Abstract
BACKGROUND:
Adhesion formation after peritoneal surgery is a major cause of postoperative bowel obstruction, infertility, and chronic pelvic pain. In this study, we compared the possible individual effects of phosphatidylcholine (PC), Seprafilm® II, and tissue plasminogen activator (t-PA) and the combined effects of phosphatidylcholine and t-PA on postoperative adhesion formation in a rat surgical model.
MATERIALS AND METHODS:
A total of 50 Wistar male rats underwent median laparotomy and standardized abrasion of the visceral and parietal peritoneum. phosphatidylcholine, Seprafilm II, and t-PA alone and phosphatidylcholine and t-PA in combination were applied intraperitoneally at the end of the surgical procedure. Seven days after surgery, a relaparotomy was performed for adhesion grading and histopathological examination.
RESULTS:
A comparison of adhesion stages demonstrated a significant difference between the control group and the study groups (p<0.001). The adhesion grade of the combined treatment group was statistically different from that of the other groups (p<0.05). In the t-PA group and the combined group, six and two rats, respectively, developed hematomas locally on the cecum.
CONCLUSIONS:
PC, t-PA, and Seprafilm II used individually reduced the adhesion grade. The t-PA and phosphatidylcholine combination was most effective in reducing adhesion formation. On the other hand, usage of t-PA alone or in combination may increase risk of bleeding. More detailed studies are needed, and future studies on the efficacy of a material for decreasing adhesion formation should include a comparison of several control materials in the same model.
Keywords: Intraabdominal adhesion, Phosphatidylcholine, Tissue plasminogen activator, Seprafilm
INTRODUCTION
Peritoneal adhesions are fibrous bands of tissue that join intraabdominal organs to each other or to the abdominal wall. These adhesions are a major complication in healing following surgery or infection and can lead to conditions such as intestinal obstruction, infertility, and chronic pain.1,2 Furthermore, the presence of dense adhesions makes reoperation technically difficult.3
Adhesions are formed when the parietal or visceral peritoneum is damaged and the basal membrane of the mesothelial layer is exposed to surrounding tissues. The sequence of adhesion formation has been reported as follows: tissue ischemia, inflammation, fibrin deposition, fibrin organization, collagen formation, and maturation with the formation of adhesions.1–4 Surgeons have used several agents to reduce adhesion formation at each of these steps. However, at least 50% of patients still develop significant adhesions.5–15 Therefore, new approaches to this problem are warranted.
Physical barriers, including both mechanical and viscous solutions, are widely used to prevent adhesion formation by limiting tissue apposition during the critical stages of mesothelial repair. Seprafilm II is a translucent bioresorbable membrane composed of chemically modified carboxymethylcellulose and sodium hyaluronate.5–7,16 Phosphatidylcholine (PC) is a viscous solution that may act as a temporary membrane by covering serosal defects. PC is the main constituent of surface active material coating the peritoneal membrane as well as an excellent lumbricant.17–21
Fibrinolytic activity of the traumatized peritoneum is another key point for preventing adhesion formation. When the fibrinolytic capacity of the peritoneum is insufficient, deposited fibrin may persist, and fibrinous adhesions may develop. The dissolution of fibrin is mediated by the fibrinolytic system. In this system, the inactive proenzyme plasminogen is converted into active plasmin by tissue plasminogen activator (t-PA). 22–25
In light of the above-mentioned subjects, we tried to test (separately and in association) agents that could potentially function as inhibitors of the formation of peritoneal adhesions endowed with different mechanisms of action. Therefore, we have compared the possible effects of PC, Seprafilm II, and tPA used in isolation, and PC and tPA combined, on postoperative adhesion formation in rats.
