regulated post-translationally through phosphorylation/dephosphorylation mechanisms involving both cAMP-dependent protein kinase (PKA) and protein kinase C (PKC) (Rothhut et al., 1996; Eilers et al., 1997; Gopalakrishnan et al., 1997; Fenster et al., 1999; Jeanclos et al., 2001; Nashmi et al., 2003; Exley et al., 2006). Initial studies using M10 fibroblasts stably transfected with chicken α4 and β2 subunit cDNAs indicated that activation of PKA by forskolin increased the number of cell surface receptors 2-fold (Rothhut et al., 1996). Furthermore, recent findings using rat α4β2 receptors expressed in tsA201 cells have suggested that this increase may be attributed to the PKA-dependent phosphorylation of α4 subunits, which promotes association with the 14-3-3 chaperone protein, increasing steady-state levels of the subunit, and resulting in a 20% increase in cell surface receptor expression (Jeanclos et al., 2001). Similar studies using mouse α4β2 receptors expressed in HEK293T cells have shown that activation of PKC also promotes the assembly and translocation of receptors to the cell surface (Nashmi et al., 2003) although the role of α4 subunit phosphorylation in mediating this action was not determined.
The phosphorylation of α4 subunits following activation of PKA and PKC has been demonstrated in several systems and evidence indicates that these kinases phosphorylate both unique and common sites located within the M3/M4 cytoplasmic domain of the subunit (Nakayama et al., 1993; Hsu et al., 1997; Wecker et al., 2001; Viseshakul et al., 1998; Guo and Wecker, 2002; Pacheco et al., 2003). Further, recent studies have suggested that these kinases may have a differential effect on the α4 subunit protein during different stages in the maturation of the α4β2 pentamer. Pollock et al. (2007) have shown that two forms of α4 subunit protein can be immunoprecipitated from SH-EP1 cells transiently transfected with the DNAs coding for human α4 and β2 subunits, a predominant 71–75 kDa protein and a less abundant protein at 80–85 kDa, the latter present most often in samples containing large amounts of protein. Further, activation of both PKA and PKC increased phosphorylation of the major 71–75 kDa protein, whereas phosphorylation of the higher molecular weight α4 protein was unaltered following activation of PKA, but increased following activation of PKC. Based on studies suggesting that the lower molecular weight α4 protein represents immature α4 subunit species while the higher molecular weight protein represents subunits isolated from mature α4β2 pentamers (Whiting and Lindstrom 1988; Vallejo et al. 2005; Sallette et al. 2005), results suggest that PKA activation may increase phosphorylation of α4 subunits prior to formation of mature receptors, whereas PKC activation may have a dual action, phosphorylating α4 subunits both prior to after formation of the mature receptor. Thus, these studies investigated the phosphorylation of α4 subunits by PKA and PKC at distinct stage(s) in the assembly and maturation of α4β2 receptors, and determined whether activation of PKA and PKC have different functional consequences.
EXPERIMENTAL PROCEDURES
Materials
The monoclonal antibodies used for immunoprecipitation included mAb299 (M218) raised against the extracellular loop of the rat nicotinic receptor α4 subunit and mAb290 (N8533) raised against the extracellular loop of the rat nicotinic receptor β2 subunit, both of which cross react with the human subunits (Sigma-Aldrich Co.). The polyclonal antibodies used for immunoblotting were H-133 (sc-5591), a rabbit polyclonal antibody raised against a recombinant protein corresponding to amino acids 342–474 of the human α4 subunit, H-92 (sc-11372), a rabbit polyclonal antibody raised against a recombinant protein corresponding to amino acids 342–433 of the human β2 subunit, and goat anti-rabbit IgG horseradish peroxidase conjugated secondary antibody (sc-2030), purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA). Sulfo-NHS-LC-Biotin was purchased from Pierce (Rockford, IL), endoglycosidase-H (Endo-H) and peptide N-glycanase F (PNGase F) were bought from New England Biolabs (Beverly, MA), and Lipofectamine™ 2000 and cell growth media were purchased from Invitrogen Corporation (Carlsbad, CA). Extravidin-HRP (E2886), Protein-G Sepharose and molecular markers were purchased from Sigma-Aldrich Co., and thin-layer chromatography (TLC) plates were obtained from VWR International Inc. (Bristol, CT). [32Pi] and [3H]cytisine were purchased from Perkin Elmer Life and Analytical Sciences Inc. (Boston, MA), polyvinylidene fluoride (PVDF) membranes (Immobilon-P) from Millipore Corporation (Bedford, MA), and XAR film from Eastman Kodak Co. (Rochester, NY). Enhanced chemiluminescence (ECL) reagents were purchased from Amersham Biosciences (Piscataway, NJ), basic electrophoresis chemicals from Bio-Rad Laboratories, Inc. (Hercules, CA), and sequencing grade trypsin from Roche Diagnostics Corp. (Indianapolis, IN). Human α4 and β2 acetylcholine receptor subunit cDNA clones in pSP64 (kindly provided by Dr. Jon Lindstrom, Department of Neuroscience, University of Pennsylvania, Philadelphia, PA) were used to make constructs in pcDNA 3.1/hygro(−) and pcDNA 3.1 zeo(+), respectively (Pollock et al., 2007). SH-EP1 and SH-EP1-hα4β2 cells were kindly provided by Dr. Ron Lukas (Barrow Neurological Institute, Phoenix, AZ).
Transfection, 32Pi labeling, kinase activation, and preparation of whole cell lysates
SH-EP1 cells were grown in 60 mm dishes at 37°C in 5% CO2 and maintained in Dulbecco’s modified Eagle’s medium with high glucose (DMEM) supplemented with 5% fetal clone II, 10% horse serum, 1 mM sodium pyruvate, 8 mM L-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, and 50 μg/ml amphotericin B. Transfections were performed with cells at 90–95% confluency in Opti-MEM. Cells were incubated with 20 μl of Lipofectamine mixed with 8 μg of DNA for α4 only, β2 only, or a mixture (α:β, 2:3) for 2–3 hours at 37°C and for an additional 3–4 hours at 32°C. The Lipofectamine was removed and cells were incubated with growth media lacking antibiotics for an additional 18–20 hours at 32°C, for a total incubation time of 24 hours.
To label endogenous ATP stores, cells were incubated with 32Pi (1 or 2 mCi/ml) for 4 hours in phosphate-free DMEM. To produce maximal activation of PKA or PKC, 10 μM forskolin or 200 nM phorbol 12,13-dibutyrate (PDBu) in 0.1% DMSO were added during the final 15 or 30 minutes, respectively. These concentrations and times of incubation were chosen based on studies indicating that these parameters led to maximal activation of PKA by forskolin and PKC by PDBu without altering the amount of radioactivity taken up by cells during the labeling period. Whole cell lysates were prepared by washing the cells (5 x) with ice-cold phosphate buffered saline (PBS) followed by incubation for 30 minutes at 4°C in 500 μl of potassium phosphate (200 mM) homogenization/lysis buffer (pH 7.4) containing 150 mM NaCl, 10 mM EDTA, 10 mM EGTA, 10 mM β-glycerophosphate, 50 mM NaF, 1 mM sodium vanadate, 0.1 mM phenylmethylsulfonyl fluoride, 1 μg/ml pepstatin A, 10 μg/ml leupeptin, 10 U/ml aprotinin, and 2% Triton X-100 or 1% Lubrol. Samples were transferred into microfuge tubes, were triturated (20 strokes) using a 1 ml syringe with a 26-gauge needle, were further solubilized for 30 minutes at 4°C on a rotator, and were centrifuged at 100,000 × g for 10 minutes; the supernatants were withdrawn and transferred to tubes for further experimentation.
Immunoprecipitations
α4 subunit protein was immunoprecipitated from whole cell lystates using both the anti-α4 monoclonal antibody mAb299 and the anti-β2 monoclonal antibody mAb290. Studies have shown that mAb299 immunoprecipitates subunits associated only with immature receptors from transfected HEK293 cells expressing α4β2 receptors, whereas mAb290 immunoprecipitates subunits associated with both immature and plasma membrane-associated mature receptors (Sallette et al., 2005). Whole cell lysates were pre-cleared with 15 μl protein-G Sepharose for 30 minutes at 4°C. The samples were incubated with primary antibody (5 μg of mAb299 or mAb290) overnight with rocking at 4°C, followed by rocking with 30 μl protein-G sepharose for 3–4 hours. The protein/antibody/bead complexes were washed five times with 50 mM MOPS buffer (pH 7.2) containing 1 mM EDTA, 1 mM EGTA, and 0.2% Triton X-100, protein was eluted by boiling in Laemmli buffer (Laemmli, 1970), and samples were centrifuged at 2,000 × g for 5 minutes to remove the protein G beads.
For sequential immunoprecipitations, samples were incubated with either mAb290 or mAb299 and protein-G sepharose as above, and centrifuged at 2,000 × g for 5 minutes. The supernatants were reincubated with antibody and protein-G sepharose and the protein/antibody/bead complexes were processed as described. This procedure was repeated 2 more times for a total of 4 sequential immunoprecipitations.
