Abstract
Background and objectives: Transplant options for type I diabetics with end-stage renal disease include simultaneous pancreas-kidney (SPKT), living donor kidney (LDKT), and deceased donor kidney transplant (DDKT). It is unclear whether SPKT offers a survival benefit over LDKT in the current era of transplantation. The authors compared outcomes of kidney transplant recipients with type I diabetes using data from the Organ Procurement and Transplant Network/United Network for Organ Sharing.
Design, setting, participants, & measurements: Adult (age 20 to 59) type I diabetics who received a solitary first-time kidney transplant between 2000 and 2007 were studied. Outcomes included overall kidney graft and patient survival. Multivariate analysis was performed using a stepwise Cox proportional hazards model.
Results: Kidney graft survival was better for recipients of LDKT compared with SPKT (P = 0.008), although patient survival was similar (P = 0.346). On multivariate analysis, LDKT was associated with lower adjusted risks over 72 mo follow-up of kidney graft failure (HR 0.71; 95% CI 0.61 to 0.83) and patient death (HR 0.78; 95% CI 0.65 to 0.94) versus SPKT. Compared with DDKT, SPKT had superior unadjusted kidney graft and patient survival, partly due to favorable SPKT donor and recipient factors.
Conclusions: Despite more transplants from older donors and among older recipients, LDKT was associated with superior outcomes compared with SPKT and was coupled with the least wait time and dialysis exposure. LDKT utilization should be considered in all type I diabetics with an available living donor, particularly given the challenges of ongoing organ shortage.
For eligible type I diabetics with end-stage renal disease (ESRD), kidney transplantation is the treatment of choice because it confers a survival advantage over maintenance dialysis (1–3). According to the 2007 United States Renal Data System Annual Report, more than 512,000 patients initiated therapy for ESRD from 2001 to 2005, and diabetes mellitus was the primary etiology of approximately 45% of these cases (4). Of these incident diabetic ESRD patients, 8.5% were type I diabetics. Kidney transplantation options for type I diabetics include simultaneous pancreas-kidney transplant (SPKT), living donor kidney transplant (LDKT), and deceased donor kidney transplant (DDKT) (5).
In type I diabetics, SPKT is associated with superior long-term kidney graft and patient survival compared with DDKT (6–10). In contrast, kidney graft and patient survival of SPKT and LDKT may be comparable (9,11). Preemptive transplantation with SPKT or LDKT in type I diabetics may result in a survival benefit over those who require pretransplant dialysis (10,12). Considering the excess mortality observed in patients waiting for a transplant, and because LDKT is more likely to minimize dialysis time, the National Kidney Foundation has recommended LDKT as the treatment of choice for most type I diabetics (5).
In the past decade, survival rates have improved for LDKT, with unadjusted 1-yr kidney graft survival rates of 95.1% in 2004 as compared with 92.6% in 1994 (13). Similarly, survival rates for SPKT have risen despite an increasingly older and higher risk recipient population, with unadjusted 1-yr kidney graft survival in 2004 of 91.4% versus 85.7% in 1994 (13,14). Advancements in surgical techniques and immunosuppressive protocols may partially explain this forward progress (15,16).
Given these recent changes in management and outcomes, we performed a large retrospective analysis of type I diabetic kidney transplant recipients undergoing SPKT, LDKT, and DDKT, using data from the Organ Procurement and Transplantation Network/United Network of Organ Sharing (OPTN/UNOS) to examine short- and long-term kidney graft and patient outcomes in the current era of transplantation.
Materials and Methods
The OPTN/UNOS database was used to select type I diabetics, age 20 to 59 yr, who received a solitary first-time kidney transplant between January 2000 and April 2007. Dual organ transplants other than SPKTs were excluded. Patients with a subsequent pancreas transplant after LDKT or DDKT were included in our study. Follow-up data were available through May 2007. Of the 11,362 patients in the study population, 5352 (47.1%), 3309 (29.1%), and 2701 (23.8%) received a SPKT, LDKT, and DDKT, respectively.
Donor, recipient, and transplant characteristics were described using means ± SD or frequencies. The Wilcoxon rank-sum was used to test for significant differences in continuous variables. The chi-square test was used to compare categorical variables. Univariate comparisons of graft and patient survival were performed using the Kaplan-Meier product limit method, with significance tested using the log rank test. Overall kidney graft survival was determined from the date of transplantation until death or return to dialysis. Patients were censored at the end of the study period.
Covariates examined on univariate analyses were kidney transplant type along with donor (age, gender, race, cause of death, diabetes, hypertension, terminal serum creatinine, body mass index), recipient (age, gender, race, hypertension, cardiovascular disease, cerebrovascular disease, peripheral vascular disease, body mass index, waiting time, dialysis time, peak panel reactive antibody), and transplant (year of transplant, distance traveled, cold ischemia time, HLA mismatch, immunosuppression type) factors. All covariates with a significance level ≥0.05 were eliminated. The remaining covariates were entered into multivariate analyses. Multivariate estimates of hazards of kidney graft loss and patient mortality were calculated using stepwise Cox proportional hazards. All reported P values were two-tailed. All analyses were conducted using STATA Statistical Software, Release 9.1 (StataCorp LP, College Station, TX).