MATERIALS AND METHODS
Animals and Anesthesia
This study was conducted after approval by the Gazi University School of Medicine (No:55-11336) Ethics Committee and supported by Gazi University Research Project (TF.01/2002-111). Fifty male Wistar rats weighing 200–250 g were used. The animals were kept in single cages under standard laboratory conditions with a balanced pellet diet and water ad libitum. The animals were housed at the Center for Laboratory Animal Care of Gazi University. The animals were acclimatized for one week before the experiments. After adaptation, the animals were randomly assigned to five different groups of equal numbers. The rats were prepared for surgery with an injection of ketamine (70 mg/kg i.m. of Ketalar; Eczaciba i, Istanbul, Turkey) anesthesia. The surgical procedures were performed under sterile conditions.
Induction of Adhesions
All of the animals were fasted for at least 12 hours immediately before surgery. After hair removal, the abdomen was cleaned with 1% antiseptic povidone-iodine solution and a 3 cm midline laparotomy was made. The cecum was exposed and abrased for 10 strokes with moderate pressure using a nylon bristle dental brush. Petechial subserosal hemorrhages developed in all cases.26
Experimental Groups
The abdominal cavity was closed by a running suture of 3-0 silk. Prior the last stitch, the following agents were deposited in the abdominal cavity.
Group 1 (Control group): 2 ml/rat of 0.9% NaCl, Intraperitoneal (i.p.). (Serum Fizyolojik, Eczaciba i, Istanbul, Turkey).
Group 2 (Seprafilm®II group): The abrased cecal area was covered by a 1.5 cm2 piece of Seprafilm® II (Genzyme Corporation, Cambridge, MA, USA).
Group 3 (PC group): 20 mg/rat of phosphatidylcholine, i.p. (Lipostabil, A. Nattermann & Cie. GmbH, Köln, Germany)
Group 4 (t-PA group): 0.001 mg/rat t-PA in 2 ml of 0.9% NaCl, i.p. (Sigma Aldrich Co, Steinheim, Germany)
Group 5 (t-PA and PC combination group): 0.001 mg/rat t-PA in 2 ml of 0.9% NaCl and 20 mg/rat phosphatidylcholine combination, i.p.
Assessment of Adhesions
Seven days after surgery, the animals were sacrificed. The abdominal cavity was opened via a U-shaped incision based in the lower abdomen for complete exploration. Blind evaluations were carried out; adhesions were graded from 0 (absent) to 4 (severe) according to the Mazuji classification27 (Table 1). In general, Grades 0 and 1 adhesions have no clinical significance, whereas Grades 3 and 4 adhesions can cause intestinal obstruction. Adhesion rate is used to describe the presence of an adhesion of any grade.
Table 1 -.
Adhesion grading according to Mazuji classification27
| Grade | Description of Grade |
|---|---|
| 0 | No adhesion |
| 1 | Very small, irregular adhesion |
| 2 | Easily separable medium intensity adhesion |
| 3 | Intense, not easily separable regular adhesion |
| 4 | Very intense, not easily separable, homogeneous adhesion |
Histology
Histopathologic examination was performed by two investigators. Adhesion-carrying tissues were excised en-block and fixed in a 10% buffered formaldehyde solution. Samples were submitted for histological analysis after being stained with hematoxylin and eosin. The parameters evaluated were fibrosis, inflammation, and vascular proliferation, rated on a modified semi-quantitative scale of 0–3.6,7,28 The amount of fibrosis was scored as follows: 0, no fibrosis; 1, minimal, loose fibrosis; 2, moderate fibrosis; and 3, florid dense fibrosis. Inflammation was scored as follows: 0, no inflammation; 1, presence of giant cells, occasional lymphocytes and plasma cells; 2, presence of giant cells, plasma cells, eosinophils and neutrophils; and 3, presence of many inflammatory cells and microabscesses. Vascular proliferation was scored as: 0, no vascular proliferation; 1, mild vascular proliferation; 2, moderate vascular proliferation; and 3; intense vascular proliferation. For identification of nerve fibers, the specimens were rinsed in a phosphate-buffered solution at pH 7.4; immunohistochemical staining with a monoclonal antibody to neurofilament was performed (S-100, Neomarkers, Fremont, California, USA). When nerve fibers were not seen, another section was made from the block, restained for neurofilament and re-examined.