To isolate α4 subunits that were not associated with β2 subunits from cells expressing α4β2 receptors, all α4β2 complexes were immunoprecipitated from pre-cleared cell lysates by sequential incubation with mAb290 and excess (10 μg) H-92, a polyclonal anti-β2 antibody that is not conformation or assembly dependent. Samples were incubated with mAb290 and protein-G sepharose as above, centrifuged at 2,000 × g for 5 minutes, and the supernatants incubated with H-92 and protein-G sepharose. Samples were centrifuged at 2,000 × g for 5 minutes, and the supernatant from this second immunoprecipitation was mixed with additional protein-G sepharose to remove excess antibody, and centrifuged. The supernatants were incubated with mAb299 to immunoprecipitate α4 subunits, and samples processed as described.
Surface protein biotinylation and endoglycosidase cleavage
Twenty-four hours following transfections, cells were washed with ice cold PBS and incubated in 2 ml of ice cold PBS (pH 7.5) containing 0.5 mM Sulfo-NHS-LC-Biotin for 30 minutes at 4°C to biotinylate cell surface receptors. Reagent was removed by washing the cells with PBS containing 100 mM glycine, transferring them to microfuge tubes and washing twice more. Samples were centrifuged at 1,200 × g for 5 minutes, the cells were resuspended in 500 μl of homogenization/lysis buffer, and whole cell lysates prepared as described. Receptor complexes were immunoprecipitated from biotinylated and non-biotinylated lysates with mAb290 and protein eluted by boiling for 10 minutes with 65 μl Laemmli buffer (Laemmli, 1970) containing 1% sodium dodecyl sulfate (SDS). The eluted samples were divided into three 20 μl aliquots. One aliquot served as the control, and the others were subjected to endoglycosidase cleavage using both PNGase, which removes all N-linked carbohydrates, and Endo-H, which removes only high mannose and some hybrid types of N-linked carbohydrates. Thus, the second aliquot was mixed with 2 μl of Endo-H, and the third was mixed with 2 μl of PNGase F and 1 μl of 10% NP-40 (nonyl phenoxylpolyethoxyethanol), a nonionic, non-denaturing detergent that protects the PNGase F from inhibition by the SDS present. The samples were mixed thoroughly and incubated at 37°C for one hour, followed by the addition of 5 μl of 20% SDS. Samples were frozen at −80°C until analysis by immunoblotting.
Autoradiography and immunoblotting
Immunoprecipitated proteins were separated on a 7.5% SDS-polyacrylamide gel (SDS-PAGE) and transferred to PVDF membranes. [32P]-labeled α4 and β2 subunit proteins were visualized by autoradiography by exposing the membranes to Kodak XAR film in the presence of intensifying screens at −80°C for 2–24 hours prior to immunoblotting.
For immunoblots, the PVDF membranes were blocked with Tris-buffered saline (TBS) containing 0.05% Tween-20 and 5% non-fat dry milk for 2 hours at 24°C. For samples in which both α4 and β2 subunit proteins were determined, membranes were cut horizontally just above the 59 kDa marker between the α4 and β2 protein bands. Membranes were incubated with H-133 and H-92 (each 200 μg/ml at 1:2000 dilution) in TBS/0.05% Tween/0.5% non-fat dry milk for 1 hour at 24°C to immunodetect the α4 and β2 subunit proteins, respectively. These polyclonal antibodies have been shown to react specifically with the proteins of interest following immunoprecipitation with the monoclonal antibodies and were used to avoid high backgrounds obtained when the monoclonal antibodies are used for both immunoprecipitation and immunodetection (Pacheco et al., 2003; Pollock et al., 2007). The membranes were washed and incubated with horseradish peroxidase conjugated goat anti-rabbit secondary antibody (400 μg/ml at 1:2000 dilution) for 30 minutes, and the α4 and β2 signals were visualized using ECL. Autoradiographs and immunoblots were quantitated by densitometry using a Bio-Rad Imaging Densitometer with Multi-Analyst software (Bio-Rad Laboratories). Relative phosphorylation was calculated by normalizing the densitometric signals from the autoradiographs to the immunoblots.
Phosphopeptide mapping
Two-dimensional (2D) phosphopeptide mapping of the immunoprecipitated [32P]-labeled α4 subunits was performed using standard techniques (Boyle et al., 1991) as described in detail by Pollock et al. (2007). Briefly, PVDF membrane bands containing the [32P]-labeled α4 protein were excised, digested with trypsin, spotted onto cellulose TLC plates, and subjected to electrophoresis in the first dimension and ascending chromatography in the second dimension. Tryptic phosphopeptide patterns were visualized by exposing the plates to XAR film with intensifying screens. Due to large differences in the amount of phosphorylated material obtained under different experimental conditions, within each set of experiments, samples containing equal counts and not equal amounts of protein were analyzed.
Radioligand binding
SH-EP1-hα4β2 cells were maintained (in 60 mm dishes) at 37°C in 5% CO2 and grown in DMEM (high glucose) containing 5% Fetal Clone II, 10% horse serum, 1 mM sodium pyruvate, 8 mM L-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 0.05 mg/ml amphotericin B, 0.5 mg/ml zeocin and 0.4 mg/ml hygromycin B. Cells were kept in continuous culture, fed every 2–3 days, and used at 85% confluency. Cells were incubated in control media or media containing 10 μM forskolin or 200 nM PDBu (dissolved in 0.1% DMSO) for 24 hours. Following incubation, cells were washed 3 times with PBS, and transferred to a Dounce homogenizer. Membrane fractions were prepared by homogenizing cells in 50 mM Tris-HCl buffer (pH 7.0) containing 120 mM NaCl, 5 mM KCl, 1 mM MgCl2 and 2.5 mM CaCl2. Cell lysates were centrifuged at 500 × g for 5 minutes at 4°C and the resultant supernatants were recentrifuged at 20,800 × g for 30 minutes at 4°C; the pellets were washed 3 times and resuspended in Tris-HCl buffer. The binding reaction was initiated by the addition of membrane preparations (5–10 μg protein) to triplicate sets of tubes containing 2.5 nM [3H]cytisine (32–35 Ci/mmole), representing 4 × Kd (Pacheco et al., 2001) and Tris-HCl buffer. Tubes were incubated for 2 hours on ice and bound ligand separated from free ligand by rapid filtration over Whatman GF/B filters, which were pre-soaked in Tris-HCl to minimize non-specific binding. Tubes and filters were rinsed twice with ice-cold Tris-HCl buffer, filters placed in scintillation vials and 8 ml of scintillation fluid added to each vial. Samples were shaken mechanically for 1 hour and radioactivity was determined in a Beckman LS-6000 scintillation counter (Beckman Counter, Fullerton, CA). Non-specific binding was measured in the presence of 10 μM nicotine and specific binding calculated as the difference between total and non-specific binding. Experiments were performed using different passages of cultured cells.
Electrophysiology
SH-EP1-hα4β2 cells were plated on glass cover slips and maintained in DMEM (high glucose) as above. Cover slips were transferred to a recording chamber (0.5 ml) mounted on an inverted phase-contrast microscope that allowed isolated cells to be identified. Membrane currents were studied under voltage-clamp mode using the amphotericin B perforated-patch variation of the whole-cell patch-clamp technique. Patch electrodes were pulled from thin-walled borosilicate glass (World Precision Instruments Inc., Sarasota, FL) using a Sutter Instrument (Novato, CA) P-87 pipette puller and had resistances of 1–1.5 MΩ. Membrane currents were amplified and filtered (5 kHz) using an Axopatch 200B patch-clamp amplifier, digitized with a Digidata 1322 B, and acquired using Clampex 8.2 (Axon Instruments, Foster City, CA). The external solution for whole-cell recordings was physiological saline solution containing 140 mM NaCl, 3 mM KCl, 2.5 mM CaCl2, 1.6 mM MgCl2, 7.7 mM glucose and 10 mM HEPES (pH 7.2 with 1 M NaOH). The intracellular solution contained 75 mM CsSO4, 55 mM CsCl, 5 mM MgSO4, 10 mM HEPES, 360 mg/ml amphotericin B, and 0.6% DMSO (pH 7.2 with N-methyl-D-glucamine). ACh was applied via a gravity-fed rapid application system. The flow of solution was controlled by solenoid valves (General Valve, Fairfield, NJ) using a Master 8 channel stimulator (A.M.P.I., Jerusalem, Israel) and was directed by a piezoelectric bimorph connected to a voltage generator (Burleigh Instruments, Inc., Fishers, NY). The rate of solution change was determined to be less than 5 msec as measured by liquid junction potential changes. Increasing concentrations of ACh were added every 4 minutes, a time interval determined to be sufficient for recovery from the desensitization induced by prior exposure to agonist.
Data Analysis
Where appropriate, data are presented as mean ± s.e.m. The Student’s t-test was used for comparisons involving two groups. ANOVA was used for more than 2 groups, and in those instances where significant (p < 0.05) main effects were noted, individual group differences were determined by Newman-Keuls test. A level of p<0.05 was accepted as evidence of a statistically significant effect.