Results
Baseline donor, recipient, and transplant characteristics are listed in Table 1. SPK donors were younger than living donors. Living donors had the highest proportion of females and the lowest proportion of African Americans. Donors with diabetes, hypertension, and serum creatinine >1.5 mg/dl were avoided in LDKT as compared with SPKT, and were most likely to be used in DDKT.
Table 1.
Baseline donor, recipient, and kidney graft characteristics
| Factors | SPKT (n = 5352) | LDKT (n = 3309) | DDKT (n = 2701) |
|---|---|---|---|
| Donor | |||
| mean age, years | 26.5 ± 10.8 | 40.8 ± 10.6 | 36.4 ± 16.6 |
| female, % | 32.2 | 59.8 | 39.1 |
| African American, % | 14.0 | 7.6 | 8.3 |
| trauma death, % | 68.1 | N/A | 46.4 |
| DM, % | 0.1 | 0* | 5.4 |
| HTN, % | 5.7 | 1.8 | 20.2 |
| terminal SCr > 1.5 mg/dl, % | 6.1 | 0 | 11.1 |
| BMI > 30 kg/m2, % | 41.4 | 49.7 | 58.1 |
| Recipient | |||
| mean age, years | 40.2 ± 7.8 | 42.2 ± 8.9 | 45.0 ± 8.7 |
| female, % | 38.7 | 42.4** | 41.7*** |
| African American, % | 13.0 | 8.6 | 20.4 |
| HTN, % | 83.3 | 85.1*** | 85.9** |
| CAD, % | 11.5 | 17.0 | 18.6 |
| CVD, % | 2.2 | 3.7 | 4.9 |
| PVD, % | 2.2 | 3.7 | 10.3 |
| BMI > 30 kg/m2, % | 10.7 | 18.0 | 27.5 |
| mean waiting time, days | 369 ± 356 | 244 ± 262 | 638 ± 579 |
| mean dialysis time, days | 744 ± 655 | 497 ± 552 | 1169 ± 817 |
| preemptive transplant, % | 20.6 | 32.0 | 11.0 |
| mean peak PRA, % | 5.8 ± 16.4 | 6.0 ± 16.9* | 11.3 ± 24.9 |
| Transplant | |||
| mean CIT, hours | 12.7 ± 5.8 | 2.3 ± 5.2 | 18.0 ± 8.0 |
| mean HLA DR mismatch, no. | 1.4 ± 0.6 | 1.0 ± 0.7 | 1.0 ± 0.8 |
P < 0.001 for all LDKT and DDKT variables as compared to SPKT except where noted.
P > 0.05.
P < 0.01 versus SPKT.
P < 0.05 versus SPKT.
DM, diabetes mellitus; HTN, hypertension; SCr, serum creatinine; BMI, body mass index; CAD, cardiac disease; CVD, cerebrovascular disease; PVD, peripheral vascular disease; PRA, panel reactive antibody; CIT, cold ischemia time; HLA, human leukocyte antigen.
Compared with LDKT patients, SPKT recipients were younger and had lower burden of underlying disease as measured by hypertension, cardiac disease, cerebrovascular disease, peripheral vascular disease, and obesity. LDKT recipients experienced the shortest waiting and dialysis time, whereas DDKT recipients had the longest wait and most pretransplant dialysis. Preemptive transplantation was more common in LDKT than SPKT and DDKT. There were more HLA mismatched transplants in SPKT than either LDKT or DDKT.
Early post-transplant outcomes are displayed in Table 2. LDKT had the lowest percentage of delayed graft function (DGF) of all groups. Rate of rejection at one year was higher in SPKT versus LDKT, although renal function as measured by serum creatinine was not different. Early post-transplant outcomes were poorest for the DDKT group who experienced the highest rate of primary nonfunction and DGF. SPKT had the longest initial hospital stay followed by DDKT and then LDKT.
Table 2.
Early post-transplant outcomes
| Transplant Outcomes | SPKT (n = 5352) | LDKT (n = 3309) | DDKT (n = 2701) |
|---|---|---|---|
| PNF, % | 0.34 | 0.30* | 0.93 |
| DGF, % | 7.6 | 4.5 | 25.0 |
| Rejection at discharge, % | 6.0 | 6.4* | 8.0** |
| Rejection at 1 year, % | 15.5 | 11.9 | 15.0* |
| Mean SCr at discharge, mg/dl | 1.7 ± 1.5 | 1.7 ± 1.2* | 3.2 ± 2.5 |
| Mean SCr at 1 year, mg/dl | 1.4 ± 0.6 | 1.4 ± 0.6* | 1.5 ± 0.7 |
| Mean initial hospital stay, days | 13.3 ± 15.7 | 6.5 ± 5.6 | 8.8 ± 9.9 |
P < 0.001 for all LDKT and DDKT variables as compared to SPKT except where noted.
P > 0.05 versus SPKT.