Statistical Analysis
Statistical analysis was performed using the Statistical Package for Social Science (SPSS for Windows, Release 9.05, Chicago, Illinois). Differences in the numbers of animals without adhesions in the different treatment groups were evaluated using a Chi-square test. A t-test was used to determine differences between two independent populations. Kruskal-Wallis and Mann-Whitney U tests were used when necessary. A value of p<0.05 was counted as significant.
RESULTS
Adhesion Grade and Rate
Throughout the investigation, no animal died during or after surgery. Adhesion rates and grades of the groups are presented in Table 2. In comparing adhesion grades, a significant difference was found between the control group and the treatment groups (p<0.0001). However, Groups 1, 2, and 3 did not show any significant differences between themselves (p>0.05). On the other hand, in Group 5 (t-PA and PC combination group), the adhesion grade differed significantly from that of the other groups (p<0.05) (Table 2). There were no adhesions in Grades 0 or 1 in the control group. Furthermore, most adhesions in the control group were Grade 3 or 4 (90%). On the other hand, there were no Grade 4 adhesions in the other groups. Group 5 showed the lowest incidence of postsurgical adhesions as compared to other treatment and control groups (p<0.05) (Table 4).
Table 2 -.
Postoperative adhesion rates, grades and side effects for groups
| Group | No. of rats | Adhesion grade
|
Adhesion rate (%) | Side effects | ||||
|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | ||||
| 1-(Control Group) | 10 | 0 | 0 | 1 | 4 | 5 | 10/10 (100) | _ |
| 2-(Seprafilm Group) | 10 | 4 | 3 | 3 | 0 | 0 | 6/10 (60) | _ |
| 3-(PC Group) | 10 | 4 | 0 | 5 | 1 | 0 | 6/10 (60) | _ |
| 4-(t-PA Group) | 10 | 4 | 1 | 3 | 2 | 0 | 6/10 (60) | Local hematoma (n = 6) |
| 5-(PC+t-PA Group)* | 10 | 9 | 1 | 0 | 0 | 0 | 1/10 (60) | Local hematoma (n = 2) |
p < 0.05 as compared to the control group and to the other treatment groups (Groups 2, 3, and 4). Adhesion rate is used to describe the presence of an adhesion in any grade
Table 4 -.
Statistical comparison of the groups according to the adhesion grade
| Groups | P-value |
|---|---|
| Group 1 vs. Group 2 | 0.0382 |
| Group 1 vs. Group 3 | 0.0382 |
| Group 1 vs. Group 4 | 0.0382 |
| Group 1 vs. Group 5 | 0.0008 |
| Group 2 vs. Group 3 | >0.05 |
| Group 2 vs. Group 4 | >0.05 |
| Group 2 vs. Group 5 | 0.031 |
| Group 3 vs. Group 4 | >0.05 |
| Group 3 vs. Group 5 | 0.031 |
| Group 5 vs. Group 4 | 0.031 |
A value of p<0.05 was taken as significant
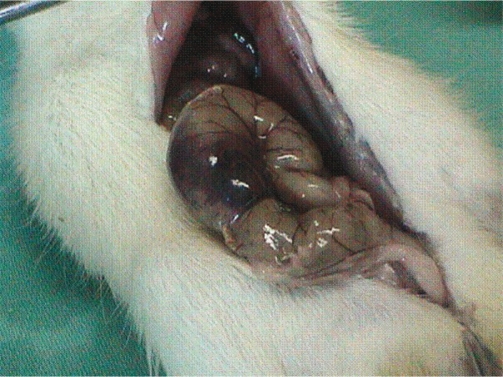
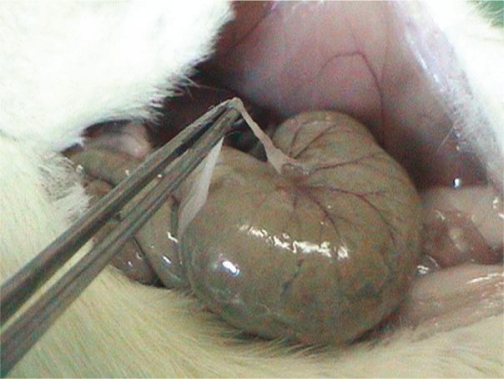
Based on these findings, it was concluded that all three products significantly reduced peritoneal adhesion development rate and adhesion grades. In Groups 4 (t-PA alone) and 5 (t-PA combined with PC), six and two rats, respectively, developed local hematomas on the cecum (Figure 1). Fifty percent of the adhesions in Group 2 (Seprafilm group) developed in the uncovered areas in the abdomen (n=3) (Figure 2).