For electrophysiological experiments, data were analyzed using SigmaPlot 8 and SigmaStat 3.1 (Systat Software Inc., Point Richmond, CA). Data were normalized to peak responses obtained for each cell at 3 mM ACh, and concentration-response data were fit using the single- [y = (Emax)([x]n)/(EC50n + [x]n)] or two-site [y = (Emax1)([x]n1)/(EC501)n1 + [x]n1) + (Emax2)([x]n2)/(EC502)n2 + [x]n2)] Langmuir-Hill Equations, where Emax represents the maximal response, [x] is the agonist concentration, EC50 is the apparent macroscopic equilibrium constant, and n is the Hill Coefficient.
RESULTS
Identification and cellular localization of α4 and β2 subunits at different stages of receptor assembly and maturation
Initial experiments established and validated conditions to separate and identify α4 and β2 subunits at distinct stages in the assembly and maturation of α4β2 receptors. The first experiment compared the ability of mAb299 and mAb290 to immunoprecipitate subunits from whole cell lysates prepared from SH-EP1 cells transfected with α4 and β2 subunit DNAs either separately or in combination. Results (Fig. 1[A]) demonstrate that when samples were immunoprecipitated with mAb299 and probed for α4, a predominant signal corresponding to an apparent molecular weight of 70–75 kDa and a minor signal at approximately 66 kDa (observed only when samples contained large amounts of protein) were apparent for samples from cells expressing either α4 subunits only (lane 1) or both α4 and β2 subunits (lane 2). In contrast, when samples were immunoprecipitated with mAb290, cells expressing both subunits (lane 4) exhibited both the 70–75 and 66 kDa α4 protein bands, as well as a higher molecular weight signal at 80–85 kDa. No α4 signals were apparent in samples from cells expressing only β2 subunits (lane 3). When samples immunoprecipitated with mAb299 were probed for β2, a single signal was apparent at 47 kDa in cells expressing both subunits (lane 2); no signal was present in cells expressing only the α4 subunit (lane 1). When mAb290 was used for immunoprecipitation, samples from cells expressing both subunits (lane 4) exhibited both the 47 kDa β2 protein and a higher molecular weight signal at 50 kDa. Samples from cells expressing only β2 subunits (lane 3) yielded a very faint signal at 47 kDa. No signals were present for either α4 or β2 in samples from untransfected cells (data not shown), confirming the specificity of the antibodies used.
Fig. 1.
Immunoprecipitation of human nicotinic receptor α4 and β2 subunits by mAb290 and mAb299. SH-EP1 cells were transfected with DNAs coding for α4 and β2 subunits alone or in combination (α4:β2, 2:3). [A] A representative immunoblot identifying α4 and β2 subunits immunoprecipitated by mAb299 versus mAb290. Whole cell lysates were prepared and proteins were immunoprecipitated with either mAb299 (directed against α4 subunits) or mAb290 (directed against β2 subunits). Samples were separated on SDS-PAGE gels and transferred to PVDF membranes. The membranes were cut just above the 59 kDa marker and the top half was probed for α4 with the H-133 antibody and the bottom half was probed for β2 with the H-92 antibody; molecular weight standards are indicated. [B] Representative immunoblots from non-biotinylated and biotinylated proteins prior to and following endoglycosidase cleavage. Surface proteins were labeled with Sulfo-NHS-LC-Biotin 24 hours following transfections, whole cell lysates prepared, α4β2 complexes immunoprecipitated with mAb290, and samples separated on SDS-PAGE gels and transferred to PVDF membranes. Non-biotinylated samples were probed for α4 and β2 subunits with H-133 and H-92, respectively, and biotinylated proteins were probed with peroxidase-conjugated extravidin. For each condition, samples were untreated (lane 1) or digested with the endoglycosidases Endo-H (lane 2) or PNGase F (lane 3). α4m and β2m represent subunits immunoprecipitated from fully processed mature pentamers present mostly in the plasma membrane, and α4i and β2i represent subunits immunoprecipitated from immature complexes located in the endoplasmic reticulum. [C] Sequential immunoprecipitations and detergent comparison. Following transfection, whole cell lysates were prepared in buffer containing either 2% Triton X-100 or 1% Lubrol. Subunits were immunoprecipitated sequentially with the indicated antibodies, and samples were separated on SDS-PAGE gels and transferred to PVDF membranes. The membranes were cut just above the 59 kDa marker and the top half was probed for α4 with the H-133 antibody and the bottom half was probed for β2 with the H-92 antibody.
It is possible that the higher molecular weight signals for both α4 and β2 subunits present in samples immunoprecipitated with mAb290 (80–85 kDa for α4 and 50 kDa for β2) correspond to subunits isolated from mature α4β2 receptors with fully processed sugars, while the lower molecular weight signals (70–75 kDa for α4 and 47 kDa for β2) arise from immature α4β2 complexes containing high mannose (Sallette et al., 2005). The minor third α4 band of 66 kDa likely corresponds to a fully deglycosylated degradation product (Sallette et al., 2005). To confirm the identity of these signals, lysates from cells expressing both subunits were prepared, and α4β2 complexes were either immunoprecipitated immediately with mAb290 followed by digestion with Endo-H or PNGase F to remove predominantly high mannose carbohydrates or all N-linked carbohydrates, respectively, or were surface biotinylated with Sulfo-NHS-LC-biotin prior to immunoprecipitation and glycosidase digestion, and probed with extravidin-conjugated peroxidase to recognize only biotinylated samples (Fig. 1[B]). Immunoblots of non-biotinylated samples indicate that the higher molecular weight species of both α4 and β2 were partially Endo-H resistant, but completely digested by PNGase F, indicating that the proteins represented by these bands contain a mixture of high mannose and complex oligosaccharides likely corresponding to subunits isolated from fully processed mature forms of the receptor associated with the trans-Golgi and plasma membrane (α4m and β2m). This was confirmed by the immunoblots for the biotinylated samples indicating that extravidin-conjugated peroxidase recognized only the higher molecular weight protein bands, corresponding to subunits isolated from cell surface, fully-processed mature receptors, in agreement with Vallejo et al. (2005). The lower molecular weight species of the subunits, which were present only in the non-biotinylated samples, exhibited identical digestion profiles for Endo-H and PNGase F, indicating that the 70–75 kDa α4 protein and 47 kDa β2 protein contain only high mannose corresponding to subunits present in immature complexes located primarily in the endoplasmic reticulum (α4i and β2i). Results further demonstrate that following PNGase F cleavage, α4i migrates at an apparent molecular weight of about 66 kDa, supporting the idea that the lowest molecular weight α4 protein signal in Fig. 1[A] corresponds to a deglycosylated form of the subunit.
To ascertain whether mAb290 immunoprecipitated α4 subunits from all immature α4β2 complexes, and determine whether solubilization in buffer containing Triton X-100 could have affected the subunit species detected (Kuryatov et al., 2005), sequential immunoprecipitations were performed in cell lysates prepared using either Triton X-100 or Lubrol, the latter less protein complex dissociating (Schuck et al., 2003). Results (Fig. 1[C] left immunoblot) indicate that a single immunoprecipitation with mAb290 from Triton-X solubilized samples complexed all α4m, but a second immunoprecipitation with mAb290 was necessary to isolate what appeared to be the remainder of the 70–75 kDa α4 species because no signal was detected upon a third immunoprecipitation. However, when the supernatant remaining after the third immunoprecipitation with mAb290 was incubated with mAb299, a prominent signal was evident at 70–75 kDa. Because mAb290 immunoprecipitates only α4 subunits coupled to β2 subunits, results suggest that the 70–75 kDa α4 band following mAb299 immunoprecipitation may represent both free α4 subunits and subunits present in immature α4β2 complexes in a conformation unable to bind mAb290. Thus, although mAb290 complexes all α4 protein present in mature α4β2 receptors upon a single immunoprecipitation, it may not bind to α4protein in all immature α4β2 complexes. The idea that immature α4β2 complexes exist in at least 2 conformations, only one of which can be complexed by mAb290 and may represent a late developmental complex, with the other bound by mAb299 representing a more juvenile subunit complex (Sallette et al., 2005), is supported by results indicating that the 47 kDa β2 signal was still apparent in samples immunoprecipitated by mAb290 three times followed by immunoprecipitation with mAb299. These findings were verified further by similar experiments in which Triton-X solubilized samples were immunoprecipitated sequentially with mAb299 three times prior to mAb290 (Fig. 1[C] right immunoblot). Lastly, when samples prepared in Triton X-100 were compared with those in Lubrol (Fig. 1[C] center immunoblot), results indicate that Triton X-100 did not affect the subunit species detected as results with Triton X-100 were identical to those obtained using Lubrol.