P < 0.01 versus SPKT.
PNF, primary nonfunction; DGF, delayed graft function defined by dialysis required in first week post-transplantation; SCr, serum creatinine.
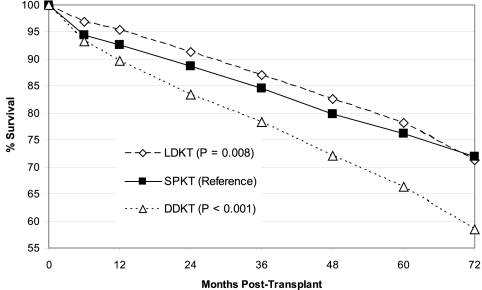
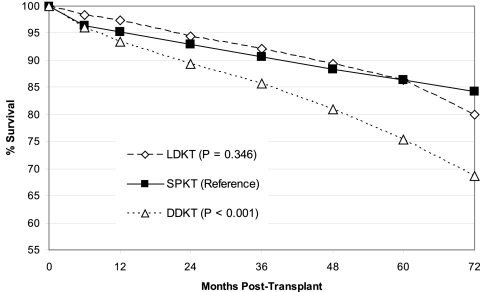
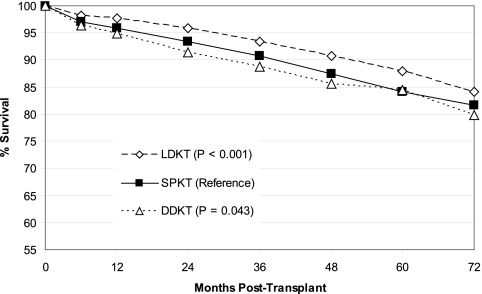
Although unadjusted kidney graft survival in LDKT was slightly better than SPKT (Figure 1), there was no significant difference in unadjusted patient survival (Figure 2) over the 72-mo follow-up. Overall kidney graft and patient survival functions, which tend to favor LDKT from months 0 to 60, do cross those of SPKT by the end of our study period. DDKT was associated with inferior unadjusted kidney graft and patient survival as compared with the other two transplant types at long-term follow-up. LDKT graft survival remained better by a small margin over SPKT on examination of unadjusted, death-censored kidney graft survival (Figure 3), and the disparity between DDKT and the other transplant groups narrowed.
Figure 1.
Unadjusted kidney graft survival by transplant type. LDKT, living donor kidney transplant; SPKT, simultaneous pancreas kidney transplant; DDKT, deceased donor kidney transplant.
Figure 2.
Unadjusted patient survival by transplant type. LDKT, living donor kidney transplant; SPKT, simultaneous pancreas kidney transplant; DDKT, deceased donor kidney transplant.
Figure 3.
Death censored kidney graft survival by transplant type. LDKT, living donor kidney transplant; SPKT, simultaneous pancreas kidney transplant; DDKT, deceased donor kidney transplant.
Donor gender, donor and recipient hypertension, donor terminal serum creatinine, recipient cerebrovascular disease, and HLA mismatch did not influence either kidney graft or patient survival on univariate analyses (P ≥ 0.05). Waiting time and peak panel reactive antibody level did not have significant univariate effects on kidney graft survival, nor did donor body mass index, recipient gender and race, year of transplant, cold ischemia time, and antibody induction on patient survival. Tables 3 and 4 list donor, recipient, and transplant factors that were significant on univariate analyses (P < 0.05) and were evaluated in multivariate models (Tables 3 and 4).
Table 3.
Multivariate Cox regression analysis for adjusted risk of graft loss
| Covariate | Reference | Hazard Ratio (95% CI) | P |
|---|---|---|---|
| Transplant type | |||
| LDKT | SPKT | 0.71 (0.61–0.83) | <0.001 |
| DDKT | 1.04 (0.91–1.18) | 0.555 | |
| Donor factors | |||
| age <20 years | 20–55 years | 0.88 (0.78–1.00) | 0.048 |
| age >55 years | 1.44 (1.21–1.72) | <0.001 | |
| African American race | Other race | 1.39 (1.20–1.61) | <0.001 |
| traumatic cause of death | Other causes | 0.87 (0.78–0.98) | 0.018 |
| history of diabetes mellitus | No disease | 1.63 (1.21–2.20) | 0.001 |
| BMI ≥30 kg/m2 | <30 kg/m2 | 1.10 (0.99–1.22) | 0.070 |
| Recipient factors | |||
| age >35 yr | ≤35 yr | 1.10 (0.99–1.23) | 0.065 |
| male sex | Female | 0.91 (0.83–1.00) | 0.054 |
| African American Race | Other race | 1.24 (1.09–1.42) | 0.001 |
| cardiovascular disease | No disease | 1.32 (1.16–1.49) | <0.001 |
| peripheral vascular disease | No disease | 1.18 (1.01–1.38) | 0.033 |
| BMI ≥30 kg/m2 | <30 kg/m2 | 1.19 (1.06–1.34) | 0.005 |
| dialysis time 1-3 years | <1 year | 1.13 (1.02–1.27) | 0.017 |
| dialysis time >3 years | 1.31 (1.15–1.49) | <0.001 | |
| Transplant factors | |||
| year transplanted prior to 2003 | 2003–2007 | 0.91 (0.82–1.01) | 0.078 |
| cold ischemia time ≥26 hr | <26 hr | 1.32 (1.07–1.61) | 0.008 |
| CNI tacrolimus | CSA or none | 0.73 (0.66–0.80) | <0.001 |
| antiproliferative MMF | Other or none | 0.70 (0.64–0.77) | <0.001 |
| induction antibody therapy | None | 0.89 (0.80–0.98) | 0.016 |
LDKT, living donor kidney transplant; SPKT, simultaneous pancreas kidney transplant; DDKT, deceased donor kidney transplant; CNI, calcineurin inhibitor; MMF, mycophenolate mofetil.