Figure 1 -.
The appearance of local hematoma on the cecum in Group 4 (t-PA group)
Figure 2 -.
The appearance of Grade 1 adhesion in Group 2 (Seprafilm group), which developed on the uncovered area on the cecum
Histology
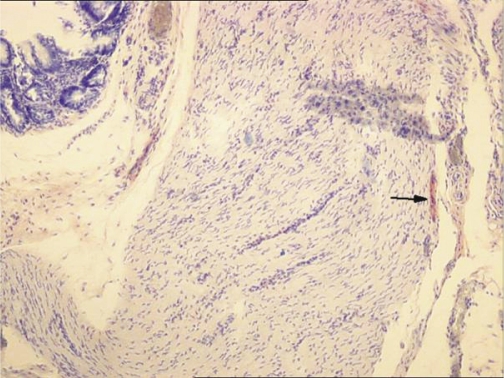
The histologic findings of adhesions differed significantly among the control group and treatment groups with respect to fibrosis (p = 0.0001), inflammation (p = 0.004), and vascular proliferation (p = 0.0001). The control group showed the highest scores for fibrosis and the lowest scores for inflammation and vascular proliferation. (Table 3). Histologic examination showed that in Groups 1 and 4, nerve fiber presence was seen in the adhesions of six rats and one rat, respectively (Figure 3). Adhesion grade influenced the presence of nerve fibers. All nerve fibers were identified in Grade 3 and 4 adhesions; there was no nerve fiber development in the other groups.
Table 3 -.
| Scores | |||||
|---|---|---|---|---|---|
| Histologic Feature | 0 | 1 | 2 | 3 | |
| Fibrosis | Group 1* | 0 | 0 | 1 | 9 |
| Group 2 | 4 | 2 | 0 | 0 | |
| Group 3 | 2 | 3 | 1 | 0 | |
| Group 4 | 3 | 2 | 1 | 0 | |
| Group 5 | 0 | 1 | 0 | 0 | |
| Inflammation | Group 1* | 8 | 1 | 1 | 0 |
| Group 2 | 1 | 0 | 2 | 3 | |
| Group 3 | 0 | 0 | 2 | 4 | |
| Group 4 | 0 | 0 | 3 | 3 | |
| Group 5 | 0 | 0 | 1 | 0 | |
| Vascular proliferation | Group 1* | 8 | 2 | 0 | 0 |
| Group 2 | 0 | 1 | 4 | 1 | |
| Group 3 | 0 | 0 | 2 | 4 | |
| Group 4 | 0 | 0 | 3 | 3 | |
| Group 5 | 0 | 0 | 1 | 0 | |
p < 0.05 compared to other treatment groups (Groups 2, 3, 4, and 5)
Figure 3 -.