α4 subunits associated with immature α4β2 complexes are phosphorylated by both PKA and PKC, whereas subunits associated with mature pentameric receptors are phosphorylated only by PKC
To investigate the possibility that activation of PKA and PKC may increase α4 subunit phosphorylation at different stages of receptor assembly and maturation, SH-EP1 cells were transfected with α4 and β2 subunit DNAs and incubated with 32Pi for 4 hours, the final 15 or 30 minutes in 10 μM forskolin or 200 nM PDBu, respectively. Whole cell lysates were incubated with mAb290 to immunoprecipitate α4β2 complexes, proteins were separated by electrophoresis and transferred to PVDF membranes, and the immunoblots probed for α4 and β2 subunit protein (Fig. 2). In the absence of exogenous stimulation of the kinases (basal state), both α4m and α4i were phosphorylated, the former to a greater extent (3 times) than the latter on a protein basis. Stimulation of PKA by forskolin did not alter the relative phosphorylation of α4m, but significantly (p<0.05) increased α4i phosphorylation 2.2-fold. In contrast, stimulation of PKC by PDBu increased phosphorylation of both α4m and α4i significantly (p<0.05) 1.6-fold for both species. Furthermore, when the possible phosphorylation of β2 subunits was investigated, results indicated that only subunits isolated from mature receptors were phosphorylated, and stimulation of either PKA or PKC increased phosphorylation of β2m significantly (p<0.05) by approximately 3-fold; immature β2 subunits were not phosphorylated under any condition (see autoradiograph and immunoblot in Fig. 2). Results demonstrate for the first time that β2 subunits associated with mature α4β2 receptors are also subject to phosphorylation upon kinase activation, albeit the relative phosphorylation of β2m in the basal state was only 1/10th that of α4m.
Fig. 2.
In vivo phosphorylation of immature and mature α4 subunits. SH-EP1 cells transfected with DNAs coding for α4 and β2 subunits (α4:β2, 2:3) were incubated with 32Pi (1 or 2 mCi/ml) for 4 hours, the final 15 or 30 minutes in 10 μM forskolin or 200 nM PDBu, respectively. Whole cell lysates were prepared, α4β2 complexes were immunoprecipitated with mAb290, and samples were separated on SDS-PAGE gels and transferred to PVDF membranes. Radioactivity was visualized by autoradiography by exposing the membranes to film prior to cutting them horizontally just above the 59 kDa marker. The top and bottom halves of the membranes were probed for subunit protein using H-133 for α4 and H-92 for β2 subunit, respectively. A representative autoradiograph and immunoblot are shown depicting α4m and β2m (subunits from fully processed pentamers) and α4i and β2i (immature subunit complexes). Relative phosphorylation was determined as the ratio of the densitometric signals from the autoradiographs to the immunoblots. Bars represent the mean + s.e.m. from 4–6 experiments. The asterisks denote significant differences relative to corresponding basal values, p<0.05.
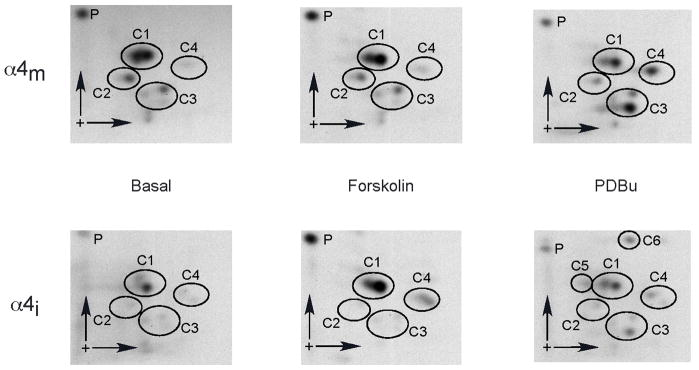
To determine whether PKA and PKC phosphorylated the same or similar residues on α4 subunits associated with mature receptors and immature complexes, the [32P]-labeled bands were excised from the PVDF membranes, digested with trypsin, and equal amounts of radioactivity (500 cpm) per sample were spotted on TLC plates and 2D phosphopeptide maps generated. Results (Fig. 3) demonstrate that under basal conditions, 4 clusters (C1–C4) of labeled phosphopeptide fragments were present in α4m, with a predominant signal at C1. This signal was also apparent for α4i, but the other signals were faint because basal phosphorylation for α4i was only 1/3 that of α4m (Fig. 2). When subunit species were isolated from samples incubated with forskolin, the phosphopeptide pattern generated for α4m did not differ from controls, whereas the map generated for α4i had increased signals associated with both C1 and C4, indicating that the substrates contained within these clusters are phosphorylated upon PKA activation prior to receptor maturation. When α4 subunit species were isolated from samples following incubation with PDBu, the map for α4m exhibited increased the phosphorylation of fragments within C3 and C4, whereas the pattern for α4i demonstrated increased phosphorylation associated with C3 and the appearance two additional phosphopeptide fragments, C5 and C6, which were not present in α4m, suggesting that these latter sites are phosphorylated upon activation of PKC prior to receptor maturation, and are dephosphorylated during the maturation process.
Fig 3.
2D phosphopeptide maps of immature and mature α4 subunits following stimulation of PKA or PKC. SH-EP1 cells were transfected, incubated and proteins immunoprecipitated and separated as in Fig. 2. The [32P]-labeled α4i and α4m bands were excised from the PVDF membranes, digested with trypsin, and spotted on TLC plates. Phosphopeptides were separated by electrophoresis in the horizontal dimension and ascending chromatography in the vertical dimension, and detected by autoradiography. Representative maps (from a total of 3 for each condition) from unstimulated cells (basal) or cells incubated in the presence of forskolin or PDBu are shown. Phosphopeptide clusters are grouped within circles.
PKA and PKC can phosphorylate α4 subunits prior to assembly with β2 subunits
Results from sequential immunoprecipitation studies suggested that the 70–75 kDa α4 protein from samples immunoprecipitated with mAb299 likely represents both free α4 subunits (either monomers or oligomers) and subunits associated with early developmental immature α4β2 complexes that can not be complexed by mAb290. To determine directly whether free α4 subunits can be phosphorylated either as monomers or oligomers, α4 subunit phosphorylation was determined in cells expressing only these subunits. SH-EP1 cells transfected with the DNA coding for α4 subunits only were incubated with 32Pi for 4 hours, the final 15 or 30 minutes in the absence or presence of forskolin or PDBu, respectively, whole cell lysates were prepared, and α4 subunit protein was immunoprecipitated with mAb299. Autoradiographs and immunoblots (Fig. 4[A]) indicate that when α4 subunits are expressed alone, a single phosphorylated protein band of approximately 70–75 kDa is apparent, and PKA or PKC stimulation increases subunit phosphorylation significantly (p<0.05) by 4.9- or 2.7-fold, respectively. The 2D phosphopeptide maps generated from these samples indicate that under basal conditions, phosphorylation is readily apparent on fragments within C1 and C4, and forskolin incubation increased phosphorylation of both of these substrates. In contrast, incubation with PDBu increased signals associated with fragments within C2, C3, C5 and C6, the latter particularly prominent. PKC stimulation did not alter the signals associated with either C1 or C4. Thus, the phosphopeptide maps for samples immunoprecipitated by mAb299 from cells expressing only α4 subunits were identical to those generated for α4i for samples immunoprecipitated by mAb290 from cells expressing both α4 and β2 subunits under basal or PKA stimulated conditions, but not following activation of PKC.
Fig 4.
In vivo phosphorylation of homomeric α4 subunits. SH-EP1 cells were transfected with the DNA coding for either [A] α4 subunits only or [B] both α4 and β2 subunits. Cells were incubated with 32Pi for 4 hours, the final 15 or 30 minutes in 10 μM forskolin or 200 nM PDBu, respectively, and whole cell lysates prepared as in Fig 2. [A] α4 subunits were immunoprecipitated with mAb299. [B] All α4 subunits assembled with β2 subunits were removed by sequential immunoprecipitation with mAb290 and excess H-92. The remaining α4 subunits (α4) were immunoprecipitated with mAb299. Samples were separated on SDS-PAGE gels and transferred to PVDF membranes. Radioactivity was visualized by autoradiography, and subunit protein probed for α4 with H-133. A representative autoradiograph and immunoblot are shown. Relative phosphorylation was calculated as the ratio of densitometric signals from the autoradiographs to the immunoblots. Each bar represents the mean + s.e.m. from 4–6 experiments. The asterisks denote significant differences relative to corresponding basal values, p<0.05. For 2D phosphopeptide maps, the [32P]-labeled α4 band was excised from the PVDF membranes, washed, digested with trypsin, and spotted on TLC plates as in Fig. 3. Phosphopeptide clusters are grouped within circles and labeled as in Fig. 3.