Table 4.
Multivariate Cox regression analysis for adjusted risk of patient death
| Covariate | Reference | Hazard Ratio (95% CI) | P |
|---|---|---|---|
| Transplant type | |||
| LDKT | SPKT | 0.78 (0.65–0.94) | 0.007 |
| DDKT | 1.16 (0.99–1.35) | 0.070 | |
| Donor factors | |||
| age >55 years | ≤55 years | 1.31 (1.05–1.63) | 0.016 |
| African American race | Other race | 1.43 (1.20–1.71) | <0.001 |
| traumatic cause of death | Other causes | 0.86 (0.74–0.98) | 0.029 |
| history of diabetes mellitus | No disease | 1.45 (0.98–2.16) | 0.063 |
| Recipient factors | |||
| age 35–50 years | ≤35 years | 1.18 (1.01–1.37) | 0.034 |
| age >50 years | 1.45 (1.21–1.74) | <0.001 | |
| cardiovascular disease | No disease | 1.42 (1.22–1.65) | <0.001 |
| peripheral vascular disease | No disease | 1.38 (1.16–1.66) | <0.001 |
| BMI ≥30 kg/m2 | <30 kg/m2 | 1.24 (1.07–1.44) | 0.004 |
| waiting time ≥1 year | <1 year | 1.15 (1.02–1.31) | 0.028 |
| dialysis time 1–3 years | <1 year | 1.22 (1.06–1.41) | 0.005 |
| dialysis time >3 years | 1.63 (1.15–1.49) | <0.001 | |
| peak PRA ≥30% | <30% | 1.24 (1.01–1.52) | 0.036 |
| Transplant factors | |||
| CNI tacrolimus | CSA or none | 0.72 (0.64–0.82) | <0.001 |
| antiproliferative MMF | Other or none | 0.73 (0.64–0.83) | <0.001 |
LDKT, living donor kidney transplant; SPKT, simultaneous pancreas kidney transplant; DDKT, deceased donor kidney transplant; PRA, panel reactive antibody; CNI, calcineurin inhibitor; CSA, cyclosporine; MMF, mycophenolate mofetil.
Multivariate analyses suggest that LDKT was associated with a lower risk of kidney graft failure (hazard ratio [HR] 0.71; 95% confidence interval 0.61 to 0.83) and patient death (HR 0.78; 95% confidence interval 0.65 to 0.94) when compared with SPKT. No significant differences between SPKT and DDKT were noted after multivariate analyses.
Discussion
SPKT is an attractive option for type I diabetics. A functional pancreas graft may improve quality of life over kidney transplant alone, eliminate the need for exogenous insulin therapy, and enhance glycemic control (17). Pancreas transplantation may also ameliorate chronic microvascular disease such as diabetic nephropathy, retinopathy, and neuropathy (18–21). Furthermore, long-term cardiovascular mortality, the leading cause of death in kidney transplant recipients, may be reduced in recipients of SPKT compared with kidney transplant alone (22,23).
Despite the aforementioned benefits, the effect of SPKT on survival outcomes is not clear, particularly compared with LDKT. Prior studies suggest that long-term kidney graft and patient survival are similar between SPKT and LDKT (9,11). Because trends in transplantation management are constantly in flux, we undertook a study of a recent cohort of type I diabetics who received a kidney transplant.
Does SPKT Lead to Improved Outcomes Compared with LDKT?
Graft Survival
Our study did not demonstrate improved outcomes with SPKT compared with LDKT. In fact, we found that LDKT was associated with slightly higher overall kidney graft survival compared with SPKT. Death-censored survival functions remain favorable for LDKT, suggesting that differences in patient mortality are not the sole reason for this finding. Our LDKT results may, in part, be explained by multiple factors that portend better post-transplant outcomes, including reduced dialysis exposure, increased rate of preemptive transplantation, a higher proportion of donors and recipients who were non-African American, shorter cold ischemia time, lower donor terminal serum creatinine, and more HLA matching (12,24–26). Nonetheless, after adjusting for these and other factors, LDKT was associated with a 29% reduction in relative risk of kidney graft loss compared with SPKT.