The arrow indicates the microscopic appearance of nerve fibers in the adhesion between the cecum and the omentum in Group 1 (control group) (S-100 × 100)
DISCUSSION
Postsurgical adhesion formation is a significant clinical problem for every surgical specialty. Evaluation of the causes and means of prevention of adhesion formation has been the major goal of many investigations. Several drugs and substances are used locally or systematically for this purpose, including mechanical barriers and physical, chemical, and pharmacological agents.5–15 However, despite positive reports, none of these have been adopted for standard therapy. Research in adhesion prevention has focused strongly on barrier films, fibrinolytic agents and phospholipids.5–7,16–21
With the barrier technique, surgically traumatized surfaces are kept covered during mesothelial regeneration, thus preventing adherence of adjacent structures and reducing adhesion formation. Seprafilm® II is a bioresorbable membrane based on a chemically modified form of hyaluronic acid and carboxymethylcellulose. Hyaluronic acid exists in all tissues and constitutes the major component of the intercellular matrix.29 Because Seprafilm® II is the only biosynthetic material that has been studied extensively in well-designed randomized controlled clinical trails,30–32 we preferred to use Seprafilm® II for comparison with the other agents used in this study. In the present study, Seprafilm® II significantly reduced adhesion formation (p<0.05). There was no adhesion formation of Grades 3 or 4 in the Seprafilm group (Group 2). In fact, most of the adhesions in Group 2 developed in uncovered areas in the abdomen. This fact underlines the necessity of using liquid anti-adhesive agents to cover all potential peritoneal lesions.
The surface-active material coating the peritoneum is chemically similar to pulmonary surfactants.17,18 Thin layer chromatography showed that 81% of these are phosphatidylcholines.19 Ar’rajab et al.21 speculated that the local formation of fibrin might be prevented by the lubricant action of PC since the tissue defects are covered. Phospholipids are composed of both hydrophobic and hydrophilic parts; it has been suggested that phospholipids adhere to the negatively charged peritoneum by their positively charged head-group in such a way that their hydrophobic tails are oriented into the cavity and coming into contact with hydrophobic components from the opposing side. Thus, by acting as a liquid barrier separating the opposite areas of the peritoneum by a very thin membrane-like film, PC may reduce adhesion formation.19,21 The present study showed that PC reduced adhesion formation when compared with the control group (p<0.05).
The idea that normal peritoneum must have inherent fibrinolytic activity that could prevent the formation of adhesions has been described previously.24,33 Since then, evaluation of changes in fibrinolytic activity after peritoneal trauma has been the subject of several studies.23–25 Injury to the peritoneum because of surgery, infection or irritation causes a local ischemia and inflammatory reaction, which leads to the formation of a serosanguineous exudate rich in fibrin. When the fibrinolytic capacity of the peritoneum is insufficient, deposited fibrin may persist, resulting in fibrinous adhesions which then become organized. As a result, these adhesions become permanent.24 Fibrin is principally degraded by plasmin, a protease converted from plasminogen by plasminogen activator (PA). Thus, an imbalance between fibrin deposition and fibrin dissolution may be the key event in adhesion formation. It was postulated that an early reduction in peritoneal PA activity might be secondary to a reduction in t-PA levels, whereas the subsequent abolition of functional fibrinolytic activity is probably caused by a dramatic increase in plasminogen activator inhibitor 1 (PAI-1) and plasminogen activator inhibitor 2 (PAI-2) concentrations.23 In the present study, we used t-PA, which is derived from human melanoma cells, to supplement the insufficient PA activity of traumatized peritoneum. This kind of t-PA is absorbed by fibrin clots in particular and only there exhibits effects; even when introduced into circulating blood, these t-PAs have little or no general side effects in hemostasis.24,25 On the contrary, in the present study, six Group 4 rats and two Group 5 rats developed local hematomas on the cecum. The hematomas were smaller in Group 5 rats. t-PA significantly reduced adhesion stage compared to the control group (p<0.05). Based on these findings, we can conclude that all three products significantly reduce peritoneal adhesion development rate and adhesion stage.