Although results suggested that α4 subunits can be phosphorylated prior to assembly with β2 subunits, α4 complexes never reach the cell surface (Ren et al., 2005) and α4 subunit expression in brain depends on β2 subunit expression (Whiteaker et al., 2006). Thus, to ascertain whether α4 subunit phosphorylation in cells expressing only α4 subunits was the same as in cells expressing both subunits, following preparation of cell lysates from cells transfected with the cDNAs for both α4 and β2 subunits and incubated as above, samples were incubated sequentially with mAb290 and excess of the anti-β2 polyclonal antibody H-92 to complex all species associated with β2 subunits. Immunoblot analysis confirmed that this procedure removed all α4β2 complexes irrespective of conformation or assembly. The free α4 subunits remaining in the samples were immunoprecipitated with mAb299. Autoradiographs and immunoblots (Fig. 4[B]) demonstrate that free α4 subunits unassociated with β2 subunits have the same molecular weight as the α4 protein isolated from cells expressing α4 subunits alone, albeit a much lower yield was obtained. Phosphorylation was apparent under basal conditions, and stimulation of PKA or PKC increased phosphorylation significantly (p<0.05) by 1.9- or 1.7-fold, respectively, similar to the respective 2.2- or 1.6-fold increases observed for α4i following stimulation of PKA or PKC (Fig. 2). Due to the low protein yield obtained from these samples, corresponding 2D phosphopeptide maps could not be generated. However, based on the similarity of the relative increase in phosphorylation of free α4 subunits and α4i following PKA and PKC stimulation, data supports the idea that α4 subunits may be phosphorylated prior to assembly with β2 subunits under basal and kinase-stimulated conditions.
Both PKA and PKC activation increase the number of membrane-associated receptors, but only PKC stimulation increases receptor function
Results indicating that both PKA and PKC phosphorylate α4 subunits prior to associating with β2 subunits, whereas only PKC phosphorylates subunits associated with fully processed mature receptors suggest that activation of these kinases may differentially affect receptor processing and function. To examine this possibility, both the number of membrane-associated receptors and magnitude of ACh-activated currents were determined in SH-EP1 cells stably transfected with human α4 and β2 subunits (SH-EP1-hα4β2 cells) and incubated with forskolin or PDBu for 24 hours. The first series of experiments determined the effects of forskolin and PDBu on receptor density. Based on studies indicting that SH-EP1-hα4β2 cells express a single population of binding sites with high affinity and 24 hours incubation with nicotine increases receptor density (Pacheco et al., 2001), the binding of 2.5 nM [3H]cytisine to membranes prepared from cells incubated in the absence or presence of 10 μM forskolin or 200 nM PDBu for 24 hours was measured. Results (Fig. 5[A]) indicate that both forskolin and PDBu increased the number of membrane-associated receptors significantly (p<0.05) by 1.6- and 1.4-fold, respectively, in agreement with studies from HEK293 cells stably expressing human α4β2 receptors (Gopalakrishnan et al., 1997). Studies investigating shorter incubation times (6 and 12 hours) with forskolin and PDBu did not detect any alterations in receptor density (data not shown).
Fig. 5.
Effects of forskolin and PDBu on α4β2 receptor density and function. [A] SH-EP1-hα4β2 cells were incubated in the absence or presence of 10 μM forskolin or 200 nM PDBu for 24 hours, membrane fractions prepared, and binding at 2.5 nM [3H]cytisine was determined. Bars represent group mean values + s.e.m. from 7 experiments. The asterisk denotes a significant difference from basal values, p<0.05. [B] Responses of cells to varying concentrations of ACh. Membrane currents were measured using the perforated patch configuration of the whole-cell voltage clamp technique. The cell was held at −60 mV and membrane currents evoked by rapid application of ACh at the indicated concentrations. The horizontal bar indicates the time during the application of ACh (400 msec). A recording from a representative cell is shown on the left and cumulative data depicting normalized peak ACh-induced inward currents as a function of ACh concentration on the right. Peak currents were normalized to currents measured in response to 3 mM ACh for each cell. Each point represents data obtained from 12–16 cells ± s.e.m. The solid line represents the results of a fit of the data to a 2-site Langmuir-Hill equation, while the dashed line shows a fit of the data to a 1-site equation. [C] Current-voltage relationship for ACh-evoked peak current amplitudes. A representative family of traces of ACh-activated current responses from a cell held at the indicated potentials and currents evoked using 2 mM ACh (400 msec). The mean peak ACh-induced inward currents as a function of holding potential are shown. Peak current amplitudes were normalized to the absolute peak measured at −60 mV for each cell in response to 2 mM ACh. Each point represents data from 7–9 cells ± s.e.m. [D] Modulation of ACh-evoked current density in SH-EP1-hα4β2 cells following activation of PKA or PKC. Cells were incubated in the absence or presence of 10 μM forskolin or 200 nM PDBu for 24 hours, washed, and membrane currents in response to 2 mM ACh were measured using the perforated patch configuration of the whole-cell voltage clamp technique; cells were held at −60 mV. Bar graphs depict group mean values + s.e.m. of current densities (normalized to controls). The control ACh current density was −103 ± 5.5 pA/pF; n=9 cells from each group. The asterisk denotes a significant difference from basal values, p<0.05.
To evaluate the consequences of kinase activation on functional responses, ACh-evoked currents were measured in these cells, which have been shown to express functional receptors with properties similar to human α4β2 receptors studied in other expression systems (Wu et al., 2004; Eaton et al., 2003). Initial experiments were conducted using the perforated-patch configuration of the whole-cell recording technique to determine the basic functional properties of the receptors expressed by these cells under conditions which preserve the intracellular milieu required for ‘normal’ kinase and phosphatase functioning. Application of ACh evoked fast activating transient inward currents that exhibited concentration-dependent amplitude and concentration- and time-dependent desensitization (Fig. 5[B]). When the normalized peak amplitude was plotted as a function of ACh concentration, a sigmoidal concentration-response relationship was apparent with data best fit by the two-site Langmuir-Hill equation with the following parameters: the high affinity site had Emax-1, EC50-1, and n1 values of 0.11, 1 μM, and 0.7, respectively; the low affinity site had Emax-2, EC50-2, and n2 values of 0.89, 161 μM, and 1.0, respectively. The voltage dependence of responses to 2 mM ACh were consistent with nicotinic receptor activation. A plot of peak ACh-evoked current amplitude as a function of holding potential demonstrated a linear current-voltage relationship at negative potentials, with a reversal potential near 0 mV, and inward rectification at positive potentials (Fig. 5[C]). When cells were incubated with 10 μM forskolin or 200 nM PDBu for 24 hours, forskolin incubation did not alter peak ACh-evoked current densities, whereas incubation with PDBu increased current density significantly (p<0.05) by nearly 2-fold (Fig. 5[D]).
To ascertain whether the ability of PDBu to increase ACh-evoked responses was attributed specifically to PKC activation, cells were incubated with PDBu in the absence or presence of the PKC inhibitor Ro-31-8220 (100 nM) for 24 hours. Results (Fig. 6[A]) indicated that Ro-31-8220 prevented the PDBu-induced increase. Incubation of cells with Ro-31-8220 alone or vehicle (0.1% DMSO) did not alter affect peak current amplitudes. Thus, the ability of PDBu to functionally up regulate α4β2 receptors could be attributed specifically to activation of PKC.
Fig 6.
Modulation of ACh-evoked currents in SH-EP1-hα4β2 cells by PDBu. Cells were incubated in the absence or presence of 200 nM PDBu or 100 nM Ro-31-8220 alone or in combination for different periods of time, and membrane currents in response to 2 mM ACh were measured following washing using the perforated patch configuration of the whole-cell voltage clamp technique; cells were held at −60 mV. The horizontal bars indicate the time of ACh application (1.5 sec). Representative current traces and bar graphs depicting group mean values + s.e.m. of current densities (normalized to controls) are shown. [A] Cells were incubated for 24 hours with the indicated compounds and washed for 10 minutes prior to recording. The control ACh current density was −71.6 ± 5.1 pA/pF (n=54). The sample size (n) for the other groups were: 17 for DMSO, Ro-31-8220 and Ro + PDBu; and 29 for PDBu alone. [B] Cells were incubated for 1 hour with the indicated compounds followed by an extensive 10 minute washout prior to recording. The control ACh current density was −57.3 ± 3.7 pA/pF (n=47). The sample sizes (n) for the other groups were 13 for DMSO and 25 for PDBu. [C] Cells were incubated for 1 hour with the indicated compounds followed by washout and 23 hours of incubation in control media prior to recording. The control ACh current density was −74.1 ± 4.6 pA/pF (n=61). The sample sizes (n) for the other groups were: 16 for DMSO; 13 for Ro-31-8220; 28 for PDBu; 19 for Ro + PDBu. The asterisks denote significant differences from control (p < 0.05).
Based on studies indicating that PKC activation for 30 minutes increases the proportion of mature α4β2 receptors on or near the surface of HEK293T cells (Nashmi et al., 2003), the time dependence of the ability of PDBu to increase ACh-activated currents was investigated. SH-EP1-hα4β2 cells were incubated with PDBu for 1 hour and peak ACh-evoked current densities were measured either immediately following a 10 minute washout or 23 hours following washout and incubation in control media. When cells were exposed to PDBu for 1 hour and responses measured following a 10 minute washout, no differences were apparent between control and PDBu exposed cells (Fig. 6[B]). In contrast, when cells were incubated with PDBu for 1 hour followed by a 23 hour incubation period in control media, a significant (p<0.05) 55% increase in ACh-evoked current density was observed, and this increase was prevented by incubating cells with Ro-31-8220 (Fig. 6[C]). These results indicate that the ability of PDBu to increase receptor function appears to be initiated by activation of PKC, but requires additional cellular processes before functional differences are manifest.