Patient Survival
There was no significant difference in overall patient survival between recipients of SPKT and LDKT. Compared with SPKT recipients, LDKT patients had several high-risk characteristics, including older donor and recipient age, and a higher rate of comorbid illness, including cardiac disease (27–30). After adjusting for these and other potential confounders on multivariate analysis, LDKT was associated with improved patient survival over SPKT. To some extent, this finding may be attributed to the complicated nature of the SPKT procedure and its higher rates of myocardial infarction, infection, and acute rejection within the early post-transplant period (31,32). SPKT patients in our study had a longer initial hospital stay and higher rates of both DGF and acute rejection at 1 yr as compared with LDKT, suggesting a more difficult early post-transplant course. The divergence of survival functions for kidney graft survival appeared greatest at 12 mo post-transplant, with a cross-sectional disparity of 2.8% favoring LDKT.
Our findings extend the results of a previous OPTN/UNOS registry analysis by Reddy et al. of type I diabetics who received a kidney transplant from 1987 to 1996 (9). In that study, SPKT was associated with a higher mortality risk (HR 2.2, P < 0.001) through 18 mo post-transplantation versus LDKT, results which are in concordance with our observations of poorer early SPKT outcomes. However, in contrast to our study, Reddy et al. found no difference in long-term outcomes, as there was a lower mortality risk of SPKT compared with LDKT after 18 mo (HR 0.86, P < 0.02) such that adjusted patient survival was equivalent at 8 yr follow-up.
The results of our current study may differ from those of Reddy et al. for a number of reasons. Most important, although the prior study accounted for various recipient characteristics such as age, gender, and race, it did not adjust for recipient comorbidities, such as cardiac disease and peripheral vascular disease. These factors often exclude SPKT candidacy given the increased risks of adverse events after transplantation (28,33,34). As stated earlier, these pre-existing disease states were more often seen in our older LDKT population and were taken into account.
Also, the study by Reddy et al. was performed using data from a different era in transplantation (1987 to 1996). Strategies for post-transplantation immunosuppression are continually in evolution (16). Since 1995, induction antibody therapy and combination tacrolimus/mycophenolate maintenance immunosuppression have been increasingly used by transplant centers. Such changes may have had more significance for LDKT outcomes than SPKT. For instance, induction antibody therapy for kidney transplant alone was prescribed 38.5% of the time in 1996 as compared with 73.6% in 2005, whereas its use in SPKT fell from 81.9% to 78.1% (13).
Does SPKT Lead to Improved Outcomes Compared with DDKT?
We found that SPKT was associated with significant long-term improvements in unadjusted kidney graft and patient survival when compared with DDKT. Superior kidney graft and patient outcomes with SPKT over DDKT have also been documented by multiple other observational studies (6–10,35). Short-term outcomes for SPKT in our study were better, with a lower rate of primary nonfunction, DGF, and acute rejection at discharge. Our death-censored graft survival curves suggest that patient death was a more substantial cause for kidney graft loss in DDKT versus the other transplant types.
After multivariate analyses, there were no differences between SPKT and DDKT with regards to kidney graft or patient survival, despite the dramatic divergence seen in our unadjusted survival curves. Likely, SPKT and DDKT recipients are two disparate populations that cannot be compared without accounting for potential confounders. Favorable donor, recipient, and transplant factors, rather than transplant type, appear to explain the superior SPKT outcomes. Compared with DDKT, SPKT had younger donors and recipients, fewer donor and recipient baseline comorbidities, lower donor terminal creatinine, and shorter dialysis and cold ischemia time. A prior study by our group looking at SPKT and DDKT outcomes for type I diabetics who received transplants from 1994 to 1997 uncovered similar findings after multivariate modeling (8).
Study Limitations/Future Directions
As with all retrospective, observational studies, our results should be interpreted with care. Our unadjusted kidney graft and patient survival curves do cross and favor SPKT over LDKT by the end of our 72-mo study period. This finding may be due to finally realized long-term benefits associated with a functioning pancreas. Euglycemia may modulate diabetic complications, including accelerated cardiovascular disease, a primary cause of post-transplant death (23). However, such chronic benefits may not be readily apparent until 5 to 10 yr post-SPKT (22). Exchange of unadjusted survival curves may also be caused by factors independent of transplant type, as multivariate analysis reveals a patient survival benefit for LDKT over SPKT that did not previously exist. We are unable to analyze outcomes at times beyond our study period, which was designed to assess current trends in transplantation management.
Our study is also limited to the information available in the OPTN/UNOS database. We cannot track the development or progression of diabetic complications such as diabetic nephropathy, retinopathy, or neuropathy. These complications may be curtailed by a functioning pancreas graft. Moreover, SPKT may enhance diabetes-associated quality of life by eliminating glucose monitoring and insulin injections (17,36,37). We were not able to assess lifestyle benefits because of the lack of such documentation. Future studies that follow diabetic complications and quality of life may be central in evaluating the role of SPKT.
In addition, we cannot identify patients with brittle type I diabetes in our analysis. Likely, most eligible brittle type I diabetics were listed for SPKT as a result of difficulty with controlling erratic blood sugars solely with medical management (5). We feel that these patients should continue to be listed for SPKT, particularly because diabetes may be exacerbated by immunosuppressive medications such as steroids and calcineurin inhibitors.