Since various attempts made at different stages of adhesion pathogenesis failed to prevent adhesion formation, we conclude that there may not be a single way to prevent adhesion formation. Instead, a combination treatment acting on different stages of adhesion pathogenesis simultaneously may be successful. Thus, for Group 5, we combined t-PA and PC. Nine rats (90%) in Group 5 were adhesion-free, and only one rat developed a Grade 1 adhesion. Comparing the groups with regard to adhesion stage shows a significant difference between Group 5 (t-PA and PC combined) and the other treatment groups. In the present study, use of t-PA, in both groups administered, caused local hematomas with various degree; the clinical result would be a possible disturbance of the delicate balance between coagulation and fibrinolysis, causing a postoperative bleeding hazard.
The histologic findings on adhesions showed that the control group had the highest fibrosis scores and the lowest inflammation and vascular proliferation scores. We did not find these results surprising because similar results have been reported previously by Kaptanoglu et al.5 and Ersoy et al.6 Several authors have noted an association between adhesion and pain.2,14,24 Sulaiman et al.34 have shown the presence of sensory nerves in all human peritoneal adhesions examined. Although nerve fibers were found in some of the adhesions in our experimental design, the pathophysiology of this result was out of the scope of the present study that requires further investigation.
In conclusion, PC, t-PA, and Seprafilm II used in isolation reduced the adhesion grade; the combination of PC and t-PA was most effective in reducing adhesion formation. More detailed studies are needed on this topic, and future studies on the efficacy of a material in decreasing adhesion formation should include a comparison of several control materials.
REFERENCES
- 1.Menzies D, Ellis H. Intestinal obstruction forms adhesions: how big is the problem? Ann R Coll Surg Eng. 1990;72:60–3. [PMC free article] [PubMed] [Google Scholar]
- 2.Ellis H, Moran BJ, Thompson JN, Parker MC, Wilson MS, Menzies D, et al. Adhesion-related hospital readmissions after abdominal and pelvic surgery: A retrospective cohort study. Lancet. 1999;353:1476–80. doi: 10.1016/S0140-6736(98)09337-4. [DOI] [PubMed] [Google Scholar]
- 3.Coleman MG, McLain AD, Moran BJ. Impact of previous surgery on time taken for incision and division of adhesions during laparotomy. Dis. Colon Rectum. 2000;43:1297–9. doi: 10.1007/BF02237441. [DOI] [PubMed] [Google Scholar]
- 4.Milligan DW, Raftery AT. Observations on the pathogenesis of peritoneal adhesions: a light and electron microscopical study. Br J Surg. 1974;61:274–80. doi: 10.1002/bjs.1800610406. [DOI] [PubMed] [Google Scholar]
- 5.Kaptanoglu L, Kucuk HF, Yegenoglu A, Uzun H, Eser M, Mentes CV, et al. Effects of seprafilm and heparin in combination on intra-abdominal adhesions. Eur Surg Res. 2008;41:203–7. doi: 10.1159/000134919. [DOI] [PubMed] [Google Scholar]
- 6.Ersoy E, Ozturk V, Yazgan A, Ozdogan M, Gundogdu H.Comparison of the two types of bioresorbable bariers to prevent intra-abdominal adhesions in rats J Gastrointest SurgDOI 10.1007/s11605-008-0678-5. [DOI] [PubMed] [Google Scholar]