DISCUSSION
The goals of this study were to determine whether activation of PKA and PKC increases the phosphorylation of α4 subunits during different stages in the assembly and maturation of α4β2 receptors, and differentially affects receptor function. Results indicate that: 1) PKA phosphorylates α4 subunits on peptide fragments contained within C1 and C4 prior to complexing with β2 subunits, and these sites remain phosphorylated during the development of immature α4β2 complexes; 2) PKC phosphorylates α4 subunits on two substrates within C5 and C6 prior to complexing with β2 subunits, and these sites remain phosphorylated during the development of immature α4β2 complexes; 3) PKC also phosphorylates a fragment within C3 on α4 subunits associated with both immature α4β2 complexes and mature receptors; and 4) activation of both kinases increases the number of membrane-associated receptors, but only activation of PKC increases ACh-evoked current density. Studies also demonstrate for the first time that β2 subunits are phosphorylated, but only in the mature α4β2 receptor in the plasma membrane.
Experiments to separateα4 species at distinct stages of receptor assembly and maturation led to several important observations, some, but not all of which are consistent with other studies. The finding that additional higher molecular weight signals for both α4 and β2 subunits were present in samples immunoprecipitated with mAb290 and not mAb299 is identical to results from metabolic labeling studies indicating that the high molecular weight forms correspond to subunits isolated from mature α4β2 receptors with fully processed sugars located on the plasma membrane, while the lower molecular weight signals (70–75 kDa for α4 and 47 kDa for β2) arise from immature α4β2 complexes containing high mannose (Sallette et al., 2005). In addition, results from the present study also confirm that mAb299 complexes α4 subunits expressed either alone or with β2 subunits (Sallette et al., 2005), and extend this finding to indicate that this antibody immunoprecipitates two species of α4 present in both conditions, both the predominant 70–75 kDa immature species, and a lower molecular weight species of 66 kDa, likely representing a non-glycosylated form of α4 (Fig. 1[A]). However, in contrast to Sallette et al. (2005), who reported that mAb290 recognizes β2 subunits only when co-expressed with α4 subunits, results from the present study indicate that mAb290 does not require β2 to be in a heterooligomeric form with α4, as a signal for β2, albeit faint, is present at 47 kDa in cells transfected with only the cDNA for β2 subunit protein. It is possible that this difference reflects differences in the cell systems used, the methods of transfection, the amount of cDNA transfected, the time experiments were performed following transfection, or the relative expression level of the subunit. Nevertheless, because the signal for the β2 subunit from cells transfected only with this subunit was so faint, results suggest that perhaps in this situation, heteromers of β2 are formed that sterically hinder the ability of mAb299 to bind to the extracellular loop of the subunit.
Results from the current experiments also support findings that the anti-α4 monoclonal antibody mAb299 obtained from Sigma-Aldrich Co. (St. Louis, MO) does not immunoprecipitate subunits associated with mature α4β2 receptors (Sallette et al., 2005). This observation was somewhat surprising as we have demonstrated that a different commercial preparation of mAb299 from Covance Inc. (Richmond, CA) did indeed complex the mature 80–85 kDa α4 species (Pollock et al., 2007). These antibodies were generated from the same clone, the only difference being that the preparation from Covance Inc. is generated and used in ascites fluid, whereas the preparation from Sigma-Aldrich Co. was generated in vitro and contains purified antibody. Although one may expect the former to contain contaminants that bind to cellular membranes thereby preventing immunodetection, this was not the case, and the reason why these preparations differ in their ability to complex α4 subunits associated with mature receptors remains unclear.
Prior studies have identified two major substrate sites for PKA on α4 subunits, Ser467 on a peptide fragment within C1, and Ser362 on a fragment within the C4 doublet (Pollock et al., 2007). Based on the phosphopeptide patterns generated by Pollock et al. (2007), the present study indicates that Ser467 is phosphorylated on α4 subunits prior to complexing with β2 subunits and remains phosphorylated throughout receptor assembly, maturation, and trafficking to the plasma membrane, suggesting that its phosphorylation plays a long-lasting determinant role. This idea is supported by studies demonstrating that forskolin stimulation of PKA increases the binding of the 14-3-3 chaperone protein to the rat α4 subunit expressed in tsA 201 cells, resulting in an increase in the steady-state level of α4 protein and the number of α4β2 cell surface receptors, and when Ser441, which corresponds to Ser467 in the human α4 subunit, was mutated to an alanine, this effect was not present (Jeanclos et al., 2001). Furthermore, studies in Xenopus oocytes have shown that when Ser467 in human α4 subunits was replaced by alanine and expressed with β2 subunits, steady-state concentrations of α4 subunit protein, the maximal density of [3H]cytisine binding sites, ACh response amplitude, and the proportion of α4β2 receptors with low sensitivity to agonist all decreased, and opposite effects were noted when Ser467 was replaced by aspartate to mimic the phosphorylated α4 subunit (Exley et al., 2006). Based on these findings, evidence suggests that phosphorylation of Ser467 on free α4 subunits prior to association with β2 subunits has long-lasting consequences increasing the stability of α4 subunits, perhaps resulting in an enhanced expression of α4β2 receptors in the low affinity (α4)3(β2)2 configuration (Nelson et al., 2003; Moroni et al., 2006).
Similarly, based on prior phosphopeptide patterns (Pollock et al., 2007), the current results indicate that Ser362 also appears to be phosphorylated by PKA prior to assembly of α4 with β2 subunits, but this residue is minimally phosphorylated on α4 subunits associated with mature receptors. This suggests that this post-translational modification plays a transient role prior to α4β2 complexes exiting the endoplasmic reticulum. Although a functional role for the PKA-mediated phosphorylation of Ser362 has not been investigated, studies have identified sequences within the large M3/M4 cytoplasmic loop of α4 subunits that are overlapping with or adjacent to Ser362 that are essential for α4β2 receptor subcellular targeting (Xu et al., 2006), export from the endoplasmic reticulum (Ren et al., 2005) or trafficking from the endoplasmic reticulum to the cell surface (Keller et al., 2001). It is possible that phosphorylation of Ser362 by PKA could play a role in regulating one or more of these functions. Although PKA is localized in the cytosol and is highly concentrated around the endoplasmic reticulum and Golgi (Nigam and Blobel, 1989), it does not appear to be available to phosphorylate any additional PKA sites on α4 subunits associated with immature complexes or fully mature receptors in the plasma membrane.
Possible substrate sites for PKC on α4 subunits include Ser550, which is present on one fragment contained within the C3 triplet (Pollock et al., 2007). Stimulation of PKC by PDBu enhanced the phosphorylation of this fragment within C3 on α4 subunits associated with both immature α4β2 complexes and mature receptors (Fig. 3), but not on free α4 subunits (Fig. 4). This suggests that phosphorylation of Ser550 may affect both the trafficking of receptors from the endoplasmic reticulum to the cell surface and receptor function rather than the expression of a specific receptor conformation. The phosphorylation of α4 subunits within mature receptors at the plasma membrane by PKC is not surprising as phorbol esters induce translocation of the enzyme to the plasma membrane (Oancea and Meyer, 1998; Almholt et al., 1999; Shirai and Saito, 2002) or other cellular membranes (Wang et al., 2000), where it can access and phosphorylate sites in the cytoplasmic domain of α4 subunits. Ser550 is unlike many other phosphorylation sites within the M3/M4 cytoplasmic domain of α4 subunits, which demonstrate a high degree of sequence homology among different species, and is present only on subunits from human and chicken, and not rat or mouse (see Fig. 1 in Pollock et al., 2007). Thus, the phosphorylation of Ser550 may be responsible for functional differences observed between human and rat receptors such as the human receptor exhibiting sensitization following sustained agonist exposure (Buisson and Bertrand, 2001; Gopalakrishnan et al., 1997) whereas the rat receptor exhibits inactivation (Fenster et al., 1999; Hsu et al., 1997).