Although our multivariate models adjusted for the documented differences in donor and recipient characteristics, there may be unaccounted risk factors that affect outcomes of the kidney transplant groups. Mortality of patients on the wait list may create a selection bias toward those who live long enough to receive a kidney transplant. This phenomenon could not be assessed given the limitations of our data set. Selection bias may also occur at the time of listing as certain LDKT and DDKT recipients may not be suitable for SPKT because of the higher surgical risk (34). There may be center-specific bias on outcomes, because only approximately 60% of all kidney transplant centers perform SPKT, and the volume of SPKT performed per year may vary from center to center. Our analysis did not take this into account because our database did not have center-specific data.
Finally, the specific effect of pancreas after kidney transplant (PAKT) was not assessed. Patients with subsequent PAKT were not separated from our study groups so as to maintain a comprehensive study population. Likely many of these patients were eligible for SPKT at the onset of transplant listing. Decision to undergo PAKT was felt to be inherent to the choice of initial LDKT or DDKT over SPKT.
Whether a successful PAKT results in a survival benefit as compared with those on the PAKT waiting list is controversial (32,38). The primary added risk of PAKT is associated with potential surgical and anesthetic complications (39). Perioperative mortality in LDKT patients with subsequent PAKT could partly account for the narrowing of the LDKT survival advantage in our latter study period. Also, PAKT recipients encounter another antigenic challenge and may require increased immunosuppression. Higher doses of calcineurin inhibitor and overall immunosuppression could also diminish kidney graft and patient survival post-PAKT.
On the other hand, PAKT in select LDKT or DDKT recipients may improve long-term outcomes as a result of endogenous insulin production. If true, PAKT after LDKT may be preferred over SPKT in those with available donors, particularly because it frees up deceased donor kidneys that are taken for SPKT. Therefore, further study is paramount to evaluate the consequences of PAKT on eligible LDKT and DDKT recipients.
Conclusions
The kidney transplant waiting list continues to grow each year. It reached almost 58,000 patients in 2004, a time in which just slightly more than 9000 deceased donor kidneys were available for transplant (14). As our organ shortage crisis progresses to staggering proportions, it is critical to consider all available sources of kidney donation. Our results may encourage the increased use of LDKT in type I diabetics with an available donor, particularly because LDKT was associated with lower adjusted risks of kidney graft loss and patient death during our study period. In those without a potential living donor, SPKT may be preferred over DDKT given the overall survival benefit and associated favorable donor characteristics with SPKT.
Disclosures
None.
Acknowledgments
Preliminary findings of this work were reported in abstract form at the 2007 American Society of Nephrology Renal Week in San Francisco, CA.
Dr. Brian Young was supported in part by National Institutes of Health Training Grant T32-DK07789. Dr. Jagbir Gill was supported in part by the Kidney Research Core Education and National Training (KRESCENT) Program and the University of British Columbia Clinical Investigator Program.
This work also was supported in part by the U.S. Department of Health and Human Services, Health Resources and Services Administration, Office of Special Programs, Division of Transplantation, under Contract 231 to 00–0115, for the operation of the Organ Procurement and Transplantation Network. The content is the responsibility of the authors alone and does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Published online ahead of print. Publication date available at www.cjasn.org.
See related editorial, “Best Option for Transplant Candidates with Type 1 Diabetes and a Live Kidney Donor: A Bird in the Hand is Worth Two in the Bush,” on pages 700–702.