- 7.Hooker GD, Taylor BM, Driman DK. Prevention of adhesion formation with use of sodium hyaluronate-based bioresorbable membrane in a rat model of ventral hernia repair with polypropylene mesh – A randomised, controlled study. Surg. 1999;125:211–6. [PubMed] [Google Scholar]
- 8.Avsar AF, Avsar FM, Sahin M, Topaloglu S, Vatansev H, Belviranli M. Diphenhydramine and hyaluronic acid derivates reduce adnexal adhesions and prevent tubal obstructions in rats. Eur J Obstet Gynecol Reprod Biol. 2003;106:50–4. doi: 10.1016/s0301-2115(02)00213-0. [DOI] [PubMed] [Google Scholar]
- 9.Kesting MR, Loeffelbein DJ, Steinstraesser L, Muecke T, Demtroeder C, Sommerer F, et al. Cryopreserved Human Amniotic membrane for soft tissue repair in rats. Ann Plast Surg. 2008;60:684–91. doi: 10.1097/SAP.0b013e31814fb9d2. [DOI] [PubMed] [Google Scholar]
- 10.Corrales F, Corrales M, Schirmer CC.Preventing intraperitoneal adhesions with vitamin E and sodium hyaluronate/carboxymethylcelulose. A comperative study in rats Acta Cir Bras[serial on the internet] 2008 Jan–Feb;23(1). Available from URL:http://scielo.br/acb [DOI] [PubMed]
- 11.Şahin Y, Saǧlam A. Synergistic effects of carboxymethylcellulose and low molecular weight heparin in reducing adhesion formation in the uterine horn model. Acta Obstet Gynecol Scand. 1994;73:70–3. doi: 10.3109/00016349409013399. [DOI] [PubMed] [Google Scholar]
- 12.Türkçapar AG, Özarslan C. The effectiveness of low molecular weight heparin on adhesion formation in experimental rat model. Int Surg. 1995;80:92–4. [PubMed] [Google Scholar]
- 13.Bulbuller N, Ilhan YS, Kirkil C, Cetiner M, Gogebakan Ö, Ilhan N. Can angiotensin converting enzyme inhibitors prevent postoperative adhesions? J Surg Res. 2005;125:94–7. doi: 10.1016/j.jss.2004.11.030. [DOI] [PubMed] [Google Scholar]
- 14.Kucuk HF, Kaptanoglu L, Kurt N, Uzun H, Eser M, Bingul S, et al. The role of simvastatin on postoperative peritoneal adhesion formation in an animal model. Eur. Surg. Res. 2007;39:98–102. doi: 10.1159/000099156. [DOI] [PubMed] [Google Scholar]
- 15.Nair SK, Bhat IK, Aurora AL. Role of proteolytic enzyme in the prevention of postoperative intraperitoneal adhesions. Arch Surg. 1974;108:849–53. doi: 10.1001/archsurg.1974.01350300081019. [DOI] [PubMed] [Google Scholar]
- 16.Hellebrekers BWJ, Trimbos-Kemper GCM, van Blitterswijk CA, Bakkum EA, Trimbos JBMZ. Effects of five different barrier materials on postsurgical adhesion formation in the rat. Human Reproduction. 2000;15:1358–63. doi: 10.1093/humrep/15.6.1358. [DOI] [PubMed] [Google Scholar]
- 17.Grahame GR, Torchia MG, Dankewich KA, Ferguson IA. Surface-active material in peritoneal affluent of CAPD patients. Perit Dial Bull. 1985;5:109–11. [PubMed] [Google Scholar]
- 18.Gotloib L. Anatomy of peritoneal membrane. Witching proceeding 1st course on peritoneal dialysis. 1982:17. [Google Scholar]
- 19.Snoj M. Intra-abdominal adhesion formation is initiated by phospholipase A2. Medical hypotheses. 1993;41:525–8. doi: 10.1016/0306-9877(93)90109-4. [DOI] [PubMed] [Google Scholar]
- 20.Becker JM, Dayton MT, Fazio VW, Beck DE, Stryker SJ, Wexner SD, et al. Prevention of postoperative abdominal adhesions by sodium hyaluronate-based bioresorbable membrane: a prospective, randomized, double blind multicenter study. J Am Coll Surg. 1996;183:297–306. [PubMed] [Google Scholar]