In addition to the PKC-mediated phosphorylation of Ser550 on immature complexes and mature receptors, stimulation of this kinase led to the appearance of two additional unidentified phosphopeptide fragments within C5 and C6 on free α4 subunits (Fig. 4), and these sites remain phosphorylated on subunits associated with immature α4β2 complexes, but not on those associated with mature receptors (Fig. 3). This pattern indicates that these residues are phosphorylated on α4 subunits in the endoplasmic reticulum prior to assembly with β2 subunits, that phosphorylation persists throughout α4β2 complex formation, but that the residues are dephosphorylated by the time the mature receptor is transported from the Golgi to the plasma membrane. Based on evidence that Ser550 is phosphorylated on subunits associated with immature complexes, in concert with findings that phosphorylation of Ser550 is greater on α4 subunits associated with mature pentamers, it is possible that dephosphorylation of the substrates on fragments within C5 and C6 must occur prior to further phosphorylation of Ser550. Thus, the phosphorylation and dephosphorylation of substrates within C5 and C6 is dependent on the state of assembly and maturation of the receptor with dephosphorylation likely involved in trafficking of receptors from the endoplasmic reticulum to the cell surface (Keller et al., 2001). A schematic summarizing the sites of phosphorylation/dephosphorylation at different stages in the assembly, maturation and trafficking of α4β2 receptors is presented in Fig. 7.
Fig. 7.
Putative sites of action of PKA and PKC on α4β2 receptors. The schematic depicts the stages in the assembly, maturation and trafficking of α4β2 receptors during which subunits may be phosphorylated (or dephosphorylated). Immature α4 subunits (α4i) are shown representing free, unassembled subunits and immature subunit complexes containing high mannose in the endoplasmic reticulum. Mature α4 subunits (α4m) are shown representing fully processed mature forms with complex carbohydrates associated with the Golgi and plasma membrane. Results from phosphorylation and differential immunoprecipitation experiments indicate that free α4 subunits are phosphorylated by both PKA and PKC, the former on Ser362 and Ser467, and the latter on two as yet unidentified serine residues (within C5 and C6). Phosphorylation at these sites promotes the assembly of subunits, followed sequentially by the PKC-mediated phosphorylation of Ser550. Although the schematic depicts all immature complexes phosphorylated by PKC, these complexes exist in at least two conformations, a juvenile form which can be immunoprecipitated with mAb299 and a late developmental form, which can be immunoprecipitated by mAb290 (Sallette et al., 2005). It is possible that phosphorylation by PKC occurs only on one form, and that provides the signal for the exiting of the receptor from the endoplasmic reticulum. Maturation of the receptor in the Golgi appears to involve the dephosphorylation of several residues, perhaps one of which enables the pentamer to exit the Golgi and insert into the plasma membrane. α4 subunits associated with the plasma membrane may be further phosphorylated on Ser550 by PKC, perhaps affecting receptor function or stabilization. β2 subunits can also be phosphorylated by PKA and PKC, but only when are part of the membrane-associated pentamer.
Evidence that PKC has a dual action affecting α4 subunits associated with mature receptors at the cell surface and as intracellular subunits in the free and immature complexed states, resembles the effects of nicotine on these receptors. Nicotine has also been postulated to have a dual action, both at the cell surface and on immature α4β2 oligomers in the endoplasmic reticulum. Several studies have supported the idea that nicotine acts intracellularly as a chaperone to promote subunit assembly (Kuryatov et al., 2005; Sallette et al., 2005), increase the transport of receptors from the endoplasmic reticulum to the plasma membrane (Darsow et al., 2005), and stabilize receptors at the cell surface (Peng et al., 1994; Wang et al., 1998; Kuryatov et al., 2005), albeit the cellular mechanism(s) involved have not been elucidated. Incubation of HEK293 cells with nicotine increases the number of receptors 2.5-fold and produces a 1.6-fold increased functional response (Sallette et al., 2005), similar to results obtained upon activation of PKC. These findings, in concert with studies demonstrating that both nicotine and PKC activation increase receptor function by increasing subunit assembly and translocation of the receptor to or near the cell surface, resulting in increased surface expression (Nashmi et al., 2003), suggests that at least some of the actions of nicotine may be mediated via activation of PKC.
Lastly, the present study indicates for the first time that β2 subunits of mature α4β2 receptors at the cell surface are phosphorylated in the basal state and can be further phosphorylated upon activation of both PKA and PKC. Although Exley et al. (2006) failed to identify any PKA phosphorylation sites on human β2 subunits, predictions for putative phosphorylation sites indicate that a PKA, but not PKC consensus sequence is present in the large M3/M4 cytoplasmic loop of these subunits (http://www.cbs.dtu.dk/services/NetPhosK/). We have obtained preliminary data that phosphorylation occurs primarily on serine residues, but have not identified specific substrates or the functional consequences of β2 subunit phosphorylation. Current experiments are determining whether receptors normally expressed in brain tissue are phosphorylated at different maturational stages upon activation of PKA and PKC as those in studies from heterologous expression systems.
Acknowledgments
These studies were supported in part by a grant from the National Institute of Drug Abuse #DA14010 (to L.W.).
Abbreviations
- 2D
two-dimensional
- ACh
acetylcholine
- ECL
enhanced chemiluminescence
- MOPS
3-(N-morpholino)propanesulfonic acid
- NP-40
nonyl phenoxylpolyethoxylethanol
- PBS
phosphate-buffered saline
- PDBu
phorbol 12,13-dibutyrate
- PKA
cyclic AMP-dependent protein kinase
- PKC
protein kinase C
- PVDF
polyvinylidene fluoride
- SDS-PAGE
sodium dodecyl sulfate polyacrylamide gel
- TBS
Tris-buffered saline
- TLC
thin layer chromatography
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Almholt K, Arkhammar POG, Thastrup O, Tullin S. Simultaneous visualization of the translocation of protein kinase Cα-green fluorescent protein hybrids and intracellular calcium concentrations. Biochem J. 1999;337:211–218. [PMC free article] [PubMed] [Google Scholar]
- Boyle WJ, van der Geer P, Hunter T. Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods Enzymol. 1991;201:110–149. doi: 10.1016/0076-6879(91)01013-r. [DOI] [PubMed] [Google Scholar]
- Buisson B, Bertrand D. Chronic exposure to nicotine upregulates the human α4β2 nicotinic acetylcholine receptor function. J Neurosci. 2001;21:1819–1829. doi: 10.1523/JNEUROSCI.21-06-01819.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darsow T, Booker TK, Piña-Crespo JC, Heinemann SF. Exocytotic trafficking is required for nicotine-induced up-regulation of α4β2 nicotinic acetylcholine receptors. J Biol Chem. 2005;280:18311–18320. doi: 10.1074/jbc.M501157200. [DOI] [PubMed] [Google Scholar]
- Eaton JB, Peng JH, Schroeder KM, George AA, Fryer JD, Krishnan C, Buhlman L, Kuo YP, Steinlein O, Lukas RJ. Characterization of human α4β2-nicotinic acetylcholine receptors stably and heterologously expressed in native nicotinic receptor-null SH-EP1 human epithelial cells. Mol Pharmacol. 2003;64:1283–1294. doi: 10.1124/mol.64.6.1283. [DOI] [PubMed] [Google Scholar]
- Eilers H, Schaeffer E, Bickler PE, Forsayeth JR. Functional deactivation of the major neuronal nicotinic receptor caused by nicotine and a protein kinase C-dependent mechanism. Mol Pharmacol. 1997;52:1105–1112. [PubMed] [Google Scholar]
- Exley R, Moroni M, Sasdelli F, Houlihan LM, Lukas RJ, Sher E, Zwart R, Bermudez I. Chaperone protein 14-3-3 and protein kinase A increase the relative abundance of low agonist sensitivity human α4β2 nicotinic acetylcholine receptors in Xenopus oocytes. J Neurochem. 2006;98:876–885. doi: 10.1111/j.1471-4159.2006.03915.x. [DOI] [PubMed] [Google Scholar]
- Fenster CP, Beckman ML, Parker JC, Sheffield EB, Whitworth TL, Quick MW, Lester RA. Regulation of α4β2 nicotinic receptor desensitization by calcium and protein kinase C. Mol Pharmacol. 1999;55:432–443. [PubMed] [Google Scholar]