References
- 1.Wolfe RA, Ashby VB, Milford EL, Ojo AO, Ettenger RE, Agodoa LY, Held PJ, Port FK: Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med 341: 1725–1730, 1999 [DOI] [PubMed] [Google Scholar]
- 2.Meier-Kriesche HU, Ojo AO, Port FK, Arndorfer JA, Cibrik DM, Kaplan B: Survival improvement among patients with end-stage renal disease: trends over time for transplant recipients and wait-listed patients. J Am Soc Nephrol 12: 1293–1296, 2001 [DOI] [PubMed] [Google Scholar]
- 3.Oniscu GC, Brown H, Forsythe JL: Impact of cadaveric renal transplantation on survival in patients listed for transplantation. J Am Soc Nephrol 16: 1859–1865, 2005 [DOI] [PubMed] [Google Scholar]
- 4.United States Renal Data System: Annual Data Report: Atlas of End-Stage Renal Disease in the United States. Bethesda MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2007
- 5.Gaston RS, Basadonna G, Cosio FG, Davis CL, Kasiske BL, Larsen J, Leichtman AB, Delmonico FL: Transplantation in the diabetic patient with advanced chronic kidney disease: a task force report. Am J Kidney Dis 44: 529–542, 2004 [PubMed] [Google Scholar]
- 6.Becker BN, Brazy PC, Becker YT, Odorico JS, Pintar TJ, Collins BH, Pirsch JD, Leverson GE, Heisey DM, Sollinger HW: Simultaneous pancreas-kidney transplantation reduces excess mortality in type 1 diabetic patients with end-stage renal disease. Kidney Int 57: 2129–2135, 2000 [DOI] [PubMed] [Google Scholar]
- 7.Ojo AO, Meier-Kriesche HU, Hanson JA, Leichtman A, Magee JC, Cibrik D, Wolfe RA, Port FK, Agodoa L, Kaufman DB, Kaplan B: The impact of simultaneous pancreas-kidney transplantation on long-term patient survival. Transplantation 71: 82–90, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Bunnapradist S, Cho YW, Cecka JM, Wilkinson A, Danovitch GM: Kidney allograft and patient survival in type I diabetic recipients of cadaveric kidney alone versus simultaneous pancreas kidney transplants: A multivariate analysis of the UNOS database. J Am Soc Nephrol 14: 208–213, 2003 [DOI] [PubMed] [Google Scholar]
- 9.Reddy KS, Stablein D, Taranto S, Stratta RJ, Johnston TD, Waid TH, McKeown JW, Lucas BA, Ranjan D: Long-term survival following simultaneous kidney-pancreas transplantation versus kidney transplantation alone in patients with type 1 diabetes mellitus and renal failure. Am J Kidney Dis 41: 464–470, 2003 [DOI] [PubMed] [Google Scholar]
- 10.Israni AK, Feldman HI, Propert KJ, Leonard M, Mange KC: Impact of simultaneous kidney-pancreas transplant and timing of transplant on kidney allograft survival. Am J Transplant 5: 374–382, 2005 [DOI] [PubMed] [Google Scholar]
- 11.Rayhill SC, D'Alessandro AM, Odorico JS, Knechtle SJ, Pirsch JD, Heisey DM, Kirk AD, Van der Werf W, Sollinger HW: Simultaneous pancreas-kidney transplantation and living related donor renal transplantation in patients with diabetes: Is there a difference in survival? Ann Surg 231: 417–423, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Becker BN, Rush SH, Dykstra DM, Becker YT, Port FK: Preemptive transplantation for patients with diabetes-related kidney disease. Arch Intern Med 166: 44–48, 2006 [DOI] [PubMed] [Google Scholar]
- 13.2006 Annual Report of the U.S. Organ Procurement and Transplantation Network and the Scientific Registry of Transplant Recipients: Transplant Data 1996–2005, Department of Health and Human Services, Health Resources and Services Administration, Healthcare Systems Bureau, Division of Transplantation, Rockville, MD; United Network for Organ Sharing, Richmond, VA; University Renal Research and Education Association, Ann Arbor, MI, 2006
- 14.Cohen DJ, St Martin L, Christensen LL, Bloom RD, Sung RS: Kidney and pancreas transplantation in the United States, 1995–2004. Am J Transplant 6: 1153–1169, 2006 [DOI] [PubMed] [Google Scholar]
- 15.Ming CS, Chen ZH: Progress in pancreas transplantation and combined pancreas-kidney transplantation. Hepatobiliary Pancreat Dis Int 6: 17–23, 2007 [PubMed] [Google Scholar]
- 16.Meier-Kriesche HU, Li S, Gruessner RW, Fung JJ, Bustami RT, Barr ML, Leichtman AB: Immunosuppression: evolution in practice and trends, 1994–2004. Am J Transplant 6: 1111–1131, 2006 [DOI] [PubMed] [Google Scholar]
- 17.Gross CR, Limwattananon C, Matthees B, Zehrer JL, Savik K: Impact of transplantation on quality of life in patients with diabetes and renal dysfunction. Transplantation 70: 1736–1746, 2000 [DOI] [PubMed] [Google Scholar]
- 18.Navarro X, Sutherland DE, Kennedy WR: Long-term effects of pancreatic transplantation on diabetic neuropathy. Ann Neurol 42: 727–736, 1997 [DOI] [PubMed] [Google Scholar]
- 19.Fioretto P, Steffes MW, Sutherland DE, Goetz FC, Mauer M: Reversal of lesions of diabetic nephropathy after pancreas transplantation. N Engl J Med 339: 69–75, 1998 [DOI] [PubMed] [Google Scholar]