- 21.Ar’Rajab A, Ahren B, Rozga J, Benbark S. Phosphatidylcholine prevents postoperative peritoneal adhesions: An experimental study in rat. J Surg Res. 1991;50:212–5. doi: 10.1016/0022-4804(91)90180-t. [DOI] [PubMed] [Google Scholar]
- 22.Sulaiman H, Dawson L, Laurent GJ, Herrick SE. Role of plasminogen activators in peritoneal adhesion formation. Biochemical Society Transactions. 2002;30:126–31. doi: 10.1042/. [DOI] [PubMed] [Google Scholar]
- 23.Scott-Coombes DN, Whawell SA, Thompson JN. The operative peritoneal fibrinolytic response to abdominal operation. Eur J Surg. 1995;161:395–9. [PubMed] [Google Scholar]
- 24.Aorons CB, Cohen PA, Gower A, Reed KL, Leeman SE, Stucchi AF, et al. Statins (HMG-CoA reductase inhibitors) decrease postoperative adhesions by increasing peritoneal fibrinolytic activity. Ann Surg. 2007;245:176–84. doi: 10.1097/01.sla.0000236627.07927.7c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Raftery AT. Effect of peritoneal trauma on peritoneal fibrinolytic activity and intraperitoneal adhesion formation. An experimental study in the rat. Eur Surg Res. 1981;13:397–401. doi: 10.1159/000128208. [DOI] [PubMed] [Google Scholar]
- 26.Rozga J, Andersson R, Srinivas U, Ahren B, Benmark S. Influence of Phosphatidylcholine on intra-abdominal adhesion formation and peritoneal macrophages. Nephron. 1990;54:134–38. doi: 10.1159/000185833. [DOI] [PubMed] [Google Scholar]
- 27.Mazuji MK, Kalambaheti K, Pawar B. Prevention of adhesions with polyvinylpyrrolidone. Preliminary report. Arch Surg. 1964;89:1011–5. doi: 10.1001/archsurg.1964.01320060079015. [DOI] [PubMed] [Google Scholar]
- 28.Koçak I, Ünlü C, Akçan Y, Yakin K. Reduction of adhesion formation with cross-linked hyaluronic acid fter peritoneal surgery in rats. Fertil steril. 1999;72:873–8. doi: 10.1016/s0015-0282(99)00368-4. [DOI] [PubMed] [Google Scholar]
- 29.Laurent TC, Laurent UB, Fraser JR. The structure and function of hyaluronan: An overview. Immunol Cell Biol. 1996;74:A1–7. doi: 10.1038/icb.1996.32. [DOI] [PubMed] [Google Scholar]
- 30.Alponat A, Lakshminara SR, Yavuz N, Goh PM. Prevention of adhesions by Seprafilm, an absorbable barrier: an incisional hernia model in rats. Am Surg. 1997;63:818–9. [PubMed] [Google Scholar]
- 31.Müller SA, Treutner KH, Jörn H, Anurov M, Oettinge AP, Schumpelick V. Adhesion prevention comparing liquid and solid barriers in the rabbit uterine horn model. Eur J Obstet Gynecol Reprod Biol. 2005;120:222–6. doi: 10.1016/j.ejogrb.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 32.Diamond MP. Reduction of adhesions after uterine myomectomy by Seprafilm membrane (HAL-F): a blinded, prospective, randomized, multicenter clinical study. Seprafilm Adhesion Study Group. Fertil Steril. 1996;66:904–10. [PubMed] [Google Scholar]
- 33.Heydrick SJ, Reed KL, Cohen PA, Aarons CB, Gower AC, Becker JM, et al. Intraperitoneal administration of methylene blue attenuates oxidative stress, increases peritoneal fibrinolysis, and inhibits intraabdominal adhesion formation. J Surg Res. 2007;143:311–9. doi: 10.1016/j.jss.2006.11.012. [DOI] [PubMed] [Google Scholar]
- 34.Sulaiman H, Gabella G, Davis C, Mutsaers SE, Boulos P, Laurent GJ, et al. Presence and distribution of sensory nevre fibers in human peritoneal adhesions. Ann Surg. 2001;324:256–61. doi: 10.1097/00000658-200108000-00016. [DOI] [PMC free article] [PubMed] [Google Scholar]