- Gopalakrishnan M, Molinari EJ, Sullivan JP. Regulation of human α4β2 neuronal nicotinic acetylcholine receptors by cholinergic channel ligands and second messenger pathways. Mol Pharmacol. 1997;52:524–534. [PubMed] [Google Scholar]
- Guo X, Wecker L. Identification of three cAMP-dependent protein kinase (PKA) phosphorylation sites within the major intracellular domain of neuronal nicotinic receptor β4 subunits. J Neurochem. 2002;82:439–447. doi: 10.1046/j.1471-4159.2002.01027.x. [DOI] [PubMed] [Google Scholar]
- Hsu YN, Edwards SC, Wecker L. Nicotine enhances the cyclic AMP-dependent protein kinase-mediated phosphorylation of α4 subunits of neuronal nicotinic receptors. J Neurochem. 1997;69:2427–2431. doi: 10.1046/j.1471-4159.1997.69062427.x. [DOI] [PubMed] [Google Scholar]
- Jeanclos EM, Lin L, Treuil MW, Rao J, DeCoster MA, Anand R. The chaperone protein 14-3-3η interacts with the nicotinic acetylcholine receptor α4 subunit. Evidence for a dynamic role in subunit stabilization. J Biol Chem. 2001;276:28281–28290. doi: 10.1074/jbc.M011549200. [DOI] [PubMed] [Google Scholar]
- Keller SH, Lindstrom J, Ellisman M, Taylor P. Adjacent basic amino acid residues recognized by the COP I complex and ubiquitination govern endoplasmic reticulum to cell surface trafficking of the nicotinic acetylcholine receptor α-subunit. J Biol Chem. 2001;276:18384–18391. doi: 10.1074/jbc.M100691200. [DOI] [PubMed] [Google Scholar]
- Kuryatov A, Luo J, Cooper J, Lindstrom J. Nicotine acts as a pharmacological chaperone to up-regulate human α4β2 acetylcholine receptors. Mol Pharmacol. 2005;68:1839–1851. doi: 10.1124/mol.105.012419. [DOI] [PubMed] [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Moroni M, Zwart R, Sher E, Cassels BK, Bermudez I. α4β2 nicotinic receptors with high and low acetylcholine sensitivity: pharmacology, stoichiometry, and sensitivity to long-term exposure to nicotine. Mol Pharmacol. 2006;70:755–768. doi: 10.1124/mol.106.023044. [DOI] [PubMed] [Google Scholar]
- Nakayama H, Okuda H, Nakashima T. Phosphorylation of rat brain nicotinic acetylcholine receptor by cAMP-dependent protein kinase in vitro. Brain Res Mol Brain Res. 1993;20:171–177. doi: 10.1016/0169-328x(93)90123-7. [DOI] [PubMed] [Google Scholar]
- Nashmi R, Dickinson ME, McKinney S, Jareb M, Labarca C, Fraser SE, Lester HA. Assembly of α4β2 nicotinic acetylcholine receptors assessed with functional fluorescently labeled subunits: effects of localization, trafficking, and nicotine-induced upregulation in clonal mammalian cells and in cultured midbrain neurons. J Neurosci. 2003;23:11554–11567. doi: 10.1523/JNEUROSCI.23-37-11554.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson ME, Kuryatov A, Choi CH, Zhou Y, Lindstrom J. Alternate stoichiometries of α4β2 nicotinic acetylcholine receptors. Mol Pharmacol. 2003;63:332–341. doi: 10.1124/mol.63.2.332. [DOI] [PubMed] [Google Scholar]
- Nigam SK, Blobel G. Cyclic AMP-dependent protein kinase in canine pancreatic rough endoplasmic reticulum. J Biol Chem. 1989;264:16927–16932. [PubMed] [Google Scholar]
- Oancea E, Meyer T. Protein kinase C as a molecular machine for decoding calcium and diacylglycerol signals. Cell. 1998;95:307–318. doi: 10.1016/s0092-8674(00)81763-8. [DOI] [PubMed] [Google Scholar]
- Pacheco MA, Pastoor TE, Wecker L. Phosphorylation of the α4 subunit of human α4β2 nicotinic receptors: role of cAMP-dependent protein kinase (PKA) and protein kinase C (PKC) Brain Res Mol Brain Res. 2003;114:65–72. doi: 10.1016/s0169-328x(03)00138-4. [DOI] [PubMed] [Google Scholar]
- Peng X, Gerzanich V, Anand R, Whiting PJ, Lindstrom J. Nicotine-induced increase in neuronal nicotinic receptors results from a decrease in the rate of receptor turnover. Mol Pharmacol. 1994;46:523–530. [PubMed] [Google Scholar]
- Picciotto MR, Caldarone BJ, Brunzell DH, Zachariou V, Stevens TR, King SL. Neuronal nicotinic acetylcholine receptor subunit knockout mice: physiological and behavioral phenotypes and possible clinical implications. Pharmacol Ther. 2001;92:89–108. doi: 10.1016/s0163-7258(01)00161-9. [DOI] [PubMed] [Google Scholar]
- Pollock VV, Pastoor TE, Wecker L. Cyclic AMP-dependent protein kinase (PKA) phosphorylates ser362 and 467 and protein kinase C phosphorylates ser550 within the M3/M4 cytoplasmic domain of human nicotinic receptor α4 subunits. J Neurochem. 2007;103:456–466. doi: 10.1111/j.1471-4159.2007.04853.x. [DOI] [PubMed] [Google Scholar]
- Ren XQ, Cheng SB, Treuil MW, Mukherjee J, Rao J, Braunewell KH, Lindstrom JM, Anand R. Structural determinants of α4β2 nicotinic acetylcholine receptor trafficking. J Neurosci. 2005;25:6676–6686. doi: 10.1523/JNEUROSCI.1079-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothhut B, Romano SJ, Vijayaraghavan S, Berg DK. Post-translational regulation of neuronal acetylcholine receptors stably expressed in a mouse fibroblast cell line. J Neurobiol. 1996;29:115–125. doi: 10.1002/(SICI)1097-4695(199601)29:1<115::AID-NEU9>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- Sallette J, Pons S, Devillers-Thiery A, Soudant M, Prado de Carvalho L, Changeux JP, Corringer PJ. Nicotine upregulates its own receptors through enhanced intracellular maturation. Neuron. 2005;46:595–607. doi: 10.1016/j.neuron.2005.03.029. [DOI] [PubMed] [Google Scholar]
- Schuck S, Honsho M, Ekroos K, Shevchenko A, Simons K. Resistance of cell membranes to different detergents. Proc Natl Acad Sci U S A. 2003;100:5795–5800. doi: 10.1073/pnas.0631579100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirai Y, Saito N. Activation mechanisms of protein kinase C: maturation, catalytic activation, and targeting. J Biochem. 2002;132:663–668. doi: 10.1093/oxfordjournals.jbchem.a003271. [DOI] [PubMed] [Google Scholar]
- Tapper AR, McKinney SL, Nashmi R, Schwarz J, Deshpande P, Labarca C, Whiteaker P, Marks MJ, Collins AC, Lester HA. Nicotine activation of α4* receptors: sufficient for reward, tolerance, and sensitization. Science. 2004;306:1029–1032. doi: 10.1126/science.1099420. [DOI] [PubMed] [Google Scholar]
- Vallejo YF, Buisson B, Bertrand D, Green WN. Chronic nicotine exposure upregulates nicotinic receptors by a novel mechanism. J Neurosci. 2005;25:5563–5572. doi: 10.1523/JNEUROSCI.5240-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viseshakul N, Figl A, Lytle C, Cohen BN. The α4 subunit of rat α4β2 nicotinic receptors is phosphorylated in vivo. Brain Res Mol Brain Res. 1998;59:100–104. doi: 10.1016/s0169-328x(98)00128-4. [DOI] [PubMed] [Google Scholar]
- Wang F, Nelson ME, Kuryatov A, Olale F, Cooper J, Keyser K, Lindstrom J. Chronic nicotine treatment up-regulates human α3β2 but not α3β4 acetylcholine receptors stably transfected in human embryonic kidney cells. J Biol Chem. 1998;273:28721–28732. doi: 10.1074/jbc.273.44.28721. [DOI] [PubMed] [Google Scholar]
- Wang QJ, Fang TW, Fenick D, Garfield S, Bienfait B, Marquez VE, Blumberg PM. The lipophilicity of phorbol esters as a critical factor in determining the pattern of translocation of protein kinase C δ fused to green fluorescent protein. J Biol Chem. 2000;275:12136–12146. doi: 10.1074/jbc.275.16.12136. [DOI] [PubMed] [Google Scholar]
- Wecker L, Guo X, Rycerz AM, Edwards SC. Cyclic AMP-dependent protein kinase (PKA) and protein kinase C phosphorylate sites in the amino acid sequence corresponding to the M3/M4 cytoplasmic domain of α4 neuronal nicotinic receptor subunits. J Neurochem. 2001;76:711–720. doi: 10.1046/j.1471-4159.2001.00041.x. [DOI] [PubMed] [Google Scholar]
- Whiteaker P, Cooper JF, Salminen O, Marks MJ, McClure-Begley TD, Brown RW, Collins AC, Lindstrom JM. Immunolabeling demonstrates the interdependence of mouse brain α4 and β2 nicotinic acetylcholine receptor subunit expression. J Comp Neurol. 2006;499:1016–1038. doi: 10.1002/cne.21181. [DOI] [PubMed] [Google Scholar]
- Wu J, Kuo YP, George AA, Xu L, Hu J, Lukas RJ. β-myloid directly inhibits human α4β2-nicotinic acetylcholine receptors heterologously expressed in human SH-EP1 cells. J Biol Chem. 2004;279:37842–37851. doi: 10.1074/jbc.M400335200. [DOI] [PubMed] [Google Scholar]
- Xu J, Zhu Y, Heinemann SF. Identification of sequence motifs that target neuronal nicotinic receptors to dendrites and axons. J Neurosci. 2006;26:9780–9793. doi: 10.1523/JNEUROSCI.0840-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoli M, Le Novere N, Hill JA, Jr, Changeux JP. Developmental regulation of nicotinic ACh receptor subunit mRNAs in the rat central and peripheral nervous systems. J Neurosci. 1995;15:1912–1939. doi: 10.1523/JNEUROSCI.15-03-01912.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]