- 20.Fioretto P, Sutherland DE, Najafian B, Mauer M: Remodeling of renal interstitial and tubular lesions in pancreas transplant recipients. Kidney Int 69: 907–912, 2006 [DOI] [PubMed] [Google Scholar]
- 21.Giannarelli R, Coppelli A, Sartini MS, Del Chiaro M, Vistoli F, Rizzo G, Barsotti M, Del Prato S, Mosca F, Boggi U, Marchetti P: Pancreas transplant alone has beneficial effects on retinopathy in type 1 diabetic patients. Diabetologia 49: 2977–2982, 2006 [DOI] [PubMed] [Google Scholar]
- 22.Biesenbach G, Konigsrainer A, Gross C, Margreiter R: Progression of macrovascular diseases is reduced in type 1 diabetic patients after more than 5 years successful combined pancreas-kidney transplantation in comparison to kidney transplantation alone. Transpl Int 18: 1054–1060, 2005 [DOI] [PubMed] [Google Scholar]
- 23.Orsenigo E, Socci C, Fiorina P, Zuber V, Secchi A, Di Carlo V, Staudacher C: Cardiovascular benefits of simultaneous pancreas-kidney transplant versus kidney alone transplant in diabetic patients. Transplant Proc 37: 3570–3571, 2005 [DOI] [PubMed] [Google Scholar]
- 24.Takemoto SK, Cho YW and Gjertson DW: Transplant risks. Clin Transpl 325–34, 1999 [PubMed]
- 25.Siddiqi N, McBride MA, Hariharan S: Similar risk profiles for post-transplant renal dysfunction and long-term graft failure: UNOS/OPTN database analysis. Kidney Int 65: 1906–1913, 2004 [DOI] [PubMed] [Google Scholar]
- 26.Giblin L, O'Kelly P, Little D, Hickey D, Donohue J, Walshe JJ, Spencer S, Conlon PJ: A comparison of long-term graft survival rates between the first and second donor kidney transplanted–the effect of a longer cold ischaemic time for the second kidney. Am J Transplant 5: 1071–1075, 2005 [DOI] [PubMed] [Google Scholar]
- 27.Terasaki PI, Gjertson DW, Cecka JM, Takemoto S, Cho YW: Significance of the donor age effect on kidney transplants. Clin Transplant 11: 366–372, 1997 [PubMed] [Google Scholar]
- 28.Woo YM, McLean D, Kavanagh D, Ward L, Aitken S, Miller GJ, Egan P, Hughes K, Clark L, Carswell K, Morris ST, Northridge DB, Rodger RS, Jardine AG: The influence of pre-operative electrocardiographic abnormalities and cardiovascular risk factors on patient and graft survival following renal transplantation. J Nephrol 15: 380–386, 2002 [PubMed] [Google Scholar]
- 29.Keith DS, Demattos A, Golconda M, Prather J, Norman D: Effect of donor recipient age match on survival after first deceased donor renal transplantation. J Am Soc Nephrol 15: 1086–1091, 2004 [DOI] [PubMed] [Google Scholar]
- 30.Kauffman HM, McBride MA, Cors CS, Roza AM, Wynn JJ: Early mortality rates in older kidney recipients with comorbid risk factors. Transplantation 83: 404–410, 2007 [DOI] [PubMed] [Google Scholar]
- 31.Douzdjian V, Abecassis MM, Corry RJ, Hunsicker LG: Simultaneous pancreas-kidney versus kidney-alone transplants in diabetics: Increased risk of early cardiac death and acute rejection following pancreas transplants. Clin Transplant 8: 246–251, 1994 [PubMed] [Google Scholar]
- 32.Gruessner RW, Sutherland DE, Gruessner AC: Mortality assessment for pancreas transplants. Am J Transplant 4: 2018–2026, 2004 [DOI] [PubMed] [Google Scholar]
- 33.Gruessner RW, Dunn DL, Gruessner AC, Matas AJ, Najarian JS, Sutherland DE: Recipient risk factors have an impact on technical failure and patient and graft survival rates in bladder-drained pancreas transplants. Transplantation 57: 1598–1606, 1994 [PubMed] [Google Scholar]
- 34.Becker BN, Odorico JS, Becker YT, Groshek M, Werwinski C, Pirsch JD, Sollinger HW: Simultaneous pancreas-kidney and pancreas transplantation. J Am Soc Nephrol 12: 2517–2527, 2001 [DOI] [PubMed] [Google Scholar]
- 35.Knoll GA, Nichol G: Dialysis, kidney transplantation, or pancreas transplantation for patients with diabetes mellitus and renal failure: A decision analysis of treatment options. J Am Soc Nephrol 14: 500–515, 2003 [DOI] [PubMed] [Google Scholar]
- 36.Sureshkumar KK, Mubin T, Mikhael N, Kashif MA, Nghiem DD, Marcus RJ: Assessment of quality of life after simultaneous pancreas-kidney transplantation. Am J Kidney Dis 39: 1300–1306, 2002 [DOI] [PubMed] [Google Scholar]
- 37.Sureshkumar KK, Patel BM, Markatos A, Nghiem DD, Marcus RJ: Quality of life after organ transplantation in type 1 diabetics with end-stage renal disease. Clin Transplant 20: 19–25, 2006 [DOI] [PubMed] [Google Scholar]
- 38.Venstrom JM, McBride MA, Rother KI, Hirshberg B, Orchard TJ, Harlan DM: Survival after pancreas transplantation in patients with diabetes and preserved kidney function. JAMA 290: 2817–2823, 2003 [DOI] [PubMed] [Google Scholar]
- 39.Hariharan S, Pirsch JD, Lu CY, Chan L, Pesavento TE, Alexander S, Bumgardner GL, Baasadona G, Hricik DE, Pescovitz MD, Rubin NT, Stratta RJ: Pancreas after kidney transplantation. J Am Soc Nephrol 13: 1109–1118, 2002 [DOI] [PubMed] [Google Scholar]